Introduction
Osteoporosis (OP) is a common skeletal disorder
characterized by a low bone mass, the deterioration of bone tissue
and damaged bone structure (1).
OP can increase the risk of fractures and other severe bone-related
diseases, leading to increased mortality and healthcare costs
worldwide (2,3). Importantly, an increased number of
OP-induced fracture cases occur in females aged >55 years
(2). Accumulating evidence now
suggests that OP in postmenopausal females is a commonly
encountered skeletal disease, which is responsible for the
increased risk of disability in thousands of individuals (4). Various complex signaling pathways
are involved in the process of OP, and the exact molecular
mechanisms responsible for the pathogenesis of OP remain
unclear.
A previous study reported that osteoblasts and
osteoclasts mediate bone formation and resorption in the process of
bone remodeling, which is vital for the maintenance of bone
homeostasis (5). Accumulating
evidence has indicated that reactive oxygen species (ROS) play a
key role in regulating bone homeostasis. An earlier study reported
that the overproduction of ROS, induced by estrogen deficiency,
suppressed osteoblast differentiation in vitro (6). Moreover, increased oxidative stress
can cause bone loss by modulating the differentiation and survival
of osteoblasts in vivo (7). As a transcription factor, nuclear
factor erythroid 2-related factor 2 (Nrf2) can exert its
cytoprotective effects under conditions of oxidative stress
(8). It has been reported that
Nrf2 exerts suppressive effects on bone resorption and subsequently
promotes bone formation (9).
However, Nrf2 overexpression has been demonstrated to be negatively
associated with the differentiation of osteoblastic MC3T3-E1 cells
(10). In addition, Nrf2
hyperactivation has been reported to be responsible for the
impaired differentiation of osteoblasts (11). Hence, the biological role of Nrf2
in bone development should be further elucidated.
It was previously reported that the kelch-like
ECH-associated protein 1 (Keap1)/cullin-3 (CUL3)/E3
ubiquitin-protein ligase RBX1 (RBX1) complex is a negative
regulator of the Nrf2 protein (12). When any component of the complex
is disrupted, the function of the Keap1/CUL3/RBX1 complex can
become dysregulated, thus inducing Nrf2 hyperactivation. Of note,
the present study discovered the interaction between CUL3 and long
non-coding RNA (lncRNA) X inactive-specific transcript (XIST) on
the Starbase website (http://starbase.sysu.edu.cn/index.php). lncRNAs are a
type of endogenous non-coding RNA with a transcriptional length of
>200 nucleotides (13).
lncRNAs exert a regulatory effect on gene expression and are
involved in various physiological processes, including nuclear
transport, epigenetics, as well as transcriptional and
post-transcriptional regulation (14). Among these lncRNAs, lncRNA XIST
is highly expressed in the serum and monocytes of patients with OP,
suggesting that lncRNA XIST promotes OP development (15). Furthermore, XIST knockdown exerts
suppressive effects on the progression of tumors by reducing cell
proliferation, invasion and migration, and promoting the apoptosis
of human glioblastoma stem cells (16). Thus, in the present study, it was
hypothesized that XIST was associated with the pathogenesis of OP
via the regulation of Nrf2 expression by targeting CUL3.
In the present study, rats underwent ovariectomy
(OVX) to construct an animal model of OP, in order to investigate
whether XIST inhibits the differentiation of osteoblasts and
promotes OP, and elucidate the role of Nrf2 in this process, in
order to determine whether XIST may serve as an effective
therapeutic candidate for OP.
Materials and methods
Animals and rat model of OP
A total of 78 female Wistar rats, aged 8 weeks and
weighing 240-250 g, were obtained from Oriental Bioservice, Inc.
These rats were acclimated for 1 week prior to the start of the
study. The animals were housed in a temperature-controlled room
(22±2°C) with a 12-h light/dark cycle and 55±10% relative humidity.
The animals were fed a standard chow diet and had access to water
ad libitum. Following anesthesia with isoflurane (induction
dose, 4%; maintenance dose, 2.5%), the rats underwent bilateral
OVX, as previously described (17,18). In breif, an incision was made in
the middle of the abdomen into the abdominal cavity under aseptic
conditions following anesthesia, the bilateral ovaries were
removed, and the same amount of greater omentum was removed in the
sham-operated group. The animal surgical procedures were performed
in accordance with the Guidelines for Animal Care and were approved
by the Ethics Committee at the Affiliated Hospital of Jining
Medical University (Jining, China, No. IACUC-2000509-5). All
operations were carried out under sterile conditions in the
specific pathogen-free animal center. Firstly, the animals were
randomly assigned into the following six groups to examine the
efficiency of transfection (n=3): i) The control group [injection
of the same volume of the vehicle control (PBS) into knee joints]
(19,20); ii) shRNA-NC group (injection of
shRNA-NC into knee joints); iii) shRNA-XIST-1 group (injection of
shRNA-XIST-1 into knee joints); iv) shRNA-XIST-2 group (injection
of shRNA-XIST-2 into knee joints); v) OverExp-NC group (injection
of overexpression-NC plasmids into knee joints); and vi)
OverExp-XIST group (injection of overexpression-XIST plasmids into
knee joints). Subsequently, the animals (different rats) were
randomly assigned into the following five groups using simple
randomization (n=12): i) The control group [sham operation +
injection of the same volume of vehicle control (PBS) into knee
joints]; ii) overexpression (OverExp)-XIST group control (sham
operation + injection of overexpression-XIST plasmids into knee
joints); iii) model group (OVX + injection of the same volume of
control vehicle (PBS) into knee joints); iv) model + short hairpin
(sh)RNA-negative control (NC) group (OVX + injection of shRNA-NC
into knee joints); and v) model + shRNA-XIST group (OVX + injection
of shRNA-XIST into knee joints). The intra-articular injection of
50 µl lentivirus or PBS was performed every 2 days for 10
weeks post-surgery, and the animals administered buprenorphine
(0.05 mg/kg, s.c.) daily and were sacrificed in two batches at 10
and 12 weeks post-surgery. All rats were rapidly euthanized by
cervical dislocation during deep anesthesia, induced by an
intraperitoneal (i.p.) injection of 3.6% chloral hydrate solution
at a concentration of 360 mg/kg at the end of the experiment. The
femurs were harvested for histological analysis and RNA
extraction.
Cell culture and treatment
The murine osteoblastic cell line MC3T3-E1
(ATCC® CRL-2594) was obtained from the American Type
Culture Collection. Cells were maintained in α-minimal essential
medium (α-MEM) containing 10% fetal bovine serum (Gibco; Thermo
Fisher Scientific, Inc.), 100 U/ml penicillin and 100 µg/ml
streptomycin (Gibco; Thermo Fisher Scientific, Inc.) at 37°C in a
humid environment with 5% CO2, and the medium was
replaced every 2 days. When cell confluency reached 80–90%,
MC3T3-E1 cells were cultured in differentiation induction medium,
which was complete medium including L-ascorbic acid (50
µg/ml) and β-glycerophosphate (5 mM). Finally, the MC3T3-E1
cells were exposed to 0.3 mM hydrogen peroxide
(H2O2) for 24 h to induce dysfunction and
oxidative stress in vitro.
Transfection
The interaction between CUL3 and lncRNA X
inactive-specific transcript (XIST) was predicted with the Starbase
website. After the cell confluency reached 80%, MC3T3-E1 cells were
serum-starved for 3 h, and then transfected with shRNA-XIST (50 nM,
shRNA-XIST-1, 5′-GCTGACTACCTGAGATTTAAG-3′; shRNA-XIST-2,
5′-GCTCTTGAACAGTTAATTTGC-3′), OverExp-XIST (100 nM), shRNA-CUL3 (50
nM, shRNA-CUL3-1, 5′-GCGAGAAGATGTACTAAATTC-3′; shRNA-CUL3-2,
5′-GCTTGGAATGATCATCAAACA-3′) or the shRNA-NC,
(5′-TTCTCCGAACGTGTCACGT-3′) using Lipofectamine® 2000
reagent (Invitrogen; Thermo Fisher Scientific, Inc.), respectively.
XIST-overexpression plasmids and control overexpression plasmids
were cloned into a pCDNA3.1 vector and provided by Shanghai
GenePharma Co., Ltd. Following 6 h of incubation at 37°C, the
serum-free medium was exchanged for medium with serum. Finally, the
MC3T3-E1 cells were cultured for a further 24 h at 37°C and then
used for subsequent experiments. The transfection efficiency was
examined by reverse transcription-quantitative PCR (RT-qPCR).
Hematoxylin and eosin (H&E)
staining
Distal femurs were collected and fixed in 10%
neutral formaldehyde for 72 h. Subsequently, the femur bones were
removed and decalcified in 10% ethylene diamine tetraacetate
solution (pH 7.4) for 3 weeks and embedded in paraffin, after which
time they were cut into 5-µm-thick sections, stained with
H&E, and the field of interest at the distal femur was
visualized under a fluorescence microscope (Olympus IX53; Olympus
Corporation) at ×200 magnification. The relative injury levels of
each group were measured by the cortical area and analyzed using
ImageJ software (version 1.48; National Institutes of Health).
Dual-energy X-ray absorptiometry (DXA)
analysis
DXA measurement with Hologic DXA equipment (Hologic
Discovery W 81507; Hologic, Inc.) was carried out to scan and
measure the bone mineral density (BMD) of each individual rat.
Results were expressed as the grams of mineral content per square
centimeter of bone area (g/cm2).
Alkaline phosphatase (ALP) staining
Distal femurs were fixed with 80% alcohol and cut
into 5-µm-thick sections using a cryostat. In addition,
MC3T3-E1 cells (1.2×104 cells/well) were seeded in
6-well plates. When the cells were at 80% confluency, they were
treated with H2O2, or transfected as
aforementioned in α-MEM for 10 days. The determination of ALP
activity was performed using ALP staining kits (Thermo Fisher
Scientific, Inc.) both in vitro and in vivo (Roche
Diagnostics GmbH) according to the manufacturer's protocols.
Subsequently, the OD value was measured at 540 nm using a
microplate reader (Infinite™ M2000; Tecan Group, Ltd.), and the
images were visualized and collected under an inverted fluorescence
microscope (Olympus Corporation) at ×200 magnification.
RT-qPCR
Total RNA was extracted using TRIzol®
reagent (Invitrogen; Thermo Fisher Scientific, Inc.), and the
Reverse Transcription System kit (Takara Bio, Inc.) was used to
generate cDNA. Subsequently, RT-qPCR was performed with an SYBR
Premix Ex Taq kit (Takara Bio, Inc.). Relative expression was
calculated using the 2−ΔΔCq method (21) and normalized to GAPDH expression.
The results are expressed as the relative ratio to the control. The
thermocycling conditions were as follows: Pre-denaturation at 95°C
for 5 min, followed by 40 cycles at 95°C for 15 sec, 60°C for 30
sec and 72°C for 30 sec. The primer sequences included were as
follows: XIST forward, 5′-TCAGCCCATCAGTCCAAGATC-3′ and reverse,
5′-CCTAGTTCAGGCCTGCTTTTCAT-3′; Keap-1 forward,
5′-CCAACTTCCTCAAGGAGCAG-3′ and reverse, 5′-CGGCGACAAATATCATCCTT-3′;
Nrf2 forward, 5′-GGCTACGTTTCAGTCACTTG-3′ and reverse,
5′-AACTCAGGAATGGATAATAG-3′; CUL3 forward, 5′-GATGAGTTCAGGCAACATC-3′
and reverse, 5′-ATGTCTTGGTGCTGGTGG-3′; ALP forward,
5′-GGACCATTCCCACGTCTTCAC-3′ and reverse,
5′-CCTTGTAGCCAGGCCCATTG-3′; bone morphogenetic protein 2 (BMP2)
forward, 5′-CCCAGCGTGAAAAGAGAGAC-3′ and reverse,
5′-GGAAGCAGCAACGCTAGAAG-3′; runt-related transcription factor 2
(Runx2) forward, 5′-TTACCCCTCCTACCTGAGCCAG-3′ and reverse,
5′-GGTGTGGTAGTGAGTGGTGG-3′; osterix (OSX) forward,
5′-GCCTACTTACCGTGACTTT-3′ and reverse, 5′-GCCCACTATTGCCAACTGC-3′;
GAPDH forward, 5′-AAGGTGAAGGTCGGAGTCAAC-3′ and reverse,
5′-GGGGTCATTGATGGCAACAATA-3′.
Western blot analysis
Bone tissues and MC3T3-E1 cells were processed for
protein extraction using RIPA lysis buffer (Takara Bio, Inc.), and
then quantified using a BCA assay kit (Bio-Rad Laboratories, Inc.).
In brief, a total of 25 µg protein samples were separated
via 10% SDS-PAGE, and separated proteins were then transferred onto
PVDF membranes. After blocking with 5% skimmed milk at room
temperature for 2 h, the membranes were incubated with primary
antibodies against Keap-1 (1:1,000, ab227828, Abcam), CUL3
(1:1,000, ab75851, Abcam), Nrf2 (1:1,000, ab76026, Abcam), and
GAPDH (1:2,000, ab8245, Abcam) overnight at 4°C, and the membranes
were then washed with PBS and incubated with the appropriate
HRP-conjugated secondary antibody (1:5,000; cat. no. 7074; Cell
Signaling Technology, Inc.) at room temperature for 2 h. Finally,
ECL solution was applied to bands to measure the protein expression
levels with Tanon-5200 Chemiluminescence Imager (Tanon Science and
Technology Co., Ltd.). The relative intensities of target proteins
were analyzed using ImageJ software (version 1.48; National
Institutes of Health) and normalized to GAPDH expression.
Cell Counting kit-8 (CCK-8) assay
CCK-8 assay was performed to determine cell
viability. MC3T3-E1 cells were collected at 36 h following culture
with CCK-8 solution (Dojindo Molecular Technologies, Inc.). The
absorbance of each sample at 450 nm was then measured using a
microplate reader (Bio-Rad Laboratories, Inc.).
Flow cytometry
MC3T3-E1 cells were seeded in 6-well plates
(1×105 cells/well) and subjected to specific
transfection under H2O2 conditions. An
Annexin V-FITC/PI apoptosis detection kit (556547, BD Biosciences)
was used to evaluate the cell apoptotic rate. Cells were
resuspended with 1X Binding Buffer (500 µl), and incubated
with 5 µl Annexin V-FITC (5 µl) and PI staining
solution (10 µl) for 15 min at room temperature in the dark.
Following incubation in the dark for 30 min at room temperature,
apoptosis was evaluated using a flow cytometer (BD Biosciences)
with BD CellQuest Pro software version 5.2.1 (BD Biosciences).
Alizarin Red staining
Following incubation for 24 h at 37°C, MC3T3-E1
cells were fixed with 4% paraformaldehyde for 15 min at room
temperature and rinsed with ddH2O. Subsequently, 1%
Alizarin Red solution was prepared and the pH was adjusted to 4.2.
Subsequently, the cells were stained with Alizarin Red staining
solution for 15 min at room temperature and captured under an
inverted fluorescence microscope (Olympus IX53; Olympus
Corporation).
Dual-luciferase reporter assay
The combination of XIST and CUL3 was evaluated by
performing a dual-luciferase reporter assay. Briefly, MC3T3-E1
cells were seeded in 6-well plates and differentiation was induced
for 24 h after the cell confluency reached 80%. The cells were then
co-transfected with pmirGLO vector (Promega Corporation) recombined
with wild-type (WT, with the binding sites in XIST)/mutant-type
(MUT, with the mutant binding sites in XIST) and WT/MUT 3′UTR of
CUL3 (CUL3-WT and CUL3-MUT) using Lipofectamine® 2000.
Following 24 h of transfection, cells were collected and the
Firefly luciferase activity was quantified using the
Dual-Luciferase Reporter Assay System (Promega Corporation). The
data were normalized to Renilla luciferase activity.
Statistical analysis
All data were obtained from three independent
experiments and are expressed as the mean ± SEM. Differences
between groups were statistically analyzed with an unpaired
Student's t-test or one-way ANOVA followed by Tukey's post hoc
test. All statistical analyses were performed with the GraphPad
Prism 5 software package (GraphPad Software, Inc.). P<0.05 was
considered to indicate a statistically significant difference.
Results
XIST promotes OP and inhibits bone
formation in rats with OVX-induced OP
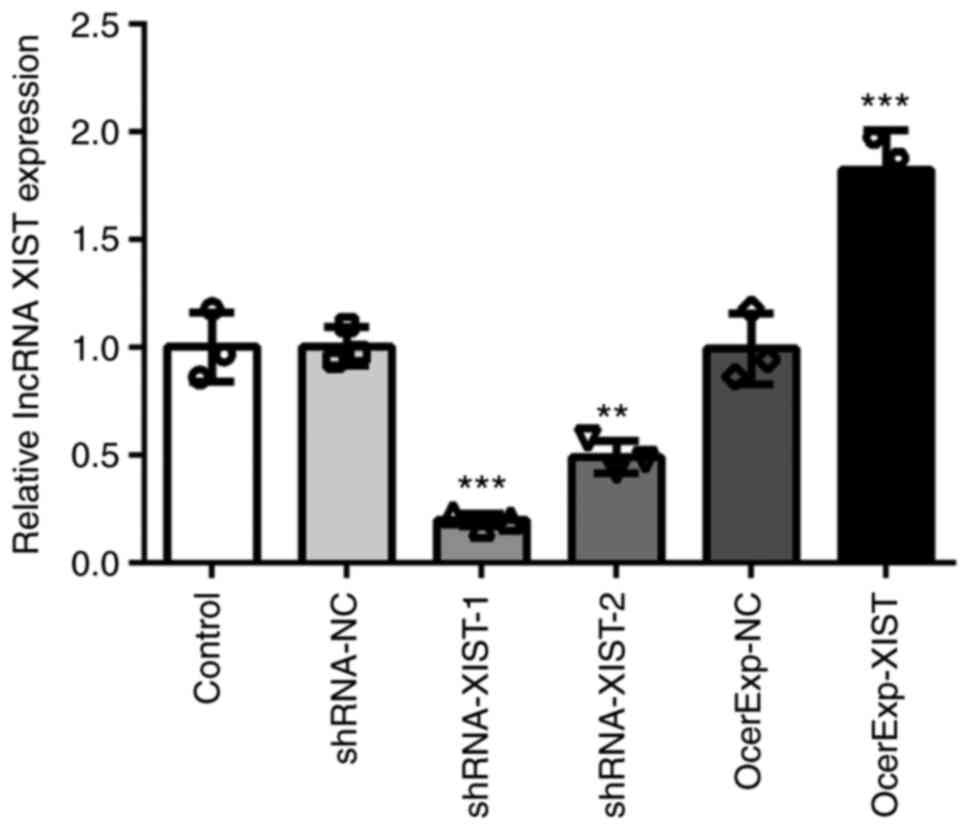
First, the shRNA-XIST and OverExp-XIST plasmids were
constructed to regulate the expression of XIST. As shown in
Fig. 1, shRNA-XIST-1 exerted a
more significant knockdown effect on XIST expression than
shRNA-XIST-2. Thus, shRNA-XIST-1 was selected for use in further
experiments. In addition, OverExp-XIST plasmids notably enhanced
the expression of XIST. The results of RT-qPCR suggested that these
plasmids could be used for subsequent experiments due to their high
transfection efficiencies. Second, OverExp-XIST and shRNA-XIST
plasmids were injected into the knee joints of rats to examine the
effects of XIST on OP pathogenesis. As demonstrated by the results
of DXA, the BMD of the rats from the OVX model group was decreased
compared with that in the control group. Following treatment with
OverExp-XIST and shRNA-XIST for 12 weeks, the BMD of the rats in
the OverExp-XIST group was decreased compared with that in the
control group, whereas the BMD of the rats in the model +
shRNA-XIST group was increased compared with that in the model
group (Fig. 2A), suggesting that
XIST accelerated the progression of OP and thus aggravated the
severity of OP.
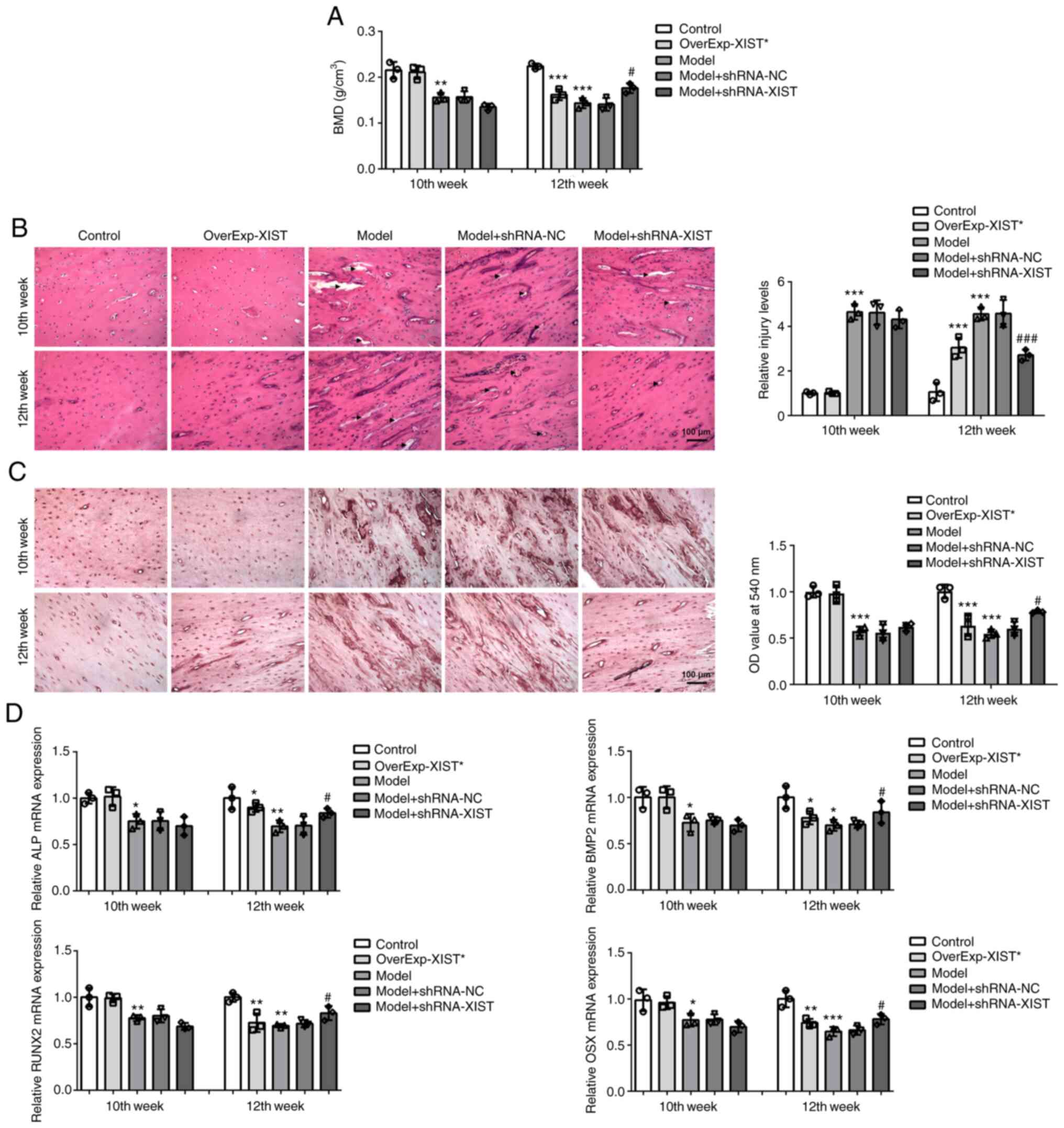 | Figure 2X inactive-specific transcript
promotes OP and suppresses bone formation in the
ovariectomy-induced OP rats. (A) Bone mineral density of rats from
each group was measured via DXA with Hologic DXA equipment. (B)
Bone histopathological changes in groups (Control, OverExp-XIST,
Model, Model+shRNA-NC, Model+shRNA-XIST) were analyzed by
hematoxylin and eosin staining. Scale bar, 100 µm. Arrows
indicate the destructive area of the cortex. (C) ALP activity was
detected by ALP staining. Scale bar, 100 µm. (D) The
relative mRNA expression levels of ALP, bone morphogenetic protein
2, runt-related transcription factor 2 and osterix in femurs were
detected via reverse transcription-quantitative PCR. Results are
presented as the mean ± SEM from three independent experiments.
*P<0.05, **P<0.01,
***P<0.001 vs. control; #P<0.05,
###P<0.01 vs. model group. OP, osteoporosis; DXA,
dual-energy X-ray absorptiometry; ALP, alkaline phosphatase. |
To further examine the effects of XIST on the
pathogenesis of OP, histopathological changes in bone and ALP
activity were determined by H&E and ALP staining, respectively.
The results of H&E staining revealed that the OVX procedure led
to a significant decrease in the cortical area in the rats in the
model group. The cortical area of the OverExp-XIST rats was notably
decreased compared with that of the control rats at the 12th week,
and thus the relative injury levels were increased. However, the
cortical area of the rats in the model + shRNA-XIST group was
significantly increased compared with that of the rats in the model
group, with a decrease in the relative injury levels (Fig. 2B). Moreover, ALP is a known
biomarker of bone formation; therefore, ALP staining was performed
to confirm the role of XIST in the progression of OP. It was
observed that ALP activity was inhibited by OVX and XIST
overexpression, and the OVX-inhibited ALP activity was increased by
XIST downregulation in vivo at the 12th week (Fig. 2C). Moreover, RT-qPCR revealed
that the mRNA expression levels of osteoblastic differentiation
makers (ALP, BMP2, RUNX2 and OSX) were suppressed by OVX and XIST
overexpression, whereas these expression levels were elevated by
XIST silencing (Fig. 2D). These
results indicated that XIST aggravated OP and hampered bone
formation in rats with OVX-induced OP.
XIST regulates the expression levels of
CUL3 and Nrf2 in rats with OVX-induced OP
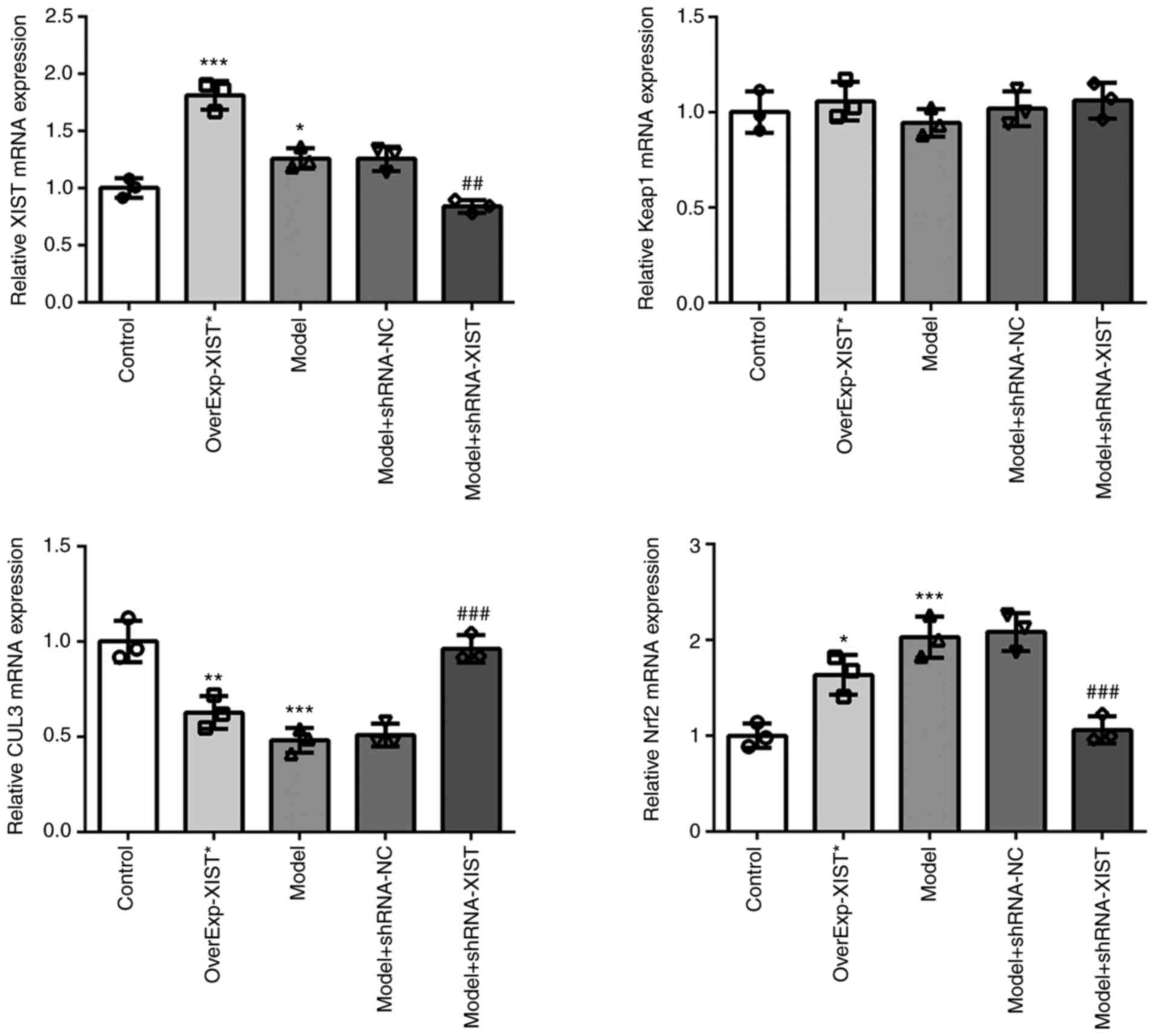
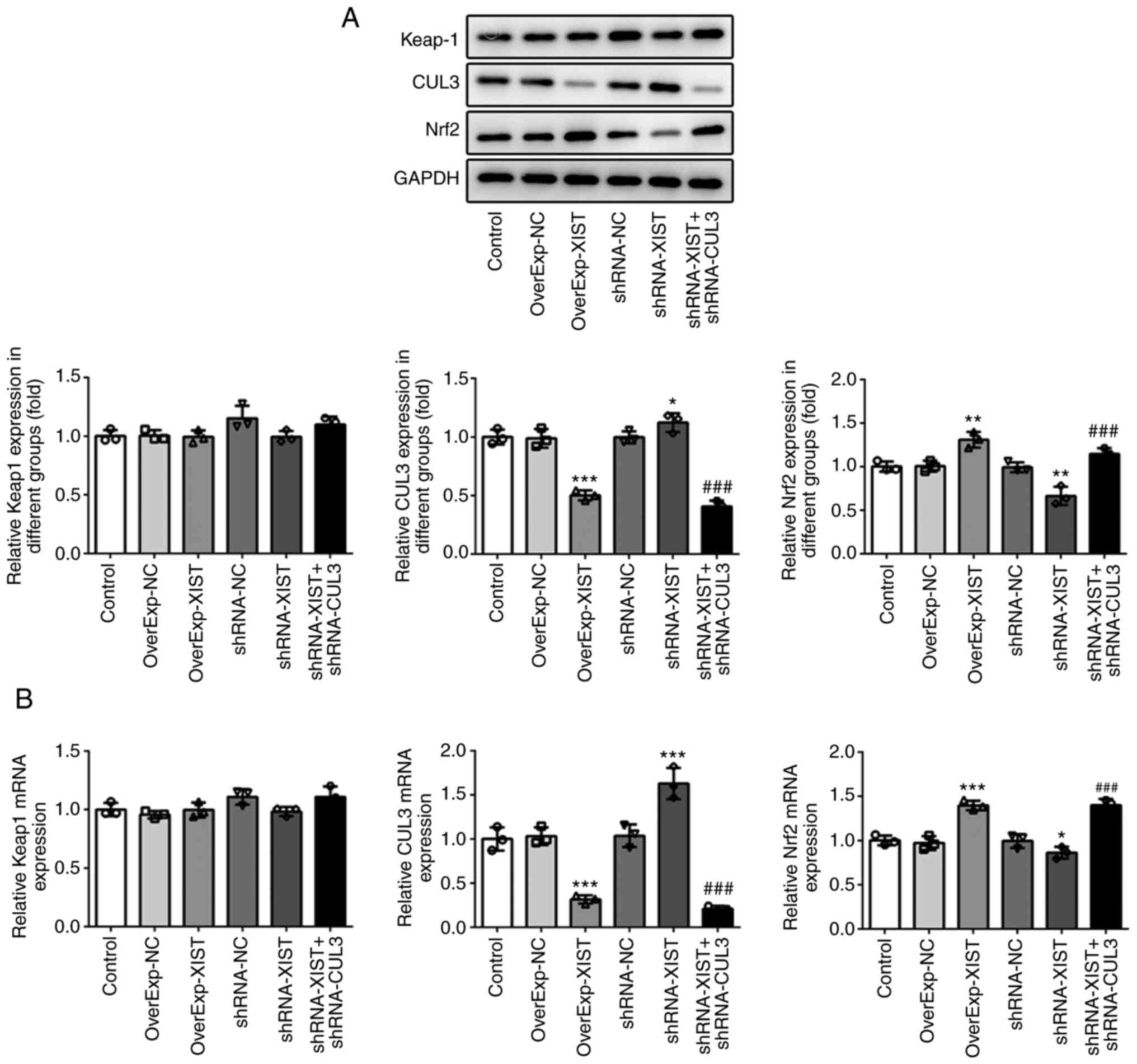
To examine the effects of XIST on Keap-1, CUL3 and
Nrf2 expression, western blot analysis and RT-qPCR were carried out
to assess the protein and mRNA expression levels of Keap-1, CUL3
and Nrf2 in rats with OP, respectively. As shown in Fig. 3, the results of RT-qPCR revealed
that OverExp-XIST transfection and OVX induced XIST overexpression,
and shRNA-XIST led to XIST downregulation. Moreover, XIST
overexpression led to a reduction in the expression of CUL3, and
induced an upregulation in the expression of Nrf2. By contrast,
XIST knockdown induced an upregulation in the expression of CUL3,
and led to the downregulation of Nrf2 expression. Additionally,
XIST did not exert any marked regulatory effects on the expression
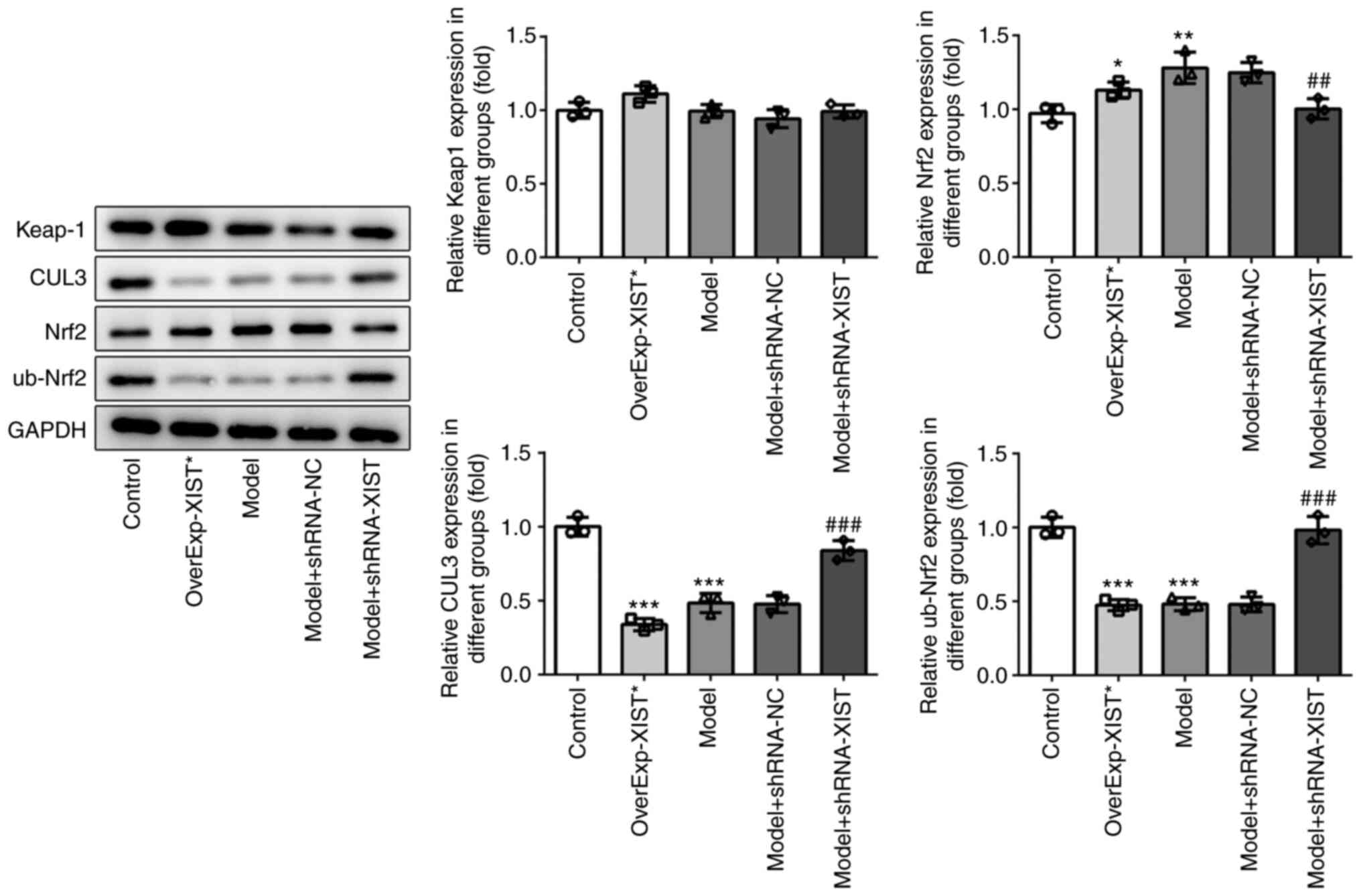
of Keap-1. As shown in Fig. 4,
the results of western blot analysis demonstrated that XIST exerted
similar effects on the protein expression of these genes. Of note,
the ubiquitination of Nrf2 was inhibited by XIST overexpression,
but promoted by XIST knockdown. These data suggested that XIST
played a regulatory role in the expression of CUL3 and Nrf2.
XIST downregulation promotes the
survival, differentiation and mineralization of
H2O2-treated MC3T3-E1 cells
To confirm the role of XIST in OP pathogenesis, a
series of functional assays were performed in vitro to
determine the effects of XIST on the cellular behaviors of MC3T3-E1
cells. Firstly, shRNA-XIST and OverExp-XIST were transfected into
the cells to achieve XIST silencing and overexpression,
respectively. As the expression of XIST in the shRNA-XIST-1 group
was lower than that in shRNA-XIST-2 group, shRNA-XIST-1 was
selected for use in subsequent experiments, as demonstrated by the
RT-qPCR results (Fig. 5A).
MC3T3-E1 cells were exposed to 0.3 mM H2O2 to
induce dysfunction and oxidative stress, which was defined as an
in vitro OP cell model. The results of CCK-8 assay revealed
that H2O2 treatment significantly reduced the
survival rate of the MC3T3-E1 cells, which was blocked by XIST
silencing (Fig. 5B).
Furthermore, the results of flow cytometry demonstrated that
H2O2 treatment promoted cell apoptosis,
whereas XIST knockdown attenuated the apoptotic rate of the
MC3T3-E1 cells in the model group (Fig. 5C and D). The aforementioned data
indicated that XIST knockdown improved the survival rate of
MC3T3-E1 cells which was decreased by H2O2
treatment.
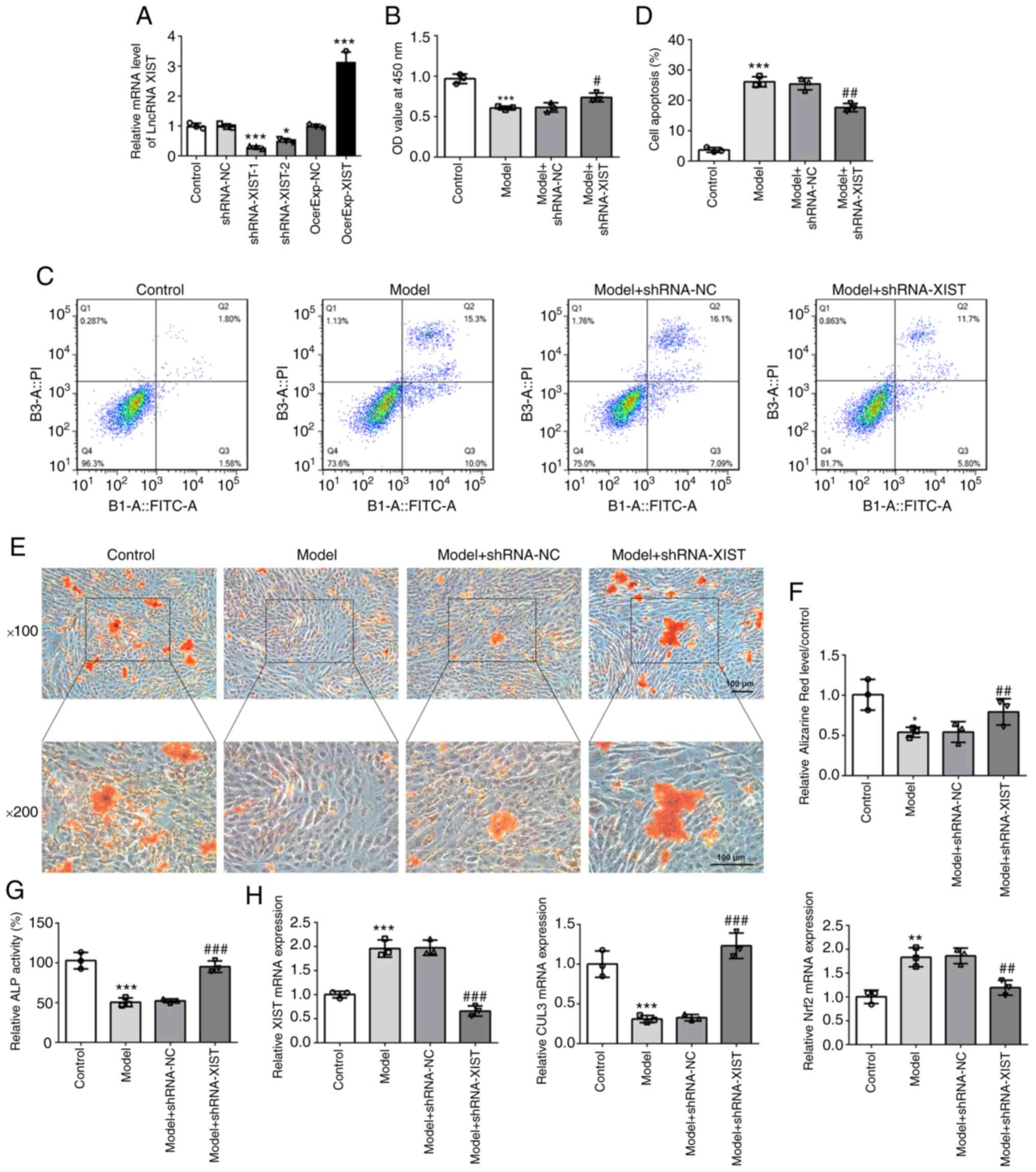 | Figure 5XIST knockdown improves the survival,
differentiation and mineralization of cells, and alters mRNA levels
of CUL3 and Nrf2 in H2O2-treated MC3T3-E1
cells. (A) The relative mRNA expression level of XIST was detected
via reverse transcription-quantitative PCR. (B) The survival rate
of cells treated with or without H2O2 was
evaluated using a Cell Counting Kit-8 assay. (C and D) The cell
apoptotic rate was determined via flow cytometry. (E and F) The
mineralization ability of MC3T3-E1 cells was determined by Alizarin
Red staining. Scale bar, 100 µm. (G) ALP activity was
detected using an ALP assay kit. (H) The relative mRNA expression
levels of XIST, CUL3 and Nrf2 were detected via reverse
transcription-quantitative PCR. Results are presented as the mean ±
SEM from three independent experiments. *P<0.05,
**P<0.01, ***P<0.001 vs. control;
#P<0.05, ##P<0.01,
###P<0.001 vs. model group. XIST, X inactive-specific
transcript; CUL3, cullin-3; Nrf2, nuclear factor erythroid
2-related factor 2; ALP, alkaline phosphatase. |
Moreover, Alizarin Red staining and ALP assays
determined the differentiation and mineralization abilities of
MC3T3-E1 cells, respectively. The Alizarin Red staining results
revealed that the number of calcium nodules was decreased following
H2O2 treatment. However, XIST silencing
significantly increased the number of calcium nodules that was
reduced by H2O2 treatment (Fig. 5E and F), suggesting that the
knockdown of XIST expression enhanced the mineralization ability of
MC3T3-E1 cells under oxidative stress conditions. Additionally, ALP
assay indicated that MC3T3-E1 cells in the model group had a lower
differentiation ability compared with the control group, and that
XIST downregulation notably enhanced the differentiation ability of
the cells compared with the model group (Fig. 5G). Finally, the results of
RT-qPCR demonstrated that H2O2 treatment
induced the upregulation of XIST and Nrf2 expression levels, and
the downregulation of CLU3 expression, which were reversed by the
silencing of XIST (Fig. 5H).
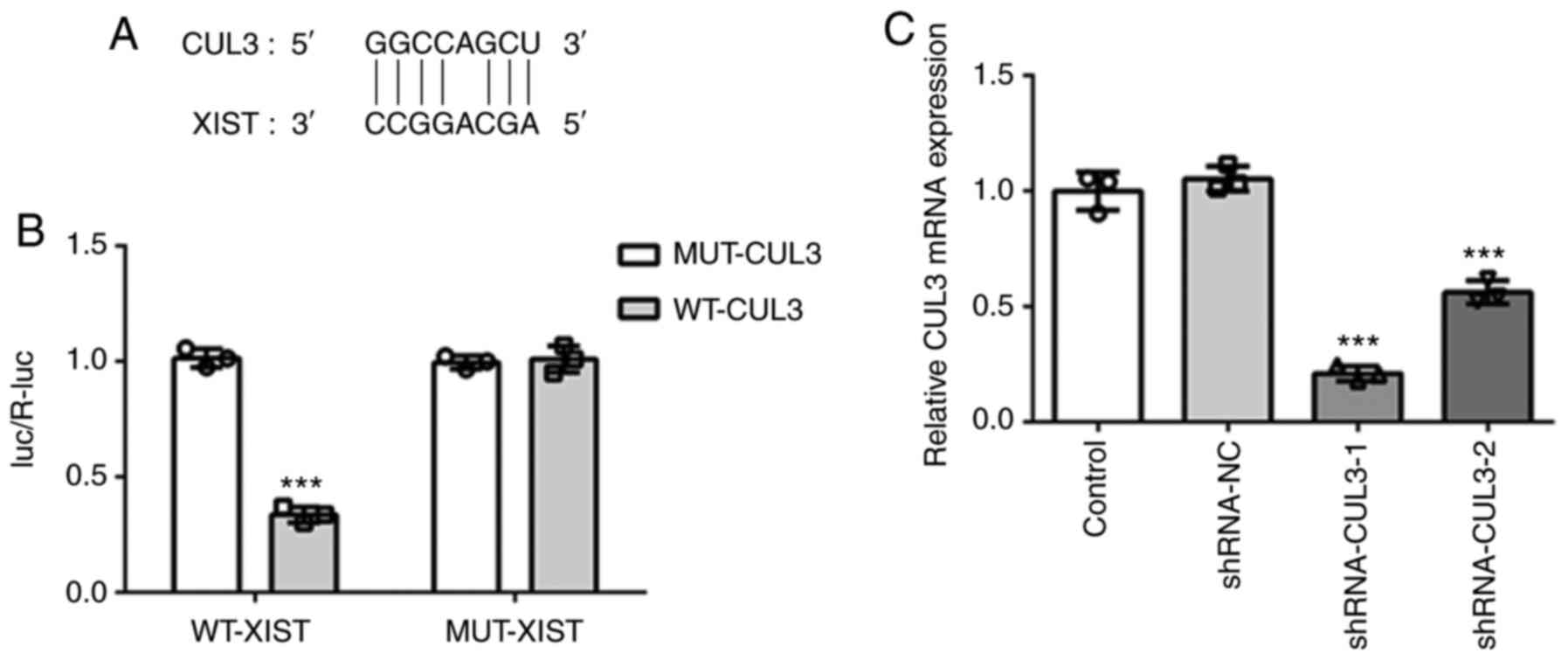
Interaction between XIST and CUL3
Subsequently, the underlying mechanisms of XIST in
OP pathogenesis were investigated. Herein, the interaction between
XIST and CUL3 was confirmed using a dual-luciferase reporter assay.
As shown in Fig. 6A and B, the
luciferase activity was notably decreased in the MC3T3-E1 cells
when the pmirGLO-CUL3-3′UTR WT plasmid was co-transfected with
WT-XIST. No differences were found in other groups. These findings
indicated that XIST directly targeted CUL3 and negatively modulated
its expression.
Downregulation of CUL3 reverses the
effects of XIST silencing on cell viability, differentiation and
mineralization, as well as the expression levels of Nrf2 and CUL3
in MC3T3-E1 cells
To knockdown the expression of CUL3, shRNA-CUL3
plasmids were constructed. As demonstrated by the results of
RT-qPCR, the decrease in the expression of CUL3 in the shRNA-CUL3-1
group was significantly higher than that in the shRNA-CUL3-2 group
(Fig. 6C). Therefore,
shRNA-CUL3-1 plasmids were employed in subsequent experiments.
To confirm the CUL3-mediated role of XIST in OP
progression, the cellular behaviors were observed when the MC3T3-E1
cells were separately transfected with shRNA-NC, shRNA-XIST and
shRNA-XIST + shRNA-CUL3. As shown by the results of CCK-8 assay,
XIST overexpression inhibited cell viability, whereas XIST
knockdown increased cell viability. However, CUL3 knockdown
abolished the promoting effects of XIST knockdown on cell viability
(Fig. 7A). As presented in
Fig. 7B and C, XIST
overexpression suppressed the differentiation and mineralization of
MC3T3-E1 cells. In addition, the promoting effects of XIST
silencing on differentiation and mineralization were eliminated by
CUL3 knockdown. Finally, the results of western blot analysis and
RT-qPCR demonstrated that XIST overexpression significantly
downregulated the expression of CUL3 and markedly promoted the
expression of Nrf2. By contrast, XIST knockdown notably increased
CUL3 expression and markedly decreased Nrf2 expression, which were
reversed by CUL3 silencing (Fig. 8A
and B). On the whole, these results illustrated that CUL3
knockdown abolished the effects of XIST silencing on the behaviors
of MC3T3-E1 cells via Nrf2.
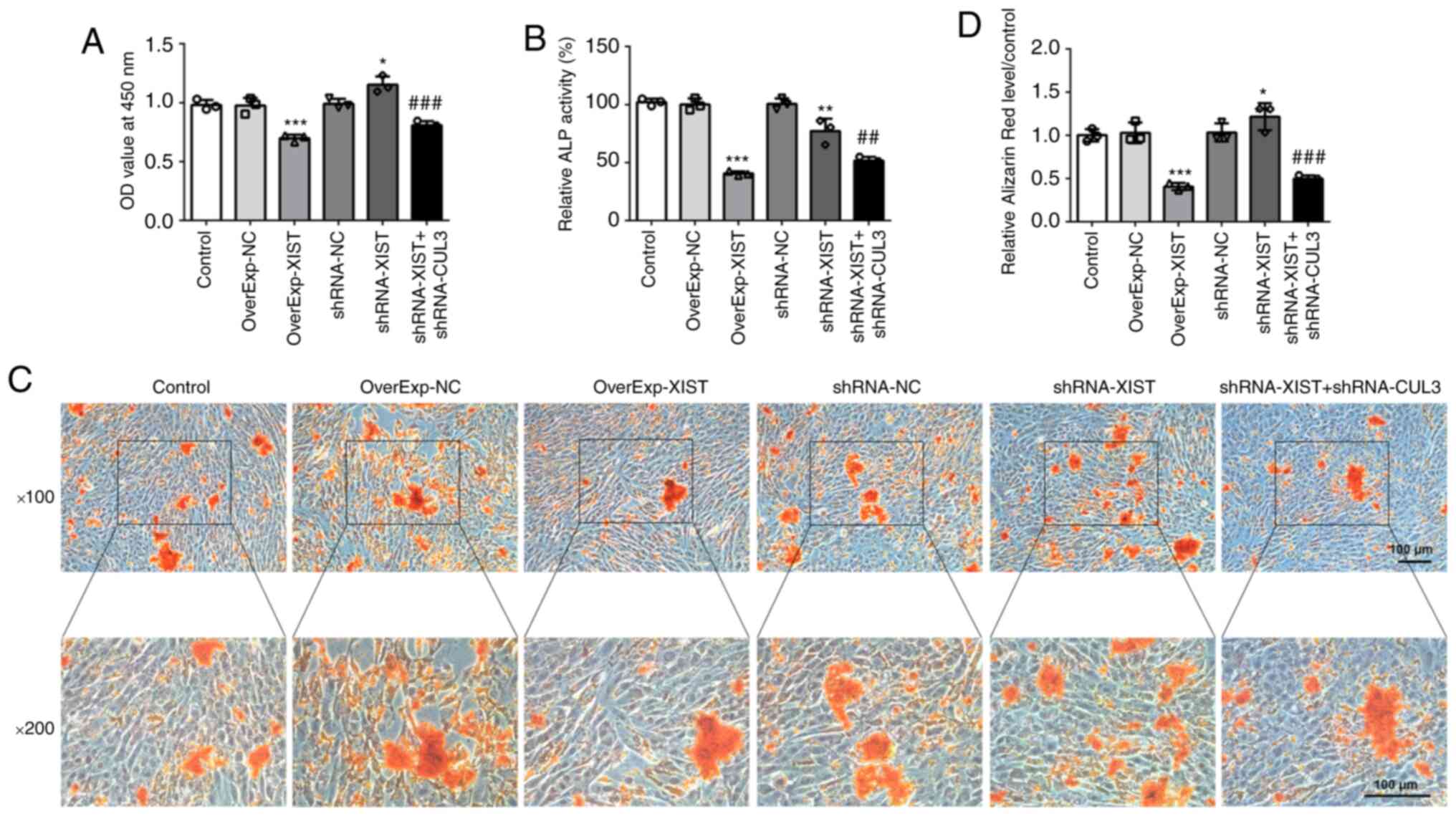 | Figure 7CUL3 downregulation reverses the XIST
knockdown-induced effects on cell viability, differentiation and
mineralization of MC3T3-E1 cells. (A) The survival rate of cells
transfected with OverExp-NC, OverExp-XIST, shRNA-NC, shRNA-XIST or
shRNA-XIST + shRNA-CUL3-1 was evaluated with a Cell Counting Kit-8
assay. (B) ALP activity was elevated, as determined using an ALP
assay kit. (C and D) The mineralization ability of MC3T3-E1 cells
was determined using Alizarin Red staining. Scale bar, 100
µm. Results are presented as the mean ± SEM from three
independent experiments. *P<0.05,
**P<0.01, ***P<0.001 vs. control;
##P<0.01, ###P<0.001 vs. shRNA-XIST.
XIST, X inactive-specific transcript; CUL3, cullin-3; ALP, alkaline
phosphatase; OverExp, overexpression; NC, negative control; shRNA,
short hairpin. |
Discussion
As a chronic skeletal disorder, OP is characterized
by damaged bone structure and low bone mass, and can reduce bone
strength and an increased susceptibility to fractures (22). It has been reported that estrogen
deficiency leads to bone loss via enhancing osteoclastic function
(23). Postmenopausal OP is a
highly prevalent disease, which contributes to a high morbidity and
mortality. Oral bisphosphonates are common therapeutic agents, and
the long-term use of these agents can reduce the risk of fractures
in patients with OP (24).
However, there are various limitations in current drug treatment
regimens, including a slow therapeutic effect and an increased risk
of adverse events when used over a prolonged period of time.
Consequently, the development of novel therapeutic strategies to
regulate bone formation in patients with OP is of utmost
importance.
A previous study reported that lncRNA XIST is
expressed at high levels in the serum and monocytes of patients
with OP (15). In the present
study, it was observed that XIST overexpression promoted OP by
decreasing BMD, the destruction of the cortex and inhibiting bone
formation in rats, whereas XIST knockdown inhibited OP progression
in rats with OVX-induced OP. Moreover, XIST knockdown enhanced cell
viability, as well as the differentiation and mineralization
abilities of H2O2-treated MC3T3-E1 cells.
These data indicated that lncRNA XIST enhanced OP development,
which was consistent with previous findings (15).
Most importantly, XIST was confirmed to directly
target CUL3 and negatively regulate its expression, as determined
by a dual-luciferase reporter assay. The Keap1/CUL3/RBX1 complex is
a negative regulator of Nrf2 protein expression (12). Under normal conditions, Nrf2
protein interacts with the Keap1/CUL3/RBX1 complex and is
maintained at low levels due to ubiquitination and proteasomal
degradation (25). Under high
levels of oxidative stress, the interaction is prevented due to
conformational changes of Keap1, resulting in the accumulation of
Nrf2 and translocation to the nucleus. Once any component of the
complex is absent or altered, Nrf2 is hyperactivated, which leads
to its accumulation and translocation into the nucleus (26). In the present study, XIST was
confirmed to inhibit CUL3 expression and enhance Nrf2 expression,
which was supported by both in vivo and in vitro
results. Moreover, XIST suppressed the ubiquitination of Nrf2 in
OVX-induced OP rats. Of note, CUL3 knockdown abolished the
inhibitory effect of XIST silencing on Nrf2 expression, which
indicated that the inhibition of XIST leads to the degradation of
Nrf2 by targeting CUL3.
A previous study confirmed that targeting the
Keap1/Nrf2 pathway could evidently relieve oxidative stress
(27). Furthermore, it is
commonly known that ROS play a crucial role in the activation of
numerous biological pathways, and are closely associated with cell
proliferation, apoptosis and differentiation (28). Estrogen deficiency initiates OP
via the overproduction of ROS and hence, antioxidants may be
effective therapeutic candidates (29). It has been reported that
increased oxidative stress negatively affects bone formation.
However, low levels of ROS suppress the mineralization ability of
osteoblasts by altering the differentiation of osteoblasts in early
stages (30). Hence, Nrf2
hyperactivation may result in low concentrations of ROS, and may
thus inhibit the differentiation of osteoblasts. In the present
study, the constitutive activation of Nrf2, as a result of CLU3
downregulation via XIST overexpression, suppressed cell viability,
differentiation and mineralization of MC3T3-E1 cells. Most
importantly, CUL3 downregulation eliminated the suppressive effects
of XIST silencing on the cellular behaviors of MC3T3-E1 cells.
Taken together, these findings indicated that XIST suppressed the
differentiation of osteoblasts and promoted OP via Nrf2
hyperactivation by targeting CUL3. However, the estrous cycle was
not analyzed following bilateral ovariectomy, and the atrophy of
uterine horns was not evaluated after the animals were sacrificed
in the present study, which will be taken into account in future
experiments. Additionally, in future studies, the authors aim to
determine the role of XIST in oxidative stress and inflammation, in
order to provide a more in-depth understanding of the OP
pathogenesis.
In conclusion, the present study demonstrated that
XIST facilitated OP and suppressed bone formation by regulating the
expression of CUL3 and Nrf2 in rats with OVX-induced OP. In
addition, XIST knockdown promoted the survival, differentiation and
mineralization of H2O2-treated MC3T3-E1
cells, which was reversed by CUL3 downregulation. Overall, XIST
inhibited the differentiation of osteoblasts and promoted OP by
inhibiting the ubiquitination and degradation of Nrf2 via targeting
CUL3.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XC and GC designed the experiments and drafted the
manuscript. XC, FM, NZ and FG performed the experiments and
analyzed the data. XC and GC reviewed the manuscript. All authors
read and approved the final manuscript. XC and GC confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
The animal surgical procedures were performed in
accordance with the Guidelines for Animal Care and were approved by
the Ethics Committee at the Affiliated Hospital of Jining Medical
University (Jining, China, no. IACUC-2000509-5).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
Funding
The present study was supported by the Research Fund for Lin
He's Academician Workstation of New Medicine and Clinical
Translation in Jining Medical University, China (grant no.
JYHL2018FMS12) and Development Project of Medical and Health
Technology in Shandong Province, China (grant no.
2018WSB34010).
References
|
1
|
Cosman F, de Beur SJ, LeBoff MS, Lewiecki
EM, Tanner B, Randall S and Lindsay R; National Osteoporosis
Foundation: Clinician's guide to prevention and treatment of
osteoporosis. Osteoporos Int. 25. pp. 2359–2381. 2014, View Article : Google Scholar
|
|
2
|
Compston JE, McClung MR and Leslie WD:
Osteoporosis. Lancet. 393:364–376. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wu W, Xiao Z, Chen Y, Deng Y, Zeng D, Liu
Y, Huang F, Wang J, Bellanti JA, Rong L and Zheng SG: CD39 produced
from Human GMSCs regulates the balance of osteoclasts and
osteoblasts through the Wnt/β-catenin pathway in osteoporosis. Mol
Ther. 28:1518–1532. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Eastell R, O'Neill TW, Hofbauer LC,
Langdahl B, Reid IR, Gold DT and Cummings SR: Postmenopausal
osteoporosis. Nat Rev Dis Primers. 2:160692016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Manolagas SC: Corticosteroids and
fractures: A close encounter of the third cell kind. J Bone Miner
Res. 15:1001–1005. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jun JH, Lee SH, Kwak HB, Lee ZH, Seo SB,
Woo KM, Ryoo HM, Kim GS and Baek JH: N-acetylcysteine stimulates
osteoblastic differentiation of mouse calvarial cells. J Cell
Biochem. 103:1246–1255. 2008. View Article : Google Scholar
|
|
7
|
Lean JM, Davies JT, Fuller K, Jagger CJ,
Kirstein B, Partington GA, Urry ZL and Chambers TJ: A crucial role
for thiol antioxidants in estrogen-deficiency bone loss. J Clin
Invest. 112:915–923. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Suzuki T, Motohashi H and Yamamoto M:
Toward clinical application of the Keap1-Nrf2 pathway. Trends
Pharmacol Sci. 34:340–346. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hyeon S, Lee H, Yang Y and Jeong W: Nrf2
deficiency induces oxidative stress and promotes RANKL-induced
osteoclast differentiation. Free Radic Biol Med. 65:789–799. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hinoi E, Fujimori S, Wang L, Hojo H, Uno K
and Yoneda Y: Nrf2 negatively regulates osteoblast differentiation
via interfering with Runx2-dependent transcriptional activation. J
Biol Chem. 281:18015–18024. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yoshida E, Suzuki T, Morita M, Taguchi K,
Tsuchida K, Motohashi H, Doita M and Yamamoto M: Hyperactivation of
Nrf2 leads to hypoplasia of bone in vivo. Genes Cells. 23:386–392.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ziros PG, Manolakou SD, Habeos IG, Lilis
I, Chartoumpekis DV, Koika V, Soares P, Kyriazopoulou VE, Scopa CD,
Papachristou DJ and Sykiotis GP: Nrf2 is commonly activated in
papillary thyroid carcinoma, and it controls antioxidant
transcriptional responses and viability of cancer cells. J Clin
Endocrinol Metab. 98:E1422–E1427. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fernandes JCR, Acuna SM, Aoki JI,
Floeter-Winter LM and Muxel SM: Long Non-Coding RNAs in the
regulation of gene expression: Physiology and disease. Noncoding
RNA. 5:172019.
|
|
14
|
Ponting CP, Oliver PL and Reik W:
Evolution and functions of long noncoding RNAs. Cell. 136:629–641.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen X, Yang L, Ge D, Wang W, Yin Z, Yan
J, Cao X, Jiang C, Zheng S and Liang B: Long non-coding RNA XIST
promotes osteoporosis through inhibiting bone marrow mesenchymal
stem cell differentiation. Exp Ther Med. 17:803–811.
2019.PubMed/NCBI
|
|
16
|
Yao Y, Ma J, Xue Y, Wang P, Li Z, Liu J,
Chen L, Xi Z, Teng H, Wang Z, et al: Knockdown of long non-coding
RNA XIST exerts tumor-suppressive functions in human glioblastoma
stem cells by up-regulating miR-152. Cancer Lett. 359:75–86. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jee WS and Yao W: Overview: Animal models
of osteopenia and osteoporosis. J Musculoskelet Neuronal Interact.
1:193–207. 2001.
|
|
18
|
Komori T: Animal models for osteoporosis.
Eur J Pharmacol. 759:287–294. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Su K, Bai Y, Wang J, Zhang H, Liu H and Ma
S: Comparison of hyaluronic acid and PRP intra-articular injection
with combined intra-articular and intraosseous PRP injections to
treat patients with knee osteoarthritis. Clin Rheumatol.
37:1341–1350. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Aytekin K, Erhan S, Erişgin Z, Esenyel CZ
and Takır S: Intra-articular injection of hydrogen sulfide
decreased the progression of gonarthrosis. Can J Physiol Pharmacol.
97:47–54. 2019. View Article : Google Scholar
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
22
|
Makitie RE, Costantini A, Kampe A, Alm JJ
and Makitie O: New insights into monogenic causes of osteoporosis.
Front Endocrinol (Lausanne). 10:702019. View Article : Google Scholar
|
|
23
|
Shen G, Ren H, Shang Q, Zhang Z, Zhao W,
Yu X, Tang J, Yang Z, Liang D and Jiang X: miR-128 plays a critical
role in murine osteoclastogenesis and estrogen deficiency-induced
bone loss. Theranostics. 10:4334–4348. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Guiducci L, Vassalle C, Parchi P and
Maffei S: Monthly intramuscular neridronate for the treatment of
postmenopausal osteoporosis: Results of a 6-year prospective
italian study. Int J Endocrinol. 2019:98028272019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Iso T, Suzuki T, Baird L and Yamamoto M:
Absolute Amounts and Status of the Nrf2-Keap1-Cul3 complex within
cells. Mol Cell Biol. 36:3100–3112. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Martinez VD, Vucic EA, Pikor LA, Thu KL,
Hubaux R and Lam WL: Frequent concerted genetic mechanisms disrupt
multiple components of the NRF2 inhibitor KEAP1/CUL3/RBX1
E3-ubiquitin ligase complex in thyroid cancer. Mol Cancer.
12:1242013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xue Y and Wang AZ: DJ-1 plays a
neuroprotective role in SH-SY5Y cells by modulating Nrf2 signaling
in response to lidocaine-mediated oxidative stress and apoptosis.
Kaohsiung J Med Sci. 36:630–639. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Migliario M, Pittarella P, Fanuli M, Rizzi
M and Reno F: Laser-induced osteoblast proliferation is mediated by
ROS production. Lasers Med Sci. 29:1463–1467. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhou X, Wang Z, Ni Y, Yu Y, Wang G and
Chen L: Suppression effect of N-acetylcysteine on bone loss in
ovariectomized mice. Am J Transl Res. 12:731–742. 2020.PubMed/NCBI
|
|
30
|
Nicolaije C, Koedam M and van Leeuwen JP:
Decreased oxygen tension lowers reactive oxygen species and
apoptosis and inhibits osteoblast matrix mineralization through
changes in early osteoblast differentiation. J Cell Physiol.
227:1309–1318. 2012. View Article : Google Scholar
|