Introduction
Head and neck squamous cell carcinomas (HNSCC),
known for their aggressive growth and propensity to metastasize,
are among the most common tumors developed in patients with Fanconi
anemia (1,2). Although morphologically the cancer is
the same, the incidence and course of HNSCC is altered in FA
patients. Incidence of HNSCC reaches 14% among Fanconi anemia
patients by age 40 years, while the incidence in the general
population is only 0.038% (3).
Furthermore, the associated risk factors of tobacco and alcohol
that is associated with 85% of the non-FA HNSCC does not play as
much of a role in FA; approximately 16% of FA HNSCC cases are
associated with these risk factors (3). HNSCC is more aggressive in patients
with FA, with early lymph node metastases and early soft tissue
invasion, resulting in poorer prognoses than in HNSCC patients
without FA (3). Secondary primary
tumors occur in 63% of FA patients compared to only 15% in non-FA
patients (3).
Furthermore, 2-year overall survival is only 49% in
FA patients compared to 70% in non-FA patients (3). HNSCC most frequently presents in the
oral cavity (65%), compared to the larynx, hypopharynx and
oropharynx, each at 10%, which differs from the HNSCC in the
general population. Due to significant toxic sequelae from use of
radiation therapy and/or chemotherapy in FA patients, surgical
treatment is the main modality used. HNSCC in the general
population is treated with radiation, chemotherapy and surgery. The
highly metastatic potential of HNSCC in FA patients and inadequate
treatment methods, leading to poor outcomes, create an urgent need
to develop more effective, less toxic treatment alternatives.
Use of natural health products to prevent, inhibit
and reverse carcinogenesis is gaining increasing importance, since
scientific evidence shows that high consumption of fruits and
vegetables are associated with reduced risk of various cancers
(4,5).
This study examines the effect of a mixture of
phytobiologicals (PB), containing quercetin, curcumin, green tea,
cruciferex, and resveratrol, on human FA HNSCC cell line OHSU-974
in vivo, in athymic nude mice bearing HNSCC xenografts, and
in vitro, evaluating cell viability, MMP secretion, invasion
and migration.
Materials and methods
Cancer cell line and culture
Human FAHNSCC cell line OHSU-974 was obtained from
Fanconi Anemia Research Fund, Oregon Health and Science University
(Portland, OR, USA). FAHNSCC cells were maintained in RPMI medium
supplemented with 20% FBS, 100 U/ml penicillin and 100 μg/ml
streptomycin. The media and sera used were obtained from ATCC, and
antibiotics (penicillin and streptomycin) were from Gibco BRL, Long
Island, NY.
Composition of the phytonutrient
mixture
The mixture of nutrients (PB) was composed of the
following in the ratio indicated: quercetin 400 mg; cruciferex 400
mg; curcumin 300 mg; resveratrol 50 mg; and standardized green tea
extract [derived from green tea leaves, was obtained from US Pharma
Lab Inc.; the certificate of analysis indicated the following
characteristics: total polyphenol 80%, catechins 60%,
epigallocatechin gallate (EGCG) 35%, and caffeine 1.0%] 300 mg.
In vivo studies
Animals
Male athymic mice (NCr-nu/nu), approximately five
weeks of age on arrival, were purchased from Simonsen Laboratories
(Gilroy, CA, USA) and maintained in microisolator cages under
pathogen-free conditions on a 12-h light/12-h dark schedule for a
week. All procedures were performed according to humane and
customary care and use of experimental animals and followed a
protocol approved by the internal institutional animal safety
review committee.
Experimental design
After housing for a week, the mice (n=12) were
inoculated subcutaneously with 3×106 OHSU-974 cells in
0.2 ml PBS and 0.1 ml Matrigel (BD Bioscience, Bedford, MA). After
injection, the mice were randomly divided into two groups; the
control group of mice was fed regular Purina mouse chow and the PB
group the regular diet supplemented with 1% PB (w/w). The regular
diet was Laboratory Rodent Diet 5001 from Purina Mills (Gray
Summit, MO, USA) LLC/Test Diet. The 1% PB diet was milled and
pressed by Purina Mills, LLC and generated by Vitatech (Tustin, CA,
USA). During the study, the mice consumed, on the average, 4 g of
their respective diets per day. Thus, the supplemented mice
received ~40 mg of PB per day. After four weeks, the mice were
sacrificed and their tumors were excised and processed for
histology.
Dimensions (length and width) of tumors were
measured using a digital caliper, and the tumor burden was
calculated using the following formula: 0.5 × length × width. Mean
weight of mice at initiation of study and termination of study did
not differ significantly between the groups.
Histology
Tissue samples were fixed in 10% buffered formalin.
All tissues were embedded in paraffin and cut at 4–5 microns.
Sections were deparaffinized through xylene and graduated alcohol
series to water and stained with hematoxylin and eosin (H&E)
for evaluation using a standard light microscope.
In vitro studies
Cell culture
Human HNSCC OHSU-974 cells were grown in RPMI,
supplemented with 20% fetal bovine serum, penicillin (100 U/ml) and
streptomycin (100 mg/ml) in 24-well tissue culture plates (Costar,
Cambridge, MA, USA). Cells were incubated with 1 ml of media at
37°C in a tissue culture incubator equilibrated with 95% air and 5%
CO2. At near confluence, the cells were treated with PB,
dissolved in media and tested at 0, 10, 25, 50, 75 and 100 μg/ml in
triplicate at each dose. Phorbol 12-myristate 13-acetate (PMA), 100
ng/ml was added to cells to induce MMP-9 secretion. The plates were
then returned to the incubator.
MTT assay
Cell viability was evaluated by MTT assay, a
colorimetric assay based on the ability of viable cells to reduce a
soluble yellow tetrazolium salt [3-(4,5-dimethylthiazol-2-yl)
2,5-diphenyl tetrazolium bromide] (MTT) to a blue formazan crystal
by mitochondrial succinate dehydrogenase activity of viable cells.
This test is a good index of mitochondrial activity and thus of
cell viability. After 24 h of incubation, the cells were washed
with phosphate buffered saline (PBS) and 500 μl of MTT (Sigma
#M-2128) 0.5 mg/ml in media was added to each well. After MTT
addition (0.5 mg/ml) the plates were covered and returned to the
37°C incubator for 2 h, the optimal time for formazan product
formation. Following incubation, the supernatant was carefully
removed from the wells, the formazan product was dissolved in 1 ml
DMSO, and absorbance was measured at 570 nm in Bio Spec 1601,
Shimadzu spectrometer. The OD570 of the DMSO solution in
each well was considered to be proportional to the number of cells.
The OD570 of the control (treatment without supplement)
was considered 100%.
Gelatinase zymography
Gelatinase zymography was performed in 10% Novex
Pre-Cast SDS Polyacrylamide Gel (Invitrogen Corp.) in the presence
of 0.1% gelatin under non-reducing conditions. Culture media (20
μl) were mixed with sample buffer and loaded for SDS-PAGE with Tris
glycine SDS buffer, as suggested by the manufacturer (Novex).
Samples were not boiled before electrophoresis. Following
electrophoresis the gels were washed twice in 2.5% Triton X-100 for
30 min at room temperature to remove SDS. The gels were then
incubated at 37°C overnight in substrate buffer containing 50 mM
Tris-HCl and 10 mM CaCl2 at pH 8.0 and stained with 0.5%
Coomassie Blue R250 in 50% methanol and 10% glacial acetic acid for
30 min and destained. Upon renaturation of the enzyme, the
gelatinases digested the gelatin, producing clear bands against an
intensely stained background. Protein standards were run
concurrently and approximate molecular weights were determined by
plotting the relative mobilities of known proteins.
Matrigel invasion
Invasion studies were conducted using Matrigel
(Becton Dickinson) inserts in 24-well plates. Suspended in medium,
OHSU-974 cells were supplemented with nutrients, as specified in
the design of the experiment and seeded on the insert in the well.
Thus, both the medium on the insert and in the well contained the
same supplements. The plates with the inserts were then incubated
in a culture incubator equilibrated with 95% air and 5%
CO2 for 24 h. After incubation, the media from the wells
were withdrawn. The cells on the upper surface of the inserts were
gently scrubbed away with cotton swabs. The cells that had
penetrated the Matrigel membrane and migrated onto the lower
surface of the Matrigel were stained with H&E and visually
counted under the microscope.
Cell migration: scratch test
To study cell migration, a 2-mm wide single
uninterrupted scratch was made from the top to bottom of culture
plates of OHSU-947 cells grown to confluence. Culture plates were
washed with PBS and incubated with PB in medium and tested at 0,
10, 25, 50 and 100 μg/ml, in triplicate at each dose for 24 h.
Cells were washed with PBS, fixed and stained with H&E and
photomicrographs were taken.
Morphology
Morphology of cells cultured for 24 h in test
concentrations of PB was evaluated by hematoxylin and eosin
(H&E) staining and observed and photographed by microscopy.
Statistical analysis
The results are expressed as means ± SD, as
indicated in the results, for the groups. Data were analyzed by
independent sample ‘t’ test.
Results
In vivo
Tumor growth and burden
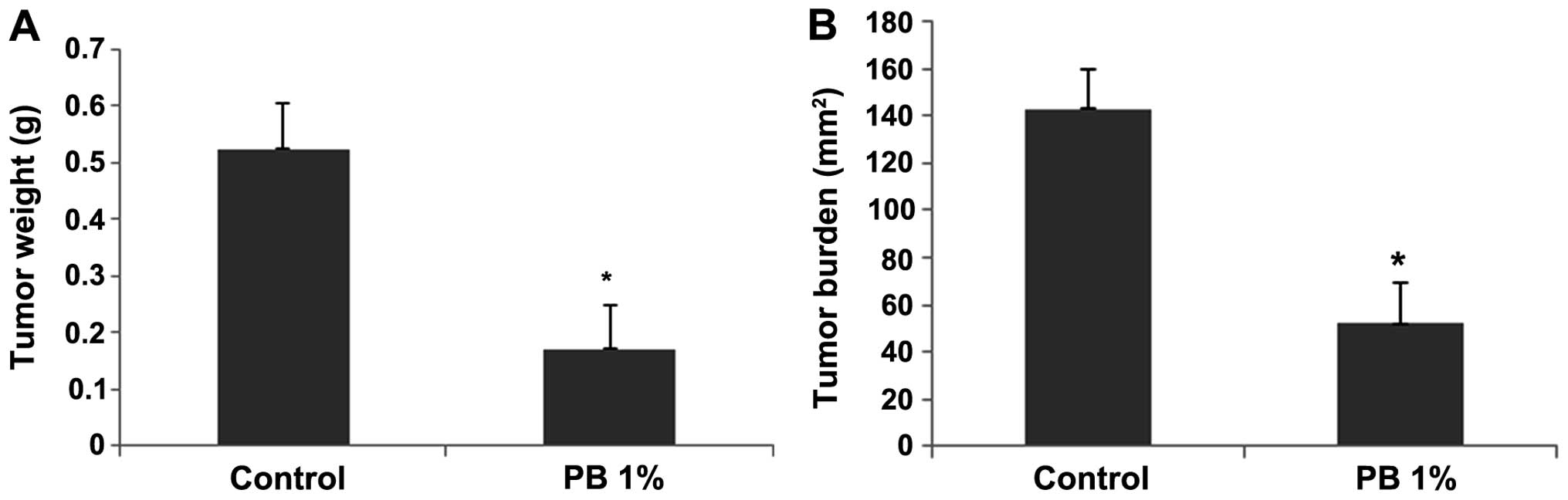
NM strongly inhibited the growth of OHSU-974
xenografts in nude mice. Mean tumor weight was inhibited by 67.6%
(p<0.0001) with PB 1% dietary supplementation, as shown in
Fig. 1A, and tumor burden was
inhibited by 63.6% (p<0.0001), as shown in Fig. 1B.
Histology
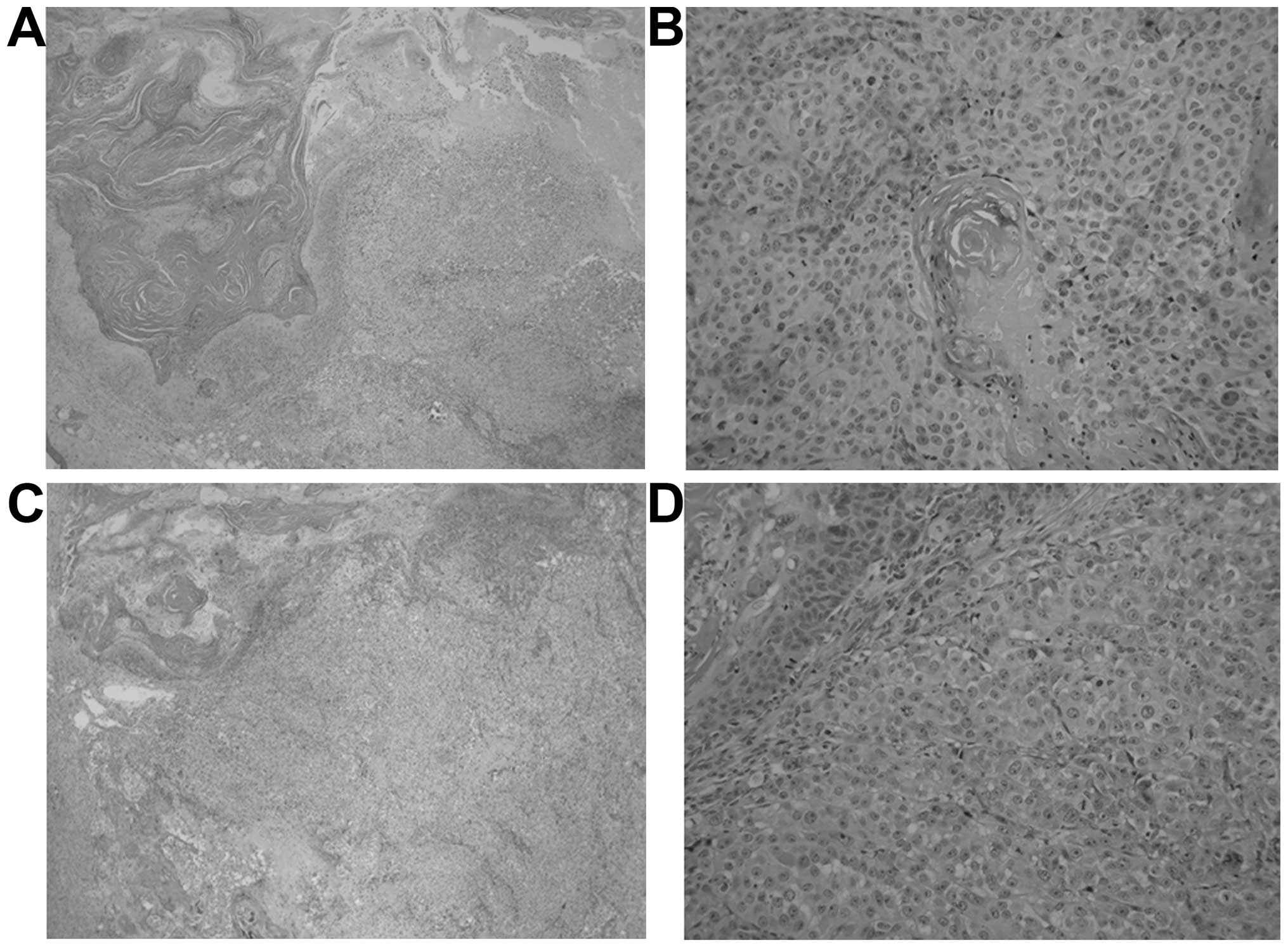
The tumors from both groups were irregularly round,
ulcerated, skin subcutaneous masses, consistent with squamous cell
carcinoma. Tumors from control and PB-supplemented mice were
similar morphologically, but the tumors from supplemented mice were
significantly smaller in size (Fig.
2).
In vitro
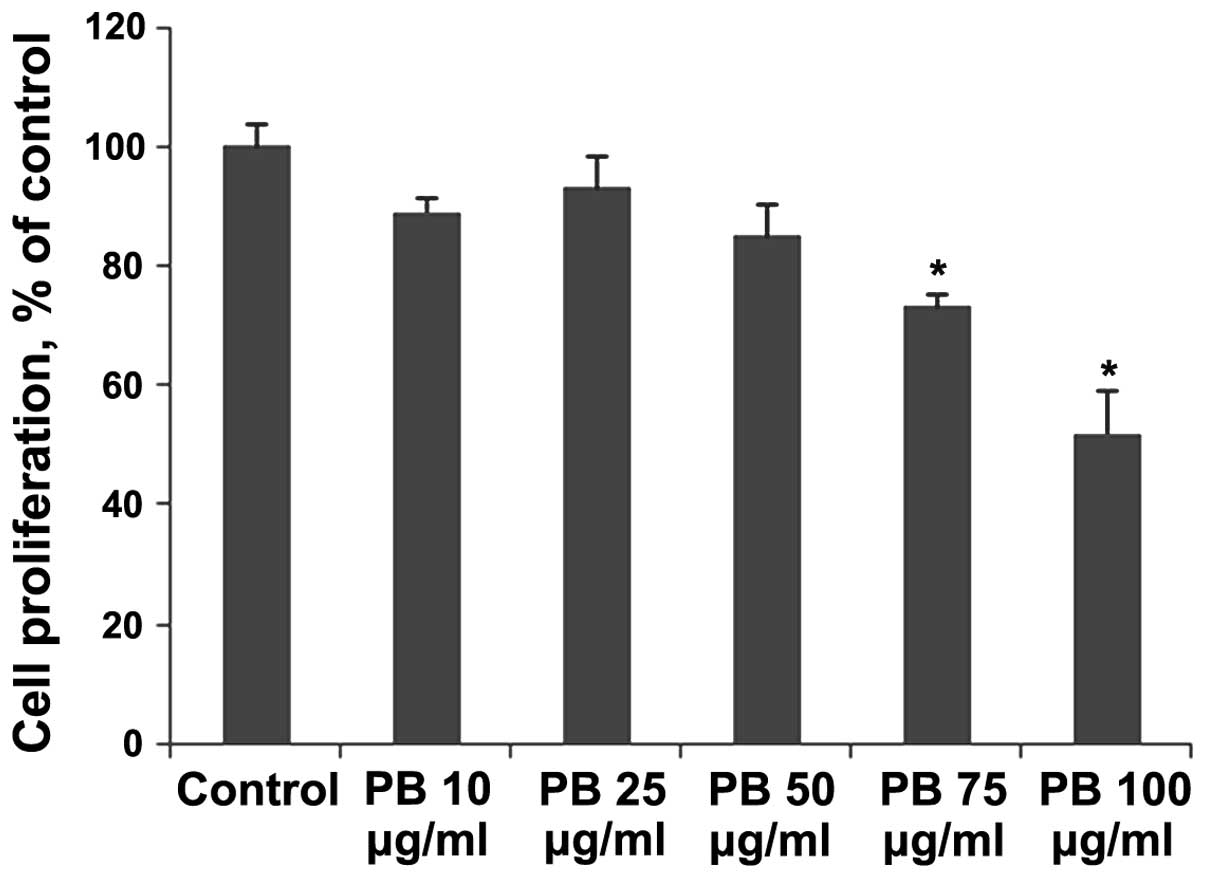
Cytotoxicity
PB exhibited dose-dependent toxicity to HNSCC
OHSU-974 cells in vitro with 27% (p=0.0003) at 75 μg/ml and
48% (p=0.0004) at 100 μg/ml, compared to the control, as shown in
Fig. 3.
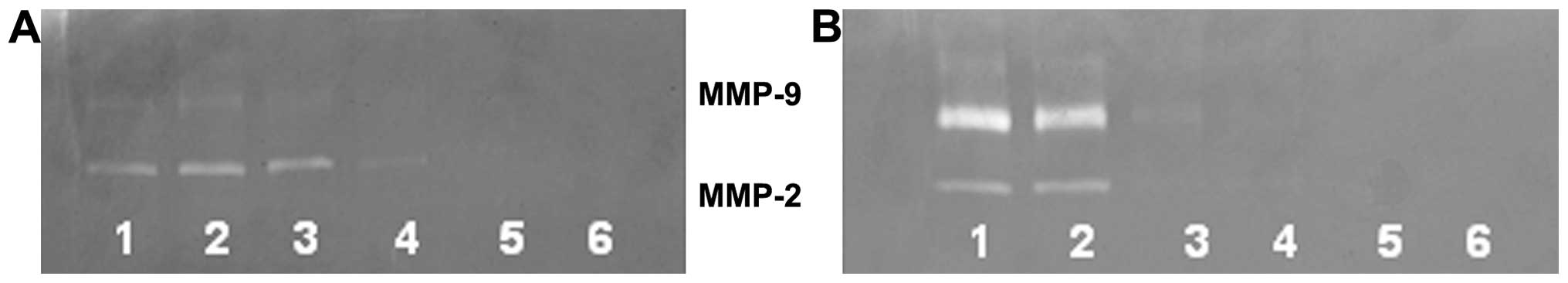
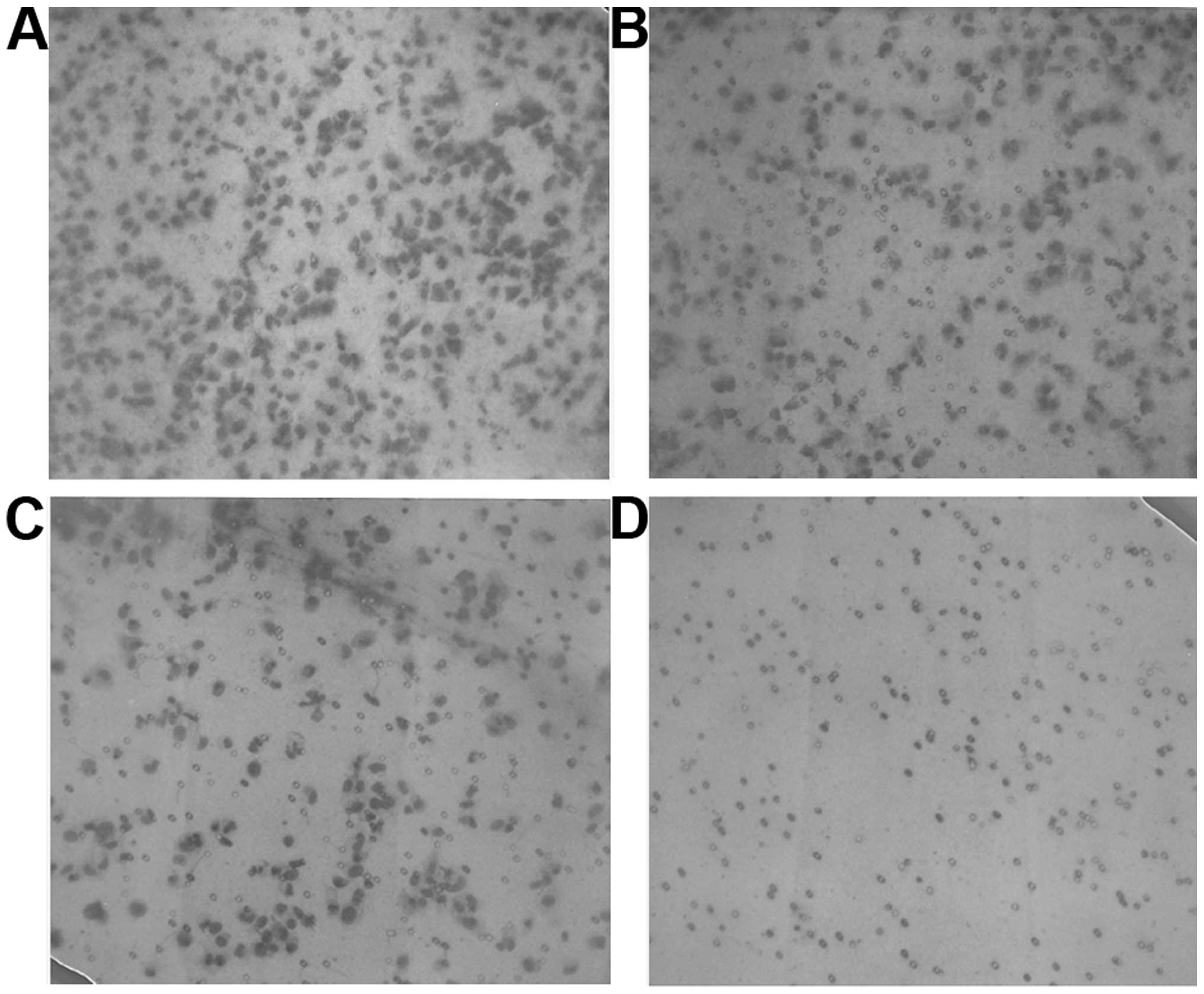
Gelatinase zymography
Zymography demonstrated MMP-2 and MMP-9 secretion by
normal and enhanced MMP-9 with PMA-treated OHSU-947 cells. PB
inhibited secretion of both MMPs in a dose-dependent fashion with
virtual total inhibition of MMP-9 and MMP-2 at 50 μg/ml, as shown
in Fig. 4A and B.
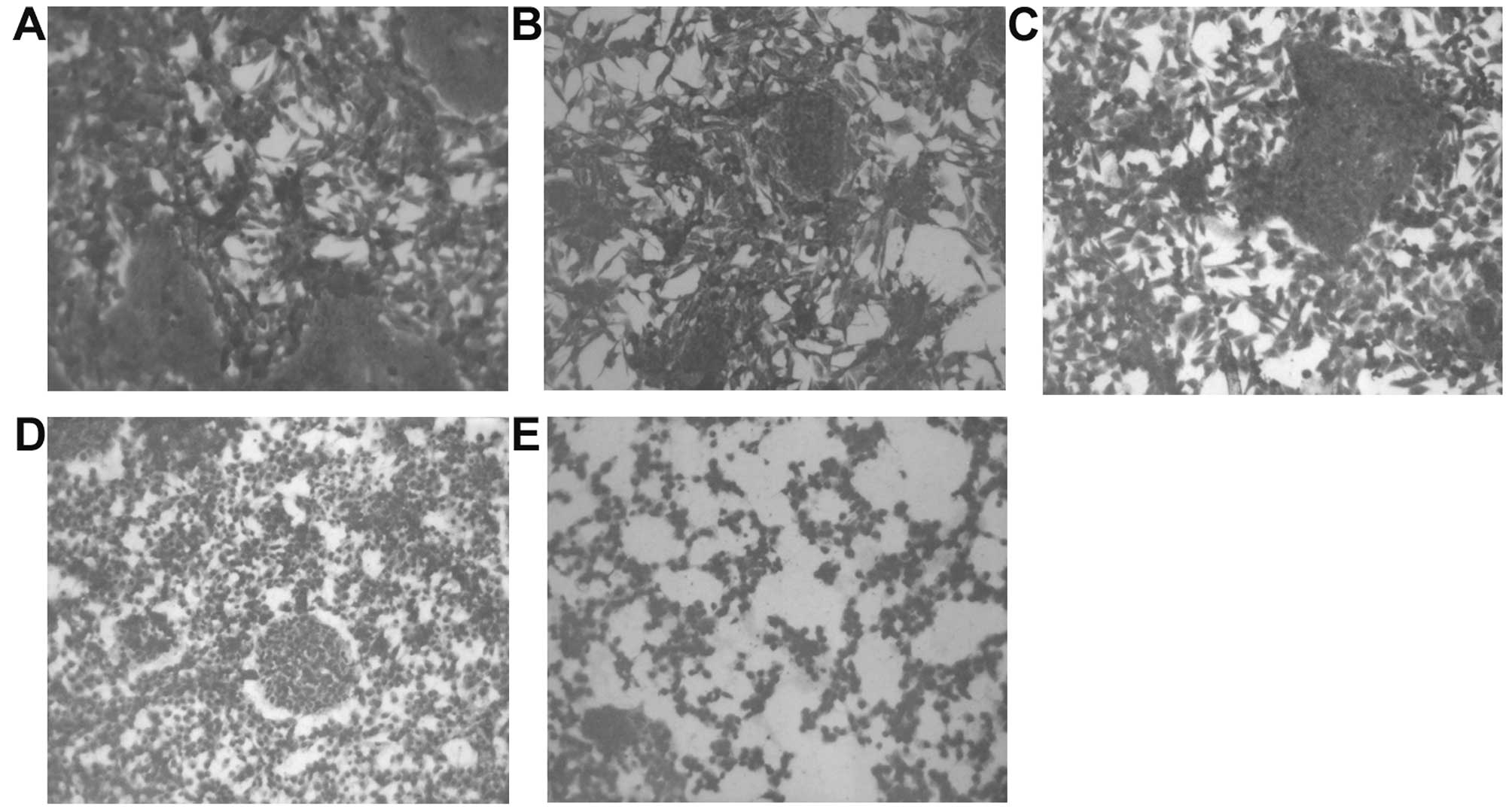
Matrigel invasion
PB significantly inhibited OHSU-974 cell invasion
through Matrigel in a dose-dependent manner, with total block at 50
μg/ml, as shown in Fig. 5
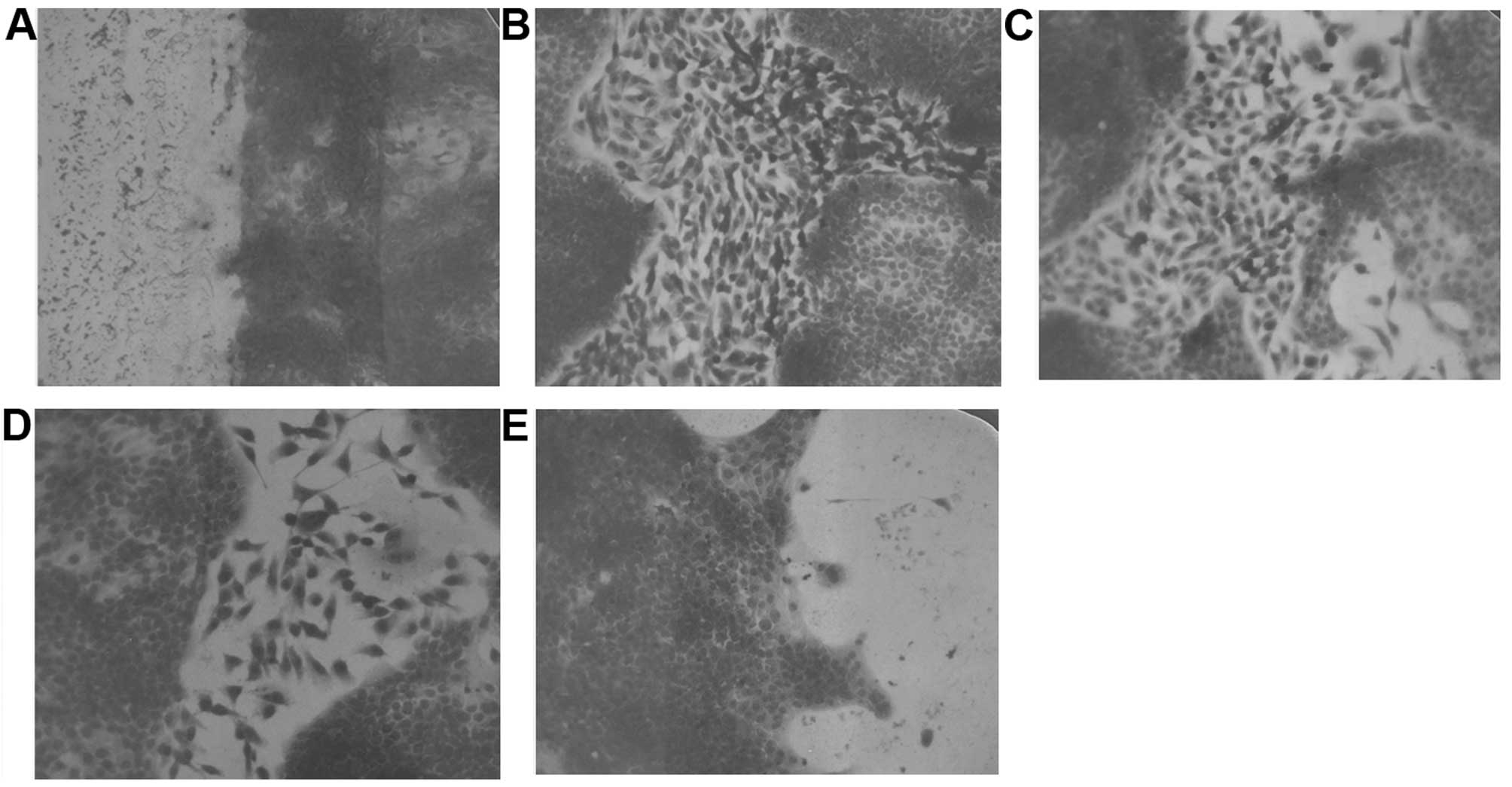
Cell migration: scratch test
PB reduced cell migration in a dose-dependent
manner, with complete block of OHSU-974 cells at 50 μg/ml.
Photomicrographs of the results for the scratch tests for OHSU-974
cells are shown in Fig. 6.
Morphology
H&E staining showed no morphological changes
below PB 50 μg/ml, as shown in Fig.
7.
Discussion
The results of the in vivo study of human
HNSCC xenografts in immune impaired (athymic) nude mice
demonstrated significant suppression of HNSCC OHSU-974 tumor growth
(67.6% inhibition of mean tumor weight and 63.6% inhibition of mean
tumor burden with PB 1% dietary supplementation). Results from the
cellular proliferation study support the in vivo findings,
as PB showed increased toxicity in OHSU-974 cells in a
dose-dependent manner, with 48% inhibition of cell growth in cells
exposed to 100 μg/ml PB.
Invasion of host tissues is dependent on tumor cell
adhesion, cell migration, and proteolytic degradation of the ECM by
MMPs (6). MMPs, especially MMP-2
and MMP-9 are prognostic for survival and metastatic potential in
head and neck squamous carcinomas. Kawamata et al (7) observed increased activity of
pro-MMP-9 and active MMP-2 in cancer cell nests in human oral
squamous cell carcinoma compared with normal surrounding gingival
tissue and significantly higher MMP-2 activity in metastatic cancer
cell nests. Patel et al (8)
reported significant elevation of latent, active and total forms of
MMP-2 and MMP-9 in malignant tissue compared with adjacent normal
tissues in oral cancer patients. In addition, MMP-2 was correlated
with lymph node metastatic development (8).
In examining a group of patients with early stage
oral squamous cell carcinoma, Katayama et al (9) found that patients who developed
regional lymph node and/or distant metastasis showed significantly
increased MMP-9 and TIMP-2 expression than patients without any
tumor metastasis; expression of MMP-9 and TIMP-2 correlated with
the worst cause-specific survival. Reidel et al (10) found that MMP-9 expression in
patients with HNSCC correlated with poor survival of patients, as
well as high VEGF expression and higher mean vessel density than
MMP-9-negative tumors, suggesting MMP-9 functions as a regulator of
tumor angiogenesis supporting endothelial cell invasion in human
head and neck cancer. Kuhara et al (11) demonstrated a significant decrease
in ECM staining (indicating loss of ECM) in invasive and metastatic
cases of oral squamous cell carcinoma with increased expression of
MMP-1, MMP-2 and MMP-9.
The results from our in vitro study of HNSCC
OSH-947 cells demonstrated potent, significant suppression of
invasive parameters by the phytonutrient mixture. PB inhibited
MMP-2 and MMP-9 secretion with total block at 50 μg/ml and 100%
inhibition of invasion of cells through Matrigel at 50 μg/ml.
Migration of cells using scratch test showed total block at PB 50
μg/ml.
Use of natural health products to prevent, inhibit
and reverse carcinogenesis is gaining increasing importance, since
scientific evidence shows that high consumption of fruits and
vegetables are associated with reduced risk of various cancers
(4,5). PB was formulated by defining critical
physiological targets in cancer progression and metastasis.
Curcumin (a derivative of turmeric), used for centuries to treat
inflammatory conditions, has been reported to affect multiple
targets in many cancers: leukemia, lymphoma, gastrointestinal
cancers, genitourinary cancers, breast cancer, ovarian cancer, head
and neck squamous cell carcinoma, lung cancer, melanoma,
neurological cancers and sarcomas (12). Curcumin has been reported to down
regulate cancer cell proliferation, invasion (MMP-9 and adhesion
molecules), angiogenesis (VEGF) and metastasis and to induce
apoptosis (12). Anticarcinogenic
properties of cruciferex (derived from cabbage, cauliflower, carrot
and broccoli) include detoxification and excretion of carcinogens,
protection against oxidative stress, inhibition of cancer cell
proliferation and increase in apoptosis resulting in inhibition of
tumor growth (13). Green tea
extract has been shown to modulate cancer cell growth, metastasis,
angiogenesis, and other aspects of cancer progression (14–19).
Resveratrol has been shown to inhibit cellular
events associated with tumor initiation, promotion and progression,
partially attributable to its antioxidant activities and inhibition
of Cox-2 and inhibition of protein kinase C, a key mediator of
tumor promotion stage of carcinogenesis (20). Among its anticarcinogenic effects,
resveratrol inhibits cell proliferation and induces apoptosis
(20). Bishayee et al
reported that resveratrol affects carcinogenesis by modulating
signal transduction pathways that control cell division and growth,
apoptosis, inflammation, angiogenesis and metastasis (21). Cancer preventive effects of
quercetin include induction of cell cycle arrest, apoptosis and
antioxidant functions (22).
Induction of apoptosis by quercetin in cancer cells during
different cell cycle stages without affecting normal cells has been
documented in various cancers in vivo and in vitro
(22).
In conclusion, current treatment methods for Fanconi
anemia-associated cancers are generally ineffective and especially
toxic to these patients. Thus, there is a need for development of
effective therapeutic agents for these cancers with minimal
toxicity. Our studies demonstrated that the mixture of the
non-toxic components of PB significantly inhibited the growth and
tumor burden of FA HNSCC cell line OHSU-974 in vivo. In
addition, invasive parameters, such as OHSU-974 cell line MMP-2 and
-9 secretion and invasion were significantly inhibited by PB in
vitro. These findings suggest potential of PB in treatment of
FA HNSCC.
Acknowledgements
This study was funded by Dr Rath Health Foundation
(Santa Clara, CA, USA), a non-profit organization. Consulting
pathologist Alexander de Paoli, DVM, PhD, IDEXX Reference
Laboratories provided histopathology slides of HNSCC OHSU-974
tumors.
References
|
1
|
Alter BP, Greene MH, Velazquez I and
Rosenberg PS: Cancer in Fanconi anemia. Blood. 101:2072–2073. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kutler DI, Auerbach AD, Satagopan J,
Giampietro PF, Batish SD, Huvos AG, Goberdhan A, Shah JP and Singh
B: High incidence of head and neck squamous cell carcinoma in
patients with Fanconi anemia. Arch Otolaryngol Head Neck Surg.
129:106–112. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Singh B: Head and neck squamous carcinoma
in Fanconi anemia patients. Fanconi Anemia: Guidelines for
Diagnosis and Management. Eiler ME, Fohnmayer D, Forhnmayer L,
Larsen K and Owen J: 3rd edition. Fanconi Anemia Research Fund,
Inc; 2008
|
|
4
|
Adlercreutz H: Western diet and Western
diseases: Some hormonal and biochemical mechanisms and
associations. Scand J Clin Lab Invest (Suppl). 201:3–23. 1990.
View Article : Google Scholar
|
|
5
|
Miller AB: Diet and cancer. A review Acta
Oncol. 29:87–95. 1990. View Article : Google Scholar
|
|
6
|
Duffy MJ: The role of proteolytic enzymes
in cancer invasion and metastasis. Clin Exp Metastasis. 10:145–155.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kawamata H, Uchida D, Hamano H,
Kimura-Yanagawa T, Nakashiro KI, Hino S, Omotehara F, Yoshida H and
Sato M: Active-MMP2 in cancer cell nests of oral cancer patients:
Correlation with lymph node metastasis. Int J Oncol. 13:699–704.
1998.PubMed/NCBI
|
|
8
|
Patel BP, Shah SV, Shukla SN, Shah PM and
Patel PS: Clinical significance of MMP-2 and MMP-9 in patients with
oral cancer. Head Neck. 29:564–572. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Katayama A, Bandoh N, Kishibe K, Takahara
M, Ogino T, Nonaka S and Harabuchi Y: Expressions of matrix
metal-loproteinases in early-stage oral squamous cell carcinoma as
predictive indicators for tumor metastases and prognosis. Clin
Cancer Res. 10:634–640. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Riedel F, Götte K, Schwalb J, Bergler W
and Hörmann K: Expression of 92-kDa type IV collagenase correlates
with angiogenic markers and poor survival in head and neck squamous
cell carcinoma. Int J Oncol. 17:1099–1105. 2000.PubMed/NCBI
|
|
11
|
Kurahara S, Shinohara M, Ikebe T, Nakamura
S, Beppu M, Hiraki A, Takeuchi H and Shirasuna K: Expression of
MMPS, MT-MMP, and TIMPs in squamous cell carcinoma of the oral
cavity: Correlations with tumor invasion and metastasis. Head Neck.
21:627–638. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Anand P, Sundaram C, Jhurani S,
Kunnumakkara AB and Aggarwal BB: Curcumin and cancer: An ‘old-age’
disease with an ‘age-old’ solution. Cancer Lett. 267:133–164. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Keck AS and Finley JW: Cruciferous
vegetables: Cancer protective mechanisms of glucosinolate
hydrolysis products and selenium. Integr Cancer Ther. 3:5–12. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Valcic S, Timmermann BN, Alberts DS,
Wächter GA, Krutzsch M, Wymer J and Guillén JM: Inhibitory effect
of six green tea catechins and caffeine on the growth of four
selected human tumor cell lines. Anticancer Drugs. 7:461–468. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mukhtar H and Ahmad N: Tea polyphenols:
Prevention of cancer and optimizing health. Am J Clin Nutr.
71(Suppl 6): S1698–S1702; discussion S1703–S1704. 2000.
|
|
16
|
Yang GY, Liao J, Kim K, Yurkow EJ and Yang
CS: Inhibition of growth and induction of apoptosis in human cancer
cell lines by tea polyphenols. Carcinogenesis. 19:611–616. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Taniguchi S, Fujiki H, Kobayashi H, Go H,
Miyado K, Sadano H and Shimokawa R: Effect of (-)-epigallocatechin
gallate, the main constituent of green tea, on lung metastasis with
mouse B16 melanoma cell lines. Cancer Lett. 65:51–54. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hara Y: Green tea: Health Benefits and
Applications. Marcel Dekker, Inc; New York, Basel: 2001, View Article : Google Scholar
|
|
19
|
Harakeh S, Abu-El-Ardat K, Diab-Assaf M,
Niedzwiecki A, El-Sabban M and Rath M: Epigallocatechin-3-gallate
induces apoptosis and cell cycle arrest in HTLV-1-positive and
-negative leukemia cells. Med Oncol. 25:30–39. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cal C, Garban H, Jazirehi A, Yeh C,
Mizutani Y and Bonavida B: Resveratrol and cancer: Chemoprevention,
apoptosis, and chemo-immunosensitizing activities. Curr Med Chem
Anticancer Agents. 3:77–93. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bishayee A: Cancer prevention and
treatment with resveratrol: From rodent studies to clinical trials.
Cancer Prev Res (Phila). 2:409–418. 2009. View Article : Google Scholar
|
|
22
|
Gibellini L, Pinti M, Nasi M, Montagna JP,
De Biasi S, Roat E, Bertoncelli L, Cooper EL and Cossarizza A:
Quercetin and cancer chemoprevention. Evid Based Complement
Alternat Med. 2011:5913562011. View Article : Google Scholar : PubMed/NCBI
|