Introduction
Esophageal cancer is the sixth leading cause of
cancer death in male and the ninth in female, with an estimated
over 400,000 new cases and deaths each year worldwide (1). The two main histological types are
esophageal squamous cell carcinoma (ESCC) and esophageal
adenocarcinoma. ESCC is the most common histological subtype in
China with 95% of all esophageal cancer cases. With new methods of
diagnosis and treatment, esophageal cancer still has a poor 5-year
survival rate (2). Most of their
mortality is not due to their primary tumors, but to their
invasiveness and metastasis (3). A
better understanding of molecular pathogenesis of ESCC is urgent
for the diagnosis and treatment of the disease.
Regulation of gene expression is a major control
point in organisms to adapt to the environment. The regulation of
mRNA stability and translation are the basis of eukaryotes in
response to intrinsic or extrinsic stimuli. Many mRNAs encode
proto-oncogenes and cytokines, the aberrant mRNA stability is
associated with inflammation and carcinogenesis (4,5). It
is recognized that approximately 16% of all human protein coding
genes are encoded by mRNAs that contain an adenylate-uridylate
(AU)-rich element (ARE) motif in their 3′UTR (6–9).
AU-rich element RNA-binding factor 1 (AUF1), also called the
heterogeneous nuclear ribonucleoprotein D (hnRNPD), is an
established RNA-binding protein. The human AUF1 locus maps to 4q21,
and it comprises a family of proteins composed of four isoforms
with different molecular masses: p37, p40, p42 and p45 (10). AUF1 isoforms express preferentially
in different tissues, which have their distinct biological
properties (10). AUF1 influences
the process of development, apoptosis and tumorigenesis via
interacting with ARE-bearing mRNAs (11).
Altered AUF1 expression level may be a promoter to
the development and progression of multiple types of cancers. It
was reported that overexpression of AUF1 remarkably affected the
stability of ARE containing mRNAs. Kumar et al (12) found that the expression level of
nuclear AUF1 was significantly higher in oral squamous cell
carcinoma than that in normal cells, and downregulated AUF1 with
RNAi resulted in decreased proliferation rates. Overexpression of
AUF1 isoform p37 developed sarcomas with deregulated c-Myc, c-Fos
and c-Jun and cyclin D1 expression in tumor tissues (13). AUF1 has been shown to facilitate
the decay of multiple target mRNAs, but occasionally to modulate
the stability and translation of several target mRNAs (14). Several studies have shown that AUF1
plays an anti-tumorigenic role in regulation of the cell cycle and
apoptosis. For example, increasing the AUF1 by UVC may promote
apoptosis by reducing ARE-containing Bcl-2 mRNA (15). Treatment with postaglandin A2
(PGA2), an anticancer agent, induces expression of AUF1 isoform p45
and leads to the destabilization of cyclin D1 mRNA in non-small
cell lung cancer cells (16). The
above evidence suggests that AUF1 may be a promoter or inhibitor in
the processes of carcinogenesis or proliferation, which vary
between different types of cancer cells.
The clinical relevance of AUF1 and its biological
function in ESCC progression have not been reported. In the present
study, we investigated the expression of AUF1 in the ESCC tissue
samles and normal samples. We further illustrated the role and
downstream effector of AUF1 in driving cell proliferation of ESCC
cells.
Materials and methods
Tissue samples
The ESCC samples were collected from 104 patients
who had not received chemotherapy or radiotherapy prior to surgery.
For these patients, if preoperative ultrasonography or computed
tomography (CT) did not show any enlarged cervical lymph nodes
(minor axis <0.5 cm), patients underwent a subtotal
esophagectomy with two-field lymphadenectomy through a right
thoracotomy, followed by a laparotomy. If enlarged cervical lymph
nodes were detected by preoperative ultrasonography or CT, patients
underwent radical oesophagectomy with cervico-thoraco-abdominal
three-field lymphadenectomy through a right thoracotomy, followed
by a laparotomy and a cervical incision. The resection extent
includes nodes along the cervical part of the esophagus and deep
cervix. Tumor adjacent tissues were obtained 3 cm away from the
tumor. Fifteen normal esophageal tissue samples were obtained from
surgical resections of trauma patients. These normal esophageal
tissue samples were obtained from upper endoscopy of trauma
patients, which were performed by the same operator. These patients
had a fish bone or a poultry bone stuck. These tissues were
obtained postoperatively between 2010 and 2013 from the
Gastrointestinal Center, Jiangyin People's Hospital, the Jiangyin
Clinical College of Xuzhou Medical University (Jiangyin, China) as
previously reported (17). All
patients provided signed and informed consents for their tissue
samples to be used for scientific research. The ethics approval for
the study was obtained from the Jiangyin People's Hospital, the
Jiangyin Clinical College of Xuzhou Medical University. All
diagnoses were based on pathological and/or cytological evidence.
The histological features of the specimens were evaluated by a
senior pathologist according to the classification criteria from
the World Health Organization (18). Tissue samples were obtained prior
to chemotherapy and radiation therapy and were immediately fixed in
10% neutral buffered formalin prior to immunohistochemistry
analysis.
Immunohistochemistry staining (IHC)
The tissue samples were fixed in 10% neutral
buffered formalin and embedded in paraffin. Three-micrometer thick
paraffin sections were deparaffinized and heat-treated with citrate
buffer (pH 6.0) for 7 min as an epitope retrieval protocol.
Endogenous peroxidase was blocked with 3% hydrogen peroxide for 15
min at room temperature and tissue non-specific binding sites were
blocked with skim milk powder at 4% applied for 30 min. Sections
were then incubated with the AUF1 and GCH1 antibodies (Santa Cruz
Biotechnology, Santa Cruz, CA, USA) for 1 h and mixed with skim
milk powder at 2% again to reduce the non-specific staining.
Biotinylated secondary antibody was then added for 30 min. IHC
staining was visualized with substrate solution containing
diaminobenzidine (DAB) and hydrogen peroxide. The counter-staining
was performed with haematoxylin. All steps were performed at room
temperature. Negative controls consisted of tissue sections
undergoing similar staining procedures in the absence of the
primary antibody. The criteria for scoring the stained sections
were as follows: negative (0), <10% of the whole tissue section
stained positive; weakly positive (+ 1), 10–25% of the whole tissue
section stained positive; moderately positive (+ 2), 25–75% of the
whole tissue section stained positive; and strongly positive (+ 3),
75% of the tissue section stained positive. Tissues with score 0 or
1 were considered as AUF1-negative, while with score 2 or 3 were
considered as AUF1-positive.
Cell culture and transfection
The human esophageal cancer cell line Eca-109 was
maintained in Dulbecco's modified Eagle's medium (DMEM)
supplemented with 10% fetal bovine serum (FBS) and antibiotics
(Gibco, Grand Island, NY, USA). Cells were grown in a 37°C
incubator with 5% CO2.
The siRNA targeting AUF1 (siRNA-AUF1) and siRNA
control (siRNA-NC) used in the present study were obtained from
Santa Cruz Biotechnology. The cells were grown in a 6-well culture
plate to 70–80% confluence and then transfected with either
siRNA-NC, siRNA-AUF1 or siRNA-GCH1 (Santa Cruz Biotechnology) using
Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) according to the
manufacturer's instructions. After 48 h, the cells were collected
for western blot analysis.
Cell viability assay
Cell viability was evaluated using the
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide
(MTT) assay. Cells were seeded in 96-well plates. The next day, the
cells were transfected with plasmids or siRNAs according to the
experimental design. The cells were then incubated with 20 μl MTT
(5 mg/ml) for 4 h. After the medium was removed, 100 μl DMSO was
added and the optical density (OD) at 490 nm was measured using a
microplate reader (Bio-Rad Laboratories, Hercules, CA, USA). The
viability index was calculated as experimental OD value/control OD
value. Three independent experiments were performed in
quadruplicate.
Focus formation
Cells transfected with the indicated vectors were
plated at low density (1,000 cells/6-cm plate), incubated for 10
days and fixed and stained with crystal violet. Foci and colonies
containing >50 cells were counted using a microscope.
Western blot analysis
After transfection, cells were harvested and lysed
in RIPA lysis buffer (50 mM Tris-HCl, pH 7.4, 150 mM NaCl, 1%
Triton X-100, 1% sodium deoxycholate, 0.1% SDS) containing protease
inhibitor cocktail (Sigma-Aldrich, St. Louis, MO, USA) for 30 min
at 4°C. Protein (40 μg) from each lysate was fractionated by 10%
SDS-PAGE and transferred to polyvinylidene difluoride membranes
(Millipore, Bedford, MA, USA). After blocking with 5% non-fat milk
in PBS-Tween-20 for 1 h at room temperature, the membranes were
blotted with AUF1 and GCH1 primary antibodies (Abcam, Cambridge,
UK). GAPDH (Beyotime Institute of Biotechnology, Nantong, China)
was used as a loading control. After washing four times with TBST,
the membranes were incubated with horseradish peroxidase
(HRP)-conjugated secondary antibody for 2 h. The proteins were
visualized using the enhanced chemiluminescence (ECL; Beyotime
Institute of Biotechnology).
Microarray analysis
Microarray-based mRNA expression profiling was
performed using the Roche-NimbleGen (135K array) array (Roche
NimbleGen, Madison, WI, USA). The microarrays contained
approximately 45,033 assay probes corresponding to all of the
annotated human mRNA sequences (NCBI HG18). Total RNA labeling and
hybridization were performed using standard conditions according to
the manufacturer's instructions. Genes with fold change of 2 or
greater were subsequently subjected to pathway analysis using
Ingenuity Pathway Analysis (Qiagen, Redwood City, CA, USA).
RNA extraction and real-time PCR
The total RNA from the esophageal tissue samples was
extracted with TRIzol (Invitrogen) and reverse transcribed to
complementary DNA (cDNA) using an oligo(dT)12 primer and
SuperScript II (Invitrogen). The SYBR-Green dye (Takara Bio, Osaka,
Japan) was used for the amplification of cDNA. The mRNA levels of
transcripts were measured using real-time quantitative PCR in
triplicate on a Prism 7500 real-time PCR machine (Applied
Biosystems, Foster City, CA, USA). Primers specific for the genes
are listed in Table I. PCR was
performed in a 20 μl reaction mixture. The thermal cycling
conditions included an initial denaturation step of 95°C for 20
sec, followed by 40 cycles at 95°C for 5 sec and annealing at 60°C
for 30 sec. The relative mRNA expression levels were normalized to
GAPDH.
 | Table IPrimer sequences for real-time PCR
analysis. |
Table I
Primer sequences for real-time PCR
analysis.
| Gene | Forward primer | Reverse primer |
|---|
| GAPDH |
5′-AAGGTGAAGGTCGGAGTC-3′ |
5′-GAAGATGGTGATGGGATTTC-3′ |
| Nalcn |
5′-CCATGAAATCTGTCGTGTGC-3′ |
5′-TGCTCACTGGACAATCAAGG-3′ |
| UGP2 |
5′-TCTGGAAACCTTCGCATCTT-3′ |
5′-TTGCTCCATCTGTGCTTTTC-3′ |
| BEX2 |
5′-TAATAAAGGGGAGCCCTTGG-3′ |
5′-ACTGCCCGCAAACTATGACT-3′ |
| Prdm16 |
5′-ACGCTTGGTTGAAGGTCACT-3′ |
5′-TGGAAAAGTCGGAATGAACC-3′ |
Luciferase assays
Two luciferase reporters containing the full-length
3′UTR fragment of the GCH1 3′UTR (+1 to +1063, relative to
the stop coden) was constructed by Shanghai Biobuy Co., Ltd.
(Shanghai, China). Then, constructs each with a deletion of the two
potential AU-rich elements in the GCH1 3′UTR were
constructed. For the luciferase assay, the reporter vector with the
GCH1 3′UTR was co-transfected with pRL-TK (Promega, Madison,
WI, USA) to correct the transfection efficiency. Luciferase
activity was measured with the Dual-luciferase reporter assay
system (Promega). Promoter activities were expressed as the ratio
of firefly luciferase to Renilla luciferase activities.
Intracellular nitric oxide (NO)
determination
Total NO concentration in cultured cells was
detected by measuring the concentration of nitrate and nitrite by
modified Griess reaction method. After lysis of cells, a total
Nitric Oxide assay kit (Beyotime Institute of Biotechnology) was
used to detect NO. The optical densities at 540 nm wavelength were
recorded using a microplate reader (BioTek Instruments, Winooski,
VT, USA) and the concentrations of NO were calculated according to
the standard curve.
Statistical analysis
Data are expressed as the means ± standard error of
the mean (SEM) of at least three independent experiments. Standard
error bars were included for all data points. The data were first
analyzed with the Kolmogorov-Smirnov test for data distribution
normality. The data were then analyzed using Student's t-test when
only two groups were present or assessed by one-way analysis of
variance (ANOVA) when more than two groups were compared. The
P-values for t-tests were performed by 2-tailed t-tests.
Correlation analysis of the mRNA expression data was performed
using the Pearson test. Statistical analysis was performed using
SPSS software (Release 19.0; SPSS, Inc., Chicago, IL, USA). Data
were considered significant at P<0.05.
Results
Upregulation of AUF1 is correlated with
clinicopathological feature in esophageal squamous cell
carcinoma
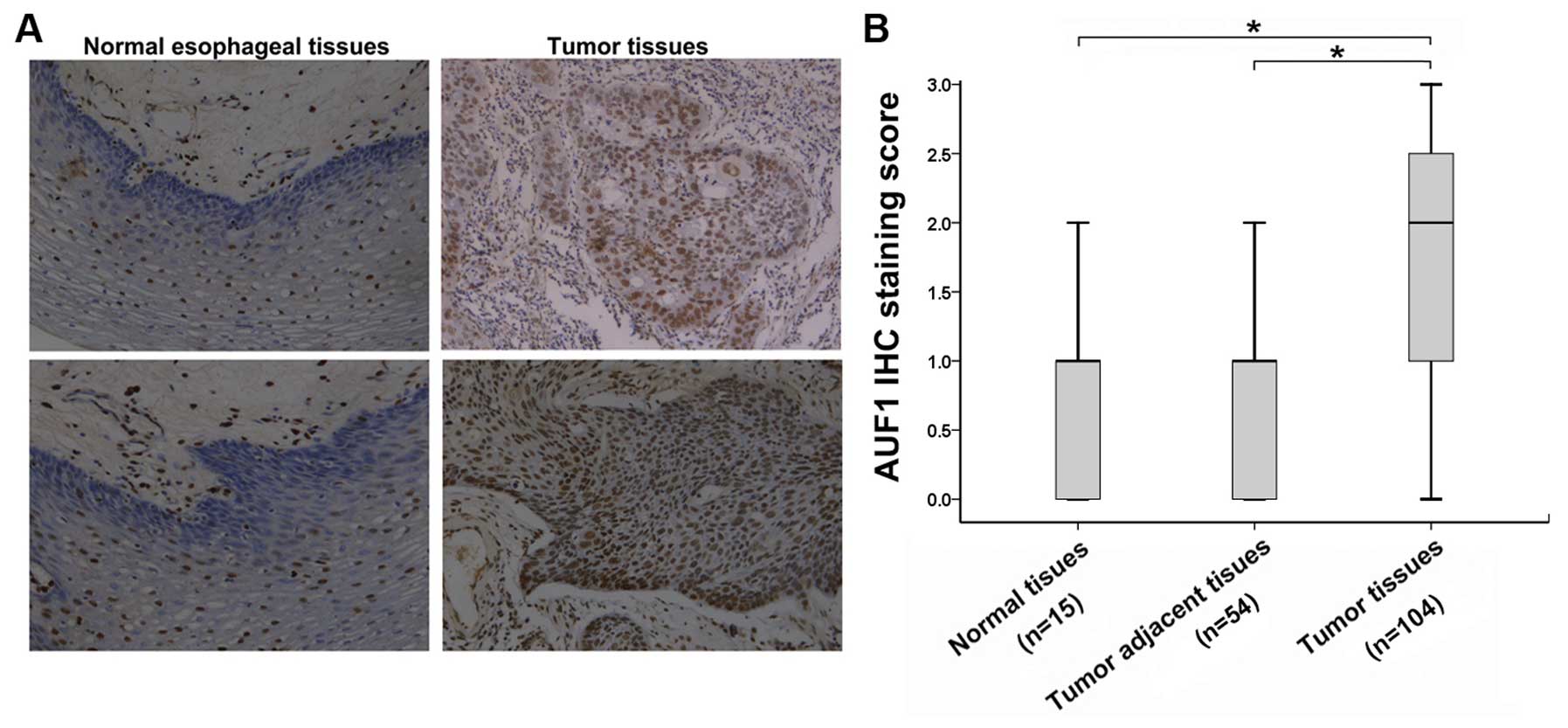
To investigate the expression of AUF1 in human ESCC
tissues, we performed immunohistochemistry analysis on tissue
section of normal esophageal tissue specimens, ESCC and tumor
adjacent tissues. Fifteen paraffin-embedded normal esophageal
tissues and 104 ESCC tissues, 54 of which had corresponding
adjacent tissues were collected. The demographic features of
patients with ESCC for the immunohistochemistry staining are
summarized in Table II. Results
from IHC staining showed that a generally stronger staining of AUF1
was observed in the nucleus of ESCC tissues, whereas very weak
staining of AUF1 was observed in the normal samples (Fig. 1A). When the samples were scored by
a pathologist, as described in Materials and methods, AUF1
expression was similar between normal esophageal tissues and tumor
adjacent tissues, while increased significantly among ESCC tissues,
and between tumor tissues and tumor adjacent tissues (P<0.05;
Fig. 1B). All the above results
indicate that AUF1 may be involved in the pathogenesis of ESCC.
 | Table IIPatient demographic features for the
IHC analysis. |
Table II
Patient demographic features for the
IHC analysis.
| Normal | Grade I | Grade II | Grade III | Grade IV | All tumors |
|---|
| Number | 15 | 15 | 52 | 33 | 4 | 104 |
| Mean age
(years) | 58.73 | 62.88 | 59.21 | 56.50 | 66.00 | 58.90 |
| Age range
(years) | 32–65 | 55–69 | 33–73 | 32–75 | 52–67 | 32–76 |
| Gender |
| Male | 10 | 9 | 37 | 23 | 4 | 73 |
| Female | 5 | 6 | 15 | 10 | 0 | 31 |
Next, the relationship between clinicopathology of
ESCC patients and AUF1 IHC staining score was analyzed. As shown in
Table III, the results showed a
non-significant correlation between AUF1 expression and age
(P=0.610), gender (P=0.637) and lymphatic invasion (P=0.096) of
ESCC patients. AUF1 expression correlated with ESCC stage (P=0.011)
and marginally correlated with lymph node metastasis (P=0.055).
These results suggest AUF1 as a characteristic of this
malignancy.
 | Table IIIRelationship between
clinicopathological parameters and the expression of AUF1 in
esophageal squamous cell carcinoma (ESCC) (n=104). |
Table III
Relationship between
clinicopathological parameters and the expression of AUF1 in
esophageal squamous cell carcinoma (ESCC) (n=104).
| Clinicopathological
parameters | Case | AUF1
expression | P-value |
|---|
|
|---|
| Negative | Positive |
|---|
| Age (years) | | | | 0.610 |
| <64 | 61 | 22 | 39 | |
| ≥64 | 43 | 13 | 30 | |
| Gender | | | | 0.637 |
| Male | 73 | 25 | 48 | |
| Female | 31 | 10 | 21 | |
| Stage | | | | 0.011 |
| 1 | 15 | 9 | 6 | |
| 2 | 52 | 18 | 34 | |
| 3 | 33 | 7 | 26 | |
| 4 | 4 | 1 | 3 | |
| pN | | | | 0.055 |
| (+) | 41 | 9 | 32 | |
| (−) | 63 | 26 | 37 | |
| Lymphatic
invasion | | | | 0.096 |
| (+) | 49 | 12 | 37 | |
| (−) | 55 | 23 | 32 | |
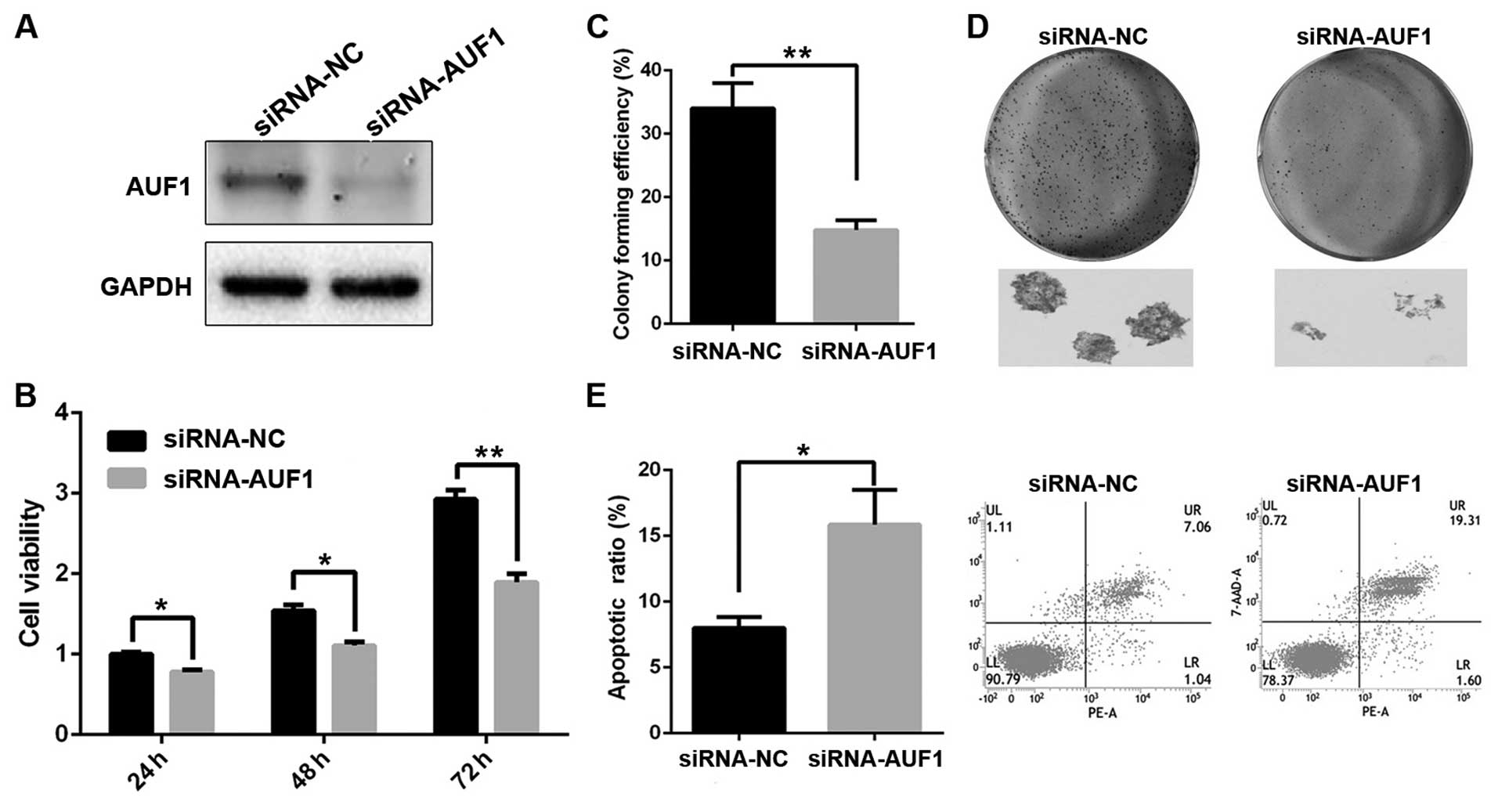
AUF1 expression affects the proliferation
and apoptosis of ESCC cells
Because AUF1 expression was associated with ESCC
progression, we sought to explore the role of AUF1 in esophageal
cancer cell growth. Eca-109 cells were transfected with a siRNA
control (siRNA-NC) or siRNA trageting AUF1 (siRNA-AUF1). As shown
in Fig. 2A, the protein level of
AUF1 was markedly reduced after transfection of siRNA-AUF1. Next,
the effect of AUF1 on cell growth was measured with MTT and colony
formation. The results showed that decreased AUF1 expression
suppressed cell growth and focus formation (Fig. 2B and C). We next investigated
whether reduced cell growth was associated with apoptosis in ESCC
cells. As shown in Fig. 2E,
silencing of AUF1 significantly increased apoptosis of Eca-109
cells. These results demonstrated that AUF1 modulates the
proliferation and apoptotic cell death of ESCC cells.
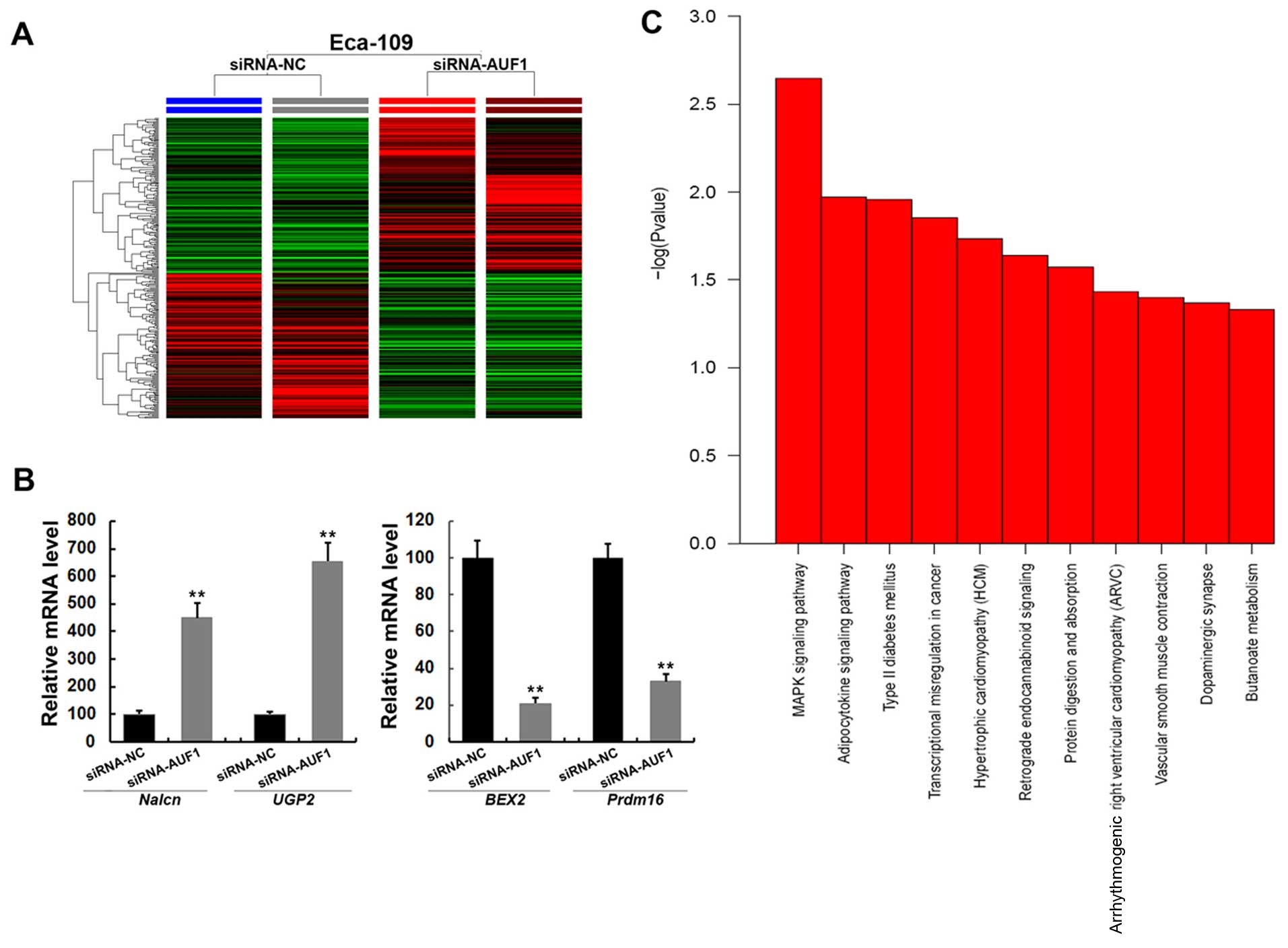
mRNA microarray screening of
AUF1-affected genes
Although many AUF1-regulated genes have been
identified, the profile of AUF1-affected genes in ESCC cells
remains elusive. To further analyze the underlying mechanisms
responsible for AUF1-mediated effects, we screened gene expression
between AUF1 silencing (siRNA-AUF1) and the control cells
(siRNA-NC) by microarray. Twenty-four hours after the transfection,
a total of 285 genes (138 upregulated and 147 downregulated genes)
were identified with an expression differential of 2-fold or
greater between the two groups (n=2; Fig. 3A and Table IV). The differentially expressed
genes included Nalcn, MMP23B, E2F1, BEX2 and GCH1. We then
validated the results from microarray analysis by RT-PCR. As shown
in Fig. 3B, compared with control
cells, several genes dysregulated in microarray data, such as
Nalcn, UGP2, BEX2 and Prdm16 showed consistent changes by real-time
PCR analysis. As expected, AUF1 appeared to have modulated the
Eca-109 cells via complex mechanisms. Pathway analysis revealed
that AUF1 affected multiple pathways, including MAPK signaling
pathway, transcriptional regulation and adipocytokine signaling
pathway (Fig. 3C). These results
suggested that AUF1 might be a wide-spectrum regulator of gene
expression in esophageal cancer cells.
 | Table IVMicorarray analysis of gene
expression changes between siRNA-NC- and siRNA-AUF1-transfected
cells. |
Table IV
Micorarray analysis of gene
expression changes between siRNA-NC- and siRNA-AUF1-transfected
cells.
| Gene name | Fold change
upregulated | Chromosome | Description |
|---|
| NALCN | 25.949 | chr13 | Sodium leak
channel, non selective |
| UGP2 | 20.386 | chr2 | UDP-glucose
pyrophosphorylase 2 |
| SCAF1 | 17.645 | chr19 | SR-related
CTD-associated factor 1 |
| KRTAP10-10 | 17.180 | chr21 | Keratin associated
protein 10-10 |
| MMP23B | 16.273 | chr1 | Matrix
metallopeptidase 23B |
| MAP7D2 | 15.894 | chrX | MAP7 domain
containing 2 |
| TMEM129 | 15.310 | chr4 | Transmembrane
protein 129, E3 ubiquitin protein ligase |
| DACT1 | 15.130 | chr14 | Dishevelled-binding
antagonist of β-catenin 1 |
| ARHGAP30 | 13.292 | chr1 | Rho GTPase
activating protein 30 |
| E2F1 | 13.152 | chr20 | E2F transcription
factor 1 |
| PCNXL2 | 12.669 | chr1 | Pecanex-like 2
(Drosophila) |
| NID1 | 12.587 | chr1 | Nidogen 1 |
| PAK6 | 11.838 | chr15 | P21 protein
(Cdc42/Rac)-activated kinase 6 |
| AMBRA1 | 11.740 | chr11 | Autophagy/Beclin-1
regulator 1 |
| TAF11 | 11.715 | chr6 | TAF11 RNA
polymerase II, TATA Box binding protein (TBP)-associated factor, 28
kDa |
| NCDN | 11.146 | chr1 | Neurochondrin |
| C14ORF101 | 11.047 | chr14 | Transmembrane
protein 260 |
| WDR20 | 10.609 | chr14 | WD repeat domain
20 |
| CCDC74A | 10.475 | chr2 | Coiled-coil domain
containing 74A |
| TRO | 10.257 | chrX | Trophinin |
|
| Gene name | Fold change
downregulated | Chromosome | Description |
|
| BEX2 | 23.010 | chrX | Brain expressed
X-linked 2 |
| PRDM16 | 19.479 | chr1 | PR domain
containing 16 |
| ARX | 18.122 | chrX | Aristaless related
homeobox |
| CPZ | 17.834 | chr4 | Carboxypeptidase
Z |
| GRM4 | 17.232 | chr6 | Glutamate receptor,
metabotropic 4 |
| PHF20L1 | 16.919 | chr8 | PHD finger protein
20-like 1 |
| ANO9 | 13.588 | chr11 | Anoctamin 9 |
| SLC24A4 | 13.212 | chr14 | Solute carrier
family 24 |
| BAZ1B | 12.677 | chr7 | Bromodomain
adjacent to zinc finger domain, 1B |
| C1ORF147 | 11.959 | chr1 | Chromosome 1 open
reading frame 147 |
| SIRT2 | 10.788 | chr19 | Sirtuin 2 |
| RCBTB1 | 10.731 | chr13 | Regulator of
chromosome condensation (RCC1) and BTB (POZ) domain containing
protein 1 |
| ISM1 | 10.317 | chr20 | Isthmin 1,
angiogenesis inhibitor |
| OAZ2 | 10.129 | chr15 | Ornithine
decarboxylase antizyme 2 |
| SNCAIP | 10.088 | chr5 | Synuclein, α
interacting protein |
| CAMK1G | 10.031 | chr1 |
Calcium/calmodulin-dependent protein
kinase IG |
| MYCT1 | 9.578 | chr6 | Myc target 1 |
| SEC14L3 | 8.865 | chr22 | SEC14-like lipid
binding 3 |
| SGCB | 8.732 | chr4 | Sarcoglycan, β |
| GCH1 | 8.726 | chr14 | GTP cyclohydrolase
1 |
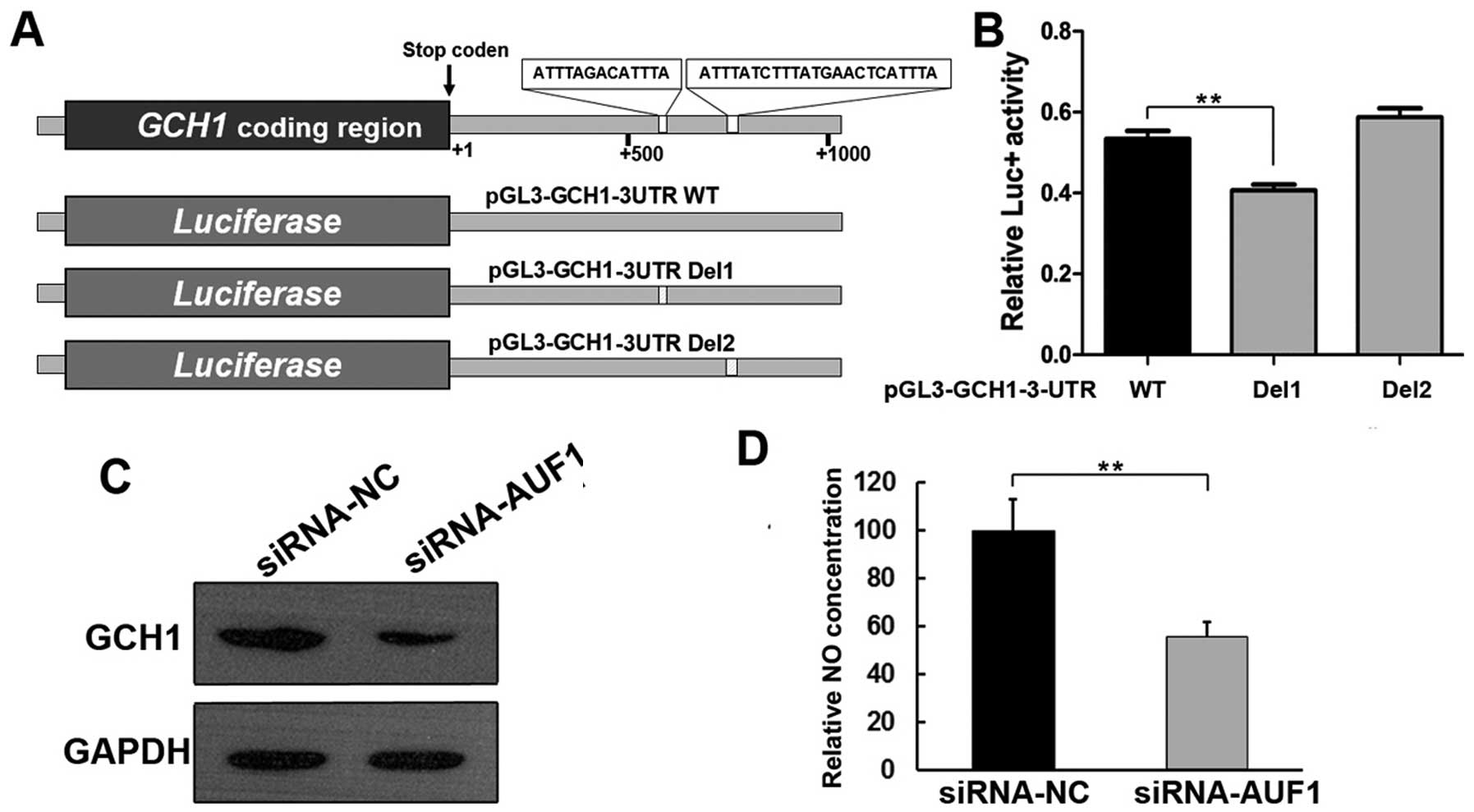
AUF1 regulated GCH1 expression through
3′UTR
Among AUF1 affected genes, we focused on GCH1, which
catalyzes the formation of 5,6,7,8-tetrahydrobiopterin (BH4), an
essential co-factor for all nitric oxide synthases (19,20).
GCH1 has been reported to contribute to cancer progression
(21). Sequence analysis of the
GCH1 3′UTR identified two potential AU-rich elements (Fig. 4A), which were likely to be
regulated by AUF1. We therefore investigated whether AUF1 modulated
the expression of GCH1 and cancer progression through GCH1. The
effect of AUF1 on the GCH1 3′UTR was also measured using
luciferase constructs containing the GCH1 3′UTR with or
without AUF1 binding sites (Fig.
4A). The results revealed that deletion of the ARE1 (pGL3-GCH1
3UTR Del1) showed significantly decreased luciferase activity
compared with that of wild-type GCH1 3′UTR in Eca-109 cells
(Fig. 4B). However, deletion of
the ARE2 (pGL3-GCH1 3UTR Del2) showed similar luciferase activity
as compared with that of wild-type GCH1 3′UTR in Eca-109 cells
(Fig. 4B). Western blot analysis
further confirmed that knockdown of AUF1 decreased the expression
of GCH1 (Fig. 4C). Because GCH1 is
an essential co-factor for all NOS, as expected, silencing of AUF1
significantly reduced cellular NO level. These results suggested
that AUF1 regulated GCH1 through ARE1 region (Fig. 4D).
Expression of AUF1 correlates with GCH1
in ESCC tissues
The above mentioned results led us to investigate
whether the expression level of AUF1 correlated with that of GCH1
in tissue samples. Immunohistochemistry was used to detect the
expression levels of GCH1 protein in 102 ESCC tissue samples.
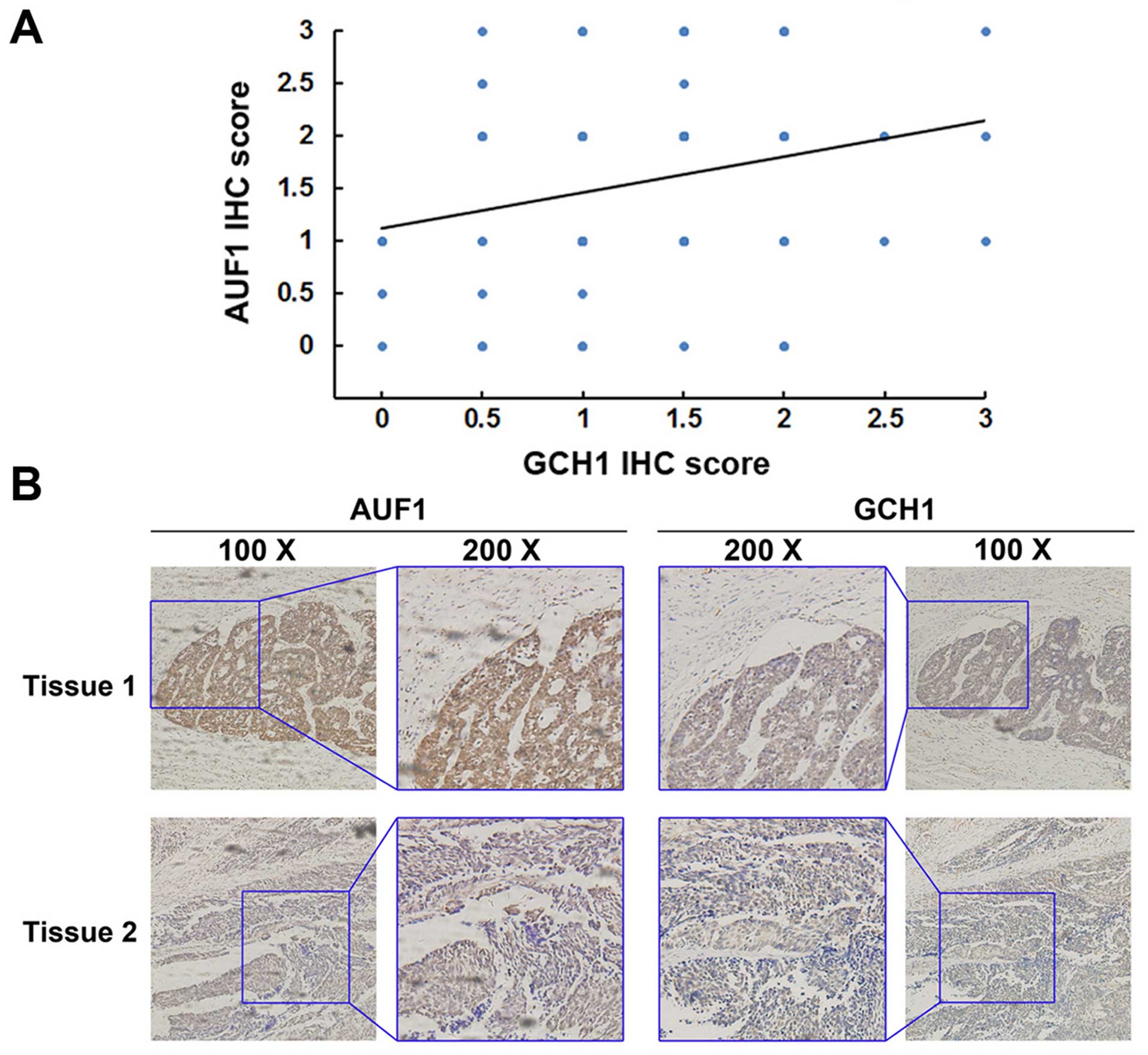
Representative IHC staining of both AUF1 and GCH1 in the same
tissue is shown in Fig. 5A. Tissue
samples with strong AUF1 staining in the nucleus exhibited positive
GCH1 staining, whereas weak AUF1 expression showed negative GCH1
staining. Statistical analysis revealed that the protein levels of
the two genes correlated with each other (Pearson r=0.251, P=0.019;
Fig. 5B), indicating that the
expression of AUF1 correlates with the expression of GCH1 in human
ESCC tissues.
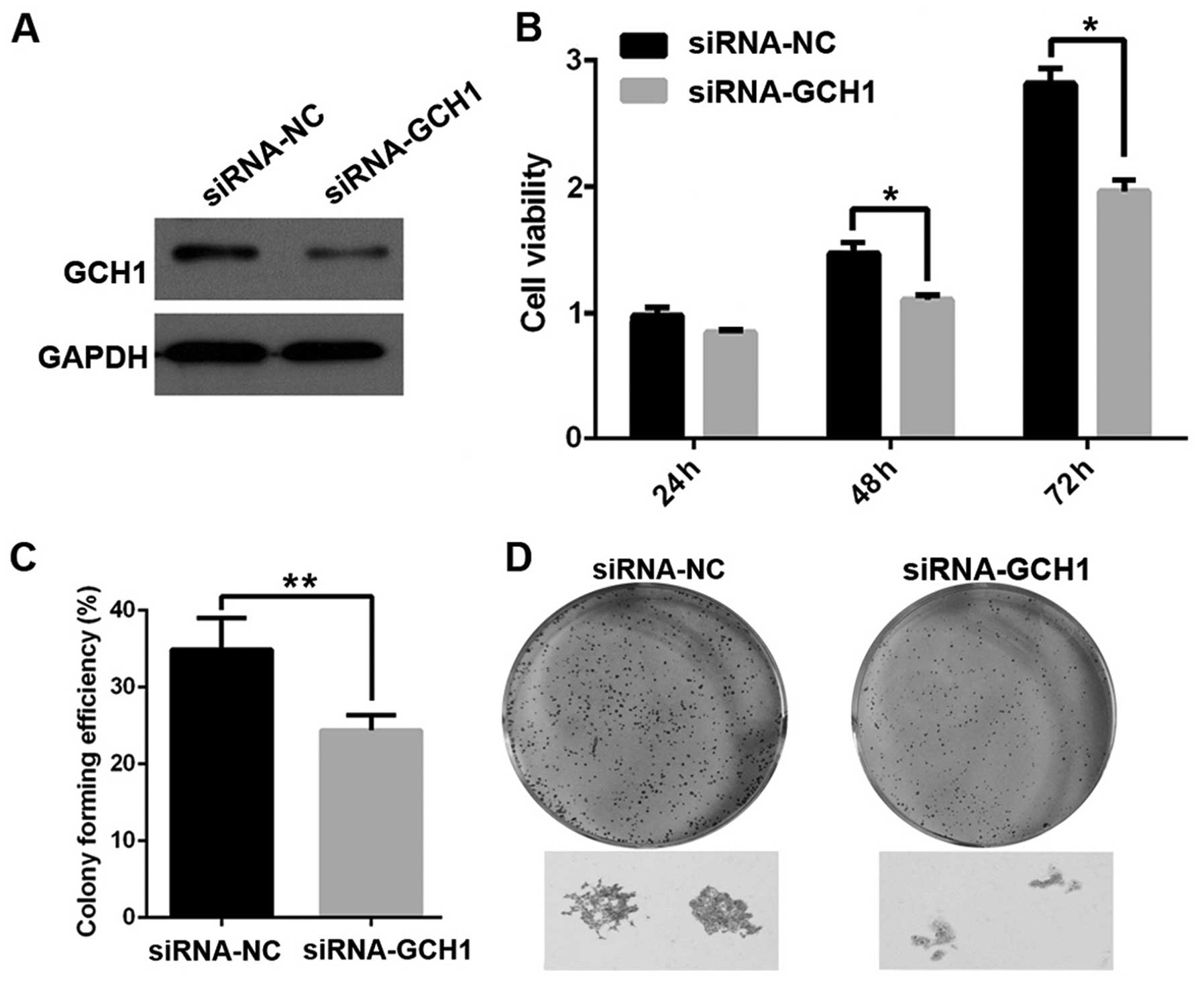
Inhibition of GCH1 suppresses the
proliferation and colony formation in ESCC cells
To explore the role of GCH1 in esophageal cancer
cell growth, cells were first transfected with a siRNA-NC or
siRNA-GCH1, and the resulting growth was measured with MTT and
colony formation assay. The results showed that GCH1 silencing
caused significantly decreased cell viability (75% of the control
group). Moreover, knockdown of GCH1 attenuated the number and the
size of colonies of Eca-109 cells. These results suggest that GCH1
promotes esophageal tumor growth in vitro. (Fig. 6).
Discussion
AUF1 plays a central role in various physiological
and pathological processes. Excessive and dysregulated AUF1
expression has been implicated in diseases including cancers.
Overexpression of AUF1 has been detected in numerous malignancies,
including breast, skin, thyroid, liver, oral squamous cell
carcinoma and sarcoma (12,13,16,22–24).
AUF1 has therefore been recognized to modulate different
cancer-related events. However, there is considerable controversy
in defining its role in cancer progression. Several studies have
shown that AUF1 has both tumor promoting and tumoricidal effects
depending on cancer types (12–16).
AUF1 has been suggested to modulate different cancer-related events
including apoptosis, tumorigenesis and development (10). However, the role of AUF1 in ESCC
progression has not been reported. In the present study, we found
that the expression of AUF1 was significantly higher in ESCC
tissues than that in normal tissues and tumor adjacent tissues. In
this study, we found that AUF1 levels are correlated with tumor
stage, indicating that AUF1 may be a predicator of malignant
degree, and related with prognosis of the malignancy. We also found
that AUF1 silencing resulted in markedly decreased cell
proliferation and focus formation and elevated cell apoptosis.
AUF1 has been implicated in regulating the
expression of numerous mRNAs containing 3′UTR ARE, including Bcl2,
cyclin D1 and SOD1 (10,13,14).
However, the regulatory profile of AUF1 may be cell-specific. To
comprehensively understand the function of AUF1 in esophageal
cancer, we performed mRNA microarray-based screening analysis in
Eca-109 cells. Through this analysis, we found that overexpression
of AUF1 affected multiple genes, such as Nalcn, UGP2, BEX2 and
Prdm16. Among AUF1 affected genes, GCH1 encoded protein is the
first and rate-limiting enzyme in tetrahydrobiopterin (BH4)
biosynthesis, catalyzing the conversion of GTP into
7,8-dihydroneopterin triphosphate (25). BH4 is an essential element required
for nitric oxide (NO) formation by all nitric oxide synthases
(NOSs) (26). NO has been reported
to modulate many cancer-related events, such as angiogenesis,
apoptosis, cell cycle, invasion and metastasis (27), and correlated with the development
of several cancers, including breast, cervical, lung, gastric and
head and neck cancer (28–33). Therefore, GCH1 is likely to play
important roles in cancer progression. Chen et al (34) reported that GCH1 promotes cancer
angiogenesis in vitro and in vivo. Consistently,
another group demonstrates that GCH1 inhibition reduces tumor
growth by: i) direct killing of tumor cells; ii) inhibiting
angiogenesis; and iii) enhancing the antitumoral immune response.
However, little is known about the functional significance of GCH1
in ESCC cancer. Here, we report that GCH1 was regulated by AUF1 and
involved in the proliferation and colony formation in ESCC cells.
The function of GCH1 in other aspects and regulatory mechnisms
merit further investigation.
In summary, we found that AUF1 expression was
significantly increased in ESCC tumor tissues, and that its
expression correlated with tumor stage in human ESCC tissue
samples. Furthermore, knockdown of AUF1 resulted in markedly
decreased proliferation and increased cell apoptosis. AUF1
silencing-affected genes were screened by microarray assay. In
particular, we demonstrated that AUF1 regulated the expression of
GCH1 through its 3′UTR. Silencing of GCH1 inhibited the growth of
ESCC cells. In conclusion, AUF1/GCH1 axis may present a novel
potential target in the treatment of ESCC.
Acknowledgements
The present study was supported by the Natural
Science Foundation of Jiangsu Province (BK20161152), the National
Natural Science Foundation of China (31670857, 81672975, 81522039
and 31400720), the Key Research and Development Program of China
(2016YFC0904702) and the Key Scientific Research Program of Wuxi
Municipal Health Bureau (Z201509).
References
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Enzinger PC and Mayer RJ: Esophageal
cancer. N Engl J Med. 349:2241–2252. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ji X, Cai J, Chen Y and Chen LQ: Lymphatic
spreading and lymphadenectomy for esophageal carcinoma. World J
Gastrointest Surg. 8:90–94. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wilusz CJ, Wormington M and Peltz SW: The
cap-to-tail guide to mRNA turnover. Nat Rev Mol Cell Biol.
2:237–246. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pont AR, Sadri N, Hsiao SJ, Smith S and
Schneider RJ: mRNA decay factor AUF1 maintains normal aging,
telomere maintenance, and suppression of senescence by activation
of telomerase transcription. Mol Cell. 47:5–15. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gruber AR, Fallmann J, Kratochvill F,
Kovarik P and Hofacker IL: AREsite: A database for the
comprehensive investigation of AU-rich elements. Nucleic Acids Res.
39(Database): D66–D69. 2011. View Article : Google Scholar :
|
|
7
|
White EJ, Brewer G and Wilson GM:
Post-transcriptional control of gene expression by AUF1:
Mechanisms, physiological targets, and regulation. Biochim Biophys
Acta. 1829:680–688. 2013. View Article : Google Scholar :
|
|
8
|
Gratacós FM and Brewer G: The role of AUF1
in regulated mRNA decay. Wiley Interdiscip Rev RNA. 1:457–473.
2010. View
Article : Google Scholar
|
|
9
|
Barker A, Epis MR, Porter CJ, Hopkins BR,
Wilce MC, Wilce JA, Giles KM and Leedman PJ: Sequence requirements
for RNA binding by HuR and AUF1. J Biochem. 151:423–437. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Moore AE, Chenette DM, Larkin LC and
Schneider RJ: Physiological networks and disease functions of
RNA-binding protein AUF1. Wiley Interdiscip Rev RNA. 5:549–564.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Abdelmohsen K, Tominaga-Yamanaka K,
Srikantan S, Yoon JH, Kang MJ and Gorospe M: RNA-binding protein
AUF1 represses Dicer expression. Nucleic Acids Res. 40:11531–11544.
2012.PubMed/NCBI
|
|
12
|
Kumar M, Matta A, Masui O, Srivastava G,
Kaur J, Thakar A, Shukla NK, RoyChoudhury A, Sharma M, Walfish PG,
et al: Nuclear heterogeneous nuclear ribonucleoprotein D is
associated with poor prognosis and interactome analysis reveals its
novel binding partners in oral cancer. J Transl Med. 13:2852015.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gouble A, Grazide S, Meggetto F, Mercier
P, Delsol G and Morello D: A new player in oncogenesis: AUF1/hnRNPD
over-expression leads to tumorigenesis in transgenic mice. Cancer
Res. 62:1489–1495. 2002.PubMed/NCBI
|
|
14
|
von Roretz C, Di Marco S, Mazroui R and
Gallouzi IE: Turnover of AU-rich-containing mRNAs during stress: A
matter of survival. Wiley Interdiscip Rev RNA. 2:336–347. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lapucci A, Donnini M, Papucci L, Witort E,
Tempestini A, Bevilacqua A, Nicolin A, Brewer G, Schiavone N and
Capaccioli S: AUF1 is a bcl-2 A + U-rich element-binding protein
involved in bcl-2 mRNA destabilization during apoptosis. J Biol
Chem. 277:16139–16146. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lin S, Wang W, Wilson GM, Yang X, Brewer
G, Holbrook NJ and Gorospe M: Down-regulation of cyclin D1
expression by prostaglandin A2 is mediated by enhanced
cyclin D1 mRNA turnover. Mol Cell Biol. 20:7903–7913. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Luo J, Zhou X, Ge X, Liu P, Cao J, Lu X,
Ling Y and Zhang S: Upregulation of Ying Yang 1 (YY1) suppresses
esophageal squamous cell carcinoma development through heme
oxygenase-1. Cancer Sci. 104:1544–1551. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sobin LH and Wittekind C: TNM
Classification of Malignant Tumor. 6. John Wiley and Sons; New
Jersey: 2002
|
|
19
|
Thöny B, Auerbach G and Blau N:
Tetrahydrobiopterin biosynthesis, regeneration and functions.
Biochem J. 347:1–16. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Werner ER, Blau N and Thöny B:
Tetrahydrobiopterin: Biochemistry and pathophysiology. Biochem J.
438:397–414. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen L, Zeng X, Kleibeuker E, Buffa F,
Barberis A, Leek RD, Roxanis I, Zhang W, Worth A, Beech JS, et al:
Paracrine effect of GTP cyclohydrolase and angiopoietin-1
interaction in stromal fibroblasts on tumor Tie2 activation and
breast cancer growth. Oncotarget. 7:9353–9367. 2016.PubMed/NCBI
|
|
22
|
Trojanowicz B, Brodauf L, Sekulla C,
Lorenz K, Finke R, Dralle H and Hoang-Vu C: The role of AUF1 in
thyroid carcinoma progression. Endocr Relat Cancer. 16:857–871.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zucconi BE and Wilson GM: Modulation of
neoplastic gene regulatory pathways by the RNA-binding factor AUF1.
Front Biosci (Landmark Ed). 16:2307–2325. 2011. View Article : Google Scholar
|
|
24
|
Vázquez-Chantada M, Fernández-Ramos D,
Embade N, Martínez-Lopez N, Varela-Rey M, Woodhoo A, Luka Z, Wagner
C, Anglim PP, Finnell RH, et al: HuR/methyl-HuR and AUF1 regulate
the MAT expressed during liver proliferation, differentiation, and
carcinogenesis. Gastroenterology. 138:1943–1953. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Antoniades C, Shirodaria C, Van Assche T,
Cunnington C, Tegeder I, Lötsch J, Guzik TJ, Leeson P, Diesch J,
Tousoulis D, et al: GCH1 haplotype determines vascular and plasma
biopterin availability in coronary artery disease effects on
vascular super-oxide production and endothelial function. J Am Coll
Cardiol. 52:158–165. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Douglas G, Hale AB, Crabtree MJ, Ryan BJ,
Hansler A, Watschinger K, Gross SS, Lygate CA, Alp NJ and Channon
KM: A requirement for Gch1 and tetrahydrobiopterin in embryonic
development. Dev Biol. 399:129–138. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ying L and Hofseth LJ: An emerging role
for endothelial nitric oxide synthase in chronic inflammation and
cancer. Cancer Res. 67:1407–1410. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Loibl S, von Minckwitz G, Weber S, Sinn
HP, Schini-Kerth VB, Lobysheva I, Nepveu F, Wolf G, Strebhardt K
and Kaufmann M: Expression of endothelial and inducible nitric
oxide synthase in benign and malignant lesions of the breast and
measurement of nitric oxide using electron paramagnetic resonance
spectroscopy. Cancer. 95:1191–1198. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Thomsen LL, Miles DW, Happerfield L,
Bobrow LG, Knowles RG and Moncada S: Nitric oxide synthase activity
in human breast cancer. Br J Cancer. 72:41–44. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Beevi SS, Rasheed MH and Geetha A:
Evidence of oxidative and nitrosative stress in patients with
cervical squamous cell carcinoma. Clin Chim Acta. 375:119–123.
2007. View Article : Google Scholar
|
|
31
|
Masri FA, Comhair SA, Koeck T, Xu W,
Janocha A, Ghosh S, Dweik RA, Golish J, Kinter M, Stuehr DJ, et al:
Abnormalities in nitric oxide and its derivatives in lung cancer.
Am J Respir Crit Care Med. 172:597–605. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Feng CW, Wang LD, Jiao LH, Liu B, Zheng S
and Xie XJ: Expression of p53, inducible nitric oxide synthase and
vascular endothelial growth factor in gastric precancerous and
cancerous lesions: Correlation with clinical features. BMC Cancer.
2:82002. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cooper RG and Magwere T: Nitric
oxide-mediated pathogenesis during nicotine and alcohol
consumption. Indian J Physiol Pharmacol. 52:11–18. 2008.PubMed/NCBI
|
|
34
|
Chen L, Zeng X, Wang J, Briggs SS, O'Neill
E, Li J, Leek R, Kerr DJ, Harris AL and Cai S: Roles of
tetrahydrobiopterin in promoting tumor angiogenesis. Am J Pathol.
177:2671–2680. 2010. View Article : Google Scholar : PubMed/NCBI
|