Introduction
Tumor angiogenesis is essential for solid tumor
growth and the key stages in the development of ovarian cancer are
the passage of the carcinoma cells through the basic membrane and
infiltration of adjacent tissues. Therefore, angiogenesis is an
important process for creation of new blood and lymphatic vessels,
which sustain the growth of the tumor (1). In neo-vascularization, it is known
that VEGF-R1 and VEGF-R2 (vascular endothelial growth factor
receptors) act as receptors for VEGF-A (vascular endothelial growth
factor) (2). Although VEGF-A can
bind both VEGF-R1 and VEGF-R2, most data suggest that binding of
VEGF-A to VEGF-R2 accounts for the majority of the stimulatory
signal for the angiogenesis observed in vivo. However, the
discovery of dozens of other pro-antigenic cytokines and their
cognate receptors have confirmed the complexity of the neo-vascular
process (3). Thus, the p53 tumor
suppressor gene has been implicated to play a central role in the
regulation of angiogenesis, with genetic inactivation of p53
resulting in upregulation of pro-angiogenic factors and
downregulation of angiogenic inhibitors (4). VEGF-A contains p53 response elements
and is involved in formation and inhibition of new blood vessels
(5).
Hypoxia is an important regulatory stimulus for
tumor growth and for a variety of different biological processes,
and tumor hypoxia also induces an ‘antigenic switch’, a change from
an avascular to a vascular tumor, which is critical for the growth
of solid tumors. Tumor hypoxia also plays an important role in
malignant progression, radio-resistance, and chemo-therapeutic drug
resistance and can further lead to changes in cell cycle arrest,
differentiation, angiogenesis, carcinogenesis and apoptosis
(6). p53 is a central component of
most cellular stress responses, including hypoxic stress. Despite
potential cross-talk mechanisms between p53 and VEGF-A, it remains
unclear how these two key cancer-signaling pathways could
functionally interact (7). The p53
status (detected by IHC or mutational analysis) in a
meta-regression analysis showed that the FIGO stage may influence
the of outcome predictive value of p53 and the prognostic
significance of p53 seems also to be more restricted to low stage
tumors (8,9). Studies on the importance of VEGF in
epithelial ovarian cancer and its relationship to prognosis have
shown inconsistent results (10,11).
In the same manner, results from studies concerning the prognostic
value of VEGF-R1 and VEGF-R2 for ovarian cancer are diverse
(1,2).
The aim of the present study was to investigate the
role of the angiogenesis regulator proteins VEGF-A, VEGF-R1 and
VEGF-R2 and their relation to the tumor suppressor p53 in ovarian
carcinoma in patients limited to FIGO-stages I–II.
Materials and methods
Study population
A total of 140 consecutive patients with FIGO-stage
I–II epithelial ovarian cancer, who underwent primary surgery and
post-surgical chemotherapy in the Uppsala-Örebro Medical Region
during the 5-year period from January 1, 2000 to December 31, 2004,
were entered into this study. All samples were collected with the
patient's informed consent and were in compliance with the Helsinki
Declaration (12) and used in
accordance with the Swedish Biobank Legislation and Ethical Review
Act [approval by Uppsala Ethical Review Board, decision ref.
(UPS-03-477)].
In total, 131 patients out of 140 patients, who
accepted to participate in the present study were included and
there were 131 available tumors for analysis of p53. For VEGF-R1
there were 129 and for VEGF-R2 130 available tumors, respectively.
For analysis of VEGF-A, there were 98 available tumors (lower
numbers because of technical issues in the staining process). The
primary surgery was performed at nine different surgical
gynecological departments and the staging procedure was done at the
time of primary surgery. Modified surgical staging according to the
EORTC surgical staging categories in early ovarian cancer (13) was undertaken in 34 (26%) out of the
131 cases, and in the remaining 77 (74%) patients surgical staging
was regarded as minimal or inadequate according to the same
guidelines. All patients underwent adjuvant chemotherapy 4–6 weeks
after primary surgery. In the total series 105 out of the 131 (80%)
of patients received paclitaxel 175 mg/m2 and
carboplatin (AUC=5) at 3-week intervals usually in four courses.
The remaining 26 patients were treated with single-drug carboplatin
in 4–6 courses.
Patient characteristics, e.g. age, BMI in two
groups, performance status (WHO), FIGO-stage and the type of tumor
(type I/II) are demonstrated in Table
I. No patients were lost from follow-up and the mean follow-up
time was 65 months (range, 5–110 months). Survival was taken as
date of confirmed histological diagnosis after primary surgery to
date of recurrence or last visit.
 | Table IThe patient characteristics. |
Table I
The patient characteristics.
| Characteristics
(n=131) | n (%) |
|---|
| Age (median) 59.0
years (range, 25–84) |
| BMI |
| BMI ≤25 | 69 (54.8) |
| BMI >25 | 57 (45.2) |
| WHO performance
status |
| 0 | 37 (28.2) |
| 1 | 66 (50.4) |
| 2 | 21 (16.0) |
| 3 | 6 (4.6) |
| FIGO-stage |
| IA | 39 (29.7) |
| IB | 6 (4.6) |
| IC | 66 (50.4) |
| II | 20 (15.3) |
| Types of ovarian
tumorsa |
| Type I | 79 (65.8) |
| Low-grade (G1)
serous | 14 |
| Mucinous
(G1+G2+G3) | 20 |
| Low-grade
endometrioid (G1+G2) | 29 |
| Clear cell | 16 |
| Type II | 52 (34.2) |
| High-grade
(G2+G3) serous | 37 |
| High-grade (G3)
endometrioid | 13 |
| Anaplastic | 2 |
Sampling and tissue microarray
construction of ovarian cancer tissue
Tissue samples of the ovarian cancers were obtained
at the primary surgery. The tissue microarrays were constructed as
previously described (14). In
brief, tumor tissues were embedded in paraffin and 5 μm sections
stained with hematoxylin-eosin (H&E) were obtained to select
representative areas for biopsies. Core tissue biopsy specimens
(diameter 0.6 mm) were taken from these areas of individual donor
paraffin blocks and precisely arrayed into a new recipient paraffin
block with a custom-built instrument. Tissue core specimens from
131 ovarian carcinomas were arranged in three recipient paraffin
blocks. Two core biopsies were obtained from each specimen. The
presence of tumour tissue on the arrayed samples was verified by
hematoxylin-eosin-stained section. The specimens were then
reviewed, classified and graded by a single pathologist.
Immunohistochemistry and
interpretation
Sections (5 μm) were cut from each block on coated
slides and dried overnight at 37°C. The sections were pre-treated
by heath-induced epitope retrieval in target-retrieval solution
(Dako), pH 6.0 or EDTA buffer pH 9.0, for 7 + 7 min in microwave
oven (99°C). Blocking with peroxidase was performed for 5 min. The
slides were counterstained with hematoxylin for 2 min. The
procedure was performed in a TechMate 500 automated machine (LSAB
detection kit; Dako ChemMate). The following monoclonal primary
antibodies were used: DO-7, directed against p53 protein (dilution
1:1,000; Dako, Glostrup, Denmark), the IgG antibody (polyclonal
rabbit) to the VEGF-A protein (Dako). For VEGF-R1 the polyclonal
rabbit antibody Flt-1 was used as primary antibody and for VEGF-R2,
the polyclonal mouse antibody Flk-1 was used as primary antibody
(both from Santa Cruz Biotechnology). The work of tissue-microarray
construction was undertaken at the Department of Pathology, the
University Hospital MAS in Malmö in South-Sweden, but the
immunohistochemical analyses and interpretation were performed at
the Department of Pathology, Halmstad Medical Central Hospital. The
IHC stains were interpreted by two of the authors (I.S. and T.S.).
At the time of evaluation no information was available on the
specific diagnosis and prognosis for the individual cases. A
semi-quantitative analysis (15)
was used and the stains were graded as negative, +, ++ and +++ for
p53, VEGF-A, VEGF-R1 and VEGF-R2, and all those markers were
dichotomized into negative and positive (+, ++ and +++) cases
(16). The staining for p53 was
considered to be positive when there was a strong, granular
staining of the nuclei of the majority of tumor cells. Positive
staining for VEGF-A, VEGF-R1 and VEGF-R2 were confined to the
membrane and cytoplasm of the tumor cells.
Statistical analyses
The Pearson's χ2 was used for testing
proportional differences in univariate analyses. The survival
curves were generated by using the Kaplan-Meier technique and
differences between these curves were tested by the log-rank test.
All tests were two-sided and the level of statistical significance
was P≤0.05. The Statistica 12.5 (StatSoftTM) statistical
package for personal computers was used for the analyses. For
multivariate analyses the logistic regression model was used with
recurrence as the endpoint and Cox regression model was used with
disease-free survival (DFS) as the endpoint.
Results
Background characteristics
Patient characteristics, including age, BMI
(dichotomized), performance status of the patients (WHO),
FIGO-stage and types of ovarian tumors (type I and type II) are
demonstrated in Table I. Primary
cure was achieved in all 131 patients. The total number of
recurrences in the complete series was 34 out of 131 (26%), and 22
of these patients (67%) died due to disease. In the complete
series, recurrent disease was significantly associated with FIGO
sub-stages (P=0.0005), FIGO-grade (P=0.030), adequate surgical
staging (P=0.033) and residual disease (P=0.001). In the complete
series the 5-year disease-free survival rate was 68%, the
disease-specific survival rate 76% and the overall survival rate
was 71%.
Expression of angiogenesis regulator
proteins and relation to clinicopathological factors and
survival
In the present study, the status of protein
expression (positivity/negativity) for the angiogenesis regulators,
VEGF-A, VEGF-R1 and VEGF-R2 (Table
II) are compared with the clinical and pathological
factors.
 | Table IIStatus of protein expression in
tumors of the VEGF-A, VEGF-R1 and VEGF-R2 vs. clinical and
pathological features (N=131). |
Table II
Status of protein expression in
tumors of the VEGF-A, VEGF-R1 and VEGF-R2 vs. clinical and
pathological features (N=131).
| Expression | VEGF-A+
n (%) | VEGF-A−
n (%) | VEGF-R1+
n (%) | VEGF-R1−
n (%) | VEGF-R2+
n (%) | VEGF-R2−
n (%) |
|---|
| Total | 60 (61) | 38 (39) | 18 (19) | 111 (86) | 100 (77) | 30 (23) |
| Histopathology |
| Serous | 28 (47) | 13 (34) | 6 (33) | 46 (41) | 43 (43) | 9 (30) |
| Non-serous | 32 (53) | 25 (66) | 12 (67) | 65 (59) | 57 (57) | 21 (70) |
| P-value
(χ2) | 0.223 | 0.515 | 0.202 |
| Tumor grade |
| G1+G2 | 34 (57) | 28 (74) | 7 (39) | 67 (60) | 56 (56) | 19 (63) |
| G3 | 26 (43) | 10 (26) | 11 (61) | 44 (40) | 44 (44) | 11 (37) |
| P-value
(χ2) | 0.088 | 0.087 | 0.476 |
| Type of tumors |
| Type I | 31 (52) | 28 (74) | 13 (72) | 64 (58) | 54 (54) | 24 (80) |
| Type II | 29 (48) | 10 (26) | 5 (28) | 47 (42) | 46 (46) | 6 (20) |
| P-value
(χ2) | 0.030 | 0.242 | 0.011 |
| FIGO-stage |
| IA-IB | 20 (33) | 16 (42) | 8 (44) | 37 (33) | 34 (34) | 11 (37) |
| IC | 31 (52) | 19 (50) | 8 (44) | 57 (52) | 49 (49) | 16 (53) |
| II | 9 (15) | 3 (8) | 2 (12) | 17 (15) | 17 (17) | 3 (10) |
| P-value
(χ2) | 0.482 | 0.644 | 0.647 |
| Recurrent
disease |
| Without | 44 (73) | 28 (74) | 17 (94) | 78 (70) | 78 (78) | 18 (60) |
| With | 16 (27) | 10 (26) | 1 (56) | 33 (30) | 22 (22) | 12 (40) |
| P-value
(χ2) | 0.969 | 0.030 | 0.049 |
VEGF-A staining was confined to the membrane and
cytoplasm. Positivity for VEGF-A was observed in 60 (61%) out of 98
available tumors for this marker. There were no differences in mean
age between the groups of patients with VEGF-A positive and VEGF-A
negative tumors (61 vs. 59 years; P=0.333). As demonstrated in
Table II, the VEGF-A status was
not related to serous/non-serous histological subtype, tumor grade,
FIGO-sub-stages or recurrent disease. However, the status of VEGF-A
was statistically significantly (P=0.030) related to the type of
tumor. Thus, positive staining for VEGF-A was seen more frequently
in type II tumors than in type I tumors (P=0.030). The VEGF-A
status alone was not related to survival.
VEGF-R1 staining was confined to the membrane and
cytoplasm and positivity for VEGF-R1 was observed in 18 (19%) out
of 129 available tumors. There were no differences in mean age
between the groups of patients with VEGF-R1 positive and VEGF-R1
negative tumors (59 years for both of the groups). As shown in
Table II, the VEGF-R1 status was
not associated with serous/non-serous tumors, tumor grade, tumor
type or FIGO-stage. However, the VEGF-R1 status was significantly
(P=0.030) associated with recurrent disease in univariate analysis.
Thus, negativity of VEGF-R1 was detected in tumors by 33 (97%) out
of the 34 patients with recurrent disease in the present study.
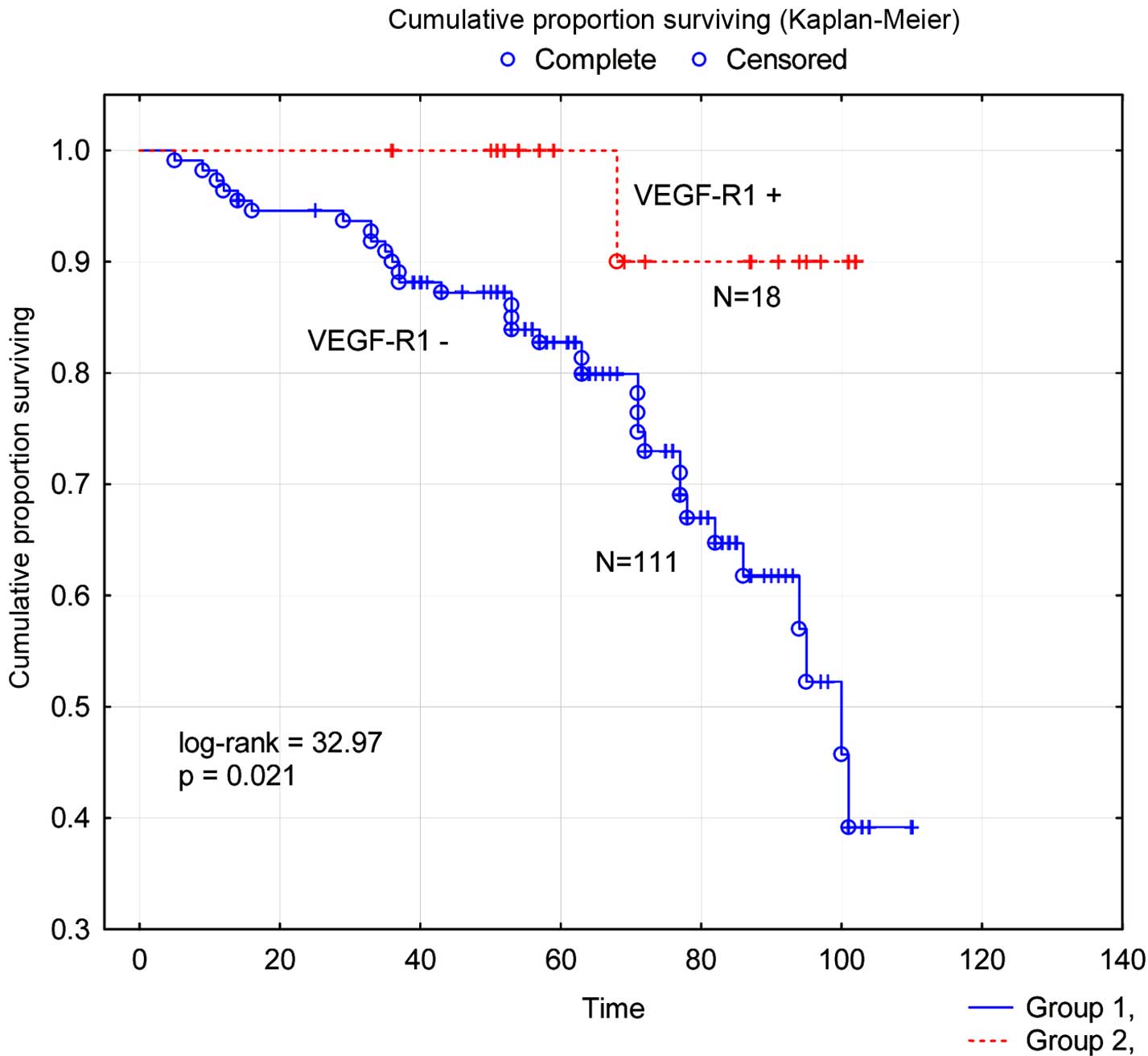
Survival analysis (Fig. 1) showed
significantly worse (P=0.021) disease-free survival for the
subgroup of patients with VEGF-R1 negative tumors compared to the
subgroup with VEGF-R1 positive tumors.
VEGF-R2 staining was confined to the membrane and
cytoplasm, and positivity for VEGF-R2 was observed in 100 (77%) out
of the 130 tumors. There were no differences in mean age between
the groups of patients with VEGF-R2-positive and VEGF-R2-negative
tumors (59 vs. 58 years; P=0.620). The VEGF-R2 status (Table II) was not associated with
serous/non-serous tumors, tumor grade or FIGO-stage. However, the
VEGF-R2 status was significantly (P=0.011) associated with type II
tumors. Furthermore, recurrent disease occurred more frequently
(P=0.049) in the subgroup of patients with VEGF-R2 negative tumors.
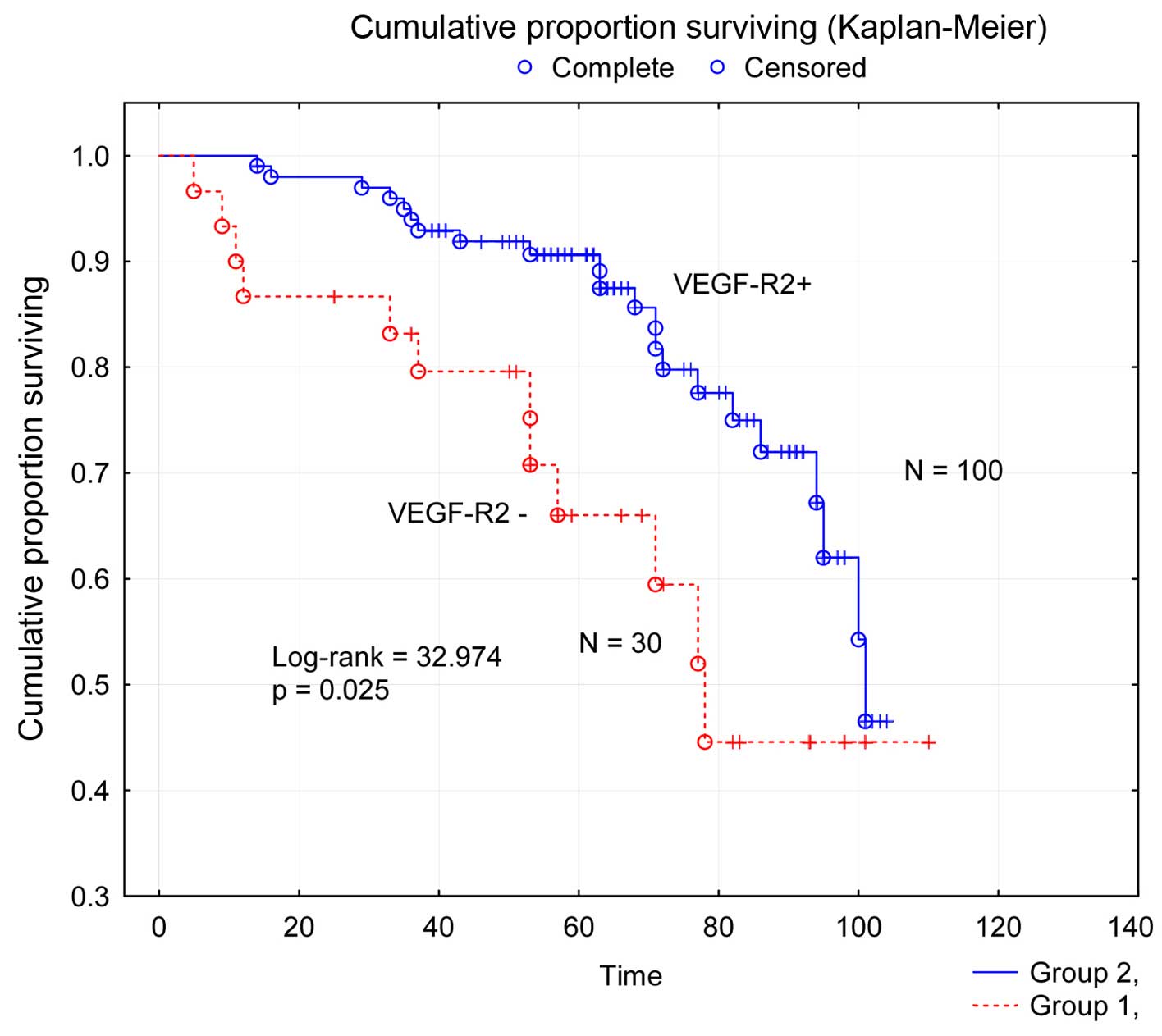
The survival analysis (Fig. 2)
showed significant worse (P=0.025) DFS for the subgroup of patients
with VEGF-R2 negative tumors compared to the subgroup with VEGF-R2
positive tumors. Thus, patients from the subgroup with VEGF-R2
negative tumors had a 5-year disease-free survival rate of 66%
compared to 90% in the other subgroup.
VEGF-A was strongly (P=0.001) related to VEGF-R2,
but 76 (70%) out of the 98 patients available for statistical
analysis had concomitant positivity for both VEGF-A and
VEGF-R2.
Results from statistical analysis of the combination
of VEGF-A and VEGF-R2 in four groups were undertaken for the same
variables as shown in Table II,
but no statistical significance was detected. Furthermore, no
relation between the status of VEGF-R1 and VEGF-A was found
(P=0.475). In separate analysis of patients with type I tumors
(n=79) and type II tumors (n=52), respectively, no statistically
significant difference for VEGF-A, VEGF-R1 and VEGF-R2 after age,
serous/non-serous histology, tumor grade or FIGO-sub-stages was
found. However, for the patients with type I tumors (n=78),
recurrent disease was strongly (P=0.008) related to VEGF-R2
negativity of tumors. In the same manner, survival analysis for the
patients with type II tumors (n=52), showed significantly worse
(log-rank=14.498; P=0.0003) DFS for the subgroup of patients with
VEGF-R2 negative tumors compared to the subgroup with VEGF-R2
positive tumors. For the subgroup of patients with type I tumors
and type II tumors, respectively, no relation was found between the
VEGF-A status of tumors and recurrent disease.
The relationship of p53 status to
angiogenesis regulators and association to clinicopathological
factors and survival
In a previous study (17) including the total series of
patients (n=131) results from IHC and interpretation for p53 have
already been presented. Positivity for p53 was found more
frequently in type II tumors (37%) compared to type I tumors (17%),
(P=0.015). Furthermore, recurrent disease (Table III) occurred more often (48%) in
the group of patients with p53 positive tumors compared with the
group of patients with p53 negative tumors (18%), (P=0.0006).
However, the p53 status alone was not related to VEGF-A (P=0.805),
VEGF-R1 (P=0.724) or VEGF-R2 (P=0.210) in the present study.
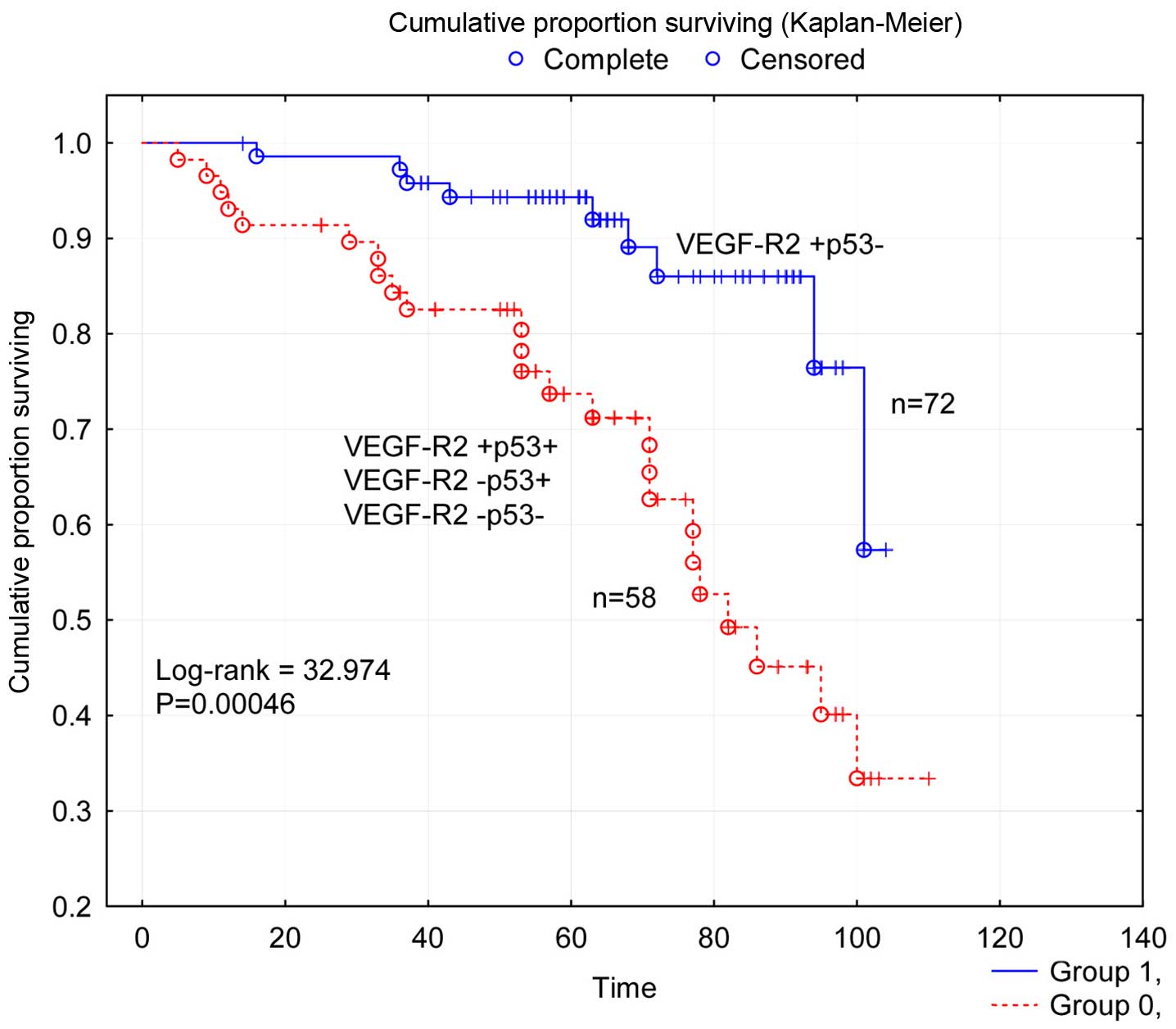
Survival analysis of the concomitant VEGF-R2 status and p53 status
in four groups was undertaken (χ2=14.360; P=0.00246) and
it was found that the subgroup of patients with concomitant
positivity for VEGF-R2 and negativity for p53 and
(VEGF-R2+p53−) of tumors had a favorable
survival compared to the other three subgroups. Therefore,
disease-free survival for patients in the first subgroup was
compared to survival for the other three collective subgroups a
further survival analysis (Fig. 3)
where the difference (log-rank=32.974; P=0.00046) was highly
significant.
 | Table IIIStatus of protein expression in
tumors of the p53,
p53−VEGF-R2+/othera
(p53+VEGF-R2+,
p53+VEGF-R2− and
p53−VEGF-R2−) and
p53+VEGF-A−/other
(p53+VEGF-A+,
p53−VEGF-A+ and
p53−VEGF-A−) vs. clinical and pathological
features (N=131). |
Table III
Status of protein expression in
tumors of the p53,
p53−VEGF-R2+/othera
(p53+VEGF-R2+,
p53+VEGF-R2− and
p53−VEGF-R2−) and
p53+VEGF-A−/other
(p53+VEGF-A+,
p53−VEGF-A+ and
p53−VEGF-A−) vs. clinical and pathological
features (N=131).
| Expression
p53+ | p53+ n
(%) | p53− n
(%) | VEGF-R2+
and p53− n (%) | Othera n
(%) | VEGF-A and
p53+ n (%) | Other n (%) |
|---|
| Total | 33 (25) | 98 (75) | 72 (56) | 58 (44) | 11 (12) | 87 (88) |
| Histopathology |
| Serous | 16 (48) | 37 (38) | 31 (43) | 32 (55) | 5 (45) | 36 (41) |
| Non-serous | 17 (52) | 61 (62) | 41 (57) | 26 (45) | 6 (55) | 51 (59) |
| P-value
(χ2) | 0.277 | 0.428 | 0.796 |
| Tumor grade |
| G1+G2 | 16 (48) | 60 (61) | 42 (58) | 32 (55) | 8 (73) | 54 (62) |
| G3 | 17 (52) | 38 (39) | 30 (42) | 26 (45) | 3 (27) | 33 (38) |
| P-value
(χ2) | 0.199 | 0.602 | 0.489 |
| Type of tumors |
| Type I | 14 (42) | 65 (66) | 42 (58) | 36 (62) | 7 (64) | 52 (60) |
| Type II | 19 (58) | 33 (34) | 30 (42) | 22 (38) | 4 (36) | 35 (40) |
| P-value
(χ2) | 0.015 | 0.665 | 0.805 |
| FIGO-stage |
| IA-IB | 9 (27) | 36 (37) | 25 (35) | 20 (35) | 3 (27) | 33 (38) |
| IC | 16 (48) | 50 (51) | 38 (53) | 27 (46) | 7 (64) | 43 (49) |
| II | 8 (24) | 12 (12) | 9 (12) | 11 (19) | 1 (9) | 11 (13) |
| P-value
(χ2) | 0.223 | 0.570 | 0.674 |
| Recurrent
disease |
| Without | 17 (52) | 80 (82) | 63 (87) | 33 (57) | 5 (45) | 67 (77) |
| With | 16 (48) | 18 (18) | 9 (13) | 25 (43) | 6 (55) | 20 (40) |
| P-value
(χ2) | 0.0006 | 0.00008 | 0.025 |
Concomitant VEGF-R2+p53− vs.
other combinations in one group were compared after the same
clinicopathological factors as before (Table III) without detection of any
significance with the exception of recurrent disease (P=0.00008).
It was found, that only 9 (15%) out of the 34 patients with
recurrent disease in this subgroup compared with 25 (85%) out of
the remaining 34 patients, who belonged to the other subgroup of
patients.
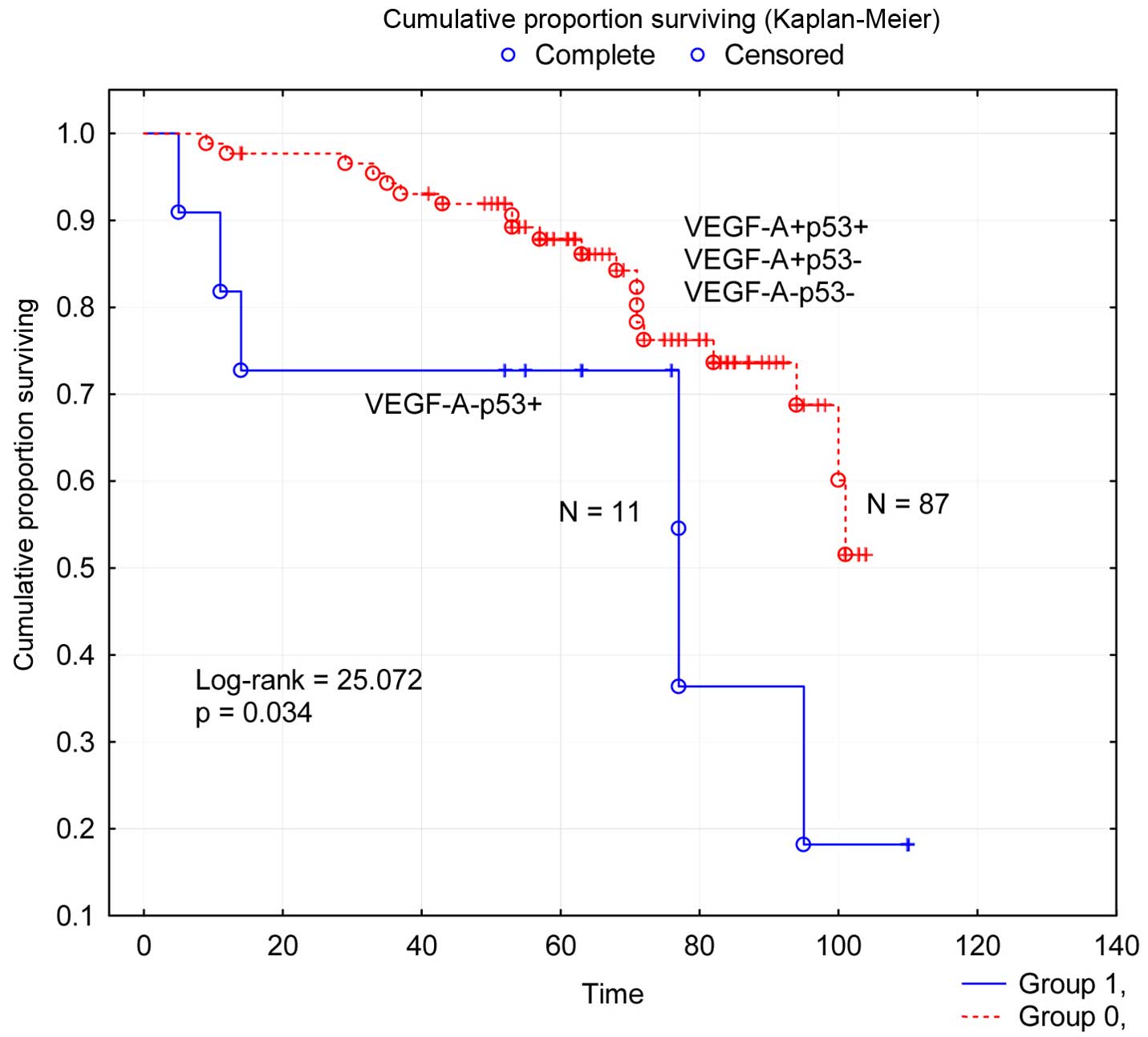
Furthermore, survival analysis according to
concomitant VEGF-A status and p53 status of tumors for available
patients (n=98) in four subgroups was undertaken and the results
reported (χ2, 8.240; P=0.0413). The subgroup of patients
with tumors of concomitant negativity for VEGF-A and positivity for
p53 (VEGF-A−p53+) (n=11) was the only one of
the four subgroups of patients, which had statistically different
survival (log-rank=25.072; P=0.034) from the other collective
subgroup (Fig. 4). The variable
VEGF-A−p53+ vs. other combinations in one
group was related to recurrent disease (P=0.025), but not to any
other of the clinicopathological factors (Table III).
Multivariate analysis
Results are shown for univariate and multivariate
Cox analysis with disease-free survival (DFS) as endpoint in
Table IVA and B. Only 98 out of
131 patients in the study could be included because of the limited
number of tumors with available information on the VEGF-A status.
In the first analysis (Table
IVA), FIGO-stage, VEGF-R2 status and p53 status, all were
significant and independent prognostic factors. For VEGF-A with a
HR=1.40, there was a trend for decreased survival for the subgroup
of patients with VEGF-A positive tumors (P=0.436). In Table IVB the variables VEGF-R2 and p53
were replaced with the new variable
VEGF-R2+p53−/three collectively in one group.
The value HR=0.237 (P=0.002) indicates that patients from that
subgroup had 86% reduced risk of dying of the disease. The variable
type (I/II) of tumor (a combination of histological subtype and
tumor grade shown in Table I), was
related to the variables VEGF-A, VEGF-R2 and p53 and therefore
could not be included in the multivariate analyses.
 | Table IVCox analysis. |
Table IV
Cox analysis.
| A, Cox analysis
(univariate and multivariate) with DFS as endpoint |
|---|
|
|---|
| Univariate
analysis | Multivariate
analysis | |
|---|
|
|
| |
|---|
| Variables | HR | 95% CI | HR | 95% CI | P-value |
|---|
| Age | 1.016 | 0.986–1.046 | 1.018 | 0.987–1.051 | 0.245 |
| Stage (I/II) | 3.318 | 1.655–6.654 | 3.834 | 1.847–7.954 | 0.0003 |
| VEGF-R1 | 0.148 | 0.019–1.116 | 0.206 | 0.027–1.568 | 0.127 |
| VEGF-R2 | 0.436 | 0.215–0.883 | 0.292 | 0.126–0.868 | 0.004 |
| VEGF-A | 0.943 | 0427–2.083 | 1.404 | 0.597–3.303 | 0.436 |
| p53 | 2.318 | 1.173–4.582 | 2.634 | 1.278–5.390 | 0.008 |
|
| B, Cox analysis
(univariate and multivariate) with DFS as endpoint |
|
| Univariate
analysis | Multivariate
analysis | |
|
|
| |
| Variables | HR | 95% CI | HR | 95% CI | P-value |
|
| Age | 1.016 | 0.986–1.046 | 1.029 | 0.994–1.066 | 0.100 |
| Stage (I/II) | 3.318 | 1.655–6.654 | 2.934 | 1.236–6.962 | 0.0145 |
| VEGF-R1 | 0.148 | 0.019–1.116 | 0.429 | 0.055–3.311 | 0.417 |
| VEGF-A | 0.943 | 0427–2.083 | 1.151 | 0.503–2.634 | 0.738 |
|
VEGF-R2+p53−/the
collective groupa | 0.278 | 0.130–0.599 | 0.237 | 0.094–0.600 | 0.002 |
In a multivariate logistic regression analysis with
recurrent disease as endpoint, FIGO-stage, VEGF-R2 status and p53
status all were independent predictive factors (Table VA). In a further logistic
regression analysis with recurrent disease as endpoint, only
FIGO-stage and status of VEGF-R2+p53− were
independent predictive factors (Table
VB).
 | Table VPredictive factors for recurrent
disease. |
Table V
Predictive factors for recurrent
disease.
| A, Predictive
factors for recurrent disease (univariate and multivariate logistic
regression analysis) |
|---|
|
|---|
| Univariate
analysis | Multivariate
analysis | |
|---|
|
|
| |
|---|
| Variables | OR | 95% CI | OR | 95% CI | P-value |
|---|
| Age | 1.013 | 0.981–1.047 | 1.017 | 0.978–1.057 | 0.395 |
| Stage (I/II) | 7.959 | 2.801–22.617 | 11.799 | 3.282–42.427 | 0.0001 |
| VEGF-R1 | 0.139 | 0.017–1.110 | 0.141 | 0.016–1.243 | 0.074 |
| VEGF-R2 | 0.423 | 0.175–1.018 | 0.229 | 0.078–0.668 | 0.006 |
| p53 | 4.179 | 1.477–12.071 | 4.709 | 1.662–13.339 | 0.003 |
|
| B, Predictive
factors for recurrent disease (univariate and multivariate logistic
regression analysis) |
|
| Univariate
analysis | Multivariate
analysis | |
|
|
| |
| Variables | OR | 95% CI | OR | 95% CI | P-value |
|
| Age | 1.013 | 0.981–1.047 | 1.014 | 0.975–1.055 | 0.468 |
| Stage (I/II) | 7.959 | 2.801–22.617 | 9.786 | 2.663–35.963 | 0.0005 |
| VEGF-R1 | 0.139 | 0.017–1.110 | 0.187 | 0.022–1.600 | 0.122 |
| Type (I/II) | 2.456 | 1.099–5.490 | 2.217 | 0.827–5.940 | 0.109 |
|
VEGF-R2+p53−/the
collective groupa | 0.278 | 0.130–0.599 | 0.169 | 0.061–0.468 | 0.005 |
Discussion
In the present study, positive staining for the
angiogenesis regulators VEGF-A and VEGF-R2 and the apoptosis
regulator p53 was found more frequently in type II tumors than in
type I tumors. Furthermore, positivity for VEGF-R1, VEGF-R2 and
concomitant positivity for VEGF-R2 and negativity for p53 vs. the
other three groups collectively, all were associated with decreased
risk of recurrent disease and better survival compared with other
alternatives. Differently, positive staining for p53 alone or
concomitant negativity for VEGF-A and positivity for p53 vs. the
collective group was related to increased risk of recurrent disease
and worse survival compared with other alternatives.
High expression for VEGF-R1 was detected in 33 (49%)
out of the 67 ovarian tumors in a study concerning the prognostic
value of antigenic markers including patients with ovarian cancer
in FIGO-stages III–IV, but VEGF-R1 was not associated with
progressions-free or overall survival (4). This could be compared with positivity
for VEGF-R1 in only 18 (19%) out of 129 available tumors from
patients in FIGO-stages I–II in the present study. The subgroup of
patients with VEGF-R1 positivity in the present study had
significantly better 5-year disease-free survival rate than
patients with VEGF-R1 negative tumors. Positivity for VEGF-R2 was
observed in 100 (77%) out of the 130 available tumors in the
present study. This is in line with results from as study by
Nishida et al (2), where
positivity for VEGF-R2 was detected by IHC in 60 (75%) out of the
80 ovarian tumors from patients in FIGO-stages I–IV. Patients, who
had high expression of VEGF-R2 in tumors had worse disease-free
survival compared to tumors with negative or low expression of
VEGF-R2 in that study. In a further study conducted by western blot
analysis, higher concentration of VEGF-R2 was found in ovarian
tumors in FIGO-stages I–II compared with FIGO-stages III–IV.
However, high expression of VEGF-R2 did not show any effect on
progressions-free or overall survival (1).
The VEGF-A status alone was not related to survival
in the present study, but positivity for VEGF-A was observed in 60
(61%) out of 98 available tumors. Patients, who had tumors with
high expression of VEGF-A tended to have longer disease-free
survival in a study by Nishida et al (2). Differently, findings in other study
including 36 patients in FIGO-stages I–II showed shorter
disease-free survival with increased expression of VEGF-A in tumors
detected by IHC in 10 (28%) out of the 36 tumors (18). High expression of VEGF-A (using
mRNA) was detected in 48% of tumors to be predictive of poor
prognosis by Shen et al (19). On the contrary, Engels et al
(20) illustrated in the subgroup
of 54 patients with macroscopic complete tumor resection among 112
patients with primary serous cancer in FIGO-stages I–IV, that high
expression of VEGF-A had improved progression-free survival for
this group of patients and this was sustained in multivariate
analysis as independent factor. Despite large numbers of studies
reporting the role of VEGF in ovarian cancer, the mechanisms by
which VEGF mediates these effects remain unclear (21). In randomized trials for primary
disease (GOG 218 and ICON7) and for recurrent platinum sensitive
disease (OCEANS and GOG213), significant improvement of progression
free-survival was observed by the addition of bevacizumab to
conventional chemotherapy. Overall survival has not, however, been
affected and bevacizumab, and >50% of patients do not show
benefit from treatment. Therefore, biomarkers that accurately
predict resistance to the drug would find immediate clinical use
(22). The development of highly
specific inhibitors of both the VEGF ligand (bevacizumab and
VEGF-Trap, ranibizumab) as well as the VEGF receptor (cediranib,
pazopanib and sorafenib) relates to the central role that this
pathway plays in ovarian cancer (3,23).
There are four positive trials with different anti-angiogenic
agents in the front line of ovarian cancer, which have different
strategies, drugs, schedules, ways of administration, and toxicity
profiles why several questions have been raised and there is room
for maximizing the effect of this therapy (24,25).
Although all trials have shown to be positive for progression-free
survival, no one has demonstrated an impact on overall survival.
For instance in a pre-planned test for interaction in predefined
groups of the ICON7 trial (24),
there was a different magnitude of benefit of anti-angiogenic
agents after subgroup. For patients defined as high-risk (stage IV,
stage III not operated, and stage III with residual disease after
surgery >1 cm), there was benefit in progression-free survival
(PFS) with HR of 0.73 and OS with HR of 0.78. While such analyses
have limitations, they can provide direction for future research
(25). In the results from a newly
published meta-analysis (26) the
interaction between three different VEGF polymorphism variants and
decreased risk for ovarian cancer was explored. It was concluded
that one of these (VEGF +936>T) may be a protective factor for
epithelial ovarian cancer among the white ethnicity.
Findings from the present study confirmed that the
angiogenesis regulators VEGF-R2 and VEGF-A correlate to the p53
status with regard to recurrent disease and survival. Thus, the
subgroup of patients with concomitant positivity for VEGF-R2 and
negativity for p53 (VEGF-R2+p53−) of tumors
had a favorable survival compared to the other collective subgroup.
In multivariate analysis the variable
(VEGF-R2+p53−) was independent predictive and
prognostic factor with recurrent disease and disease-free survival
as endpoint, respectively. Negativity of p53 (p53−)
detected by IHC means sustained function of the tumor suppressor
p53. Differently, p53 positivity (p53+) means a defect
function of p53 (mutant p53) in the present study. Disease-free
survival for patients with tumors of positivity for VEGF-R2 alone
was unchanged (95% disease-free survival after 5 years) after
addition of p53 with sustained function in the present study. The
question is whether patients with concomitant positivity of VEGF-R2
and negativity of p53 of tumors exhibit higher rate of clinical
benefits because of unknown tumor-biological properties or could it
be explained with effect of the given post-surgical
chemotherapy.
The subgroup of patients with tumors of concomitant
negativity for VEGF-A and positivity for p53
(VEGF-A−p53+) vs. the collective group had
increased risk for recurrent disease and worse survival. The
variable (VEGF-A−p53+) was not an independent
prognostic or predictive factor in a multivariate analysis.
However, Secord et al (4)
demonstrated that p53 over-expression (p53 positivity) was
associated with low expression of VGEF-A (or VEGF-A negativity) and
their findings could partly confirm results from the present study
study. However, there are conflicting reports in the literature
regarding associations between p53 and VEGF protein expression in
tumor specimens. Thus, a study (7)
with in vitro experiments was performed to understand
cross-talk between p53 and VEGF-A regulation under hypoxic
conditions better. The authors concluded, that there is growing
evidence that the p53 tumor suppressor (wild-type) down-regulates
VEGF-A expression under hypoxia, and according to a further in
vitro study (6) the p53 tumor
suppressor (mutant type) showed decreased apoptosis in response to
sustained chronic relative hypoxia and increased release of VEGF-A.
The development of targeted agents has accelerated in ovarian
cancer during recent years. However, selecting drugs for testing
needs greater knowledge of the biology of ovarian cancer, and new
predictive and prognostic biomarkers so that novel therapeutics can
be introduced more rationally and effectively (11). The initial aim of anti-angiogenic
therapy by using the VEGF/VEGF-receptor axis was to inhibit growth
of new vessel of tumors, usually in combination with chemotherapy.
Studies with various anti-VEGF/VEGF-receptor therapies have shown
that these agents, when used in combination with chemotherapy,
significantly improve survival and response rates in patients
(23,27). A number of studies have shown that
blockade of VEGF or its receptor VEGF-R2 have normalized tumor
vasculature and increased oxygen tension or improved drug
penetration. The increased neo-vascular damage might explain
improved anti-tumor activity when combining VEGF-targeted agents
with chemotherapy (28). This
hypothesis could not explain improved survival rate for patients in
the subgroup with VEGF-R2 positivity alone or with concomitant
negativity for p53 in tumors in the present study. However, in
studies by Adham et al (29,30)
on chemoresistance, it was explored whether a more complete
blockade of VEGF signaling would be an effective strategy for
ovarian cancer control by knocking down VEGF-R2 expression in
chemo-resistant OVCAR-3 EOC cells. Thus, cells with VEGF-R2
knockdown demonstrated more aggressive subcutaneous growth in
vivo. NRP-1 (Neuropilin-1), a co-receptor for VEGF-R2 is also a
receptor for VEGF-A, and enhances the antigenic signal for VEGF-R2,
but the cells lacking VEGF-R2 showed increased NRP-1 expression.
Evaluation of 80 clinical cases of EOC for NRP-1 versus VEGF-R2
expression showed a significantly higher NRP-1:VEGF-R2 ratio with
cancer progression, which means higher NRP-1 expression and/or
decreased VEGF-R2 expression. Therefore, it could be concluded that
overexpression of VEGF-R2 (positivity) in the tumor cells could
protect cancer progression and this findings could partly explain
improved survival rate for patients in the subgroup with VEGF-R2
positivity in the present study.
Limitations of the present study corresponds to the
relatively limited number of patients included, the tissue
microarray technology and the method of semi-quantitative analysis
was used for the interpretation, where all markers were
dichotomized into negative and positive groups. Findings from the
present study confirmed that the angiogenesis regulators VEGF-R2
and VEGF-A have association with the p53 status with regard to
recurrent disease and survival. However, some findings from the
present study are not in line with results from different previous
studies on the regulation of angiogenesis. Although many trials
with anti-angiogenic agents in the front line of ovarian cancer
treatment have shown positive results for progression-free
survival, none has demonstrated an impact on overall survival.
Therefore, one of the greatest challenges in ovarian cancer
research, is to discover predictive and prognostic biomarkers.
References
|
1
|
Klasa-Mazurkiewicz D1, Jarząb M, Milczek
T, Lipińska B and Emerich J: Clinical significance of VEGFR-2 and
VEGFR-3 expression in ovarian cancer patients. Pol J Pathol.
62:31–40. 2011.PubMed/NCBI
|
|
2
|
Nishida N, Yano H, Komai K, Nishida T,
Kamura T and Kojiro M: Vascular endothelial growth factor C and
vascular endothelial growth factor receptor 2 are related closely
to the prognosis of patients with ovarian carcinoma. Cancer.
101:1364–1374. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kieran MW, Kalluri R and Cho YJ: The VEGF
pathway in cancer and disease: Responses, resistance, and the path
forward. Cold Spring Harb Perspect Med. 2:a0065932012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Secord AA, Darcy KM, Hutson A, Lee PS,
Havrilesky LJ, Grace LA and Berchuck A; Gynecologic Oncology Group
study. Co-expression of angiogenic markers and associations with
prognosis in advanced epithelial ovarian cancer: A Gynecologic
Oncology Group study. Gynecol Oncol. 106:221–232. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goodheart MJ, Ritchie JM, Rose SL,
Fruehauf JP, De Young BR and Buller RE: The relationship of
molecular markers of p53 function and angiogenesis to prognosis of
stage I epithelial ovarian cancer. Clin Cancer Res. 11:3733–3742.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kamat CD, Green DE, Warnke L, Thorpe JE,
Ceriello A and Ihnat MA: Mutant p53 facilitates pro-angiogenic,
hyperproliferative phenotype in response to chronic relative
hypoxia. Cancer Lett. 249:209–219. 2007. View Article : Google Scholar
|
|
7
|
Farhang Ghahremani M, Goossens S, Nittner
D, Bisteau X, Bartunkova S, Zwolinska A, Hulpiau P, Haigh K,
Haenebalcke L, Drogat B, et al: p53 promotes VEGF expression and
angiogenesis in the absence of an intact p21-Rb pathway. Cell Death
Differ. 20:888–897. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Le Page C, Huntsman DG, Provencher DM and
Mes-Masson AM: Predictive and prognostic protein biomarkers in
epithelial ovarian cancer: Recommendation for future studies.
Cancers (Basel). 2:913–954. 2010. View Article : Google Scholar
|
|
9
|
de Graeff P, Crijns AP, de Jong S, Boezen
M, Post WJ, de Vries EG, van der Zee AG and de Bock GH: Modest
effect of p53, EGFR and HER-2/neu on prognosis in epithelial
ovarian cancer: A meta-analysis. Br J Cancer. 101:149–159. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hata K, Watanabe Y, Nakai H, Hata T and
Hoshiai H: Expression of the vascular endothelial growth factor
(VEGF) gene in epithelial ovarian cancer: an approach to anti-VEGF
therapy. Anticancer Res. 31:731–738. 2011.PubMed/NCBI
|
|
11
|
Ledermann JA, Marth C, Carey MS, Birrer M,
Bowtell DD, Kaye S, McNeish I, Oza A, Scambia G, Rustin G, et al:
Role of molecular agents and targeted therapy in clinical trials
for women with ovarian cancer. Int J Gynecol Cancer. 21:763–770.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
WMA Declaration of Helsinki - Ethical
Principles for Medical Research Involving Human Subjects.
[http://www.wma.net/en/30publications/10policies/b3/index.html.
|
|
13
|
Trimbos JBVI, Vergote I, Bolis G,
Vermorken JB, Mangioni C, Madronal C, Franchi M, Tateo S, Zanetta
G, Scarfone G, et al; EORTC-ACTION collaborators. European
Organisation for Research and Treatment of Cancer-Adjuvant
ChemoTherapy in Ovarian Neoplasm. Impact of adjuvant chemotherapy
and surgical staging in early-stage ovarian carcinoma: European
Organisation for Research and Treatment of Cancer-Adjuvant
ChemoTherapy in Ovarian Neoplasm trial. J Natl Cancer Inst.
95:113–125. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kononen J, Bubendorf L, Kallioniemi A,
Bärlund M, Schraml P, Leighton S, Torhorst J, Mihatsch MJ, Sauter G
and Kallioniemi OP: Tissue microarrays for high-throughput
molecular profiling of tumor specimens. Nat Med. 4:844–847. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Seidal T, Balaton AJ and Battifora H:
Interpretation and quantification of immunostains. Am J Surg
Pathol. 25:1204–1207. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Köbel M, Kalloger SE, Boyd N, McKinney S,
Mehl E, Palmer C, Leung S, Bowen NJ, Ionescu DN, Rajput A, et al:
Ovarian carcinoma subtypes are different diseases: Implications for
biomarker studies. PLoS Med. 5:e2322008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Skirnisdottir I and Seidal T: Association
of p21, p21 p27 and p21 p53 status to histological subtypes and
prognosis in low-stage epithelial ovarian cancer. Cancer Genomics
Proteomics. 10:27–34. 2013.PubMed/NCBI
|
|
18
|
Paley PJ, Staskus KA, Gebhard K, Mohanraj
D, Twiggs LB, Carson LF and Ramakrishnan S: Vascular endothelial
growth factor expression in early stage ovarian carcinoma. Cancer.
80:98–106. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shen GH, Ghazizadeh M, Kawanami O, Shimizu
H, Jin E, Araki T and Sugisaki Y: Prognostic significance of
vascular endothelial growth factor expression in human ovarian
carcinoma. Br J Cancer. 83:196–203. 2000.PubMed/NCBI
|
|
20
|
Engels K, du Bois A, Harter P,
Fisseler-Eckhoff A, Kommoss F, Stauber R, Kaufmann M, Nekljudova V
and Loibl S: VEGF-A and i-NOS expression are prognostic factors in
serous epithelial ovarian carcinomas after complete surgical
resection. J Clin Pathol. 62:448–454. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li J, Li L, Li Z, Gong G, Chen P, Liu H,
Wang J, Liu Y and Wu X: The role of miR-205 in the VEGF-mediated
promotion of human ovarian cancer cell invasion. Gynecol Oncol.
137:125–133. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bast RC Jr: Biomarkers for ovarian cancer:
new technologies and targets to address persistently unmet needs.
Dis Markers. 8:161–166. 2010.
|
|
23
|
Burger RA: Role of vascular endothelial
growth factor inhibitors in the treatment of gynecologic
malignancies. J Gynecol Oncol. 21:3–11. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Perren TJ, Swart AM, Pfisterer J,
Ledermann JA, Pujade-Lauraine E, Kristensen G, Carey MS, Beale P,
Cervantes A, Kurzeder C, et al: A phase 3 trial of bevacizumab in
ovarian cancer. N Engl J Med. 365:2484–2496. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Abu-Rustum N, Chi D, Coleman RL, del Campo
JM, Fotopoulou C, Frumovitz M, Gershenson DM, Gonzalez A, Jhingran
A, Ledermann J, et al: Summary of the 2014 MD Anderson
International Meeting in Gynecologic Oncology: Emerging therapies
in gynecologic cancer. Gynecol Oncol. 134:6–9. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang X, Qin J and Qin A: Three
polymorphisms of vascular endothelial growth factor (936C > T,
-460C > T, and -2578C > A) and their susceptibility to
ovarian cancer: A meta-analysis. Int J Gynecol Cancer. 25:779–785.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tang C, Hess K, Jardim DL, Gagliato Dde M,
Tsimberidou AM, Falchook G, Fu S, Janku F, Naing A, Piha-Paul S, et
al: Synergy between VEGF/VEGFR inhibitors and chemotherapy agents
in the phase I clinic. Clin Cancer Res. 20:5956–5963. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hicklin DJ and Ellis LM: Role of the
vascular endothelial growth factor pathway in tumor growth and
angiogenesis. J Clin Oncol. 23:1011–1027. 2005. View Article : Google Scholar
|
|
29
|
Adham S, Sher I and Coomber BL: Abstract
#4284: Blockade of VEGFR2/KDR increases malignancy in human
epithelial ovarian carcinoma. Cancer Res. 69:42842009.
|
|
30
|
Adham SA, Sher I and Coomber BL: Molecular
blockade of VEGFR2 in human epithelial ovarian carcinoma cells. Lab
Invest. 90:709–723. 2010. View Article : Google Scholar : PubMed/NCBI
|