Introduction
Although advanced techniques are being developed to
treat breast cancer, which is the most common cancer among women,
its mortality is still steadily increasing each year (1). Previous studies demonstrated that
mortality was closely associated with metastasis: The 5-year
survival rate of breast cancer patients diagnosed with
non-metastasized tumor was ≥90%, while the survival rate of
metastatic breast cancer patients was ~27% (2,3).
Therefore, it is necessary to understand the mechanism of
metastasis in cancer to improve the 5-year survival rate and lower
the risk of recurrence.
Metastasis entails a number of complex steps,
including detachment of primary cancer cells, migration, invasion,
and attachment of detached cancer cells at secondary sites
(4). During these steps, matrix
metalloproteinases (MMPs), urokinase plasminogen activator (uPA),
and uPA receptor (uPAR) play important roles in the degradation of
extracellular matrix (ECM) and basement membrane (5,6).
Among MMPs, MMP-9 plays an important role in breast cancer
metastasis. Several investigations have revealed the relationship
between the MMP-9 level and metastasis in breast cancer patients or
breast cancer cell models (6-10).
Certain studies observed a high metastatic potential in
MMP-9-overexpressing breast cancer cells (11,12).
Furthermore, other researchers demonstrated that the inhibition of
MMP-9 expression decreased the metastatic potential in breast
cancer cells, implicating that the inhibition of MMP-9 expression
may be a useful strategy for the prevention of metastasis in breast
cancer (7,13). uPA, which activates MMP-9 and
degrades ECM and BM via catalyzing plasmin production, is also a
key proteolytic enzyme for breast cancer metastasis (6). Furthermore, certain clinical studies
showed that the prognosis and recurrence rate of breast cancer were
linked with the uPA level in tumor tissue or serum, indicating that
uPA is an important prognostic factor in breast cancer (5,14,15).
Taken together, these previous studies demonstrated that the
monitoring of MMP-9 and uPA is an important strategy for the
prevention of metastasis in breast cancer.
A number of studies demonstrated that the metastatic
potential in various cancer models was predominantly linked with
the transcriptional regulation of MMP-9 and uPA, mediated by
activator protein-1 (AP-1) and/or NF-κB (16,17).
Furthermore, several investigations revealed that natural
substances and/or chemicals reversed the
12-O-tetradecanoylphorbol-13-acetate (TPA)-induced metastatic
potential via the regulation of MMP-9 and/or uPA expression through
the regulation of c-Jun and/or c-Fos expression in breast cancer
cells (7,18,19).
These investigations imply that AP-1 is an important transcription
factor for the regulation of MMP-9 and uPA expression in breast
cancer cells.
Fibronectin plays an important role in MMP-9
secretion and is a potent marker of poor breast cancer prognosis
(20,21). When cancer cells are released from
the primary tumor and invade other tissues, several proteases, such
as MMP-9, uPA, uPAR and MMP-7, are involved in this process.
Fibronectin participates in metastasis via stimulating MMP-9
secretion and increasing the metastatic potential (22-27).
Furthermore, migration and invasion of MCF-7 cells were triggered
by fibronectin treatment with the EMT-like morphological change
(20). The aforementioned
investigations demonstrate the importance of fibronectin in the
regulation of metastatic potential in cancer.
Plant flavonoid 2′,3,4′,5,7-pentahydroxyflavone
(morin hydrate), naturally occurring in almonds, bilberries,
Moraceae family and leaves of Cudranaia tricuspidata Buread,
has various physiological properties, including antihypertensive,
antiangiogenic, hepatoprotective, and anti-inflammatory activities
(28-30). However, the physiological activity
of morin hydrate in hormone receptor-positive breast cancer has not
been fully elucidated to date. The current study investigated the
effects of morin hydrate on the TPA-induced metastatic potential in
MCF-7 hormone receptor-positive human breast cancer cells.
Materials and methods
Materials
Morin hydrate, bicinchoninic acid (BCA) solution,
TPA, hematoxylin, eosin, and sulforhodamine B (SRB) were purchased
from Sigma-Aldrich; Merck KGaA. DMEM, insulin solution, trypsin and
antibiotic/antimycotic solution were acquired from Welgene, Inc.
Trichloroacetic acid (TCA) was purchased from Samchun Pure Chemical
Co., Ltd., and 30% polyacrylamide solution, protease inhibitor
cocktail, and phosphatase inhibitor cocktail were obtained from
GenDEPOT. Primary antibodies for Akt (cat. no. 4691), phospho-Akt
(cat. no. 4060), ERK1/2 (cat. no. 4695), phospho-ERK1/2 (cat. no.
4370), JNK (cat. no. 9258), phospho-JNK (cat. no. 4668), p38
mitogen-activated protein kinase (MAPK; cat. no. 8690), phospho-p38
MAPK (cat. no. 4511), NF-κB (cat. no. 8242), phospho-NF-κB (cat.
no. 3033), c-Jun (cat. no. 9165), c-Fos (cat. no. 2250), glycogen
synthase kinase (GSK)-3β (cat. no. 5676), phospho-GSK-3α/β (cat.
no. 9331) and GAPDH (cat. no. 5174) were purchased from Cell
Signaling Technology, Inc., and goat anti-rabbit immunoglobulin
G-horseradish peroxidase-conjugated antibody (cat. no. 31460) was
from Thermo Fisher Scientific, Inc. PI3K/Akt inhibitors LY294002
and wortmannin were obtained from LC Laboratories and Cayman
Chemical Company, respectively. Primers for reverse
transcription-quantitative PCR (RT-qPCR) were synthesized by
Bioneer Corporation.
Cell culture
MCF-7 human breast cancer cells were obtained from
the Korean Cell Line Bank. The cells were grown in DMEM
supplemented with 10% FBS (American Type Culture Collection), 10
µg/ml insulin and 1% antimycotic/antibiotic mixture at 37°C
in 5% CO2 atmosphere. The cells were serum-starved for
24 h prior to the addition of conditioned DMEM containing morin
hydrate (0-200 µM) and/or 100 nM TPA at 37°C in 5%
CO2 atmosphere. All cell culture experiments were
performed at 37°C in a 5% CO2 atmosphere in DMEM.
Analysis of cytotoxic effect of morin
hydrate
A total of 5,000 cells were seeded into a 96-well
plate in each well and attached for 24 h in DMEM supplemented with
10% FBS. Then, DMEM supplemented with 10% FBS in each well was
replaced with conditioned DMEM containing 5% FBS and various
concentration of morin hydrate. The cells were further incubated
for 24, 48 or 72 h; then the conditioned DMEM was removed. The
cells were fixed with 20% TCA solution for 1 h, washed with tap
water, and dried for 2 h, all at room temperature. To stain the
dried cells, 0.4% SRB solution was added into each well and stored
at room temperature for 30 min. The stained cells were rinsed with
1% acetic acid and dried at room temperature. Then, 100 µl
of 10 mM Tris-HCl (pH 5.0) buffer was added in each well to
dissolve SRB, and absorption was measured at a wavelength of 510 nm
by a SpectraMax® M2e microplate reader (Molecular
Devices, LLC).
Wound healing cell migration assay
A total of 5×105 cells/well were seeded
into collagen-coated six-well plates and attached for 24 h in DMEM
supplemented with 10% FBS. Then, the cell monolayers were scratched
by 1 ml micropipette tip and DMEM supplemented with 10% FBS was
removed by aspiration. Then, the cell monolayers were washed by PBS
to remove detached cells and serum-starved for 24 h in serum-free
DMEM. Then, serum-free DMEM was removed and the cell monolyers were
treated with various concentration of morin hydrate dissolved in
DMEM containing 1% FBS for 1 h prior to taking photographs. Then,
the cells were further administrated with TPA to trigger cell
migration for 24 h and the scratch zones were observed and
photographed. The change of the width of the scratch zone was
observed and images were captured with Nikon light microscope
(Nikon Corporation).
Matrigel invasion assay
A total of 3×104 cells were plated in the
upper chambers of 24-well transwell plates (8-µm-pore;
Corning Inc.) in cold serum-free DMEM. For the invasion assay,
plates were coated with the Matrigel® (BD Biosciences)
for 1 h. DMEM supplemented 10% FBS were added in the lower
chambers; then, the cells were cultured for 24 h in the upper
chamber. The media in the upper chambers were replaced with
serum-free DMEM and various concentrations of morin hydrate. The
media in lower chambers were replaced with new DMEM supplemented
with 10% FBS. After 1 h, TPA was added into the upper chambers as
an inducer for invasion. The cells were further incubated for 24 h
at 37°C in 5% CO2 atmosphere. The cells that migrated
through or invaded the polycarbonate filter were fixed with 100%
methanol and stained with heamatoxylin and eosin Y solution for 10
min. The stained cells were observed with light microscope and
images were captured using an inverted optical microscope. ImageJ
(version 1.6.0_20) was used for data analysis (National Institutes
of Health).
Analysis of MMP-9 activity by gelatin
zymography
A total of 5×105 cells/well were attached
for 24 h and then further cultured for 24 h in FBS-free DMEM for
serum-starvation. The cells were treated with various
concentrations of morin hydrate with 100 nM TPA for 24 h. The DMEM
were electrophoresed with 8% non-denaturing SDS-PAGE containing
0.1% (v/v) gelatin. The gel was washed with 0.25% Triton X-100
solution to remove SDS and incubated at 37°C in a reaction buffer
[5 mM CaCl2, 0.04% NaN3 and 50 mM Tris-HCl]
overnight to confirm the gelatinase activity of MMP-9. Bands were
visualized by Coomassie brilliant blue R staining for 1 h and
images were captured with LAS-4000 image analyzer (Fujifilm).
ImageJ was used for data analysis.
Analysis of the change of nuclear c-Fos
level by PI3K/Akt inhibitors
A total of 5×105 cells/well were attached
for 24 h and then further cultured for 24 h in FBS-free DMEM for
serum-starvation. The cells were pretreated for 24 h with various
concentrations of morin hydrate and further incubated with or
without Akt inhibitors, LY294002 (20 µM) and wortmannin (20
µM), for 30 min prior to TPA treatment for 1 h at 37°C.
Then, the cells were harvested and analyzed by western
blotting.
RNA isolation and RT-qPCR
The cells were treated with morin hydrate and 100 nM
TPA for 24 h after 24-h attachment and 24-h serum-starvation. To
isolate total RNA, the cells were harvested by trypsinization,
collected by centrifugation at room temperature for 3 min at 1,000
× g and lysed with easy-BLUE™ Total RNA Extraction kit (Intron
Biotechnology, Inc.). The extraction of total RNA from the lysed
cells was performed in accordance with the manufacturer’s protocol.
Total RNA (1 µg) was used for cDNA synthesis. RT reaction
was performed to synthesize cDNA in 1X GoScript™ reaction buffer
containing 2 mM MgCl2 with GoScript Reverse
Transcriptase, 0.5 mM dNTPs (Promega Corporation) and pd(N)9 Random
Primers (Takara Bio, Inc.). qPCR was performed using Q Green Sybr
Green Master Mix kit (CellSafe Co., Ltd.) using Eco™ Real-Time PCR
system (Illumina, Inc.). The following thermocycling conditions
were used: Initial denaturation at 95°C for 5 min; 45 cycles of
95°C for 10 sec, 60°C for 20 sec and 72°C for 20 sec. Relative mRNA
expression was automatically calculated using EcoStudy software
(version 5.0.49; Illumina, Inc.). The primer sequences for PCR
amplification are presented in Table
I.
 | Table INucleotide sequences for primers used
in quantitative PCR. |
Table I
Nucleotide sequences for primers used
in quantitative PCR.
| Target | Primer sequence
(5′-3′)
|
|---|
| Forward | Reverse |
|---|
| Fibronectin |
CAGTGGGAGACCTCGAGAAG |
TCCCTCGGAACATCAGAAAC |
| MMP-7 |
GGGACTCCTACCCATTTG |
CAGCGTTCATCCTCATCG |
| MMP-9 |
GGGACGCAGACATCGTCATC |
TCGTCATCGTCGAAATGGGC |
| uPA |
GGAGATGAAGTTTGAGGTGG |
GGTCTGTATAGTCCGGGATG |
| uPAR |
CACAAAACTGCCTCCTTCCT |
AATCCCCGTTGGTCTTACAC |
| E-cadherin |
CGACCCAACCCAAGAATCTA |
AGGCTGTGCCTTCCTACAGA |
| N-cadherin |
TGAGCCTGAAGCCAACCTTA |
AGGTCCCCTGGAGTTTTCTG |
| GAPDH |
CTGCTCCTCCTGTTCGACAGT |
CCGTTGACTCCGACCTTCAC |
Nuclear fractionation
A total of 5×105 cells/well were attached
for 24 h and then further cultured for 24 h in FBS-free DMEM for
serum-starvation. The cells were pretreated with or without morin
hydrate for 24 h and then further incubated with or without TPA for
1 h with serum-free DMEM. Then, DMEM was removed, the cells were
washed twice with ice-cold PBS and treated with a hypotonic buffer
(20 mM Tris-HCl pH 7.4, 10 mM NaCl and 3 mM MgCl2)
containing phosphatase and protease inhibitor cocktails. The cells
were collected using a rubber policeman scraper, transferred to new
microtubes and incubated for 15 min on ice. Then, 1/8 vol 10% NP-40
(Sigma-Aldrich; Merck KGaA) was added and the cells were vortexed
for 10 sec at the highest speed setting. The cells were incubated
on ice for 10 min and centrifuged at 2,500 × g for 10 min at 4°C.
The supernatants were removed as the cytosolic fraction, and the
pellet was lysed for 30 min on ice with the Cell Extraction Buffer
(Invitrogen; Thermo Fisher Scientific, Inc.) containing phosphatase
and protease inhibitor cocktails. The lysates were separated by
centrifugation at 14,000 × g for 30 min at 4°C and the supernatants
were removed as the nuclear fraction.
Western blotting
A total of 5×105 cells/well were attached
for 24 h and then further cultured for 24 h in FBS-free DMEM for
serum-starvation. The cells were pretreated with or without morin
hydrate for 24 h and then further incubated with or without TPA for
30 min with serum-free DMEM. Then, DMEM was removed, the cells were
washed twice with ice-cold PBS, and whole cell lysates were
prepared with radioimmunoprecipitation assay lysis buffer
(Biosesang) supplemented with protease inhibitor and phosphatase
inhibitor cocktails. The lysed cells were centrifuged at 14,000 × g
for 10 min at 4°C, and the supernatants were collected as the whole
cell lysate and stored at -80°C until further use. Equal amounts
(20 µg) of protein form the whole cell lysate, nuclear
fraction or cytosolic fraction were subjected to 8 or 10% SDS-PAGE
and transferred to PVDF membranes (Pall Life Sciences). Blocking
was performed with 5% non-fat dry milk or 1% bovine serum albumin
(BSA; Santa Cruz Biotechnology, Inc.) in Tris-buffered saline-Tween
(TBST; 50 mM Tris-HCl, 150 mM NaCl, 0.1% Tween-20) solution for 1 h
at room temperature. The primary antibodies for target proteins
were diluted at 1:3,000 in 5% non-fat dry milk or 1% BSA in TBST
solution, and the reactions for primary antibodies were conducted
overnight at 4°C. Subsequently, the membrane was washed by TBST
solution, covered with secondary antibody diluted at 1:5,000 in TBS
solution and incubated for 1 h at room temperature. The target
protein bands were detected by a chemiluminescent substrate [100 mM
Tris (pH 8.5; BioShop Canada, Inc.), 1.25 mM luminol, 198 µM
coumaric acid and 0.01% hydrogen peroxide (all Sigma-Aldrich; Merck
KGaA)] and images were captured using LAS-4000 image analyzer. The
band densities were analyzed with Scion Image software (Alpha
4.0.3.2; Scion Corporation).
Statistical analysis
Data were analyzed using one-way ANOVA followed by
Turkey post hoc test, using SPSS software (version 20.0; IBM
Corp.). The values are presented as the mean ± SD. P<0.05 was
considered to indicate a statistically significant difference.
Results
Effect of morin hydrate on MCF-7 human
breast cancer cell viability
Previous studies showed that morin hydrate had no
cytotoxic activity in KEL FIB human fibroblast cells and EA.hy 926
human umbilical endothelial cells (31,32).
Therefore, the current study firstly tested the effects of morin
hydrate on cell viability in MCF-7 human breast cancer cells using
an SRB colorimetric assay. MTT assay was not used, as the
absorbance of insoluble formazan produced by MTT reagents is
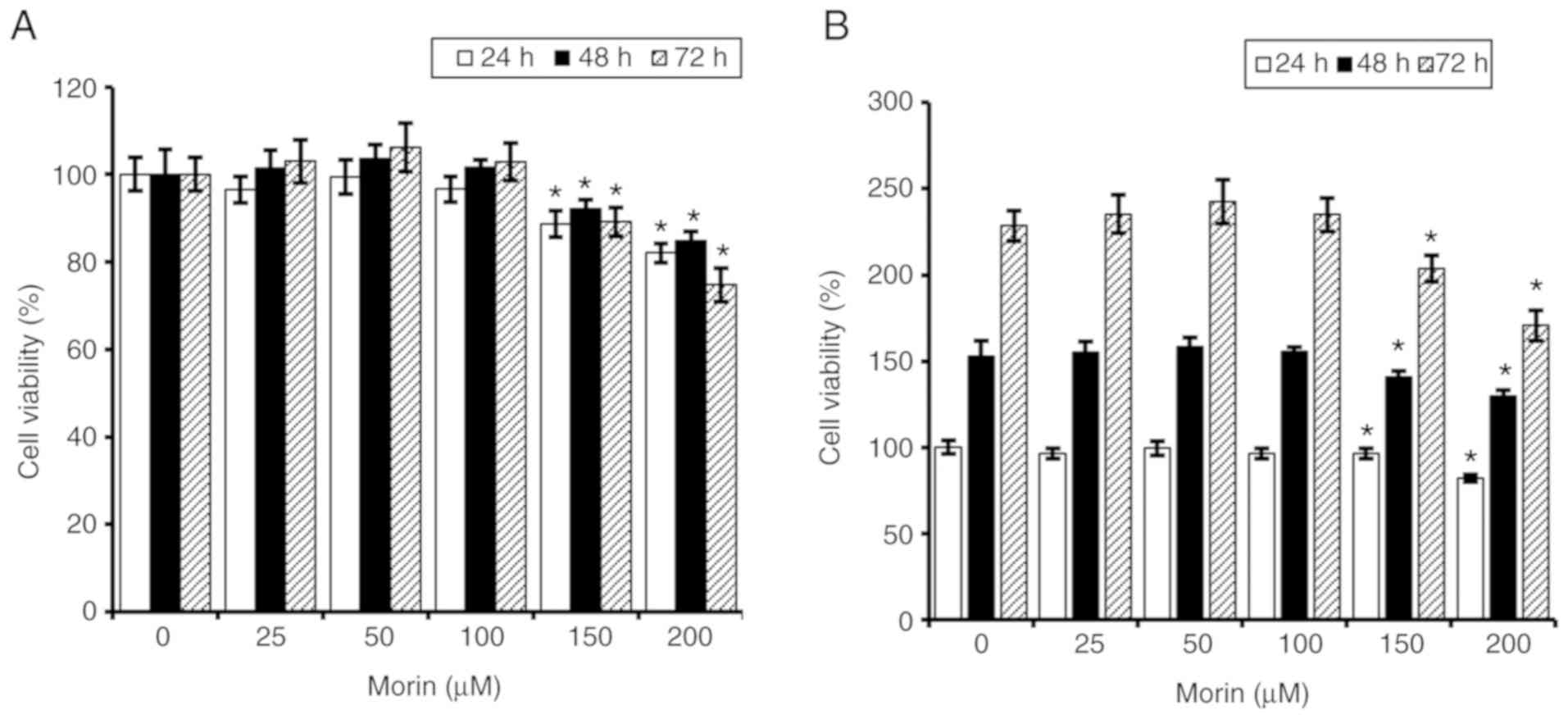
affected by morin hydrate. The result of the current study showed
that cell viability of MCF-7 cells was slightly decreased after
treatment with 0-200 µM morin hydrate and the maximum
reduction rate of >20% was observed after treatment with 200
µM morin hydrate for 72 h (Fig.
1A). Furthermore, the reduction of cell viability was
associated with the delayed cell growth (Fig. 1B). These results suggested that
morin hydrate had a weak antiproliferative activity when
administered at a concentration range of 150-200 µM in MCF-7
hormone receptor-positive human breast cancer cells.
Effect of morin hydrate on TPA-induced
migration and invasion of MCF-7 human breast cancer cells
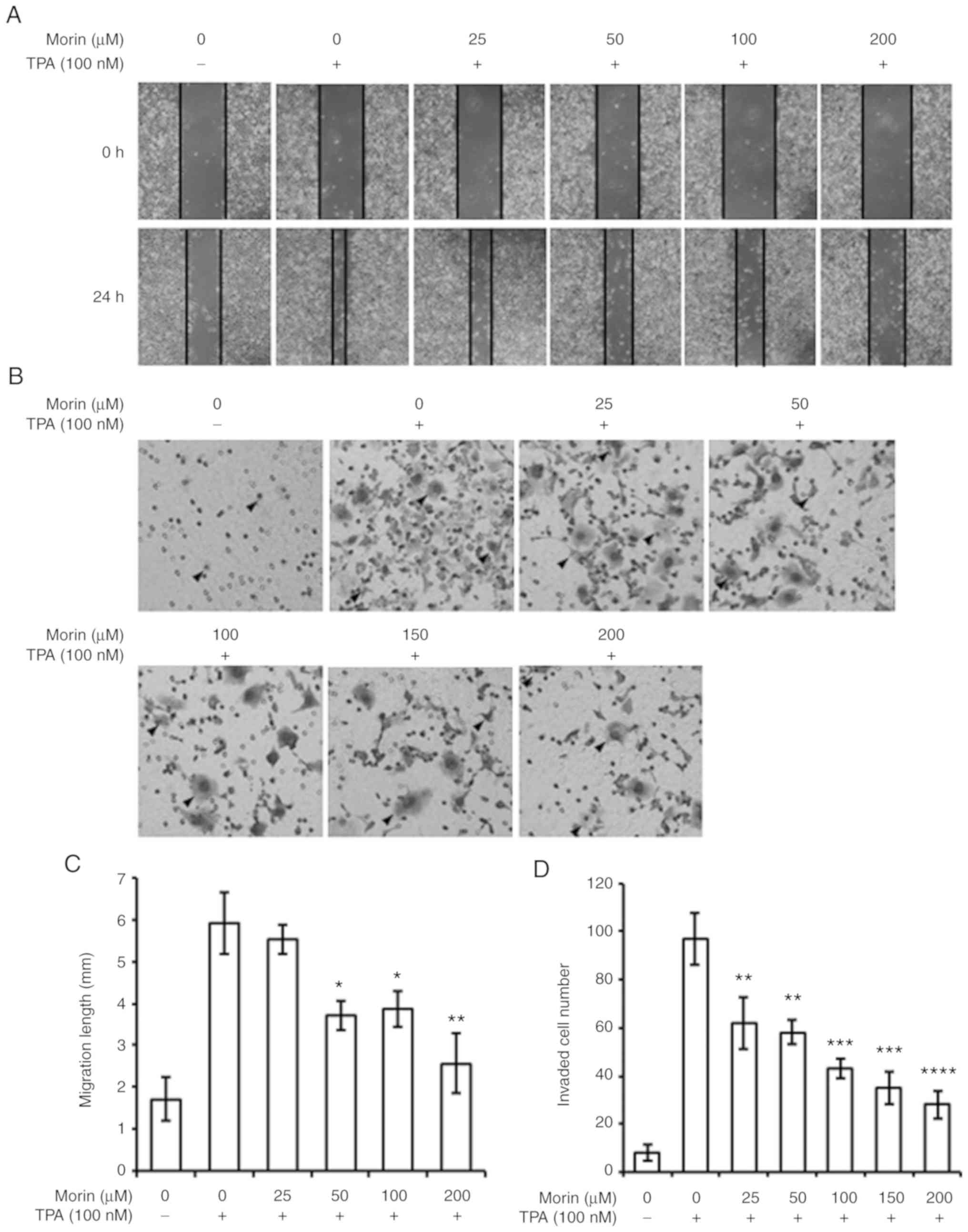
The current study assessed whether morin hydrate
could prevent TPA-induced metastatic potential in MCF-7 human
breast cancer cells using cell migration and invasion assays. The
result showed that TPA-enhanced cell migration was decreased by
morin hydrate treatment in a concentration-dependent manner
(Fig. 2A and C). Furthermore, the
invasiveness promoted by TPA was also reversed by the presence of
morin hydrate in MCF-7 cells in a concentration-dependent manner
(Fig. 2B and D). These results
suggest that morin hydrate could suppress the TPA-induced
metastatic potential in MCF-7 cells.
Effect of morin hydrate on metastatic
factors in TPA-treated MCF-7 cells
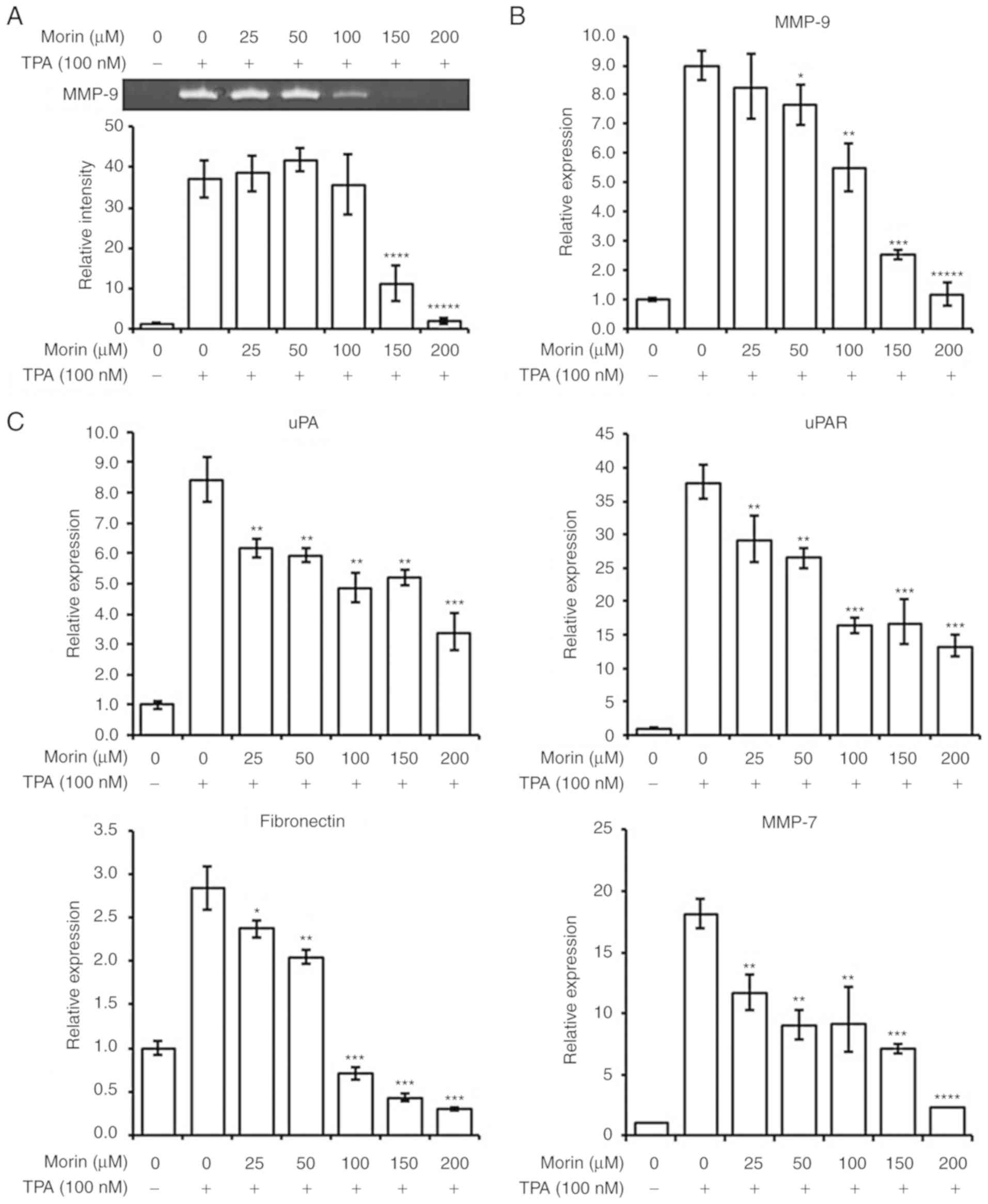
As indicated above, morin hydrate decreased the
invasiveness of MCF-7 cells induced by TPA. Therefore, the current
study further evaluated the effects of morin hydrate on MMP-9
activity and expression levels of MMP-9, uPA, uPAR, fibronectin and
MMP-7. According to the gelatin zymographic analysis, the
proteolytic activity of MMP-9 enhanced by TPA was significantly
suppressed by 150 and 200 µM morin hydrate treatment in
MCF-7 cells (Fig. 3A).
Furthermore, MMP-9, uPA, uPAR and MMP-7 transcription levels were
also decreased by morin hydrate supplementation in a
concentration-dependent manner (Fig.
3B and C). In addition, gene expression of fibronectin, a known
EMT factor, was significantly reduced by morin hydrate treatment
(Fig. 3C). However, TPA did not
affect the expression of E-cadherin and N-cadherin which are also
EMT factors (data not shown). The results suggested that morin
hydrate could prevent the participation of MMP-9 activity induced
by TPA via negative regulation of MMP-9, uPA, uPAR, fibronectin and
MMP-7.
Morin hydrate inhibits TPA-induced
phosphorylation of Akt and GSK-3β and the expression of c-Fos
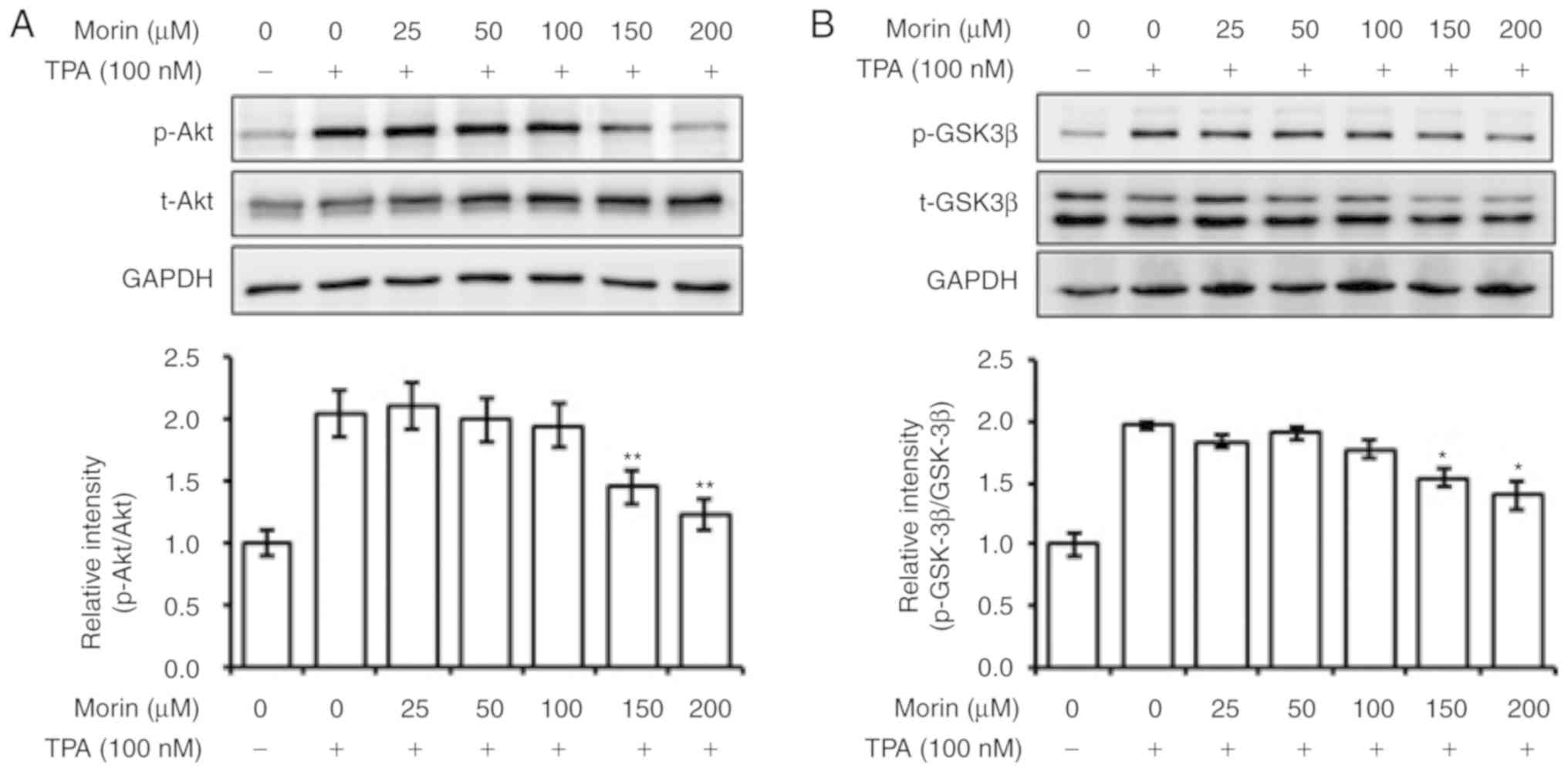
The current study evaluated the effects of morin
hydrate on the phosphorylation levels of Akt, MAPK and GSK3β, as
well as on the expression levels of AP-1 subunits c-Jun and c-Fos.
The result showed that morin hydrate suppressed the phosphorylation
of Akt and GSK-3β, in addition to the expression of c-Fos induced
by TPA, in a concentration-dependent manner (Fig. 4A-C). By contrast, phosphorylation
levels of ERK1/2 and NF-κB, and the expression of c-Jun, which are
also involved in the regulation of expression of
metastasis-enhancing factors, were not affected by morin hydrate
treatment (Fig. 4C and D). JNK and
p38 phosphorylation levels were also not altered by treatment with
morin hydrate (data not shown). These results suggest that the
inhibition of expression of metastasis-enhancing factors by morin
hydrate may be associated with the reversal of Akt and GSK3β
phosphorylation and inhibition of c-Fos expression.
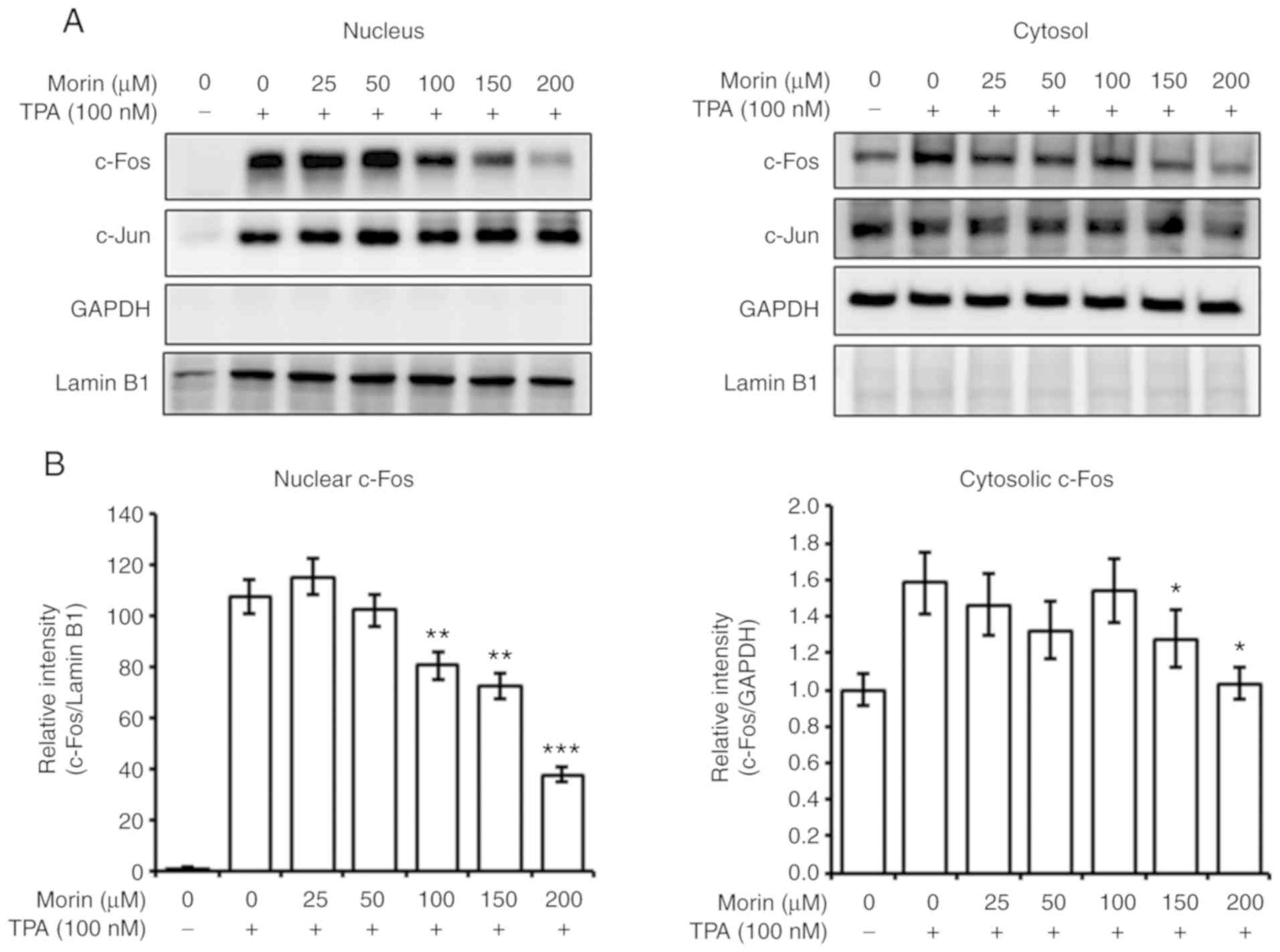
Morin hydrate decreases the TPA-induced
nuclear c-Fos level
Therefore, the current study assessed the effect of
morin hydrate on the nuclear localization of c-Fos and c-Jun. Morin
hydrate decreased the nuclear c-Fos level increased by TPA, in a
concentration-dependent manner (Fig.
5A and B). However, the nuclear c-Jun level was not changed by
morin hydrate treatment (Fig. 5A).
These results imply that morin hydrate could inhibit AP-1
transcriptional activity by decreasing the c-Fos level in the
nucleus. The change of the nuclear c-Fos level was also associated
with altered whole cell c-Fos expression.
Akt inhibitors suppress GSK-3β
phosphorylation and c-Fos expression
The results of the current study revealed that morin
hydrate inhibited Akt and GSK-3β phosphorylation, and c-Fos
expression (Fig. 6A), without the
change of ERK and NF-κB phosphorylation (Fig. 4D). Therefore, the current study
tested the effects of PI3K/Akt inhibitors, LY294002 and wortmannin,
on Akt and GSK-3β phosphorylation and c-Fos expressions. The
PI3K/Akt inhibitors downregulated the TPA-induced Akt and GSK-3β
phosphorylation as well as the c-Fos expression. These results
suggested that the inhibitory effect of morin hydrate on the
metastatic potential may be regulated by the inhibition of the
PI3K/Akt-mediated GSK-3β phosphorylation and c-Fos expression
(Fig. 6B).
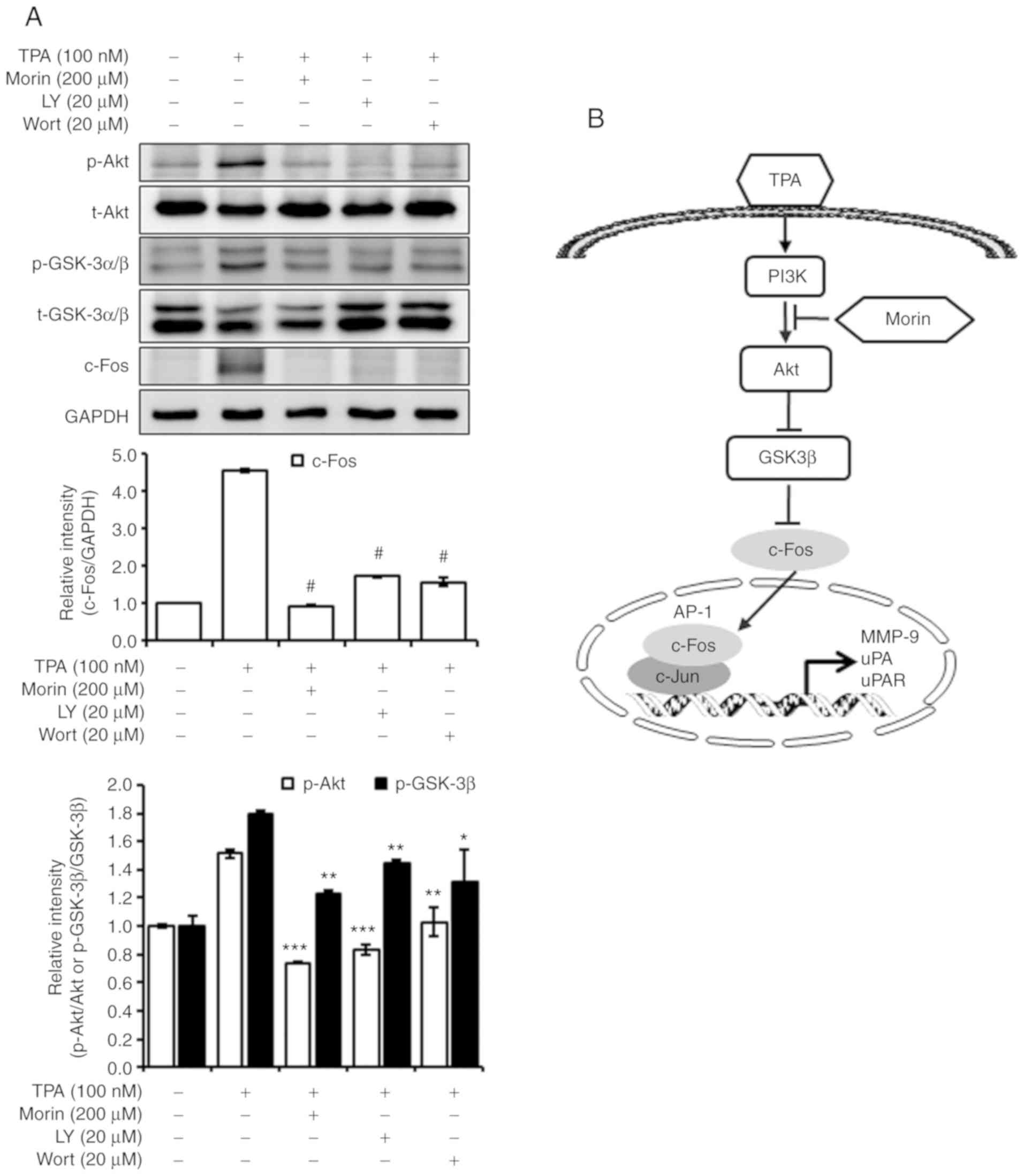 | Figure 6Decrease in nuclear c-Fos level by
morin hydrate is regulated by the Akt-mediated signaling pathway.
(A) Akt inhibitors, LY294002 and wort-mannin, and morin hydrate,
inhabited the phosphorylation of Akt and GSK-3β as well as the
expression of c-Fos induced by TPA. The cells were pretreated with
morin hydrate for 24 h and treated with Akt inhibitors, LY (20
µM) and wort (20 µM), for 30 min prior to TPA
treatment for 1 h at 37°C. The whole cell lysates were subjected to
10% SDS-PAGE. Bands were developed by a chemiluminescent substance
and images were captured using LSA-4000 image analyzer. GAPDH was
used as the internal control. Three independent experiments were
performed. Data are presented as the mean ± SD.
*P<0.05, **P<0.01,
***P<0.001 and #P<0.0001 vs. TPA only.
(B) Proposed mechanism of morin hydrate-mediated inhibition of
TPA-induced expression of metastasis-enhancing factors. TPA,
12-O-tetradecanoylphorbol-13-acetate; GSK, glycogen synthase
kinase. LY, LY294002; Wort, wortmannin; p, phospho; t, total; MMP,
matrix metalloproteinase; uPA, urokinase plasminogen activator;
uPAR, uPA receptor. |
Discussion
Metastasis is one of the major causes of poor
prognosis of patients with cancer. Therefore, prevention of
metastasis is an important strategy for effective cancer therapy.
Metastasis is primarily regulated by a number of factors, including
MMPs, uPA and uPAR. Numerous investigations revealed that the
inhibition of metastasis was linked with the regulation of MMPs,
uPA and/or uPAR activities and/or expression levels (5,6,18,33-35).
In the present study, morin hydrate suppressed cell migration and
invasiveness in TPA-treated MCF-7 human breast cancer cells, and
delayed cell growth. Furthermore, MMP-9 activity was markedly
decreased by morin hydrate. In addition, the expression levels of
metastasis-enhancing factors, such as MMP-7, MMP-9, uPA, and uPAR,
were decreased by morin hydrate, in a concentration-dependent
manner. MMP-9 is a secretory protein activated by plasmin produced
by the uPA/uPAR system, and its secretion is triggered by
fibronectin (21,36). MMP-9 activity is closely associated
with its gene expression (13,35).
In addition, Wang et al (37) reported that enhanced cell invasion
and increased MMP-2 and MMP-9 secretion were shown in
MMP-7-transfected MCF-7 cells, suggesting that MMP-7 participates
in MMP-9 activation. In the present study, morin hydrate only
reduced MMP-9 expression but also prevented MMP-9 activation and
secretion via the inhibition of MMP-7, fibronectin, uPA and uPAR
gene expression. Consequently, the results demonstrated that the
inhibition of TPA-induced metastatic potential by morin hydrate in
MCF-7 human breast cancer cells was linked with the downregulation
of metastasis-enhancing factor expression and the diminishment of
MMP-9 activity.
The expression of metastasis-enhancing factors, such
as MMP-9, uPA and uPAR, is predominantly regulated by Akt and MAPK
and involves the JNK, ERK, and p38 signaling pathways. GSK-3β is
also a key protein involved in the EMT, Wnt signaling and
metastasis in breast cancer (38).
Furthermore, the activity of AP-1, which is an important
transcription factor regulating MMP-9, uPA, and uPAR expression, is
directly regulated by GSK-3β (39). Previous studies showed that
enhanced migration of MCF-7 human breast cancer cells was mediated
by the induction of GSK-3β phosphory-lation (40,41).
These studies implied that the inhibition of metastatic potential
was linked with the suppressive regulation of GSK-3β
phosphorylation. In the current study, morin hydrate suppressed not
only cell migration and invasion, but also the phosphorylation of
GSK-3β induced by TPA in MCF-7 cells, in a concentration-dependent
manner. A previous study revealed that the activation of protein
kinase C (PKC) by TPA lead to the inactivation of GSK-3β, inducing
the activation of AP-1 and promotion of tumorigenesis (42). In the current study, morin hydrate
inhibited the expression of AP-1 subunit c-Fos induced by TPA and
this inhibition was associated with the inhibition of GSK-3β
phosphorylation. Furthermore, nuclear level of c-Fos was also
markedly decreased by morin hydrate, in a concentration-dependent
manner. Therefore, morin hydrate may attenuate the metastatic
potential through the inhibition of metastasis-enhancing factors,
such as MMP-9, uPA, and uPAR, via downregulation of AP-1
transcriptional activity and activation of GSK-3β. Consequently,
these results demonstrate that the suppressive effect of morin
hydrate on the TPA-induced metastatic potential is linked with the
GSK-3β-controlled inhibition of metastasis-enhancing factors.
TPA is a PKC activator that enhances cancer
metastasis on the cellular level. TPA-mediated activations of Akt
and MAPK play important roles in the induction of metastatic
potential (43). Akt is a
well-known regulator of GSK-3β activity (44,45)
and c-Fos expression may also be regulated by Akt (46). The present investigation showed
that TPA-induced Akt phosphorylation was reversed by morin hydrate
along with the suppression of GSK-3β phosphorylation, c-Fos
expression, and nuclear c-Fos level. Furthermore, LY294002 and
wortmannin, which are Akt/PI3K inhibitors, inhibited TPA-induced
GSK-3β phosphorylation and c-Fos expression. By contrast, morin
hydrate did not affect to phosphorylation of ERK and NF-κB induced
by TPA. The transcriptional activity of AP-1 is closely linked with
the nuclear localization of its subunits, c-Jun and c-Fos. These
results demonstrated that the inhibition of TPA-induced metastatic
potential by morin hydrate in MCF-7 cells was regulated via the
Akt/GSK-3β/c-Fos signaling pathway. Therefore, this investigation
demonstrated that Akt acted as a key regulator of morin
hydrate-mediated prevention of metastasis in MCF-7 human breast
cancer cells.
The current study revealed the antimetastatic
potential of morin hydrate in TPA-treated MCF-7 human breast cancer
cells. This effect was shown at high concentrations (150-200
µM) of morin hydrate. At doses of 150 and 200 µM,
morin hydrate inhibited the growth of MCF-7 cells. Gene expression
levels of metastatic factors, such as MMP-7, MMP-9, fibronectin,
uPA and uPAR, were downregulated at lower morin hydrate
concentrations (50-200 µM); however, TPA-induced MMP-9
activity, Akt and GSK-3β phosphorylation and c-Fos expression were
significantly reversed at 150 and 200 µM morin hydrate. It
may be hypothesized that the inhibition of metastatic potential by
morin hydrate in TPA-treated MCF-7 cells was closely linked with
the suppression of cell growth.
Although anticarcinogenic and anticancer activities
of morin hydrate were previously evaluated in animal and cancer
cells and no toxicity of morin hydrate (10 and 50 mg/kg) was
revealed in a mouse model following daily injection for 7 days, the
effects of morin hydrate on metastasis remain to be fully assessed
(30). The present investigation
showed the antimeta-static activity of morin hydrate in TPA-treated
MCF-7 cells and it was revealed that the antimetastatic potential
was induced by the Akt/GSK-3β-mediated inhibition of c-Fos
expression. In conclusion, the present investigation demonstrated
that morin hydrate, at the concentration range of 150-200
µM, is a potent substance that may prevent the metastasis of
breast cancer. However, further research in patient-derived
xenografts or 3D tumor models is required to assess the potential
for clinical use.
Funding
This research was supported by the Basic Science
Research Program through the National Research Foundation of Korea
funded by the Ministry of Education (grant no.
2018R1D1A1B07047758).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Author’s contributions
KSL and KSN designed the experiments. KSL, GSN and
JB performed experiments. KSL, SK and KSN analyzed the data. KSL
wrote the manuscript. KSL, SK and KSN reviewed the manuscript. All
authors confirmed and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
References
|
1
|
Ghoncheh M, Pournamdar Z and Salehiniya H:
Incidence and mortality and epidemiology of breast cancer in the
world. Asian Pac J Cancer Prev. 17:43–46. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Howlader N, Noone AM, Krapcho M, Garshell
J, Miller D, Altekruse SF, Kosary CL, Yu M, Ruhl J, Tatalovich Z,
Mariotto A, et al: SEER Cancer Statistics Review, 1975-2012.
National Cancer Institute; Bethesda, MD: 2015, https://seer.cancer.gov/archive/csr/1975_2012/.
Accessed July 5, 2019.
|
|
3
|
Jung KW, Won YJ, Kong HJ, Oh CM, Cho H,
Lee DH and Lee KH: Cancer statistics in Korea: Incidence,
mortality, survival, and prevalence in 2012. Cancer Res Treat.
47:127–141. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Oskarsson T: Extracellular matrix
components in breast cancer progression and metastasis. Breast.
22(Suppl 2): S66–S72. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Janicke F, Prechtl A, Thomssen C, Harbeck
N, Meisner C, Untch M, Sweep CG, Selbmann HK, Graeff H and Schmitt
M; German N0 Study Group: Randomized adjuvant chemotherapy trial in
high-risk, lymph node-negative breast cancer patients identified by
urokinase-type plasminogen activator and plasminogen activator
inhibitor type 1. J Natl Cancer Inst. 93:913–920. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kwon YS, Lee KS, Chun SY, Jang TJ and Nam
KS: Suppressive effects of a proton beam on tumor growth and lung
metastasis through the inhibition of metastatic gene expression in
4T1 orthotopic breast cancer model. Int J Oncol. 49:336–342. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lee KS, Shin JS and Nam KS: Starfish
polysaccharides downregulate metastatic activity through the MAPK
signaling pathway in MCF-7 human breast cancer cells. Mol Biol Rep.
40:5959–5966. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li X, Kong X, Wang Y and Yang Q: BRCC2
inhibits breast cancer cell growth and metastasis in vitro and in
vivo via down-regulating AKT pathway. Cell Death Dis. 4:e7572013.
View Article : Google Scholar
|
|
9
|
Yang F, Hu M, Lei Q, Xia Y, Zhu Y, Song X,
Li Y, Jie H, Liu C, Xiong Y, et al: Nifuroxazide induces apoptosis
and impairs pulmonary metastasis in breast cancer model. Cell Death
Dis. 6:e17012015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Daniele A, Zito AF, Giannelli G, Divella
R, Asselti M, Mazzocca A, Paradiso A and Quaranta M: Expression of
metal-loproteinases MMP-2 and MMP-9 in sentinel lymph node and
serum of patients with metastatic and non-metastatic breast cancer.
Anticancer Res. 30:3521–3527. 2010.PubMed/NCBI
|
|
11
|
Choi JY, Jang YS, Min SY and Song JY:
Overexpression of MMP-9 and HIF-1α in breast cancer cells under
hypoxic conditions. J Breast Cancer. 14:88–95. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mehner C, Hockla A, Miller E, Ran S,
Radisky DC and Radisky ES: Tumor cell-produced matrix
metalloproteinase 9 (MMP-9) drives malignant progression and
metastasis of basal-like triple negative breast cancer. Oncotarget.
5:2736–2749. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Moirangthem A, Bondhopadhyay B, Mukherjee
M, Bandyopadhyay A, Mukherjee N, Konar K, Bhattacharya S and Basu
A: Simultaneous knockdown of uPA and MMP9 can reduce breast cancer
progression by increasing cell-cell adhesion and modulating EMT
genes. Sci Rep. 6:219032016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Harbeck N, Schmitt M, Kates RE, Kiechle M,
Zemzoum I, Jänicke F and Thomssen C: Clinical utility of
urokinase-type plasminogen activator and plasminogen activator
inhibitor-1 determination in primary breast cancer tissue for
individualized therapy concepts. Clin Breast Cancer. 3:196–200.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Look MP: Pooled analysis of uPA and PAI-1
for prognosis in primary breast cancer patients. EORTC receptor and
biomarker study group. Int J Biol Markers. 15:70–72. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sato H and Seiki M: Regulatory mechanism
of 92 kDa type IV collagenase gene expression which is associated
with invasiveness of tumor cells. Oncogene. 8:395–405.
1993.PubMed/NCBI
|
|
17
|
Takahra T, Smart DE, Oakley F and Mann DA:
Induction of myofibroblast MMP-9 transcription in three-dimensional
collagen I gel cultures: Regulation by NF-kappaB, AP-1 and Sp1. Int
J Biochem Cell Biol. 36:353–363. 2004. View Article : Google Scholar
|
|
18
|
Kim HS, Kim MJ, Kim EJ, Yang Y, Lee MS and
Lim JS: Berberine-induced AMPK activation inhibits the metastatic
potential of melanoma cells via reduction of ERK activity and COX-2
protein expression. Biochem Pharmacol. 83:385–394. 2012. View Article : Google Scholar
|
|
19
|
Kim S, Kim SH, Hur SM, Lee SK, Kim WW, Kim
JS, Kim JH, Choe JH, Nam SJ, Lee JE and Yang JH: Silibinin prevents
TPA-induced MMP-9 expression by down-regulation of COX-2 in human
breast cancer cells. J Ethnopharmacol. 126:252–257. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li CL, Yang D, Cao X, Wang F, Hong DY,
Wang J, Shen XC and Chen Y: Fibronectin induces
epithelial-mesenchymal transition in human breast cancer MCF-7
cells via activation of calpain. Oncol Lett. 13:3889–3895. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Thant AA, Nawa A, Kikkawa F, Ichigotani Y,
Zhang Y, Sein TT, Amin AR and Hamaguchi M: Fibronectin activates
matrix metal-loproteinase-9 secretion via the MEK1-MAPK and the
PI3K-Akt pathways in ovarian cancer cells. Clin Exp Metastasis.
18:423–428. 2000. View Article : Google Scholar
|
|
22
|
Mierke CT, Frey B, Fellner M, Herrmann M
and Fabry B: Integrin α5β1 facilitates cancer cell invasion through
enhanced contractile forces. J Cell Sci. 124:369–383. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nam JM, Onodera Y, Bissell MJ and Park CC:
Breast cancer cells in three-dimensional culture display an
enhanced radio-response after coordinate targeting of integrin
alpha5beta1 and fibronectin. Cancer Res. 70:5238–5248. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wei PL, Kuo LJ, Huang MT, Ting WC, Ho YS,
Wang W, An J and Chang YJ: Nicotine enhances colon cancer cell
migration by induction of fibronectin. Ann Surg Oncol.
18:1782–1790. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kenny HA, Chiang CY, White EA, Schryver
EM, Habis M, Romero IL, Ladanyi A, Penicka CV, George J, Matlin K,
et al: Mesothelial cells promote early ovarian cancer metastasis
through fibronectin secretion. J Clin Invest. 124:4614–4628. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Knowles LM, Gurski LA, Engel C, Gnarra JR,
Maranchie JK and Pilch J: Integrin αvβ3 and fibronectin upregulate
Slug in cancer cells to promote clot invasion and metastasis.
Cancer Res. 73:6175–6184. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang JP and Hielscher A: Fibronectin: How
its aberrant expression in tumors may improve therapeutic
targeting. J Cancer. 8:674–682. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kang DG, Moon MK, Sohn EJ, Lee DH and Lee
HS: Effects of morin hydrate on blood pressure and metabolic
changes in fructose-induced hypertensive rats. Biol Pharm Bull.
27:1779–1783. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jung HJ, Kim SJ, Song YS, Park EH and Lim
CJ: Evaluation of the antiangiogenic, anti-inflammatory, and
antinociceptive activities of morin hydrate. Planta Med.
76:273–275. 2010. View Article : Google Scholar
|
|
30
|
Lee HS, Jung KH, Park IS, Kwon SW, Lee DH
and Hong SS: Protective effect of morin hydrate on
dimethylnitrosa-mine-induced hepatic fibrosis in rats. Dig Dis Sci.
54:782–788. 2009. View Article : Google Scholar
|
|
31
|
Jin H, Lee WS, Eun SY, Jung JH, Park HS,
Kim G, Choi YH, Ryu CH, Jung JM, Hong SC, et al: Morin hydrate, a
flavonoid from Moraceae, suppresses growth and invasion of the
highly metastatic breast cancer cell line MDA-MB-231 partly through
suppression of the Akt pathway. Int J Oncol. 45:1629–1637. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lee YJ, Kim WI, Kim SY, Cho SW, Nam HS,
Lee SH and Cho MK: Flavonoid morin hydrate inhibits proliferation
and induces apoptosis of melanoma cells by regulating reactive
oxygen species, Sp1 and Mcl-1. Arch Pharm Res. 42:531–542. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kim S, Chun SY, Lee DH, Lee KS and Nam KS:
Mineral-enriched deep-sea water inhibits the metastatic potential
of human breast cancer cell lines. Int J Oncol. 43:1691–1700. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Park SY, Kim YH, Kim Y and Lee SJ:
Aromatic-turmerone attenuates invasion and expression of MMP-9 and
COX-2 through inhibition of NF-κB activation in TPA-induced breast
cancer cells. J Cell Biochem. 113:3653–3662. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lee SO, Jeong YJ, Im HG, Kim CH, Chang YC
and Lee IS: Silibinin suppresses PMA-induced MMP-9 expression by
blocking the AP-1 activation via MAPK signaling pathways in MCF-7
human breast carcinoma cells. Biochem Biophys Res Commun.
354:165–171. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li S, Lu J, Chen Y, Xiong N, Li L, Zhang
J, Yang H, Wu C, Zeng H and Liu Y: MCP-1-induced ERK/GSK-3β/Snail
signaling facilitates the epithelial-esenchymal transition and
promotes the migration of MCF-7 human breast carcinoma cells. Cell
Mol Immunol. 14:621–630. 2017. View Article : Google Scholar
|
|
37
|
Wang Y and Zhou BP: Epithelial-mesenchymal
transition-a hallmark of breast cancer metastasis. Cancer Hallm.
1:38–49. 2013. View Article : Google Scholar
|
|
38
|
Jin H, Yu Y, Zhang T, Zhou X, Zhou J, Jia
L, Wu Y, Zhou BP and Feng Y: Snail is critical for tumor growth and
metastasis of ovarian carcinoma. Int J Cancer. 126:2102–2111.
2010.
|
|
39
|
Mishra R: Glycogen synthase kinase 3 beta:
Can it be a target for oral cancer. Mol Cancer. 9:1442010.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Luo J: Glycogen synthase kinase 3 beta
(GSK3 beta) in tumori-genesis and cancer chemotherapy. Cancer Lett.
273:194–200. 2009. View Article : Google Scholar
|
|
41
|
Weng CJ, Chau CF, Hsieh YS, Yang SF and
Yen GC: Lucidenic acid inhibits PMA-induced invasion of human
hepatoma cells through inactivating MAPK/ERK signal transduction
pathway and reducing binding activities of NF-kappa B and AP-1.
Carcinogenesis. 29:147–156. 2008. View Article : Google Scholar
|
|
42
|
Ma C, Wang J, Gao Y, Gao TW, Chen G, Bower
KA, Odetallah M, Ding M, Ke Z and Luo J: The role of glycogen
synthase kinase 3beta in the transformation of epidermal cells.
Cancer Res. 67:7756–7764. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Park SY, Kim YH, Kim Y and Lee SJ:
Frondoside A has an anti-invasive effect by inhibiting TPA-induced
MMP-9 activation via NF-κB and AP-1 signaling in human breast
cancer cells. Int J Oncol. 41:933–940. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Cross DA, Alessi DR, Vandenheede JR,
McDowell HE, Hundal HS and Cohen P: The inhibition of
glycogen-synthase kinase-3 by insulin or insulin-like growth-factor
1 in the rat skeletal-muscle cell-line-L6 is blocked by wortmannin,
but not by rapamycin: Evidence that wortmannin blocks activation of
the mitogen-activated protein-kinase pathway in l6-cells between
Ras and Raf. Biochem J. 303:21–26. 1994. View Article : Google Scholar
|
|
45
|
Cross DA, Alessi DR, Cohen P, Andjelkovich
M and Hemmings BA: Inhibition of glycogen-synthase kinase-3 by
insulin-mediated by protein-kinase B. Nature. 378:785–789. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kim J, Montagne K, Nemoto H, Ushida T and
Furukawa KS: Hypergravity down-regulates c-fos gene expression via
ROCK/Rho-GTP and the PI3K signaling pathway in murine ATDC5
chondroprogenitor cells. PLos One. 12:e01853942017. View Article : Google Scholar : PubMed/NCBI
|