Introduction
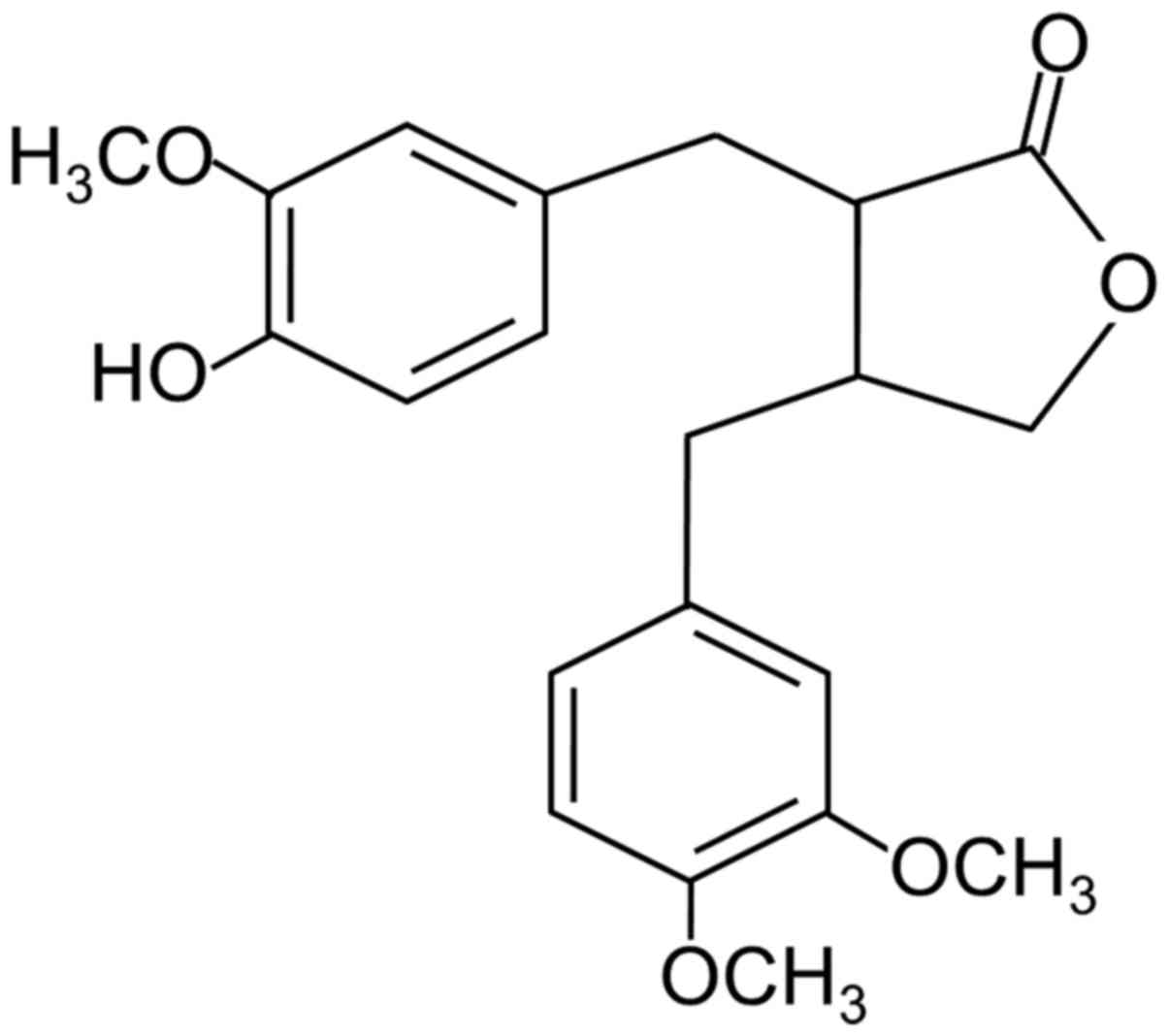
Arctigenin is a phenylpropanoid dibenzyl
butyrolactone lignan (Fig. 1)
found in members of the Asteraceae family eg: Arctium lappa
(1). Arctium lappa,
commonly known as burdock, and its extracts have long been consumed
in Europe, North America and Asia. It is also traditionally used in
folk medicine for treating sore throat and various infections.
Several studies have shown that arctigenin possesses therapeutic
effects for anti-viral (2–4), anti-inflammatory (5,6),
immune modulatory (7–10) and antitumor activities.
Particularly, the antitumor activity of arctigenin has drawn much
scientific interest and it has been tested in various human cancer
cell lines, showing promising results in cancer cells of the
stomach (11,12), liver (12), pancreas (13,14),
intestine (15–17), lung (12,18,19),
bladder (20) and ovaries
(21). Regarding the effects of
arctigenin in breast cancers, Hsieh et al (22) showed that arctigenin markedly
inhibited the growth of ER-negative MDA-MB-231 cells by triggering
the mitochondrial caspase-independent apoptotic pathway. However,
as lignans have been identified as phytoestrogens, arctigenin has
also been reported to have estrogenic properties (23), making it an interesting candidate
as breast and other hormone responsive cancer treatments.
When breast cancers express estrogen receptors,
estrogen is considered to promote tumor growth. Since
phytoestrogens are plant-derived chemicals that are structurally
and/or functionally similar to estrogen, the role of phytoestrogens
in breast cancer therapy is a highly debated topic (24,25).
Although their affinities to estrogen receptors are lower than that
of estrogen, it is still of concern that a high intake of
phytoestrogen may promote estrogen-dependent transcription, leading
to increased risk in breast cancer patients, especially those
treated with hormone therapy to reduce estrogen effects within the
breast tissue. However, there is growing evidence that
phytoestrogens have a protective effect on the initiation of breast
cancers by inhibiting the enzymes involved in the synthesis of
estrogen, resulting in the reduced production of estrogen (26–28).
Moreover, the low rates of breast cancer in Asia imply the health
benefit of soy phytoestrogen, which is abundant in traditional
Asian diet, in reducing the risk of breast cancer. Thus, it is
still questionable whether phytoestrogens have beneficial effects
against the risk of breast cancers, and determine the necessity for
breast cancer patients to increase the intake of arctigenin or
arctigenin-enriched foods.
Despite the successful treatment of the primary
tumor, metastasis is a major cause of lethality in cancer patients.
It was reported that 30–75% of patients undergoing surgery and
adjuvant treatment developed recurrent metastatic disease and the
median survival of patients with metastatic breast cancer is
approximately 2 years (29,30).
Zhang et al (15) reported
that arctigenin inhibited HCT116 colon cancer cell migration and
invasion through the regulation of the H1F4A and Wnt/β catenin
pathways. However, to the best of our knowledge, the
anti-metastatic effect of arctigenin has not been clearly evaluated
on breast cancer cells to date. In this study, we assessed the
anti-metastatic effects of arctigenin on breast cancer cells. In
order to check if the effects of the phytoestrogen arctigenin
depended on the status of ER expression we employed both estrogen
receptor (ER)-positive MCF-7 and ER-negative MDA-MB-231 human
breast cancer cell lines.
Materials and methods
Cell culture
MCF-7 and MDA-MB-231 human breast cancer cell lines
were purchased from the Korean Cell line Bank (Seoul, Korea). MCF-7
cells were cultured in Dulbecco's modified Eagle's medium (DMEM)
supplemented with 10% fetal bovine serum (FBS) and 10 µg/ml
insulin (all from welgene Inc., Daegu, Korea). MDA-MB-231 cells
were cultured in DMEM supplemented with 10% FBS only. All media
also contained 1% antibiotic-antimycotic solution (welgene).
Wound healing assay
The cells were seeded in 6-well plates coated with
collagen type I (Corning Life Sciences, Bedford, MA, USA) and grown
to reach 100% confluence. The monolayers of cells were scratched
with a 200 µl pipette tip to create a wound. The cells were
then treated with conditioned media containing 0, 10, 50 and 200
µM arctigenin (Santa Cruz Biotechnology, Inc., Santa Cruz,
CA, USA). A total of 100 nM
12-O-tetradecanoylphorbol-13-acetate (TPA; Sigma-Aldrich,
St. Louis, MO, USA) was used to induce cell migration in MCF-7
cells, while the treatment of TPA was omitted in MDA-MB-231 cells
due to their inherent invasive properties. Two independent
experiments were done in duplicate wells and optical microscopic
images were captured from two different areas of each well at 0 and
24 h. The area between the wound edges was measured at each
time-point using ImageJ software as described before (31). Wound closure was quantified as
previously described (32).
Matrigel invasion assay
The effect of arctigenin on the invasiveness of
human breast cancer cells were determined using Matrigel and
Transwell chambers (Corning Life Sciences) with 8.0-µm pore
polycarbonate filter inserts in 24-well plates. The lower face of
the polycarbonate filter (Transwell insert) was coated with
Matrigel for 1 h at 37°C. After coating, 3×104 cells
were seeded into the Transwell chamber and 750 µl culture
media was added to the lower chamber. After 24 h, cells were
treated with conditioned media containing 2% FBS and 0, 10, 50 and
200 µM arctigenin (Santa Cruz Biotechnology). TPA (100 nM)
was used to induce only MCF-7 cells and not the innately invasive
MDA-MB-231 cells. The media in the bottom chamber was also changed
to media containing 10% FBS and incubated at 37°C in 5%
CO2 atmosphere for 24 h. Cells that migrated across the
membrane were then fixed and stained using hematoxylin and eosin
(H&E) staining and photographed under an inverted microscope
using ×200 magnification.
Protein extraction and western blot
analysis
The cells were treated with conditioned media
containing 0, 10, 25, 50, 100 and 200 µM arctigenin for 24
h. For induction of invasive characteristics, 100 nM TPA was added
for 1 h and then cells were harvested for protein extraction. The
cells were lysed with RIPA buffer (50 mM Tris-HCl pH 7.5 with 150
mM NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% sodium
dodecyl sulphate-SDS and 2 mM EDTA) containing phosphatase and
protease inhibitor cocktail (GenDEPOT, Barker, TX, USA). Lysates
were cleared of debris at 13,000 rpm for 20 min at 4°C and protein
concentrations were determined using bicinchoninic acid reagent
(BCA; Sigma-Aldrich). The proteins were separated by SDS-PAGE and
transferred onto the polyvinylidene fluoride (PVDF) membranes at
100 V for 40 min. The membranes were blocked in 5% skim milk in
Tris-buffered saline Tween-20 buffer (10 mM Tris-HCl, 150 mM NaCl
and 0.1% Tween-20) for 1 h at room temperature. All primary
antibodies were from Cell Signaling Technology (Beverly, MA, USA),
were diluted 3,000 times and incubated with blots overnight at 4°C:
phospho (#4060) and total (#4691) Akt, phospho (#3033) and total
(#8242) NF-κB, phospho (#4370) and total (#4695) ERK1/2, phospho
(#4511) and total (#8690) p38, phospho (#4668) and total (#9258)
JNK 1/2, c-jun (#9165), c-fos (#2250) and GAPDH (#5174).
Corresponding HRP-conjugated secondary antibody (#sc-2004; Santa
Cruz Biotechnology) diluted 5,000 times was incubated with blots
for 1 h at room temperature. The blots were developed and imaged
using luminescent Image Analyzer LAS-4000 (Fujifilm, Tokyo, Japan).
Densitometric analysis was performed using Scion Image (Scion
Corp., Frederick, MD, USA) with data from three independent
experiments.
Zymography
Conditioned media were collected and electrophoresed
on 8% SDS-PAGE containing 0.1% (v/v) gelatin under non-reducing
conditions. To remove SDS, the gel was washed with 2.5% Triton
X-100 for 1 h at room temperature. Gelatinase reaction was carried
out at 37°C in reaction buffer (50 mM Tris-HCl, pH 7.5, 10 mM
CaCl2, 0.04% NaN3) for 24 h in the case of
MDA-MB-231 cells and 48 h in the case of MCF-7 cells. The gel was
then stained with staining solution (0.05% Coomassie brilliant blue
in 45% methanol and 10% acetic acid) and destained with destaining
solution (10% methanol and 10% acetic acid). Proteolysis was
detected as clear bands in a dark blue field. Densitometric
analysis was performed using Scion Image with data from three
independent experiments.
RNA isolation and reverse transcriptase
polymerase chain reaction (RT-PCR)
The cells were treated with conditioned media
containing 0, 10, 25, 50, 100 and 200 µM arctigenin and 100
nM TPA for 24 h. The cells were then harvested for RNA isolation.
Total RNA was isolated using easy-BLUE™ Total RNA Extraction kit
(iNtRoN Biotechnology, Inc., Sungnam, Korea) and the RNA
concentrations were determined using NanoDrop (Schimadzu Scientific
Instruments, Columbia, MD, USA). The synthesis of cDNA and PCR
reactions were performed as previously described (33). The primer sequences for the target
genes and thermal cycling conditions are given in Table I. Densitometric analysis was
performed using Scion Image with data from three independent
experiments.
 | Table IPrimer sequences and thermal cycling
parameters used for RT-PCR. |
Table I
Primer sequences and thermal cycling
parameters used for RT-PCR.
| Gene | Primer
sequence | PCR conditions
(temperature and time)
|
|---|
| Denaturation | Annealing | Extension |
|---|
| MMP-9 | Forward: 5′-TTC ATC
TTC CAA GGC CAA TC-3′ | | | |
| Reverse: 5′-CTT GAC
GCT GTC AAA GTT CG-3′ | 94°C, 30 sec | 55°C, 30 sec | 72°C, 30 sec |
| uPA | Forward: 5′-CCA ATT
AGG AAG TGT AAC AGC-3′ | | | |
| Reverse: 5′-GCC AAG
AAA GGG ACA TCT ATG-3′ | 94°C, 30 sec | 55°C, 30 sec | 72°C, 45 sec |
| uPAR | Forward: 5′-CAT GCA
GTG TAA GAC CAA CGG GGA-3′ | | | |
| Reverse: 5′-AAT AGG
TGA CAG CCC GGC CAG AGT-3′ | 94°C, 30 sec | 65°C, 30 sec | 72°C, 45 sec |
| GAPDH | Forward: 5′-ATC CCA
TCA CCA TCT TCC AG-3′ | | | |
| Reverse: 5′-TTC TAG
ACG GCA GGT CAG GT-3′ | 94°C, 30 sec | 58°C, 30 sec | 72°C, 60 sec |
| 18S rRNA | Forward: 5′-GTA ACC
CGT TGA ACC CCA TT-3′ | | | |
| Reverse: 5′-CCA TCC
AAT CGG TAG TAG CG-3′ | 94°C, 30 sec | 58°C, 30 sec | 72°C, 30 sec |
Statistical analysis
Statistical significance was determined using the
Student's t-test on Microsoft office Excel 2010 (Microsoft,
Albuquerque, NM, USA) and results are presented as mean ± SD.
P<0.05 were considered statistically significant.
Results
Arctigenin inhibits cell migration and
invasion of human breast cancer cells
The effects of arctigenin against metastasis were
assessed by the ability of the cells to move and close the gap
after creating uniform wounds with a 200 µl pipette in the
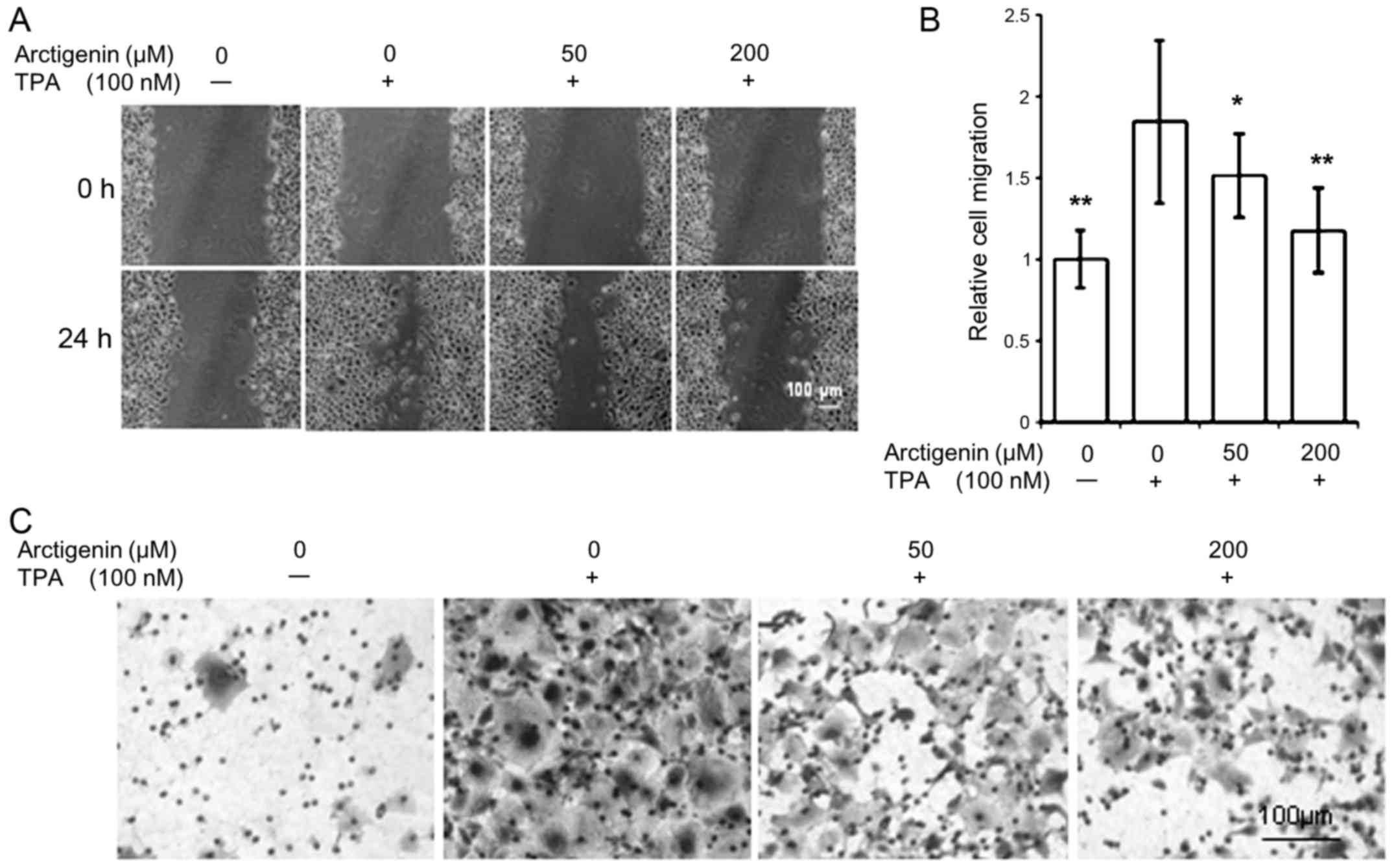
wound healing assay. Since MCF-7 cells are non-invasive ER-positive
breast cancer cells, representing the ER/PR-positive luminal
subtype, they were stimulated with the tumor promoter TPA to induce
cell migration. However, treatment of TPA was omitted in MDA-MB-231
cells due to their inherent invasive properties. Our results show
that arctigenin inhibited cell migration in both cell types.
ER-positive MCF-7 control cells showed almost no cell migration. On
TPA treatment, however, cell migration and almost complete wound
closure were observed within 24 h (Fig. 2A). This TPA-induced cell migration
was inhibited by arctigenin in a dose-dependent manner; exerting
almost 90% inhibition of cell migration with 200 µM
arctigenin treatment (Fig. 2B).
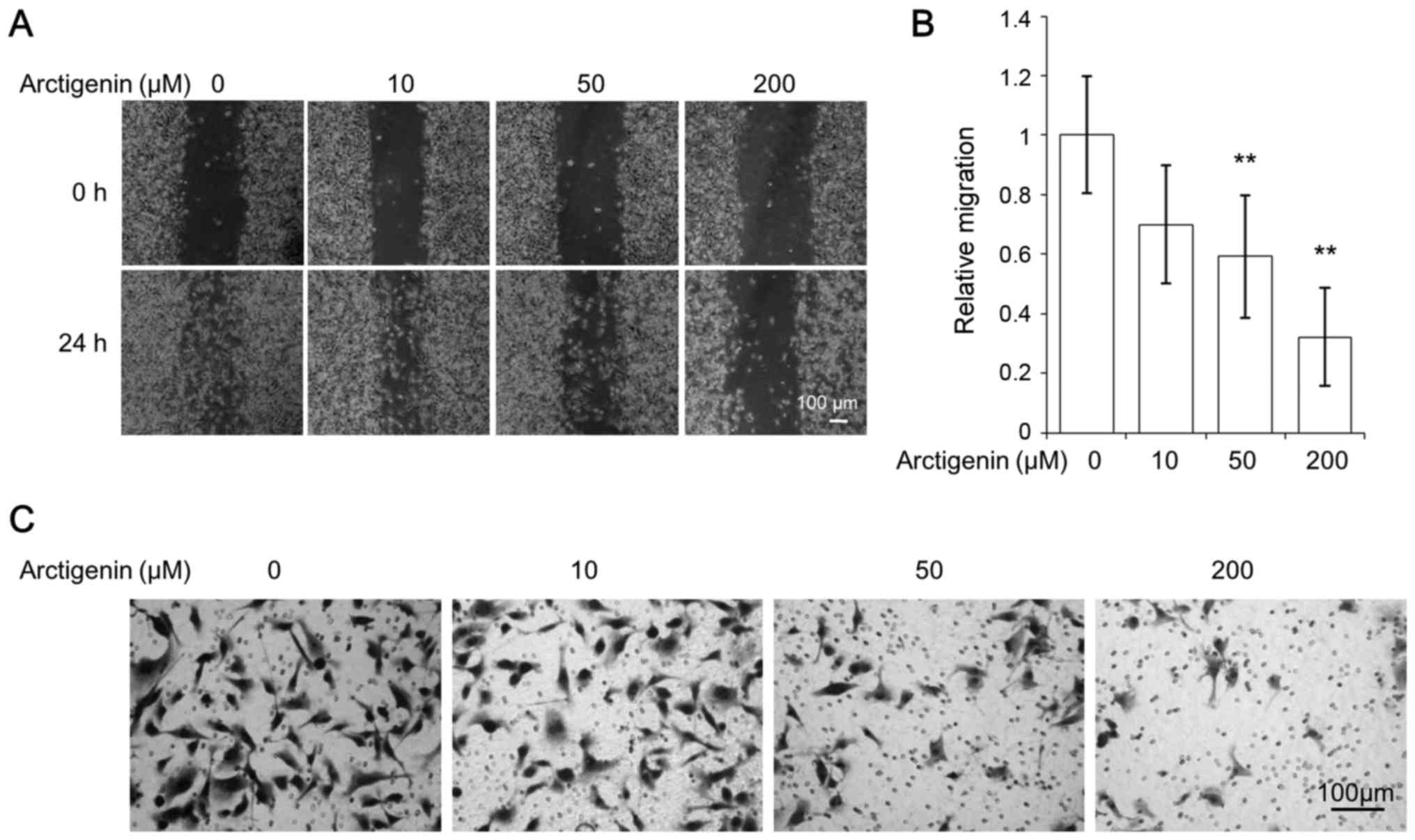
MDA-MB-231 cells exhibited high migratory characteristics, showing
rapid wound closure, even in the control cells (Fig. 3A). However, similar to the effect
of artigenin on MCF-7 cells, arctigenin inhibited the cell
migration of MDA-MB-231 cells in a dose-dependent manner (Fig. 3B). The ability of cancer cells to
invade and move across matrices were also evaluated using the
Matrigel invasion assay. Arctigenin inhibited the TPA-induced
invasive characteristics of MCF-7 cells (Fig. 2C) as well as the innate
invasiveness of MDA-MB-231 cells (Fig.
3C) in a dose-dependent manner. Thus, our data suggested that
arctigenin inhibited cell migration and invasion of human breast
cancer cells regardless of the presence of ER.
Arctigenin inhibits the crucial
proteolytic enzyme MMP-9
For the cancer cells to metastasize, it has to first
cross the physical barriers, i.e. the basement membrane and
extracellular matrix. This is made possible by several protease
enzymes, primarily the matrix metalloproteases (MMPs) that are
capable of disrupting the extracellular matrix. The gelatinase
MMP-9 is a highly regulated enzyme that is upregulated in the case
of invasive breast cancers (34).
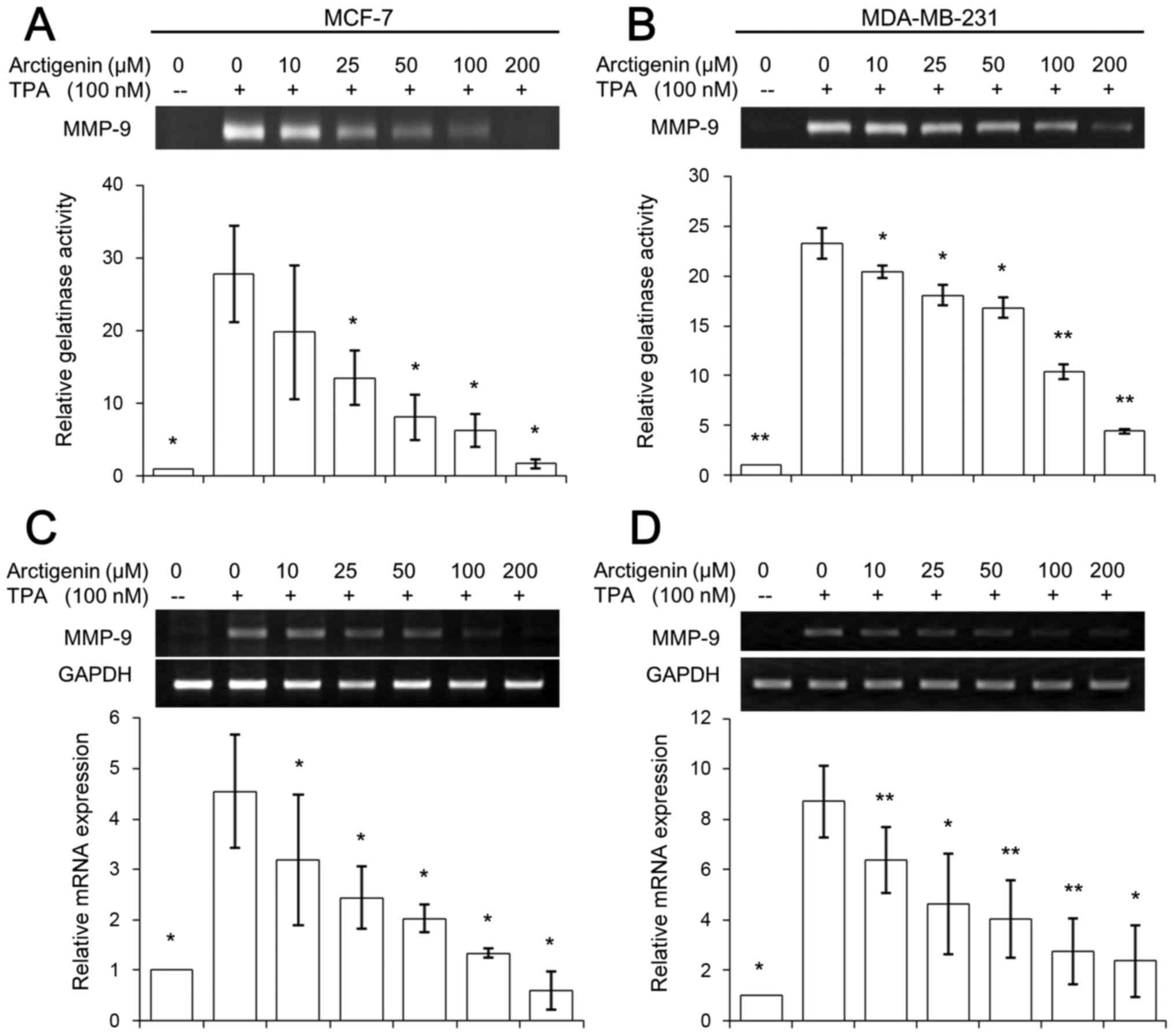
Therefore, we looked at the effect of arctigenin on MMP-9, at both
the activity and transcriptional levels. Since the synthesis and
secretion of MMP-9 are induced by a variety of stimuli, including
cytokines and TPA, both cell lines were treated with 100 nM TPA for
24 h to induce detectable levels of MMP-9. First, we measured the
activity of secreted MMP-9 in the conditioned media using
zymography.
The collected media after 24 h treatment with
various doses of arctigenin was run on SDS-PAGE gels containing
0.1% gelatin and the gelatinolytic activity could be quantified
from the clear bands due to proteolysis after staining the gel with
Coomassie brilliant blue. The endogenous level of MMP-9 activities
were barely detectable in MCF-7 or MDA-MB-231 cells, while TPA
treatment amplified MMP-9 activity ~20 times in both MCF-7
(Fig. 4A) and MDA-MB-231 (Fig. 4B) cell lines. However, arctigenin
exerted a dose-dependent inhibitory effect in both cell lines,
exhibiting almost complete inhibition at 200 µM arctigenin
treatment (Fig. 4A and B). The
gene expression of MMP-9 at the transcription level was also
studied using RT-PCR. TPA treatment amplified MMP-9 mRNA levels ~3
times in both MCF-7 (Fig. 4C) and
MDA-MB-231 (Fig. 4D) cell lines.
Similar to the effect of arctigenin on MMP-9 activity, the
treatment with arctigenin showed a clear inhibitory effect on the
expression of MMP-9 gene; in particular, the cells treated with
conditioned media containing 200 µM arctigenin showed almost
basal level expression of MMP-9 in both cell lines (Fig. 4C and D). Therefore, our data
suggested that arctigenin inhibited cell migration of human breast
cancer cells by regulating the activity of MMP-9 and its gene
expression regardless of the ER status.
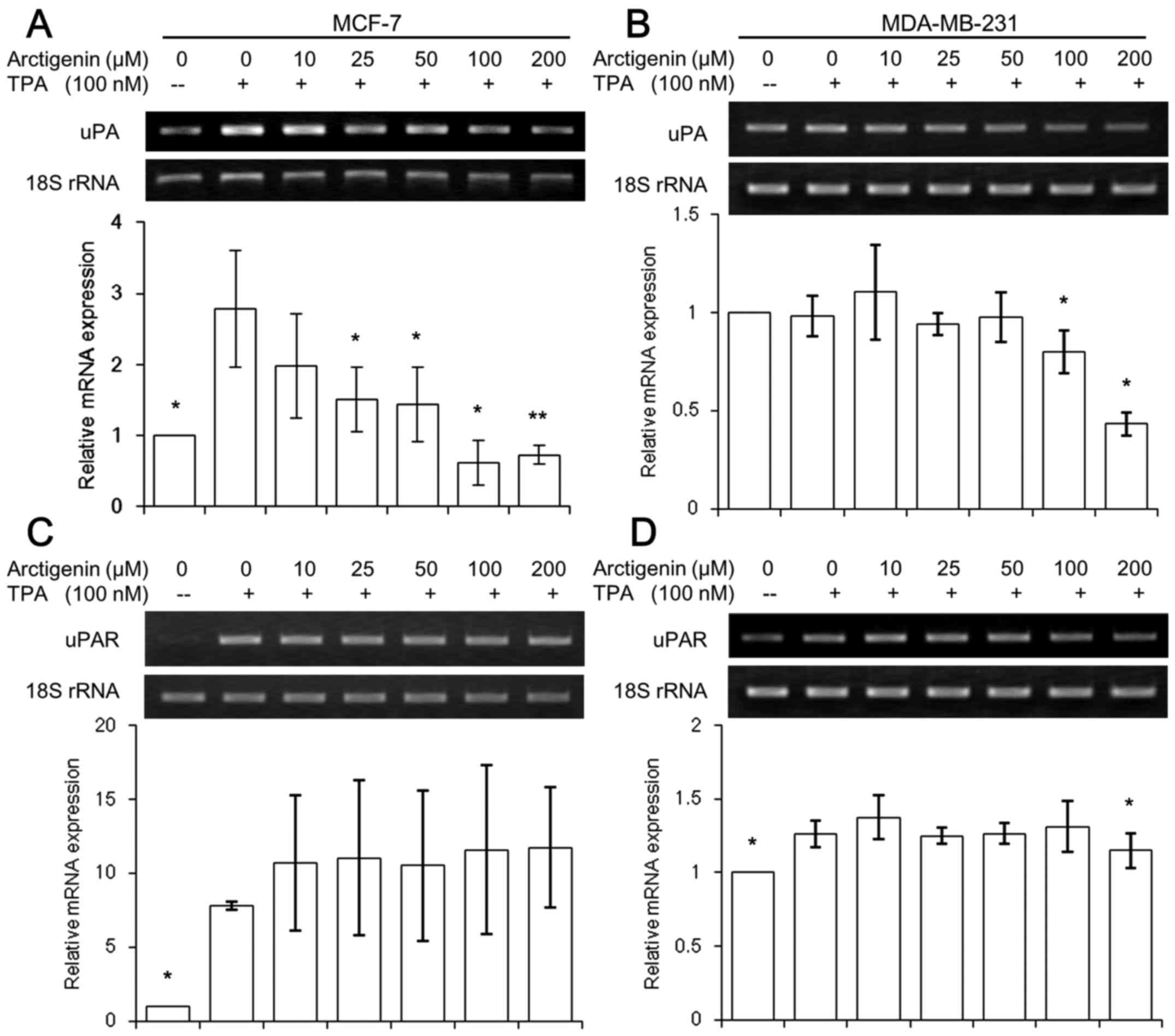
Arctigenin suppressed the expression of
uPA
It is well known that the activation of MMP-9 is
mediated by the active serine protease urokinase-type plasminogen
activator (uPA) and its receptor, uPAR (35). The cleavage of plasminogen to
generate active plasmin was catalyzed by uPA/uPAR, which
facilitates the release of several proteolytic enzymes, including
MMPs. Thus, we tested whether the inhibitory effects of arctigenin
on MMP-9 enzyme results from the suppressed expression of uPA and
uPAR. TPA significantly induced both uPA and uPAR expressions in
MCF-7 cells (Fig. 5A and C), while
the endogenous levels in invasive MDA-MB-231 cells were relatively
high (Fig. 5B and D) and the
effect of TPA in inducing uPA and uPAR expression was very mild
compared with MCF-7 cells. However, both cell lines showed that the
expression of uPA was inhibited by high concentrations of
arctigenin (Fig. 5A and B),
whereas the uPAR levels remained unchanged (Fig. 5C and D).
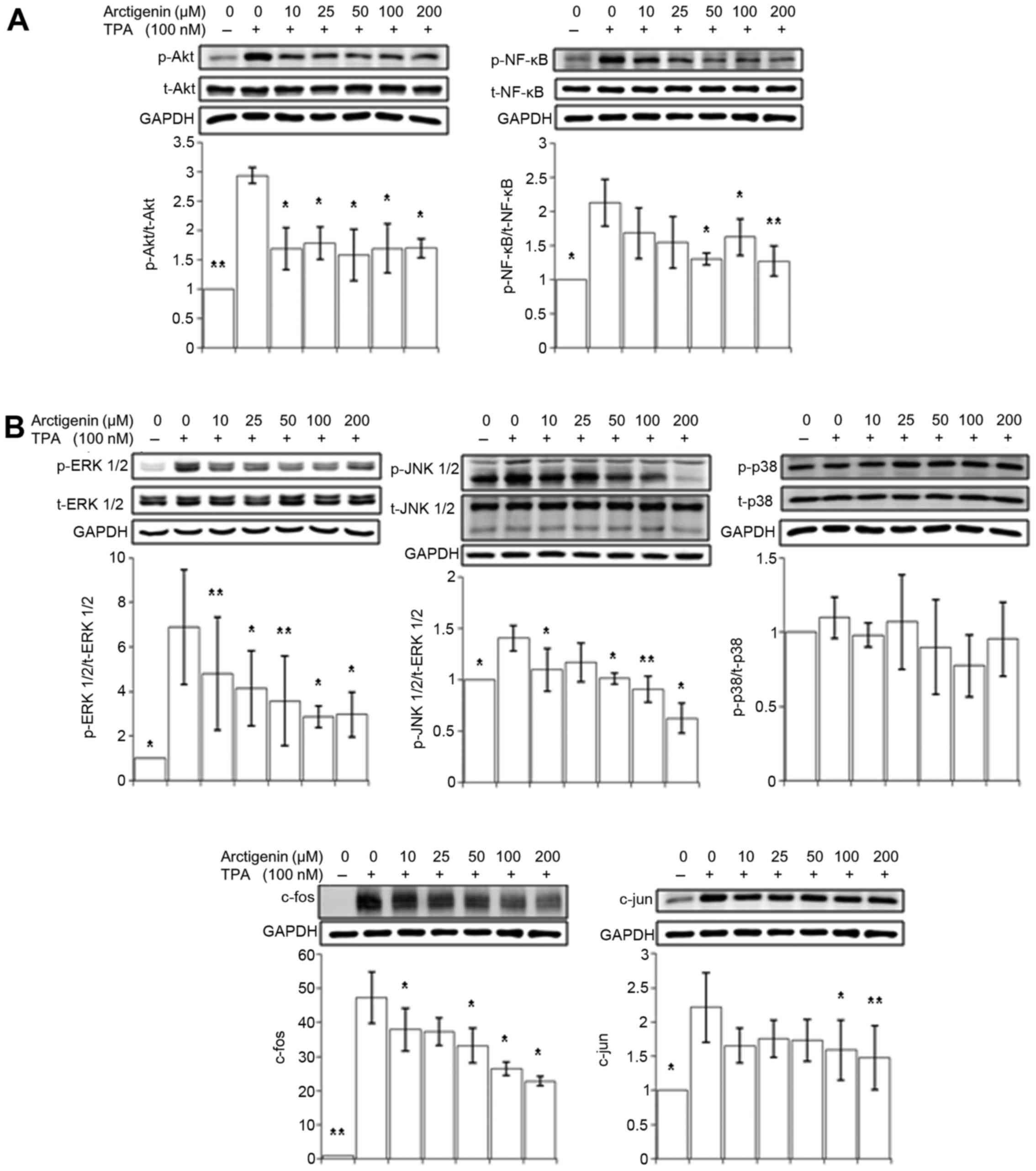
Arctigenin inhibits TPA-induced Akt/NF-κB
and MAPK signaling in MCF-7 human breast cancer cells
Our data so far showed that arctigenin inhibited
cell migration in human breast cancer cells by inhibiting MMP-9 and
uPA. The expression of MMP-9 and uPA can be modulated by various
intracellular upstream signaling cascades, particularly the
PI3K/Akt pathway and MAPK pathways (36). The activation of protein kinase C
(PKC) by TPA induces the activation of two transcription factors,
NF-κB and AP-1; of which, activations are regulated by the PI3K/Akt
and MAPK pathways. To understand the mechanism underlying the
suppression of MMP-9 and uPA by arctigenin, we examined whether the
treatment with arctigenin alter the PI3K/Akt and MAPK pathways.
Since Akt activates the NF-κB transcription factor that
translocates into the nucleus and initiates the transcription of
MMP-9 and uPA, we first tested the inhibitory effect of arctigenin
on the phosphorylation of Akt and NF-κB. As shown in Fig. 6A, the activated/phosphorylated
forms of both Akt and NF-κB, which increased with the TPA
treatment, were efficiently inhibited by arctigenin.
The MAPK pathway consists of the ERK 1/2, JNK 1/2
and p38 signaling pathways. Its activation leads to the
translocation of c-fos and c-jun into the nucleus to form the
transcription factor AP-1, resulting in increased transcription of
uPA and MMP-9. As shown in Fig.
6B, the phosphorylation of ERK 1/2 and JNK 1/2 was
significantly attenuated by arctigenin treatment in a
dose-dependent manner, subsequently leading to a decrease in the
expression of c-fos and c-jun. However, the phosphorylation of p38
was not altered by the treatment with arctigenin, suggesting that
AP-1 transcription activity is suppressed by arctigenin through ERK
1/2 and JNK 1/2 MAPK pathways, rather than through p38 MAPK
pathway.
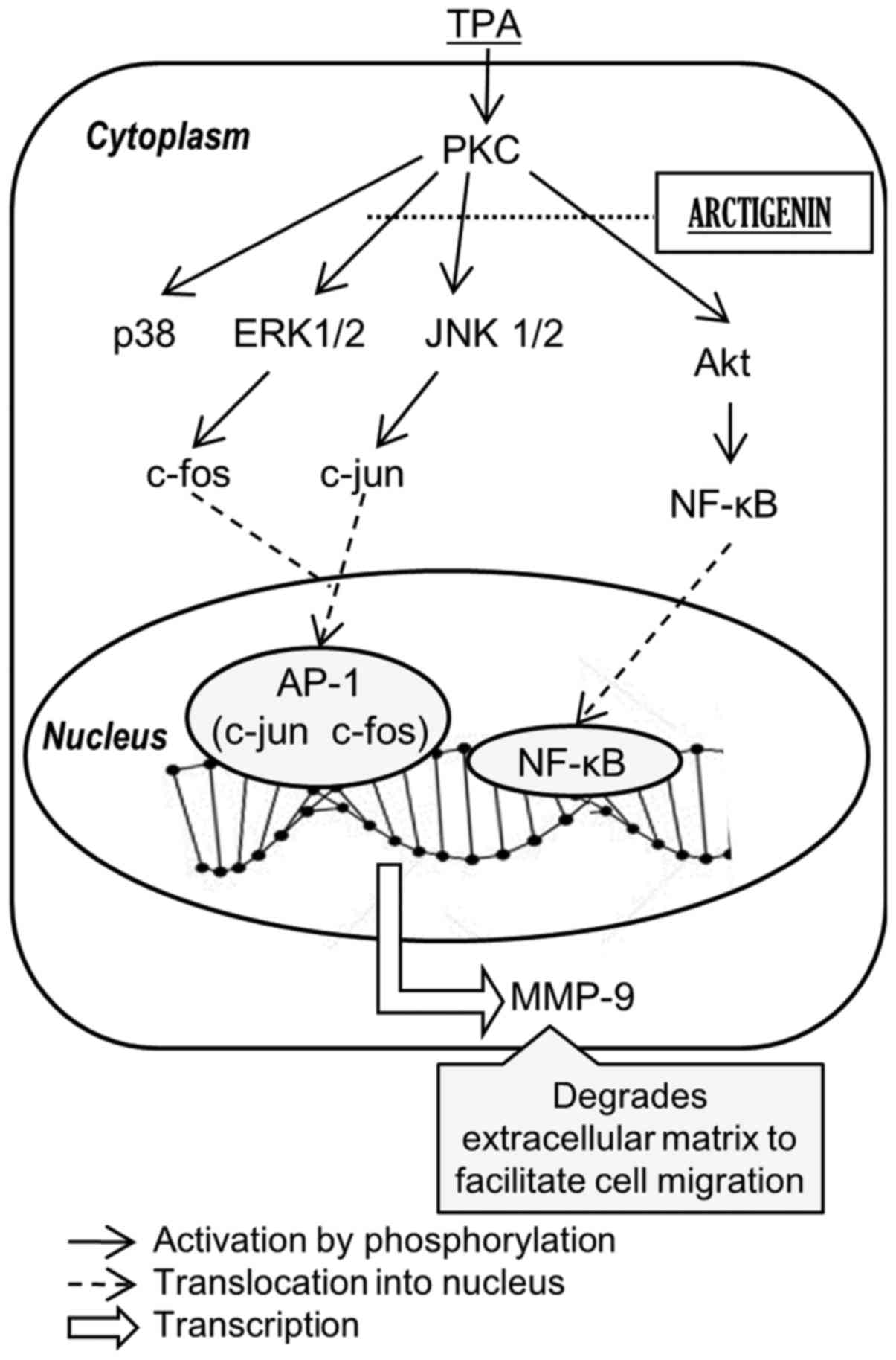
Taken together, our data suggested that the
inhibitory effect of arctigenin on MMP-9 and uPA appears to be
mediated through the inhibition of AP-1 and NF-κB activity, via
suppressing the phosphorylation of Akt, ERK 1/2 and JNK 1/2
(Fig. 7).
Discussion
According to their chemical structures,
phytoestrogens are classified into isoflavones and lignans.
Isoflavones are major constituents in soy products while lignans
are widely distributed in fruits and vegetables. Arctigenin is a
lignan phytoestrogen extracted from greater burdock and is consumed
worldwide as foods and beverages. It is also used in traditional
medicines for its health benefits. Several scientific studies
report the beneficial effects of arctigenin and these include
anti-oxidant, anti-inflammatory and antitumor activities. It has
been shown that arctigenin inhibits the growth of various cancer
cells by inhibiting the mitochondrial respiration (18), induction of apoptosis (11,14,16,20–22)
and cell cycle arrest (11,19–21).
Studies also show that it kills cancer cells, specifically while
sparing the normal cells (37).
However, the effect of arctigenin on breast cancer cells has not
been fully understood, although arctigenin, as a phytoestrogen, has
been reported to have estrogenic properties. Recent studies
reported that arctigenin possesses anticancer effect on ER-negative
MDA-MB-231 cells by the mitochondrial caspase-independent apoptotic
pathway (22). However, they
observed that arctigenin showed neither a proliferative nor an
anti-proliferative effects on ER-positive MCF-7 cells, suggesting
that the antitumor effect of arctigenin may not be related to the
ER signaling pathway.
In the present study, we demonstrated that
arctigenin confers anti-metastatic effects on human breast cancer
cells regardless of the presence of ER. Arctigenin efficiently
inhibited cell migration in highly invasive MDA-MB-231 cells and
also the TPA-induced cell migration in MCF-7 cells. This inhibition
was shown to be dose-dependent, with as little as 10 µM
arctigenin exerting significant inhibition and complete inhibition
with 200 µM arctigenin. This is in accordance with the
anti-migratory capacity of arctigenin previously reported by Zhang
et al (15), where 5
µM arctigenin was found to inhibit cell migration in human
colon cancer cell lines.
Cell migration and invasion are complex cellular
behaviors that require coordination of many signaling pathways.
However, critical proteolytic events must occur early in the
metastatic cascade for the degradation of the basement membrane and
successful intravasation. Almost all cells in tumor and host
environment overexpress one or more of the proteolytic enzymes
required for this purpose.
MMPs are a family of zinc- and calcium-dependent
endoproteinases that are divided into four subclasses based on
their substrate; collagenase, gelatinase, stromelysin and
membrane-associated MMPs. MMP-2 and MMP-9 are the key enzymes due
to their ability to degrade type IV collagen which is a major
component of the basement membrane. It has been reported that the
induction of MMP-9, also known as gelatinase-B and 92-kDa type IV
collagenase, is particularly important for the invasiveness of
human cancers, including breast cancer (38). Therefore, a blockade of
MMP-9-mediated invasion suppresses the metastasis of breast cancer
cells to the other organs. Synthesis and secretion of MMP-9 can be
stimulated by a variety of growth factors and inflammatory
cytokines during pathological processes and by agents, such as the
tumor promoter TPA. The activation of MMP-9 is mediated by active
serine protease uPA and uPAR (35,39).
The cleavage of plasminogen to generate active plasmin is catalyzed
by uPA/uPAR, which facilitates the release of several proteolytic
enzymes, including MMPs.
Our data show that arctigenin decreased MMP-9 at the
transcriptional level with a corresponding suppression in its
activity level, which was observed with gelatinase zymography. The
other metastatic protease uPA was also inhibited by arctigenin,
whereas the uPAR levels remained unchanged. Zhang et al
(15) have previously reported the
inhibition of cell migration and invasion via the Wnt/β-catenin
pathway in human colon cancer cells. They also showed that
arctigenin inhibited MMP-9 along with MMP-2. The activity of MMP-9
in various tumor cells is tightly controlled, with regulation
occurring primarily at the transcription level. The promoter of
human MMP-9 contains cis-acting regulatory elements
that bind with transcription factors such as nuclear factor-kappa B
(NF-κB) or activator protein-1 (AP-1) (36). NF-κB and AP-1 are ubiquitous
eukaryotic transcription factors that can be induced by multiple
stimuli. NF-κB, a heterodimer of p50 and p65, is sequestered in the
cytoplasm under normal conditions due to its association with the
inhibitory protein, IκBα. Stimulation by inflammatory cytokines or
tumor promoters leads to the dissociation of IκBα from NF-κB and
the activated NF-κB translocates into the nucleus, binding to the
promoter region of MMP-9 and leading to gene expression.
AP-1 is a nuclear transcription factor that is comprised of
homodimers and heterodimers of the members of the Fos and Jun
family proteins. NF-κB and AP-1 dependent MMP-9 expression is
regulated by Akt and MAPKs, respectively. In this study, we proved
that arctigenin can inhibit metastasis of breast cancer cells
through these pathways (Fig. 7).
The activated Akt and NF-κB, within 1 h of TPA treatment, was
significantly inhibited by arctigenin. Furthermore, the
phosphorylation of ERK 1/2 and JNK 1/2 were significantly
attenuated by the treatment with arctigenin in a dose-dependent
manner, which subsequently led to a decrease in the expression of
c-fos and c-jun. However, the phosphorylation of p38 was not
altered by the treatment with arctigenin. Although TPA is known to
activate all three MAPKs i.e. ERK 1/2, JNK1/2 and p38, several
studies showed that the activation of p38 depends on concentration
or time-points of TPA treatment or cell lines (40,41).
Since activation of p38 was not observed in this study, we presumed
that TPA treatment for 24 h was unable to induce p38 signaling in
MCF-7 human breast cancer cells. This suggests that the AP-1
transcription activity is suppressed by arctigenin through ERK 1/2
and JNK 1/2 MAPK pathways, rather than through the p38 MAPK
pathway.
Taken together, these data suggest that arctigenin
confers anti-metastatic effects by inhibiting MMP-9 and uPA via the
Akt, NF-κB and MAPK signaling pathways on breast cancers,
regardless of ER expression. Therefore, we propose that consumption
of burdock tea and arctigenin-enriched food products maybe a safe
supplement for breast cancer patients in preventing metastasis.
Abbreviations:
|
ER
|
estrogen receptor
|
|
RT-PCR
|
reverse transcriptase-polymerase chain
reaction
|
|
TPA
|
12-O-tetradecanoylphorbol-13-acetate
|
|
MMP
|
matrix metalloprotease
|
|
TIMP
|
tissue inhibitor of
metalloprotease
|
|
uPA
|
urokinase plasminogen activator
|
|
uPAR
|
urokinase plasminogen activator
receptor
|
|
Akt
|
protein kinase B
|
|
NF-κB
|
nuclear factor kappa B
|
|
MAPK
|
mitogen-activated protein kinase
|
|
ERK
|
extracellular signal-regulated
kinase
|
|
JNK
|
c-Jun N-terminal kinase
|
|
AP-1
|
activator protein-1
|
|
PKC
|
protein kinase C
|
|
GAPDH
|
glyceraldehyde-3-phosphate
dehydrogenase
|
Acknowledgments
The present study was supported by the Basic Science
Research Program through the National Research Foundation of Korea
(NRF) funded by the Ministry of Education
(2015R1D1A1A01058841).
References
|
1
|
Gao H, Li G, Zhang J and Zeng J:
Arctigenin: A lignan from Arctium lappa. Acta Crystallogr Sect E
Struct Rep Online. 64:o15382008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kim Y, Hollenbaugh JA, Kim DH and Kim B:
Novel PI3K/Akt inhibitors screened by the cytoprotective function
of human immunodeficiency virus type 1 Tat. PLoS One. 6:e217812011.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hayashi K, Narutaki K, Nagaoka Y, Hayashi
T and Uesato S: Therapeutic effect of arctiin and arctigenin in
immunocompetent and immunocompromised mice infected with influenza
A virus. Biol Pharm Bull. 33:1199–1205. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Swarup V, Ghosh J, Mishra MK and Basu A:
Novel strategy for treatment of Japanese encephalitis using
arctigenin, a plant lignan. J Antimicrob Chemother. 61:679–688.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lee JY and Kim CJ: Arctigenin, a
phenylpropanoid dibenzylbutyrolactone lignan, inhibits type I–IV
allergic inflammation and pro-inflammatory enzymes. Arch Pharm Res.
33:947–957. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kang HS, Lee JY and Kim CJ:
Anti-inflammatory activity of arctigenin from Forsythiae Fructus. J
Ethnopharmacol. 116:305–312. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yao X, Li G, Lü C, Xu H and Yin Z:
Arctigenin promotes degradation of inducible nitric oxide synthase
through CHIP-associated proteasome pathway and suppresses its
enzyme activity. Int Immunopharmacol. 14:138–144. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tsai WJ, Chang CT, Wang GJ, Lee TH, Chang
SF, Lu SC and Kuo YC: Arctigenin from Arctium lappa inhibits
interleukin-2 and interferon gene expression in primary human T
lymphocytes. Chin Med. 6:122011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cho MK, Park JW, Jang YP, Kim YC and Kim
SG: Potent inhibition of lipopolysaccharide-inducible nitric oxide
synthase expression by dibenzylbutyrolactone lignans through
inhibition of I-kappaBalpha phosphorylation and of p65 nuclear
translocation in macrophages. Int Immunopharmacol. 2:105–116. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cho MK, Jang YP, Kim YC and Kim SG:
Arctigenin, a phenylpropanoid dibenzylbutyrolactone lignan,
inhibits MAP kinases and AP-1 activation via potent MKK inhibition:
The role in TNF-alpha inhibition. Int Immunopharmacol. 4:1419–1429.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jeong JB, Hong SC, Jeong HJ and Koo JS:
Arctigenin induces cell cycle arrest by blocking the
phosphorylation of Rb via the modulation of cell cycle regulatory
proteins in human gastric cancer cells. Int Immunopharmacol.
11:1573–1577. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Susanti S, Iwasaki H, Itokazu Y, Nago M,
Taira N, Saitoh S and Oku H: Tumor specific cytotoxicity of
arctigenin isolated from herbal plant Arctium lappa L. J Nat Med.
66:614–621. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Awale S, Lu J, Kalauni SK, Kurashima Y,
Tezuka Y, Kadota S and Esumi H: Identification of arctigenin as an
antitumor agent having the ability to eliminate the tolerance of
cancer cells to nutrient starvation. Cancer Res. 66:1751–1757.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim JY, Hwang JH, Cha MR, Yoon MY, Son ES,
Tomida A, Ko B, Song SW, Shin-ya K, Hwang YI, et al: Arctigenin
blocks the unfolded protein response and shows therapeutic
antitumor activity. J Cell Physiol. 224:33–40. 2010.PubMed/NCBI
|
|
15
|
Zhang S, Li J, Song S, Li J, Tong R, Zang
Z, Jiang Q and Cai L: Integrated in silico and experimental methods
revealed that Arctigenin inhibited angiogenesis and HCT116 cell
migration and invasion through regulating the H1F4A and
wnt/β-catenin pathway. Mol Biosyst. 11:2878–2884. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hausott B, Greger H and Marian B:
Naturally occurring lignans efficiently induce apoptosis in
colorectal tumor cells. J Cancer Res Clin Oncol. 129:569–576. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gu Y, Scheuer C, Feng D, Menger MD and
Laschke MW: Inhibition of angiogenesis: A novel antitumor mechanism
of the herbal compound arctigenin. Anticancer Drugs. 24:781–791.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gu Y, Qi C, Sun X, Ma X, Zhang H, Hu L,
Yuan J and Yu Q: Arctigenin preferentially induces tumor cell death
under glucose deprivation by inhibiting cellular energy metabolism.
Biochem Pharmacol. 84:468–476. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Susanti S, Iwasaki H, Inafuku M, Taira N
and Oku H: Mechanism of arctigenin-mediated specific cytotoxicity
against human lung adenocarcinoma cell lines. Phytomedicine.
21:39–46. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang S, Ma J, Xiao J, Lv X, Li X, Yang H,
Liu Y, Feng S and Zhang Y: Arctigenin anti-tumor activity in
bladder cancer T24 cell line through induction of cell-cycle arrest
and apoptosis. Anat Rec (Hoboken). 295:1260–1266. 2012. View Article : Google Scholar
|
|
21
|
Huang K, Li LA, Meng YG, You YQ, Fu XY and
Song L: Arctigenin promotes apoptosis in ovarian cancer cells via
the iNOS/NO/STAT3/survivin signalling. Basic Clin Pharmacol
Toxicol. 115:507–511. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hsieh CJ, Kuo PL, Hsu YC, Huang YF, Tsai
EM and Hsu YL: Arctigenin, a dietary phytoestrogen, induces
apoptosis of estrogen receptor-negative breast cancer cells through
the ROS/p38 MAPK pathway and epigenetic regulation. Free Radic Biol
Med. 67:159–170. 2014. View Article : Google Scholar
|
|
23
|
Jin JS, Lee JH and Hattori M: Ligand
binding affinities of arctigenin and its demethylated metabolites
to estrogen receptor alpha. Molecules. 18:1122–1127. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Matsumura A, Ghosh A, Pope GS and Darbre
PD: Comparative study of oestrogenic properties of eight
phytoestrogens in MCF7 human breast cancer cells. J Steroid Biochem
Mol Biol. 94:431–443. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Rice S and Whitehead SA: Phytoestrogens
and breast cancer–promoters or protectors? Endocr Relat Cancer.
13:995–1015. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lephart ED: Modulation of aromatase by
phytoestrogens. Enzyme Res. 2015:5946562015. View Article : Google Scholar
|
|
27
|
Whitehead SA and Lacey M: Phytoestrogens
inhibit aromatase but not 17beta-hydroxysteroid dehydrogenase (HSD)
type 1 in human granulosa-luteal cells: Evidence for FSH induction
of 17beta-HSD. Hum Reprod. 18:487–494. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Low YL, Taylor JI, Grace PB, Dowsett M,
Scollen S, Dunning AM, Mulligan AA, Welch AA, Luben RN, Khaw KT, et
al: Phytoestrogen exposure correlation with plasma estradiol in
postmenopausal women in European Prospective Investigation of
Cancer and Nutrition-Norfolk may involve diet-gene interactions.
Cancer Epidemiol Biomarkers Prev. 14:213–220. 2005.PubMed/NCBI
|
|
29
|
DeSantis C, Ma J, Bryan L and Jemal A:
Breast cancer statistics, 2013. CA Cancer J Clin. 64:52–62. 2014.
View Article : Google Scholar
|
|
30
|
Jung KW, Won YJ, Kong HJ, Oh CM, Seo HG
and Lee JS: Cancer statistics in Korea: Incidence, mortality,
survival and prevalence in 2010. Cancer Res Treat. 45:1–14. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Straatman K: Wound healing assay.
University of Leicester, College of Medicine, Biological Sciences
and Psychology, Advanced Imaging Facilities; 2008
|
|
32
|
Loughlin DT and Artlett CM: Modification
of collagen by 3-deoxyglucosone alters wound healing through
differential regulation of p38 MAP kinase. PLoS One. 6:e186762011.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lee K, Shin J, Kwon Y, Moon D and Nam K:
Suppression of cancer progression and metastasis in HT-29 human
colorectal adenocarcinomas by deep sea water. Biotechnol Bioprocess
Eng. 18:194–200. 2013. View Article : Google Scholar
|
|
34
|
Mehner C, Hockla A, Miller E, Ran S,
Radisky DC and Radisky ESR: Tumor cell-produced matrix
metalloproteinase 9 (MMP-9) drives malignant progression and
metastasis of basal-like triple negative breast cancer. Oncotarget.
5:2736–2749. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Holst-Hansen C, Johannessen B,
Høyer-Hansen G, Rømer J, Ellis V and Brünner N: Urokinase-type
plasminogen activation in three human breast cancer cell lines
correlates with their in vitro invasiveness. Clin Exp Metastasis.
14:297–307. 1996.PubMed/NCBI
|
|
36
|
Lungu G, Covaleda L, Mendes O,
Martini-Stoica H and Stoica G: FGF-1-induced matrix
metalloproteinase-9 expression in breast cancer cells is mediated
by increased activities of NF-kappaB and activating protein-1. Mol
Carcinog. 47:424–435. 2008. View Article : Google Scholar
|
|
37
|
Moritani S, Nomura M, Takeda Y and
Miyamoto K: Cytotoxic components of bardanae fructus (goboshi).
Biol Pharm Bull. 19:1515–1517. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
McCawley LJ and Matrisian LM: Matrix
metalloproteinases: Multifunctional contributors to tumor
progression. Mol Med Today. 6:149–156. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Duffy MJ: The urokinase plasminogen
activator system: Role in malignancy. Curr Pharm Des. 10:39–49.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Cheepala SB, Yin W, Syed Z, Gill JN,
McMillian A, Kleiner HE, Lynch M, Loganantharaj R, Trutschl M, Cvek
U, et al: Identification of the B-Raf/Mek/Erk MAP kinase pathway as
a target for all-trans retinoic acid during skin cancer promotion.
Mol Cancer. 8:272009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lilleholt LL, Johansen C, Arthur JS,
Funding A, Bibby BM, Kragballe K and Iversen L: Role of p38
mitogen-activated protein kinase isoforms in murine skin
inflammation induced by 12-O-tetradecanoylphorbol 13-acetate. Acta
Derm Venereol. 91:271–278. 2011. View Article : Google Scholar : PubMed/NCBI
|