Introduction
Hepatocellular carcinoma (HCC) is the fifth most
frequent cancer type and the second most common cause of
cancer-associated mortality globally (1), and HCC cases in China account for
~55% of all HCC cases worldwide (2). Every year, >780,000 novel HCC
cases are reported globally, causing ~745,000 mortalities (3,4).
Recent advances in the treatment of HCC, including surgical
excision, liver transplantation, targeted drug therapy and
chemoradiotherapy, have considerably improved the quality of life
of the patient and their prognosis (5). However, the long-term clinical
outcomes of patients with HCC is one of the shortest among all
human cancer types (6). For
instance, the survival rate of patients with HCC is <5%, with
>50% cases experiencing recurrence and metastasis (7). Multiple risk factors, such as
hepatitis B and C virus infections, long-term alcoholism,
non-alcoholic fatty liver disease and consumption of food
contaminated by Aspergillus flavus, are closely associated
with HCC and its progression (8,9).
However, the processes underlying HCC pathogenesis remain largely
unknown and require further elucidation. Therefore, it is important
to acquire an in-depth understanding of the mechanisms associated
with HCC progression, which may help identify a promising strategy
for tumor prevention and therapy.
Long non-coding RNAs (lncRNAs) are a diverse group
of non-coding RNA transcripts comprising >200 nucleotides
(10). lncRNAs do not encode
proteins but are widely distributed in somatic cells, and can
interact with DNA, RNA or proteins to modulate gene expression at
transcriptional and post-transcriptional levels (11). A previous review has shown that
lncRNAs serve important roles in numerous biological processes,
including differentiation, proliferation and metabolism, and also
have crucial functions in disease pathogeneses, particu-larly
cancer (12). Differentially
expressed lncRNAs have been observed in HCC by several studies
(13–15), suggesting a crucial role of
lncRNAs in HCC. Moreover, lncRNAs exhibit cancer-inhibiting or
cancer-promoting activities and are implicated in the regulation of
pathological processes in malignant cells (16).
MicroRNAs (miRNAs/miRs) are widely expressed in
eukaryotes and are a family of endogenous, highly conserved and
single-stranded small non-coding RNA molecules comprising ~23
nucleotides (17). miRNAs
effectively regulate genes by directly binding to the
3′-untranslated regions (UTRs) of their target genes, resulting in
mRNA degradation and/or translation inhibition (18). Previous studies have reported that
numerous miRNAs contribute to HCC oncogenesis and progression
(19–21). For example, miR-375 (22), miR-369-3p (23) and miR-499 (24) are weakly expressed in HCC, and
execute anti-oncogenic actions. It has been revealed that lncRNAs
harbor miRNA-response elements and can serve as competing
endogenous RNAs (ceRNAs) or molecular sponges to modulate miRNAs
and liberate their binding to target mRNAs (25).
The present study measured alterations in muskelin 1
antisense RNA (MKLN1-AS) expression in HCC and evaluated its
clinical value in patients with HCC. In addition, the current study
investigated the role of MKLN1-AS in regulating the malignant
features of HCC cells, and the detailed molecular mechanisms
responsible for the cancer-promoting actions of MKLN1-AS in HCC
were elucidated.
Materials and methods
Tissue specimens
The present study was approved by the Ethics
Committee of Shibo High-Tech Hospital and was performed in
accordance with the Declaration of Helsinki. Written informed
consent was obtained from all participants. HCC tissues and
corresponding adjacent healthy tissues (≥3 cm) were collected from
65 patients (39 males; 26 females; age range, 48–72 years) admitted
to Shibo High-Tech Hospital between June 2013 and February 2015.
None of the patients were previously diagnosed with other human
cancer types or received anticancer therapies. All tissue specimens
were snap-frozen in liquid nitrogen (−196°C) immediately after
tissue excision and immersed in liquid nitrogen.
Cell culture
Transformed Human Liver Epithelial-3 cells (THLE-3)
and HCC cell lines (SNU-182, HuH7 and Hep3B) were acquired from the
Cell Bank of the Chinese Academy of Sciences. THLE-3 cells were
cultured in Bronchial Ep ithelial Growth Medium (Clonetics
Corporation) that was supplemented with 10% heat-inactivated FBS
(Gibco; Thermo Fisher Scientific, Inc.), 5 ng/ml epidermal growth
factor and 70 ng/ml Phosphoethanolamine. The HCC cell line SNU-398
(American Type Culture Collection) was cultured in RPMI-1640
(Gibco; Thermo Fisher Scientific, Inc.) containing 10% FBS (Gibco;
Thermo Fisher Scientific, Inc.), 100 U/ml penicillin and 100 ng/ml
streptomycin (Gibco; Thermo Fisher Scientific, Inc.). HuH7 and
SNU-182 cells were cultured in DMEM (Gibco; Thermo Fisher
Scientific, Inc.) and RPMI-1640, respectively, which were
supplemented with 10% FBS, 1% GlutaMAX, 1% non-essential amino
acids, 100 U/ml penicillin and 100 ng/ml streptomycin. Minimal
essential media (Gibco; Thermo Fisher Scientific, Inc.)
supplemented with 10% FBS, 1% GlutaMAX, 1% non-essential amino
acids, 1% sodium pyruvate 100 mM solution, 100 U/ml penicillin and
100 ng/ml streptomycin was used to culture Hep3B cells. All cells
were maintained at 37°C in a humidified atmosphere containing 5%
CO2.
Cell transfection
miR-654-3p mimic and miR-654-3p inhibitor were
acquired from Shanghai GenePharma Co., Ltd., and were used to
upregulate and downregulate miR-654-3p expression, respectively.
Negative control (NC) miRNA mimic (miR-NC) and NC inhibitor served
as the control for miR-654-3p mimic and miR-654-3p inhibitor,
respectively. The miR-654-3p mimics sequence was
5′-UUCCACUACCAGUCGUCUGUAU-3′ and the miR-NC sequence was
5′-UUGUACUACACAAAAGUACUG-3′. The miR-654-3p inhibitor sequence was
5′-AAGGUGAUGGUCAGCAGACAUA-3′ and the NC inhibitor sequence was
5′-ACUACUGAGUGACAGUAGA-3′. Small interfering RNAs (siRNAs)
specifically targeting MKLN1-AS (si-MKLN1-AS) and NC siRNA (si-NC)
were chemically synthesized by Guangzhou RiboBio Co., Ltd. The
si-MKLN1-AS sequences were as follows: si-MKLN1-AS #1,
5′-TACTAAAAATACAAAAAATTAGC-3′; si-MKLN1-AS #2,
5′-AAACACTTTCAGGATATAATTGG-3′; and si-MKLN1-AS #3,
5′-GACCAAAAATGGGGATCTTTGA-3′. The si-NC sequence was
5′-CACGATAAGACAATGTATTT-3′. The hepatoma-derived growth factor
(HDGF) overexpression plasmid pcDNA3.1-HDGF (pc-HDGF) and empty
pcDNA3.1 plasmid were designed and generated by Shanghai GenePharma
Co., Ltd.
After plating in 6-well plates with a density of
6×105 cells and incubating overnight, cells were
transfected with the miRNA mimic (100 pmol), miRNA inhibitor (100
pmol), siRNA (100 pmol) or plasmid (4 μg) using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) in accordance with the product specifications.
Reverse-transcription quantitative PCR (RT-qPCR), flow cytometry
analysis, migration and invasion assays and western blotting were
conducted after 48 h incubation. A Cell Counting Kit-8 (CCK-8)
assay was performed at 24 h post-transfection.
RT-qPCR
Total RNA isolation was performed using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.), and total RNA was quantified using a NanoDrop 1000
spectrophotometer (NanoDrop Technologies; Thermo Fisher Scientific,
Inc.). For MKLN1-AS and HDGF detection, total RNA was
reverse-transcribed into first-strand cDNA using PrimeScript™ RT
reagent kit (Takara Biotechnology Co., Ltd.). The temperature
protocol for reverse transcription was as follows: 37°C for 15 min
and 85°C for 5 sec. Then, cDNA was subjected to qPCR using
SYBR® Premix Ex Taq™ (Takara Biotechnology Co., Ltd.).
The thermocycling conditions were as follows: Initial denaturation
for 5 min at 95°C, followed by 40 cycles of 95°C for 30 sec and
65°C for 45 sec.
To determine miR-654-3p expression, cDNA synthesis
and qPCR were performed using the miScript RT kit (Qiagen GmbH) and
miScript SYBR Green PCR kit (Qiagen GmbH), respectively. The
temperature protocols for reverse transcription were as follows:
37°C for 60 min, 95°C for 5 min and kept at 4°C. The thermocycling
conditions for qPCR were as follows: Initial denaturation at 95°C
for 2 min, followed by 40 cycles at 95°C for 10 sec, 55°C for 30
sec and 72°C for 30 sec. GAPDH served as the internal reference for
MKLN1-AS and HDGF, and miR-654-3p expression was normalized to U6
small nuclear RNA expression. All data were analyzed using the
2-ΔΔCq method (26).
The qPCR primer sequences were as follows: MKLN1-AS
forward, 5′-AAAGAGTATGTCGCTTATTGTCTAAGA-3′ and reverse,
5′-ATCCTGCTGACTTACTCCAGATGT-3′; HDGF forward,
5′-AATCAACAGCCAACAAATACCAAGT-3′ and reverse,
5′-AGCCTTGACAGTAGGGTTGTTCTC-3′; GAPDH forward,
5′-CGGAGTCAACGGATTTGGTCGTAT-3′ and reverse,
5′-AGCCTTCTCCATGGTGGTGAAGAC-3′; miR-654-3p forward,
5′-TCGGCAGGUGGUGGGCCGCAG-3′ and reverse,
5′-CACTCAACTGGTGTCGTGGA-3′; and U6 forward,
5′-GCTTCGGCAGCACATATACTAAAAT-3′ and reverse,
5′-CGCTTCACGAATTTGCGTGTCAT-3′.
Nuclear/cytoplasmic fractionation
HCC cells (2×106 cells) were collected,
and cytoplasmic and nuclear fractions were separated using a
Cytoplasmic and Nuclear RNA Purification kit (Norgen Biotek Corp.).
RT-qPCR was performed as aforementioned to determine relative
MKLN1-AS expression in cytoplasmic and nuclear fractions.
CCK-8 assay
CCK-8 assay (Dojindo Molecular Technologies, Inc.)
was performed according to the manufacturer's instructions.
Transfected cells were harvested after being incubated for 24 h. A
cell suspension was prepared, and its concentration was adjusted to
2×104 cells/ml. Cells were seeded into 96-well plates at
a volume of 100 μl and cultured at 37°C with 5%
CO2 for 0, 24, 48 and 72 h. A total of 10 μl
CCK-8 reagent was added into each well, followed by an additional
incubation for 2 h at 37°C with 5% CO2. The absorbance
was then measured at 450 nm using a microplate reader (Bio-Rad
Laboratories, Inc.).
Flow cytometry analysis
Transfected cells were digested with EDTA acid-free
0.25% trypsin and rinsed with PBS at 4°C. The cells were
centrifuged at room temperature for 5 min at 12,000 × g, the
supernatant was discarded and apoptosis of the collected cells was
determined using an Annexin V-FITC apoptosis detection kit
(Biolegend, Inc.). Cells were suspended in 100 μl binding
buffer and then stained with 10 μl Annexin V-FITC and 5
μl PI at room temperature for 15 min in the dark. The
percentage of early + late apoptotic cells were detected using a
flow cytometer (FACScan; BD Biosciences). Data was analyzed with
the CellQuest software (version 2.9; BD Biosciences).
Migration and invasion assays
For migration assays, transfected cells were
collected at 48 h post-transfection and resuspended in basal medium
without FBS. The upper compartments of Transwell chambers (size, 8
μM diameter; Corning, Inc.) were loaded with 200 μl
cell suspension containing 5×104 cells, while 600
μl culture medium supplemented with 20% FBS was added into
the lower compartments. After culturing for 24 h, the non-migrated
cells remaining in the upper surface of the membrane were removed
with a cotton swab, and the migrated cells were fixed with 100%
methanol at room temperature for 30 min and stained with 0.5%
crystal violet at room temperature for 30 min. After extensive
washing with PBS, the stained cells in five random fields at ×200
magnification were imaged and counted using an inverted light
microscope (Olympus Corporation). For invasion assays, 40 μl
Matrigel (BD Biosciences) was used to pre-coat the Transwell
chambers at 37°C for 2 h, and the aforementioned steps were
repeated.
Tumor xenograft experiments
To obtain a MKLN1-AS stable knockdown HCC cell line,
lentiviral expression vector stably expressing MKLN1-AS-short
hairpin RNA (shRNA; sh-MKLN1-AS) and NC-shRNA (sh-NC) was acquired
from Shanghai GenePharma Co., Ltd., mixed with polybrene (5
μg/ml; Sigma-Aldrich; Merck KGaA) and transfected into
SNU-398 cells with MOI=5. Subsequently, 1 μg/ml puromycin
was applied at 37°C to incubate the transfected cells for 4 weeks,
and select MKLN1-AS stably silenced SNU-398 cells.
The experimental procedures for animal studies were
approved by the Animal Experiment Administration Committee of Shibo
High-Tech Hospital. Male BALB/c nude mice (n=6; age, 4–6 weeks;
weight, 20 g) were obtained from Hunan SJA Laboratory Animal Co.,
Ltd (Hunan, China) and raised in a specific pathogen-free
environment at 25°C with 50% humidity, with a 10/14-h light/dark
cycle and ad libitum food and water access. A total of
5×106 SNU-398 cells stably expressing sh-MKLN1 or sh-NC
were collected, resuspended in PBS and subcutaneously injected into
the flanks of nude mice. The growth of tumor xenografts was
monitored weekly using a caliper, and their volume was analyzed
using the following formula: Volume (mm3) = 0.5 × width
× length2. All nude mice were euthanized via cervical
dislocation at 4 weeks post-cell injection, and tumor xenografts
were resected and weighed.
Bioinformatics analysis
Gene Expression Profiling Interactive Analysis
version 2.0 (GEPIA2; http://gepia.cancer-pku.cn), which included the data
from The Cancer Genome Atlas (TCGA) and Genotype Tissue Expression
(GTEx) projects, was used to assess the expression pattern of
MKLN1-AS in HCC. The location of MKLN1-AS in cells was predicted
using lncLocator (http://www.csbio.sjtu.edu.cn/bioinf/lncLocator/).
The online database StarBase version 2.0 (http://star-base.sysu.edu.cn/) was used to identify
the target miRNAs of MKLN1-AS. The putative targets of miR-654-3p
were predicted using miRDB (Verison 6.0; http://mirdb.org/), TargetScan (Release 7.2: March
2018; http://www.targetscan.org/vert_72) and StarBase
version 2.0.
RNA immunoprecipitation (RIP) assay
RIP assays were performed using the Magna RIP™ RNA
Binding Protein Immunoprecipitation kit (EMD Millipore). HCC cells
(~1×107) were collected and lysed in RIP lysis buffer.
After centrifugation 10,000 × g at 4°C for 5 min, cell lysates were
incubated overnight at 4°C with magnetic beads conjugated to human
Argonaute-2 (Ago2) or control immunoglobulin G (IgG) antibodies
(1:5,000; both from cat. no. 03–110; EMD Millipore). The magnetic
beads were treated with 0.5 mg/ml protease K 30 min at 55°C to
digest the protein. The extracted immunoprecipitated RNA was
subjected to RT-qPCR analysis for MKLN1-AS and miR-654-3p
enrichment determination.
Luciferase reporter assay
The target fragments of MKLN1-AS containing the
wild-type (WT) miR-654-3p binding site and mutant (MUT) MKLN1-AS
fragments were constructed and subcloned into the pmirGLO
luciferase reporter plasmid (Promega Corporation) to generate the
recombinant luciferase reporter plasmids, MKLN1-AS-WT and
MKLN1-AS-MUT. The recombinant luciferase reporter plasmids HDGF-WT
and HDGF-MUT were also generated using the same experimental
protocol. For reporter assays, WT or MUT luciferase reporter
plasmids (0.2 μg) were transfected into HCC cells in the
presence of miR-654-3p mimic (20 pmol) or miR-NC (20 pmol) using
Lipofectamine® 2000. At 48 h after transfection,
luciferase activity was determined using a dual-luciferase reporter
assay system (Promega Corporation), and normalized to that of
Renilla luciferase activity.
Western blot analysis
Transfected cells or homogenized tumor xenografts
were lysed in RIPA lysis buffer (Beyotime Institute of
Biotechnology), and then subjected to centrifugation 10,000 × g at
4°C for 5 min to harvest the supernatant and extract total protein.
Total protein quantification was performed with a BCA Protein Assay
kit (Sangon Biotech Co., Ltd.). Equal amounts of protein (30
μg) were subjected to 10% SDS-PAGE. Then, the targeted
proteins were transfected to PVDF membranes. After blocking in 5%
non-fat dried milk diluted in TBS-0.05% Tween-20 at room
temperature for 2 h, the membranes were incubated overnight at 4°C
with primary antibodies targeting HDGF (cat. no. ab128921; 1:1,000;
Abcam) or GAPDH (cat. no. ab8245; 1:1,000; Abcam). The goat
anti-mouse (cat. no. ab205719; 1:5,000; Abcam) and goat anti-rabbit
(cat. no. ab6721; 1:5,000; Abcam) IgG horseradish
peroxidase-conjugated secondary antibody was incubated with
membranes at room temperature for 2 h. Subsequently, positive bands
were detected using an ECL Advance western blotting detection kit
(Cytiva). Quantity One software version 4.62 (Bio-Rad Laboratories,
Inc.) was used for densitometry.
Statistical analysis
All data from three biological replicates for each
experiment are presented as the mean ± SD. Both paired and unpaired
Student's t-test was used for comparing differences between the two
groups. Comparisons among multiple groups were conducted using
one-way ANOVA followed by Tukey's test. Survival curves were
plotted using the Kaplan-Meier method, after which the curves were
compared using the log-rank test. The correlation between MKLN1-AS
and miR-654-3p expression was analyzed using Pearson's correlation
coefficient. P<0.05 was considered to indicate a statistically
significant difference.
Results
MKLN1-AS is upregulated in HCC and
associated with poor prognosis
The GEPIA2 database, which includes TCGA and GTEx
data, was used to assess the expression profile of MKLN1-AS in HCC.
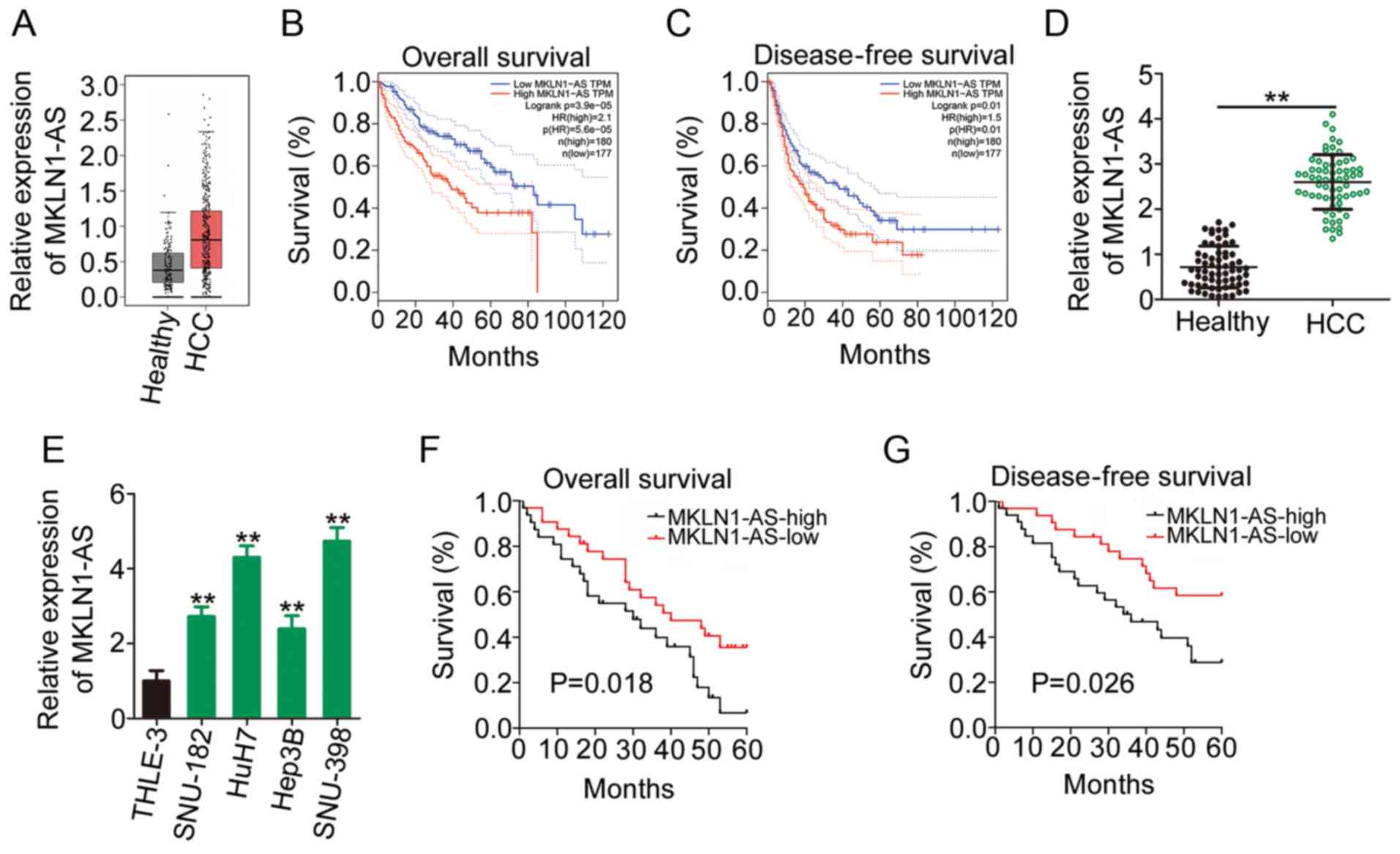
The outcomes of the box plots (Fig.
1A) demonstrated that MKLN1-AS was highly expressed in HCC
tissues (n=369) compared with that in healthy liver tissues
(n=160). Additionally, patients with HCC exhibiting high MKLN1-AS
expression had shorter overall survival (Fig. 1B) and disease-free survival rates
(Fig. 1C) compared with those
exhibiting low MKLN1-AS expression, as indicated by the GEPIA2
database.
To confirm the aforementioned findings, 65 pairs of
HCC tissues and corresponding adjacent healthy tissues were
collected and used for MKLN1-AS quantification. RT-qPCR analysis
identified that MKLN1-AS expression was higher in HCC tissues
compared with adjacent healthy tissues (Fig. 1D). Similarly, MKLN1-AS expression
in four HCC cell lines (SNU-182, HuH7, Hep3B and SNU-398) was
increased compared with that in THLE-3 cells (Fig. 1E).
To further evaluate the clinical significance of
MKLN1-AS in HCC, the median value of MKLN1-AS expression was
regarded as the cutoff, and all 65 patients with HCC were
classified into either MKLN1-AS-low or MKLN1-AS-high expression
groups. The overall survival periods were shorter (Fig. 1F) and disease-free survival rates
were lower (Fig. 1G) in the
MKLN1-AS-high expression group compared with the MKLN1-AS-low
expression group. Thus, these results indicated that MKLN1-AS may
serve a key role in HCC oncogenesis and progression.
MKLN1-AS knockdown inhibits the
proliferation, migration and invasion, as well as promotes the
apoptosis of HCC cells
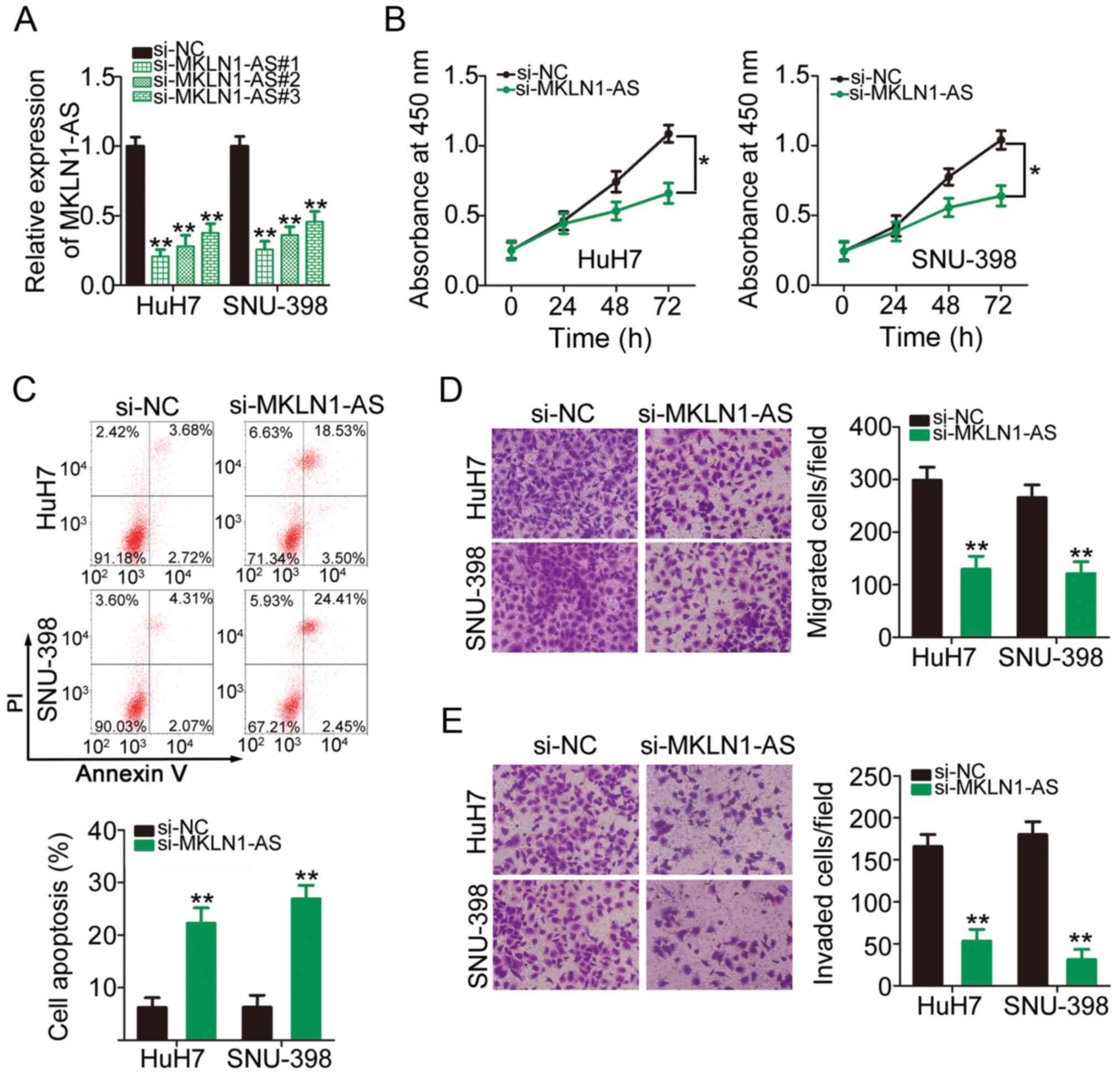
To determine the detailed role of MKLN1-AS in HCC,
MKLN1-AS was knockdown in HuH7 and SNU-398 cells via transfection
with si-MKLN1-AS (Fig. 2A). In
particular, si-MKLN1-AS#1 exhibited the highest silencing efficacy
among the three siRNAs and was therefore selected for subsequent
functional assays. A CCK-8 assay was conducted to determine HCC
cell proliferation. Knockdown of MKLN1-AS expression significantly
suppressed HuH7 and SNU-398 cell proliferation compared with the NC
(Fig. 2B). Cell apoptosis
analysis demonstrated that the apoptosis of HuH7 and SNU-398 cells
was significantly increased following MKLN1-AS knockdown (Fig. 2C). Moreover, the role of MKLN1-AS
in the migratory and invasive abilities of HCC cells was assessed
using migration and invasion assays. It was found that the
migration (Fig. 2D) and invasion
(Fig. 2E) of HuH7 and SNU-398
cells were inhibited after MKLN1-AS knockdown. Collectively, these
results suggested that MKLN1-AS exerts tumor-promoting activities
in HCC cells.
MKLN1-AS acts a molecular sponge for
miR-654-3p in HCC cells
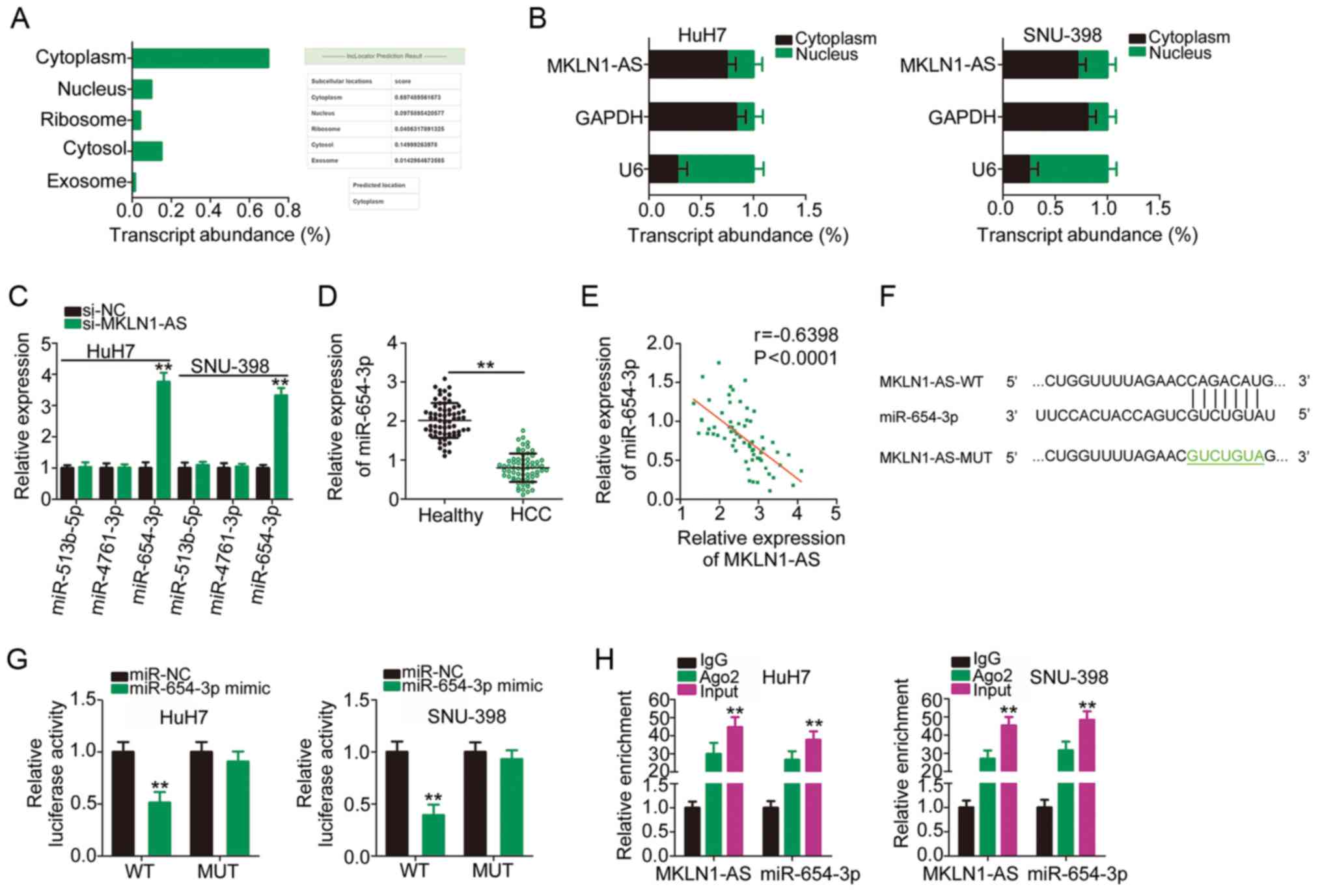
Previous studies have reported that lncRNAs execute
their roles in the cytoplasm of human cancer cells by acting as
small molecular sponges or ceRNAs (27,28). To determine the localization of
MKLN1-AS expression, an lncRNA subcellular localization predictor,
lncLocator, was used. MKLN1-AS was predicted to be mainly located
in the cytoplasm (Fig. 3A), which
was confirmed by the nuclear/cytoplasmic fractionation experiments
(Fig. 3B). These results
suggested a theoretical basis for MKLN1-AS as a sponge of miRNAs.
StarBase 2.0 was used to identify the putative binding miRNAs of
MKLN1-AS. It was found that MKLN1-AS has elements complementary to
three miRNAs, including miR-513b-5p, miR-4761-3p and miR-654-3p.
RT-qPCR analysis demonstrated that, compared with si-NC, only
miR-654-3p expression was significantly increased when MKLN1-AS was
knockdown (Fig. 3C). Accordingly,
miR-654-3p was selected for further experimental verification.
miR-654-3p expression was detected in the 65 pairs
of HCC tissues and corresponding adjacent healthy tissues using
RT-qPCR, and it was identified that miR-654-3p was downregulated in
HCC tissues compared with healthy tissues (Fig. 3D). Additionally, a moderate
negative correlation between miR-654-3p and MKLN1-AS expression
levels was identified in the 65 HCC tissues (r=−0.6398;
P<0.0001; Fig. 3E). Fig. 3F presents the predicted binding
sequences of miR-654-3p within MKLN1-AS, and a luciferase reporter
assay was used to validate the binding relationship between
miR-654-3p and MKLN1-AS in HCC cells. The results indicated that
miR-654-3p overexpression significantly decreased the luciferase
activity of MKLN1-AS-WT in HuH7 and SNU-398 cells; however, a
mutation at the binding site abrogated the inhibitory effect
(Fig. 3G). Furthermore, compared
with IgG antibody, miR-654-3p and MKLN1-AS were significantly
enriched in the compound precipitated by anti-Ago2, as identified
by the RIP assay (Fig. 3H).
Therefore, MKLN1-AS acted as a molecular sponge for miR-654-3p in
HCC cells.
HDGF is directly targeted by miR-654-3p
in HCC cells and is under positive modulation of MKLN1-AS via
competitive sponging of miR-654-3p
To investigate the biological roles of miR-654-3p in
HCC cells, miR-654-3p was overexpressed in HuH7 and SNU-398 cells
via transfection with miR-654-3p mimic (Fig. 4A). CCK-8 assay and flow cytometry
analysis results demonstrated that transfection with miR-654-3p
mimic in HuH7 and SNU-398 cells significantly decreased
proliferation (Fig. 4B) and
increased apoptosis (Fig. 4C).
Using migration and invasion assays, it was observed that the
number of migrated (Fig. 4D) and
invaded (Fig. 4E) cells was
declined in HuH7 and SNU-398 cells after miR-654-3p
overexpression.
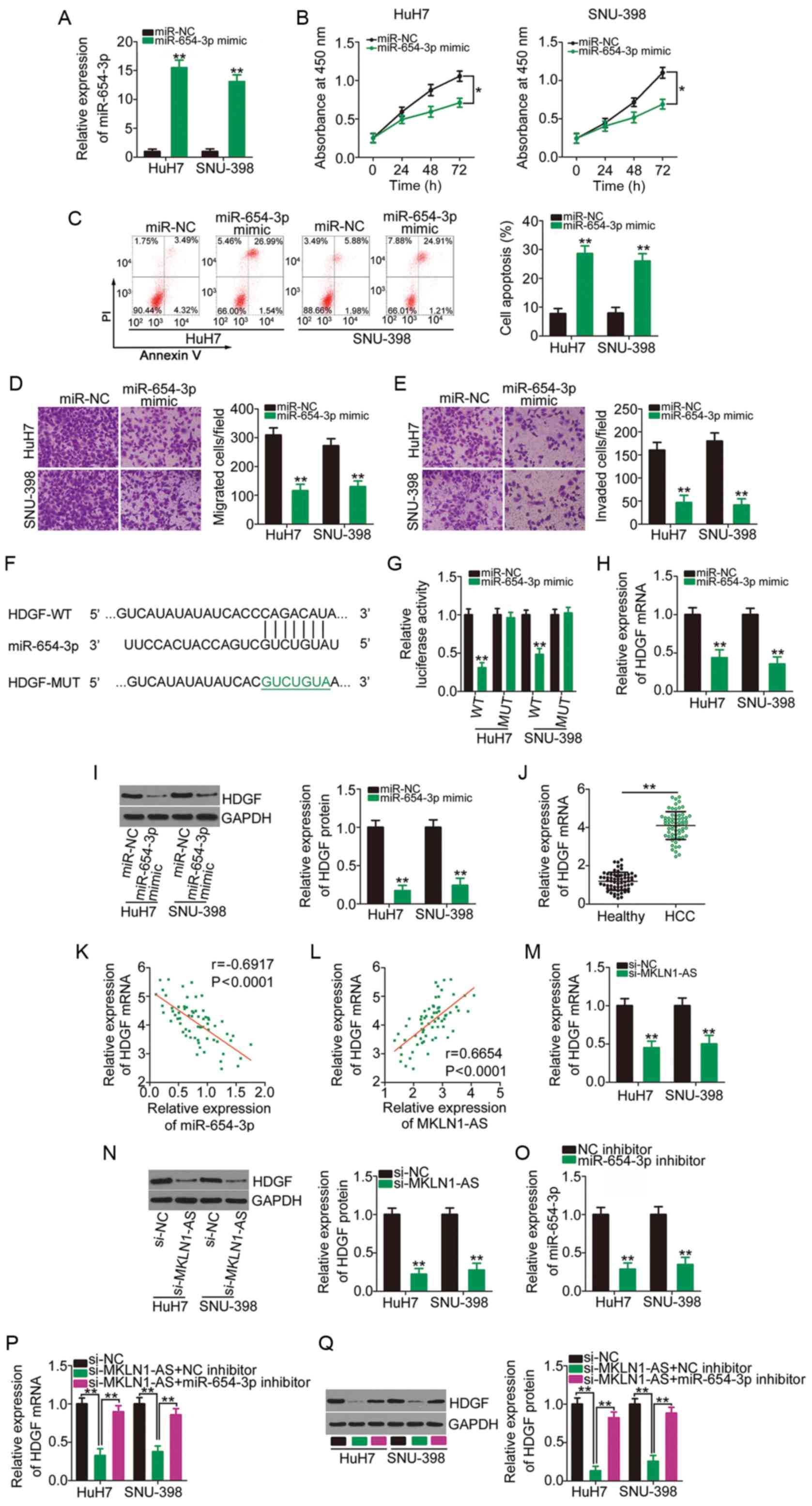 | Figure 4MKLN1-AS positively regulates HDGF
expression in HCC cells via competitive sponging of miR-654-3p. (A)
HuH7 and SNU-398 cells were transfected with miR-654-3p mimic or
miR-NC and subjected to RT-qPCR to determine transfection
efficiency. **P<0.01 compared with miR-NC. (B)
Proliferation and (C) apoptosis of miR-654-3p-overexpressing HuH7
and SNU-398 cells were evaluated using the Cell Counting Kit-8
assay and flow cytometry analysis, respectively.
*P<0.05 compared with miR-NC. **P<0.01
compared with miR-NC. (D) Migration and (E) invasion assays were
used to examine the migratory and invasive capacities of HuH7 and
SNU-398 cells after overexpression of miR-654-3p.
**P<0.01 compared with miR-NC. (F) Schematic
representation of the WT and MUT complementary base pairs between
miR-654-3p and the 3′-untranslated region of HDGF. (G) Luciferase
activity of HDGF-WT or HDGF-MUT was determined in HuH7 and SNU-398
cells transfected with miR-654-3p mimics or miR-NC.
**P<0.01 compared with miR-NC. (H) HDGF mRNA and (I)
protein expression levels were quantified in HuH7 and SNU-398 cells
transfected with miR-654-3p mimics or miR-NC.
**P<0.01 compared with miR-NC. (J) RT-qPCR was
performed to analyze HDGF mRNA expression in 65 pairs of HCC
tissues and corresponding adjacent healthy tissues.
**P<0.01 compared with adjacent healthy tissues. (K)
Correlation between miR-654-3p and HDGF mRNA expression levels in
the 65 HCC tissues was examined via Pearson's correlation
coefficient. (L) Pearson's correlation coefficient analysis of
MKLN1-AS and HDGF mRNA expression levels in the 65 HCC tissues.
Changes in HDGF (M) mRNA and (N) protein expression levels in
MKLN1-AS-depleted HuH7 and SNU-398 cells were determined using
RT-qPCR and western blotting, respectively. **P<0.01
compared with si-NC. (O) Silencing efficiency of miR-654-3p
inhibitor in HuH7 and SNU-398 cells was assessed using RT-qPCR.
**P<0.01 compared with NC inhibitor. miR-654-3p
inhibitor or NC inhibitor was transfected into HuH7 and SNU-398
cells in the presence of si-MKLN1-AS. After transfection, the (P)
mRNA and (Q) protein expression levels of HDGF were measured using
RT-qPCR and western blotting. **P<0.01 compared with
group si-MKLN1-AS + miR-654-3p inhibitor and group si-NC. MKLN1-AS,
muskelin 1 antisense RNA; NC, negative control; si, small
interfering RNA; WT, wild-type; MUT, mutation; miR, microRNA;
RT-qPCR, reverse transcription-quantitative PCR; HDGF,
hepatoma-derived growth factor. |
To elucidate the mechanism underlying the
anti-oncogenic roles of miR-654-3p, the putative targets of
miR-654-3p were predicted using miRDB, TargetScan and StarBase 2.0.
HDGF (Fig. 4F) was identified to
be a putative target of miR-654-3p, and was chosen for further
study considering its oncogenic activities during HCC pathogenesis
(29-31). A luciferase reporter assay was
conducted to confirm the binding of miR-654-3p to the 3′-UTR of
HDGF. The luciferase activity driven by HDGF-WT was significantly
repressed by miR-654-3p overexpression in HuH7 and SNU-398 cells,
while the luciferase activity of HDGF-MUT was unaffected in the two
cell lines after co-transfection with miR-654-3p mimic (Fig. 4G). Furthermore, the results of
RT-qPCR and western blotting indicated that the overexpression of
miR-654-3p significantly decreased HDGF mRNA (Fig. 4H) and protein (Fig. 4I) expression in HuH7 and SNU-398
cells. It was also found that HDGF mRNA expression was
significantly increased in HCC tissues (Fig. 4J) and had a moderate negative
correlation with miR-654-3p expression (r=-0.6917; P<0.0001;
Fig. 4K).
To investigate the ceRNA network of MKLN1-AS, the
association among MKLN1-AS, miR-654-3p and HDGF was examined.
First, the correlation between MKLN1-AS and HDGF mRNA expression
levels was determined, and the results indicated that MKLN1-AS was
moderately, positively correlated with HDGF mRNA expression in HCC
tissues (Fig. 4L; r=0.6654;
P<0.0001). HuH7 and SNU-398 cells were transfected with
si-MKLN1-AS or si-NC, and RT-qPCR and western blotting were used to
determine the changes in HDGF expression. The mRNA (Fig. 4M) and protein (Fig. 4N) expression levels of HDGF were
significantly downregulated in HuH7 and SNU-398 cells after
silencing MKLN1-AS.
A miR-654-3p inhibitor was used in follow-up rescue
experiments, and its transfection efficiency was confirmed using
RT-qPCR. Compared with the NC inhibitor, transfection with
miR-654-3p inhibitor led to a significant decrease in miR-654-3p
expression in HuH7 and SNU-398 cells (Fig. 4O). si-MKLN1-AS, in parallel with
miR-654-3p inhibitor or NC inhibitor, was introduced into HuH7 and
SNU-398 cells, and HDGF mRNA and protein expression levels in each
group were determined. The mRNA (Fig.
4P) and protein (Fig. 4Q)
expression levels of HDGF were significantly decreased following
the transfection with si-MKLN1-AS, which was reversed by miR-654-3p
inhibitor co-transfection. These data suggested that MKLN1-AS
functioned as a ceRNA in HCC cells and competitively binds to
miR-654-3p to increase the expression of the downstream target gene
HDGF.
miR-654-3p inhibition and HDGF
upregulation reverse the anti-oncogenic actions of MKLN1-AS
knockdown in HCC cells
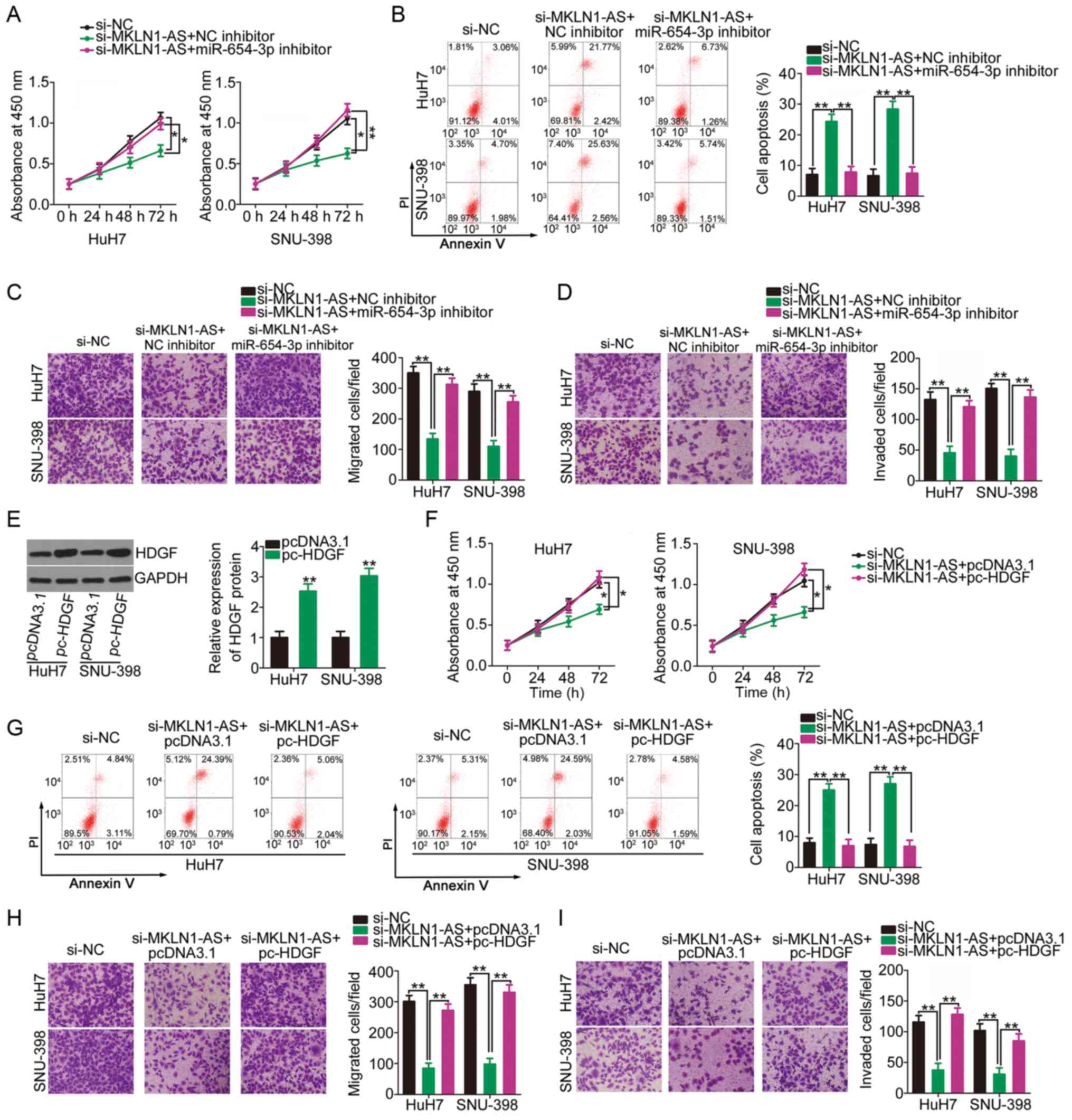
Rescue experiments were performed to examine whether
the biological actions of MKLN1-AS in HCC cells were executed by
regulating the miR-654-3p/HDGF axis. miR-654-3p inhibitor or NC
inhibitor were transfected into MKLN1-AS-deficient HuH7 and SNU-398
cells. Knockdown of MKLN1-AS resulted in a significant decrease in
HuH7 and SNU-398 cell proliferation (Fig. 5A) and an increase in cell
apoptosis (Fig. 5B); however,
these effects were alleviated by co-transfection with miR-654-3p
inhibitor. Furthermore, transfection with si-MKLN1-AS significantly
impaired the migration (Fig. 5C)
and invasion (Fig. 5D) of HuH7
and SNU-398 cells, while these effects were reversed by the
introduction of miR-654-3p inhibitor.
The HDGF overexpression plasmid pc-HDGF was
transfected into HuH7 and SNU-398 cells, resulting in HDGF
upregulation (Fig. 5E).
MKLN1-AS-depleted HuH7 and SNU-398 cells were further transfected
with pc-HDGF or empty pcDNA3.1 plasmid. Changes in proliferation,
apoptosis, migration and invasion were then evaluated. CCK-8 assay
and flow cytometry analysis demonstrated that the suppressed cell
proliferation (Fig. 5F) and
enhanced cell apoptosis (Fig. 5G)
in cells transfected with si-MKLN1-AS were abrogated by HDGF
overexpression. Additionally, the inhibitory activities of MKLN1-AS
knockdown on migration (Fig. 5H)
and invasion (Fig. 5I) of cells
were recovered via co-transfection with pc-HDGF. Collectively, it
was indicated that MKLN1-AS regulated the oncogenicity of HCC cells
via the miR-654-3p/HDGF axis.
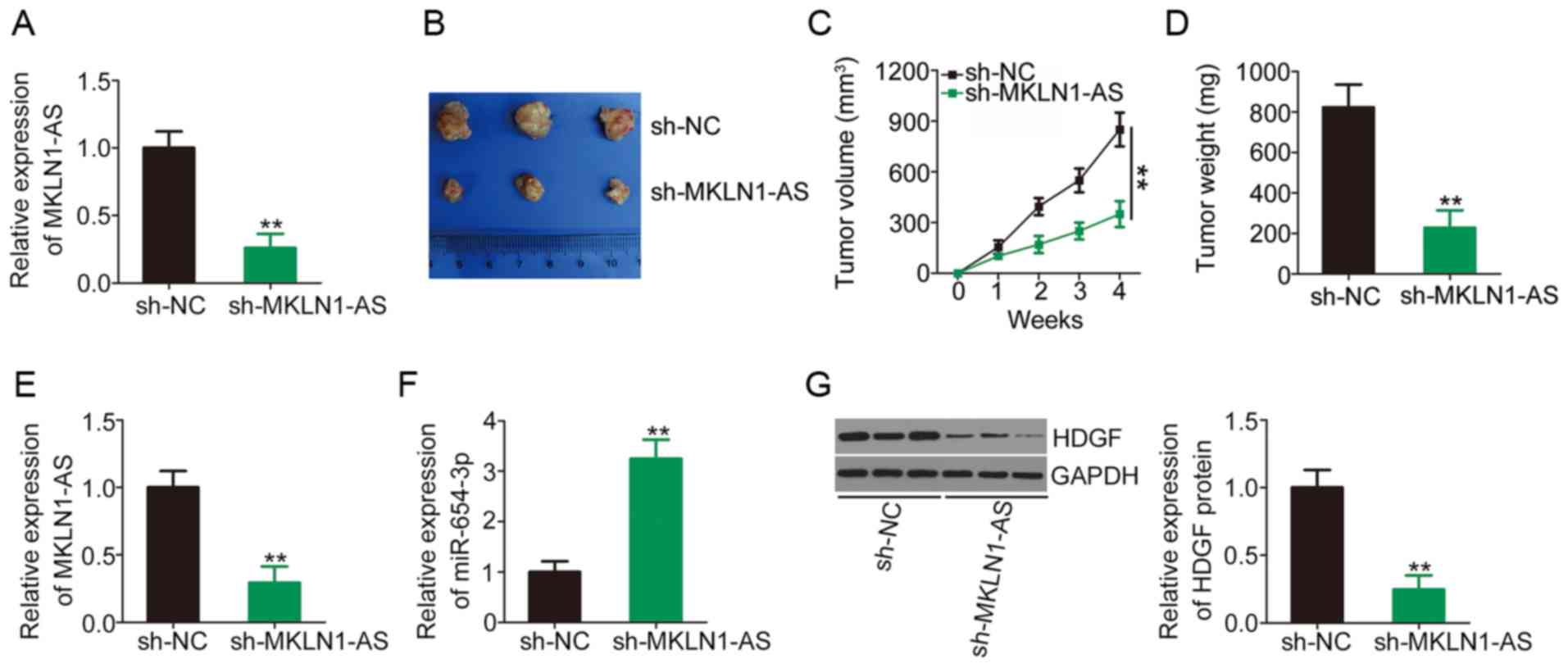
Knockdown of MKLN1-AS inhibits HCC tumor
growth in vivo
To determine the functions of MKLN1-AS in HCC
tumorigenesis in vivo, SNU-398 cells stably expressing sh-NC
or sh-MKLN1-AS were subcutaneously inoculated into nude mice and
allowed to grow for 4 weeks. The efficiency of sh-MKLN1-AS in
silencing MKLN1-AS expression was determined via RT-qPCR and the
results are presented in Fig. 6A.
Tumor growth (Fig. 6B and C) and
weight (Fig. 6D) were
significantly lower in the sh-MKLN1-AS group compared with the
sh-NC group. At 4 weeks post-injection, all mice were euthanized
and tumor xenografts were excised to determine MKLN1-AS, miR-654-3p
and HDGF expression levels. The tumor xenografts developed from
MKLN1-AS depleted-SNU-398 cells had lower MKLN1-AS (Fig. 6E) and higher miR-654-3p (Fig. 6F) expression levels compared with
those of the sh-NC-transfected group. Western blotting detection
indicated that HDGF protein expression was significantly lower in
the tumor xenografts derived from SNU-398 cells stably expressing
sh-MKLN1-AS compared with the sh-NC group (Fig. 6G). These results demonstrated that
MKLN1-AS silencing suppressed HCC tumor growth in vivo.
Discussion
lncRNAs have received increasing attention in the
past decade due to their important roles in human cancer types
(32,33). Numerous lncRNAs, such as ubiquitin
conjugating enzyme E2 R2-AS1 (34), colorectal neoplasia differentially
expressed (35) and snail family
transcriptional repressor 3-AS1 (36), are differentially expressed in HCC
and are closely associated with clinicopathological factors and
prognosis of patients with HCC (37–39). For instance, these lncRNAs
participate in the regulation of various aggressive cellular
features during hepatocarcinogenesis and cancer progression
(40–42). Hence, identifying HCC-relevant
lncRNAs, as well as understanding their detailed roles are critical
for developing effective targets for the diagnosis, prognosis and
therapy of HCC. However, to date, the functions of most
cancer-associated lncRNAs in HCC remain unknown. Therefore, the
aims of the present study were to determine the expression and
clinical significance of MKLN1-AS in HCC, and examine the detailed
roles and mechanisms of MKLN1-AS in HCC cells.
MKLN1-AS has been reported to be closely associated
with the prognosis of HCC (43).
In the present study, the relationship between MKLN1-AS and the
clinicopathological parameters of patients with HCC was initially
analyzed. It was identified that MKLN1-AS expression was
upregulated in HCC, which is consistent with TCGA dataset. Analysis
of TCGA data and clinical specimens also demonstrated that patients
with HCC exhibiting high MKLN1-AS expression had shorter overall
survival and disease-free survival rates compared with those
exhibiting low MKLN1-AS expression, suggesting that high MKLN1-AS
expression was associated with poor clinical outcomes. The roles of
MKLN1-AS in regulating the malignant features of HCC cells in
vitro and in vivo were also examined. The results
suggested that MKLN1-AS exerts a carcinogenic role in HCC
progression by promoting cell proliferation, migration and
invasion, and inhibiting cell apoptosis in vitro.
Additionally, knockdown of MKLN1-AS expression led to a decrease in
HCC tumor growth in vivo. Collectively, the current results
indicated that MKLN1-AS served a stimulatory role in HCC as an
oncogene.
The potential mechanisms underlying the
tumor-promoting activities of MKLN1-AS in HCC were investigated in
the present study. It is well-established that the roles of lncRNAs
depend on the cellular compartments in which they are located
(44). The lncLocator database
and results of nuclear/cytoplasmic fractionation experiments
demonstrated that MKLN1-AS was located in the cytoplasm, which is
commonly associated with ceRNA function. In general, miRNAs can be
complementary to target mRNAs, leading to mRNA degradation and/or
protein synthesis suppression (45). Moreover, lncRNAs can competitively
bind to miRNAs and consequently modulate miRNA-mediated restriction
of target mRNAs (46).
The present bioinformatics analysis identified that
MKLN1-AS has complementary binding sequences for miR-654-3p. Using
RT-qPCR analysis, miR-654-3p expression was found to be
significantly increased after MKLN1-AS knockdown. In addition,
miR-654-3p expression was downregulated in HCC tissues and
negatively correlated with MKLN1-AS expression. Further study using
luciferase reporter and RIP assays validated the direct binding
between miR-654-3p and MKLN1-AS in HCC cells. Mechanistic
investigations also demonstrated that miR-654-3p targeting of HDGF
was positively modulated by MKLN1-AS, and that a miR-654-3p
inhibitor partially abrogated this regulatory effect in HCC cells.
Collectively, it can be concluded that MKLN1-AS increased HDGF
expression in HCC cells by acting as a ceRNA and competitively
binding to miR-654-3p.
miR-654-3p is downregulated in ovarian cancer
(47), gastric cancer (48) and papillary thyroid cancer
(49), but is upregulated in
osteosarcoma (50). Moreover,
miR-654-3p exerts cancer-inhibiting (47–49) or cancer-promoting roles (50), and is implicated in cancer
oncogenesis and progression (47–50). A recent study reported that
miR-654-3p expression was down-regulated in HCC, which was
associated with poor patient prognosis, and that overexpression of
miR-654-3p restricted cell proliferation, migration and invasion in
HCC (51). HDGF, a
heparin-binding growth factor, was first extracted from conditioned
culture medium collected from the hepatoma cell line HuH7 (52) and was identified as a direct
downstream target of miR-654-3p in HCC cells. HDGF performs
pro-oncogenic roles in HCC and contributes to a number of malignant
characteristics during HCC genesis and progression (29–31). The present results suggested that
HDGF expression was directly regulated by the MKLN1-AS/miR-654-3p
axis in HCC. Functional rescue experiments indicated that the
miR-654-3p/HDGF axis was essential for MKLN1-AS-mediated promotion
of HCC oncogenicity. Therefore, the MKLN1-AS/miR-654-3p/HDGF
functional network was implicated in the development of HCC and was
identified to perform important functions in various malignant
processes.
In conclusion, MKLN1-AS expression is upregulated in
HCC and is associated with unfavorable clinical outcomes.
Additionally, it was found that knockdown of MKLN1-AS expression
suppressed HCC progression. The results suggested that MKLN1-AS
functioned as a ceRNA for miR-654-3p, thus increasing HDGF
expression. Collectively, the present study identified a new
mechanism in HCC pathogenesis and may contribute to the development
of novel strategies for the diagnosis, prognosis, prevention and
treatment of HCC.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
WJG and XHC conceived and designed the study. WJG,
XHC, WC and MX carried out the experiments. WJG and XHC wrote the
paper. All authors reviewed and edited the manuscript. All authors
read and approved the manuscript and agree to be accountable for
all aspects of the research in ensuring that the accuracy or
integrity of any part of the work are appropriately investigated
and resolved.
Ethics approval and consent to
participate
This study was approved by the Ethics Committee of
Shibo High-Tech Hospital. The experimental procedures for animal
studies were approved by the Animal Experiment Administration
Committee of Shibo High-Tech Hospital. Written informed consent was
obtained from all participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
Not applicable.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Goyal L, Qadan M and Zhu AX: Another
treatment option for advanced hepatocellular carcinoma with portal
vein thrombosis in China. JAMA Oncol. 5:938–939. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ferlay J, Parkin DM and Steliarova-Foucher
E: Estimates of cancer incidence and mortality in Europe in 2008.
Eur J Cancer. 46:765–781. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Medavaram S and Zhang Y: Emerging
therapies in advanced hepatocellular carcinoma. Exp Hematol Oncol.
7:172018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ikeda K: Recent advances in medical
management of hepatocellular carcinoma. Hepatol Res. 49:14–32.
2019. View Article : Google Scholar
|
|
7
|
Zhang Q, Wang S, Qiao R, Whittaker MR,
Quinn JF, Davis TP and Li H: Recent advances in magnetic
nanoparticle-based molecular probes for hepatocellular carcinoma
diagnosis and therapy. Curr Pharm Des. 24:2432–2437. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sartorius K, Makarova J, Sartorius B, An
P, Winkler C, Chuturgoon A and Kramvis A: The regulatory role of
microRNA in hepatitis-B virus-associated hepatocellular carcinoma
(HBV-HCC) pathogenesis. Cells. 8:15042019. View Article : Google Scholar
|
|
9
|
Zakharia K, Luther CA, Alsabbak H and
Roberts LR: Hepatocellular carcinoma: Epidemiology, pathogenesis
and surveillance-implications for sub-Saharan Africa. S Afr Med J.
108:35–40. 2018.PubMed/NCBI
|
|
10
|
Hu G, Niu F, Humburg BA, Liao K, Bendi S,
Callen S, Fox HS and Buch S: Molecular mechanisms of long noncoding
RNAs and their role in disease pathogenesis. Oncotarget.
9:18648–18663. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li Y, Egranov SD, Yang L and Lin C:
Molecular mechanisms of long noncoding RNAs-mediated cancer
metastasis. Genes Chromosomes Cancer. 58:200–207. 2019. View Article : Google Scholar
|
|
12
|
Liz J and Esteller M: lncRNAs and
microRNAs with a role in cancer development. Biochim Biophys Acta.
1859:169–176. 2016. View Article : Google Scholar
|
|
13
|
Li W, He Y, Chen W, Man W, Fu Q, Tan H,
Guo H, Zhou J and Yang P: Knockdown of LINC00467 contributed to
Axitinib sensitivity in hepatocellular carcinoma through
miR-509-3p/PDGFRA axis. Gene Ther. Mar 27–2020.Epub ahead of print.
View Article : Google Scholar
|
|
14
|
Lu Z, Yu Y, Ding X, Jin D, Wang G, Zhou Y,
Zhu Y, Na L, He Y and Wang Q: LncRNA FLJ33360 accelerates the
metastasis in hepatocellular carcinoma by targeting miRNA-140/MMP9
axis. Am J Transl Res. 12:583–591. 2020.PubMed/NCBI
|
|
15
|
Liu C, Zhang M, Zhao J, Zhu X, Zhu L, Yan
M, Zhang X and Zhang R: LncRNA FOXD3-AS1 mediates AKT pathway to
promote growth and invasion in hepatocellular carcinoma through
regulating RICTOR. Cancer Biother Radiopharm. 35:292–300. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen X, Tang FR, Arfuso F, Cai WQ, Ma Z,
Yang J and Sethi G: The emerging role of long non-coding RNAs in
the metastasis of hepatocellular carcinoma. Biomolecules.
10:662019. View Article : Google Scholar
|
|
17
|
Xu X, Tao Y, Shan L, Chen R, Jiang H, Qian
Z, Cai F, Ma L and Yu Y: The role of microRNAs in hepatocellular
carcinoma. J Cancer. 9:3557–3569. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang T, Yang Z, Kusumanchi P, Han S and
Liangpunsakul S: Critical role of microRNA-21 in the pathogenesis
of liver diseases. Front Med (Lausanne). 7:72020. View Article : Google Scholar
|
|
20
|
Weidle UH, Schmid D, Birzele F and
Brinkmann U: MicroRNAs involved in metastasis of hepatocellular
carcinoma: Target candidates, functionality and efficacy in animal
models and prognostic relevance. Cancer Genomics Proteomics.
17:1–21. 2020. View Article : Google Scholar :
|
|
21
|
Pratama MY, Pascut D, Massi MN and
Tiribelli C: The role of microRNA in the resistance to treatment of
hepatocellular carcinoma. Ann Transl Med. 7:5772019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li D, Wang T, Sun FF, Feng JQ, Peng JJ, Li
H, Wang C, Wang D, Liu Y, Bai YD, et al: MicroRNA-375 represses
tumor angiogenesis and reverses resistance to sorafenib in
hepatocarcinoma. Cancer Gene Ther. July 3–2020.Epub ahead of print.
View Article : Google Scholar
|
|
23
|
Xu Q and Liu K: MiR-369-3p inhibits
tumorigenesis of hepatocellular carcinoma by binding to PAX6. J
Biol Regul Homeost Agents. Jun 30–2020.Epub ahead of print.
|
|
24
|
Wang J, Li J, Chen L, Fan Z and Cheng J:
MicroRNA-499 suppresses the growth of hepatocellular carcinoma by
downregulating astrocyte elevated gene-1. Technol Cancer Res Treat.
19:15330338209202532020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Song X, Cao G, Jing L, Lin S, Wang X,
Zhang J, Wang M, Liu W and Lv C: Analysing the relationship between
lncRNA and protein-coding gene and the role of lncRNA as ceRNA in
pulmonary fibrosis. J Cell Mol Med. 18:991–1003. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
27
|
Shi SH, Jiang J, Zhang W, Sun L, Li XJ, Li
C, Ge QD and Zhuang ZG: A novel lncRNA HOXC-AS3 acts as a
miR-3922-5p sponge to promote breast cancer metastasis. Cancer
Invest. 38:1–12. 2020. View Article : Google Scholar
|
|
28
|
Tian Y, Xia S, Ma M and Zuo Y: LINC00096
promotes the proliferation and invasion by sponging miR-383-5p and
regulating RBM3 expression in triple-negative breast cancer. Onco
Targets Ther. 12:10569–10578. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang GY, Zhang AQ, Wang J, Li CH, Wang XQ,
Pan K, Zhou C and Dong JH: Hepatoma-derived growth factor promotes
growth and metastasis of hepatocellular carcinoma cells. Cell
Biochem Funct. 34:274–285. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Enomoto H, Nakamura H, Liu W, Iwata Y,
Nishikawa H, Takata R, Yoh K, Hasegawa K, Ishii A, Takashima T, et
al: Down-regulation of HDGF inhibits the growth of hepatocellular
carcinoma cells in vitro and in vivo. Anticancer Res. 35:6475–6479.
2015.PubMed/NCBI
|
|
31
|
Enomoto H, Nakamura H, Liu W and
Nishiguchi S: Hepatoma-derived growth factor: Its possible
involvement in the progression of hepatocellular carcinoma. Int J
Mol Sci. 16:14086–14097. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Agostini M, Ganini C, Candi E and Melino
G: The role of noncoding RNAs in epithelial cancer. Cell Death
Discov. 6:132020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tsagakis I, Douka K, Birds I and Aspden
JL: Long non-coding RNAs in development and disease: Conservation
to mechanisms. J Pathol. 250:480–495. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wu Z, Wei ZH and Chen SH: LncUBE2R2-AS1
acts as a microRNA sponge of miR-302b to promote HCC progression
via activation EGFR-PI3K-AKT signaling pathway. Cell Cycle. Aug
23–2020.Epub ahead of print. View Article : Google Scholar
|
|
35
|
Xie SC, Zhang JQ, Jiang XL, Hua YY, Xie
SW, Qin YA and Yang YJ: LncRNA CRNDE facilitates epigenetic
suppression of CELF2 and LATS2 to promote proliferation, migration
and chemoresistance in hepatocellular carcinoma. Cell Death Dis.
11:6762020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li Y, Guo D, Lu G, Mohiuddin Chowdhury
ATM, Zhang D, Ren M, Chen Y, Wang R and He S: LncRNA SNAI3-AS1
promotes PEG10-mediated proliferation and metastasis via decoying
of miR-27a-3p and miR-34a-5p in hepatocellular carcinoma. Cell
Death Dis. 11:6852020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Shang R, Wang M, Dai B, Du J, Wang J, Liu
Z, Qu S, Yang X, Liu J, Xia C, et al: Long noncoding RNA SLC2A1-AS1
regulates aerobic glycolysis and progression in hepatocellular
carcinoma via inhibiting the STAT3/FOXM1/GLUT1 pathway. Mol Oncol.
14:1381–1396. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chi Y, Gong Z, Xin H, Wang Z and Liu Z:
Long noncoding RNA lncARSR promotes nonalcoholic fatty liver
disease and hepatocellular carcinoma by promoting YAP1 and
activating the IRS2/AKT pathway. J Transl Med. 18:1262020.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mao LH, Chen SY, Li XQ, Xu F, Lei J, Wang
QL, Luo LY, Cao HY, Ge X, Ran T, et al: LncRNA-LALR1 upregulates
small nucleolar RNA SNORD72 to promote growth and invasion of
hepatocellular carcinoma. Aging (Albany NY). 12:4527–4546. 2020.
View Article : Google Scholar
|
|
40
|
Jin J, Xu H, Li W, Xu X, Liu H and Wei F:
LINC00346 acts as a competing endogenous RNA regulating development
of hepatocellular carcinoma via modulating CDK1/CCNB1 axis. Front
Bioeng Biotechnol. 8:542020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chen S, Wang G, Tao K, Cai K, Wu K, Ye L,
Bai J, Yin Y, Wang J, Shuai X, et al: Long noncoding RNA
metastasis-associated lung adenocarcinoma transcript 1 cooperates
with enhancer of zeste homolog 2 to promote hepatocellular
carcinoma development by modulating the microRNA-22/Snail family
transcriptional repressor 1 axis. Cancer Sci. 111:1582–1595. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhao JT, Chi BJ, Sun Y, Chi NN, Zhang XM,
Sun JB, Chen Y and Xia Y: LINC00174 is an oncogenic lncRNA of
hepatocellular carcinoma and regulates miR-320/S100A10 axis. Cell
Biochem Funct. Mar 3–2020.Epub ahead of print.
|
|
43
|
Xiao JR, Wang K, Liu Y, Li ZW, Zhou YJ,
Wang HZ, Lu JY, Cheng SS and Wei S: Exploring of a prognostic long
non-coding RNA signature of hepatocellular carcinoma by using
public database. Zhonghua Liu Xing Bing Xue Za Zhi. 40:805–809.
2019.In Chinese. PubMed/NCBI
|
|
44
|
Ma Y, Zhang J, Wen L and Lin A:
Membrane-lipid associated lncRNA: A new regulator in cancer
signaling. Cancer Lett. 419:27–29. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Niu ZS, Wang WH, Dong XN and Tian LM: Role
of long noncoding RNA-mediated competing endogenous RNA regulatory
network in hepatocellular carcinoma. World J Gastroenterol.
26:4240–4260. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ye Y, Shen A and Liu A: Long non-coding
RNA H19 and cancer: A competing endogenous RNA. Bull Cancer.
106:1152–1159. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Niu YC, Tong J, Shi XF and Zhang T:
MicroRNA-654-3p enhances cisplatin sensitivity by targeting QPRT
and inhibiting the PI3K/AKT signaling pathway in ovarian cancer
cells. Exp Ther Med. 20:1467–1479. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Deng G, Mou T, He J, Chen D, Lv D, Liu H,
Yu J, Wang S and Li G: Circular RNA circRHOBTB3 acts as a sponge
for miR-654-3p inhibiting gastric cancer growth. J Exp Clin Cancer
Res. 39:12020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Geraldo MV, Nakaya HI and Kimura ET:
Down-regulation of 14q32-encoded miRNAs and tumor suppressor role
for miR-654-3p in papillary thyroid cancer. Oncotarget.
8:9597–9607. 2017. View Article : Google Scholar :
|
|
50
|
Zhou X, Li J, Teng J, Liu Y, Zhang D, Liu
L and Zhang W: Long noncoding RNA BSN-AS2 induced by E2F1 promotes
spinal osteosarcoma progression by targeting miR-654-3p/SYTL2 axis.
Cancer Cell Int. 20:1332020. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yang J, Chen S, Dou W, Xie R and Gao J:
miR-654-3p predicts the prognosis of hepatocellular carcinoma and
inhibits the proliferation, migration, and invasion of cancer
cells. Cancer Biomark. 28:73–79. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Huang JS, Chao CC, Su TL, Yeh SH, Chen DS,
Chen CT, Chen PJ and Jou YS: Diverse cellular transformation
capability of overexpressed genes in human hepatocellular
carcinoma. Biochem Biophys Res Commun. 315:950–958. 2004.
View Article : Google Scholar : PubMed/NCBI
|