Renewed interest has recently arisen for both
functional foods and the investigation of the scientific value of
traditional medical treatments. The evaluation of mushroom
derivatives and their medical properties are important part of
these studies. Polysaccharides, including the glucans, have been
described as biologically active molecules (1–4).
Certain glucose polymers, such as (1→3), (1→6)-β-glucans, were
recently proposed as potent immunomodulation agents (3–5).
Even though glucans can be extracted from the cell walls of yeast,
oat, barley, seaweeds, algae and bacteria, the foremost source of
medical glucans turns out to be fungal cell walls which consist
either of polysaccharides such as chitin, cellulose, (1→3)-,
(1→6)-β-glucans and (1→3)-α-glucans, or polysaccharide-protein
complexes (6). The β-glucans are
the most studied within these polysaccharides and are principally
obtained from the fruit body of various types of mushrooms. Used
especially in traditional Oriental medicine (7), they are reported to be found in fruit
bodies, cultured mycelium and cultured broth from higher
Basidiomycetes mushrooms (as resulted from ∼700 species of
investigated higher Hetero- and Homobasidiomycetes). According to
traditional medicine assertions, as well as some scientific
studies, glucans have been reported to produce anti-tumor,
immunomodulating, antioxidant, radical scavenging, cardiovascular,
antihypercholesterolemia, antiviral, antibacterial, antiparasitic,
antifungal, detoxification, hepatoprotective and antidiabetic
effects (8,9). Growing interest of Western science
into biologically active polysaccharides can be considered to start
after the publication of Pillemer and Ecker in 1941 (10). They described a crude yeast cell
wall preparation, later named zymosan, able to modulate
non-specific immunity (complement) (11). It was unknown at that time which
component of this preparation was stimulating the immune response.
Later on, β-glucan was identified by Riggi and Di Luzio as the
immune-activating compound within the components of zymosan (after
testing its lipid and mannan components) (7). Since then, a large number of studies
have been performed to clarify the immunomodulating potential of
glucans and their possible antitumor effects (12–18).
The discovery of specific receptors for glucans on immune cells,
the recent advances in understanding host immune responses against
infectious agents and cancer and the importance of the innate
immunity (inflammation) in these responses have renewed the
interest toward glucans as perspective immunotherapeutic
molecules.
The glucans are D-glucose-based polysaccharides.
With their glucose anomeric structure, they can be α-D-glucans,
β-D-glucans and mixed α,β-D-glucans. They present different types
of glycosidic bonds originating either (1→3)-, (1→6)-β-glucans
(e.g., zymosan, laminarin, lentinan, pleuran), or (1→3)-, (1→4)-,
(1→6)-α-glucans (e.g., dextran, glycogen, starch). Finally,
depending on their constitution, they are indicated as homoglucans
(only glucose molecules) or heteroglucans (not only glucose
molecules) (19,20).
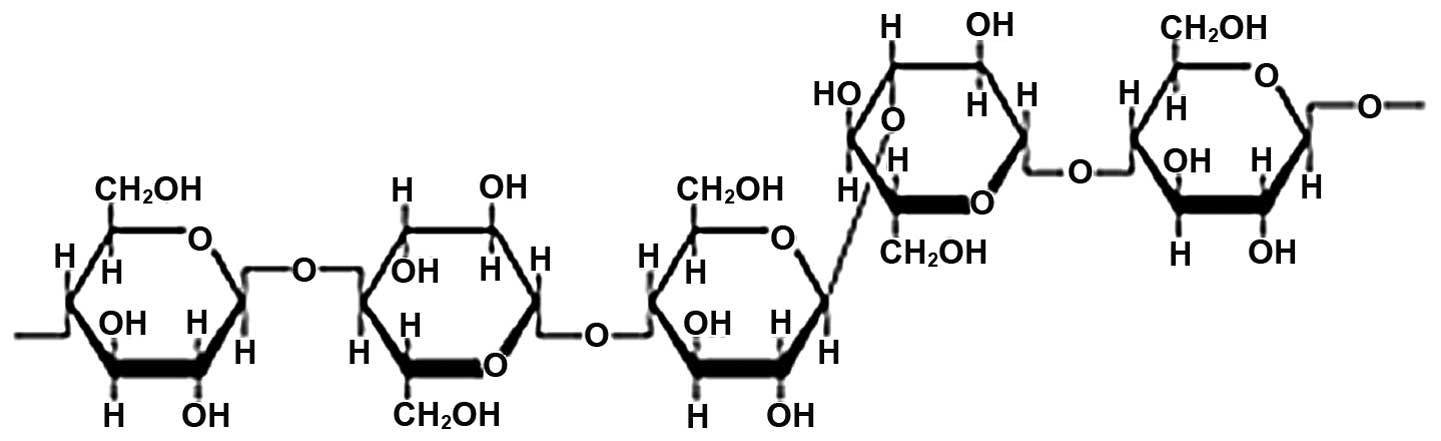
The β-glucans consist of linear unbranched
polysaccharides of β-D-glucose. The basic β-D-glucan is a repeating
structure with the β-D-glucose units joined together in linear
chains by β-bonds. These can extend either from carbon 1 of one
saccharide ring to carbon 3 of the next (β1→3) (Fig. 1), or from carbon 1 to carbon 4
(β1→4), or from carbon 1 to carbon 6 (β1→6) (1). The β-D-glucans can form large
cylindrical molecules containing up to 250,000 glucose units.
As reported above, the sources of glucans are
various, including fungi (e.g., mushrooms), yeast and seaweed, as
well as barley. Medical glucans (as the ones used by traditional
medicine) are principally obtained from edible fungi. By boiling
and treating with enzymes from one of the cited sources, glucans
can be extracted in crude form yielding soluble and insoluble
products (19,21,22).
There are many forms of soluble β-glucans evaluated for possible
antitumor activity, such as (1→3)-β-D-glucan, SSG obtained from
Sclerotina sclerotiorum IFO 9395 (23), SPG (also Schizophyllan, sizofiran,
sonifilan) from Schizophyllum commune(24) and GRN (also Grifolan) from
Grifola frondosa(25) and
they often exist as a linear triple-helical structure in an aqueous
solution (26). Insoluble glucans
have been isolated for the first time from the mushroom Lentinus
edodes(27). They were also
isolated from the cell wall of yeast by using the combination of
NaClO oxidation and dimethylsulfoxide (DMSO) extraction (28). To improve their solubility,
derivatization by phosphorylation, either sulfation or amination
can be used. However, insoluble β-glucans were found to possess
higher immunostimulating activity than soluble ones and are
administered orally. Factors that can greatly influence the
antitumor and immunodulatory activities of the glucans are their
structure, molecular weight, degree of branching and conformation
(17,29–31).
The molecular weight of glucans is dependent upon their source and
extraction method (32). For
example, the average molecular weight of Krestin (PSK), Lentinan,
Schizophyllan (SPG) and PGG-glucan are, respectively, reported as
100,000, 500,000, 450,000 and 170,000 Da (33–35).
As stated above, the immunostimulatory properties of
fungal β-glucans were studied and described almost 50 years ago
(36). Shortly afterwards, their
effects against tumor development in experimental animals were also
described (37) and finally
glucans were reported to modulate other conditions (e.g.,
cholesterol levels, glucose tolerance) (38,39).
Since these early studies, it has been demonstrated
that β-D-glucans increase the resistance of mammalians against
several bacterial, fungal, viral and protozoal pathogens (40–43).
A recent study compared the effects of soluble oat glucan versus
Pleurotan, an insoluble β-D-glucan isolated from the mushroom
Pleurotus ostreatus. They were administered as a food
supplement for athletes and the β-D-glucan isolated from the
mushroom resulted in significantly reducing the incidence of upper
respiratory tract infection. Interestingly, the Pleurotan
administration was associated with an increased number of
circulating natural killer cells as well as a preventive effect on
the reduction of natural killer cell activity. These latter
findings may explain the reduced infectivity risk in the treated
athletes (29). Since the soluble
oat glucan supplementation did not produce effects on the incidence
of respiratory tract infections, it was suggested that solubility
and structural factors (e.g., backbone structure and degree of
branching) can deeply affect the immunomodulatory capacity of
β-D-glucans (17). Many studies
have reported the ability of (1→3)-β-D-glucans to activate innate
immunity with effects also on adaptive immunity, inducing humoral
and cell-mediated immune responses. The (1→3)-β-D-glucans were
found to increase the antimicrobial activity of mononuclear cells
and neutrophils (7,44,45)
and enhance the functional activity of macrophages (46,47).
It has been reported that the (1–6)-branched type glucans, with high
molecular weight and (1→3)-β-D-glucans are especially effective in
inducing nitric oxide production by macrophages (21,47,48).
Moreover, ex vivo experiments with macrophages obtained from
animals treated with (1→3)-β-D-glucans showed enhanced esterase
release and cytostatic effect on tumor cells when challenged with
L-929 tumor cells (49).
(1→3)-β-D-glucans were also reported to have hematopoietic
activities, according to their conformation (single and triple
helix) and to stimulate the proliferation of monocytes and
macrophages (50–52). Relating to their role in triggering
innate immunity responses, insoluble and derivatized
(1→3)-β-D-glucans, according to their source, were also found to
stimulate the production of proinflammatory molecules such as
complement components, IL-1α/β, TNF-α, IL-2, IFN-γ and eicosanoids
as well as IL-10, and IL-4 (53–59).
The innate immunity cells are provided of a complex
network of germ line-encoded pattern-recognition receptors (PRRs).
They can identify pathogens by binding to carbohydrates, lipids and
proteins expressed by the microorganism, including fungi (68–71).
As reported above, in vivo administration of pure glucans
induces the activation a wide range of responses by innate immunity
(70,72). In particular, glucans have been
found to react with one or multiple of the following cell surface
receptors: complement receptor-3 (CR3), lactosylceramides,
scavenger receptors and dectin-1 (73–76).
Dectin-1 is considered the main β-D-glucan receptor. The β-D-glucan
binding to myeloid cell receptors triggers, according to the bound
receptor, a series of signaling events that modulate innate and
subsequently adaptive immune responses, mainly through release of
pro-inflammatory cytokines (IL-1α/β, IL-6, IL-8, IL-12, TNF-α) as
well as cytotoxic molecules working also as inflammatory mediators
[nitric oxide (NO) and hydrogen peroxide
(H2O2)], as cited in the previous paragraph.
The activation of macrophages performed by (1→3)-β-D-glucans is
thought to be consequent to binding of the polymer to CR3
(CD11/CD18) receptors (42). The
receptor-glucan interaction triggers phagocytosis, respiratory
burst and secretion of cytokines such as TNF-α in addition to IL-10
(77,78). For an adequate use of glucans as
immune enhancers, it is necessary to point out that glucan polymers
derived from various sources can largely differ in binding affinity
with specific receptors (from 24 μM to 11 nM). Consequently,
different biological effects can be promoted according to the
source of the chosen molecule (68). Human monocytes (but also
fibroblasts) express many glucan receptors which can differentiate
between the polymers of (1→3)-β-D-glucan (68,79).
Neutrophils exhibit lactosylceramide that mediates the response to
PGG-glucan and CR3 mediates cytotoxicity for iC3b-opsonized target
cells (35,80). CR3 receptor is also represented on
natural killer cells (NK). Consequently, the triggering of
complement alternative activation pathway by β-D-glucans with the
availability of iC3b fragment elicits a high-avidity link of
iC3b-opsonized cells (tumor cells or pathogens) to the receptors
for iC3b and stimulates phagocytosis by monocytes and cytotoxic
degranulation by NK cells (81).
Macrophage/monocytes present on their surface scavenger receptors
and dectin-1 recognizing (1→3)-β-D-glucans and non-opsonic zymosan.
Dectin-1 is also represented on dendritic cells (see below)
(82,83).
Some studies have suggested the complement receptor
type 3 (CR3, also CD11b/CD18) is a prime candidate for β-D-glucan
receptor on human monocytes, neutrophils and NK cells (80). More recently, dectin-1 was
definitively identified as the most important β-D-glucan receptor
(84). Human and murin dectin-1
mostly show a similar structure and function (85). Dectin-1 is a small type II
transmembrane glycoprotein receptor containing one lectin-like
carbohydrate recognition domain which is able to recognize (1→3)-β-
and/or (1→6) β-D-glucans as well as fungi particles (86). This receptor is highly expressed on
macrophages and granulocytes, but also on dendritic cells with
effects on T and B cell responses (75,87,88).
Dectin-1 presents two ligand-binding sites, one able to recognize
the endogenous ligand on T cells and the other for exogenous
carbohydrate (89). It has been
shown that dectin-1 is able to mediate inflammatory cellular
responses to β-D-glucans. The release of TNF-α, after interaction
of β-D-glucans with the superficial part of the receptor, needs the
cytoplasmic tail and immunoreceptor tyrosine activation motif of
Dectin-1 as well as Toll-like receptor (TLR)-2 and Myd88 (71,73,90,91).
The role of dectin-1 is important on dendritic cells (DCs)
(73,75). Recent studies have shown the
capability of DCs to stimulate antigen specific CD8+ T
cell responses after dectin-1 is bound by the anti-dectin-1
antibody. The receptor-Ab interaction triggers a Syk-dependent
pathway with upregulation of costimulatory molecules, secretion of
cytokines and chemokines. This induces enhancement of antigen
presentation, priming and expansion of antigen specific
CD8+ T cells. A similar effect can be hypothesized after
dectin-1 bounding to β-glucans (92).
Moreover, glucan-dependent dectin-1 signaling in
macrophages and bone marrow-derived dendritic cells has been found
to trigger the formation of LC3II, a central component in
autophagy, as well as recruitment of LC3II to phagosomes. Here also
Syk is involved. This promoted presentation of fungal-derived
antigens to CD4 T cells occurs by facilitation of MHC class II
molecule recruitment to phagosomes (93,94).
Polysaccharides from fruiting bodies, cultured
mycelia and cultured filtrates of basidiomycetes have been reported
to present antitumor activity. These antitumor polysaccharides are
different in their chemical composition depending on their
molecular weight, purity and degree of branches (3,82).
As quoted by Bulmer et al(95), the first reports on the antitumor
properties of extracts from fungi were published by Ringler in 1955
(a PhD thesis) and Lukas et al(96). Since then, many antitumor
polysaccharides were isolated from fungi and extensively studied,
especially in Japan (12,17,97–99).
As has been emphasized, the therapeutic efficacy of these
polysaccharides can greatly differ according to their chemical
composition, configuration and physical properties. A wide range of
glucans extending from homopolymers to highly complex
heteropolymers were found to exhibit antitumor activity and most of
the antitumor polysaccharides presented the same basic β-D-glucan
structure with different types of glycosidic bounds. Glucans with
high molecular weight appear to be more effective than those with
low molecular weight (3,99). Differences in the effectiveness of
mushroom glucan preparations are related to the type of polymer
(according to the type of β-backbone) but also to the presence and
proportion of various products in the same preparation. The
simultaneous presence of different products may elicit multiple
stimulatory activities with possible enhancement of the
immunomodula-tory effects. A clear example of this possible
collaboration, related to products obtained from Agaricus
blazei, is reported by Borchers et al in their review on
mushrooms as anticancer immune modulators (100). They assert that the mushroom
Agaricus blazei contains more compounds [an antitumor glucan
with a (1→6)-β-backbone, an (1→6)-α- and (1→4)-α-D-glucan complex
and a glucomannan with a main chain of (1→2) β-linked
D-mannopyranosyl residues] that were found to inhibit tumorigenesis
(101–103). The preparation by aqueous
extraction from powdered, dry fruiting body was less efficient than
the direct administration of the complete dry powdered form. In
rats fed with either aqueous extract or dry powdered preparation,
the complete dry powder developed a better antimutagenic activity
(104). Similar results were
found also for diets containing powdered Lentinula edodes
(shiitake) (105,106). The interpretation of Borchers
et al is that different polysaccharides can cooperate by
targeting different cell subsets by different receptors.
Consequently, a more complex and effective stimulation would be
more easily elicited when whole-mushroom extracts are used
(100,102,107–109).
Polysaccharides or polysaccharide-protein complexes
obtained from natural sources are generally reported to not produce
direct cytotoxic action on tumor cells, but to induce host-mediated
antitumor immune responses. However, the complete absence of direct
effects on tumor cells cannot be totally excluded according to some
recent studies (110–112). Pioneering studies of Di Luzio
et al, using intravenous injection of soluble or particulate
glucan, documented significant regressions of a syngeneic
anaplastic mammary carcinoma and B16F10 melanoma in A/J and C57BL/6
mice, respectively (113). It has
also been demonstrated that orally administrated yeast-derived as
well as mushroom-derived β-(1–3)
glucan had significant inhibitory effects on the growth of
metastatic cancer cells using in vivo models of cancer
(114,115). Animals that received treatment
with PSK, β-(1→4)-D-glucans with (1→6)-β-glycopyranosidic side
chains showed an increased number of neutrophils and a significant
decrease in the size and number of lung metastasis (116). Therefore, the effects may not be
limited only for use in the early stages of carcinogenesis or tumor
development as suggested by the enhancement of immune responses
(IL-1β, IFN-γ, TNF-α and IL-12 production, NK cell increase,
macrophages activation), an increase of the host's antioxidant
capacity and upregulation of phase I and phase II enzymes involved
in the metabolic transformation as well as detoxification of
mutagenic compounds (117,118).
Finally, the efficacy of some types of fungal derivatives like
lentinan, pachymaran, scleroglucan, curdland, grifolan and
Agaricus blazei (1→3)-β-D-glucan resulted particularly high
in various in vivo models of cancer. According to the
reports, the tumor inhibition ratio in animal models range from
90.4 (scleroglucan) to 99.6% (lentinan) (119–122).
Glucans have also been proposed as an adjuvant. Some
examples in animal models suggest an increasing of chemo- or
immunotherapy efficacy when they are associated to polysaccharides,
mainly glucans. The combination of an anti-MUC1 mAb with β-glucans
significantly increased 20% the rate of RMA-S-MUC1 tumor regression
in C57BL/6 mice (14).
Ganoderma lucidum polysaccharides were also able to prolong
the survival of Lewis carcinoma bearing C57BL/6 mice and to enhance
antitumor activities of cytotoxic drugs and immunomodulators
(123). Of particular interest is
the possibility of using glucans for triggering
complement-dependent antitumor cytotoxicity.
As previously cited, complement is a relevant
mediator of antitumor β-D-glucan effects even after oral
administration. Complement is an important part of the innate
immunity against microorganisms that exhibit β-D-glucans as a
surface component. These molecules are not expressed by tumor cells
and, consequently, tumor cells cannot trigger CR3-dependent
cellular cytotoxicity (CR3-DCC) (124). Oral administration of β-D-glucans
may modify this condition. Glucan, in insoluble form, can be
processed by gastrointestinal macrophages to soluble form. Once the
soluble form is delivered, it can reach CR3 of bone marrow
granulocytes and tissue macrophages making iC3b fragments
available. In this way, the promotion of cytotoxity against tumor
cells could be the result of contemporary presence of iC3b
fragments and antitumor antibodies (125). Complement activation and
deposition of iCR3 on tumor cells needs the presence of antitumor
antibodies to produce a synergistic effect. Such an effect, leading
to tumor regression, was evidenced by various authors using
administration of β-D-glucans together with monoclonal antibodies
against GD2 ganglioside, G250 protein, CD20 protein, respectively
in experimental neuroblastoma, carcinoma and CD20+
lymphoma (126–128). Evidence of the dependence of this
approach from complement involvement was given by failures of
therapy in mice deficient in CR3 (CD11b−/−) or C3
(C3−/−) (129–131). This approach, since the
progressively larger use of monoclonal antibodies in anticancer
therapies, results in a particularly appealing and prospective
application of β-D-glucans as effective enhancers of antitumor
responses, as also demonstrated by recent literature (18,132).
A substantial amount of literature has been
accumulated in past decades on the medical potential of
polysaccharides, particularly the β-D-glucans, from medical
mushrooms used by the traditional medicine. Especially in recent
years, the interest in these molecules or compounds has arisen
together with the understanding of innate immunity implications
during carcinogenesis and cancer development. Unfortunately, many
clinical reports lack a specific rationale or simply describe
effects according to traditional medicine application. However,
some recent studies on gastric and colorectal cancer patients
indicate the possible efficacy of these saccharides (133–135). Experimental studies have in large
part clarified the basic mechanisms involved in the immune
stimulation produced by β-D-glucans, especially with the knowledge
on dectin-1 and C3-iCR3 involvement. A clear definition of the
biologically active molecules and a more detailed chemical and
biological characterization of the glucans from different sources
appear necessary to better define the rationale of their
application in anticancer therapies as well as other suitable
pathologies. For example, it was suggested by Hamuro and Chihara
that only extracts able to deactivate protein helices (as tested on
bovine serum albumin) were active against tumors (136). Furthermore, β-D-glucans also
appear suitable for use in nanomedicine for preparation of natural
nanocarriers for drug or biological molecule delivery (137–139). The creation of gels or lattices
based on β-D-glucans has also been proposed for various
utilizations (e.g., in wound healing by stimulating macrophage
activation and collagen deposition) (140,141). The addition of new areas of
application, apart from the immunological use in oncology, opens
new interesting perspectives and makes the study of β-D-glucan
chemical and biological properties a prospective field of
research.
We thank the support of: ARPA
Foundation, Pisa, (IT), RVO 61388971 and RVO 67985904 (CZ).
|
1.
|
Tzianabos A: Polysaccharide
immunomodulators as therapeutic agents: structural aspects and
biologic function. Clin Microbiol Rev. 13:523–533. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Han SB, Lee CW, Jeon YJ, Hong ND, Yoo ID,
Yang KH and Kim HM: The inhibitory effect of polysaccharides
isolated from Phellinus liteus on tumor growth and metastasis.
Immunopharmacology. 41:157–164. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Ooi VE and Liu F: Immunomodulation and
anticancer activity of polysaccharide-protein complex. Curr Med
Chem. 7:715–729. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Falch BH, Espevik T, Ryan L and Stokke BT:
The cytokine stimulating activity of (1→3)-beta-D-glucans is
dependent on the triple helix conformation. Carbohydr Res.
329:587–596. 2000.
|
|
5.
|
Barsanti L, Passarelli V, Evangelista V,
Frassanito AM and Gualtieri P: Chemistry, physico-chemistry and
applications linked to biological activities of β-glucans. Nat Prod
Rep. 28:457–466. 2011.PubMed/NCBI
|
|
6.
|
Yap AT and Ng ML: An improved method for
the isolation of lentina from the edible and medicinal shiitake
mushroom, Lentinus edodes(Berk.) Sing.
(Agaricomycetideae). Int J Med Mushr. 3:6–19. 2001.
|
|
7.
|
Riggi SJ and Di Luzio NR: Identification
of a reticuloendothelial stimulating agent in zymosan. Am J
Physiol. 200:297–300. 1961.PubMed/NCBI
|
|
8.
|
Wasser SP: Current findings, future trends
and unsolved problems in studies of medicinal mushrooms. Appl
Microbiol Biotechnol. 89:1323–1332. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Chang ST and Wasser S: The role of
culinary-medicinal mushrooms on human welfare with a pyramid model
for human health. Int J Med Mushr. 14:95–134. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Pillemer L and Ecker EE: Anticomplementry
factor in fresh yeast. J Biol Chem. 137:139–142. 1941.
|
|
11.
|
Pillemer L, Blum L, Pensky J and Lepow IH:
The requirement for magnesium ions in the inactivation of the third
component of human complement (C'3) by insoluble residues of yeast
cells (zymosan). J Immunol. 71:331–338. 1953.PubMed/NCBI
|
|
12.
|
Ohano N, Miura NN, Nakajima M and Yadomae
T: Antitumor 1,3-beta-glucan from cultured fruit body of
Sparassis crispa. Biol Pharm Bull. 7:866–872. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Novak M and Vetvicka V: Glucans as
biological response modifiers. Endocr Metab Immune Disord Drug
Targets. 9:67–75. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Hong F, Hansen RD, Yan J, Allendorf DJ,
Baran JT, Ostroff CR and Ross GD: Beta-glucans functions as an
adjuvant for monoclonal antibody immunotherapy by recruiting
tumoricidal granulocytes as killer cells. Cancer Res. 63:9023–9031.
2003.PubMed/NCBI
|
|
15.
|
Kamiryo Y, Yajima T, Saito K, Nishimura H,
Fushimi T, Ohshima Y, Tsukamoto Y, Naito S and Yoshikai Y: Soluble
branched (1,4)- β-D-glucans from Acetobacter species enhance
antitumor activitiese against MHC class I-negative and postitive
malignant melanoma through augmented NK activity and cytotoxic
T-cell response. Int J Cancer. 115:769–776. 2005.
|
|
16.
|
Ryoyama K, Kidachi Y, Yamaguchi H, Kajiura
H and Takata H: Antitumor activity of an enzymatically synthesized
α-1,6 branched α-1,4-glucan, glycogen. Biosci Biotechnol Biochem.
68:2332–2340. 2004.
|
|
17.
|
Chan GC, Chan WK and Sze DM: The effects
of beta-glucan on human immune and cancer cells. J Hematol Oncol.
2:252009. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Aleem E: β-glucans and their applications
in cancer therapy: focus on human studies. Anticancer Agents Med
Chem. Nov 5–2012, (Epub ahead of print).
|
|
19.
|
Usui S, Tomono Y, Sakai M, Kiho T and Ukai
S: Preparation and antitumor activities of beta-(1→6) branched
(1→3)-beta-D-glucan derivatives. Biol Pharm Bull. 18:1630–1636.
1995.
|
|
20.
|
Synytsya A and Novák M: Structural
diversity of fungal glucans. Carbohydr Polym. 92:792–809. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Sawai M, Adachi Y, Kanai M, Matsui S and
Yadomae T: Extraction of conformationally stable (1–6)–branched
(1–3)-β-D-glucans from premixed edible mushroom powders by cold
alkaline solution. Int J Med Mushr. 4:197–205. 2002.
|
|
22.
|
Kitamura S, Hori T, Kurita K, Takeo K,
Hara C, Itoh W, Tabata K, Elgsaeter A and Stokke BT: An antitumor,
branched (1→3)-beta-D-glucan from a water extract of fruiting
bodies of Cryptoporus volvatus. Carbohydr Res. 263:111–121.
1994.
|
|
23.
|
Ohno N, Kurachi K and Yadomae T: Antitumor
activity of a highly branched (1–3)-beta-D-glucan, SSG, obtained
from Sclerotinia sclerotiorum IFO 9395. J Pharmacobiodyn.
10:478–486. 1987.
|
|
24.
|
Fujimoto S, Furue H, Kimura T, Kondo T,
Orita K, Taguchi T, Yoshida K and Ogawa N: Clinical outcome of
postoperative adjuvant immunochemotherapy with sizofiran for
patients with resectable gastric cancer: a randomised controlled
study. Eur J Cancer. 27:1114–1118. 1991. View Article : Google Scholar
|
|
25.
|
Ishibashi K, Miura NN, Adachi Y, Ohno N
and Yadomae T: Relationship between solubility of grifolan, a
fungal 1,3-beta-D-glucan and production of tumor necrosis factor by
macrophages in vitro. Biosci Biotechnol Biochem. 65:1993–2000.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Blecher P and Mackin W: Betafectin
PGG-glucan: a novel carbohydrate immunodulator with anti-infective
properties. J Biotechnol Healthcare. 2:207–222. 1995.
|
|
27.
|
Chihara G, Maeda Y, Hamuro J, Sasaki T and
Fukuoka F: Inhibition of mouse sarcoma 180 by polysaccharides from
Lentinus edodes. Nature. 222:687–696. 1969. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Ohno N, Uchiyama M, Tsuzuki A, Tokunaka K,
Miura NN, Adachi Y, Aizawa MW, Tamura H, Tanaka S and Yadomae T:
Solubilization of yeast cell-wall β-(1–3)-D-glucans by sodium
hypochlorite oxidation and dimethyl sulfoxide extraction. Carbohydr
Res. 316:161–172. 1999.
|
|
29.
|
Majtan J: Pleuran (β-Glucan from
Pleurotus ostreatus): an effective nutritional supplement
against upper respiratory tract infections? Med Sport Sci.
59:57–61. 2013.
|
|
30.
|
Volman JJ, Helsper JP, Wei S, Baars JJ,
van Griensven LJ, Sonnenberg AS and Plat J: Effects of
mushroom-derived beta-glucan-rich polysaccharide extracts on nitric
oxide production by bone marrow-derived macrophages and nuclear
factor-kappaB transactivation in Caco-2 reporter cells: can effects
be explained by structure? Mol Nutr Food Res. 54:268–276. 2010.
View Article : Google Scholar
|
|
31.
|
Zhang P, Zhang L and Cheng S: Chemical
structure and molecular weights of α-(1,3)-D-glucan from
Lentinus edodes. Biosci Biotechnol Biochem. 63:1197–1202.
1999.
|
|
32.
|
Brown GD and Gordon S: Fungal β-glucans
and mammalian immunity. Immunity. 19:311–315. 2003.
|
|
33.
|
Sasaki T and Takasuka N: Further study of
the structure of lentinan, an antitumor polysaccharide from
Lentinus edodes. Carbohydr Res. 47:99–104. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Zhang M, Cui SW, Cheung PCK and Wang Q:
Antitumor polysaccharides from mushrooms: a review on their
isolation process, structural characteristics and antitumor
activity. Trends Food Sci Technol. 18:4–19. 2007. View Article : Google Scholar
|
|
35.
|
Zimmerman JW, Lindermuth J, Fish PA,
Palace GP, Stevenson TT and DeMong DE: A novel
carbohydrate-glycosphingolipid interaction between a
beta-(1–3)-glucan immunomodulator, PGG-glucan and lactosylceramide
of human leukocytes. J Biol Chem. 273:22014–22020. 1998.PubMed/NCBI
|
|
36.
|
Zimmermann G and Krueger K:
Ultracentrifuge studies on properdin sera of zymosan-vaccinated
cattle, rabbits, guinea pigs, rats and mice and on bovine properdin
serum fractions. Acta Biol Med Ger. 11:902–917. 1963.(In
German).
|
|
37.
|
Cassone A, Bistoni F, Cenci E, Pesce CD,
Tissi L and Marconi P: Immunopotentiation of anticancer
chemotherapy by Candida albicans, other yeasts and insoluble
glucan in an experimental lymphoma model. Sabouraudia. 20:115–125.
1982.PubMed/NCBI
|
|
38.
|
Haggård L, Andersson M and Punga AR:
β-glucans reduce LDL cholesterol in patients with myasthenia
gravis. Eur J Clin Nutr. 67:226–227. 2013.
|
|
39.
|
Zhang Y, Xia L, Pang W, Wang T, Chen P,
Zhu B and Zhang J: A novel soluble β-1,3-d-glucan Salecan reduces
adiposity and improves glucose tolerance in high-fat diet-fed mice.
Br J Nutr. 13:1–9. 2012.
|
|
40.
|
Di Luzio NR: Update on the
immunomodulating activities of glucans. Springer Semin
Immunopathol. 8:387–400. 1985.PubMed/NCBI
|
|
41.
|
Di Luzio NR: Immunopharmacology of glucan:
a broadspectrum enhancer of host defence mechanisms. Trends
Pharmacol Sci. 4:344–347. 1983.PubMed/NCBI
|
|
42.
|
Kaneko Y and Chihara G: Potentiation of
host resistance against microbal infections by lentinan and its
related polysaccharides. Microbal Infections: Role of Biological
Response Modifers. Friedman H, Klein TW and Yamaguchi H: Plenum
Press; New York, NY: pp. 201–215. 1992, View Article : Google Scholar : PubMed/NCBI
|
|
43.
|
Williams DL, Browder IW and Di Luzio NR:
Immunotherapeutic modification of Escherichia coli-induced
experimental peritonitis and bacteremia by glucan. Surgery.
93:448–454. 1983.
|
|
44.
|
Ling J, Melican D, Cafro L, Palace G,
Fisette L, Armstrong R and Patchen ML: Enhanced clearance of a
multiple antibiotic resistance Staphilococcus aureus in rats
treated with PGG-glucans is associated with increased leukocyte
counts and increased neutrophil oxidative burst activity. Int J
Immunopharmacol. 20:595–614. 1998.
|
|
45.
|
Wakshull E, Brunke-Reese D, Lindermuth J,
Fisette L, Nathans RS, Crowley JJ, Tufts JC, Zimmerman J, Mackin W
and Adams DS: PGG-glucan, a soluble beta-(1,3)-glucan, enhances the
oxidative burst response, microbicidal activity and activates an
NF-kappa B-like factor in human PMN: evidence for a
glycosphingolipid beta-(1,3)-glucan receptor. Immunopharmacology.
41:89–107. 1999. View Article : Google Scholar
|
|
46.
|
Burgaletta C, Territo MC, Quan SG and
Golde DW: Glucan activated macrophages: functional characteristics
and surface morphology. J Reticuloendothel Soc. 23:195–104.
1978.PubMed/NCBI
|
|
47.
|
Chanput W, Reitsma M, Kleinjans L, Mes JJ,
Savelkoul HF and Wichers HJ: β-glucans are involved in
immune-modulation of THP-1 macrophages. Mol Nutr Food Res.
56:822–833. 2012.
|
|
48.
|
Adachi Y, Ohno N, Ohsawa S and Yadomae T:
Change biological activities of (1–3)-beta-D-glucan from Grifola
frondosa upon molecular weight reduction by heat treatment. Chem
Pharm Bull (Tokyo). 38:477–481. 1990.
|
|
49.
|
Selijelid R, Bogwald J, Rasmussen LT, Larm
O, Hoffman J, Berge A and Ugelstad J: In vivo activation of mouse
macrophages with beta-1,3-D-glucan-derivatized plastic beads. Scand
J Immunol. 6:601–605. 1985.PubMed/NCBI
|
|
50.
|
Patchen ML and Lotzova E: Modulation of
murine hemopoiesis by glucans. Exp Hematol. 8:409–422.
1980.PubMed/NCBI
|
|
51.
|
Hofer M and Pospíšil M: Modulation of
animal and human hematopoiesis by β-glucans: a review. Molecules.
16:7969–7979. 2011.
|
|
52.
|
Tsuzuki A, Tateishi T, Ohno N, Adachi Y
and Yadomae T: Increase of hematopoietic responses by triple or
single helical conformer of an antitumor (1→3)-beta-D-glucan
preparation, Sonifilan, in cyclophosphamide-induced leukopenic
mice. Biosci Biotechnol Biochem. 63:104–110. 1999.
|
|
53.
|
Browder W, Williams D, Preutes H, Olivero
G, Enrichens F, Mao P and Franchello A: Beneficial effect of
enhanced macrophage function in the trauma patient. Ann Surg.
211:605–613. 1990.PubMed/NCBI
|
|
54.
|
Czop JK and Austen KF: Generation of
leukotrienes by human monocytes upon stimulation of their
beta-glucan receptor during phagocytosis. Proc Natl Acad Sci USA.
82:2751–2755. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
55.
|
Goodridge HS, Wolf AJ and Underhill DM:
Beta-glucan recognition by the innate immune system. Immunol Rev.
230:38–50. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
56.
|
Czop JK and Austen KF: Properties of
glycans that activate the human alternative complement pathway and
interact with human monocyte beta-glucan receptor. J Immunol.
135:3388–3393. 1985.PubMed/NCBI
|
|
57.
|
Doita M, Rasmussen LT, Seljelid R and
Lipsky PE: Effect of soluble aminated beta-1,3-D-polyglucose on
human monocytes: stimulation of cytokine and prostaglandin E2
production but not antigen-presenting function. J Leukoc Biol.
49:342–351. 1991.PubMed/NCBI
|
|
58.
|
Konopski Z, Seljelid R and Eskeland T:
Cytokines and PGE2 modulate the phagocytic function of beta-glucan
receptor in macrophages. Scand J Immunol. 37:587–592. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
59.
|
Volman JJ, Ramakers JD and Plat J: Dietary
modulation of immune function by beta-glucans. Physiol Behav.
94:276–284. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
60.
|
Senoglu N, Yuzbasioglu MF, Aral M, Ezberci
M, Kurutas EB, Bulbuloglu E, Ezberci F, Oksuz H and Ciragil P:
Protective effects of N-acetylcysteine and beta-glucan pretreatment
on oxidative stress in cecal ligation and puncture model of sepsis.
J Invest Surg. 21:237–243. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
61.
|
Bedirli A, Kerem M, Pasaoglu H, Akyurek N,
Tezcaner T, Elbeg S, Memis L and Sakrak O: Beta-glucan attenuates
inflammatory cytokine release and prevents acute lung injury in an
experimental model of sepsis. Shock. 27:397–401. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
62.
|
Sener G, Toklu H, Ercan F and Erkanli G:
Protective effect of beta-glucan against oxidative organ injury in
a rat model of sepsis. Int Immunopharmacol. 5:1387–1396. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
63.
|
Williams DL, Sherwood ER, Browder IW,
McNamee RB, Jones EL, Rakinic J and DiLuzio NR: Effect of glucan on
neutrophil dynamics and immune function in Escherichia coli
peritonitis. J Surg Res. 44:54–61. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
64.
|
Onderdonk AB, Cisneros RL, Hinkson PL and
Ostroff GR: Anti-infective effect of poly β1-6
glucotriosyle-β1-3glucopyranose glucan in vivo. Infect Immun.
60:1642–1647. 1992.
|
|
65.
|
Stashenko P, Wang CY, Riley E, Wu Y,
Ostroff G and Niederman R: Reduction of infection-stimulated
periapical bone resorption by the biological response modifier
PGG-glucan. J Dent Res. 74:323–330. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
66.
|
Berovic M, Habijanic J, Zore I, Wraber B,
Hodzar D, Boh B and Pohleven F: Submerged cultivation of Ganoderm
lucidum biomass and immunostimulatory effects of fungal
polysaccharides. J Biotecnol. 103:77–86. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
67.
|
Dellinger EP, Babineau TJ, Bleicher P,
Kaiser AB, Seibert GB, Postier RG, Vogel SB, Norman J, Kaufman D,
Galandiuk S and Condon RE: Effect of PGG-glucan on the rate of
serious postoperative infection or death observed after high-risk
gastrointestinal operations. Betafectin Gastrointestinal Study
Group Arch Surg. 134:977–983. 1999.
|
|
68.
|
Mueller A, Raptis J, Rice PJ, Kalbffleisch
JH, Stout RD, Ensley HE, Browder W and Williams DL: The influence
of glucan polymer structure and solution conformation on binding to
(1–3)-β-D-glucans receptors in human monocyte-like cell line.
Glycobiology. 10:339–346. 2000.
|
|
69.
|
Williams DL, Mueller A and Browder W:
Glucan-based macrophages stimulators: a review of their
anti-infective potential. Clin Immunother. 5:392–399. 1996.
View Article : Google Scholar
|
|
70.
|
Hardison SE and Brown GD: C-type lectin
receptors orchestrate antifungal immunity. Nat Immunol. 13:817–822.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
71.
|
Saijo S and Iwakura Y: Dectin-1 and
Dectin-2 in innate immunity against fungi. Int Immunol. 23:467–472.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
72.
|
Kapetanovic R and Cavaillon JM: Early
events in innate immunity in the recognition of microbial
pathogens. Expert Opin Biol Ther. 7:907–918. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
73.
|
Tsoni SV and Brown GD: beta-glucans and
dectin-1. Ann NY Acad Sci. 1143:45–60. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
74.
|
Murphy EA, Davis JM and Carmichael MD:
Immune modulating effects of β-glucan. Curr Opin Clin Nutr Metab
Care. 13:656–661. 2010.
|
|
75.
|
Albeituni SH and Yan J: The effects of
β-glucans on dendritic cells and implications for cancer therapy.
Anticancer Agents Med Chem. Oct 18–2012, (Epub ahead of print).
|
|
76.
|
Józefowski S, Yang Z, Marcinkiewicz J and
Kobzik L: Scavenger receptors and β-glucan receptors participate in
the recognition of yeasts by murine macrophages. Inflamm Res.
61:113–126. 2012.
|
|
77.
|
Chen JT and Hasumi K: Activation of
peritoneal macrophages in patients with gynecological malignancies
by sizofiran and recombinant interferon-gamma. Biotherapy.
6:189–194. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
78.
|
Kelly EK, Wang L and Ivashkiv LB:
Calcium-activated pathways and oxidative burst mediate
zymosan-induced signaling and IL-10 production in human
macrophages. J Immunol. 184:5545–5552. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
79.
|
Kougias P, Wei D, Rice PJ, Ensley HE,
Kalbfleisch J, Williams DL and Browder IW: Normal human fibroblasts
express pattern recognition receptors for fungal
(1→3)-beta-D-glucans. Infect Immun. 69:3933–3938. 2001.PubMed/NCBI
|
|
80.
|
Vetvicka V, Thornton BP and Ross GD:
Soluble β-glucan polysaccharide binding to the lectin site of
neutrophil or natural killer cell complemet receptor type 3
(CD11b/CD18) generates a primed state of the receptor capable of
mediating cytotoxicity of iC3b-opsonized target cells. J Clin
Invest. 98:50–61. 1998.
|
|
81.
|
Ross GD, Vetvicka V, Yan J, Xia Y and
Vetvickova J: Therapeutic intervention with complement and
beta-glucan in cancer. Immunopharmacology. 42:61–74. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
82.
|
Rice PJ, Kelley JL, Kogan G, Ensley HE,
Kalbfleisch JH, Browder IW and Williams DL: Human monocyte
scavenger receptors are pattern recognition receptors for
(1→3)-beta-D-glucans. J Leukoc Biol. 72:140–146. 2002.
|
|
83.
|
Brown GD, Taylor PR, Reid DM, Willment JA,
Williams DL, Martinez-Pomares L, Wong SY and Gordon S: Dectin-1 is
a major beta-glucan receptor on macrophages. J Exp Med.
196:407–412. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
84.
|
Ariizumi K, Shen GL, Shikano S, Xu S,
Ritter R III, Kumamoto T, Edelbaum D, Morita A, Bergstresser PR and
Takashima A: Identification of a novel, dendritic cell-associated
molecule, dectin-1, by subtractive cDNA cloning. J Biol Chem.
275:20157–20167. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
85.
|
Willment JA, Gordon S and Brown GD:
Characterization of the human beta-glucan receptor and its
alternatively spliced isoforms. J Biol Chem. 276:43818–43823. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
86.
|
Brown GD and Gordon S: Immune recognition:
a new receptor for beta-glucans. Nature. 413:36–37. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
87.
|
Taylor PR, Brown GD, Reid DM, Willment JA,
Martinez-Pomares L, Gordon S and Wong SY: The beta-glucan receptor,
dectin-1, is predominantly expressed on the surface of cells of the
monocyte/macrophage and neutrophil lineages. J Immunol.
169:3876–3882. 2002. View Article : Google Scholar
|
|
88.
|
Agrawal S, Gupta S and Agrawal A: Human
dendritic cells activated via dectin-1 are efficient at priming
Th17, cytotoxic CD8 T and B cell responses. PLoS One. 5:e134182010.
View Article : Google Scholar : PubMed/NCBI
|
|
89.
|
Ozment-Skelton TR, Goldman MP, Gordon S,
Brown GD and Williams DL: Prolonged reduction of leukocyte
membrane-associated Dectin-1 levels following beta-glucan
administration. J Pharmacol Exp Ther. 318:540–546. 2006. View Article : Google Scholar
|
|
90.
|
Brown GD, Herre J, Williams DL, Willment
JA, Marshall AS and Gordon S: Dectin-1 mediates the biological
effects of beta-glucans. J Exp Med. 197:1119–1124. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
91.
|
Taylor PR, Tsoni SV, Willment JA, Dennehy
KM, Rosas M, Findon H, Haynes K, Steele C, Botto M, Gordon S and
Brown GD: Dectin-1 is required for beta-glucan recognition and
control of fungal infection. Nat Immunol. 8:31–38. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
92.
|
Ni L, Gayet I, Zurawski S, Duluc D, Flamar
AL, Li XH, O'Bar A, Clayton S, Palucka AK, Zurawski G, Banchereau J
and Oh S: Concomitant activation and antigen uptake via human
dectin-1 results in potent antigen-specific CD8+ T cell
responses. J Immunol. 185:3504–3513. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
93.
|
Ma J, Becker C, Lowell CA and Underhill
DM: Dectin-1-triggered recruitment of light chain 3 protein to
phagosomes facilitates major histocompatibility complex class II
presentation of fungal-derived antigens. J Biol Chem.
287:34149–34156. 2012. View Article : Google Scholar
|
|
94.
|
Dragicevic A, Dzopalic T, Vasilijic S,
Vucevic D, Tomic S, Bozic B and Colic M: Signaling through
Toll-like receptor 3 and Dectin-1 potentiates the capability of
human monocyte-derived dendritic cells to promote T-helper 1 and
T-helper 17 immune responses. Cytotherapy. 14:598–607. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
95.
|
Bulmer GS, Beneke ES and Stevens JA:
Studies on Calvatia gigantea. III. Antitumor substances
produced by mycelium from germinated spores and parent
basidiocarps. Mycologia. 54:621–625. 1962.
|
|
96.
|
Lukas EH, Ringler RL, Byerrum RU, Stevens
JA, Clarke DA and Stock CC: Tumor inhibitors in Boletus
edulis and other holobasidiomycetes. Antibiot Chemother.
7:1–14. 1957.
|
|
97.
|
Tukonaka K, Ohano N, Adachi Y, Tanaka S,
Tamura H and Yadomae T: Immunopharmacological and
immunotoxicological activities of a water-soluble (1–3)-β-glucan,
CSBG from Candiada spp. Int J Immunopharmacol. 5:383–394.
2000.PubMed/NCBI
|
|
98.
|
Suzuki I, Takeyama T, Ohano N, Oikawa S,
Sato K, Suzuki Y and Yadomae T: Antitumor effect of polysaccharide
griofolan NMF-5N on syngeneic tumor in mice. J Pharmacobiodyn.
2:72–77. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
99.
|
Ren L, Perera C and Hemar Y: Antitumor
activity of mushroom polysaccharides: a review. Food Funct.
3:1118–1130. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
100.
|
Borchers AT, Keen CL and Gershwin ME:
Mushrooms, tumors and immunity: an update. Exp Biol Med.
229:393–406. 2004.PubMed/NCBI
|
|
101.
|
Itoh H, Ito H, Amano H and Noda H:
Inhibitory action of a (1→6)-beta-D-glucan-protein complex (F
III-2-b) isolated from Agaricus blazei Murill
(‘himematsutake’) on Meth A fibrosarcoma-bearing mice and its
antitumor mechanism. Jpn J Pharmacol. 66:265–271. 1994.
|
|
102.
|
Mizuno M, Morimoto M, Minato K and
Tsuchida H: Polysaccharides from Agaricus blazei stimulate
lymphocyte T-cell subsets in mice. Biosci Biotechnol Biochem.
62:434–437. 1998.
|
|
103.
|
Mizuno M, Minato K, Ito H, Kawade M, Terai
H and Tsuchida H: Antitumor polysaccharide from the mycelium of
liquid-cultured Agaricus blazei mill. Biochem Mol Biol Int.
47:707–714. 1999.PubMed/NCBI
|
|
104.
|
Pinheiro F, Faria RR, de Camargo JLV,
Spinardi-Barbisan ALT, da Eira AF and Barbisan LF: Chemoprevention
of preneoplastic liver foci by dietary mushroom Agaricus
blazei Murill in the rat. Food Chem Toxicol. 41:1543–1550.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
105.
|
Alves de Lima PL, Delmanto RD, Sugui MM,
da Eira AF, Salvadori DM, Speit G and Ribeiro LR: Letinula
edodes(Berk.) Pegler (Shiitake) modulates genotoxic and
mutagenic effects induced by alkylating agents in vivo. Mutat Res.
496:23–32. 2001.
|
|
106.
|
Sugui MM, Alves de Lima PL, Delmanto RD,
da Eira AF, Salvadori DMF and Ribeiro LR: Antimutagenic effect of
Lentinula edodes(BERK.) Pegler mushroom and possible
variation among lineages. Food Chem Toxicol. 41:555–560. 2003.
|
|
107.
|
Zhang Y, Mills GL and Nair MG:
Cyclooxygenase inhibitory and antioxidant compounds from the
mycelia of the edible mushroom Grifola frondosa. J Agric
Food Chem. 50:7581–7585. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
108.
|
Prescott SM and Fitzpatrick FA:
Cyclooxygenase-2 and carcinogenesis. Biochim Biophys Acta.
1470:M69–M78. 2000.PubMed/NCBI
|
|
109.
|
Kodama N, Komuta K, Sakai N and Nanba H:
Effects of D-fraction, a polysaccharide from Grifola frondosa on
tumor growth involve activation of NK cells. Biol Pharm Bull.
25:1647–1650. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
110.
|
Lin ZB and Zhang HN: Anti-tumor and
immunoregulatory activities of Ganoderma lucidum and its
possible mechanisms. Acta Pharmacol Sin. 25:1387–1395.
2004.PubMed/NCBI
|
|
111.
|
Youn MJ, Kim JK, Park SY, Kim Y, Park C,
Kim ES, Park KI, So HS and Park R: Potential anticancer properties
of the water extract of Inonotus [corrected] obliquus by induction
of apoptosis in melanoma B16-F10 cells. J Ethnopharmacol.
121:221–228. 2009.PubMed/NCBI
|
|
112.
|
Lavi I, Levinson D, Peri I, Tekoah Y,
Hadar Y and Schwartz B: Chemical characterization,
antiproliferative and antiadhesive properties of polysaccharides
extracted from Pleurotus pulmonarius mycelium and fruiting
bodies. Appl Microbiol Biotechnol. 85:1977–1990. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
113.
|
Di Luzio NR, Williams DL, McNamee RB,
Edwads BF and Kitahama A: Comparative tumor inhibitory and
anti-bacterial activity of soluble and particlate glucan. Int J
Cancer. 24:773–779. 1979.PubMed/NCBI
|
|
114.
|
Yoon TJ, Kim TJ, Lee H, Shin KS, Yun YP,
Moon WK, Kim DW and Lee KH: Anti-tumor metastatic activity of
beta-glucan purified from mutated Saccharomyces cerevisiae.
Int Immunopharmacol. 8:36–42. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
115.
|
Yamamoto K, Kimura T, Sugitachi A and
Matsuura N: Anti-angiogenic and anti-metastatic effects of
beta-1,3-D-glucan purified from Hanabiratake, Sparassis
crispa. Biol Pharm Bull. 32:259–263. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
116.
|
Ishihara Y, Fujii T, Iijima H, Saito K and
Matsunaga K: The role of neutrophils as cytotoxic cells in lung
metastasis: suppression of tumor cell metastasis by a biological
response modifier (PSK). In Vivo. 12:175–182. 1998.PubMed/NCBI
|
|
117.
|
Lee IS and Nishikawa A: Polyozellus
multiplex, a Korean wild mushroom, as a potent chemopreventive
agent against stomach cancer. Life Sci. 73:3225–3234. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
118.
|
Fujimiya Y, Suzuki Y, Oshiman K, Kobori H,
Moriguchi K, Nakashima H, Matumoto Y, Takahara S, Ebina T and
Katakura R: Selective tumoricidal effect of soluble proteoglucan
extracted from the basidiomycete, Agaricus blazei Murill,
mediated via natural killer cell activation and apoptosis. Cancer
Immunol Immunother. 46:147–159. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
119.
|
Chihara G, Hamuro J, Maeda Y, Arai Y and
Fukuoka F: Antitumor polysaccharide derived chemically from natural
glucan (pachyman). Nature. 225:943–944. 1970. View Article : Google Scholar : PubMed/NCBI
|
|
120.
|
Ohno N, Furukawa M, Miura NN, Adachi Y,
Motoi M and Yadomae T: Antitumor betaglucan from the cultured fruit
body of Agaricus blazei. Biol Pharm Bull. 24:820–828. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
121.
|
Sasaki T, Abiko N, Sugino Y and Nitta K:
Dependence on chain length of antitumor activity of
(1→3)-β-D-glucan from Alcaligenes faecalis var.
myxogenes, IFO 13140 and its acid-degraded products. Cancer
Res. 38:379–383. 1978.
|
|
122.
|
Iino K, Ohno N, Suzuki I, Miyazaki T and
Yadomae T: Structural characterization of a neutral antitumour
beta-D-glucan extracted with hot sodium-hydroxide from cultured
fruit bodies of Grifola Frondosa. Carbohyd Res. 141:111–119.
1985. View Article : Google Scholar
|
|
123.
|
Furusawa E, Chou SC, Furusawa S, Hirazum A
and Dang Y: Antitumor activity of Gonoderma lucidum and
edible mushroom, on interaperitoneally implanted Lewis lung
carcinoma in synergeneic mice. Phytother Res. 6:300–304. 1992.
|
|
124.
|
Gelderman KA, Tomlinson S, Ross GD and
Gorter A: Complement function in mAb-mediated cancer immunotherapy.
Trends Immunol. 25:158–164. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
125.
|
Akramiene D, Kondrotas A, Didziapetriene J
and Kevelaitis EP: Effects of beta-glucans on the immune system.
Medicina (Kaunas). 43:597–606. 2007.PubMed/NCBI
|
|
126.
|
Cheung NV and Modal S: Oral
(1–3),(1–4)-β-D-Glucan synergizes with antiganglioside GD2
monoclonal antibody 3F8 in the therapy of neuroblastoma. Clin
Cancer Res. 8:1217–1223. 2002.
|
|
127.
|
Sier CFM, Gelderman KA, Prins FA and
Gorter A: Beta-glucan enhanced killing of renal cell carcinoma
micrometastases by monoclonal antibody G250 directed complement
activation. Int J Cancer. 109:900–908. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
128.
|
Modak S, Koehne G, Vickers A, O'Reilly RJ
and Cheung NV: Rituximab therapy of lymphoma is enhanced by orally
administered (1–3),(1–4)-D-β-glucan. Leuk Res. 29:679–683.
2005.PubMed/NCBI
|
|
129.
|
Hong F, Yan J, Baran JT, Allendorf DJ,
Hansen RD, Ostroff GR, Xing PX, Cheung NK and Ross GD: Mechanism by
which orally administered beta-1,3-glucans enhance the tumoricidal
activity of antitumor monoclonal antibodies in murine tumor models.
J Immunol. 173:797–806. 2004. View Article : Google Scholar
|
|
130.
|
Liu J, Gunn L, Hansen R and Yan J:
Combined yeast-derived beta-glucan with anti-tumor monoclonal
antibody for cancer immunotherapy. Exp Mol Pathol. 86:208–214.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
131.
|
Huang H, Ostroff GR, Lee CK, Agarwal S,
Ram S, Rice PA, Specht CA and Levitz SM: Relative contributions of
dectin-1 and complement to immune responses to particulate
β-glucans. J Immunol. 189:312–317. 2012.PubMed/NCBI
|
|
132.
|
Xiang D, Sharma VR, Freter CE and Yan J:
Anti-tumor monoclonal antibodies in conjunction with β-glucans: a
novel anti-cancer immunotherapy. Curr Med Chem. 19:4298–4305.
2012.
|
|
133.
|
Ina K and Ando T: The use of Lentinan for
treating gastric cancer. Anticancer Agents Med Chem. Oct
18–2012.(Epub ahead of print).
|
|
134.
|
Kataoka H, Shimura T, Mizoshita T, Kubota
E, Mori Y, Mizushima T, Wada T, Ogasawara N, Tanida S, Sasaki M,
Togawa S, Sano H, Hirata Y, Ikai M, Mochizuki H, Seno K, Itoh S,
Kawai T and Joh T: Lentinan with S-1 and paclitaxel for gastric
cancer chemotherapy improve patient quality of life.
Hepatogastroenterology. 56:547–550. 2009.PubMed/NCBI
|
|
135.
|
Chen J, Zhang XD and Jiang Z: The
application of fungal beta-glucans for the treatment of colon
cancer. Anticancer Agents Med Chem. Dec 24–2012.(Epub ahead of
print).
|
|
136.
|
Hamuro J and Chihara G: Effect of
antitumour polysaccharides on the higher structure of serum
protein. Nature. 245:40–41. 1973. View Article : Google Scholar : PubMed/NCBI
|
|
137.
|
Soto ER, Caras AC, Kut LC, Castle MK and
Ostroff GR: Glucan particles for macrophage targeted delivery of
nanoparticles. J Drug Deliv. 2012:1435242012.PubMed/NCBI
|
|
138.
|
Huang H, Ostroff GR, Lee CK, Specht CA and
Levitz SM: Robust stimulation of humoral and cellular immune
responses following vaccination with antigen-loaded beta-glucan
particles. MBio. 1:e00164–102010.
|
|
139.
|
Lehtovaara BC and Gu FX: Pharmacological,
structural and drug delivery properties and applications of
1,3-β-glucans. J Agric Food Chem. 59:6813–6828. 2011.
|
|
140.
|
Browder W, Williams D, Lucore P, Pretus H,
Jones E and Mcnamee R: Effect of enhanced macrophage function on
early wound-healing. Surgery. 104:224–230. 1988.PubMed/NCBI
|
|
141.
|
Portera CA, Love EJ, Memore L, Zhang L,
Müller A, Browder W and Williams DL: Effect of macrophage
stimulation on collagen biosynthesis in the healing wound. Am Surg.
63:125–131. 1997.PubMed/NCBI
|