Introduction
Prostaglandin I2 (PGI2) is an
important vascular prostanoid that provides an important balance in
tumor angiogenesis (1,2). Honn et al were the first to
demonstrate that PGI2 strongly reduced the number of
lung tumor metastases using an artificial lung metastasis model
(3). Since the initial reports of
the anti-metastatic action of PGI2 and its analogs, a
wide variety of tumor cell lines have been studied in models of
artificial metastasis (4–15). However, the relationship between
PGI2-prostacyclin receptor (IP) signaling and tumor
angiogenesis, including endothelial cells and pericyte interaction,
remains to be clarified.
Tumor blood vessels are structurally and
functionally abnormal, in that they lack the normal hierarchical
arrangement of arterioles, capillaries, and venules (16). Tumor endothelial cells are often
loosely connected to each other and are covered by fewer and more
abnormal mural pericytes (16–18).
In clinical data, low pericyte coverage of tumor blood vessels is
related to poor patient prognosis (19–21),
and pericyte dysfunction is suggested to increase metastasis
(22).
Our recent studies have revealed novel effects of
PGI2 on its target cells, such as endothelial and
endothelial progenitor cells (23), which suggested that
PGI2-IP signaling attenuates vascular maturation through
endothelial and pericyte interaction. In this study, we evaluated
whether activation of PGI2-IP signals of tumor blood
vessels by a stable PGI2 analog, beraprost sodium (BPS),
enhanced pericyte adhesion to endothelial cells, induced maturation
of tumor blood vessels, decreased hypoxic areas in the metastatic
tumors, and resulted in suppression of lung metastasis in lung
cancer.
Materials and methods
Lung cancer cell line and reagents
Lewis lung carcinoma (LLC; non-small cell lung
cancer derived from C57BL/6 mice) cells were purchased from
American Type Culture Collection (Manassas, VA, USA) and maintained
at 37°C in 5% CO2 using RPMI-1640 medium (Life
Technologies, Grand Island, NY, USA) containing 2 mM L-glutamine,
50 U/ml penicillin, and 50 μg/ml streptomycin, supplemented with
10% fetal bovine serum (complete medium). BPS was provided by Toray
Industries, Inc. (Chiba, Japan).
Mouse lung metastasis model
Female C57BL/6 mice, 8-.to 10-weeks-old (20–25 g),
were obtained from Charles River Laboratories Japan, Inc.
(Kanagawa, Japan). LLC cells (5.0×106 cells) in 500 μl
phosphate-buffered saline (PBS) were injected into the tail veins
of mice (5 mice/group) to generate lung tumor metastases. The day
after LLC cell injection, an Alzet mini-osmotic pump (Durect Corp.,
Cupertino, CA, USA) filled with BPS (20 μg/ml) or deionized
distilled water (DDW) was implanted under the skin of each mouse.
BPS or DDW was continuously administered for 3 weeks. To assess the
hypoxic area in metastatic tumors, mice were orally administered 15
mg/ml Hypoxyprobe-1 (pimonidazole HCl; Hypoxyprobe, Inc.,
Burlington, MA, USA) 1 h before sacrifice.
Immunohistochemistry
α-SMA (Abcam, Cambridge, UK) and NG2 (Millipore,
Billerica, MA, USA) as pericyte markers and Endomucin (Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA) as an endothelial cell
marker were studied by immunofluorescence to evaluate angiogenesis
in metastatic lung tumors. Hypoxic areas were evaluated by a
Hypoxyprobe-1 kit.
Zinc-fixed lung specimens were sectioned (4 μm
thickness), mounted onto slides, and air-dried for 30 min. The
sections were deparaffinized in xylene and rehydrated via a series
of graded alcohols. The slides were rinsed with PBS, and antigen
retrieval was enhanced by microwaving in 10 mM citrate buffer pH
6.0 for 20 min. They were incubated in 1% bovine serum albumin +
PBS-T (Triton) for 20 min at room temperature and then incubated
overnight at 4°C with a 1:100 dilution of rabbit anti-mouse α-SMA,
1:200 dilution of rabbit anti-mouse NG2, or 1:50 dilution of MAb1
(mouse monoclonal anti-pimonidazole antibody). They were rinsed
with PBS and then incubated for 1 h with a goat anti-rabbit Alexa
Fluor 488 antibody (diluted 1:1,000) or a donkey anti-mouse Alexa
Fluor 488 antibody (diluted 1:1,000) at room temperature. Slides
were rinsed with PBS, incubated for 20 min in 1% bovine serum
albumin + PBS-T, and then incubated with a 1:50 dilution of rat
anti-mouse Endomucin at 4°C overnight. Slides were rinsed with PBS
and then incubated for 1 h with a rabbit anti-rat Alexa Fluor 594
antibody (diluted 1:500) at room temperature. After rinsing, slides
were incubated with Hoechst 33258 (diluted 1:1,000; Invitrogen Life
Technologies, Carlsbad, CA, USA) for 30 min at room temperature.
Slides were mounted with Fluoromount (Diagnostic Biosystems,
Pleasanton, CA, USA) to prevent fluorescent bleaching.
Quantification of lung metastasis
Excised mouse lungs were fixed in zinc fixative (BD
Biosciences Pharmingen, Inc., San Diego, CA, USA) and embedded in
paraffin. Tumor metastasis to the lungs was assessed by hematoxylin
and eosin (H&E) staining. Light photomicrographs of the left
lobe of the lungs were taken at magnification, ×40 (5 visual
fields/section and 5 sections/mouse) using a light microscope
(BX51; Olympus, Tokyo, Japan). The number of metastatic nodules was
counted, and the metastatic area was quantified using ImageJ
software (NIH, Bethesda, MD, USA).
DDW- and BPS-treated mice within an experimental set
(5 mice/group) were analyzed with the same threshold and results as
reported below. Tumors were selected at random from each slide (4
tumors/mouse and 5 mice/group) at magnification, ×200 observed with
a fluorescence microscope (BX51; Olympus). The tumor area, number
of Endomucin+ cells in the tumor, and number of
α-SMA+ (or NG2+)/Endomucin+
double-positive cells in the tumor were quantified. Results were
reported as number of vessel-associated pericytes per
Endomucin+ cell per tumor area (/mm2). The
area of hypoxia was analyzed as described below. Tumors observed
with a fluorescence microscope were selected at random from each
slide (4 tumors/mouse and 2 mice/group) at original magnification,
×200. The tumor area and area of MAb1+ cells in each
tumor were quantified by ImageJ software. The result is reported as
the ratio of hypoxic area to tumor area.
Scanning electron microscopy
Tissue preparation for scanning electron microscopy,
the potassium hydroxide (KOH) digestion method was described
previously (24). Anesthetized
control and treated mice were perfused with physiological saline
followed by a mixture of 0.5% glutar-aldehyde (GA)-0.5%
paraformaldehyde (PFA) in 0.1 M phosphate buffer solution (PB), pH
7.4. After fixation by perfusion, lungs were cut and immersed in 2%
GA in 0.1 M PB for 2 weeks at 4°C. Then the tissue blocks were
washed thoroughly with 0.1 M PB, immersed in 30% KOH solution for
8–10 min at 60°C to remove the extracellular matrix around tumor
blood vessels. After KOH-digested tissue blocks were rinsed five or
six times in 0.1 M PB, they were conductively stained by treating
with 1% tannic acid in 0.1 M PB (2 h, 20°C) and 1% OsO4
in 0.1 M PB (2 h, 20°C). After conductive staining, the samples
were dehydrated in graded ethanols, transferred to isoamyl acetate,
and dried in a critical point dryer (HCP-2; Hitachi Koki Co., Ltd.,
Tokyo, Japan) using liquid CO2. The dried samples were
mounted onto a metal plate, coated with platinum-palladium using an
ion-sputter coater (E1010; Hitachi Koki Co., Ltd.), and then
observed with a field emission type scanning electron microscope
(S-4100; Hitachi High-Technologies Corp., Tokyo, Japan).
Clonogenic growth assay
LLC cells (1.5×103 cells) were incubated
in 6-well plates for 24 h. Subsequently, growth medium was changed
to complete medium containing the indicated concentrations of BPS.
Treated cells were incubated under normoxic condition for 10 days.
After incubation, colonies in a 6-well plate were stained with 0.5%
crystal violet (Wako, Osaka, Japan) in 0.5% methanol. The number of
colonies was determined by a colony counter and software (Microtec
Nition, Chiba, Japan).
Cell proliferation assay
We used BrdU assays [Cell Proliferation ELISA, BrdU
(colorimetric); Roche, Tokyo, Japan] to assess cell proliferation.
LLC cells (3.0×103 cells) were incubated in 96-well
plates. The next day, the medium was replaced by complete medium
containing the indicated concentrations of BPS, and LLC cells were
incubated for 3 days. After a 3-day BPS treatment, the BrdU assay
was performed according to the manufacturer’s protocol. BrdU was
added in the medium, and LLC cells were incubated for 2 h. The
BrdU-uptake in the treated cells was assessed using a microplate
luminometer (Thermo Fisher Scientific, Waltham, MA, USA).
Antibody array
LLC cells (2.5×103 cells) were incubated
in 100 mm dishes for 24 h. Subsequently, the growth medium was
changed to complete medium containing 0 or 10 nM BPS. Treated cells
were incubated under the normoxic condition for 4 days. After
incubation, we measured the cytokine spectrum in the supernatants
using the Proteome Profiler™ Mouse Angiogenesis Array kit (R&D
Systems, Minneapolis, MN, USA), which detects 53 cytokines,
chemokines, and growth factors simultaneously. Array membranes were
processed following the manufacturer’s recommendations. The signal
intensity was measured on the LAS-3000 luminescence detector, and
the resulting images were analyzed using Multi Gauge (Version 2.2;
both from Fujifilm, Tokyo, Japan). To compare the luminescence
intensities of the samples, we subtracted the background staining
and normalized the data to the positive controls on the same
membrane.
Statistical analysis
The measurements are presented as means ± SEM.
Results were analyzed by Student’s t-test using Microsoft Excel.
Two-sided p<0.05 was considered to be statistically
significant.
Results
BPS treatment reduces lung
metastasis
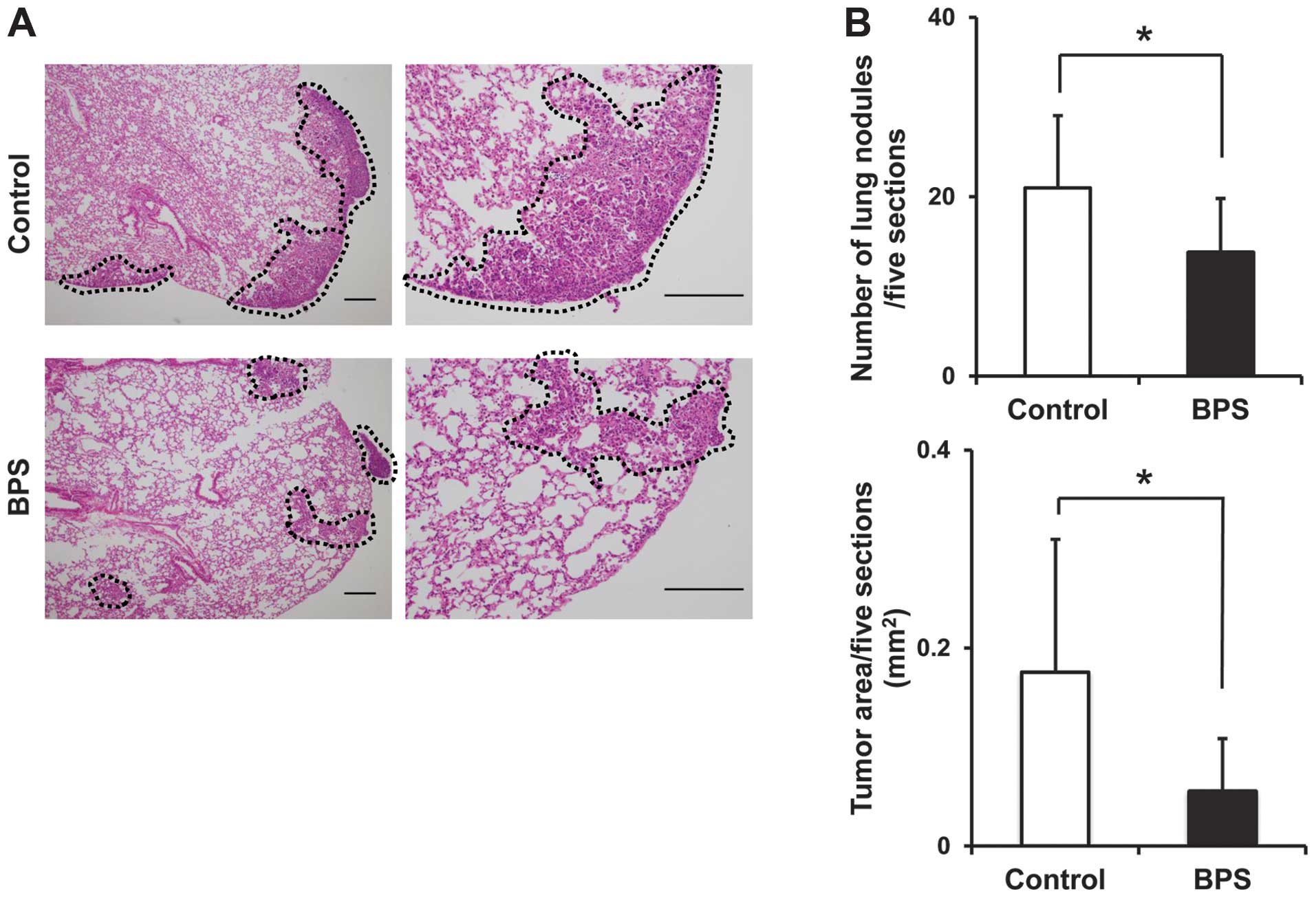
To evaluate the effect of BPS on lung metastasis, we
employed an experimental lung metastasis model. After tumor cell
inoculation, mice were treated with BPS for 3 weeks; then they were
sacrificed, and lung metastases were counted in the H&E-stained
lung sections. In control groups, tumors with wide-spread pleural
dissemination of colonized metastases were observed, while tumors
with randomly clustered small metastatic nodules were observed in
the BPS-treated group (Fig. 1A).
The median number of metastatic nodules in the BPS-treated groups
was significantly reduced compared with that in the control group
(13.8 vs. 21.0, respectively, p<0.05) (Fig. 1B). The median area of metastatic
nodules in BPS-treated groups was significantly smaller than that
in control groups (0.06 and 0.18/mm2, respectively,
p<0.05) (Fig. 1B). These
results suggest that administration of BPS significantly reduced
the number of lung metastases in our mouse lung metastasis
model.
BPS enhances pericyte and endothelial
interaction in tumor microvasculature
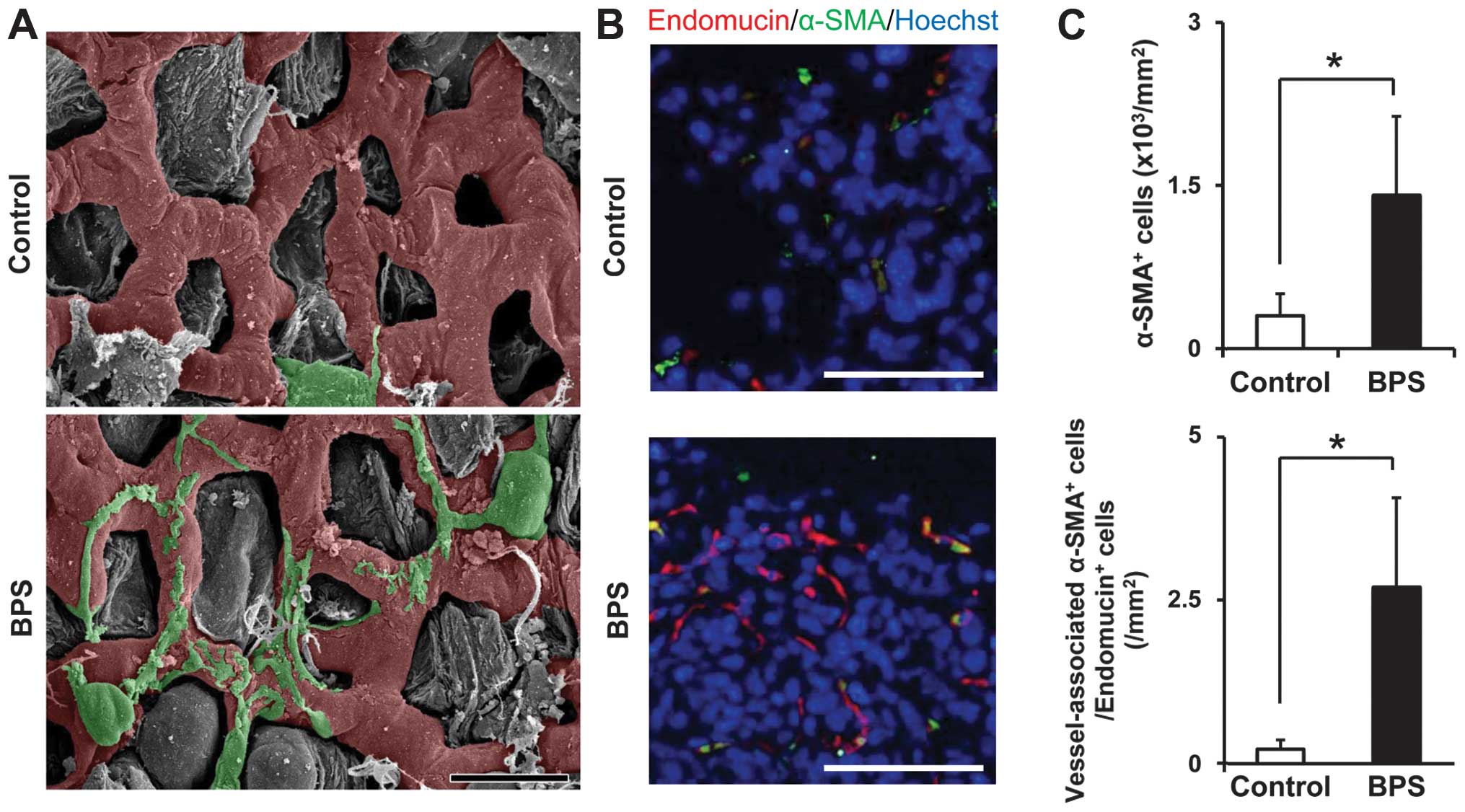
Next, to evaluate vascular maturation through
endothelial and pericyte interaction, we analyzed the structure of
tumor blood vessels in the mouse metastatic tumors using scanning
electron microscopy. In the BPS-treated group, pericyte bodies
(green) attached to endothelial capillary tubes (red) along with
processes, and the diameters of tumor blood vessels were decreased,
while in the control group, pericytes were absent or loosely
connected (Fig. 2A).
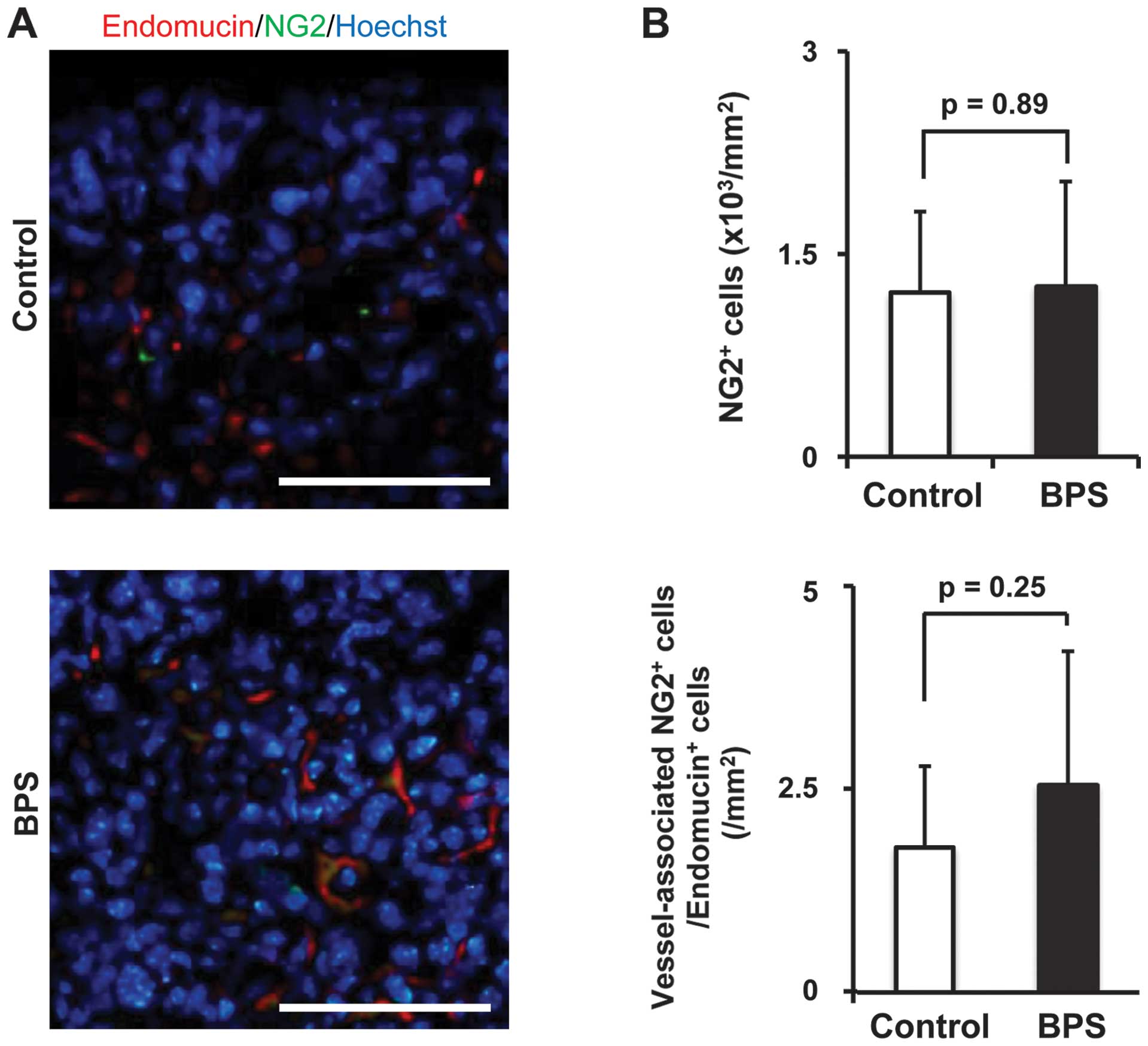
To evaluate the effects of BPS on tumor
angiogenesis, including pericyte association at the metastatic
site, pericytes (α-SMA+ or NG2+ cells) and
endothelial cells (Endomucin+ cells) in the metastatic
tumor site were analyzed by immunofluorescence (Figs. 2B and 3A). In the control group,
α-SMA+ or NG2+ cells were mostly located
randomly in the metastatic tumor sites, and they did not colocalize
with Endomucin+ cells (Figs. 2B and 3A), while in the BPS-treated group,
α-SMA+ or NG2+ cells coexisted regularly
beside Endomucin+ cells, and most of these cells were
merged with Endomucin+ cells (Figs. 2B and 3A), which revealed that the number of
mature pericytes had increased, and moreover, the number of
endothelial-attached pericytes was increased compared with that in
the control group (Figs. 2C and
3B). These results suggested that
BPS strongly enhanced pericyte and endothelial interaction in the
tumor microvasculature.
BPS induces vascular functional
maturation in metastatic tumors
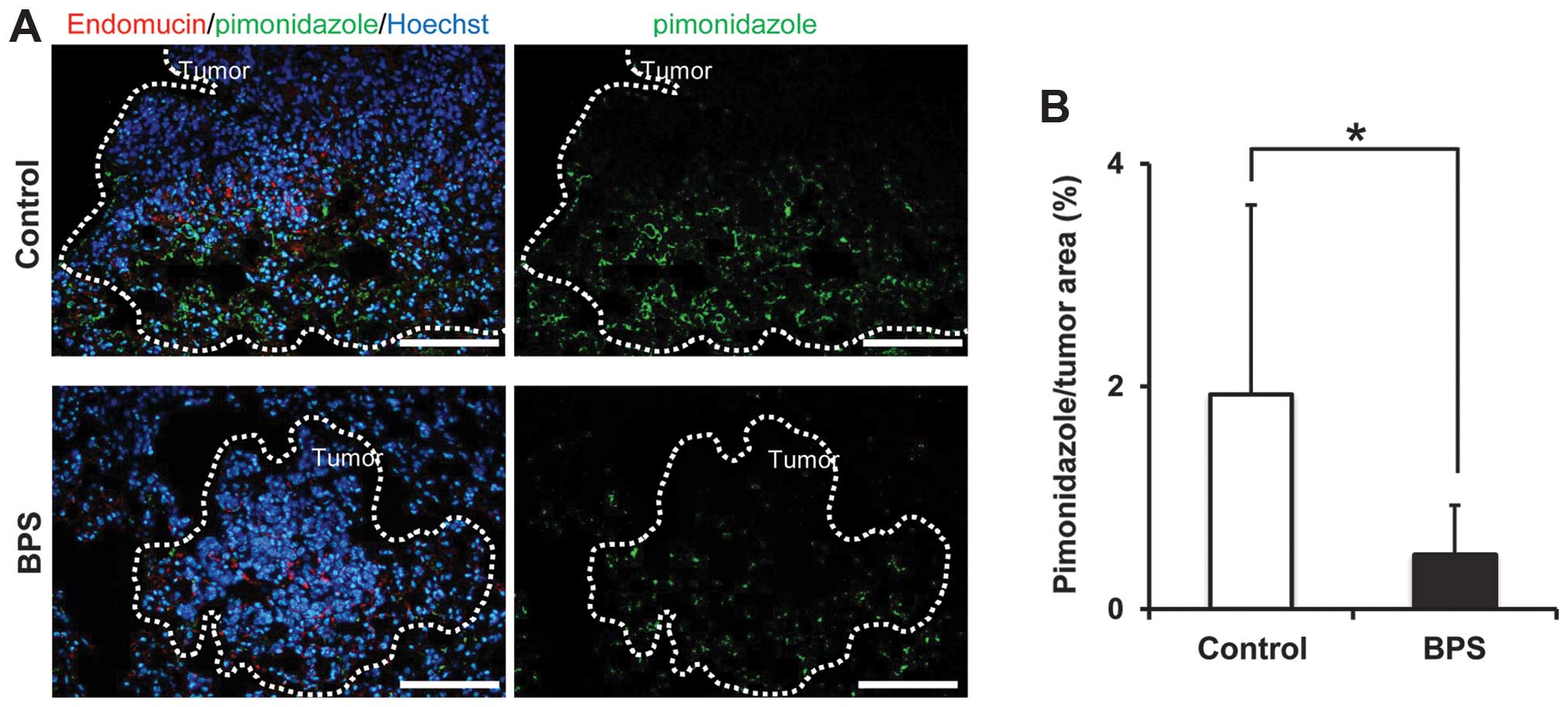
We hypothesized that functionally mature vasculature
without hypoxic regions abrogate tumor metastasis because
pericyte-covered endothelial cells are considered ‘mature
vasculature,’ which is rarely observed in the tumor area. In order
to evaluate the tumor vascular maturation, we measured hypoxia
levels in the metastatic tumors by immunohistochemistry using
pimonidazole as a hypoxia marker. Pimonidazole stained the inside
of the metastatic tumors in the control group widely, while it
stained scattered and diminished areas in the BPS-treated group
(Fig. 4A). Pimonidazole staining
per tumor area in the BPS-treated group was 0.5%, while in the
control group it was 1.9%. The area stained by pimonidazole was
significantly decreased in the BPS-treated group (p<0.05)
(Fig. 4B). These results suggested
that BPS induced maturation of vascular function in metastatic
tumors.
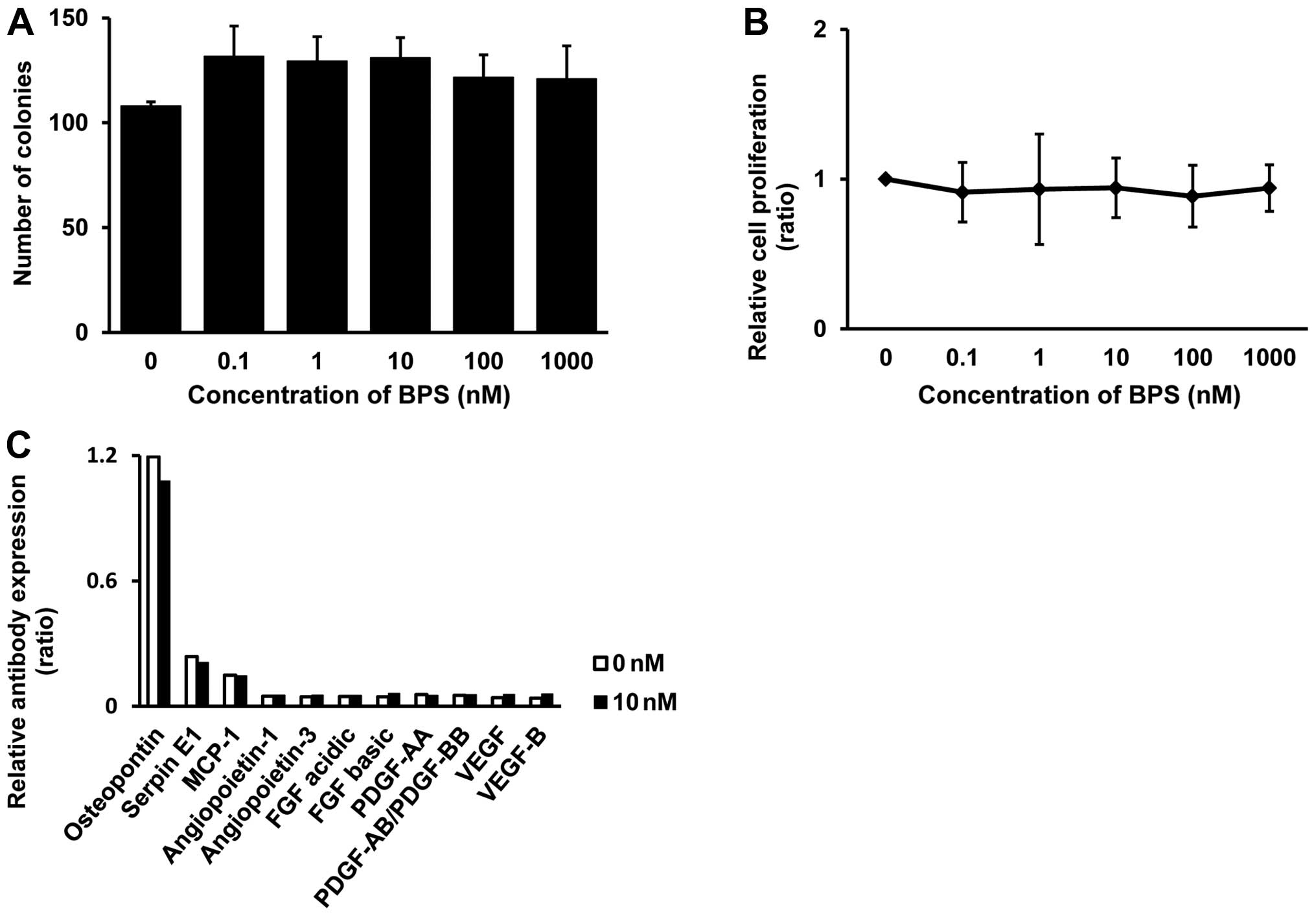
Antitumor effects of BPS against cancer
cells
Because a direct inhibitory effect of a
PGI2 analog on tumor cells, has been reported (25–27),
we evaluated the antitumor effects of BPS on LLC cells. The cell
growth was assessed by the BrdU cell growth assay or clonogenic
assay (Fig. 5A and B). In our
results, BPS did not inhibit tumor growth. To examine the autocrine
factors related to tumor angiogenesis, we analyzed the effects of
conditioned media with or without BPS on LLC cells using an
antibody array that simultaneously detected the relative
concentrations of 53 angiogenesis-related proteins (Fig. 5C). In this assay, osteopontin,
serpin E1, and MCP-1 were elevated in both BPS-treated and
untreated groups, but the change in these factors were not
significant. Based on these results, BPS did not affect LLC cell
growth directly.
Discussion
In the present study, the number of lung metastases
was significantly reduced by BPS treatment in our mouse model.
Since the first report from Honn and colleagues demonstrating that
PGI2 and its analogs strongly reduced the number of lung
metastases (3), studies of the
PGI2 effect on metastasis have been repeated using a
wide variety of tumor cell lines (4–15).
Because multistep processes of metastasis formation are responsible
for the tumor spread, the effect of BPS on tumors and the tumor
microenvironment needs to be analyzed. Most studies have examined:
i) tumor cell-induced platelet aggregation and its inhibition by
PGI2 (5,6,28–30);
ii) prevention of endothelial cell retraction (31–33);
iii) modulation of immune systems (4,7); or
iii) direct inhibitory effects on tumor cells (25–27).
However, the mechanisms of tumor vessel maturation by
PGI2 was not previously examined.
In this study, we demonstrated that BPS strongly
enhanced pericyte and endothelial interaction in the tumor
microvasculature, which is a novel anti-metastatic mechanism of a
PGI2 analog. The present results suggested that BPS
induced structural changes in tumor vessels and led to endothelial
maturation, which is consistent with our previous results (34). Consequently, tumor vessel
maturation was induced, and hypoxia levels in the metastatic tumors
decreased, resulting in BPS-induced vascular normalization in the
tumor microenvironment. These results seem paradoxical because
improving circulation in the tumor leads to tumor shrinkage, but
clinically used anti-angiogenic therapies that successfully target
tumor vessels are believed to increase microenvironment-induced
tumor shrinkage. Bevacizumab, an anti-angiogenic drug, is now used
in combination with cytotoxic agent for vascular normalization.
In the analysis of immunohistochemistry,
α-SMA+ cells merged with Endomucin+ cells
significantly increased in the BPS-treated group (Fig. 2C), while NG2+ cells
merged with Endomucin+ cells did not significantly
increase in the BPS-treated group (Fig. 3B). This result seems to be
inconsistent. However, in general, it is known that α-SMA expresses
in more mature status of pericytes than NG2 (35), and BPS induced more mature
pericytes in this study. The number of endothelial cells in the
tumors increased in the BPS-treated group with or without pericytes
(data not shown). These results indicate that BPS promotes the
maturation of tumor blood vessels.
Finally, to rule out the possibility of a direct
tumor effect of BPS on LLC cells, we investigated tumor growth
inhibition by treatment of cultured cells with BPS at
concentrations up to 1 μM. The angiogenic factors produced by LLC
cells themselves were not changed. These results suggested that BPS
did not affect tumors directly, but affected the tumor
microenvironment.
Altering the tumor microenvironment by addition of
PGI2 analogs that affect endothelium-pericyte
interaction may yield strategies for targeted angiogenesis
therapies. However, additional clinical studies are needed to
clarify the potential benefits and risks associated with
anti-metastatic treatment by PGI2 analogs.
Acknowledgements
The authors thank Dr Fumitaka Ushikubi for advice
and kindly providing experimental equipment. This study was
partially supported by funding from the JSPS Grant-in-Aid for
Scientific Research (C) (KAKENHI) Grant no. 20590910.
References
|
1
|
Turner EC, Mulvaney EP, Reid HM and
Kinsella BT: Interaction of the human prostacyclin receptor with
the PDZ adapter protein PDZK1: role in endothelial cell migration
and angiogenesis. Mol Biol Cell. 22:2664–2679. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhu W, Saddar S, Seetharam D, et al: The
scavenger receptor class B type I adaptor protein PDZK1 maintains
endothelial monolayer integrity. Circ Res. 102:480–487. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Honn KV, Cicone B and Skoff A:
Prostacyclin: a potent antimetastatic agent. Science.
212:1270–1272. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gorelik E, Bere WW and Herberman RB: Role
of NK cells in the antimetastatic effect of anticoagulant drugs.
Int J Cancer. 33:87–94. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Menter DG, Harkins C, Onoda J, et al:
Inhibition of tumor cell induced platelet aggregation by
prostacyclin and carbacyclin: an ultrastructural study. Invasion
Metastasis. 7:109–128. 1987.PubMed/NCBI
|
|
6
|
Niitsu Y, Ishigaki S, Kogawa K, et al:
Effect of combined administration of a prostacyclin analogue and
adriamycin against the artificial metastasis of Meth A cell.
Invasion Metastasis. 8:57–72. 1988.PubMed/NCBI
|
|
7
|
Sava G, Perissin L, Zorzet S, Piccini P
and Giraldi T: Antimetastatic action of the prostacyclin analog
iloprost in the mouse. Clin Exp Metastasis. 7:671–678. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Honn KV: Inhibition of tumor cell
metastasis by modulation of the vascular prostacyclin/thromboxane
A2 system. Clin Exp Metastasis. 1:103–114. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Karpatkin S, Ambrogio C and Pearlstein E:
Lack of effect of in vivo prostacyclin on the development of
pulmonary metastases in mice following intravenous injection of
CT26 colon carcinoma, Lewis lung carcinoma, or B16 amelanotic
melanoma cells. Cancer Res. 44:3880–3883. 1984.PubMed/NCBI
|
|
10
|
Mahalingam M, Ugen KE, Kao KJ and Klein
PA: Functional role of platelets in experimental metastasis studied
with cloned murine fibrosarcoma cell variants. Cancer Res.
48:1460–1464. 1988.PubMed/NCBI
|
|
11
|
Lapis K, Timár J, Pápay J, Paku S, Szende
B and Ladányi A: Experimental metastasis inhibition by pretreatment
of the host. Arch Geschwulstforsch. 60:97–102. 1990.PubMed/NCBI
|
|
12
|
Kato S, Kobari M, Matsuno S and Sato T:
Inhibitory effect of anti-platelet prostaglandin on liver
metastasis of hamster pancreatic cancer. Nihon Geka Gakkai Zasshi.
90:745–752. 1989.(In Japanese). PubMed/NCBI
|
|
13
|
Schwalke MA, Tzanakakis GN and Vezeridis
MP: Effects of prostacyclin on hepatic metastases from human
pancreatic cancer in the nude mouse. J Surg Res. 49:164–167. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tzanakakis GN, Agarwal KC and Vezeridis
MP: Inhibition of hepatic metastasis from a human pancreatic
adenocarcinoma (RWP-2) in the nude mouse by prostacyclin,
forskolin, and ketoconazole. Cancer. 65:446–451. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Costantini V, Fuschiotti P, Giampietri A,
et al: Effects of a stable prostacyclin analogue on platelet
activity and on host immunocompetence in mice. Prostaglandins.
39:581–599. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pasqualini R, Arap W and McDonald DM:
Probing the structural and molecular diversity of tumor
vasculature. Trends Mol Med. 8:563–571. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Minami Y, Sasaki T, Kawabe J and Ohsaki Y:
Accessory cells in tumor angiogenesis - tumor-associated pericytes.
Research Directions in Tumor Angiogenesis. Chai J: InTech; Croatia:
pp. 73–89. 2013
|
|
18
|
Morikawa S, Baluk P, Kaidoh T, Haskell A,
Jain RK and McDonald DM: Abnormalities in pericytes on blood
vessels and endothelial sprouts in tumors. Am J Pathol.
160:985–1000. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
O’Keeffe MB, Devlin AH, Burns AJ, et al:
Investigation of pericytes, hypoxia, and vascularity in bladder
tumors: association with clinical outcomes. Oncol Res. 17:93–101.
2008.
|
|
20
|
Stefansson IM, Salvesen HB and Akslen LA:
Vascular proliferation is important for clinical progress of
endometrial cancer. Cancer Res. 66:3303–3309. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yonenaga Y, Mori A, Onodera H, et al:
Absence of smooth muscle actin-positive pericyte coverage of tumor
vessels correlates with hematogenous metastasis and prognosis of
colorectal cancer patients. Oncology. 69:159–166. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xian X, Håkansson J, Ståhlberg A, et al:
Pericytes limit tumor cell metastasis. J Clin Invest. 116:642–651.
2006. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kawabe J, Yuhki K, Okada M, et al:
Prostaglandin I2 promotes recruitment of endothelial
progenitor cells and limits vascular remodeling. Arterioscler
Thromb Vasc Biol. 30:464–470. 2010. View Article : Google Scholar
|
|
24
|
Ushiki T and Murakumo M: Scanning electron
microscopic studies of tissue elastin components exposed by a
KOH-collagenase or simple KOH digestion method. Arch Histol Cytol.
54:427–436. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tennis MA, Van Scoyk M, Heasley LE, et al:
Prostacyclin inhibits non-small cell lung cancer growth by a
frizzled 9-dependent pathway that is blocked by secreted
frizzled-related protein 1. Neoplasia. 12:244–253. 2010.PubMed/NCBI
|
|
26
|
Honn KV and Meyer J: Thromboxanes and
prostacyclin: positive and negative modulators of tumor growth.
Biochem Biophys Res Commun. 102:1122–1129. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tang DG, Grossi IM, Chen YQ, Diglio CA and
Honn KV: 12(S)-HETE promotes tumor-cell adhesion by increasing
surface expression of alpha V beta 3 integrins on endothelial
cells. Int J Cancer. 54:102–111. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Honn KV, Busse WD and Sloane BF:
Prostacyclin and thromboxanes. Implications for their role in tumor
cell metastasis. Biochem Pharmacol. 32:1–11. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Menter DG, Onoda JM, Moilanen D, Sloane
BF, Taylor JD and Honn KV: Inhibition by prostacyclin of the tumor
cell-induced platelet release reaction and platelet aggregation. J
Natl Cancer Inst. 78:961–969. 1987.PubMed/NCBI
|
|
30
|
Menter DG, Onoda JM, Taylor JD and Honn
KV: Effects of prostacyclin on tumor cell-induced platelet
aggregation. Cancer Res. 44:450–456. 1984.PubMed/NCBI
|
|
31
|
Honn KV, Tang DG, Grossi IM, et al:
Enhanced endothelial cell retraction mediated by 12(S)-HETE: a
proposed mechanism for the role of platelets in tumor cell
metastasis. Exp Cell Res. 210:1–9. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Honn KV, Grossi IM, Diglio CA,
Wojtukiewicz M and Taylor JD: Enhanced tumor cell adhesion to the
subendothelial matrix resulting from 12(S)-HETE-induced endothelial
cell retraction. FASEB J. 3:2285–2293. 1989.PubMed/NCBI
|
|
33
|
Honn KV, Tang DG, Grossi I, et al: Tumor
cell-derived 12(S)-hydroxyeicosatetraenoic acid induces
microvascular endothelial cell retraction. Cancer Res. 54:565–574.
1994.PubMed/NCBI
|
|
34
|
Aburakawa Y, Kawabe J, Okada M, et al:
Prostacyclin stimulated integrin-dependent angiogenic effects of
endothelial progenitor cells and mediated potent circulation
recovery in ischemic hind limb model. Circ J. 77:1053–1062. 2013.
View Article : Google Scholar
|
|
35
|
Cipriani P, Marrelli A, Benedetto PD, et
al: Scleroderma Mesenchymal Stem Cells display a different
phenotype from healthy controls; implications for regenerative
medicine. Angiogenesis. 16:595–607. 2013. View Article : Google Scholar : PubMed/NCBI
|