Introduction
Lung cancer is the leading cause of cancer-related
mortality worldwide, with 1.8 million deaths predicted in 2018
(1). Approximately 85% of all lung
cancers are non-small cell lung cancer (NSCLC), with adenocarcinoma
(AC) being the major histological subtype (2). In NSCLC, the primary cause of
mortality is distant metastasis resulting from hematogenous
dissemination of cancer cells (3).
Through this route, NSCLC cells spread to the brain, contralateral
lung, bones, liver and the suprarenal glands, with consequent
impact on patient's survival (3,4). The
high mortality of NSCLC means there is an urgent requirement to
identify mechanisms associated with NSCLC progression, in order to
aid early determination of its metastatic potential and proper
staging. Cancer metastasis is a multifaceted process that comprises
multiple steps. After invading the stroma, tumour cells require the
establishment of new vasculature from the pre-existing vasculature
via angiogenesis to provide nutrition and an oxygen supply
(5). Fast tumour growth requires
that invasive tumour cells adapt to the hostile hypoxic
microenvironment and protect from immune cell attack; otherwise,
they do not survive (6). These
factors lead to dramatic changes in cell signalling and protein
expression that enables the cells to surpass the challenges of
leaving the primary tumour, migrate to distant sites and establish
a metastatic focus (7). One of the
most critical steps for metastasis is the ability of circulating
cancer cells to adhere the vascular endothelium (Fig. 1A). The initial adhesive
interactions are dictated by the calcium-dependent binding of
circulating cancer cells to endothelial E-selectins expressed in
microvasculature at inflammatory sites (8,9).
E-selectin ligands are terminal lactosaminyl tetrasaccharides,
prototypically the sialyl Lewis x (sLex) and sialyl
Lewis a (sLea) glycan antigens, displayed on cell
surface protein or lipid scaffolds (10). In proteins, these structures are
found at terminal ends on the β-1,6 branching of N-glycans
or O-glycans. Patients with NSCLC are reported to
overexpress sLex and sLea antigens on tumour
tissues and serum proteins (11,12).
The higher expression of sLex and sLea in
NSCLC has been associated with enhanced metastatic activity and
poor prognosis (13,14); however, the contribution of these
antigens to metastasis remains unclear. Additionally, the mechanism
driving sLex and sLea overexpression in NSCLC
is poorly understood. Nevertheless, in cancer, the expression of
the glycosyltransferases involved in sLex and
sLea biosynthesis is controlled by elements in the
tumour microenvironment, such as hypoxia and oncogene expression
(15). This suggests that the
hostile microenvironment of a primary tumour triggers
sLex and sLea expression, enabling cancer
cells to adapt and establish adhesive interactions with
endothelium. sLex and sLea are isomers whose
structure consists in α2,3-sialylated, and α1,3/4-fucosylated type
2 or type 1 lactosamine chains, respectively (Fig. 1B) (16,17).
The α2,3-sialyltransferases (ST3GAL3, ST3GAL4, and ST3GAL6) and the
α1,3-fucosyltransferases (FUT3, FUT4, FUT5, FUT6 and FUT7) can
respectively catalyse terminal sialylation and fucosylation steps
(18). The same enzymes may also
be involved in other glycosylation steps that compete with
sLex and sLea antigen biosynthesis, as FUT4
efficiently catalyses the synthesis of non-sialylated antigens,
such as Lewis x and Lewis y (19,20).
The activity of each enzyme is generally tissue-specific and
depends mostly on its expression level.
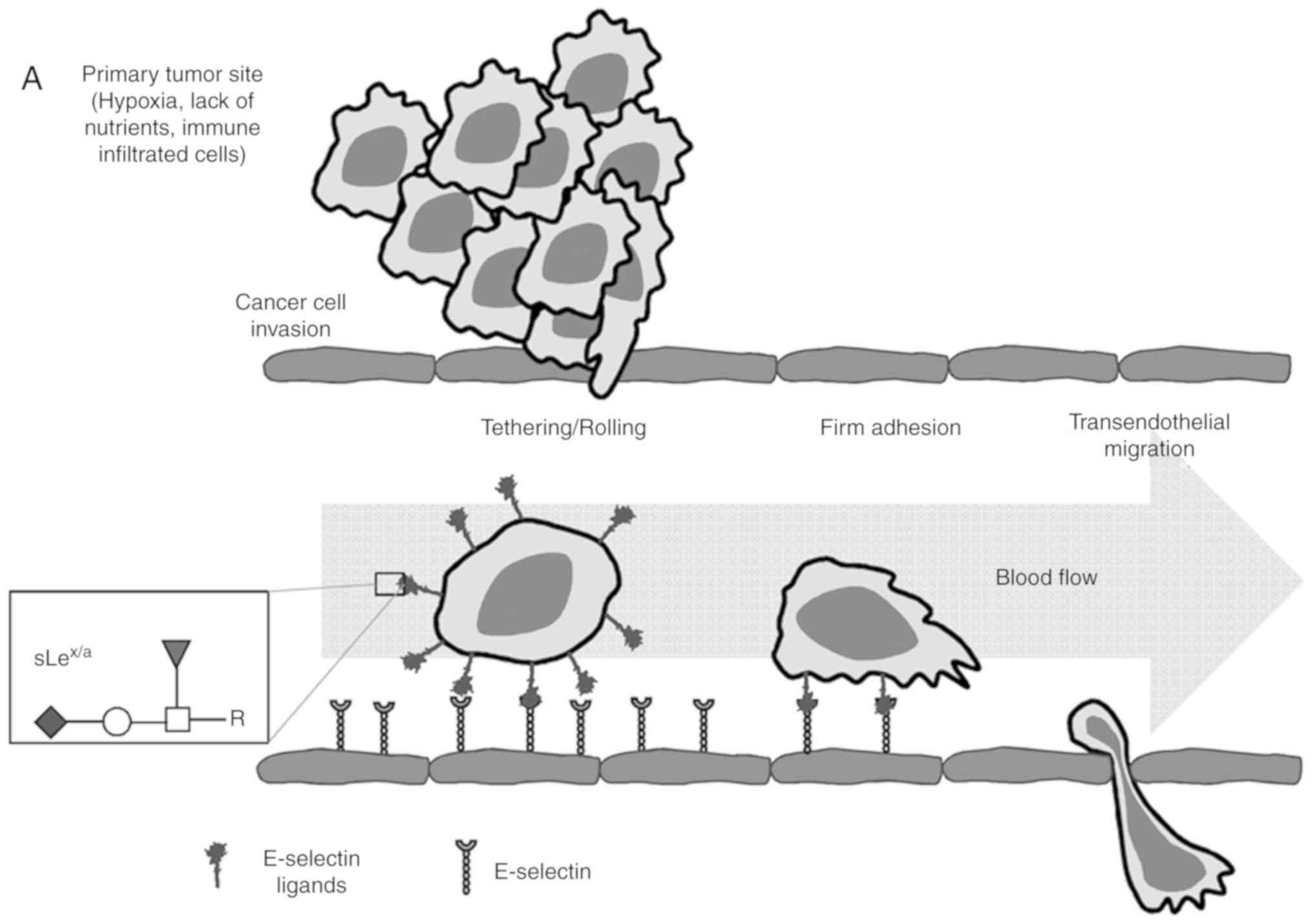 | Figure 1Schematic representation of the
multistep metastatic process in cancer and main structures
involved. (A) Contribution of
sLex/sLea antigens in facilitating cell
adhesion to E-selectins. There are four main steps in the
extravasation process, including tethering and rolling, integrin
activation, firm adhesion and transendothelial migration.
Metastatic dissemination is facilitated by interactions between
tumour cells and endothelium in distant tissues. These interactions
are mediated by E-selectins expressed by activated endothelial
cells and the corresponding E-selectin ligands expressed on the
cell surface of cancer cells. These ligands are prototypically the
sLex and sLea antigens, displayed on cell
surface proteins or lipid scaffolds. (B) Structure and schematic
representation of the biosynthesis of sialyl Lewis antigens.
sLea and sLex antigens are sialofucosylated
isomer tetrasaccharides, derived from type 1 or type 2 sugar
chains, respectively, attached to N- or O-glycan
residues. The two types of structures are sialylated by the action
of the indicated α2,3-sialyltransferases and successively
fucosylated by the action of the FUT3 (in type 1 chains), and FUT3,
4, 5, 6 or 7 (in type 2 chains). FUT, fucosyltransferase;
sLea/x, sialyl Lewis a/x. |
As aforementioned, in spite of the overexpression of
sLex and sLea in NSCLC cells, it is unknown
whether they confer adhesive capacity of cancer cells to the
endothelium. sLex and sLea functional roles
depend on their density and availability at the cell surface,
specifically their presentation by specific scaffolds to
potentially be recognised by E-selectins expressed on activated
endothelial cells. In the present study, it was hypothesised that
sLex/sLea glycans serve a role in NSCLC cell
adhesion to E-selectins. It was further hypothesised that it is
carried by protein scaffolds specific to NSCLC. To address this, in
this study, sLex/sLea expression and
E-selectin reactivity were compared in patient-derived tumour and
matched normal lung tissues. Furthermore, the expression levels of
fucosyltransferases and sialyltransferases involved in the
biosynthesis of sLex and sLea glycans were
analysed. Additionally, the ability of these ligands to adhere to
E-selectin was evaluated, and carcinoembryonic antigen (CEA) was
identified as a protein scaffold of E-selectin ligands in
NSCLC.
Improved understanding of the glycosidic alterations
occurring throughout tumour progression, as well as the
pathophysiological role of sLex/sLea
glycans-associated protein scaffolds in tumour cell adhesion to
E-selectins, will aid the identification of potential therapeutic
targets and may enable improved prediction of tumour progression
and metastasis formation in NSCLC.
Materials and methods
Patient and tissue specimens
The present study involved 18 consecutive patients
with NSCLC who underwent lobectomy due to lung cancer between July
2011 and January 2012 at the Thoracic Surgery Department of
Hospital Pulido Valente (Lisbon, Portugal). The inclusion criteria
for the patients enrolled in this study were as follows: i)
Patients with age ≥18 years; ii) suspected or proven lung cancer
with indication for pulmonary resection; and iii) histology of lung
adenocarcinoma or squamous cell carcinoma. The exclusion criteria
were: i) Patients with age <18 years; ii) previous chemotherapy
or radiotherapy; iii) histology compatible with small cell lung
cancer, large cell lung cancer, lung metastases or benign disease;
and iv) pregnant women. A total of 14 patients were further
diagnosed with AC and four with squamous cell carcinoma (SCC). The
median age of the patients was 65 years (48-83 years); 13 patients
were male. For each patient, fragments of pulmonary tumour tissue
and normal lung tissue were collected and immediately stored in
liquid nitrogen until further processing. The determination of the
histological type was performed by the Pathology Department from
the same hospital. Tumour staging was classified according to the
TNM classification system based on the International Union Against
Cancer 8th edition (21). A 5-year
follow-up of the patients after the initial surgical procedure was
conducted, and clinical data were collected into a database.
For bone metastasis analysis, sections of two cases
of lung cancer with the corresponding metastasis, from male
patients aged 58 and 86 years, were used. Samples were collected,
in the first case from the left lung and in the second case from
the right lower lobe. Bone metastasis tissues were taken from a
lumbar spine injury (L5-S1) and from the right clavicle,
respectively.
The study was approved by the Ethics Committee of
Centro Hospitalar Lisboa Norte. Written informed consent was
obtained from all patients. A summary of the clinical data is
available in Table SI.
Immunoblotting and immunoprecipitation
analysis
Whole tissue lysates were obtained by homogenising
patient tissues in lysis buffer [150 mM NaCl, 2 mM
CaCl2, 2% Nonidet P-40 and protease inhibitors (Roche
Diagnostics)]. After centrifugation for 2 min at 17,000 × g and
4°C, the quantity of protein within the supernatant was estimated
using a Pierce™ Bicinchoninic Acid Protein Assay kit (Pierce;
Thermo Fisher Scientific, Inc.) and stored for further use.
Immunoprecipitated CEA was obtained by pre-clearing tissue lysates
with protein G-agarose, followed by incubation for 2 h at 4°C with
1 µg/µl of anti-human CEA (CD66E) monoclonal antibody
(mAb; cat. no. 21278661; ImmunoTools GmbH). The immunoprecipitate
was collected with protein G-agarose beads, boiled and the released
proteins were analysed via western blotting.
After an extensive search in the literature and
several attempts with the present cohort, it was concluded that
there is no consensus regarding the optimal loading control to use
for NSCLC studies, with marked upregulation or downregulation of
some of the most used housekeeping genes (β-tubulin, β-actin,
GAPDH) in human lung tumour tissues compared with normal lung
tissues (22-24). For this reason and due to sample
limitation, these loading controls were not used in dot blotting
and western blotting experiments. As the best possible alternative,
an exact amount of protein was always loaded for all
experiments.
For dot blot analysis, 10 µg of tissue
lysates were applied to a nitrocellulose membrane (GE Healthcare
Life Sciences). For western blot analysis, 20 µg of tissue
lysates or immuno-precipitates were electrophoresed under reducing
conditions on an 8% polyacrylamide gel and transferred to a
polyvi-nylidene difluoride membrane (Bio-Rad Laboratories, Inc.).
Membranes were incubated in blocking solution [10% non-fat milk
diluted in PBS-0.1% Tween 20 for dot blotting, and TBS containing
0.1% Tween-20 (TBS-T) for western blotting experiments] overnight
at 4°C under agitation. For sLex/sLea and CEA
detection, nitrocellulose or PVDF membranes were stained with
HECA-452 mAb (1:1,000; cat. no. 321302; BioLegend, Inc.) or
anti-human CD66E mAb (1:1,000; cat. no. 21278661; ImmunoTools
GmbH), followed by staining with a horseradish peroxidase
(HRP)-conjugated secondary mAb [anti-rat IgM-HRP for HECA-452
staining (1:2,500; cat. no. 3080-05; SouthernBiotech); anti-mouse
Ig-HRP for CD66E staining (1:2,500; cat. no. 554002; BD Pharmingen;
BD Biosciences)]. E-selectin ligand staining was performed using a
3-step protocol, in the presence of 2 mM CaCl2, which
included staining with soluble mouse E-selectin-human Fc Ig chimera
(E-Ig; 1:500; cat. no. 575-ES-100; R&D Systems, Inc.), followed
by the addition of rat anti-mouse E-selectin (CD62E) mAb (1:1,000;
cat. no. 550290; BD Pharmingen; BD Biosciences) and then
HRP-conjugated anti-rat IgG (1:2,000; cat. no. 3030-05;
SouthernBiotech) (32). Membranes
were incubated with Lumi-Light Western Blotting Substrate (Roche
Diagnostics) according to the manufacturer's protocols and detected
with autoradiography film. All blots were replicated at least
twice. Image analysis was performed using ImageJ 1.48v software
(National Institutes of Health), and arbitrary units were defined
based on the intensity detected by the software.
Gene expression measurements
Tissue samples were homogenised and total RNA was
isolated following the instructions of the NZY Total RNA Isolation
kit (NZYTech). cDNA synthesis was performed using a High-Capacity
cDNA Reverse Transcription kit (Applied Biosystems; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocols.
Reverse transcription-quantitative (RT-q)PCR was performed using
TaqMan probes methodology (6-carboxyfluorescein as a
fluorescent dye) and TaqMan Fast Universal PCR Master Mix (Applied
Biosystems; Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocols and in triplicates. The thermal cycling
conditions used were as follows: 1 cycle of 2 min at 50°C, 1 cycle
of 10 min at 95°C, and 50 cycles of 15 sec at 95°C and 1 min at
60°C. For each primer/probe set, the Assay ID (Applied Biosystems;
Thermo Fisher Scientific, Inc.) was the following: FUT3,
Hs00356857_m1; FUT4, Hs01106466_s1; FUT5,
Hs00704908_s1; FUT6, Hs00173404_m1; FUT7,
Hs00237083_m1; ST3GAL3, Hs00196718_m1; ST3GAL4,
Hs00272170_m1; and ST3GAL6, Hs00196086_m1. mRNA expression
was normalised using the geometric mean of the expression of the
endogenous controls, ACTB (Hs99999903_m1) and GAPDH
(Hs99999905_m1). The relative mRNA level of expression was computed
as a permillage fraction (‰), calculated using the 2−ΔCq
×1,000 formula (25-27), which infers the number of mRNA
molecules of the gene of interest, for every 1,000 molecules of
endogenous controls. RT-qPCR was performed in a 7500 Fast Real-Time
PCR System (Applied Biosystems; Thermo Fisher Scientific, Inc.),
and the results were analysed using Sequence Detection Software
version 1.3 (Applied Biosystems; Thermo Fisher Scientific,
Inc.).
α1,3-FUT activity assay
α1,3-FUT activity was measured in whole tissue
lysates. The assay mixture contained 50 mM Na/cacodylate buffer pH
6.5, 15 mM MnCl2, 0.5% Triton X-100, 5 mM ATP, 0.1 mM
unlabelled GDP-fucose from Sigma-Aldrich (Merck KGaA), 55000 dpm
GDP-[14C] fucose (PerkinElmer, Inc.) and 300 µg
fetuin (Sigma-Aldrich; Merck KGaA), theoretically corresponding to
0.8 mM Siaα2,3Galβ1,4GlcNAc-R acceptor sites. The enzyme reaction
was performed in triplicate at 37°C for 2 h, and then the products
were precipitated, washed and counted by liquid scintillation.
Controls without the acceptor (fetuin) were run in parallel, and
the incorporation was subtracted. Homogenates of COLO-205 cells
(28) (kindly provided by
Professor Fabio Dall'Olio, University of Bologna, Italy) were used
as positive controls.
Adhesion assays
Chinese hamster ovary (CHO) cells transfected either
with cDNA encoding the full length of the human E-selectin (CHO-E)
or with a mock empty pMT2 vector (CHO-mock; kindly provided by
Professor Robert Sackstein, Brigham And Women's Hospital, Harvard
Medical School) (29) were grown
in a humidified atmosphere of 5% CO2 at 37°C in Minimum
Essential Medium (MEM; Sigma-Aldrich; Merck KGaA) supplemented with
10% of heat-inactivated fetal bovine serum, 2 mM of L-glutamine,
100 µg/ml of penicillin/streptomycin, 1 mM of sodium
pyruvate and 0.1 mM of MEM non-essential amino acids solution (all
from Gibco; Thermo Fisher Scientific, Inc.). Adhesion assays were
performed based on the modified Stamper-Woodruff binding assay that
mimics blood flow interactions (30,31).
Briefly, total tissue lysates or immunoprecipitated proteins were
spotted on glass slides, dried and blocked with 1% BSA
(Sigma-Aldrich; Merck KGaA) for 1 h at room temperature (RT).
CHO-mock or CHO-E cells, resuspended in Hank's Balanced Salt
Solution (HBSS) containing 2 mM CaCl2 (HBSS-Ca) or 5 mM
EDTA (HBSS-EDTA; negative control), were overlaid onto a protein
spot on the glass slides. In certain cases, CHO-E cells were
previously incubated with 20 µg/ml of function-blocking
anti-CD62E (clone 68-5H11; cat. no. 555648; BD Biosciences) or
isotype control mAb (clone mopc-21; cat. no. 400102; BioLegend,
Inc.). Slides were then incubated with orbital rotation at 80 RPM
for 30 min at 4°C, and subsequently placed in HBSS-Ca or HBSS-EDTA
to drain non-adherent cells. After fixing the cells with 3%
glutaraldehyde for 10 min at 4°C, the adherent cells were examined
under a phase contrast microscope (Nikon Digital Eclipse C1 system;
magnification, ×100; Nikon Corporation), and representative
photomicrographs (3 frames per sample) were acquired for analysis.
The number of cells adherent to glass slides and observed in each
photomicrograph was counted using ImageJ 1.48v software (32).
Flow cytometry: The cell surface expression of
E-selectin was analysed in CHO-E and CHO-mock cells using
anti-E-selectin monoclonal antibody (5 µl; clone 68-5H11,
cat. no. 555648; BD Biosciences), followed by a anti-mouse Ig-FITC
secondary antibody (1 µl; cat. no. f0479; Dako; Agilent
Technologies, Inc.). Antibody staining was performed for 30 min at
4°C followed by incubation with fluorescent-labelled secondary
antibody (1 µl) for 15 min at RT in the dark. Background
levels were determined in control assays by incubating cell
suspensions with isotype control mAb (5 µl; clone mopc-21;
cat. no. 400102; BioLegend, Inc.) and fluorescent-labelled
secondary antibody. The experiments were performed in an
Attune® Acoustic Focusing Cytometer (Applied Biosystems;
Thermo Fisher Scientific, Inc.) and the results were analysed using
the program FlowJo v10.0.7 (FlowJo, LLC).
Immunohistochemistry
Tissue specimens were fixed in 10% formalin for 24 h
at 4°C and after embedded in paraffin. Paraffin-embedded sections
of tumour tissue (2 µm) were submitted to antigen retrieval
by heating at 94°C in Trilogy pre-treatment solution (Cell Marque;
Merck KGaA) for 20 min. After incubation with peroxidase block
solution (Atom Scientific Ltd.), sections were stained with
anti-CD66E mAb (1:100; cat. no. 21278661; ImmunoTools GmbH) or
anti-sLex/sLea HECA-452 mAb (1:50; cat. no.
321302; Biolegend, Inc.) for 1 h in Diamond Antibody Reagent (cat.
no. 938B-09; Cell Marque) containing 1% BSA. For the washings,
TBS-T was used. For E-selectin staining, E-Ig chimera was used (0.5
µg/100 µl; cat. no. 575-ES-100; R&D Systems,
Inc.) for 30 min, followed by staining with anti-CD62E mAb (1:50;
cat. no. 550290; BD Pharmingen; BD Biosciences) for 30 min, all in
Diamond Antibody Reagent containing 1% BSA. In this case, TBS-T
containing 2 mM CaCl2 (TBS-T-Ca) was used for the
washings (33). All antibodies
were incubated at RT. Slides were then stained using HiDef
Detection HRP Polymer System (Cell Marque; Merck KGaA) for 10 min
at RT and the colour was developed using 3,3'-diaminobenzidine
solution (ScyTek Laboratories, Inc.). After nuclear contrast
staining with haematoxylin (3 min at RT) and mounting with Quick-D
mounting medium, the slides were visualised under a light
microscope with coupled camera by two certified independent
pathologists. A semi-quantitative approach was established for
tissue slide evaluation (34), to
calculate the immunoreactive score (IRS). The IRS is calculated by
multiplying two scores: The cell proportion score that is 0 if all
cells were negative, or 1 if <25%, 2 if 26-50%, 3 if 51-75% and
4 if >75% cells were stained; and the staining intensity score
that is 0 when no stain was found, or 1 if weak, 2 if intermediate
and 3 if strong staining intensity was observed. All images were
acquired with magnification, ×10, and for the semi-quantitative
analysis, 4 fields per section were evaluated.
Statistical analysis
Data from normal tissues were paired with data from
matched tumour tissues and statistical differences were analysed
using paired t-test (data with a normal distribution) or Wilcoxon
matched-pairs signed rank test (data with a non-normal
distribution). The correlations between data were analysed using
Spearman correlation and categorized as weak (r>0.3), moderate
(r>0.5) and strong (r>0.7). To investigate associations
between gene mRNA, sLex/sLea and E-selectin
ligand expression, and clinical features the Fisher's exact
statistical test was used. The multivariate survival model used was
the Cox proportional hazards model, performed with R 3.6.0
(‘survival' package; https://github.com/therneau/survival). In case of
multiple comparisons, one-way ANOVA tests were performed to test
the statistical difference between the groups of the study, with
Tukey's multiple comparison post hoc test. Overall survival was
defined as the time from diagnosis to the date of death (months).
Patients alive at the end of the study or who succumbed to clearly
non-cancer-related causes were censored. Tests were considered
statistically significant when P<0.05 and marginally significant
when 0.05<P<0.1. Statistical analysis was performed using
GraphPad Prism 6 (GraphPad Software, Inc.).
Results
E-selectin ligands and
sLex/sLea antigens are overexpressed in NSCLC
tumour tissues
To ascertain the E-selectin ligand expression in
NSCLC, 18 normal and matched tumoral lung tissues were compared for
reactivity to HECA-452 mAb, an antibody that recognises both
sLex and sLea structures, and E-Ig, an
E-selectin chimera that recognises E-selectin ligands.
Immunoblotting revealed that all tissues showed reactivity with
HECA-452 mAb and E-Ig, which was significantly higher (2.2- and
1.8-fold) in tumour samples compared with matched normal samples
(Fig. 2A and B). Furthermore, the
intensity of the reactivity of both stainings exhibit a strong
positive correlation (r=0.748, P<0.001; Fig. 2C). Immunohistochemistry revealed
that tumoral tissue also exhibited stronger reactivity to both
HECA-452 mAb (IRS, 4-9 vs. 1) and E-Ig (IRS, 6-9 vs. 2) compared
with normal tissue (Fig. 2D).
These results suggested that sLex/sLea
antigens and E-selectin ligands in NSCLC exhibit increased
expression in tumour tissues compared with normal tissues.
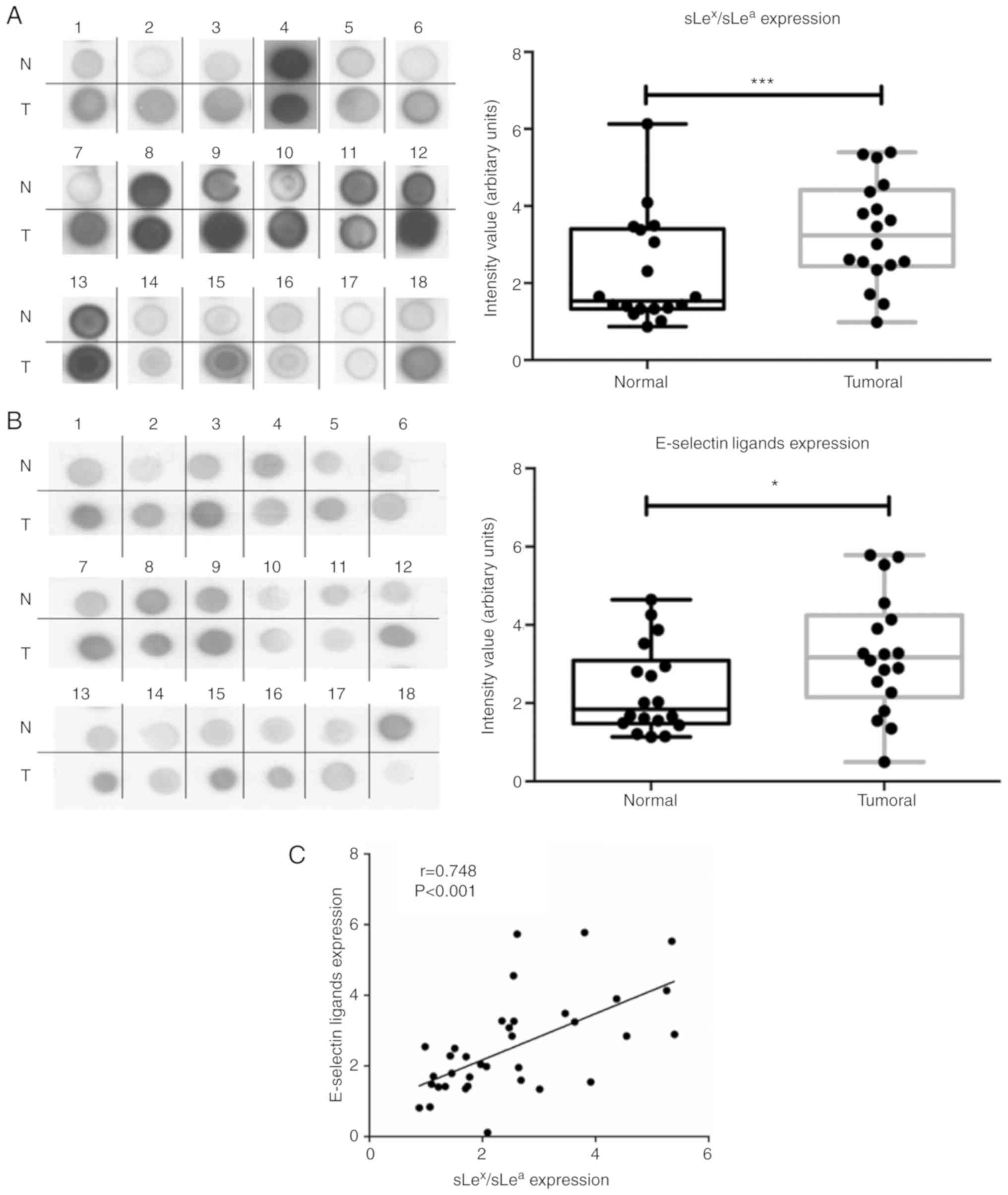 | Figure 2NSCLC has increased
anti-sLex/sLea mAb and E-selectin reactivity.
(A) Dot blot analysis of anti-sLex/sLea mAb
reactivity in matched normal and tumour proteins. Total lysates of
N or T tissues were spotted on nitrocellulose membrane and stained
with HECA-452 mAb. Left panel, representative dot blot analysis of
the HECA-452 mAb reactivity. Right, the intensity of each dot blot
spot, determined using ImageJ 1.48v software and expressed as
arbitrary units. Box-and-whisker plots represent median, and lower
and upper quartile values (boxes), ranges and all values for each
group (black dots). (B) Dot blot analysis of E-selectin reactivity
in matched normal and tumour proteins. Total lysates of N or T
tissues were spotted on nitrocellulose membrane and stained with
E-Ig. Left, representative dot blot analysis of E-Ig reactivity.
Right, the intensity of each dot blot spot expressed as arbitrary
units. Box-and-whisker plots represent median values, and lower and
upper quartiles (boxes), ranges and all values for each group
(black dots). *P<0.05, ***P<0.001. (C)
Correlation between anti-sLex/sLea mAb and
E-Ig staining intensity in tumour tissue. Correlation was analysed
using Spearman's correlation coefficient. NSCLC has increased
anti-sLex/sLea mAb and E-selectin reactivity.
(D) Anti-sLex/sLea mAb and E-Ig reactivity in
paraffin-embedded normal, tumour and metastasis sections.
Sequential sections from representative normal, non-small cell lung
cancer and bone metastasis tissues were stained with HECA-452 mAb
(top) and E-Ig chimera (bottom) via immunohistochemistry. Nuclei
stained with haematoxylin. Magnification, ×10. E-Ig, mouse
E-selectin-human Fc Ig chimera; mAb, monoclonal antibody; N,
normal; sLea/x, sialyl Lewis a/x; T, tumour. |
The associations between
sLex/sLea and E-selectin ligand expression,
and clinical features such as histological type, stage of disease,
gender, age, metastatic site and smoking habits were assessed.
Considering the expression of sLex/sLea in
tumour and normal tissue, it was found that patients who developed
bone metastasis had a higher tumour/normal (T/N) ratio compared
with patients who developed metastasis in other sites (P<0.05;
Table I). Other clinical features
showed no statistically significant association with
sLex/sLea or E-selectin ligands.
 | Table IRelation between expression of
sLex/sLea and
E-selectin ligands with patient's clinical features. |
Table I
Relation between expression of
sLex/sLea and
E-selectin ligands with patient's clinical features.
| Clinical
feature | sLex/a
expression (mean T/N ratio ± SEM) | P-value | E-Selectin ligands
expression (mean T/N ratio ± SEM) | P-value |
|---|
| Histological
type | | 0.577 | | >0.999 |
| Adenocarcinoma
(N=14) | 1.710±0.165 | | 1.557±0.185 | |
| Squamous cell
carcinoma (N=4) | 1.448±0.220 | | 1.633±0.321 | |
| Stage of
disease | | 0.335 | | <0.999 |
| I + II (N=11) | 1.500±0.222 | | 1.556±0.189 | |
| III (N=7) | 1.748±0.176 | | 1.585±0.234 | |
| Gender | | >0.999 | | <0.999 |
| Male (N=13) | 1.670±0.182 | | 1.562±0.216 | |
| Female (N=5) | 1.604±0.171 | | 1.604±0.108 | |
| Metastatic
site | | 0.033a | | 0.500 |
| Bones (N=3) | 2.353±0.307 | | 1.663±0.156 | |
| Other sites
(N=7) | 1.270±0.162 | | 1.173±0.292 | |
| Age | | 0.347 | | 0.153 |
| <median age
(N=9) | 1.483±0.213 | | 1.190±0.195 | |
| ≥median age
(N=9) | 1.820±0.166 | | 1.958±0.172 | |
| Smoking habits | | <0.999 | | <0.999 |
| Smoker (N=11) | 1.625±0.186 | | 1.715±0.215 | |
| Non-smoker
(N=7) | 1.693±0.214 | | 1.351±0.210 | |
Considering the correlation between
sLex/sLea and E-selectin ligand expression,
and the association between sLex/sLea
expression and the development of bone metastasis, the expression
of sLex/sLea and E-selectin ligands in bone
metastasis tissue derived from patients with NSCLC was then
assessed. Weak positive reactivity was observed in metastasized
cells (IRS, 1-2; Fig. 2D),
suggesting an important role for these structures in cancer
progression and the promotion of metastasis. When considering the
IRS, it is important to note that these samples were from bone
metastases, which require a decalcification process for
immunohistochemistry purposes. This process may result in a partial
loss of antigen, considering the calcium-binding dependence of
E-selectin binding (partially recovered by the addition of
CaCl2). Furthermore, the primary tissue has a strong
expression of E-selectin ligands that potentiates migration and
leads to the formation of metastasis in distant sites. After
E-selectin binding, cancer cells pass through the endothelium to
colonize new sites, forming metastasis. During metastatic
establishment in these new sites, the expression of biomarkers that
potentiate migration is expected to decrease. Hence, a staining
with a lower IRS (or equivalent to the normal section) was
expected. Thus, an IRS of 1-2 in these tissues was considered a
high score indicative of a role for E-selectin ligands in bone
metastasis.
FUT3, FUT6 and FUT7 are upregulated in
NSCLC tumour tissues
To gain further insight into the molecular basis of
augmented E-selectin ligands in NSCLC, the expression of enzymes
critical to the biosynthesis of sLex/sLea
were subsequently compared in matched normal and tumour tissues
(Fig. 1B). First, the expression
of the genes that encode for the α1,3/4-fucosyltransferases
(FUT3, FUT4, FUT5, FUT6 and
FUT7) and α2,3-sialyltransferases (ST3GAL3,
ST3GAL4 and ST3GAL6), which add fucose and sialic
acid residues, respectively, to type 1 or type 2 glycan precursors
of sLex/sLea, were evaluated via RT-qPCR
analysis (Fig. 1B). With few
exceptions, all genes were expressed in all samples and presented a
broad range of expression levels. It was observed that FUT3
showed a marginally significant 2.3-fold increase in expression in
tumour compared to normal tissue (0.05<P<0.1; Fig. 3A). FUT6 and FUT7
expression levels were significantly increased in NSCLC tissues
samples compared with matched normal tissues (5.3- and 2-fold,
respectively; P<0.001 and P<0.05, respectively; Fig. 3A). These results suggested
increased α1,3-FUT activity in tumour tissue. A comparison of the
relative levels of expression of α1,3-FUTs revealed that
FUT7 is poorly expressed, compared with FUT6, and
particularly with FUT3 and FUT4. Conversely,
FUT4, ST3GAL4 and ST3GAL6 expression levels
were significantly decreased by 3.1, 2 and 2.5 times, respectively,
in tumour compared with normal tissues (P<0.001, P<0.05 and
P<0.05, respectively). ST3GAL3 expression was not
statistically different between normal and tumour tissue (Fig. 3A). FUT5 expression was not
detected in the present study.
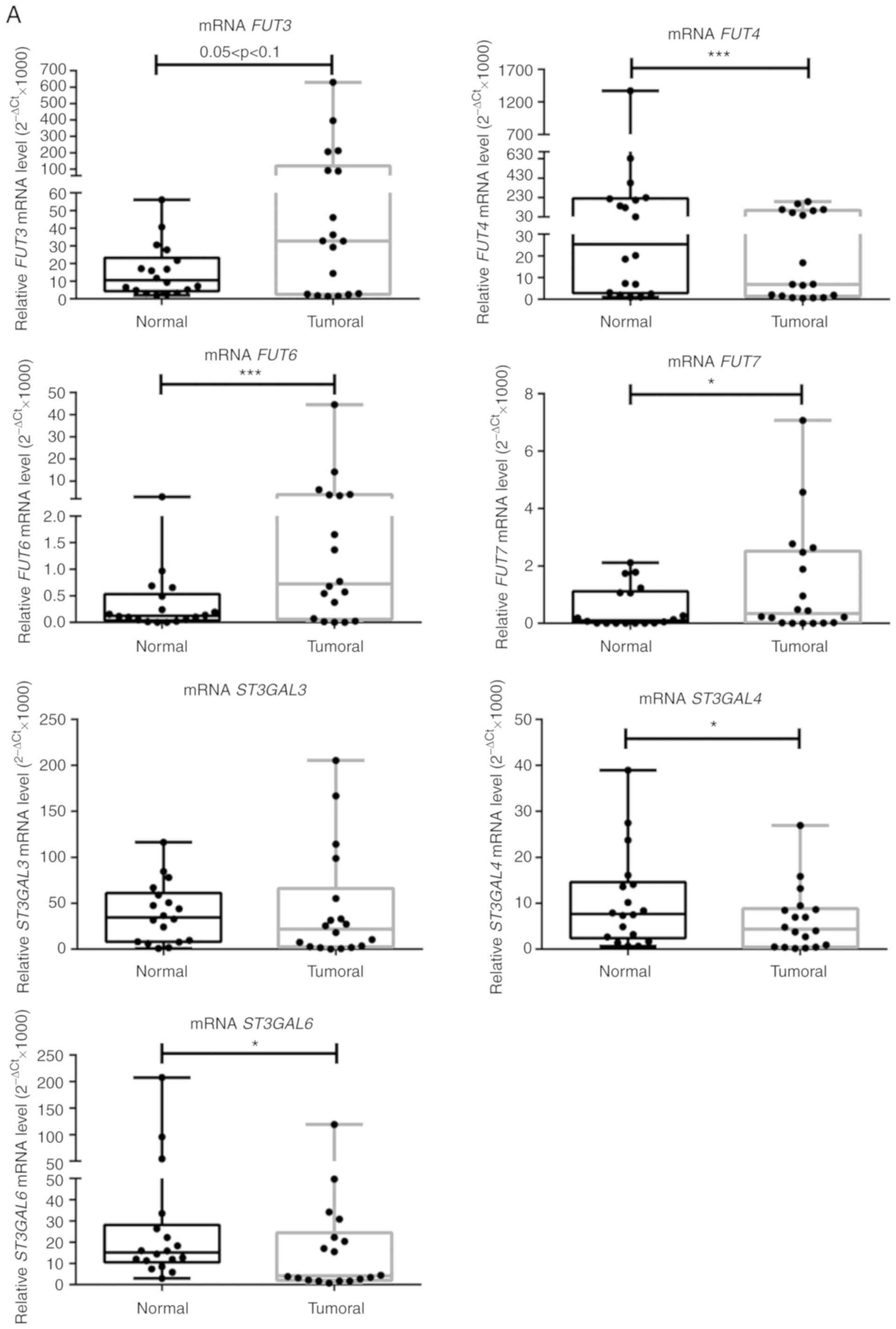 | Figure 3NSCLC tissues show upregulation of
FUT3, FUT6 and FUT7 and increased α1,3-FUT
activity. (A) Relative mRNA levels of FUT3, FUT4,
FUT6, FUT7, ST3GAL3, ST3GAL4 and
ST3GAL6 in normal and tumour tissues from patients with
NSCLC, as determined via reverse transcription-quantitative PCR
analysis. Values indicate the number of mRNA molecules of a
certain gene per 1,000 molecules of the average of the endogenous
controls (ACTB and GAPDH). Box-and-whisker plots
represent median values, and lower and upper quartiles (boxes),
ranges and all values for each group (black dots).
*P<0.05, ***P<0.001. 0.05<P<0.1,
marginally significant. NSCLC tissues show upregulation of
FUT3, FUT6 and FUT7 and increased α1,3-FUT
activity. (B) Evaluation of α1,3-FUT activity in tissue lysates.
α1,3-FUT enzymatic activity was evaluated in lysates obtained from
normal and NSCLC tissues derived from 7 patients. Activity was
measured using fetuin as an acceptor. 0.05<P<0.1, marginally
significant; *P<0.05. (C) Relationship between
overall survival and FUT4 expression ratio. Curve plots of
overall survival in 18 patients (vertical tick marks indicate
censored cases). Patients with high FUT4 T/N ratios (n=9)
and patients with low FUT4 T/N ratios (n=9) were separated
based on the median value. The multivariate survival model used was
the Cox proportional hazards model. 0.05<P<0.1, marginally
significant. FUT, fucosyltransferase; NSCLC, non-small cell lung
cancer; ST3GAL, α2,3-sialyltransferase; T/N, tumour/normal. |
Then, the enzyme activity of total α1,3-FUT was
measured in matched samples. Due to sample limitation, only samples
from 7 patients (namely patients 1, 2, 3, 5, 6, 7 and 9) were used.
As presented in Fig. 3B, α1,3-FUT
activity in tumour tissues was significantly increased compared
with in matched normal tissues (P<0.05), confirming the gene
expression data.
It was also assessed whether there was any
correlation between the analysed genes and E-Ig staining in tumour
tissues samples (data not shown). A moderate positive correlation
was observed between the expression levels of FUT3 and
FUT6 (r=0.517, P<0.05), FUT3 and FUT7
(r=0.624, P<0.01), and FUT6 and FUT7 (r=0.680,
P<0.01), suggesting that the expression of these FUTs is
regulated similarly (data not shown). Of note, a weak correlation
was observed between the expression of E-selectin ligands, and the
expression ratios of FUT3/FUT4 (r=0.369, P<0.01) and FUT7/FUT4
(r=0.366, P<0.05; data not shown). These data suggested that
overexpression of FUT3 and FUT7 enzymes, and concomitant
downregulation of FUT4 enzymes may promote E-selectin ligands
expression in NSCLC.
Patients were then categorised in two groups
(n=9/group), according to the FUT4 mRNA expression; those
with FUT4 mRNA <10 and those ~30 (Fig. 3A). The group with lower FUT4
expression exhibited higher expression levels of E-selectin
compared with the group with higher FUT4 expression (3.974±1.102
vs. 2.837±1.135; P<0.05; data not shown). Of note, the former
group of patients displayed a lower overall survival (hazard
ratio=0.16; 95% CI; 0.018-1.4; 0.05<P<0.1), suggesting that a
lower FUT4 T/N expression ratio was a potential biomarker for poor
prognosis (Fig. 3C). None of the
other variables tested proved to be significant in the multivariate
survival model used.
When subdividing the patients according to their
tumours' histological type, it was possible to observe a marginally
significantly higher ratio of FUT3 expression in patients
with AC compared with patients with SCC (0.05<P<0.1) and a
significantly higher ratio of FUT7 expression in female
compared with male patients (P<0.05; Fig. S1). Additionally, a marginally
higher T/N ratio of FUT6 was observed in non-smoker patients
compared with smoker patients (0.05<P<0.1; Fig. S1).
CEA is expressed in NSCLC samples with
high expression of sLex/sLea glycans
CEA is a glycoprotein involved in cell adhesion,
showing high levels of expression in lung cancer compared with
adult normal lung tissues (35).
It was hypothesised that CEA was expressed in these samples as a
potential scaffold of sLex/sLea in NSCLC. Via
western blot analysis, it was observed that CEA was not detectable
in normal lung tissue. In contrast, it was verified that 8 out of
the 18 NSCLC samples expressed CEA, presenting a characteristic
band of ~180 kDa (specifically, samples 1, 2, 7, 9, 10 and 15, with
weaker bands also detected in samples 16 and 18; Fig. 4A). Of note, all samples from
patients with bone metastasis expressed CEA (Table SI). Comparing the CEA-negative and
CEA-positive NSCLC samples, it was observed that CEA-positive
samples had a significantly higher expression of
sLex/sLea glycans (Fig. 4B). To investigate a potential
association between CEA, sLex/sLea and
E-selectin ligands, immunohistochemistry analysis was performed
using five NSCLC sections from tissues which exhibited higher
levels of CEA in western blotting. Paraffin-embedded sections were
stained with anti-CEA mAb, anti-sLex/sLea and
E-Ig chimera. Normal tissue sections showed no reactivity with
anti-CEA, anti-sLex/sLea or E-Ig (IRS, 0),
except for inflammatory cells, which stained with E-Ig and
anti-sLex/sLea (IRS, 1-2; data not shown). In
contrast, tumour sections showed general high staining for
sLex/sLea (IRS, 4-9), E-selectin ligands
(IRS, 6-9) and CEA (IRS, 4-9), with cytoplasmic and cell membrane
localisation (Fig. 4C).
Furthermore, areas showing reactivity for CEA also stained with
sLex/sLea and E-Ig, suggesting
co-localization of CEA and sLex/sLea in these
tissues. These results suggested that CEA is a potential protein
scaffold for sLex/sLea in NSCLC and has a
possible role as an E-selectin ligand.
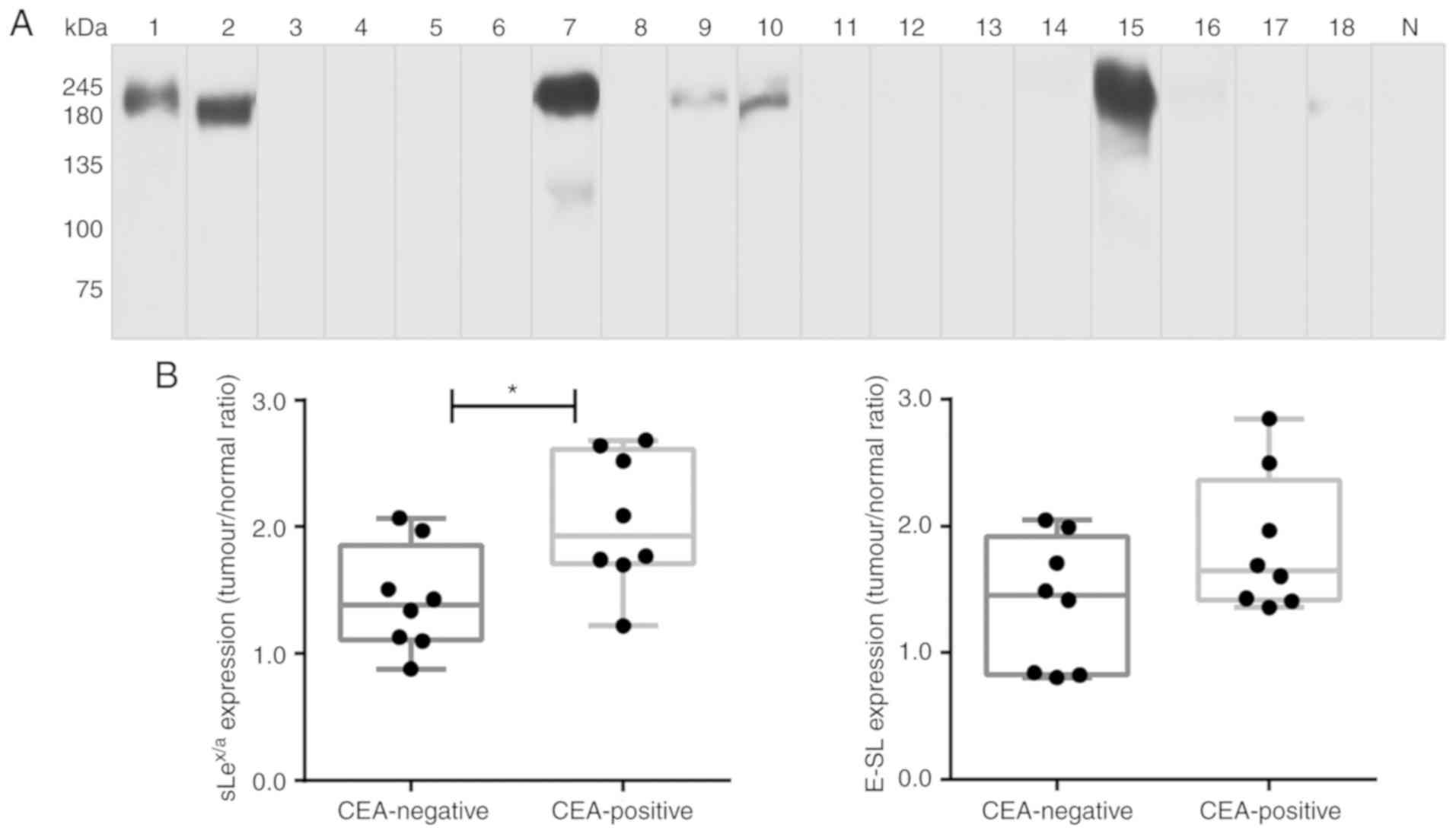 | Figure 4CEA is expressed in patients with
NSCLC. (A) Western blot analysis of CEA glycoprotein in tumour
lysates. Protein (20 µg) obtained from NSCLC patients'
tissues were ran in reduced SDS-PAGE gels and blotted with anti-CEA
mAb. The numbers above each lane represent the patient number. The
lane N corresponds to a representative normal tissue lysate from a
patient with NSCLC (patient number 7), showing negative anti-CEA
reactivity. This figure shows a blot with tracks from samples
analysed separately. (B) sLex/sLea and
E-selectin ligands expression in CEA-negative and CEA-positive
patients. The tumour/normal ratios of
sLex/sLea expression (left) and E-selectin
ligands expression (right) were calculated and separated for
CEA-negative and CEA-positive patients. Box-and-whisker plots
represent median values, and upper and lower quartiles (boxes),
ranges and all values for each group (black dots).
*P<0.05. (C) sLex/sLea,
E-selectin ligands and CEA have overlapping staining profiles in
NSCLC tissues. Sequential paraffin-embedded NSCLC tissue
sections from patients 2, 7, 10 and 15 were stained with HECA-452
mAb (left), E-Ig chimera (middle), and anti-CEA mAb (right) via
immunohistochemistry. Nuclei were stained with haematoxylin.
Magnification, ×10. CEA, carcinoembryonic antigen; E-Ig, mouse
E-selectin-human Fc Ig chimera; E-SL, E-selectin; mAb, monoclonal
antibody; N, normal; NSCLC, non-small cell lung cancer;
sLea/x, sialyl Lewis a/x. |
CEA is an E-selectin ligand in NSCLC
tissues
To study whether CEA in NSCLC is associated with
sLex/sLea and recognized by E-selectin,
protein lysates from the five NSCLC tissues were electrophoresed
and analysed via western blotting, using E-Ig, and
anti-sLex/sLea and anti-CEA mAbs. Among the
glycoproteins detected by anti-sLex/sLea mAb
and E-Ig chimera, one displaying the same molecular weight as CEA
(180 kDa) was present in all five tumour samples examined (Fig. 5A). Other glycoproteins reactive
with anti- sLex/sLea mAb and/or E-Ig chimera
of higher molecular weight (MW), such as mucins with MW >245
kDa, and lower MW have yet to be identified.
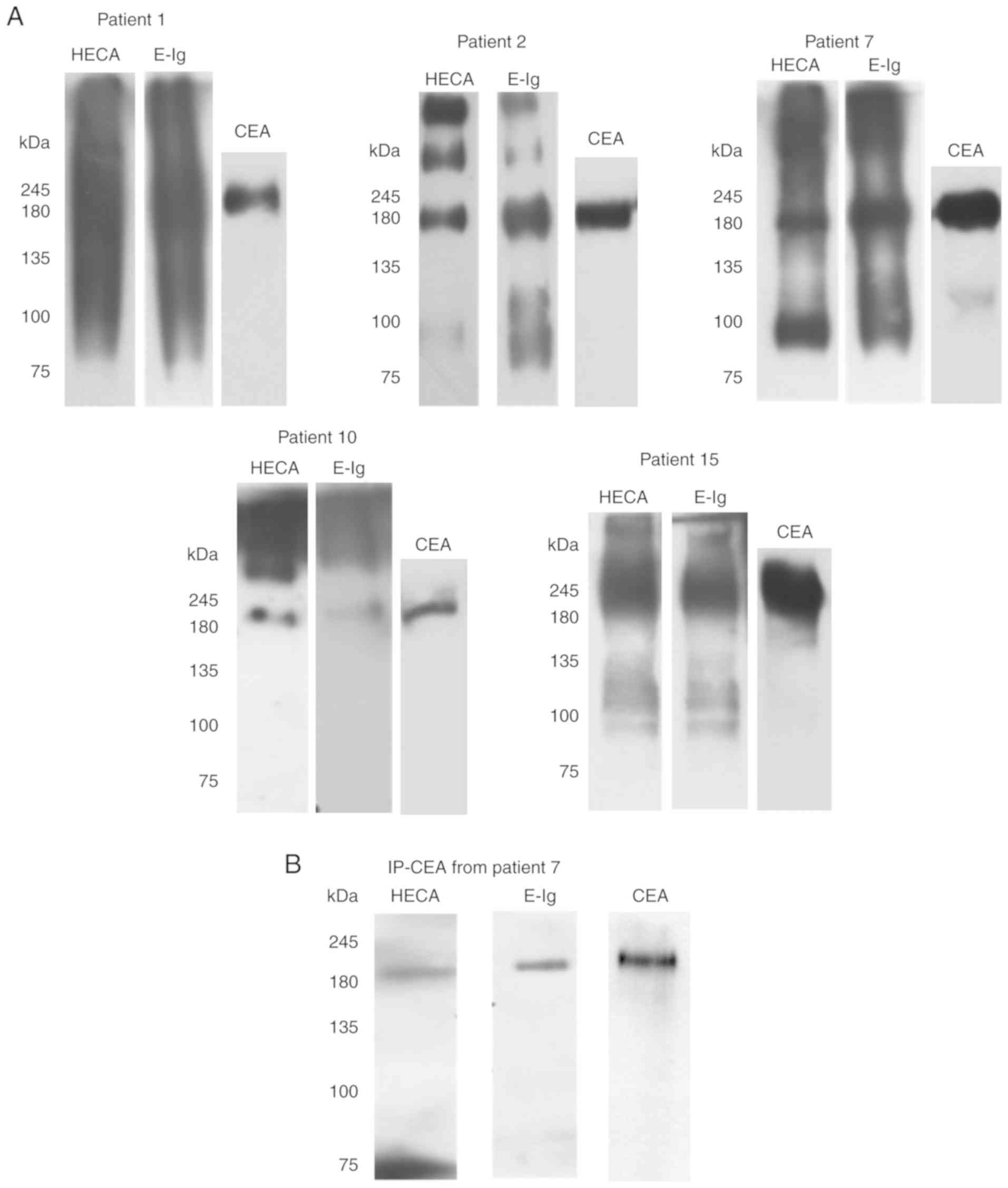 | Figure 5CEA from NSCLC associates with
sLex/sLea and has reactivity with E-selectin.
(A) Western blot analysis of tumour lysates from CEA-positive NSCLC
tissues. CEA-positive NSCLC tissues were resolved via SDS-PAGE and
immunoblotted with HECA-452 mAb, E-Ig and anti-CEA mAb. (B) Western
blot analysis of immunoprecipitated CEA. CEA was immunoprecipitated
from the tumour lysate of patient 7, and then resolved via SDS-PAGE
and blotted. Left, HECA-452 staining; middle, E-Ig staining; right,
anti-CEA mAb staining. CEA, carcinoembryonic antigen; E-Ig, mouse
E-selectin-human Fc Ig chimera; HECA, HECA-452; IP,
immunoprecipitation; mAb, monoclonal antibody; NSCLC, non-small
cell lung cancer; sLea/x, sialyl Lewis a/x. |
To further confirm that CEA reacted with
E-selectin, CEA was immunoprecipitated from the protein lysates of
patient 7, one of the patients with the highest CEA expression as
determined by western blotting (Fig.
4A). Immunoprecipitated CEA was analysed via western blotting
to verify whether this protein was stained with
anti-sLex/sLea mAb and/or E-Ig chimera. As
shown in Fig. 5B,
immunoprecipitated CEA was recognised by both, confirming that CEA
is a sLex/sLea antigens carrier and an
E-selectin ligand in NSCLC.
CEA is a functional E-selectin ligand in
flow conditions
The primary role of E-selectin engagement during
transendothe-lial migration is to slow down the leukocytes
circulating in the bloodstream, in order to promote their adhesion
to the endothelium (36). To infer
the ability of NSCLC cells to bind to E-selectin in flow
conditions, whether the proteins isolated from NSCLC tissue were
able to support rolling interactions with cells expressing
E-selectin on their surface (CHO-E cells; Fig. S2) was assessed. By using an
alternative Stamper-Woodruff assay to mimic blood flow conditions
(37), it was observed that the
tumour proteins were able to specifically bind CHO-E cells in the
presence of calcium-containing buffer (Fig. 6A). Conversely, in the presence of
EDTA-containing buffer or function-blocking mAbs against
E-selectin, binding to CHO-E cells was significantly reduced
(Fig. 6A), indicating that this
cell adhesion was mediated specifically by E-selectin interactions.
E-selectin is a calcium-binding-dependent lectin, thus the
requirement to use calcium-containing buffer for this assay
(9). To further demonstrate that
CEA protein scaffold is one of the functional E-selectin ligands in
NSCLC, the ability of CEA immunoprecipitated from tumour proteins
to bind to CHO-E and CHO-mock cells under flow conditions was
assessed. As shown in Fig. 6B,
immunoprecipitated CEA was able to bind to CHO-E cells, but not
CHO-mock, and the interaction was abrogated by EDTA-containing
buffer. Collectively, these data indicated that CEA expressed by
NSCLC is a functional E-selectin ligand.
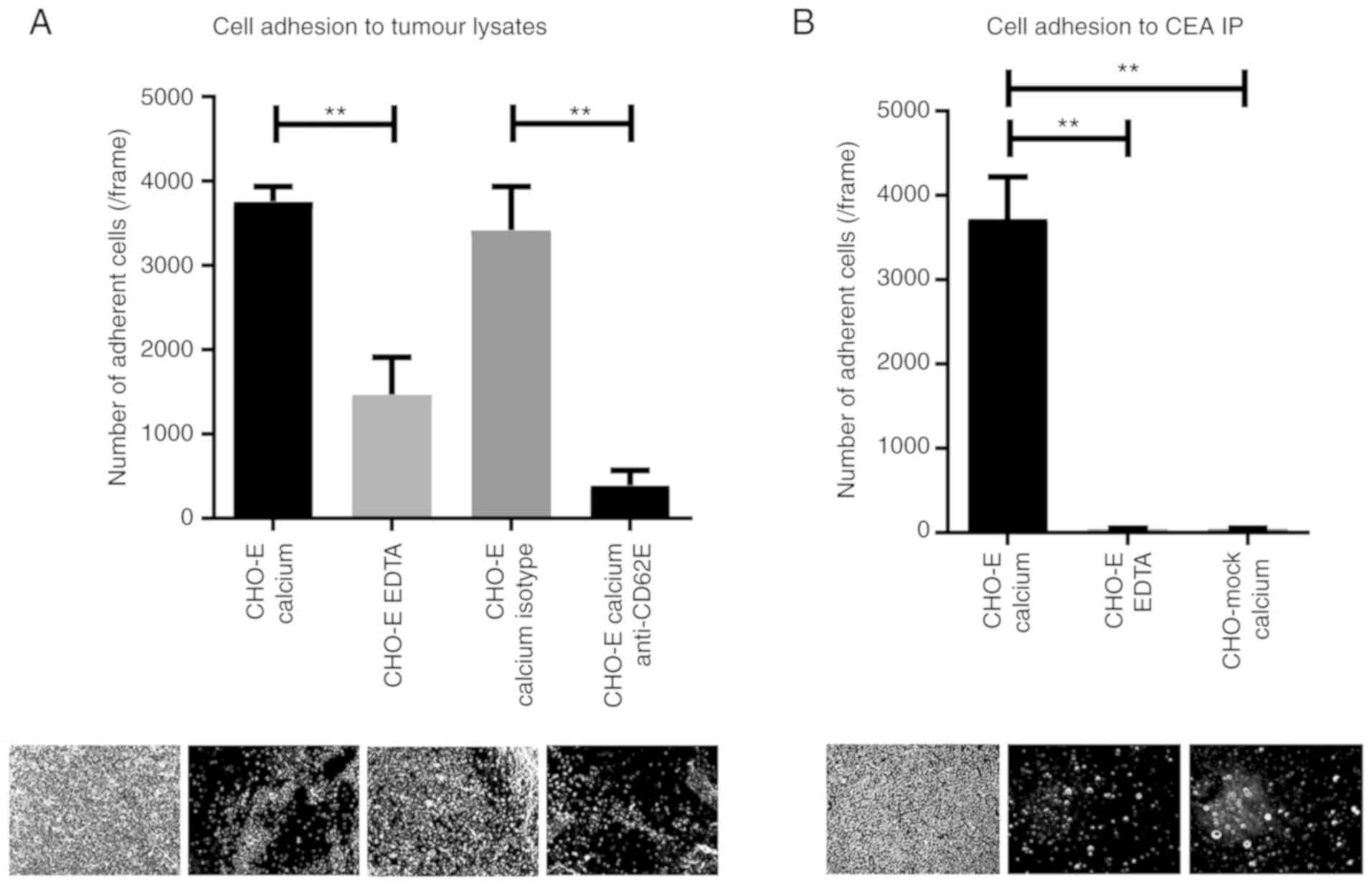 | Figure 6CEA glycoprotein is a functional
E-selectin ligand in NSCLC. (A) Adhesion of E-selectin-expressing
cells to NSCLC lysates. Lysates of NSCLC tissue, derived from
patient 7, were spotted on glass slides, and the adhesion of CHO-E
cells was tested using a modified Stamper-Woodruff binding assay
test. As E-selectin interactions are calcium-dependent, CHO-E cells
were resuspended in calcium buffer. EDTA buffer was used as a
control. Cells were pre-incubated with 20 µg/ml of specific
mAbs prior to the adhesion experiment; isotype control or
function-blocking anti-E-selectin mAb. (B) Adhesion of
E-selectin-expressing cells to CEA immunoprecipitate. CEA was
immunoprecipitated from NSCLC tissue derived from patient 7 and
spotted on glass slides. Adhesion of CHO-E or CHO-mock cells to CEA
IP was tested using a modified Stamper-Woodruff binding assay.
CHO-E and CHO-mock cells were resus-pended in calcium buffer, and
CHO-E cells resuspended in EDTA buffer were used as a negative
control. One-way ANOVA followed by Tukey's post hoc test was used
for analysis. **P<0.01. Representative pictures
captured for each condition (magnification, ×100) are represented
below the respective graphs. CD62E, E-selectin; CEA,
carcinoembryonic antigen; CHO, Chinese hamster ovary cells; CHO-E,
E-selectin-expressing CHO; CHO-mock, empty vector-transfected CHO;
isotype, isotype control Ab; mAb, monoclonal antibody. |
Discussion
Several studies have reported the expression of
sLex/sLea glycans in NSCLC tissues and cell
lines (35,38,39).
The discovery of these glycans in the serum of patients soon
motivated the quest to validate these as prognostic biomarkers
(40). Elevated levels of
sLex/sLea glycans have been associated with
metastasis by several studies (39,41,42).
In cell lines, their contributions to the adhesion of cancer cells
to selectins expressed by vascular endothelium (43), including brain endothelium
(44), have been shown. These
findings highlight the potential role of
sLex/sLea as E-selectin ligands contributing
to the adhesion of cancer cells to endothelium and subsequent
metastasis (45,46). However, little is known regarding
the molecular basis of sLex/sLea expressed by
the primary tissue, particularly its protein scaffold and whether
they also enable the cells to establish adhesive interactions with
endothelium selectins.
The present study showed that
sLex/sLea glycans are significantly
overexpressed in NSCLC tissues in comparison with matched normal
lung tissues. These results are in agreement with previous studies
showing increased expression of sLex/sLea
glycans in both serum and biopsies from patients with NSCLC
(14,42,47).
The data also showed that NSCLC has high reactivity with E-selectin
compared with normal tissue, whose intensity is correlated with
anti-sLex/sLea mAb reactivity. Although
sLex/sLea glycans are known as the
prototypical ligands of E-selectin, their functional binding also
depends on accessibility at the cell surface (48). In fact, the mere expression of the
glycan ligands does not imply E-selectin binding. Therefore, this
study provides the first evidence, to our knowledge, that NSCLC
tissue has E-selectin reactivity.
The present study showed that increased
sLex/sLea expression in NSCLC tissue was
observed in patients who later developed bone metastasis.
Furthermore, it was found that metastatic tissue derived from NSCLC
primary tissues also expressed sLex/sLea and
E-selectin ligands, which were expressed in metastasized cancer
cells. In humans, bone marrow microvasculature constitutively
expresses E-selectin, as well as all vascular endothelium following
stimulation with inflammatory cytokines (49,50).
Therefore, this observation suggested that
sLex/sLea overexpression in NSCLC cells is an
adaptation to enable cells to adhere to vascular E-selectins and
metastasise to other locations, such as the bone. In addition to
E-selectin engagement, sLex/sLea can also
alter the immune homeostasis of the mucous membrane by impairing
tumour cell recognition by the immune system and favouring cancer
progression (51-53).
The present data also showed that the median values
of FUT3, FUT6 and FUT7 mRNA expression, and
the overall mean α1,3-FUT activity were higher in NSCLC tumour
tissues, whereas that of FUT4 was lower and that of
FUT5 was not detected. The observed FUT3, FUT6
and FUT7 gene expression profiles are consistent with
previous reports showing that FUT3 is abundantly and markedly
expressed in lung cancer tissues, whereas FUT6 and FUT7 are
detected at lower levels but also upregulated compared with normal
tissues (16,54,55).
These significant alterations in the expression of FUT genes may be
the underlying cause of the observed overexpression of
sLex/sLea; however, to prove that altered FUT
gene expression is causative of altered
sLex/sLea expression, other studies will have
to be performed.
sLex/sLea biosynthesis is a
highly convoluted process with several glycosyltransferases
involved (56). For example,
sLex antigens biosynthesis is a very complex process, of
which the addition of fucose is the last step. Fucosyltransferases
compete with each other for similar substrates. Consistent with
previous reports (54), in this
study the expression of FUT4, an enzyme involved in non-sialylated
Lewis biosynthesis, was found to be lower in tumour tissue.
Notably, the ratios of FUT3/FUT4 and
FUT7/FUT4 expression correlated with E-selectin
ligand expression, highlighting the concomitant and competitive
role of these enzymes in E-selectin ligand biosynthesis.
Furthermore, the FUT4 T/N expression ratio enabled marginally
significant evaluation of the overall survival of patients; thus,
it is proposed that the relative level of FUT4 serves as a
protective factor and is therefore a clinically relevant prognostic
biomarker.
sLex/sLea was also identified
in normal cells, but at significantly reduced levels. Nonetheless,
changes in the expression of these glycans due to malignant
transformation may depend not only on the expression of fucosyl-
and sialyltransferases, but also on the activity of other enzymes
that can lead to more complex or alternative structures (57,58).
Further studies are required to fully understand the alterations of
glycan biosynthesis occurring in NSCLC.
As the functional binding of
sLex/sLea antigens with E-selectins is
crucially dependent on their presentation by scaffold proteins
(59-61), their mere detection has limited
prognostic value in different cancers (62-64).
Thus, the identification of sLex/sLea
-decorated protein scaffolds is expected to provide more specific
and reliable clinical biomarkers. While carriers of E-selectin
ligands have been identified in several types of cancer, including
the CD44 glycoform known as hematopoietic cell E-/L-selectin ligand
in colon cancer, and CEA in colon and prostate cancer (65,66),
no functionally defined E-selectin ligands were identified in NSCLC
to date. Here, the data indicated that CEA decorated by
sLex/sLea acts as a functionally relevant
E-selectin ligand. Thus, CEA interactions with endothelial
selectins may the mediate tethering, rolling and adhesion of tumour
cells to the endothelium, consequently promoting cancer metastasis.
CEA is a glycoprotein with low and limited expression in normal
tissues, but is detected at high levels in tumours with epithelial
origin, including NSCLC, gastric carcinoma and colorectal cancer,
for which it is used as a tumour biomarker (67). Serum CEA in patients with NSCLC
correlates with advanced stage of the disease, poor therapeutic
response, early relapse and shorter survival (35,68).
Notably, besides cell adhesion, CEA also serves key functions in
intracellular and intercellular signalling involved in cancer
progression, inflammation, angiogenesis and metastasis (69). Thus, CEA-E-selectin interactions
may also contribute to these malignant features. Although CEA and
sLex/sLea have been extensively suggested as
clinical biomarkers in NSCLC (12,70,71),
to our knowledge, this is the first study addressing the functional
relevance of sLex/sLea-associated CEA as an
E-selectin ligand in primary NSCLC tissue.
Unlike other reports that used cell lines, the
present findings were obtained in primary NSCLC and paired
non-tumour pulmonary tissues. The use of primary tissue offers
undeniably greater relevance to the findings, as it reflects the
in vivo NSCLC microenvironment. Yet, the use of primary
tissue posed limitations on sample quantity and sample size that
affected reproducibility, and requires validation in larger
cohorts. The expression of E-selectin ligands in lung tissue
derived from healthy subjects also remains to be established. A
further limitation of the present study was the use of normal
pulmonary tissues derived from patients with NSCLC. Non-cancerous
tissues surrounding the tumour is a common control for these types
of studies; however, it can be affected by the presence of
infiltrating cancer cells or the so-called ‘field effect' of tumour
growth (72,73).
Overall, the present findings indicated that
sLex/sLea and E-selectin ligands are
overexpressed in NSCLC tissue compared with adjacent control
tissue, alongside increased a1,3-FUT activity and
fucosyltransferase transcript expression. The highest
sLex/sLea levels were detected in the primary
tissues of patients with bone metastasis, hinting that these
antigens may promote metastasis to this site. Furthermore, the
presence of sLex/sLea and E-selectin ligands
on CEA suggests that mechanistically, CEA may facilitate adhesion
to endothelium selectins and consequently promote cancer
metastasis. This study also pinpoints the usefulness of
sLex/sLea-modified CEA as a potential
therapeutic target and diagnostic biomarker in NSCLC, a finding
that requires validation in future studies.
Supplementary Data
Funding
The authors acknowledge the financial support from
the LPCC/Pfizer 2011, and Tagus TANK award 2018 (grant no. 1/2018)
from Universidade Nova de Lisboa and José de Mello Saúde.
Additionally, scholarship funding was provided by the Portuguese
Foundation for Science and Technology (FCT) [grant nos.
SFRH/BD/100970/2014 and SFRH/BPD/108686/2015].
Availability of data and materials
The datasets used and/or analysed during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
The study was conceived by PV, who designed and
supervised all research and drafted the manuscript. IGF and MC
contributed equally to the work in designing and performing the
experiments, collecting and analysing data, and drafting the
manuscript. AGM performed experiments, prepared results and drafted
the manuscript. AB collected patient samples and conceived
experiments. PB designed and interpreted immunohistochemistry data.
ZS performed experiments and drafted the manuscript, and FDO
analysed data and drafted the manuscript. All authors reviewed the
final version of the manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Centro Hospitalar Lisboa Norte. Written informed consent was
obtained from all patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Abbreviations:
|
AC
|
adenocarcinoma
|
|
CEA
|
carcinoembryonic antigen
|
|
CHO
|
Chinese hamster ovary
|
|
mAb
|
monoclonal antibody
|
|
NSCLC
|
non-small cell lung cancer
|
|
SCC
|
squamous cell carcinoma
|
|
sLex
|
sialyl Lewis x
|
|
sLea
|
sialyl Lewis a
|
Acknowledgments
The authors acknowledge both Dr Analisa Ribeiro and
Dr Madalena Ramos (Pathology service, Hospital Pulido Valente) for
their assessment of immunohistochemistry samples, and Dr Francisco
Félix, Dr Paulo Calvinho, Dr Cristina Rodrigues and nurses
(Thoracic service, Hospital Pulido Valente) for their involvement
in the collection of lung tissue samples. We also thank Ms Ana
Raquel Henriques (technician from the Pathology service, Faculty of
Medicine, University of Lisbon) for providing paraffin-embedded
sections of lung tissues samples.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Youlden DR, Cramb SM and Baade PD: The
international epidemiology of lung cancer: Geographical
distribution and secular trends. J Thorac Oncol. 3:819–831. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Riihimäki M, Hemminki A, Fallah M, Thomsen
H, Sundquist K, Sundquist J and Hemminki K: Metastatic sites and
survival in lung cancer. Lung Cancer. 86:78–84. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tamura T, Kurishima K, Nakazawa K,
Kagohashi K, Ishikawa H, Satoh H and Hizawa N: Specific organ
metastases and survival in metastatic non-small-cell lung cancer.
Mol Clin Oncol. 3:217–221. 2015. View Article : Google Scholar
|
|
5
|
Nishida N, Yano H, Nishida T, Kamura T and
Kojiro M: Angiogenesis in cancer. Vasc Health Risk Manag.
2:213–219. 2006. View Article : Google Scholar
|
|
6
|
Noman MZ, Messai Y, Muret J, Hasmim M and
Chouaib S: Crosstalk between CTC, immune system and hypoxic tumor
microenvironment. Cancer Microenviron. 7:153–160. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Popper HH: Progression and metastasis of
lung cancer. Cancer Metastasis Rev. 35:75–91. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gout S, Tremblay PL and Huot J: Selectins
and selectin ligands in extravasation of cancer cells and organ
selectivity of metastasis. Clin Exp Metastasis. 25:335–344. 2008.
View Article : Google Scholar
|
|
9
|
Ding D, Yao Y, Zhang S, Su C and Zhang Y:
C-type lectins facilitate tumor metastasis. Oncol Lett. 13:13–21.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Barthel SR, Gavino JD, Descheny L and
Dimitroff CJ: Targeting selectins and selectin ligands in
inflammation and cancer. Expert Opin Ther Targets. 11:1473–1491.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ogawa J, Sano A, Inoue H and Koide S:
Expression of Lewis-related antigen and prognosis in stage I
non-small cell lung cancer. Ann Thorac Surg. 59:412–415. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mizuguchi S, Nishiyama N, Iwata T, Nishida
T, Izumi N, Tsukioka T, Inoue K, Kameyama M and Suehiro S: Clinical
value of serum cytokeratin 19 fragment and sialyl-Lewis X in
non-small cell lung cancer. Ann Thorac Surg. 83:216–221. 2007.
View Article : Google Scholar
|
|
13
|
Komatsu H, Mizuguchi S, Izumi N, Chung K,
Hanada S, Inoue H, Suehiro S and Nishiyama N: Sialyl Lewis X as a
predictor of skip N2 metastasis in clinical stage IA non-small cell
lung cancer. World J Surg Oncol. 11:3092013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Satoh H, Ishikawa H, Kamma H, Yamashita
YT, Takahashi H, Ohtsuka M and Hasegawa S: Elevated serum sialyl
Lewis X-i antigen levels in non-small cell lung cancer with lung
metastasis. Respiration. 65:295–298. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gomes C, Osório H, Pinto MT, Campos D,
Oliveira MJ and Reis CA: Expression of ST3GAL4 leads to SLe(x)
expression and induces c-Met activation and an invasive phenotype
in gastric carcinoma cells. PLoS One. 8:pp. e667372013, View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Dall'Olio F, Malagolini N, Trinchera M and
Chiricolo M: Sialosignaling: Sialyltransferases as engines of
self-fueling loops in cancer progression. Biochim Biophys Acta.
1840:2752–2764. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dall'Olio F, Malagolini N, Trinchera M and
Chiricolo M: Mechanisms of cancer-associated glycosylation changes.
Front Biosci (Landmark Ed). 17:pp. 670–699. 2012
|
|
18
|
Sperandio M: Selectins and
glycosyltransferases in leukocyte rolling in vivo. FEBS J.
273:4377–4389. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nakayama F, Nishihara S, Iwasaki H, Kudo
T, Okubo R, Kaneko M, Nakamura M, Karube M, Sasaki K and Narimatsu
H: CD15 expression in mature granulocytes is determined by
α1,3-fucosyltransferase IX, but in promyelocytes and monocytes by
α1,3-fucosyltransferase IV. J Biol Chem. 276:16100–16106. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tian L, Shen D, Li X, Shan X, Wang X, Yan
Q and Liu J: Ginsenoside Rg3 inhibits epithelial-mesenchymal
transition (EMT) and invasion of lung cancer by down-regulating
FUT4. Oncotarget. 7:1619–1632. 2016. View Article : Google Scholar :
|
|
21
|
Brierley JD, Gospodarowicz MK and
Wittekind C: TNM classification of malignant tumours. John Wiley
& Sons, Ltd; 2017
|
|
22
|
Guo C, Liu S and Sun MZ: Novel insight
into the role of GAPDH playing in tumor. Clin Transl Oncol.
15:167–172. 2013. View Article : Google Scholar
|
|
23
|
Guo C, Liu S, Wang J, Sun MZ and Greenaway
FT: ACTB in cancer. Clin Chim Acta. 417:39–44. 2013. View Article : Google Scholar
|
|
24
|
Cucchiarelli V, Hiser L, Smith H,
Frankfurter A, Spano A, Correia JJ and Lobert S: β-tubulin isotype
classes II and V expression patterns in nonsmall cell lung
carcinomas. Cell Motil Cytoskeleton. 65:675–685. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Videira PA, Correia M, Malagolini N,
Crespo HJ, Ligeiro D, Calais FM, Trindade H and Dall'Olio F:
ST3Gal.I sialyltransferase relevance in bladder cancer tissues and
cell lines. BMC Cancer. 9:3572009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Carrascal MA, Severino PF, Guadalupe
Cabral M, Silva M, Ferreira JA, Calais F, Quinto H, Pen C, Ligeiro
D, Santos LL, et al: Sialyl Tn-expressing bladder cancer cells
induce a tolerogenic phenotype in innate and adaptive immune cells.
Mol Oncol. 8:753–765. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bustin SA, Benes V, Garson JA, Hellemans
J, Huggett J, Kubista M, Mueller R, Nolan T, Pfaffl MW, Shipley GL,
et al: The MIQE Guidelines: Minimum Information for Publication of
Quantitative Real-Time PCR Experiments. Clin Chem. 55:611–622.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Trinchera M, Zulueta A, Caretti A and
Dall'Olio F: Control of glycosylation-related genes by DNA
methylation: The intriguing case of the B3GALT5 gene and its
distinct promoters. Biology (Basel). 3. pp. 484–497. 2014
|
|
29
|
Dimitroff CJ, Lee JY, Rafii S, Fuhlbrigge
RC and Sackstein R: CD44 is a major E-selectin ligand on human
hematopoietic progenitor cells. J Cell Biol. 153:1277–1286. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Oxley SM and Sackstein R: Detection of an
L-selectin ligand on a hematopoietic progenitor cell line. Blood.
84:3299–3306. 1994.PubMed/NCBI
|
|
31
|
Dimitroff CJ, Lee JY, Schor KS, Sandmaier
BM and Sackstein R: Differential L-selectin binding activities of
human hematopoietic cell L-selectin ligands, HCELL and PSGL-1. J
Biol Chem. 276:47623–47631. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Schneider CA, Rasband WS and Eliceiri KW:
NIH Image to ImageJ: 25 years of image analysis. Nat Methods.
9:671–675. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Carrascal MA, Talina C, Borralho P,
Gonçalo Mineiro A, Henriques AR, Pen C, Martins M, Braga S,
Sackstein R and Videira PA: Staining of E-selectin ligands on
paraffin-embedded sections of tumor tissue. BMC Cancer. 18:4952018.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lin F and Prichard J: Handbook of
Practical Immunohistochemistry. Lin F and Prichard J: 2nd edition.
Springer; New York, NY: 2015
|
|
35
|
Grunnet M and Sorensen JB:
Carcinoembryonic antigen (CEA) as tumor marker in lung cancer. Lung
Cancer. 76:138–143. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
McEver RP: Selectins: Initiators of
leucocyte adhesion and signalling at the vascular wall. Cardiovasc
Res. 107:331–339. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Dimitroff CJ, Kupper TS and Sackstein R:
Prevention of leukocyte migration to inflamed skin with a novel
fluorosugar modifier of cutaneous lymphocyte-associated antigen. J
Clin Invest. 112:1008–1018. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Holmes EH, Ostrander GK and Hakomori S:
Biosynthesis of the sialyl-Lex determinant carried by type 2 chain
glycosphin-golipids (IV3NeuAcIII3FucnLc4, VI3NeuAcV3FucnLc6, and
VI3NeuAcIII3V3Fuc2nLc6) in human lung carcinoma PC9 cells. J Biol
Chem. 261:3737–3743. 1986.PubMed/NCBI
|
|
39
|
Shimizu T, Yonezawa S, Tanaka S and Sato
E: Expression of Lewis X-related antigens in adenocarcinomas of
lung. Histopathology. 22:549–555. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Shimizu S, Suzuki H and Noguchi E: Sialyl
Lewis X-i (SLX) in the bronchoalveolar lavage fluid from patients
with lung cancer. Nihon Kyobu Shikkan Gakkai Zasshi. 30:815–820.
1992.In Japanese. PubMed/NCBI
|
|
41
|
Mizuguchi S, Inoue K, Iwata T, Nishida T,
Izumi N, Tsukioka T, Nishiyama N, Uenishi T and Suehiro S: High
serum concentrations of sialyl lewisx predict multilevel N2 disease
in non-small-cell lung cancer. Ann Surg Oncol. 13:1010–1018. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Fukuoka K, Narita N and Saijo N: Increased
expression of sialyl Lewis (x) antigen is associated with distant
metastasis in lung cancer patients: Immunohistochemical study on
bronchofiber-scopic biopsy specimens. Lung Cancer. 20:109–116.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Takada A, Ohmori K, Yoneda T, Tsuyuoka K,
Hasegawa A, Kiso M and Kannagi R: Contribution of carbohydrate
antigens sialyl Lewis A and sialyl Lewis X to adhesion of human
cancer cells to vascular endothelium. Cancer Res. 53:354–361.
1993.PubMed/NCBI
|
|
44
|
Jassam SA, Maherally Z, Smith JR, Ashkan
K, Roncaroli F, Fillmore H and Pilkington GJ: CD15s/CD62E
interaction mediates the adhesion of non-small cell lung cancer
cells on brain endothelial cells: Implications for cerebral
metastasis. Int J Mol Sci. 18:E14742017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Bendas G and Borsig L: Cancer cell
adhesion and metastasis: Selectins, integrins, and the inhibitory
potential of heparins. Int J Cell Biol. 2012:6767312012. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Witz IP: The selectin-selectin ligand axis
in tumor progression. Cancer Metastasis Rev. 27:19–30. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Satoh H, Ishikawa H, Yamashita YT, Ohtsuka
M and Sekizawa K: Serum sialyl Lewis X-i antigen in lung
adenocarcinoma and idiopathic pulmonary fibrosis. Thorax.
57:263–266. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Trinchera M, Aronica A and Dall'Olio F:
Selectin ligands sialyllewis a and sialyl-Lewis X in
gastrointestinal cancers. Biology (Basel). 6:E162017.
|
|
49
|
Schweitzer KM, Dräger AM, van der Valk P,
Thijsen SF, Zevenbergen A, Theijsmeijer AP, van der Schoot CE and
Langenhuijsen MM: Constitutive expression of E-selectin and
vascular cell adhesion molecule-1 on endothelial cells of
hematopoietic tissues. Am J Pathol. 148:165–175. 1996.PubMed/NCBI
|
|
50
|
Weninger W, Ulfman LH, Cheng G, Souchkova
N, Quackenbush EJ, Lowe JB and von Andrian UH: Specialized
contributions by alpha(1,3)-fucosyltransferase-IV and FucT-VII
during leukocyte rolling in dermal microvessels. Immunity.
12:665–676. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Kannagi R, Sakuma K, Miyazaki K, Lim KT,
Yusa A, Yin J and Izawa M: Altered expression of glycan genes in
cancers induced by epigenetic silencing and tumor hypoxia: Clues in
the ongoing search for new tumor markers. Cancer Sci. 101:586–593.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Chachadi VB, Bhat G and Cheng PW:
Glycosyltransferases involved in the synthesis of MUC-associated
metastasis-promoting selectin ligands. Glycobiology. 25:963–975.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Yoshihama N, Yamaguchi K, Chigita S, Mine
M, Abe M, Ishii K, Kobayashi Y, Akimoto N, Mori Y and Sugiura T: A
novel function of CD82/KAI1 in sialyl Lewis antigen-mediated
adhesion of cancer cells: Evidence for an anti-metastasis effect by
down-regulation of sialyl Lewis antigens. PLoS One.
10:e01247432015. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Togayachi A, Kudo T, Ikehara Y, Iwasaki H,
Nishihara S, Andoh T, Higashiyama M, Kodama K, Nakamori S and
Narimatsu H: Up-regulation of Lewis enzyme (Fuc-TIII) and
plasma-type alpha1,3fucosyltransferase (Fuc-TVI) expression
determines the augmented expression of sialyl Lewis x antigen in
non-small cell lung cancer. Int J Cancer. 83:70–79. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Barthel SR, Wiese GK, Cho J, Opperman MJ,
Hays DL, Siddiqui J, Pienta KJ, Furie B and Dimitroff CJ: Alpha 1,3
fucosyltransferases are master regulators of prostate cancer cell
trafficking. Proc Natl Acad Sci USA. 106:19491–19496. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Kannagi R, Yin J, Miyazaki K and Izawa M:
Current relevance of incomplete synthesis and neo-synthesis for
cancer-associated alteration of carbohydrate
determinants-Hakomori's concepts revisited. Biochim Biophys Acta.
1780:525–531. 2008. View Article : Google Scholar
|
|
57
|
Malagolini N, Santini D, Chiricolo M and
Dall'Olio F: Biosynthesis and expression of the Sda and sialyl
Lewis x antigens in normal and cancer colon. Glycobiology.
17:688–697. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Groux-Degroote S, Wavelet C,
Krzewinski-Recchi MA, Portier L, Mortuaire M, Mihalache A,
Trinchera M, Delannoy P, Malagolini N, Chiricolo M, et al: B4GALNT2
gene expression controls the biosynthesis of Sda and sialyl Lewis X
antigens in healthy and cancer human gastrointestinal tract. Int J
Biochem Cell Biol. 53:442–449. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Shinagawa T, Hoshino H, Taga M, Sakai Y,
Imamura Y, Yokoyama O and Kobayashi M: Clinicopathological
implications to micropapillary bladder urothelial carcinoma of the
presence of sialyl Lewis X-decorated mucin 1 in stromafacing
membranes. Urol Oncol. 35:606.e17–606.e23. 2017. View Article : Google Scholar
|
|
60
|
Burdick MM, Chu JT, Godar S and Sackstein
R: HCELL is the major E- and L-selectin ligand expressed on LS174T
colon carcinoma cells. J Biol Chem. 281:13899–13905. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Woodman N, Pinder SE, Tajadura V, Le
Bourhis X, Gillett C, Delannoy P, Burchell JM and Julien S: Two
E-selectin ligands, BST-2 and LGALS3BP, predict metastasis and poor
survival of ER-negative breast cancer. Int J Oncol. 49:265–275.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Sackstein R: The lymphocyte homing
receptors: Gatekeepers of the multistep paradigm. Curr Opin
Hematol. 12:444–450. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Hidalgo A, Peired AJ, Wild M, Vestweber D
and Frenette PS: Complete identification of E-selectin ligands on
neutrophils reveals distinct functions of PSGL-1, ESL-1, and CD44.
Immunity. 26:477–489. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Mitoma J, Miyazaki T, Sutton-Smith M,
Suzuki M, Saito H, Yeh JC, Kawano T, Hindsgaul O, Seeberger PH,
Panico M, et al: The N-glycolyl form of mouse sialyl Lewis X is
recognized by selectins but not by HECA-452 and FH6 antibodies that
were raised against human cells. Glycoconj J. 26:511–523. 2009.
View Article : Google Scholar :
|
|
65
|
Thomas SN, Zhu F, Schnaar RL, Alves CS and
Konstantopoulos K: Carcinoembryonic antigen and CD44 variant
isoforms cooperate to mediate colon carcinoma cell adhesion to E-
and L-selectin in shear flow. J Biol Chem. 283:15647–15655. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Burdick MM, Henson KA, Delgadillo LF, Choi
YE, Goetz DJ, Tees DF and Benencia F: Expression of E-selectin
ligands on circulating tumor cells: Cross-regulation with cancer
stem cell regulatory pathways? Front Oncol. 2:1032012. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Duffy MJ: Carcinoembryonic antigen as a
marker for colorectal cancer: Is it clinically useful? Clin Chem.
47:624–630. 2001.PubMed/NCBI
|
|
68
|
Wang J, Ma Y, Zhu ZH, Situ DR, Hu Y and
Rong TH: Expression and prognostic relevance of tumor
carcinoembryonic antigen in stage IB non-small cell lung cancer. J
Thorac Dis. 4:490–496. 2012.PubMed/NCBI
|
|
69
|
Beauchemin N and Arabzadeh A:
Carcinoembryonic antigen-related cell adhesion molecules (CEACAMs)
in cancer progression and metastasis. Cancer Metastasis Rev.
32:643–671. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Holdenrieder S, Wehnl B, Hettwer K, Simon
K, Uhlig S and Dayyani F: Carcinoembryonic antigen and
cytokeratin-19 fragments for assessment of therapy response in
non-small cell lung cancer: A systematic review and meta-analysis.
Br J Cancer. 116:1037–1045. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Li X, Asmitananda T, Gao L, Gai D, Song Z,
Zhang Y, Ren H, Yang T, Chen T and Chen M: Biomarkers in the lung
cancer diagnosis: A clinical perspective. Neoplasma. 59:500–507.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Kadara H and Wistuba II: Field
cancerization in non-small cell lung cancer: Implications in
disease pathogenesis. Proc Am Thorac Soc. 9:38–42. 2012. View Article : Google Scholar
|
|
73
|
Lochhead P, Chan AT, Nishihara R, Fuchs
CS, Beck AH, Giovannucci E and Ogino S: Etiologic field effect:
Reappraisal of the field effect concept in cancer predisposition
and progression. Mod Pathol. 28:14–29. 2015. View Article : Google Scholar
|




















