Introduction
According to the most recent GLOBOCAN estimates,
cervical cancer is one of the most common cancers among women
worldwide and accounts for 529,800 new cases annually (1). Moreover, it is the fourth most common
cause of cancer death in women worldwide (275,100 deaths in 2008)
(1). In response, the World Health
Organization has recommended the global prioritization of
prevention and control measures for cervical and breast cancers.
Carcinogenesis of the cervix almost exclusively involves infection
caused by human papilloma viruses (HPV) such as HPV-16 or HPV-18
(2). Thus, the incidence of
cervical cancer can be reduced to a considerable extent worldwide
by vaccination against HPV. Despite such efforts, women in Japan
continue to succumb to invasive cervical cancer.
Radical treatment strategies against cervical cancer
primarily include surgery, chemotherapy, and radiotherapy. For
advanced cervical cancer, concurrent radiotherapy and chemotherapy
are commonly administered. However, the 5-year survival rate
remains at only ~60%, indicating the need for significant
improvements in clinical outcomes (3).
The concept of cancer stem cells (CSCs) implies that
not all cells forming a tumor have the same ability to
differentiate or self-renew in order to sustain proliferation
(4,5). Because CSCs reportedly exhibit both
radio- and chemoresistance, residual or recurrent cancer after
radio- or chemotherapy may indicate treatment-induced selection of
CSCs (6). Therefore, the
development of a strategy to effectively eradicate CSCs can
theoretically improve patient outcomes. The following have been
identified as markers of CSCs in cervical cancer: the transcription
factor Nanog, the cell cycle-regulating protein nucleostemin, the
neural RNA-binding protein Musashi-1, the glycoprotein cluster of
differentiation 133, cell surface antigens
CD44+/CK17+, and the enzyme aldehyde
dehydrogenase 1 (7–10). The currently available method to
detect these markers is limited to fluorescence-activated cell
sorting (FACS); however, it is difficult to employ FACS for
real-time monitoring of CSCs both in vitro and in
vivo. A recent study reported the development of a
visualization system to evaluate CSCs in breast cancer and gliomas
using the green fluorescent protein ZsGreen fused to the degron
motif of ornithine decarboxylase (ODC), which is decomposed not by
ubiquitin but by the 26S proteasome (11). According to recent reports, 26S
proteasome activity in CSCs is lower than that in non-CSCs;
therefore, it is possible to observe differences in the
accumulation of ZsGreen in cancer cells. In this study, we
investigated optical imaging parameters to evaluate CSCs using two
human cervical cancer cell lines: CaSki and HeLa.
Materials and methods
Cell lines and culture conditions
The human cervical cancer cell lines CaSki and HeLa
were purchased from the Japanese Collection of Research
Bioresources Cell Bank (Ibaraki, Japan) and were cultured in
Dulbecco’s modified Eagle’s medium (DMEM; Sigma-Aldrich, St. Louis,
MO, USA) supplemented with 10% fetal bovine serum (FBS; Hyclone,
Logan, UT, USA) and penicillin-streptomycin (Sigma-Aldrich) in an
atmosphere of 5% CO2 at 37°C.
The retroviral vector pQCXIN-ZsGreen-cODC encodes
the ZsGreen-cODC fluorescent fusion protein (12). The Platinum-A Retroviral Packaging
Cell Line (Plat-A), optimized to produce high retrovirus titers,
was purchased from Cell Biolabs (San Diego, CA, USA). Plat-A cells
were cultured in DMEM supplemented with 10% FBS, 100 U/ml
penicillin and streptomycin each (Life Technologies, Gaithersburg,
MD, USA), 1 μg/ml of puromycin (Sigma-Aldrich), and 10 μg/ml of
blasticidin (Sigma-Aldrich). The retroviral vector was transfected
into Plat-A cells using the FuGENE6 transfection reagent (Promega
Corp., Madison, WI, USA) to generate retroviruses. The medium was
changed 1 day after transfection, and the supernatant including the
retroviruses was collected 2 days after transfection. To induce
cancer cell formation, virus-containing supernatant and 6 mg/ml of
polybrene (Sigma-Aldrich) were added to DMEM containing the
cultured cancer cells. Cells with high ZsGreen-cODC (high-Zs)
expression levels were selected after two rounds of FACS.
FACS analysis
HeLa and CaSki cells were washed using
phosphate-buffered saline (PBS), trypsinized with 0.25%
trypsin-ethylenediaminetetraacetic acid (EDTA; Life Technologies),
and subjected to FACS analysis using the BD FACSAria II cell sorter
system (Becton-Dickinson, Franklin Lakes, NJ, USA).
Time-lapse analysis
Cancer cells were seeded into 35-mm dishes
containing 3 ml of DMEM. After incubation for 1 day, fluorescence
was observed under the BioStudio fluorescent microscope equipped
with a high-quality charge-coupled device camera (Nikon Engineering
Co., Yokohama, Kanagawa, Japan).
Proteasome assay
The 26S proteasome exhibits three primary catalytic
functions: caspase-like, chymotrypsin-like, and trypsin-like. Each
catalytic activity was measured using the Proteasome-Glo™
Cell-Based Assay kit (Promega Corp.). Briefly, cancer cells (5,000
cells/well) were trypsinized using StemPro Accutase (Life
Technologies) and then seeded into a white-walled 96-well plate
containing 100 μl/well of DMEM; subsequently, 100 μl/well of
Proteasome-Glo™ Cell-based reagents (caspase-, chymotrypsin- or
trypsin-like) were added to the wells. The plate was covered with
aluminum foil and placed on a plate shaker at 700 rpm for 2 min,
followed by incubation at room temperature for 10 min. The
luminescence of each well was measured using a
GloMax®-Multi+ Microplate Multimode reader (Promega
Corp.).
Sphere-forming assay
Cancer cells were trypsinized using StemPro Accutase
in order to prepare single-cell suspensions. Two hundred cancer
cells per well were seeded into ultra-low-attachment 96-well plates
containing 200 μl of the DMEM/F12 medium (Sigma-Aldrich)
supplemented with 20 ng/ml of EGF (Life Technologies) and 20 ng/ml
of basic fibroblast growth factor (Peprotech, Offenbach, Germany).
After 3 weeks, sphere numbers were counted under a microscope.
Differentiation capacity
Cancer cells of high-Zs populations and
differentiated cells (i.e., the differentiated population) isolated
using FACS were incubated for ~1 month and then subjected to
several rounds of FACS analysis.
Clonogenic survival assay
Cancer cells were trypsinized using 0.25%
trypsin-EDTA, and appropriate numbers of cancer cells were seeded
into 6- or 10-cm dishes, followed by irradiation with 0, 2, 4, or 8
Gy. After 2 weeks, the number of colonies stained with crystal
violet was counted.
Tetrazolium dye (MTT) cell proliferation
assay
The MTT cell proliferation assay was performed using
the Cell Counting Kit-8 (Dojindo, Kumamoto, Japan). Briefly, after
trypsinization with 0.25% trypsin-EDTA, 3,000 cancer cells/well
were seeded into 96-well plates containing 100 μl of DMEM
supplemented with 10% FBS and penicillin-streptomycin. After
incubation for 24 h, the cancer cell populations were exposed to
various concentrations of cisplatin and paclitaxel. After 3 days,
10 μl/well of Cell Counting Kit-8 solution was added to each well
of the 96-well plate. After 2 h, the absorbance of each well was
measured using a microplate absorbance reader (Bio-Rad, Hercules,
CA, USA).
Tumorigenicity assay
To assess tumorigenicity, we purchased 5-week-old
female non-obese diabetic/severe combined immunodeficiency mice
from Oriental Yeast Co., Ltd. (Itabashi, Tokyo, Japan). The animal
research protocol was approved by the Animal Experiments Committee
of Osaka University (Suita, Osaka, Japan). Cells with high and low
fusion protein expression, sorted by FACS, were suspended in PBS
and Corning Matrigel Basement Membrane Matrix (Corning Incorporated
Life Sciences, Tewksbury, MA, USA) at a 1:1 ratio, and then
103, 104 or 105 cells were
subcutaneously injected into the mice.
Statistical analysis
Including the MTT assay, the Student’s t-test was
used for all statistical analysis. For the MTT assay, statistical
analysis was performed using two-way analysis of variance (ANOVA).
A p-value of <0.05 was considered statistically significant.
Results
Visualization and FACS analysis of
CSC-like cell populations
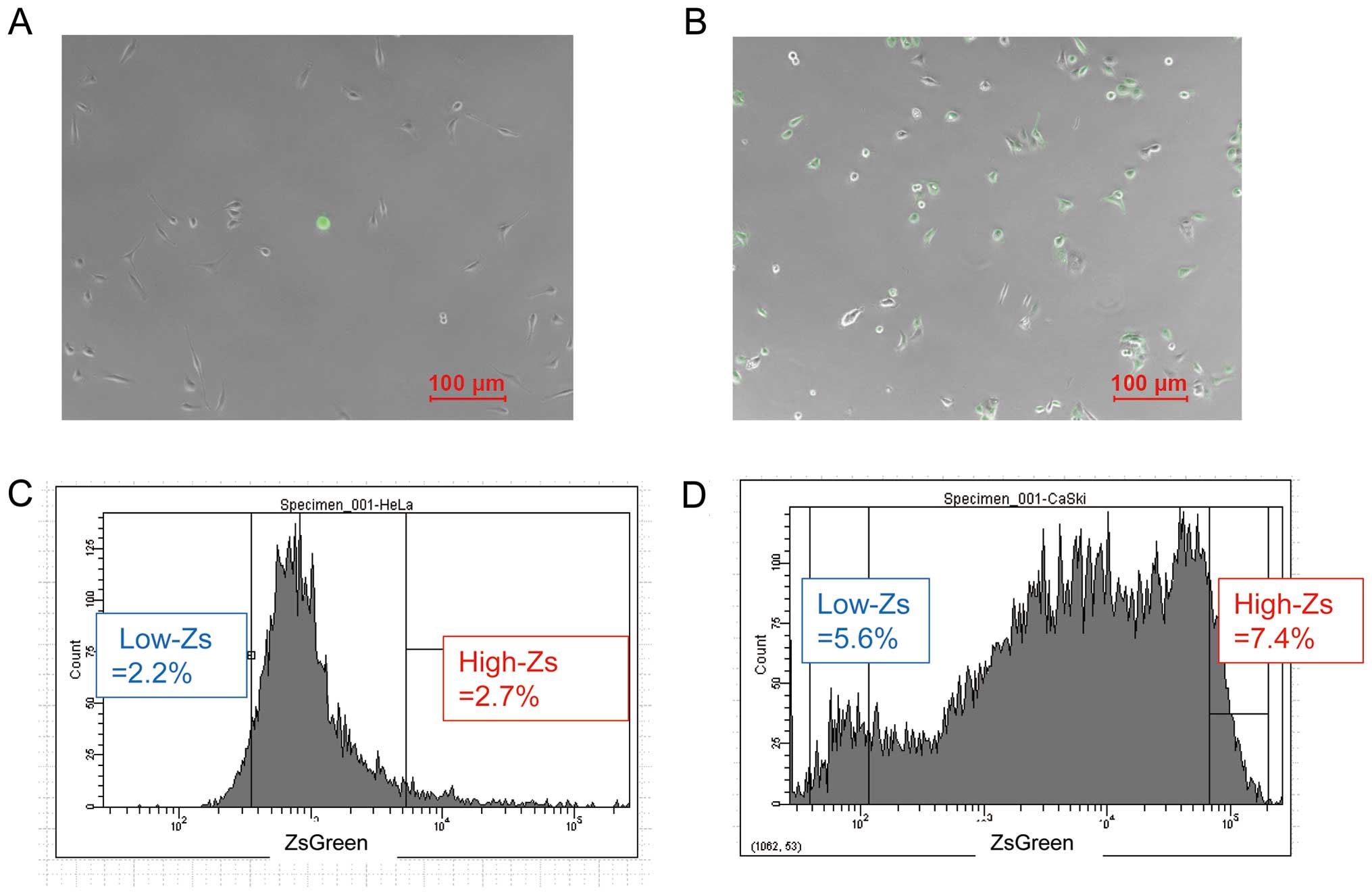
Cells with high-Zs expression levels were
established as shown in Fig. 1A and
B. The distributions of ZsGreen-cODC expression in cell
populations sorted by FACS are shown in Fig. 1C and D. Most CaSki cells were
fluorescent, whereas most HeLa cells were not. We defined
populations as cell types having high-Zs and low ZsGreen-cODC
(low-Zs) expression levels (Fig. 1C
and D).
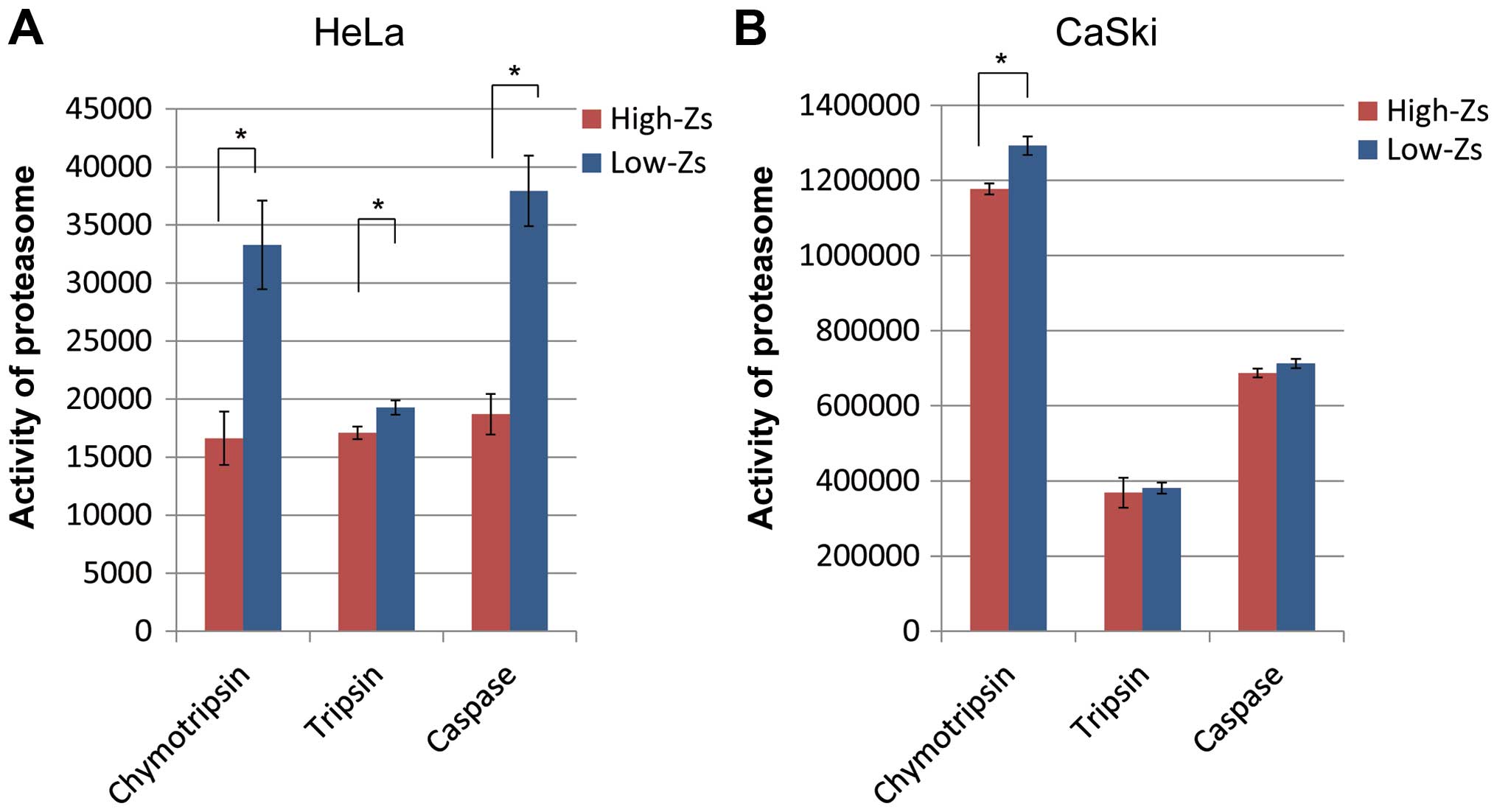
26S proteasome activities
The activity of the 26S proteasome, which decomposes
the cODC degron motif, was confirmed. The chymotrypsin-, trypsin-,
and caspase-like activities of the 26S proteasome in HeLa and CaSki
cells (populations with high- and low-Zs expression levels,
respectively) sorted by FACS are shown in Fig. 2A and B, respectively. In HeLa
cells, proteasome activities of cell populations with high-Zs
expression levels were significantly lower than those with low-Zs
expression levels. With regard to CaSki cells, only
chymotrypsin-like activity in the high-Zs population was
significantly lower than that of the low-Zs population (p<0.05),
and both trypsin- and caspase-like activities tended to be lower
than those of the low-Zs population. These results showed
comparatively low 26S proteasome activities in both HeLa and CaSki
cells.
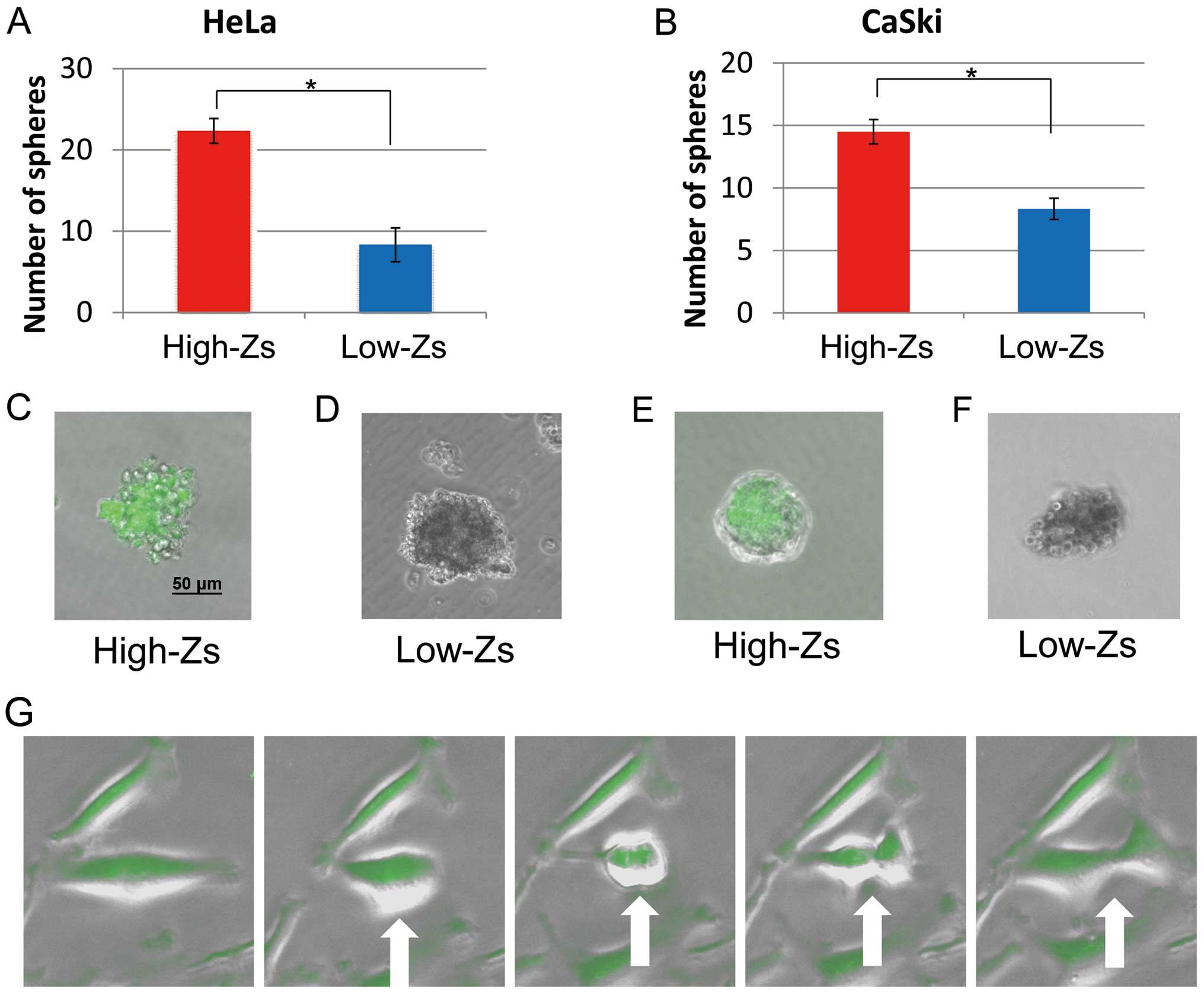
Self-renewal ability
To assess the self-renewal ability of different
populations of CSCs, a sphere-forming assay was performed using
high- and low-Zs populations sorted by FACS. Both populations
formed spheres, the numbers of which are shown in Fig. 3A and B, respectively. The number of
spheres formed by high-Zs populations of both HeLa and CaSki cells
was significantly higher than that formed by low-Zs populations.
Spheres formed by high-Zs populations of both HeLa and CaSki cells
exhibited fluorescence (Fig. 3C and
E, respectively), whereas those formed by low-Zs populations
did not (Fig. 3D and F,
respectively). The imaging of high-Zs populations supports the
concept that CSCs give rise to additional CSCs (Fig. 3G). Furthermore, these findings
indicate that, compared with low-Zs populations, high-Zs
populations of both HeLa and CaSki cells have higher ability for
self-renewal, which accords with the proposed characteristics of
CSC populations.
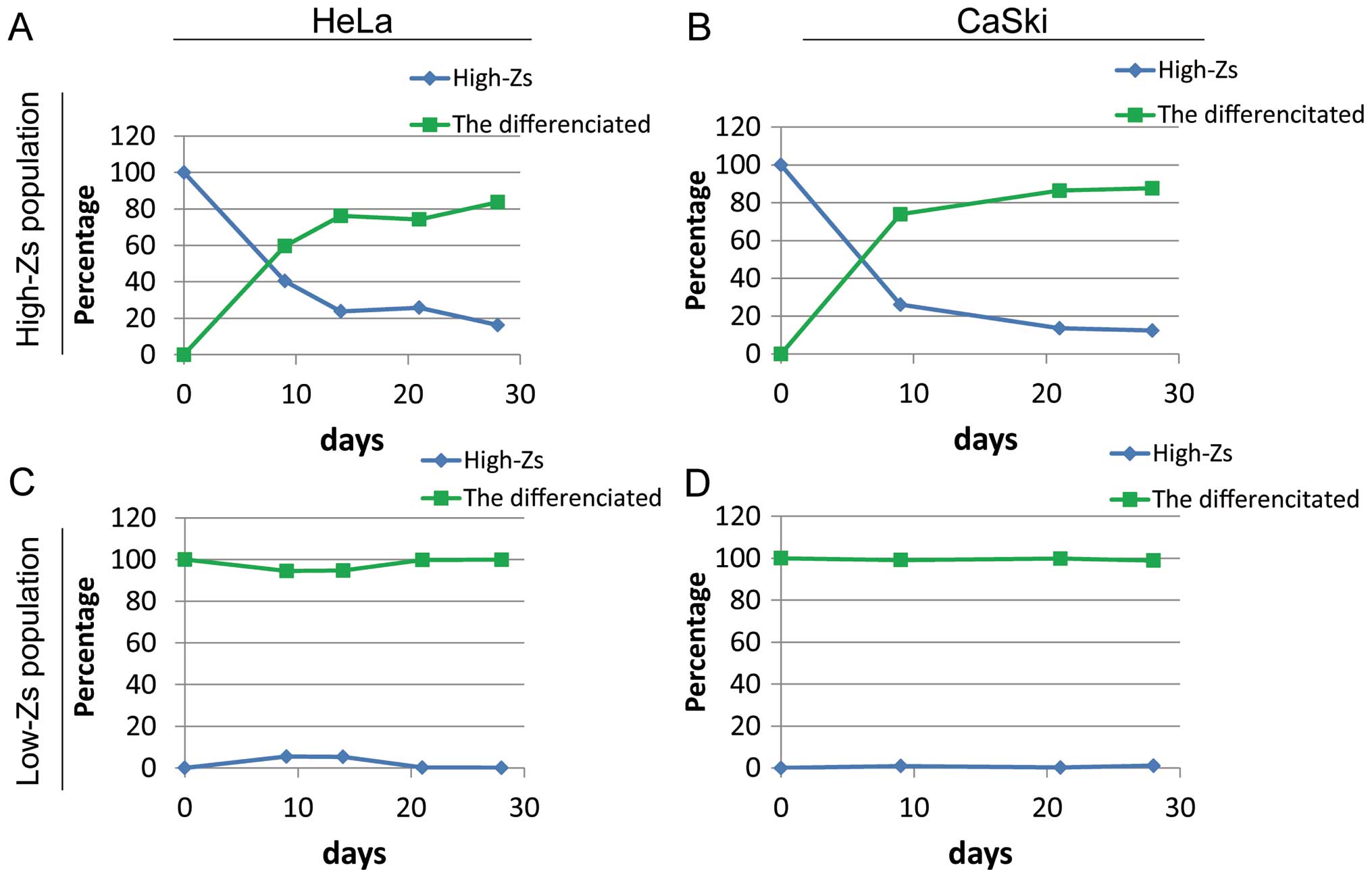
Differentiation capacity
To identify the differentiation capacity of CSCs, we
analyzed whether differentiated cells (the differentiated
population) were generated from high- or low-Zs populations of HeLa
and CaSki cells sorted by FACS. The ratios of high-Zs populations
decreased with time, but the ratios of the differentiated
population increased (Fig. 4A and
B). However, in low-Zs populations, the ratios of high-Zs and
the differentiated populations remained unchanged (Fig. 4C and D). Taken together, these
results indicate that only high-Zs populations of both HeLa and
CaSki cells have the capacity to generate differentiated
populations i.e., high-Zs populations were capable of asymmetrical
division.
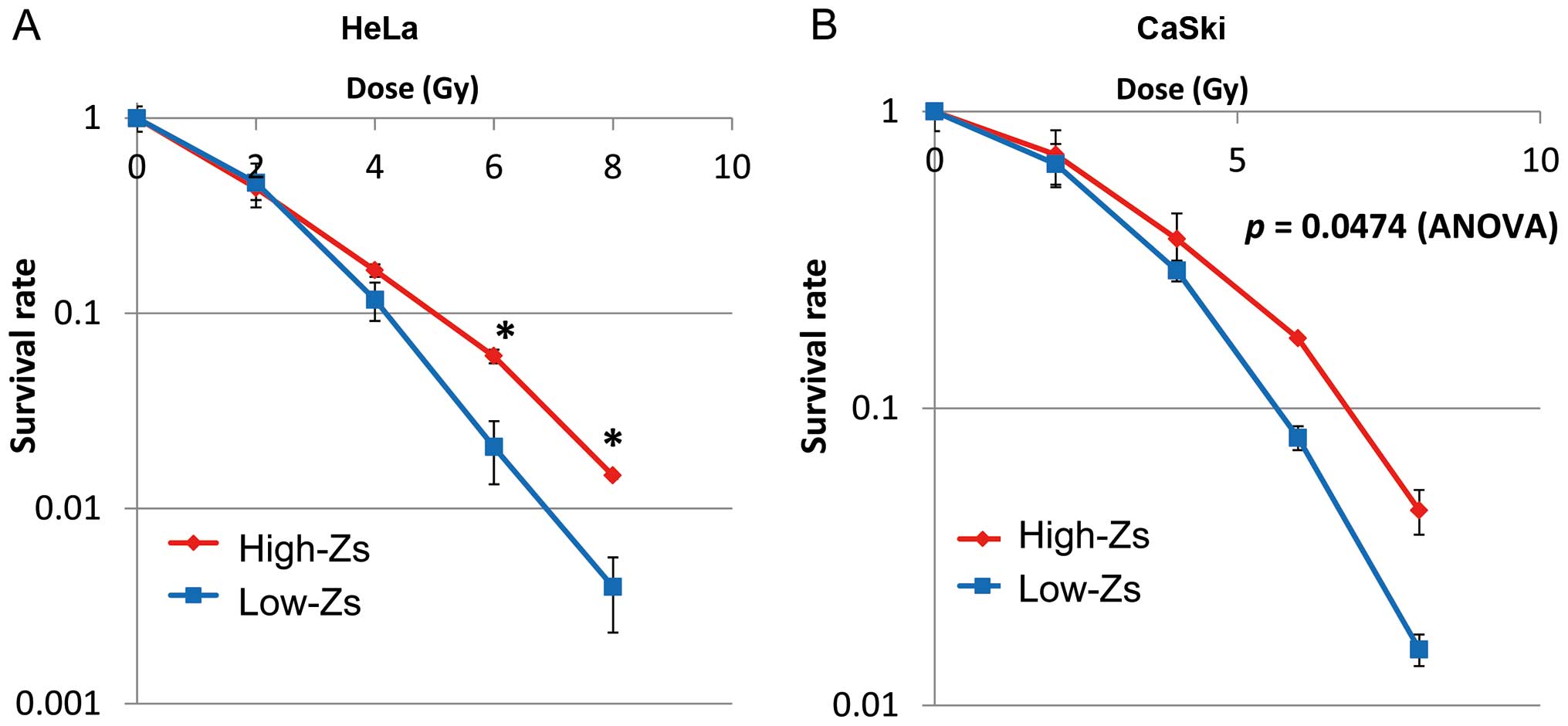
Radiosensitivity
We investigated the radiosensitivity characteristics
of CSCs by a clonogenic survival assay. Briefly, high- and low-Zs
populations of HeLa and CaSki cells were irradiated and sorted by
FACS. The survival rate of the high-Zs population of CaSki cells
was significantly greater than that of the low-Zs population using
two-way ANOVA (Fig. 5). There was
no significant difference between high-Zs populations of both HeLa
and CaSki cells by two-way ANOVA. However, the high-Zs population
of HeLa cells exhibited significantly greater radiosensitivity than
the low-Zs population, particularly at high doses, as indicated by
Student’s t-test (p<0.001). These results confirmed the
radioresistance of the high-Zs population of both HeLa and CaSki
cells.
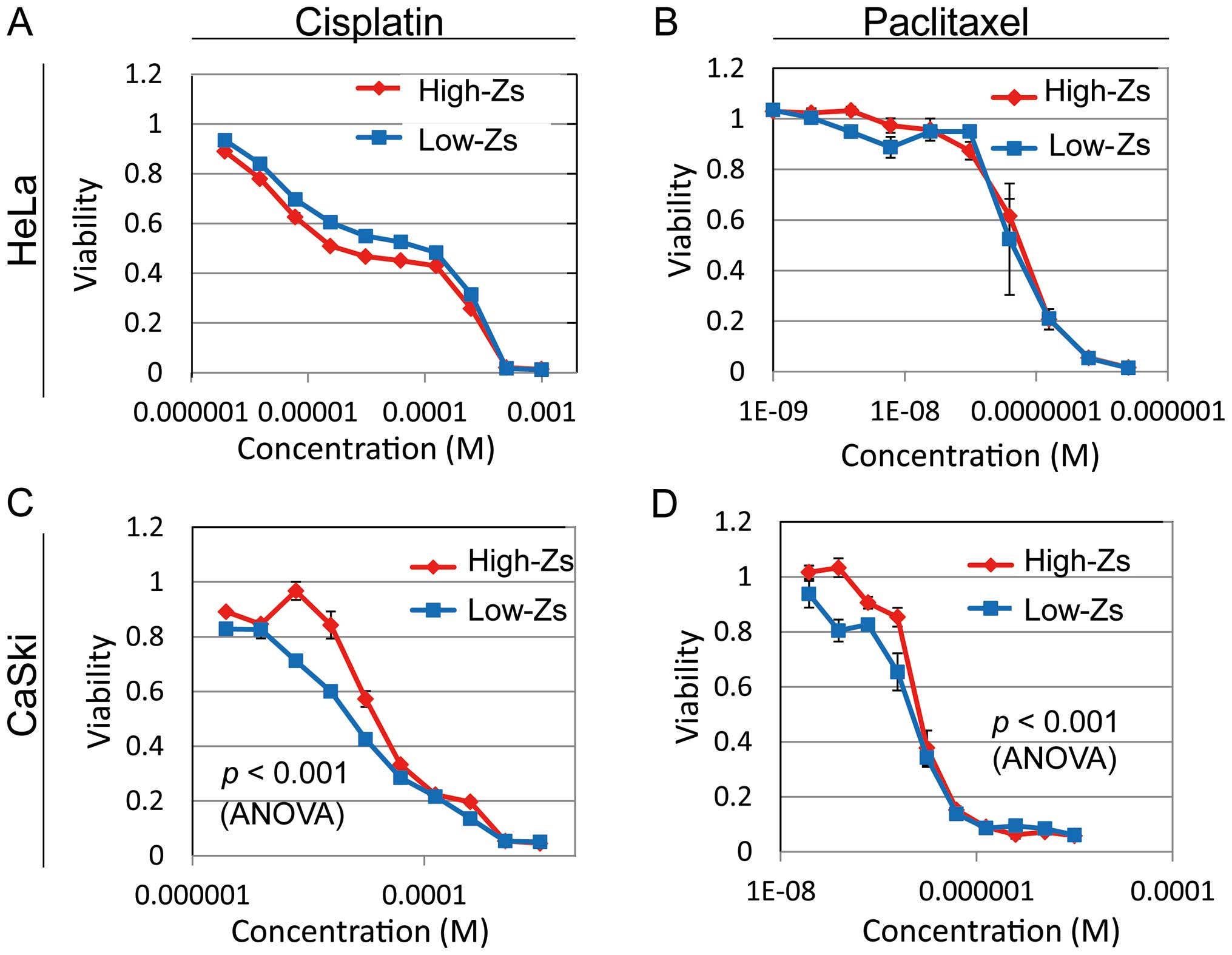
Drug sensitivity
Drug resistance, an important defining
characteristic of CSCs, was analyzed by the MTT assay. We selected
cisplatin and paclitaxel for the assay because each is an
established clinical agent for the treatment of advanced cervical
cancer. We analyzed the drug sensitivity of high- and low-Zs
populations of HeLa and CaSki cells. Cell viability of the high-Zs
population of CaSki cells after treatment with both cisplatin and
paclitaxel was significantly higher than the low-Zs population by
two-way ANOVA (Fig. 6). There was
no significant difference in the high-Zs population of HeLa cells
by two-way ANOVA. These results confirmed chemoresistance of the
high-Zs population of CaSki cells.
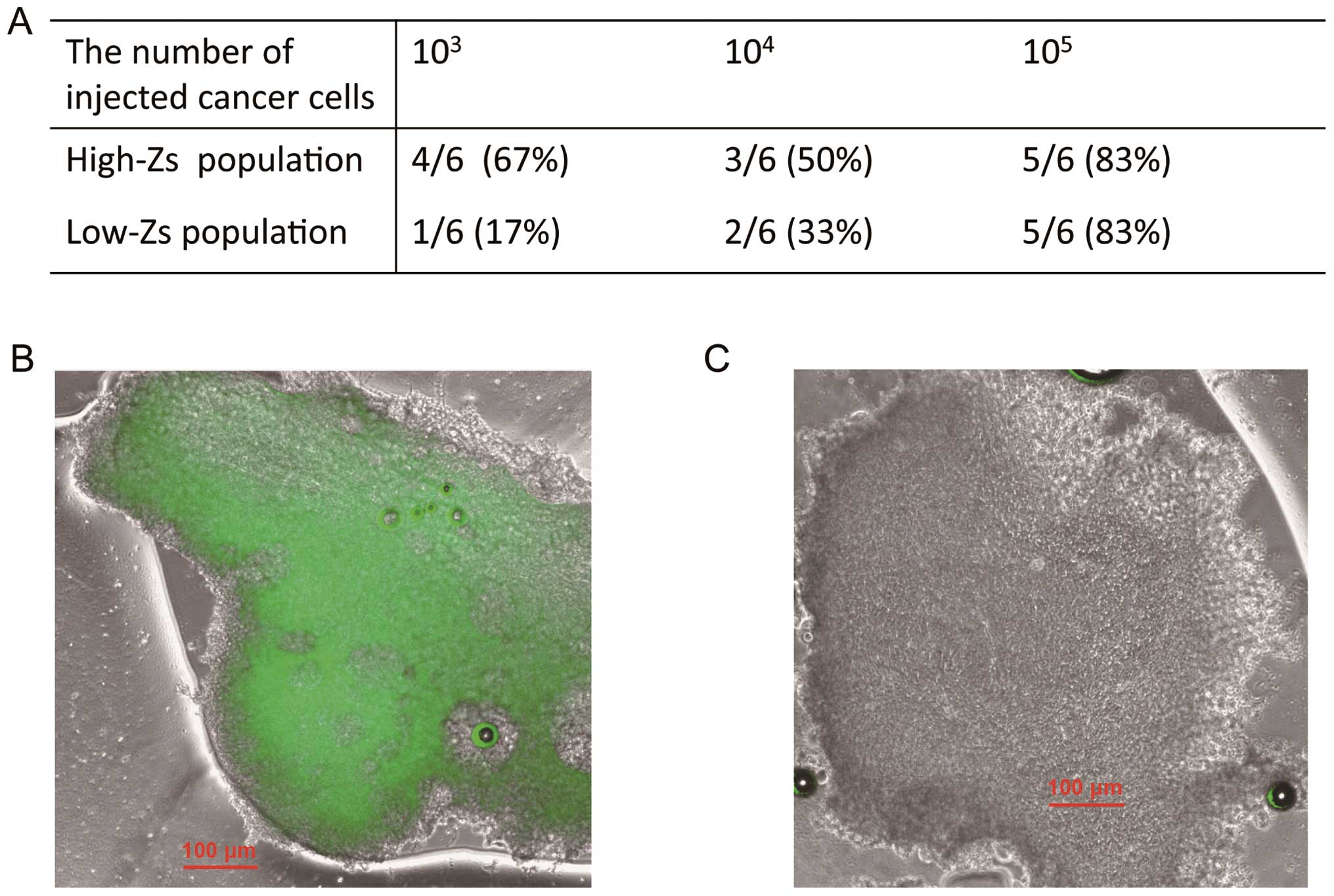
Tumorigenicity
Mice were injected with high- and low-Zs populations
of HeLa cells to assess the ability of the cells to form tumors
in vivo. The number of injected cancer cells required to
form a tumor in more than half of the mice was at least
103 and 105 for high- and low-Zs populations,
respectively (Fig. 7A), i.e., the
tumor-initiating ability of the high-Zs population was ~100-fold
greater than that of the low-Zs population. We observed resected
tumors using fluorescence microscopy (Fig. 7B and C) and found that tumors in
high-Zs populations exhibited fluorescence, whereas those in low-Zs
populations did not. These results showed that the high-Zs
population exhibited higher tumorigenic activity compared with the
low-Zs population.
Discussion
In the present study, the human cervical cancer cell
lines HeLa and CaSki were transfected with a retroviral vector
encoding the ZsGreen-cODC fluorescent fusion protein. We defined
high- and low-Zs populations of HeLa and CaSki cells sorted by FACS
and validated whether the high-Zs population satisfied the criteria
for CSCs, which is defined as those cells that have self-renewal
ability, differentiation capacity, and sustained proliferation
(4,5). First, the self-renewal ability of
high-Zs populations was confirmed using a time-lapse imaging
system. Moreover, the results of the sphere-forming assay showed
that the self-renewal ability of the high-Zs population was
significantly higher than that of the low-Zs population. Second,
the FACS analysis showed that the high-Zs population was capable of
asymmetrical division, indicating that it had the capacity for cell
differentiation. Third, the tumorigenicity assay validated that the
high-Zs population had high tumorigenic potential in vivo,
which was characterized by sustained proliferation. Moreover, the
chemo- and radioresistant properties of the high-Zs population were
validated by the MTT and clonogenic survival assays, in accordance
with the characteristics of CSCs described elsewhere (6,13).
Taken together, these results indicate that high-Zs populations of
both the HeLa and CaSki cell lines possess CSC-like properties and
therapeutic resistance.
Recent reports have shown the development of
visualization systems to assess CSCs in breast cancer, gliomas,
lung cancer, prostate cancer, hepatic carcinoma, esophageal cancer,
and pancreatic cancer using ZsGreen-cODC (11,12,14–17).
However, no previous reports address the use of ZsGreen-cODC in
cervical cancer. Thus, our results indicate a new application of
ZsGreen-cODC. In conclusion, we successfully visualized CSC-like
cells using a fluorescent protein system.
Acknowledgements
The authors would like to thank the members of our
laboratories for fruitful discussions. This study was supported in
part by a Grant-in-Aid for Scientific Research, a grant from the
Platform for Drug Discovery, Informatics, and Structural Life
Science from the Ministry of Education, Culture, Sports, Science
and Technology, and a Grant-in-Aid from the Third Comprehensive
10-Year Strategy for Cancer Control, Ministry of Health, Labor, and
Welfare, Japan. H.I., M.K., N.N., and J.K. received partial
financial support from Chugai Co., Ltd., Yakult Honsha Co., Ltd.,
Merck Co., Ltd., and Taiho Therapeutic Co., Ltd. via institutional
endowments.
References
|
1
|
Ahmedin J, Freddie B, Melissa M, et al:
Globan cancer statistics. CA Cancer J Clin. 61:69–90. 2011.
View Article : Google Scholar
|
|
2
|
Walboomers JM, Jacobs MV, Manos MM, et al:
Human papillomavirus is a necessary cause of invasive cervical
cancer worldwide. J Pathol. 189:12–19. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rose PG, Ali S, Watkins E, et al:
Long-term follow-up of a randomized trial comparing concurrent
single agent cisplatin, cisplatin-based combination chemotherapy,
or hydroxyurea during pelvic irradiation for locally advanced
cervical cancer: a Gynecologic Oncology Group Study. J Clin Oncol.
25:2804–2810. 2007. View Article : Google Scholar
|
|
4
|
Jordan CT, Guzman ML and Noble M: Cancer
stem cells. N Engl J Med. 355:1253–1261. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rich JN: Cancer stem cells in radiation
resistance. Cancer Res. 67:8980–8984. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bao S, Wu Q, McLendon RE, et al: Glioma
stem cells promote radioresistance by preferential activation of
the DNA damage response. Nature. 444:756–760. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Feng D, Peng C, Li C, et al:
Identification and characterization of cancer stem-like cells from
primary carcinoma of the cervix uteri. Oncol Rep. 22:1129–1134.
2009.PubMed/NCBI
|
|
8
|
Wang K, Zeng J, Luo L, et al:
Identification of a cancer stem cell-like side population in the
HeLa human cervical carcinoma cell line. Oncol Lett. 6:1673–1680.
2013.PubMed/NCBI
|
|
9
|
Rao Q-X, Yao T-T, Zhang B-Z, et al:
Expression and functional role of ALDH1 in cervical carcinoma
cells. Asian Pac J Cancer Prev. 13:1325–1331. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ye F, Zhou C, Cheng Q, Shen J and Chen H:
Stem-cell-abundant proteins Nanog, Nucleostemin and Musashi1 are
highly expressed in malignant cervical epithelial cells. BMC
Cancer. 8:1082008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vlashi E, Kim K, Lagadec C, et al: In vivo
imaging, tracking, and targeting of cancer stem cells. J Natl
Cancer Inst. 101:350–359. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kano Y, Konno M, Kawamoto K, et al: Novel
drug discovery system for cancer stem cells in human squamous cell
carcinoma of the esophagus. Oncol Rep. 31:1133–1138.
2014.PubMed/NCBI
|
|
13
|
Dean M, Fojo T and Bates S: Tumour stem
cells and drug resistance. Nat Rev Cancer. 5:275–284. 2005.
View Article : Google Scholar
|
|
14
|
Muramatsu S, Tanaka S, Mogushi K, et al:
Visualization of stem cell features in human hepatocellular
carcinoma reveals in vivo significance of tumor-host interaction
and clinical course. Hepatology. 58:218–228. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Adikrisna R, Tanaka S, Muramatsu S, et al:
Identification of pancreatic cancer stem cells and selective
toxicity of chemotherapeutic agents. Gastroenterology. 143:234–245.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pan J, Zhang Q, Wang Y and You M: 26S
proteasome activity is down-regulated in lung cancer stem-like
cells propagated in vitro. PLoS One. 5:e132982010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Della Donna L, Lagadec C and Pajonk F:
Radioresistance of prostate cancer cells with low proteasome
activity. Prostate. 72:868–874. 2012.PubMed/NCBI
|