Introduction
Oral squamous cell carcinoma (OSCC) represents ∼1–3%
of all human cancers, and is the 6th most frequent cancer in the
world (1,2). OSCC continues to show a poor
prognosis and remains a lethal disease for >50% of cases
diagnosed annually (3). In
particular, its local recurrence is strongly related to a poor
prognosis (4–6). Even when the surgical margins are
diagnosed as tumor free by histopathological examination, the local
recurrence rate is still 6.9–22.0% (4,5,7,8).
There are two explanations for the mechanism of local recurrence
(9). First, there is the
possibility that residual cancer cells remain undetected in the
surgical margins. In the majority of these cases routine
histopathological investigation of the resection specimen will
detect tumor tissue at the resection margin, making it likely that
residual cancer is the cause of local recurrence. However, the
number of residual cancer cells is too small to be detected by
routine histopathology, a phenomenon also known as minimal residual
cancer (MRC) (9,10). Secondly, it is possible that
tumor-related mucosal precursor lesions, ‘fields’ of genetically
altered cells may be left behind, and these might give rise to new
invasive carcinomas. A subgroup of these fields can be recognized
clinically as leukoplakia or erythroplakia, but the majority can
only be diagnosed by conventional histology, immunohistochemical
staining, or genetic methods (9,11,12).
Recently, we showed that the deep surgical margin
was resected very close to the tumor site in many neoadjuvant
chemotherapy (NAC)-treated OSCC patients, suggesting that NAC may
lead to local recurrence and poor outcomes (6,13).
In these studies (6,13), therefore, we speculated that the
most invasive front became unclear after modification by NAC, which
may have resulted in resection with an insufficient safety margin.
Moreover, we estimated that not only the mechanism for the closed
margin but also the residual cancer cells after NAC were involved
in the mode of local recurrence. Most recently, some researchers
reported that these residual cancer cells after NAC possess the
characteristics of cancer stem cells (CSC) (14,15).
Yoshikawa et al (15)
showed that CD44-expressing cancer cells in the clinical samples of
human head and neck cancers selectively survive and increase in
number after NAC. CD44 is a single-pass type I transmembrane
glycoprotein and functions as a cellular adhesion molecule for
hyaluronic acid, a major component of the extracellular matrix
(16,17). Expression of CD44, especially
variant isoforms (CD44v6), is highly expressed in CSCs derived from
solid tumors, including brain (18), colon (19), pancreas (20) and head and neck (21) cancer. ATP-binding cassette,
subfamily G, member 2 (ABCG2), is a member of the ABC transporter
protein superfamily that produces multiple drug-resistant cancer.
It is also known as a molecular determinant for maintaining the
side population (SP) phenotype in stem cells (22). This phenotype has been observed in
OSCC (23,24). Increasing evidence therefore
suggests that ABCG2 is recognized as a CSC marker.
However, it is unclear whether NAC is associated
with the expression of CD44v6 or ABCG2, or whether the expression
of these CSC markers is clinically important in NAC-surviving cells
(MRC cells after NAC). We therefore investigated whether NAC is
associated with the expression of CD44v6 and ABCG2 using
immunohistochemical staining, and evaluated the association with
clinicopathological factors and the clinical outcome of oral tongue
squamous cell carcinoma (OTSCC) patients.
Materials and methods
Patients
We retrospectively reviewed the records of 89
patients who underwent radical surgery for OTSCC between January
2001 and December 2011. The study cohort included patients with
histologically confirmed diagnoses of OTSCC and a minimum follow-up
of 12 months. All study patients underwent extensive pretreatment
evaluations, including blood chemistry, complete blood cell count,
chest X-ray, computed tomography (CT) and/or magnetic resonance
imaging (MRI) of the head and neck area, and thoracoabdominal CT,
and provided informed consent to participate in the study. Data
collected included demographic information, site, TNM stage at
diagnosis, tumor histologic grade, pattern of tumor invasion,
treatment, depth of tumor invasion, status of the margin,
histopathological response after NAC, recurrence, and the adjuvant
therapy used for disease control.
Tumor stage was classified according to the TNM
classification of the International Union against Cancer. Tumor
histologic grade was defined according to the WHO classification.
Pattern of invasion (POI) was examined at the host/tumor interface.
POI types 1–4 have been previously defined by Bryne et al
(25): POI type 1, tumor invasion
in a broad pushing manner with a smooth outline. POI type 2, tumor
invasion with broad pushing ‘fingers’ or separate large tumor
islands, with a stellate appearance. POI type 3, invasive islands
of tumor >15 cells per island. POI type 4, invasive tumor
islands <15 cells per island. This includes cord-like and single
cell invasion. The evaluation was performed using biopsy
specimens.
Treatment and pathologic examination
In our institute, the modality of surgery alone was
preferred for the treatment of patients with oral cancer; however,
patients who hesitated to consent to surgical intervention or for
whom surgery was not available because of a busy schedule were
selected for NAC. During the period of NAC, patients were
encouraged to undergo surgery after completion of chemotherapy. All
patients who received NAC underwent radical surgery. The regimen of
NAC consisted of a combination of cisplatin or carboplatin-based
multidrugs. In most patients, modifications of chemotherapy dosages
were made for toxicity as indicated using standard criteria.
Primary tumors were excised with ≥1.5-cm safety
margins (both superficial and deep margins). The extent of surgery
was not modified according to the tumor response with NAC. All
patients underwent glossectomy with curative intent. Elective neck
dissection was not performed routinely in our institutions. Local
recurrence was defined as lesions arising in the oral cavity
relative to the primary tumor beyond 6 weeks within the first 5
years after the first definitive treatment. Recurrence arising at
both the primary site and in the neck was also considered to be
local recurrence. Recurrence arising only in the neck was defined
as regional recurrence. Recurrence arising either at the primary
site or in the neck was defined as locoregional recurrence.
Patients with recurrences that were considered potentially curable
and operable underwent salvage surgery and radiotherapy. Patients
with recurrences that were considered incurable were treated with
palliative chemotherapy.
For pathological examination of the invasion depth
and surgical margin, all permanent section histological slides of
primary resection specimens were reviewed retrospectively. Depth of
invasion (DOI) was measured as the infiltrative portion of the
tumor which extended below the surface of the adjacent mucosa. The
keratin layer was not included in the measurements. Where the
epithelium was destroyed (since the majority of the tumor showed
ulceration), it was measured after reconstructing a virtual
surface. Previous studies demonstrated that DOI ≥4 mm had a
predictive value for cervical lymph node metastasis in patients
with OTSCC (26–30); therefore, in this study, DOI was
classified as ≥4 and <4 mm. Likewise, regarding the surgical
margin, a large cohort study demonstrated that a pathological
margin distance ≤4 mm was significantly associated with
locoregional recurrence (31);
therefore, in this study, the status of the surgical margin was
classified as >4 and ≤4 mm for the superficial and deep margins,
respectively. The degree of histopathological response after NAC
was classified into four categories, based on the method in
previous studies (32,33): grade I, >50% vital residual
tumor cells (VRTC); grade II, 10–50% VRTC; grade III, nearly
complete response (NCR) with <10% VRTC; and grade IV, complete
response. All pathological assessments were performed by expert
pathologists who were unaware of the clinical outcomes.
Immunohistochemistry (IHC)
Immunohistochemical staining was performed on
4-μm-thick formalin-fixed, paraffin-embedded tissue sections.
Deparaffinized sections in xylene were soaked in 10 mM citrate
buffer (pH 6.0) and placed in an autoclave at 121°C for 5°min for
antigen retrieval. Endogenous peroxidase was blocked using 0.3%
H2O2 in methanol for 30 min.
Immunohistochemistry was performed by the EnVision method
(EnVision+; Dako, Glostrup, Denmark). The primary
antibodies used were directed against CD44v6 (VFF-18; Abcam,
Cambridge, UK; 1:100 dilution) and ABCG2 (BXP-21; Abcam; 1:50
dilution). The sections were incubated with the antibodies
overnight at 4°C. Reaction products were visualized by immersing
the sections in diaminobenzidine (DAB) solution, and the samples
were counterstained with Myer’s hematoxylin and mounted.
The membranous expression of CD44v6 or ABCG2 was
estimated by immunopositivity mainly in the tumor invasive front
closed surgical margin. The average number of stained cells in 5
visual fields was regarded as the percent ratio of positively
stained cells in each section. Staining with these antibodies was
considered positive if >10% of tumor cells stained, because a
10% cutoff level has been used in several previous studies
(34,35).
Statistical analysis
Statistical analyses were performed using StatMate
IV (Atms Co., Tokyo, Japan). The categorical data were assessed by
the χ2 test or Fisher’s exact test, as appropriate. The
disease-specific survival (DSS) rate was calculated using the
Kaplan-Meier method. Significance was evaluated using the log-rank
test. A multiple regression study was performed using Cox
proportional hazard analysis. Univariate logistic and multivariate
logistic regression analyses were used to identify independent
predictive factors for local recurrence. Predictors that were not
associated with DSS or local recurrence in univariate analysis were
not included in multivariate analysis. P<0.05 was considered
significant.
Results
Patient characteristics
Patient demographic characteristics are summarized
in Table I. The male-to-female
ratio was 1.28 with 50 male subjects. The mean age at diagnosis was
63.4 years (range, 28–88 years). Twenty-two (24.7%) patients
received NAC. NAC regimens were CDDP (CDDP 10 mg/m2,
days 1–5; 2 courses with 2-week intervals), CDDP/S-1 (S-1 40
mg/m2 twice a day orally for 21 days, CDDP 60
mg/m2 on day 8; 2 courses with 2-week interval periods),
or CBDCA/5-FU (CBDCA 300 mg/m2 on day 1, 5-FU 600 mg on
days 1–5; 2 courses with 3-week interval periods). In NAC-treated
patients, 9 patients (40.9%) had stage III or IV disease, and 13
patients (59.1%) had stage I or II (Table II). Toxicity to NAC was acceptable
(CTCAE v. 4.0 grade 1/2) and related to nausea, mucositis and
hematologic toxicity.
 | Table I.Demographic characteristics of 89
patients with oral tongue squamous cell carcinoma. |
Table I.
Demographic characteristics of 89
patients with oral tongue squamous cell carcinoma.
|
Characteristics | No. of cases
(%) |
|---|
| Gender | |
| Male | 50 (56.2) |
| Female | 39 (43.8) |
| Age | |
| ≥64 | 48 (53.9) |
| ≤63 | 41 (46.1) |
| pT stage | |
| T1 | 39 (43.8) |
| T2 | 43 (48.4) |
| T3 | 5 (5.6) |
| T4 | 2 (2.2) |
| pN stage | |
| N0 | 63 (70.8) |
| N1 | 12 (13.5) |
| N2 | 14 (15.7) |
| N3 | 0 (0) |
| Histological
grade | |
| Well | 82 (92.1) |
| Moderately | 5 (5.6) |
| Poorly | 2 (2.3) |
| POI | |
| 1 | 6 (6.7) |
| 2 | 25 (28.2) |
| 3 | 40 (44.9) |
| 4 | 18 (20.2) |
| Treatment | |
| Surgery
alone | 67 (75.3) |
| NAC +
surgery | 22 (24.7) |
| DOI | |
| <4 mm | 55 (61.8) |
| ≥4 mm | 34 (38.2) |
| Surgical
margin | |
| >4 mm | 68 (76.4) |
| ≤4 mm | 21 (23.6) |
| Perineural
invasion | |
| No | 65 (73.0) |
| Yes | 24 (27.0) |
| Local
recurrence | |
| No | 78 (87.6) |
| Yes | 11 (12.4) |
 | Table II.Neoadjuvant chemotherapy regimen and
disease stage of oral tongue squamous cell carcinoma. |
Table II.
Neoadjuvant chemotherapy regimen and
disease stage of oral tongue squamous cell carcinoma.
| Treatment | Stage I | Stage II | Stage III | Stage IV | Total |
|---|
| Surgery alone | 35 | 23 | 4 | 4 | 67 |
| NAC + surgery | 2 | 11 | 5 | 4 | 22 |
| NAC regimen | | | | | |
| CDDP | 1 | 1 | 0 | 0 | 2 |
| CDDP/S-1 | 0 | 7 | 1 | 1 | 9 |
| CBDCA/5-FU | 1 | 3 | 4 | 3 | 11 |
Local recurrence developed in 11 patients (12.4%)
during the follow-up period. The mean follow-up period was 44.7
months for the whole series (range, 3–125 months).
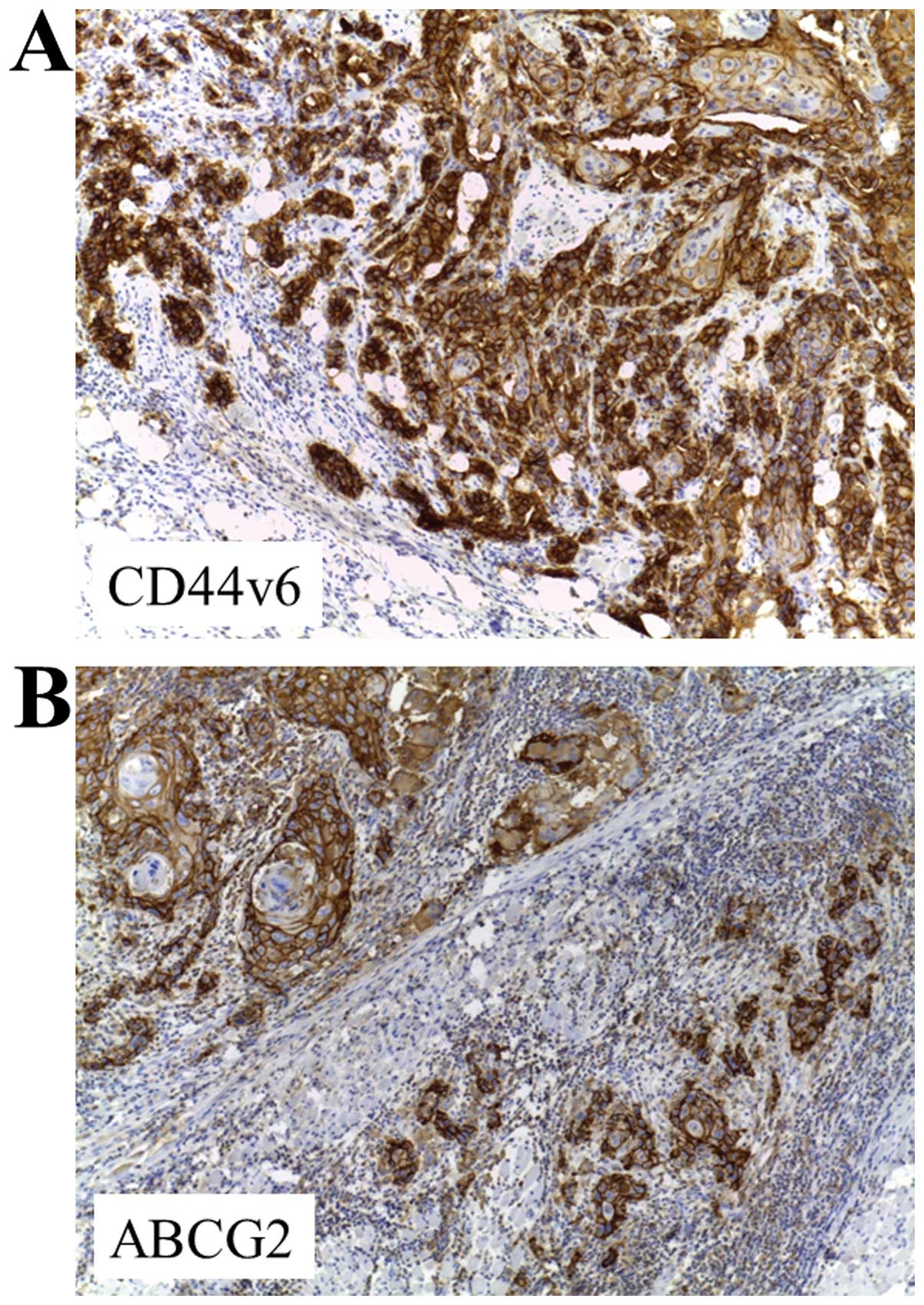
CD44v6 and ABCG2 expression in OTSCC by
IHC
The positive rates of CD44v6 and ABCG2 in 89 OTSCC
patients detected by immunohistochemical staining were 24.7 and
13.5%, respectively. Staining for CD44v6 and ABCG2 showed a mixed
membranous and cytoplasmic pattern of staining in OTSCC cells
(Fig. 1). Both markers were mainly
characterized by higher invasive front localization.
Association of CD44v6 and ABCG2
expression with clinicopathological factors and survival
CD44v6 expression was significantly associated with
lymph node metastasis, POI, NAC, DOI, perineural invasion, and
local recurrence. Meanwhile, ABCG2 was significantly associated
with lymph node metastasis, POI, DOI, perineural invasion and local
recurrence. Univariate analysis using the log-rank test revealed
that T-stage (P=0.010), lymph node metastasis (P=0.003), tumor
differentiation (P=0.011), POI (P<0.001), NAC (P<0.001), DOI
(P=0.002), perineural invasion (P<0.001) and local recurrence
(P<0.001) were significantly associated with 5-year DSS
(Table III).
 | Table III.Association of CD44v6 and ABCG2
expression with clinicopathological factors and survival. |
Table III.
Association of CD44v6 and ABCG2
expression with clinicopathological factors and survival.
|
Characteristics | n | CD44v6 | P-value | ABCG2 | P-value | 5-year DSS (%) | P-value |
|---|
|
|
|---|
| + | − | + | − |
|---|
| Gender | | | | 0.417 | | | 0.305 | | 0.422 |
| Male | 50 | 14 | 36 | | 9 | 41 | | 84.3 | |
| Female | 39 | 8 | 31 | | 4 | 35 | | 86.2 | |
| Age | | | | 0.122 | | | 0.231 | | 0.222 |
| ≥64 | 48 | 15 | 33 | | 9 | 39 | | 83.2 | |
| ≤63 | 41 | 7 | 34 | | 4 | 37 | | 88.7 | |
| pT | | | | 0.505 | | | 0.980 | | 0.010 |
| T1+T2 | 82 | 21 | 61 | | 12 | 70 | | 90.0 | |
| T3+T4 | 7 | 1 | 6 | | 1 | 6 | | 47.6 | |
| pN | | | | <0.001 | | | <0.001 | | 0.003 |
| N0 | 63 | 8 | 55 | | 4 | 59 | | 94.5 | |
| N1+N2+N3 | 26 | 14 | 12 | | 9 | 17 | | 63.9 | |
|
Differentiation | | | | 0.806 | | | 0.276 | | 0.011 |
| Well | 82 | 20 | 62 | | 11 | 71 | | 88.4 | |
| Moderate +
poor | 7 | 2 | 5 | | 2 | 5 | | 57.1 | |
| POI | | | | <0.001 | | | <0.001 | | <0.001 |
| 1+2+3 | 71 | 7 | 64 | | 5 | 66 | | 89.4 | |
| 4 | 18 | 15 | 3 | | 8 | 10 | | 69.1 | |
| Treatment | | | | 0.042 | | | 0.214 | | <0.001 |
| Surgery
alone | 67 | 13 | 54 | | 8 | 59 | | 96.0 | |
| NAC +
surgery | 22 | 9 | 13 | | 5 | 17 | | 63.3 | |
| DOI | | | | <0.001 | | | <0.001 | | 0.002 |
| <4 mm | 55 | 4 | 51 | | 2 | 53 | | 98.0 | |
| ≥4 mm | 34 | 18 | 16 | | 11 | 23 | | 70.2 | |
| Surgical
margin | | | | 0.640 | | | 0.510 | | 0.077 |
| >4 mm | 68 | 16 | 52 | | 9 | 59 | | 86.2 | |
| ≤4 mm | 21 | 6 | 15 | | 4 | 17 | | 81.0 | |
| Perineural
invasion | | | | <0.001 | | | 0.018 | | <0.001 |
| No | 65 | 10 | 56 | | 6 | 59 | | 96.7 | |
| Yes | 24 | 12 | 12 | | 7 | 17 | | 63.5 | |
| Local
recurrence | | | | <0.001 | | | 0.002 | | <0.001 |
| No | 78 | 14 | 64 | | 8 | 70 | | 88.2 | |
| Yes | 11 | 8 | 3 | | 5 | 6 | | 47.7 | |
Univariate analysis using the log-rank test and
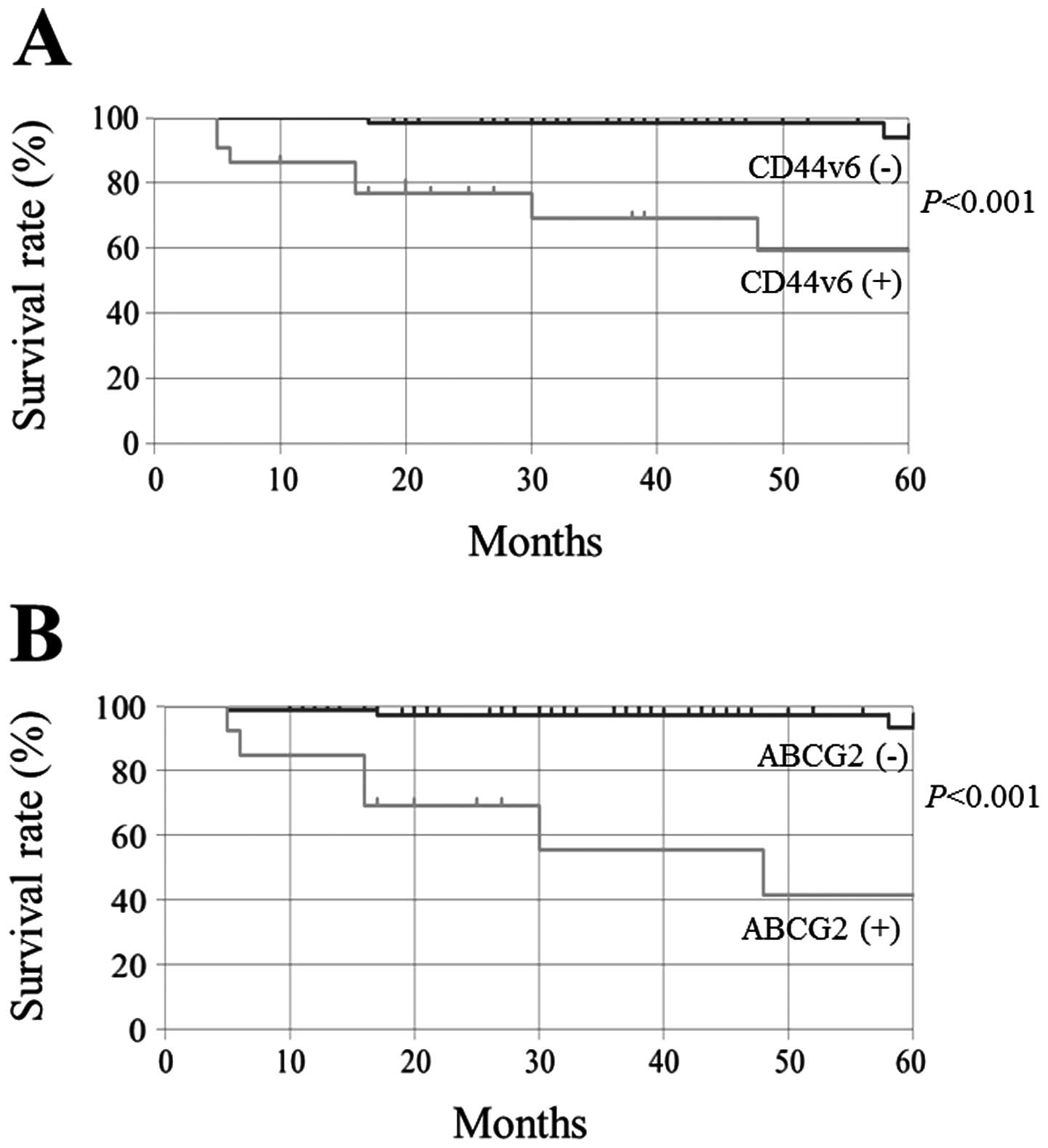
Kaplan-Meier method showed that CD44v6 and ABCG2 expression was
significantly associated with 5-year DSS (Fig. 2, P<0.001, respectively).
Predictors that were associated with 5-year DSS in univariate
analysis were included in the Cox proportional hazard model, and
this multivariate analysis showed that local recurrence was only
one independent predictor of 5-year DSS in OTSCC patients. The odds
ratio of local recurrence was 17.54 (95% confidence interval,
0.36–5.37; P=0.025).
Association with local recurrence
Univariate logistic analysis showed a significant
association of tumor local recurrence with POI (P<0.001), NAC
(P=0.038), DOI (P=0.029), surgical margin (P=0.003), CD44v6
(P<0.001) and ABCG2 (P=0.008) (Table IV). Multivariate logistic
regression was performed for each predictor of the local recurrence
and revealed that only the surgical margin (odds ratio, 10.58; 95%
confidence interval, 1.75–63.89; P=0.010) was an independent factor
for local recurrence.
 | Table IV.Clinicopathological factors and
CD44v6 and ABCG2 expression in relation to local recurrence. |
Table IV.
Clinicopathological factors and
CD44v6 and ABCG2 expression in relation to local recurrence.
|
Characteristics | n | Local
recurrence | Odds ratio | 95% CI | P-value |
|---|
|
|---|
| + (n=11) (%) | − (n=78) |
|---|
| Gender | | | | | 0.17–2.17 | 0.659 |
| Male | 50 | 5 (10.0) | 45 | 0.61 | | |
| Female | 39 | 6 (15.4) | 33 | 1.00 | | |
| Age | | | | | 0.29–3.65 | 0.780 |
| ≥64 | 48 | 6 (12.5) | 42 | 1.03 | | |
| ≤63 | 41 | 5 (12.2) | 36 | 1.00 | | |
| pT | | | | | 0.13–11.02 | 0.662 |
| T1+T2 | 82 | 10 (12.2) | 72 | 1.00 | | |
| T3+T4 | 7 | 1 (14.3) | 6 | 1.20 | | |
| pN | | | | | 0.22–6.89 | 0.833 |
| N0 | 63 | 7 (11.1) | 56 | 1.00 | | |
| N1+N2+N3 | 26 | 4 (15.4) | 22 | 1.24 | | |
|
Differentiation | | | | | 0.55–19.24 | 0.448 |
| Well | 82 | 9 (11.0) | 73 | 1.00 | | |
| Moderate +
poor | 7 | 2 (28.6) | 5 | 3.24 | | |
| POI | | | | | 2.67–42.5 | <0.001 |
| 1+2+3 | 71 | 4 5.6) | 67 | 1.00 | | |
| 4 | 18 | 7 (38.9) | 11 | 10.66 | | |
| Treatment | | | | | 1.26–17.20 | 0.038 |
| Surgery
alone | 67 | 5 (9.1) | 62 | 1.00 | | |
| NAC +
surgery | 22 | 6 (21.7) | 16 | 4.65 | | |
| DOI | | | | | 1.30–21.80 | 0.029 |
| <4 mm | 55 | 3 (5.5) | 52 | 1.00 | | |
| ≥4 mm | 34 | 8 (21.1) | 26 | 5.33 | | |
| Surgical
margin | | | | | 2.06–31.10 | 0.003 |
| >4 mm | 68 | 4 (5.9) | 64 | 1.00 | | |
| ≤4 mm | 21 | 7 (33.3) | 14 | 8.00 | | |
| Perineural
invasion | | | | | 0.71–9.44 | 0.267 |
| No | 65 | 6 (9.2) | 59 | 1.00 | | |
| Yes | 24 | 5 (20.8) | 19 | 2.59 | | |
| CD44v6 | | | | | 2.87–51.84 | <0.001 |
| Positive | 22 | 8 (36.4) | 14 | 12.19 | | |
| Negative | 67 | 3 (4.5) | 64 | 1.00 | | |
| ABCG2 | | | | | 1.81–29.40 | 0.008 |
| Positive | 13 | 5 (38.5) | 8 | 7.29 | | |
| Negative | 76 | 6 (7.9) | 70 | 1.00 | | |
Association of NAC with cancer stem cell
markers (CD44v6 and ABCG2)
As mentioned above, the positive rate of CD44v6 in
NAC-treated patients was significantly higher than that in surgery
alone-treated patients. Likewise, the local recurrence rate in
NAC-treated patients was significantly higher than that in surgery
alone-treated patients.
Therefore, to examine the effect of NAC on local
recurrence, the relationship between cancer stem cell markers
(CD44v6 and ABCG2) and local recurrence was analyzed by subgroup
assay. There was a significant correlation between local recurrence
with CD44v6 or ABCG2 positivity in the NAC-treated group but not
the surgery alone-treated group (Table
V). Then, the correlation of the degree of histopathological
response after NAC with local recurrence or these cancer stem cell
marker expressions was not significant (Table VI).
 | Table V.Relationship between CD44v6, ABCG2
and local recurrence by subgroup (surgery alone and NAC) assay. |
Table V.
Relationship between CD44v6, ABCG2
and local recurrence by subgroup (surgery alone and NAC) assay.
| Factors | n | Surgery
alone-treated group | P-value | NAC-treated
group | P-value |
|---|
|
|
|---|
| (n=67) Local
recurrence | (n=22) Local
recurrence |
|---|
|
|
|---|
| + | − | + | − |
|---|
| CD44v6 | | | | 0.275 | | | <0.001 |
| Positive | 22 | 2 | 12 | | 6 | 2 | |
| Negative | 67 | 3 | 50 | | 0 | 14 | |
| ABCG2 | | | | 0.563 | | | 0.003 |
| Positive | 13 | 1 | 7 | | 4 | 1 | |
| Negative | 76 | 4 | 55 | | 2 | 15 | |
 | Table VI.Correlation of the histopathological
response after NAC with local recurrence or cancer stem cell marker
expressions. |
Table VI.
Correlation of the histopathological
response after NAC with local recurrence or cancer stem cell marker
expressions.
| Factors | n | Histopathological
response | P-value |
|---|
|
|---|
| I | II | III | IV |
|---|
| Local
recurrence | | | | | | 0.422 |
| + | 6 | 1 | 3 | 2 | 0 | |
| − | 16 | 1 | 14 | 1 | 0 | |
| CD44v6 | | | | | | 0.133 |
| Positive | 8 | 2 | 4 | 2 | 0 | |
| Negative | 14 | 0 | 13 | 1 | 0 | |
| ABCG2 | | | | | | 0.816 |
| Positive | 5 | 1 | 3 | 1 | 0 | |
| Negative | 17 | 1 | 14 | 2 | 0 | |
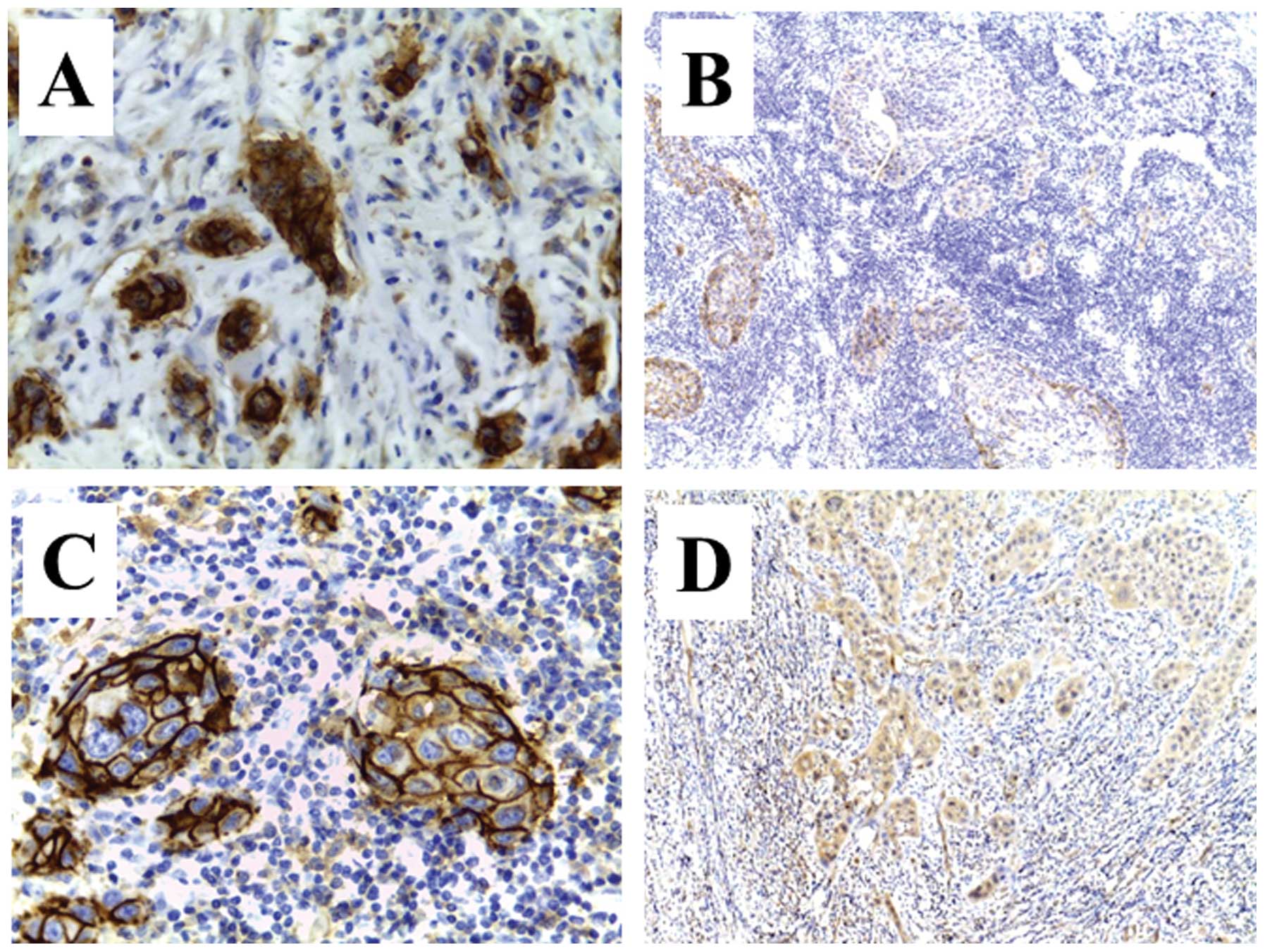
Immunopositivity of CD44v6 or ABCG2 showed
significantly high intensity in NAC-treated patients compared with
surgery alone-treated patients. In particular, the immunopositivity
of these cancer stem cell markers was apparent at the tumor/host
interface in NAC-treated patients with local recurrence. In
contrast, immunopositivity in NAC-treated patients without local
recurrence revealed pale or faint staining (Fig. 3).
Discussion
Locoregional recurrence in patients with OSCC
remains a problem and can affect their survival (4–6). In
particular, local recurrence is a high potential risk factor for
survival (4,6). Correspondingly, in our study, tumor
local recurrence was only one predictive factor of the 5-year DSS
rate by multivariate analysis. Previous studies reported that local
recurrence occurred in 6.9–22.0% (4,5,7,8),
similar to the 12.4% rate in the present series. Several studies
have demonstrated that demographic variables such as site, TNM
stage, and simultaneous disorder, and pathological variables such
as DOI, surgical margin, and POI could be reliable parameters for
local recurrence (4–7). In our study, POI, NAC, DOI, surgical
margin, and perineural invasion were significantly associated with
local recurrence, and in particular, the surgical margin (≤4 mm)
was a predictive factor of local recurrence. Based on data similar
to these results, we previously proposed that the most invasive
front became unclear after modification by NAC, which may have
resulted in resection with an insufficient safety margin (6,13).
It was also hypothesized in the mechanism of tumor local recurrence
that this elevated risk may be due to undetected tumor cells
present in surgical margins thought to be negative by conventional
light microscopy (9–11). Recently, to improve the detection
of residual cancer cells or tumor-related precursor lesions
remaining in histologically negative margins, molecular margin
analysis was proposed and has now been used to observe and predict
local recurrence of head and neck cancers (9,12,36).
We therefore focused on the expression of molecules relevant to
CSCs in tumor satellites at the tumor/host interface.
The presence of CSCs in the primary tumor appears to
have prognostic significance in various types of cancer including
OSCC (24,37,38).
Since the concept of CSC has been proposed to explain tumor cell
heterogeneity, some research has suggested that current therapies
fail to prevent cancer relapse and metastasis because of a small,
surviving population of CSC (37–41).
We previously demonstrated that the side population cells isolated
from an oral cell line possessed the characteristics of cancer stem
cells, chemoresistance, and high proliferation ability (24). Moreover, in another study, we
estimated that not only the mechanism for the closed margin but
also residual cancer cells exhibiting the characteristic of CSC
after NAC are involved in the mode of local recurrence (13). We focused on the expressions of
CD44v6 and ABCG2 as CSC markers in OTSCC, because we hypothesized
that residual cancer cells exhibiting the characteristics of CSC
would be relevant to the mechanism of local recurrence in
NAC-treated OTSCC patients.
CD44 is a cell surface glycoprotein known to be a
hyaluronate receptor, which may function in lymphocyte homing and
cancer metastasis (16,17). CD44 is encoded by a single gene
which contains 20 exons and is located on the short arm of
chromosome 11 (11p13) (16). Exons
1–5 and 16–19 are spliced together to form the transcript for the
standard isoform of CD44 (CD44s), which is expressed in a wide
range of normal tissues as well as in tumors of ectodermal origin
(16). Alternative splicing of
exons 6–15 (variant exons 1–10) gives rise to numerous variant
forms of CD44 (CD44v), in which an additional segment encoded by
one or more of the variant exons is inserted in the extracellular
domain of CD44s, which is encoded by exons 1–5 and exons 16–20
(16,42,43).
The variant 6 isoform (CD44v6), in particular, is associated with
several cancer types but not in somatic cells (15,18–21).
Moreover, CD44v6 is widely known as a marker of CSCs (18,21,36,44–46).
Recently, Zhao et al reported that immunohistochemical
assessment of CD44v6 in a negative surgical margin would be a
valuable approach for predicting recurrence and survival after
curative surgery for laryngeal cancer (36). Some authors showed that CD44v6
could be associated with cancer progression, metastasis, and
recurrence (36,46). Our data indicate that CD44v6
expression is associated with regional lymph node metastasis, POI,
NAC, DOI, perineural invasion, and local recurrence in OTSCC.
CD44v6-positive patients revealed a significant poor survival rate
compared with CD44v6-negative patients. Furthermore, CD44v6
expression was a predictive factor for local recurrence (odds
ratio, 12.19), as well as diffuse POI, NAC modality, deep (≥4 mm)
DOI, closed (≤4 mm) surgical margin in univariate logistic
analysis. These results support the idea that CD44v6-positive
cancer cells exhibiting the characteristics of CSC take part in the
mechanism of local recurrence, and CD44v6 is a useful marker of
tumor progression in OTSCC.
ABCG2 has been shown to be a major mediator of the
SP phenotype, which refers to a transmembrane transporter that
carries out the important biological function of the efflux of
multiple endogenous and exogenous substances out of cells (22,23).
Elevated expression of ABCG2 has been observed in a number of
putative CSCs from various cancers (24,34,35).
AGCG2 is a well-known marker useful for identifying and isolating
CSCs (22–24,34,35,47).
In our study, ABCG2 expression was 14.6% in OTSCC patients, similar
to the 12.4% in esophageal squamous cell carcinoma (35). Our data indicate that ABCG2
expression is associated with regional lymph node metastasis, POI,
DOI, perineural invasion, and local recurrence in OTSCC.
ABCG2-positive patients revealed a significant poor survival rate
compared with ABCG2-negative patients. Furthermore, ABCG2
expression was a predictive factor for local recurrence (odds
ratio, 7.29) in univariate logistic analysis. As well as CD44v6,
these results support the idea that ABCG2 is relevant to the
mechanism of local recurrence in OTSCC.
Previously, we reported that the surgical margins
were resected very close to the site in many NAC-treated cases,
suggesting that NAC would lead to local recurrence and a poor
outcome in OSCC (6,13). Correspondingly, in this current
study, we demonstrated that NAC was a significant predictive factor
for local recurrence. NAC, which is the use of systemic
chemotherapy before definitive surgery and/or radiotherapy, is an
attractive approach in the management of HNSCC. The benefits of NAC
for patients with locally advanced head and neck cancer, as
demonstrated by several clinical studies, include a reduction in
distant metastasis and the preservation of organ function (48–51);
however, NAC has failed to demonstrate any significant improvement
of survival in several randomized control trials (49,50).
The efficacy of NAC is controversial, and no report supports NAC
with a high evidence level.
Interestingly, in this study, the positive rate of
CD44v6 in NAC-treated cases was significantly higher than that in
surgery alone-treated cases. Moreover, there was significant
correlation between local recurrence and CD44v6 positivity in the
NAC-treated group. On the other hand, this correlation in the
surgery-alone group was not significant. These results indicate the
possibility that the upregulation of CD44v6 expression may play an
important role in local recurrence in patients with OTSCC who are
receiving NAC. Several studies have reported that NAC could
significantly modify the expression of CSC markers such as CD44v6,
CD133, Nanog, Oct4, or EpCAM in various types of cancers (15,52–55).
In rectal cancer, some authors demonstrated that CD133 expression
in residual cancer cells after chemoradiotherapy was significantly
higher than matched pre-chemoradiotherapy CD133 expression in
biopsy specimens (53–55). In HNSCC, Yoshikawa et al
reported that the relative area occupied by CD44v-expressing cells
was significantly larger in the tumors of patients who received NAC
than in those of patients who did not, suggesting that
CD44-positive HNSCC cells are resistant to chemotherapy compared
with CD44v-negative cells (15).
Thus, we conclude that CD44v6 could be induced by NAC, and
CD44v6-expressing residual cancer cells exhibiting the
characteristics of CSC could be relevant to the mechanism of local
recurrence in NAC-treated OTSCC patients. On the other hand, in
this study, there was no significant difference in the positive
rate of ABCG2 between NAC-treated cases and surgery alone-treated
cases. In subgroup analysis, however, there was a significant
correlation between local recurrence and ABCG2 positivity in the
NAC-treated group, but not in the surgery alone-treated group.
These results suggest that ABCG2-expressing cells are resistant to
NAC, thereby increasing the fraction of ABCG2-expressing cells so
that ABCG2-expressing residual cancer cells would be relevant to
the mechanism of local recurrence in NAC-treated OTSCC patients.
Some studies have also shown that patients whose disease responded
to NAC had better local control and survival than those who
received ineffective NAC (48,51).
In our study, however, the relationship between the histological
response after NAC and local recurrence or CSC marker expression
was unclear.
Taken together, the expression of CSC markers
(CD44v6 and ABCG2) was correlated with lymph node metastasis, tumor
progression, and local recurrence, thereby decreasing survival. We
conclude that the expression of these CSC markers plays an
important role in local recurrence in patients with OTSCC who are
receiving NAC. Therefore, we propose that NAC leads to the
selection and/or residue of more aggressive CSCs. Accordingly, at
present, we cannot recommend the modality of NAC for patients with
OTSCC. Detection of the CD44v6 and ABCG2 status in the surgical
margin may contribute to the identification of patients with high
risk factors of local recurrence and should be addressed in future
studies.
References
|
1.
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global Cancer Statistics 2011. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2.
|
Warnakulasuriya S: Global epidemiology of
oral and oropharyngeal cancer. Oral Oncol. 45:309–316. 2009.
View Article : Google Scholar
|
|
3.
|
Rogers SN, Brown JS, Woolgar JA, et al:
Survival following primary surgery for oral cancer. Oral Oncol.
45:201–211. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Brandwein-Gensler M, Teixeira MS, Lewis
CM, et al: Oral squamous cell carcinoma. Histologic risk
assessment, but not margin status, is strongly predictive of local
disease-free and overall survival. Am J Surg Pathol. 29:167–178.
2005.
|
|
5.
|
Kernohan MD, Clark JR, Gao K, Ebrahimi A
and Milross CG: Predicting the prognosis of oral squamous cell
carcinoma after first recurrence. Arch Otolaryngol Head Neck Surg.
136:1235–1239. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Yanamoto S, Yamada S, Takahashi H, et al:
Clinicopathological risk factors for local recurrence in oral
squamous cell carcinoma. Int J Oral Maxillofac Surg. 41:1195–1200.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Liao CT, Chang JTC, Wang HM, et al:
Analysis of risk factors of predictive local tumor control in oral
cavity cancer. Ann Surg Oncol. 15:915–922. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Ord RA, Kolokythas A and Reynolds MA:
Surgical salvage for local and regional recurrence in oral cancer.
J Oral Maxillofac Surg. 64:1409–1414. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Braakhuis BJM, Bloemena E, Leemans CR and
Brakenhoff RH: Molecular analysis of surgical margins in head and
neck cancer: more than a marginal issue. Oral Oncol. 46:485–491.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Partridge M, Li SR, Pateromichelakis S, et
al: Detection of minimal residual cancer to investigate why oral
tumors recur despiteseemingly adequate treatment. Clin Cancer Res.
6:2718–2725. 2000.PubMed/NCBI
|
|
11.
|
van Houten VMM, Leemans CR, Kummer JA, et
al: Molecular diagnosis of surgical margins and local recurrence in
head and neck cancer patients: a prospective study. Clin Cancer
Res. 10:3614–3620. 2004.PubMed/NCBI
|
|
12.
|
Bilde A, von Buchwald C, Dabelsteen E,
Therkildsen MH and Dabelsteen S: Molecular markers in the surgical
margin of oral carcinomas. J Oral Pathol Med. 38:72–78. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Yanamoto S, Yamada S, Takahashi H, et al:
Predictors of locoregional recurrence in T1-2N0 tongue cancer
patients. Pathol Oncol Res. 19:795–803. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Kim JJ and Tannock IF: Repopulation of
cancer cells during therapy: an important cause of treatment
failure. Nat Rev Cancer. 5:516–525. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Yoshikawa M, Tsuchihashi K, Ishimoto T, et
al: xCT-expressing tumor cells that are resistant to EGFR-targeted
therapy in head and neck squamous cell carcinoma. Cancer Res.
73:1855–1866. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Naor D, Sionov RV and Ish-Shalom D: CD44:
structure, function, and association with the malignant process.
Adv Cancer Res. 71:241–319. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Nagano O, Okazaki S and Saya H: Redox
regulation in stem-like cancer cells by CD44 variant isoforms.
Oncogene. 32:5191–5198. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Jijiwa M, Demir H, Gupta S, et al: CD44v6
regulates growth of brain tumor stem cells partially through the
AKT-mediated pathway. PLoS One. 6:e242172011. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Lee HJ, Eom DW, Kang GH, et al: Colorectal
micropapillary carcinomas are associated with poor prognosis and
enriched in markers of stem cells. Mod Pathol. 26:1123–1131. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Gaviraghi M, Tunici P, Valensin S, et al:
Pancreatic cancer spheres are more than just aggregates of stem
marker-positive cells. Biosci Rep. 31:45–55. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Mack B and Gires O: CD44s and CD44v6
expression in head and neck epithelia. PLoS One. 3:e33602008.
View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Zhou S, Schuetz JD, Bunting KD, et al: The
ABC transporter Bcrp1/ABCG2 is expressed in a wide variety of stem
cells and is a molecular determinant of the side-population
phenotype. Nat Med. 67:1028–1034. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Zhang P, Zhang Y, Mao L, Zhang Z and Chen
W: Side population in oral squamous cell carcinoma possesses tumor
stem cell phenotypes. Cancer Lett. 277:227–234. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Yanamoto S, Kawasaki G, Yamada S, et al:
Isolation and characterization of cancer stem-like side population
cells in human oral cancer cells. Oral Oncol. 47:855–860. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Bryne M, Koppang HS, Lilleng R and
Kjaerheim A: Malignancy grading of the deep invasive margins of
oral squamous cell carcinoma has high prognostic value. J Pathol.
166:375–381. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Asakage T, Yokose T, Mukai K, et al: Tumor
thickness predicts cervical metastasis in patients with stage I/II
carcinoma of the tongue. Cancer. 82:1443–1448. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Kurokawa H, Yamashita Y, Takeda S, Zhang
M, Fukuyama H and Takahashi T: Risk factors for late cervical lymph
node metastases in patients with stage I or II carcinoma of the
tongue. Head Neck. 24:731–736. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Lim SC, Zhang S, Ishii G, et al:
Predictive markers for late cervical metastasis in stage I and II
invasive squamous cell carcinoma of the oral tongue. Clin Cancer
Res. 10:166–172. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Sparano A, Weinstein G, Chalian A, Yodul M
and Weber R: Multivariate predictors of occult neck metastasis in
early oral tongue cancer. Otolaryngol Head Neck Surg. 131:472–476.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Huang SH, Hwang D, Lockwood G, Goldstein
DP and O’Sullivan B: Predictive value of tumor thickness for
cervical lymph-node involvement in squamous cell carcinoma of the
oral cavity. A meta-analysis of reported studies Cancer.
115:1489–1497. 2009.PubMed/NCBI
|
|
31.
|
Fan KH, Wang HM, Kang CJ, et al: Treatment
results of postoperative radiotherapy on squamous cell carcinoma of
the oral cavity: coexistence of multiple minor risk factors results
in higher recurrence rates. Int J Radiat Oncol Biol Phys.
77:1024–1029. 2010. View Article : Google Scholar
|
|
32.
|
Schneider PM, Baldus SE, Metzger R, et al:
Histomorphologic tumor regression and lymph node metastases
determine prognosis following neoadjuvant radiochemotherapy for
esophageal cancer. Ann Surg. 242:684–692. 2005. View Article : Google Scholar
|
|
33.
|
Bollschweiler E, Besch S, Drebber U, et
al: Influence of neoadjuvant chemoradiation on the number and size
of analyzed lymph nodes in esophageal cancer. Ann Surg Oncol.
17:3187–3194. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Ota S, Ishii G, Goto K, et al:
Immunohistochemical expression of BCRP and ERCC1 in biopsy specimen
predicts survival in advanced non-small-cell lung cancer treated
with cisplatin-based chemotherapy. Lung Cancer. 64:98–104. 2009.
View Article : Google Scholar
|
|
35.
|
Hang D, Dong HC, Ning T, Dong B, Hou DL
and Xu WG: Prognostic value of the stem cell markers CD133 and
ABCG2 expression in esophageal squamous cell carcinoma. Dis
Esophagus. 25:638–644. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Zhao H, Ren J, Zhuo X, Ye H, Zou J and Liu
S: Prognostic significance of surviving and CD44v6 in laryngeal
cancer surgical margins. J Cancer Res Clin Oncol. 134:1051–1058.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Dalerba P, Cho RW and Clarke MF: Cancer
stem cells: models and concepts. Annu Rev Med. 58:267–284. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Visvader JE and Lindeman GJ: Cancer stem
cells in solid tumours: accumulating evidence and unresolved
questions. Nat Rev Cancer. 8:755–768. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39.
|
Graziano A, d’Aquino R, Tirino V,
Desiderio V, Rossi A and Pirozzi G: The stem cell hypothesis in
head and neck cancer. J Cell Biochem. 103:408–412. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40.
|
Prince ME and Ailles LE: Cancer stem cells
in head and neck squamous cell cancer. J Clin Oncol. 26:2871–2875.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
41.
|
Sinha N, Mukhopadhyay S, Das DN, Panda PK
and Bhutia SK: Relevance of cancer initiating/stem cells in
carcinogenesis and therapy resistance in oral cancer. Oral Oncol.
49:854–862. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42.
|
Screaton GR, Bell MV, Jackson DG, Cornelis
FB, Gerth U and Bell JI: Genomic structure of DNA encoding the
lymphocyte homing receptor CD44 reveals at least 12 alternatively
spliced exons. Proc Natl Acad Sci USA. 89:12160–12164. 1992.
View Article : Google Scholar
|
|
43.
|
Goodison S, Urquidi V and Tarin D: CD44
cell adhesion molecules. Mol Pathol. 52:189–196. 1999. View Article : Google Scholar
|
|
44.
|
Wang SJ, Wong G, de Heer AM, Xia W and
Bourguignon LY: CD44 variant isoforms in head and neck squamous
cell carcinoma progression. Laryngoscope. 119:1518–1530. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
45.
|
Yang YM and Chang JW: Bladder cancer
initiating cells (BCICs) are among (initiating) cells. Cancer
Invest. 26:725–733. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46.
|
Shi J, Zhou Z, Di W and Li N: Correlation
of CD44v6 expression with ovarian cancer progression and
recurrence. BMC Cancer. 13:1822013. View Article : Google Scholar : PubMed/NCBI
|
|
47.
|
Shen B, Dong P, Li D and Gao S: Expression
and function of ABCG2 in head and neck squamous cell carcinoma and
cell lines. Exp Ther Med. 2:1151–1157. 2011.PubMed/NCBI
|
|
48.
|
Lore JM, Diaz-Ordaz E, Spaulding M, et al:
Improved survival with preoperative chemotherapy followed by tumor
response for advanced squamous cell carcinoma of the head and neck.
Am J Surg. 170:506–511. 1995. View Article : Google Scholar
|
|
49.
|
Pignon JP, Bourhis J, Domenge C and
Designe L: Chemotherapy added to locoregional treatment for head
and neck squamous cell carcinoma: three meta-analyses of updated
individual data. Lancet. 355:949–955. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
50.
|
Licitra L, Grandi C, Guzzo M, et al:
Primary chemotherapy in resectable oral cavity squamous cell
carcinoma: a randomized controlled trial. J Clin Oncol. 21:327–333.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
51.
|
Olasz L, Szalma J, Orsi E, Tornoczky T,
Marko T and Nyarady Z: Neoadjuvant chemotherapy: does it have
benefits for surgeon in the treatment of advanced squamous cell
cancer of the oral cavity? Pathol Oncol Res. 16:207–212. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
52.
|
Costa S, Terzano P, Bovicelli A, et al:
CD44 isoform 6 (CD44v6) is a prognostic indicator of the response
to neoadjuvant chemo-therapy in cervical carcinoma. Gynecol Oncol.
80:67–73. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
53.
|
Saigusa S, Tanaka K, Toiyama Y, et al:
Clinical significance of CD133 and hypoxia inducible factor-1α gene
expression in rectal cancer after preoperative chemotherapy. Clin
Oncol. 23:323–332. 2011.PubMed/NCBI
|
|
54.
|
Kawamoto A, Tanaka K, Saigusa S, et al:
Clinical significance of radiation-induced CD133 expression in
residual rectal cancer cells after chemoradiotherapy. Exp Ther Med.
3:403–409. 2012.
|
|
55.
|
Sprenger T, Conradi L, Beissbarth T, et
al: Enrichment of CD133-expressing cells in rectal cancers treated
with preoperative radiochemotherapy is an independent marker for
metastasis and survival. Cancer. 119:26–35. 2013. View Article : Google Scholar
|