Introduction
Lung cancer is the leading cause of cancer-related
mortality, accounting for 1.8 million deaths worldwide in 2018
(1). Over the past 2 decades, the
use of targeted therapies and immunotherapies has achieved survival
benefits in a proportion of patients (2-4).
However, the 5-year overall survival rate of all stages combined
non-small lung cancer (NSCLC) and small cell lung cancer (SCLC) is
currently 18.6% (5) and 6%
(6), respectively. Therefore, the
development of novel treatment approaches, including rationally
designed combination therapies, is urgently required in order to
improve the clinical outcomes of patients.
Natural products (NPs) are major sources of new
drugs due to their chemical structure diversity (7-9).
In 1940 and 2014, the US Food and Drug Administration (FDA)
approved 175 anticancer small molecules, of which 63% were either
NPs or directly derived from NPs (10,11). Ursolic acid (UA) is a pentacyclic
triterpenoid natural compound initially identified in the
epicuticular waxes of apples in the 1920s and is widely distributed
in the peel of numerous fruits, as well as in medicinal herbs and
other plants (12,13). UA displays a wide range of
pharmacological effects, such as anti-inflammatory (14,15), antioxidant (16), antitumor (17,18), cardioprotective (19) and immunomodulatory (20,21) activities. UA has been shown to
possess a variety of anticancer capabilities in vitro and
in vivo, including the ability to inhibit the proliferation
of multiple types of tumor cells, induce apoptosis, induce cell
cycle arrest, suppress tumor invasion, metastasis and angiogenesis
(22-27). However, the underlying anticancer
mechanisms of UA in lung cancer are not yet fully understood.
Autophagy is an evolutionarily conserved process in
which intracellular membrane structures undergo dynamic
morphological changes that lead to the degradation of cellular
proteins and cytoplasmic organelles (28). Autophagy is a double-edged sword
and can either repress or promote cell apoptosis, depending on the
cell type, nutrient supply and stimulus (29,30). Recent studies have revealed that
several signaling pathways regulate autophagy, such as the
mammalian target of rapamycin (mTOR) pathway and the extracellular
signal-regulated kinase 1/2 (ERK1/2) pathway (28,31,32). It has been reported that UA
induces autophagy in some types of cancer cells (33,34). However, the underlying mechanisms
of UA-induced autophagy are not yet fully understood.
According to the theory of Traditional Chinese
Medicine (TCM), lung cancer is the result of 'heat and toxin
accumulation' within the body, and medicinal herbs with the
'heat-clearing and detoxification' activity are considered to have
anti-lung cancer activity. In the present study, compounds isolated
from the 'heat-clearing and detoxification' medicinal herbs were
collected and the effects of 63 previously untested compounds
(Table SI) on the human NSCLC
cell line, A549, were examined. As breast cancer is the most
commonly diagnosed type of cancer and the leading cause of
cancer-related mortality among women (1), the human breast cancer cell line
MCF-7, was also examined. It was reported that UA exhibited potent
anticancer activity against these cells. Further experiments
revealed that UA suppressed the proliferation, and induced the
apoptosis and autophagy of lung cancer cells. It was identified
that the inhibition of the mTOR signaling pathway, rather than the
activation of the ERK1/2 signaling pathway, was one of the
mechanisms responsible for the UA-induced autophagy of NSCLC cells.
Of note, the inhibition of autophagy by chloroquine (CQ) or siRNA
for autophagy-related gene 5 (ATG5) enhanced the UA-induced
inhibition of proliferation and the induction of apoptosis,
suggesting that UA-induced autophagy plays a pro-survival role in
NSCLC cells. These findings suggest that the combined use of UA and
autophagy inhibitors may be a promising therapeutic strategy for
lung cancer.
Materials and methods
Reagents and antibodies
UA and the custom compound library (cat. no. LC00)
containing 63 plant-sourced natural products (Table SI) were purchased from Target
Molecule Corp.; chloroquine (CQ) and 4,6-diamidino-2-phenylindole
(DAPI) were purchased from Sigma-Aldrich Merck KGaA; LY294002 and
3BDO were obtained from MedChemExpress; U0126 was obtained from
Cell Signaling Technology, Inc. The cells were treated with 5
µM CQ for 25 or 49 h. The cells were treated with 10
µM LY294002 or 5 µM U0126 for 25 h. The cells were
treated with 5 or 10 µM 3BDO for 25 h. Antibodies used
included rabbit anti-microtubule-associated protein 1 light chain 3
(LC3; cat. no. L7543, 1:1,000 for western blot analysis) and mouse
anti-actin (cat. no. A5441, 1:5,000 for western blot analysis) from
Sigma-Aldrich Merck KGaA; rabbit anti-poly(ADP-ribose) polymerase
(PARP; cat. no. 9542, 1:1,000 for western blot analysis), rabbit
anti-B-cell lymphoma 2 (Bcl-2; cat. no. 2870, 1:1,000 for western
blot analysis), rabbit anti-ATG5 (cat. no. 12994, 1:1,000 for
western blot analysis), rabbit anti-S6K (cat. no. 9202, 1:1,000 for
western blot analysis), rabbit anti-phospho-S6K (Thr389) (cat. no.
9205, 1:1,000 for western blot analysis), rabbit anti-phospho-S6
ribosomal protein (Ser240/244) (cat. no. 5364, 1:1,000 for western
blot analysis), rabbit anti-S6 ribosomal protein (cat. no. 2217,
1:1,000 for western blot analysis), rabbit anti-eukaryotic
translation initiation factor 4E-binding protein 1 (4E-BP1; cat.
no. 9644, 1:1,000 for western blot analysis), rabbit
anti-phospho-4E-BP1 (Ser65) (cat. no. 9451, 1:1,000 for western
blot analysis) and rabbit anti-phospho-ERK1/2 (Thr202/Tyr204) (cat.
no. 4370, 1:2,000 for western blot analysis) from Cell Signaling
Technology, Inc.; rabbit anti-ERK2 (cat. no. ab32081, 1:1,000 for
western blot analysis) from Abcam; rabbit anti-phospho-Akt (Ser473)
(cat. no. sc-7985-R, 1:500 for western blot analysis) and rabbit
anti-Akt (cat. no. sc-8312, 1:500 for western blot analysis) from
Santa Cruz Biotechnology, Inc..
Cells and cell culture
The human NSCLC cell lines, H460, H1975 (harboring
EGFR-L858R/T790M mutations), A549, H1299 and H520, the human SCLC
cell lines, H82 and H446, the human breast cancer cell line, MCF-7,
and the C57BL/6 murine Lewis lung carcinoma cell line, LLC, were
obtained from the American Type Culture Collection (ATCC). The
cells were cultured in DMEM or RPMI-1640 supplemented with 10%
fetal bovine serum (FBS; Gibco, Thermo Fisher Scientific, Inc.) at
37°C in a humidified atmosphere containing 5% CO2.
Cell viability assay
Cell viability was determined by
3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-
2H-tetrazolium, inner salt (MTS) assay, according to the protocol
of the CellTiter 96®AQueous One Solution Cell
Proliferation Assay kit (Promega Corporation). A total of 100
µl of cell suspension were seeded onto each well of 96-well
plates. The cells were co-incubated with UA for 24 to 72 h, and 20
µl of CellTiter 96®AQueous One Solution reagent
was added to each well, and the cells were then incubated at 37°C
for 1-2 h. The absorbance was measured at 490 nm using a microplate
reader (Bio-Tek Instruments, Inc.). The half maximal inhibitory
concentration (IC50) values were calculated from
appropriate dose-response curves.
Colony formation assay
The H460 and H1975 cells were seeded into 6-well
plates and cultured in RPMI-1640 supplemented with 10% FBS at 37°C
in a humidified atmosphere containing 5% CO2 overnight.
The cells were then exposed to 10 and 15 µM UA for
approximately 7 days until the cells grew to visible colonies. The
colonies were fixed in methanol and stained with 0.005% crystal
violet (Sigma-Aldrich Merck KGaA) for 30 min at room temperature.
Colonies comprising >50 cells were counted using an Olympus
inverted phase-contrast microscope (Olympus Corporation).
Apoptosis assay
Cell apoptosis was analyzed using the Annexin V-FITC
Apoptosis Detection kit (BD Biosciences) according to the
manufacturer's instructions. The cells were treated with the
indicated protocols, collected, resuspended in Annexin-V binding
buffer, followed by incubation with Annexin V-FITC and propidium
iodide (PI) for 15 min at room temperature in the dark, and were
examined by flow cytometry using a FACSCalibur flow cytometer (BD
Biosciences).
Western blot analysis
The preparation of whole cell protein lysates and
western blot analysis were performed as previously described
(35). The membranes were
incubated with the indicated primary antibodies at 4°C overnight,
followed by incubation with the horseradish peroxidase-conjugated
anti-mouse (cat. no. 115-035-003, Jackson ImmunoResearch
Laboratories, Inc.; 1:10,000) or anti-rabbit (cat. no. 111-035-003,
Jackson ImmunoResearch Laboratories, Inc.; 1:10,000) secondary
antibodies for 2 h at room temperature. The densitometric
quantification of protein expression was performed using ImageJ
software (version 1.4.3.67; National Institutes of Health).
Cell morphological analysis
The H460, H1975 and A549 cells were seeded in
12-well plates and incubated at 37°C in a humidified atmosphere
containing 5% CO2 overnight for cell attachment. The
following day, the cells were treated with UA at 10, 15 and 20
µM. Following incubation at 37°C in a humidified atmosphere
containing 5% CO2 for 24 h, the morphology of the cells
was observed and photographed using an Olympus inverted
phase-contrast microscope (Olympus Corporation).
Immunofluorescence assay
The H460 and H1975 cells were seeded on the cover
slides with 1% gelatin in 6-well plates and cultured in RPMI-1640
supplemented with 10% FBS at 37°C in a humidified atmosphere
containing 5% CO2 overnight. The cells were then treated
with UA at 15 µM. Following incubation at 37°C in a
humidified atmosphere containing 5% CO2 for 16 h, the
cells were fixed with 4% paraformaldehyde for 20 min, washed with
150 mM glycine in PBS, and permeabilized with 0.1% Triton X-100 in
PBS for 15 min at room temperature. After blocking with 5% BSA for
1 h at room temperature, the cells were incubated with LC3 antibody
(cat. no. L7543, Sigma-Aldrich Merck KGaA; 1:100) at 4°C overnight
and then incubated with FITC-conjugated secondary antibody (cat.
no. ZF-0311, ZSGB-BIO; 1:100) for 2 h at room temperature. Nuclei
were stained with DAPI for 2 min at room temperature. Images were
acquired using a laser confocal scanning microscope (N-STORM, Nikon
Corporation).
Small interfering RNAs (siRNAs) and
transfection
The specific target sequences of ATG5 (sense, 5′-GAC
GUU GGU AAC UGA CAA ATT-3′ and antisense, 5′-UUU GUC AGU UAC CAA
CGU CTT-3′) (36) and the
negative control (sense, 5′-UUC UCC GAA CGU GUC ACG UTT-3′ and
antisense, 5′-ACG UGA CAC GUU CGG AGA ATT-3′) were synthesized by
GenePharma. The cells were transfected with the 50 nM siRNA using
Lipofectamine 3000 (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's instructions. The H460 and H1975
cells were transfected with the siRNAs for 36 h, and then treated
with 0-25 µM of UA for 24 or 48 h.
Statistical analysis
The results are presented as the means ± standard
deviation (means ± SD). A two-tailed Student's t-test or one-way
ANOVA with Bonferroni's post hoc test was used to determine
statistically significant differences. P-values <0.05 were
considered to indicate statistically significant differences.
Results
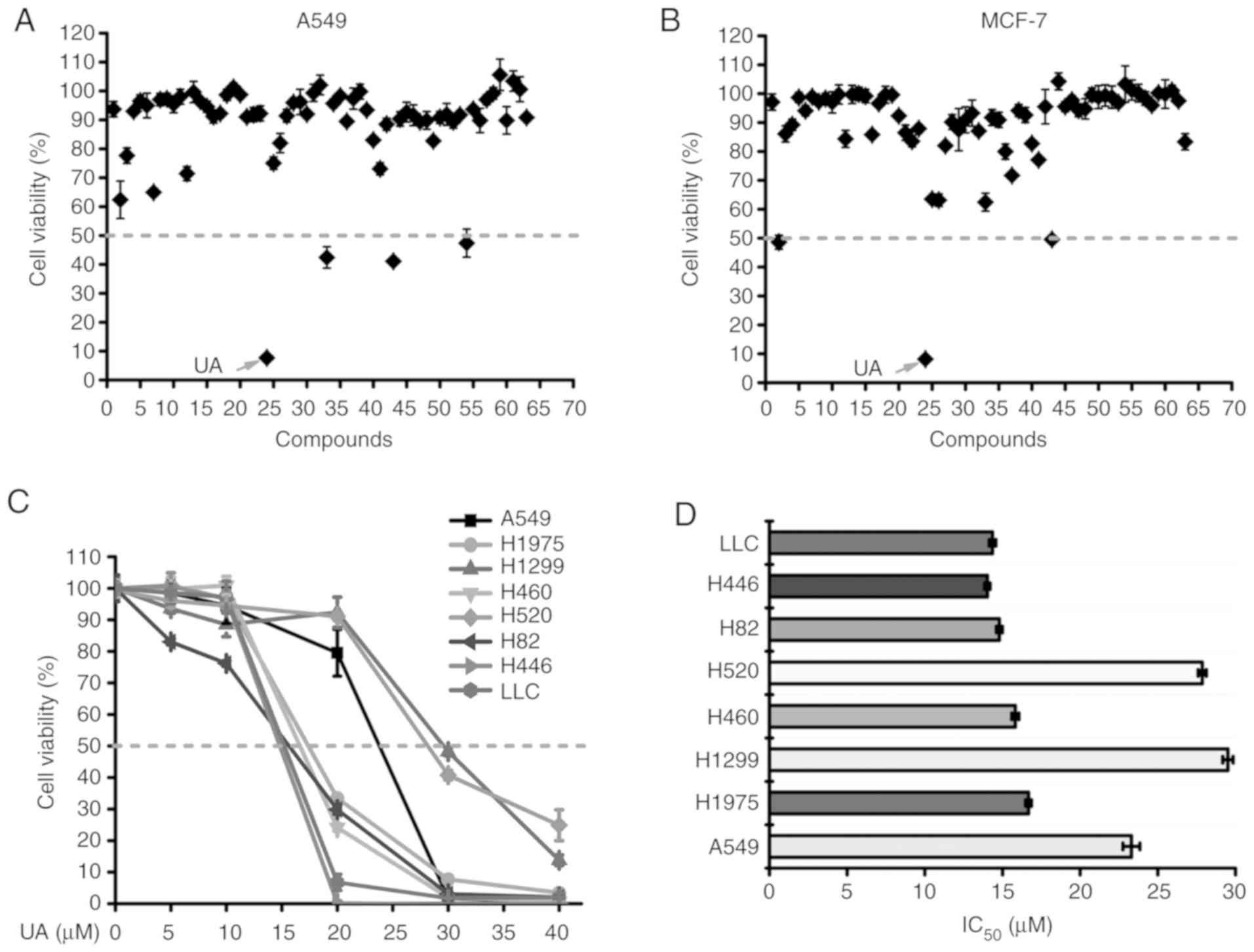
Identification of UA as a potent
anticancer compound
Compounds from medicinal herbs usually have moderate
anticancer activity. In the present study, in order to examine the
anticancer activity of the compounds, the A549 and MCF-7 cells were
treated with the compounds and cell viability was determined by MTS
assay. To reach a high efficacy in the identification of candidate
compounds, a relatively high concentration (40 µM) was used
for each molecule in the initial screening. For compounds that
exhibited a significant anticancer activity, further experiments
using lower concentrations were carried out; for those that did not
exhibit a satisfactory activity (inhibition rate <50%), no
further experiments were performed. As shown in Fig. 1A and B, UA exhibited the most
potent cytotoxicity against these 2 cancer cell lines among the 63
natural compounds tested (Table
SI). The inhibitory effect of UA on the proliferation of 8 lung
cancer cell lines (5 NSCLC cell lines: A549, H1975, H1299, H460 and
H520; and 2 SCLC cell lines: H82, H446; and the murine Lewis lung
carcinoma cell line, LLC) was further examined by MTS assay by
treating the cells with various concentrations of UA. Treatment
with UA (0-40 µM) for 48 h suppressed the proliferation of
these 8 lung cancer cell lines in a dose-dependent manner (Fig. 1C). The half maximal inhibitory
concentration (IC50) values of UA for these 8 cell lines
were 23.3±0.54, 16.68±0.18, 29.51±0.34, 15.83±0.23, 27.86±0.26,
14.8±0.2, 14.02±0.17 and 14.36±0.21 µM, respectively
(Fig. 1D).
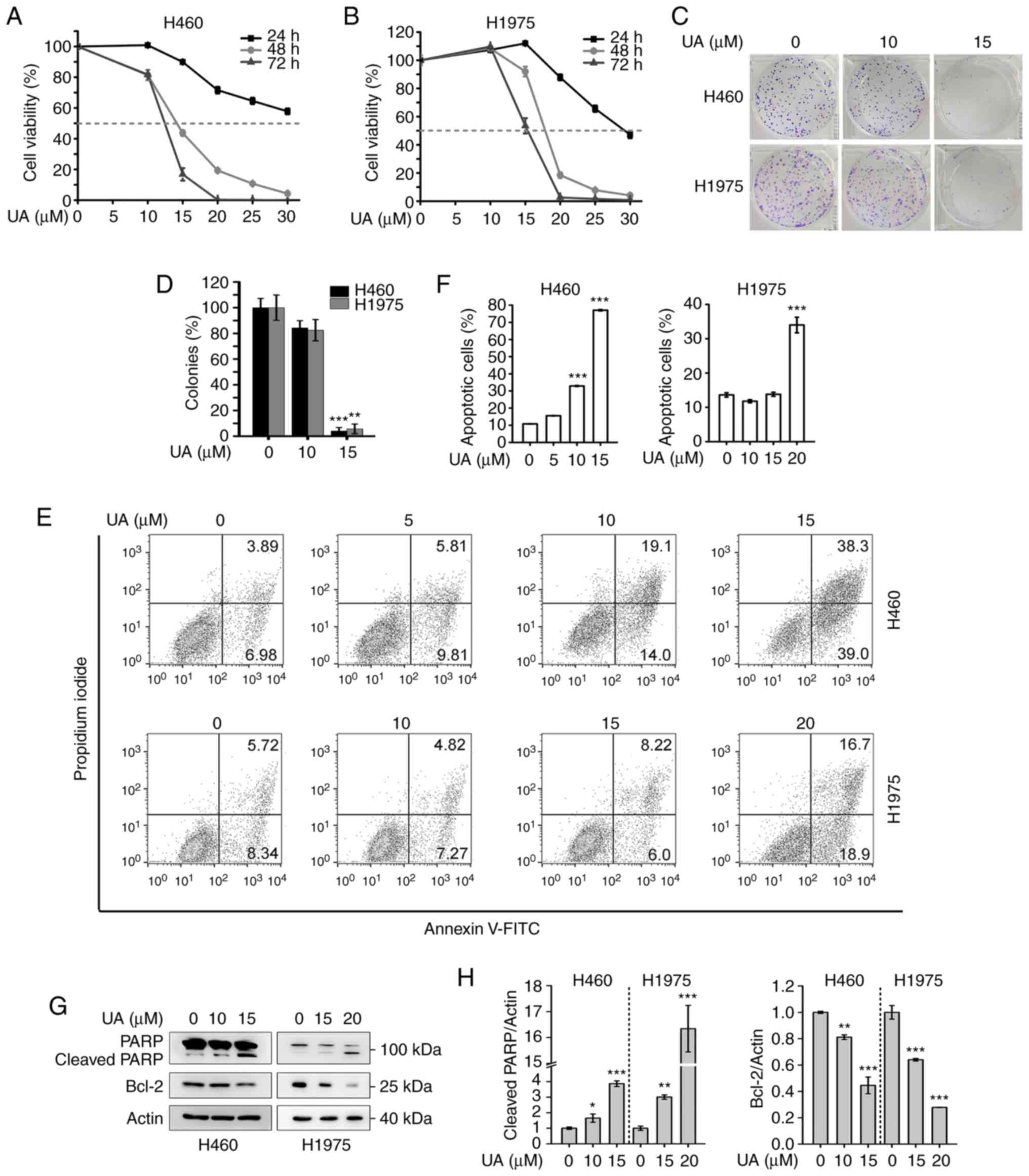
UA inhibits the proliferation and induces
the apoptosis of H460 and H1975 cells
By using MTS assay, it was found that UA
significantly suppressed the proliferation of the H460 and H1975
cells in a dose- and time-dependent manner (Fig. 2A and B). Furthermore, UA markedly
suppressed the colony formation activity of the H460 and H1975
cells, as assessed by colony formation assay (Fig. 2C and D).
To investigate whether UA induces the apoptosis of
the cells, the H460 and H1975 cells were treated with UA at the
indicated concentrations for 48 h and apoptosis was detected by
Annexin V-FITC/PI double staining and flow cytometric analysis. The
upper right and lower right quadrants of the dot plots were counted
as apoptotic cells. As shown in Fig.
2E and F, UA markedly increased the percentage of Annexin V (+)
apoptotic cells compared with the control group. In addition, the
expression of PARP, a well-known cellular substrate of caspases,
and that of Bcl-2, the major regulator of the intrinsic
(mitochondrial) apoptotic pathway (37), were examined by western blot
analysis. As shown in Fig. 2G and
H, treatment of the H460 and H1975 cells with UA for 48 h
increased the level of cleaved PARP and decreased the expression of
Bcl-2.
UA induces autophagy and increases the
autophagic flux in NSCLC cells
By observation with an inverted phase-contrast
microscopy, it was found that treatment of the H460, H1975 and A549
cells with UA for 24 h induced vacuoles in the cytoplasm,
suggesting that UA regulates autophagy in these cells (Fig. 3A). LC3 is associated with the
formation of the autophagosome membrane and usually displays a
molecular form conversion of cytoplasmic LC3-I to the lapidated
LC3-II that is generally used as a marker to monitor autophagy
(38,39). In the present study, western blot
analysis revealed that UA markedly elevated the level of LC3-II in
a concentration-dependent manner in the H460, H1975 and A549 cells
(Fig. 3B). The distribution of
endogenous LC3 in the H460 and H1975 cells was also analyzed by
immunofluorescence assay. As shown in Fig. 3C, the amount of LC3 puncta,
representative of autophagosomes, increased in the UA-treated
cells, whereas the control group cells exhibited a diffuse
distribution of LC3. ATG5 is involved in vesicle formation and the
lipidation of LC3 and plays pivotal roles in the process of
autophagy (40,41). The present study demonstrated that
the knockdown of ATG5 by siRNA reduced the UA-induced LC3-II
accumulation in the H460 and H1975 cells (Fig. 3D). In addition, the autophagic
flux was evaluated by analyzing the level of LC3-II in the presence
of CQ, which inhibits late-stage autophagy by blocking the fusion
of autophagosomes and lysosomes (39). In the H460, H1975 and A549 cells,
the LC3-II levels were elevated upon combination treatment with UA
and CQ compared to single treatment alone (Fig. 3E), indicating that UA promoted the
autophagic flux in these cells. Taken together, these results
demonstrate that UA induces autophagy and increases the autophagic
flux in NSCLC cells.
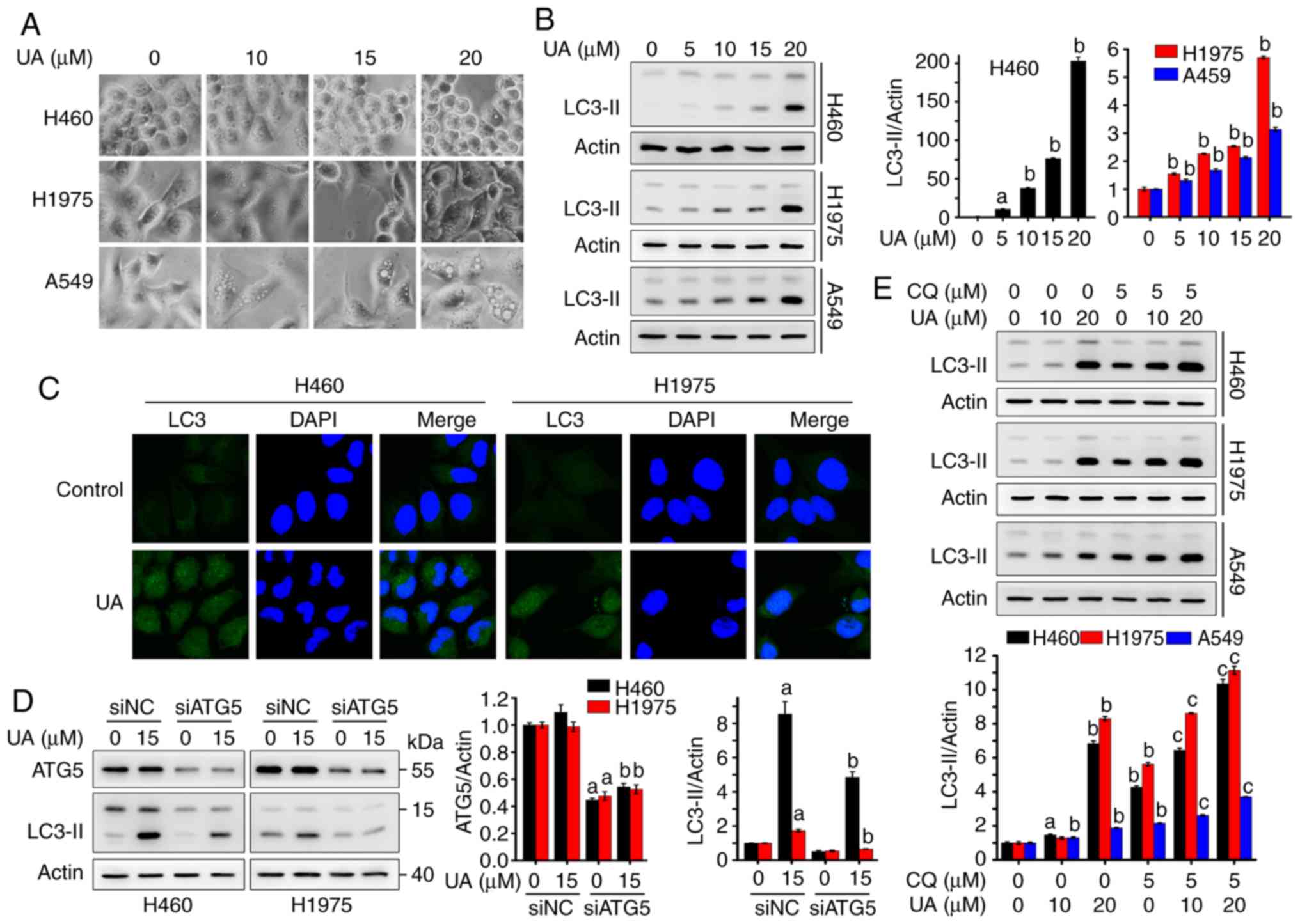 | Figure 3UA induces autophagy and increases
the autophagic flux in NSCLC cells. (A) H460, H1975 and A549 cells
were treated with UA at 10, 15 and 20 µM for 24 h, followed
by obtaining images under an Olympus inverted phase-contrast
microscope. (B) NSCLC cells were treated with the indicated
concentrations of UA for 24 h, and the expression levels of LC3
were analyzed by western blot analysis (left panel). Relative
densitometric quantification of LC3-II expression was performed
using western blot bands and data are presented as the mean ± SD
(n=3; right panel). aP<0.01 and
bP<0.001. (C) H460 and H1975 cells were treated with
15 µM UA for 16 h, cells were then analyzed by
immunofluorescence assay using LC3 antibody (green) and DAPI
(blue). (D) H460 and H1975 cells were transfected with siNC or
siATG5 for 36 h, then treated with UA at 15 µM for 24 and 48
h respectively, lysed, and whole cell lysates were subjected to
western blot analysis using the indicated antibodies. Relative
densitometric quantification of protein expression and data are
presented as the means ± SD (n=3). aP<0.001 vs. siNC
0 µM UA, bP<0.001 vs. siNC 15 µM UA.
(E) H460, H1975 and A549 cells were pre-treated without or with 5
µM CQ for 1 h, followed by UA treatment at 10 and 20
µM for 24 h, and the expression levels of LC3 were examined
by western blot analysis. Relative densitometric quantification of
the LC3-II expression and data are presented as the means ± SD
(n=3). aP<0.01 vs. vehicle, bP<0.001
vs. vehicle, cP<0.001 vs. 5 µM CQ. UA, ursolic
acid; CQ, chloroquine. |
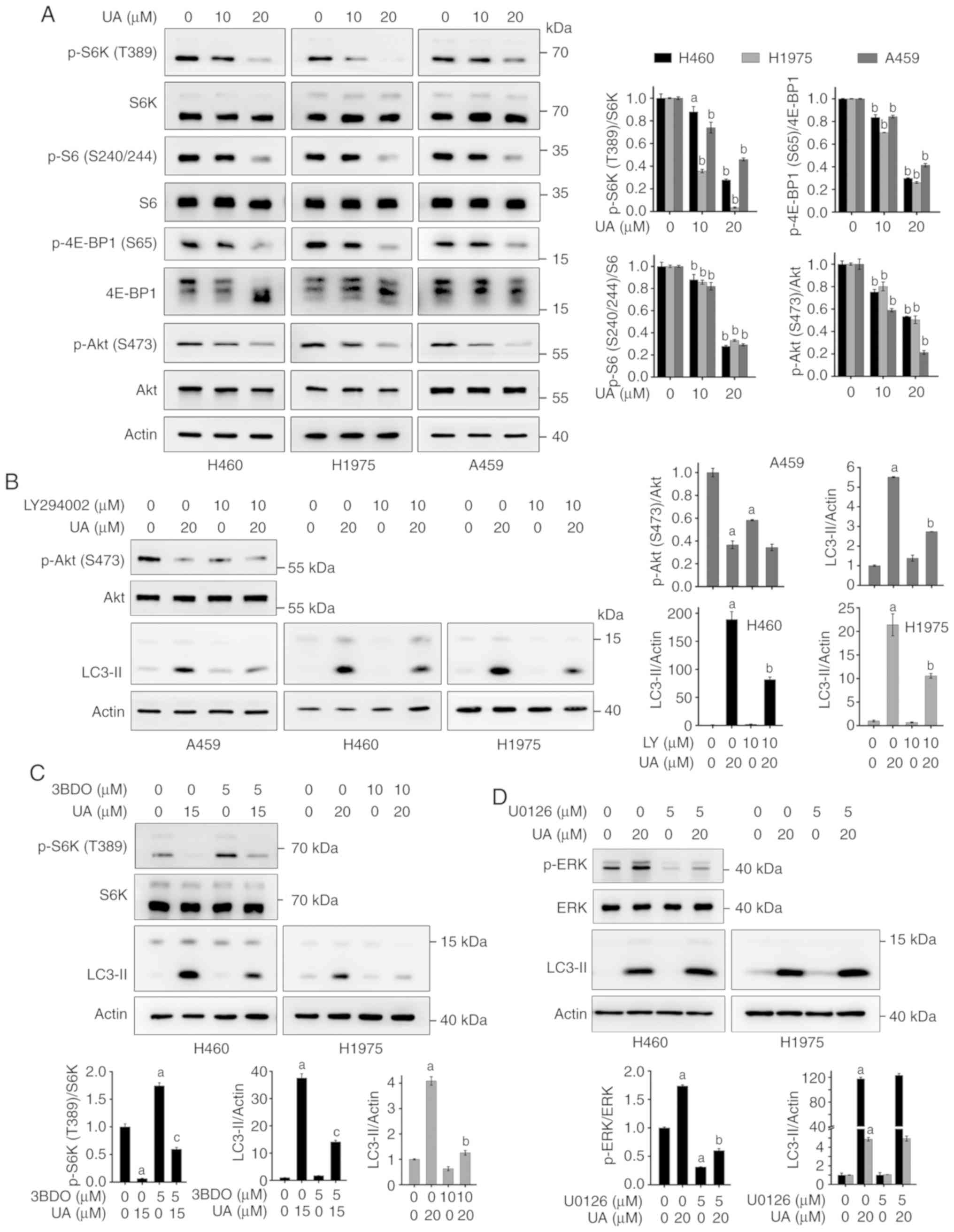
UA-induced autophagy is attributed to the
inhibition of the PI3K/Akt/mTOR signaling pathway
Since the mTOR signaling pathway is a main negative
regulator of autophagy (42-44), the present study examined whether
UA-induced autophagy is associated with the inhibition of this
signaling pathway. As shown in Fig.
4A, UA treatment suppressed the phosphorylation of S6K (p-S6K
T389) and 4E-BP1 (p-4E-BP1 S65), two of the most well-characterized
downstream effector molecules of mTOR complex 1 (mTORC1) (43). UA also suppressed the
phosphorylation of S6 (p-S6 S240/244), a substrate of S6K (Fig. 4A). These results indicated that UA
suppressed mTORC1 signaling. mTOR forms two structurally and
functionally distinct multi-protein complexes: The mTORC1 and
mTORC2 (45). mTORC2
phosphorylates Akt on S473 (46).
We examined the effects of UA on mTORC2 signaling and found that UA
markedly decreased the phosphorylation of Akt (S473) in the tested
cells (Fig. 4A), suggesting that
UA inhibits mTORC2 signaling.
To further confirm the role of the mTOR signaling
pathway in UA-induced autophagy, phosphatidylinositol 3-kinase
(PI3K) inhibitor, LY294002 (47),
and the mTOR activator, 3BDO (48), were employed. It was found that
pre-treatment with LY294002 suppressed the phosphorylation of Akt
(S473) (Fig. 4B), whereas 3BDO
upregulated p-S6K (T389) (Fig.
4C). LY294002 and 3BDO evidently attenuated the UA-induced
expression of LC3-II in the NSCLC cells (Fig. 4B and C), demonstrating that the
PI3K/Akt/mTOR signaling pathway is involved in UA-induced
autophagy. The ERK1/2 signaling pathway is another major regulator
of autophagy (28,49,50); Thus, the present study wished to
determine whether ERK1/2 signaling plays a role in UA-induced
autophagy. It was found that a selective inhibitor of ERK1/2,
U0126, downregulated p-ERK and suppressed the UA-induced
upregulation of p-ERK (Fig. 4D).
Pre-treatment with U0126 did not affect the UA-induced expression
of LC3-II (Fig. 4D), suggesting
that ERK1/2 signaling is not involved in UA-induced autophagy.
Autophagy inhibition enhances the
anti-NSCLC activity of UA
Whether cancer therapy-induced autophagy is a
pro-survival response or an anticancer effect, remains
controversial (28,51). In the present study, to assess the
role of UA-induced autophagy in its antitumor effect, the autophagy
inhibitor, CQ, was used in cells treated with UA. MTS assay
revealed that the suppression of the late autophagic process with
CQ (5 µM) significantly potentiated the UA-induced
inhibition of the proliferation of the H460, H1975 and A549 cells
(Fig. 5A). Moreover, Annexin
V-FITC/PI double staining demonstrated that CQ markedly enhanced
the UA-induced apoptosis of H460 and H1975 cells (Fig. 5B and C). Consistently, CQ
potentiated the UA-induced downregulation of the anti-apoptotic
protein, Bcl-2, in the H460 cells (Fig. 5D) and the UA-induced cleavage of
PARP in H1975 cells (Fig. 5E).
The blockage of the autophagosome formation by LY294002 (10
µM) also enhanced the UA-induced cleavage of PARP in the
H1975 cells (Fig. 5F). It was
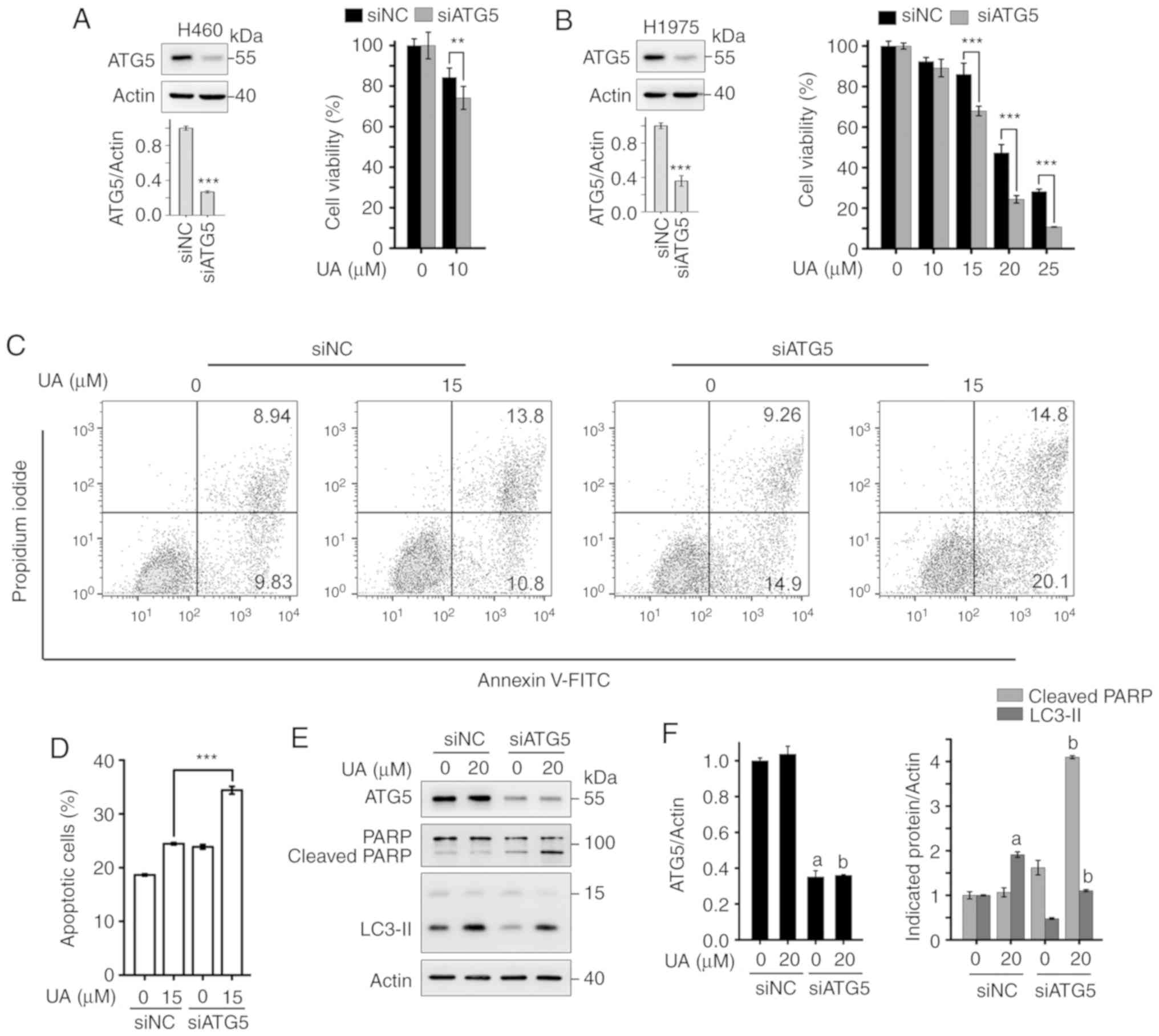
further demonstrated that the silencing of ATG5 by siRNA enhanced
the UA-induced inhibition of the proliferation of H460 and H1975
cells (Fig. 6A and B). In
addition, siATG5 potentiated the UA-induced apoptosis of H1975
cells (Fig. 6C and D). The
silencing of ATG5 resulted in the accumulation of cleaved PARP in
the UA-treated H1975 cells (Fig. 6E
and F). Collectively, these results suggest that UA-induced
autophagy plays a protective role in NSCLC cells, and the
suppression of autophagy enhances the anti-NSCLC activity of
UA.
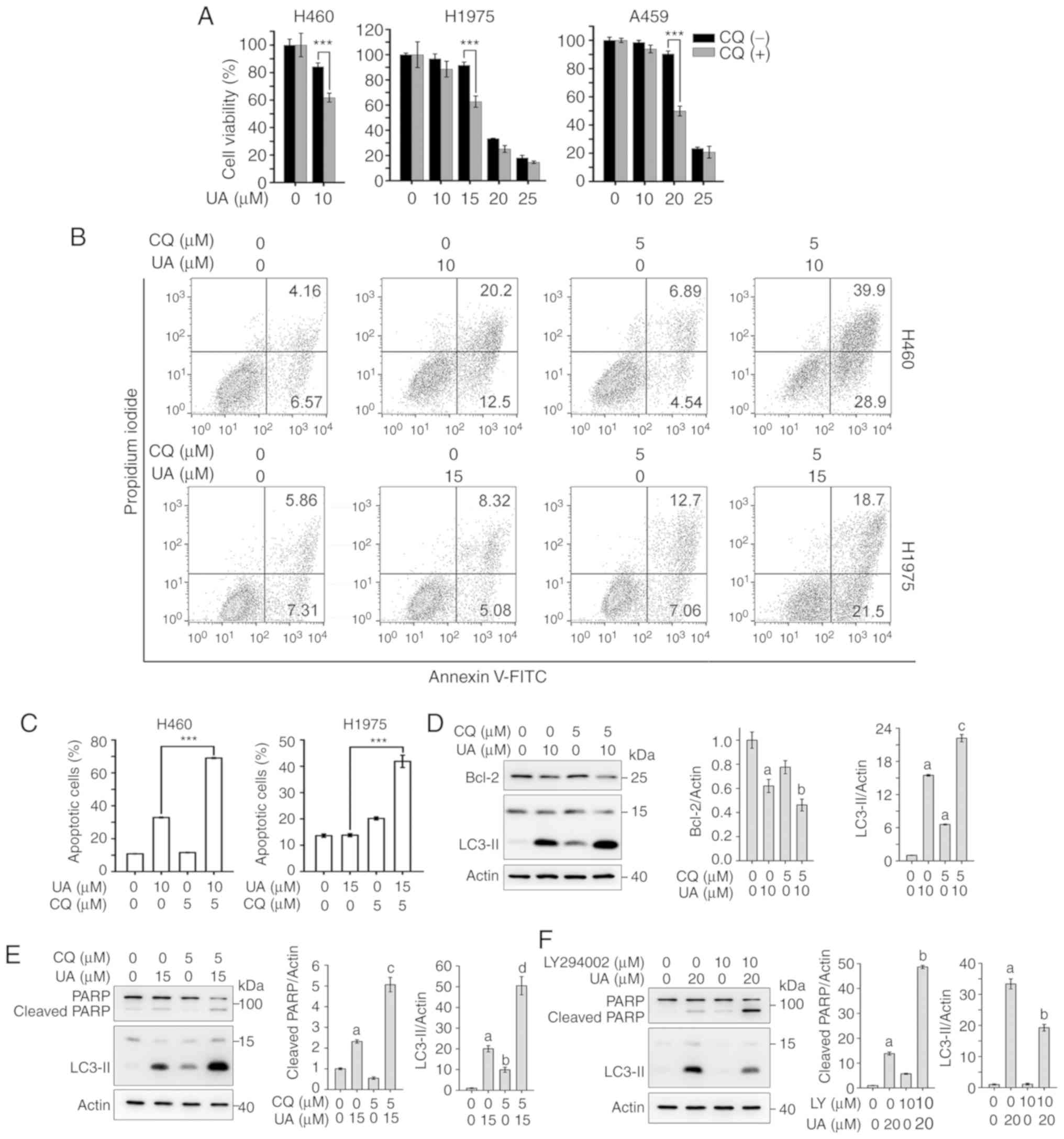 | Figure 5Autophagy inhibition by CQ enhances
the UA-induced inhibition of the proliferation and promotes the
UA-induced apoptosis of NSCLC cells. (A) NSCLC cells were treated
with the indicated concentrations of UA without or with 5 µM
CQ for 48 h, cell viability was assessed by MTS assay and data are
presented as relative viability to the control.
***P<0.001. (B) H460 and H1975 cells were pre-treated
without or with 5 µM CQ for 1 h, followed by treatment with
UA at the indicated concentrations for 48 h, cell apoptosis was
analyzed by flow cytometry using Annexin V-FITC/PI staining.
Representative images are shown. (C) Quantification of flow
cytometric analysis of apoptosis. ***P<0.001. (D)
H460 cells were pre-treated without or with 5 µM CQ for 1 h,
then incubated with 10 µM UA for 48 h, lysed, and whole cell
lysates were subjected to western blot analysis using the indicated
antibodies. Actin was used as a loading control (left panel).
Relative densitometric quantification of protein expression was
performed and data are presented as the means ± SD (n=3; right
panel). aP<0.001 vs. vehicle, bP<0.05
vs. 10 µM UA, cP<0.001 vs. 5 µM CQ. (E)
H1975 cells were pre-treated without or with 5 µM CQ for 1
h, then incubated with UA at 15 µM for 48 h, whole cell
lysates were subjected to western blot analysis with the indicated
antibodies (left panel). Relative densitometric quantification of
protein expression was performed and data are presented as the
means ± SD (n=3; right panel). aP<0.001 vs. vehicle,
bP<0.01 vs. vehicle, cP<0.001 vs. 15
µM UA, dP<0.001 vs. 5 µM CQ. (F) H1975
cells were pre-treated without or with 10 µM LY294002 for 1
h, followed by UA treatment at 20 µM for 24 h. The cells
were harvested and subjected to western blot analysis using the
indicated antibodies (left panel). Relative densitometric
quantification of protein expression was performed and data are
presented as the means ± SD (n=3; right panel).
aP<0.001 vs. vehicle, bP<0.001 vs. 20
µM UA. UA, ursolic acid; CQ, chloroquine; LY, LY294002. |
Discussion
UA is a natural pentacyclic triterpenoid that
possesses anticancer activities against a variety of cancers in
vitro and in vivo (17,18,22,24,26). For example, UA has been shown to
significantly suppress xenograft tumor growth in a human lung
cancer H1975 xenograft mouse model (24). UA exhibits a low toxicity in human
normal lung epithelial BEAS-2B cells, and exerts minimal toxic
effects on the kidney and liver tissues in mice (24). Furthermore, UA has recently been
promoted to enter clinical trials to investigate its effects on
insulin sensitivity (phase II study) and muscle function in human
sarcopenia (phase II and III studies) (52,53). However, the underlying anti-lung
cancer mechanisms of UA are not yet fully understood. In the
present study, it was demonstrated that UA inhibited the
proliferation of various lung cancer cells, including the human
NSCLC cells, H460, H1975, A549, H1299 and H520, the human SCLC
cells H82 and H446, and murine LLC cells (Fig. 1). Of note, UA exerted inhibitory
effects on gefitinib-resistant H1975 cells that bear
EGFR-L858R/T790M mutations and on H460 cells with wild-type EGFR,
as well as on the SCLC cells H82 and H446 that harbor TP53 and RB1
mutations (Fig. 1). These
findings indicate that UA possesses therapeutic potential in both
NSCLC and SCLC, which warrants further investigation.
Previous studies have demonstrated that UA induces
autophagy in some types of cancer cells, such as prostate (54), cervical (55), breast (56), gliomas (33) and oral (34) cancer cells. In the present study,
it was found that UA increased the expression level of LC3-II and
induced autophagosome accumulation in NSCLC cells (Fig. 3). However, both the upregulation
of LC3-II and increased autophagosome formation can act as
autophagy inducers or autophagy inhibitors (57,58), which can be distinguished by the
knockdown of ATG proteins or treatment with CQ (58-60). The presents study demonstrated
that the knockdown of ATG5 by siRNA reduced the UA-induced
accumulation of LC3-II in H460 and H1975 cells (Fig. 3). The LC3-II levels further
increased upon the combined use of UA and CQ (Fig. 3). These results demonstrated that
UA-induced autophagy was ATG5-dependent, and the upregulation of
LC3-II and increased autophagosome formation may be autophagy
inducers in the cells.
Several signaling molecules, such as mTOR, PI3Ks and
mitogen-activated protein kinases (MAPKs), have been shown to play
a role in regulating autophagy (31,61). The serine/threonine kinase mTOR
plays central roles in a number of fundamental cell processes, and
abnormalities in this signaling pathway have been implicated in
cancers (62). The class I PI3K
activates the downstream effector Akt, leading to activation of
mTORC1 and inhibition of autophagy (28,43). MAPKs, including ERK1/2, Jun
N-terminal kinase (JNK) and p38 MAPK, belong to the family of
serine/threonine kinases that control a variety of cellular events,
such as proliferation, apoptosis and autophagy (50). In the present study, it was
identified that the inhibition of the PI3K/Akt/mTOR signaling
pathway, rather than the activation of the ERK1/2 signaling
pathway, was a mechanism of UA-induced autophagy in NSCLC cells
(Fig. 4).
Increasing evidence implicates a dual role of
autophagy in cancer therapy, i.e., protecting cell survival and
promoting cell apoptosis (51,63). It has been reported that the
blocking of autophagy increases erianin or betulinic acid-induced
apoptosis (64,65). In the present study, it was found
that the inhibition of autophagy by CQ or siATG5 enhanced the
UA-induced inhibition of cell proliferation and promoted the
UA-induced apoptosis (Figs. 5 and
6), indicating that UA-induced
autophagy plays a pro-survival role in NSCLC cells; combined
treatment with UA and autophagy inhibitor may have therapeutic
potentials for this lethal disease. However, further investigations
with multiple animal experiments are required prior to the clinical
application of the present results.
Supplementary Data
Funding
The present study was supported by the Key Project
of the National Natural Science Foundation of China (81830093), the
National Natural Science Funds for Distinguished Young Scholar
(81425025), the CAMS Innovation Fund for Medical Sciences (CIFMS;
2019-I2M-1-003), and the National Natural Science Foundation of
China (81672765, 81802796). The study sponsors had no role in the
design of the study; the data collection, analysis, or
interpretation; the writing of the article; or the decision to
submit for publication.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article or are available from the
corresponding author on reasonable request.
Authors' contributions
The project was conceived and designed by GBZ and
MW. The experiments were conducted by MW, HY, RW, ZYC, QH, YFZ and
SHG. Data were analyzed by GBZ and MW. The manuscript was written
by GBZ and MW. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
Not applicable.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Herbst RS, Morgensztern D and Boshoff C:
The biology and management of non-small cell lung cancer. Nature.
553:446–454. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hirsch FR, Scagliotti GV, Mulshine JL,
Kwon R, Curran WJ Jr, Wu YL and Paz-Ares L: Lung cancer: Current
therapies and new targeted treatments. Lancet. 389:299–311. 2017.
View Article : Google Scholar
|
|
4
|
Ackermann CJ, Reck M, Paz-Ares L, Barlesi
F and Califano R: First-line immune checkpoint blockade for
advanced non-small-cell lung cancer: Travelling at the speed of
light. Lung Cancer. 134:245–253. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gazdar AF, Bunn PA and Minna JD:
Small-cell lung cancer: What we know, what we need to know and the
path forward. Nat Rev Cancer. 17:725–737. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li JW and Vederas JC: Drug discovery and
natural products: End of an era or an endless frontier? Science.
325:161–165. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Newman DJ and Cragg GM: Natural products
as sources of new drugs from 1981 to 2014. J Nat Prod. 79:629–661.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gordaliza M: Natural products as leads to
anticancer drugs. Clin Transl Oncol. 9:767–776. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Boufridi A and Quinn RJ: Harnessing the
properties of natural products. Annu Rev Pharmacol Toxicol.
58:451–470. 2018. View Article : Google Scholar
|
|
11
|
Harvey AL, Edrada-Ebel R and Quinn RJ: The
re-emergence of natural products for drug discovery in the genomics
era. Nat Rev Drug Discov. 14:111–129. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Luo J, Hu YL and Wang H: Ursolic acid
inhibits breast cancer growth by inhibiting proliferation, inducing
autophagy and apoptosis, and suppressing inflammatory responses via
the PI3K/AKT and NF-κB signaling pathways in vitro. Exp Ther Med.
14:3623–3631. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang Y, Huang L, Shi H, Chen H, Tao J,
Shen R and Wang T: Ursolic acid enhances the therapeutic effects of
oxaliplatin in colorectal cancer by inhibition of drug resistance.
Cancer Sci. 109:94–102. 2018. View Article : Google Scholar :
|
|
14
|
Lu J, Wu DM, Zheng YL, Hu B, Zhang ZF, Ye
Q, Liu CM, Shan Q and Wang YJ: Ursolic acid attenuates
D-galactose-induced inflammatory response in mouse prefrontal
cortex through inhibiting AGEs/RAGE/NF-κB pathway activation. Cereb
Cortex. 20:2540–2548. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kashyap D, Sharma A, Tuli HS, Punia S and
Sharma AK: Ursolic acid and oleanolic acid: Pentacyclic Terpenoids
with promising anti-inflammatory activities. Recent Pat Inflamm
Allergy Drug Discov. 10:21–33. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liobikas J, Majiene D, Trumbeckaite S,
Kursvietiene L, Masteikova R, Kopustinskiene DM, Savickas A and
Bernatoniene J: Uncoupling and antioxidant effects of ursolic acid
in isolated rat heart mitochondria. J Nat Prod. 74:1640–1644. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shanmugam MK, Manu KA, Ong TH,
Ramachandran L, Surana R, Bist P, Lim LH, Kumar AP, Hui KM and
Sethi G: Inhibition of CXCR4/CXCL12 signaling axis by ursolic acid
leads to suppression of metastasis in transgenic adenocarcinoma of
mouse prostate model. Int J Cancer. 129:1552–1563. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yang LJ, Tang Q, Wu J, Chen Y, Zheng F,
Dai Z and Hann SS: Inter-regulation of IGFBP1 and FOXO3a unveils
novel mechanism in ursolic acid-inhibited growth of hepatocellular
carcinoma cells. J Exp Clin Cancer Res. 35:592016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Senthil S, Chandramohan G and Pugalendi
KV: Isomers (oleanolic and ursolic acids) differ in their
protective effect against isoproterenol-induced myocardial ischemia
in rats. Int J Cardiol. 119:131–133. 2007. View Article : Google Scholar
|
|
20
|
Raphael TJ and Kuttan G: Effect of
naturally occurring triterpenoids ursolic acid and glycyrrhizic
acid on the cell-mediated immune responses of metastatic
tumor-bearing animals. Immunopharmacol Immunotoxicol. 30:243–255.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jiménez-Arellanes A, Luna-Herrera J,
Cornejo-Garrido J, López-García S, Castro-Mussot ME, Meckes-Fischer
M, Mata-Espinosa D, Marquina B, Torres J and Hernández-Pando R:
Ursolic and oleanolic acids as antimicrobial and immunomodulatory
compounds for tuberculosis treatment. BMC Complement Altern Med.
13:2582013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mancha-Ramirez AM and Slaga TJ: Ursolic
acid and chronic disease: An overview of UA's effects on prevention
and treatment of obesity and cancer. Adv Exp Med Biol. 928:75–96.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kashyap D, Tuli HS and Sharma AK: Ursolic
acid (UA): A metabolite with promising therapeutic potential. Life
Sci. 146:201–213. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yang K, Chen Y, Zhou J, Ma L, Shan Y,
Cheng X, Wang Y, Zhang Z, Ji X, Chen L, et al: Ursolic acid
promotes apoptosis and mediates transcriptional suppression of
CT45A2 gene expression in non-small-cell lung carcinoma harbouring
EGFR T790M mutations. Br J Pharmacol. 176:4609–4624. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen CJ, Shih YL, Yeh MY, Liao NC, Chung
HY, Liu KL, Lee MH, Chou PY, Hou HY, Chou JS and Chung JG: Ursolic
acid induces apoptotic cell death through AIF and Endo G release
through a mitochondria-dependent pathway in NCI-H292 human lung
cancer cells in vitro. In Vivo. 33:383–391. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Xu CG, Zhu XL, Wang W and Zhou XJ: Ursolic
acid inhibits epithelial-mesenchymal transition in vitro and in
vivo. Pharm Biol. 57:169–175. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Huang CY, Lin CY, Tsai CW and Yin MC:
Inhibition of cell proliferation, invasion and migration by ursolic
acid in human lung cancer cell lines. Toxicol In Vitro.
25:1274–1280. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kondo Y, Kanzawa T, Sawaya R and Kondo S:
The role of autophagy in cancer development and response to
therapy. Nat Rev Cancer. 5:726–734. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen Q, Kang J and Fu C: The independence
of and associations among apoptosis, autophagy, and necrosis.
Signal Transduct Target Ther. 3:182018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Maiuri MC, Zalckvar E, Kimchi A and
Kroemer G: Self-eating and self-killing: Crosstalk between
autophagy and apoptosis. Nat Rev Mol Cell Biol. 8:741–752. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hasima N and Ozpolat B: Regulation of
autophagy by polyphenolic compounds as a potential therapeutic
strategy for cancer. Cell Death Dis. 5:e15092014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Maes H, Rubio N, Garg AD and Agostinis P:
Autophagy: Shaping the tumor microenvironment and therapeutic
response. Trends Mol Med. 19:428–446. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shen S, Zhang Y, Zhang R, Tu X and Gong X:
Ursolic acid induces autophagy in U87MG cells via ROS-dependent
endoplasmic reticulum stress. Chem Biol Interact. 218:28–41. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lin CW, Chin HK, Lee SL, Chiu CF, Chung
JG, Lin ZY, Wu CY, Liu YC, Hsiao YT, Feng CH, et al: Ursolic acid
induces apoptosis and autophagy in oral cancer cells. Environ
Toxicol. 34:983–991. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang M, Zhou A, An T, Kong L, Yu C, Liu J,
Xia C, Zhou H and Li Y: N-Hydroxyphthalimide exhibits antitumor
activity by suppressing mTOR signaling pathway in BT-20 and LoVo
cells. J Exp Clin Cancer Res. 35:412016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tang ZH, Chen X, Wang ZY, Chai K, Wang YF,
Xu XH, Wang XW, Lu JH, Wang YT, Chen XP and Lu JJ: Induction of
C/EBP homologous protein-mediated apoptosis and autophagy by
licochalcone A in non-small cell lung cancer cells. Sci Rep.
6:262412016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pistritto G, Trisciuoglio D, Ceci C,
Garufi A and D'Orazi G: Apoptosis as anticancer mechanism: Function
and dysfunction of its modulators and targeted therapeutic
strategies. Aging (Albany NY). 8:603–619. 2016. View Article : Google Scholar
|
|
38
|
Kabeya Y, Mizushima N, Ueno T, Yamamoto A,
Kirisako T, Noda T, Kominami E, Ohsumi Y and Yoshimori T: LC3, a
mammalian homologue of yeast Apg8p, is localized in autophagosome
membranes after processing. EMBO J. 19:5720–5728. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Janku F, McConkey DJ, Hong DS and Kurzrock
R: Autophagy as a target for anticancer therapy. Nat Rev Clin
Oncol. 8:528–539. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Luo S and Rubinsztein DC: Atg5 and Bcl-2
provide novel insights into the interplay between apoptosis and
autophagy. Cell Death Differ. 14:1247–1250. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Mizushima N, Yoshimori T and Ohsumi Y: The
role of Atg proteins in autophagosome formation. Annu Rev Cell Dev
Biol. 27:107–132. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Fleming A, Noda T, Yoshimori T and
Rubinsztein DC: Chemical modulators of autophagy as biological
probes and potential therapeutics. Nat Chem Biol. 7:9–17. 2011.
View Article : Google Scholar
|
|
43
|
Shimobayashi M and Hall MN: Making new
contacts: The mTOR network in metabolism and signalling crosstalk.
Nat Rev Mol Cell Biol. 15:155–162. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Munson MJ and Ganley IG: MTOR, PIK3C3, and
autophagy: Signaling the beginning from the end. Autophagy.
11:2375–2376. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zoncu R, Efeyan A and Sabatini DM: mTOR:
From growth signal integration to cancer, diabetes and ageing. Nat
Rev Mol Cell Biol. 12:21–35. 2011. View Article : Google Scholar
|
|
46
|
Sarbassov DD, Guertin DA, Ali SM and
Sabatini DM: Phosphorylation and regulation of Akt/PKB by the
rictor-mTOR complex. Science. 307:1098–1101. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Blommaart EF, Krause U, Schellens JP,
Vreeling-Sindelárová H and Meijer AJ: The phosphatidylinositol
3-kinase inhibitors wortmannin and LY294002 inhibit autophagy in
isolated rat hepatocytes. Eur J Biochem. 243:240–246. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Ge D, Han L, Huang S, Peng N, Wang P,
Jiang Z, Zhao J, Su L, Zhang S, Zhang Y, et al: Identification of a
novel MTOR activator and discovery of a competing endogenous RNA
regulating autophagy in vascular endothelial cells. Autophagy.
10:957–971. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Aoki H, Takada Y, Kondo S, Sawaya R,
Aggarwal BB and Kondo Y: Evidence that curcumin suppresses the
growth of malignant gliomas in vitro and in vivo through induction
of autophagy: Role of Akt and extracellular signal-regulated kinase
signaling pathways. Mol Pharmacol. 72:29–39. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Cagnol S and Chambard JC: ERK and cell
death: Mechanisms of ERK-induced cell death-apoptosis, autophagy
and senescence. FEBS J. 277:2–21. 2010. View Article : Google Scholar
|
|
51
|
Wirawan E, Vanden Berghe T, Lippens S,
Agostinis P and Vandenabeele P: Autophagy: For better or for worse.
Cell Res. 22:43–61. 2012. View Article : Google Scholar :
|
|
52
|
Li Y, Xing D, Chen Q and Chen WR:
Enhancement of chemotherapeutic agent-induced apoptosis by
inhibition of NF-kappaB using ursolic acid. Int J Cancer.
127:462–473. 2010.
|
|
53
|
Shan J, Xuan Y, Zhang Q, Zhu C, Liu Z and
Zhang S: Ursolic acid synergistically enhances the therapeutic
effects of oxaliplatin in colorectal cancer. Protein Cell.
7:571–585. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Shin SW, Kim SY and Park JW: Autophagy
inhibition enhances ursolic acid-induced apoptosis in PC3 cells.
Biochim Biophys Acta. 1823:451–457. 2012. View Article : Google Scholar
|
|
55
|
Leng S, Hao Y, Du D, Xie S, Hong L, Gu H,
Zhu X, Zhang J, Fan D and Kung HF: Ursolic acid promotes cancer
cell death by inducing Atg5-dependent autophagy. Int J Cancer.
133:2781–2790. 2013.PubMed/NCBI
|
|
56
|
Zhao C, Yin S, Dong Y, Guo X, Fan L, Ye M
and Hu H: Autophagy-dependent EIF2AK3 activation compromises
ursolic acid-induced apoptosis through upregulation of MCL1 in
MCF-7 human breast cancer cells. Autophagy. 9:196–207. 2013.
View Article : Google Scholar :
|
|
57
|
Mizushima N and Yoshimori T: How to
interpret LC3 immunoblotting. Autophagy. 3:542–545. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Galluzzi L, Bravo-San Pedro JM, Levine B,
Green DR and Kroemer G: Pharmacological modulation of autophagy:
Therapeutic potential and persisting obstacles. Nat Rev Drug
Discov. 16:487–511. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Wiedmer T, Blank A, Pantasis S, Normand L,
Bill R, Krebs P, Tschan MP, Marinoni I and Perren A: Autophagy
inhibition improves sunitinib efficacy in pancreatic neuroendocrine
tumors via a lysosome-dependent mechanism. Mol Cancer Ther.
16:2502–2515. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang L, Qiang P, Yu J, Miao Y, Chen Z, Qu
J, Zhao Q, Chen Z, Liu Y, Yao X, et al: Identification of compound
CA-5f as a novel late-stage autophagy inhibitor with potent
anti-tumor effect against non-small cell lung cancer. Autophagy.
15:391–406. 2019. View Article : Google Scholar :
|
|
61
|
Deng S, Shanmugam MK, Kumar AP, Yap CT,
Sethi G and Bishayee A: Targeting autophagy using natural compounds
for cancer prevention and therapy. Cancer. 125:1228–1246. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Saxton RA and Sabatini DM: mTOR signaling
in growth, metabolism, and disease. Cell. 169:361–371. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Wu WK, Coffelt SB, Cho CH, Wang XJ, Lee
CW, Chan FK, Yu J and Sung JJ: The autophagic paradox in cancer
therapy. Oncogene. 31:939–953. 2012. View Article : Google Scholar
|
|
64
|
Wang H, Zhang T, Sun W, Wang Z, Zuo D,
Zhou Z, Li S, Xu J, Yin F, Hua Y and Cai Z: Erianin induces
G2/M-phase arrest, apoptosis, and autophagy via the ROS/JNK
signaling pathway in human osteosarcoma cells in vitro and in vivo.
Cell Death Dis. 7:e22472016. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Wang S, Wang K, Zhang C, Zhang W, Xu Q,
Wang Y, Zhang Y, Li Y, Zhang Y, Zhu H, et al: Overaccumulation of
p53-mediated autophagy protects against betulinic acid-induced
apoptotic cell death in colorectal cancer cells. Cell Death Dis.
8:e30872017. View Article : Google Scholar : PubMed/NCBI
|