Introduction
Colorectal cancer (CRC) is the third most common
type of cancer, with ~1.4 million cases diagnosed worldwide in 2012
(1). The prognosis of the disease
largely depends on the stage of the tumor at diagnosis (2). The disease is relatively
heterogenous, and is classified into 4 different consensus
molecular subtypes. The main characteristic of CRC is genetic
instability, which can be due to either chromosomal instability
(CIN) (3) or microsatellite
instability (MSI) due to a defective DNA mismatch repair (dMMR)
system (4). Additionally, CpG
island methylation phenotype (CIMP) is a feature that induces
epigenetic instability by silencing through promoter
hypermethylation of a range of tumor suppressor genes, including
mutL homolog 1 (MLH1) (5).
Apart from the surgical removal of the tumor,
usually followed by adjuvant 5-fluoruracil (5-FU)-based
chemotherapy, various immunotherapeutic approaches are currently
being investigated as alternative options for the treatment of the
disease (6,7). Latest encouraging developments in
cancer immunotherapy, which involve priming the host's natural
immune defenses to recognize, target and destroy cancer cells
effectively, have brought some glimpse of hope for combatting CRC
(8-11). To this end, tremendous progress has
been made in the understanding of the immune microenvironment of
CRC (12). The deciphering of the
immunological and molecular landscape of the tumor may help
determine subsets of immunogenic CRC, and determine potential
predictive markers to help select patients for immunotherapeutic
approaches (11). Apart from
malignant cells, a solid CRC tissue contains various other innate
immune cells (granulocytes, mast cells and monocytes/macrophages),
adaptive immune cells (T-cells and B-cells), fibroblasts and
endothelial cells. Working together, these cells contribute to the
inflammatory and/or immunological status of the tumor tissue via
cell-to-cell contact and/or the production of cytokines and
chemokines. Tumor-infiltrating lymphocytes (TILs) are mixtures of
T-cells, B-cells, natural killer (NK) cells, macrophages and other
innate cells in variable proportions, with T-cells being the most
abundant (13). They can
infiltrate the solid tumor, and are used as signals of the immune
system, in its attempt to attack the cancer cells. Primarily, TILs
appear in the human body to indicate the existence of the host,
thus reflecting the dynamic process of cancer immunization
(14,15).
The present study aimed to investigate whether a
high mutation rate is associated with distinct expression profiles
of various immune checkpoint molecules. To this end, the
association between the TIL load and overall survival of patients
with CRC was first examined, determining the expression of various
immune checkpoint molecules, including programmed death-ligands 1
and 2 (PD-L1/2), cytotoxic T-lymphocyte-associated protein 4
(CTLA-4) and indoleamine 2, 3-dioxygenase 1 (IDO1).
In addition, the TIL load and the expression of such immune
checkpoint molecules in each tumor, including the tumor's mutation
rate, were evaluated. Finally, 9 immune-related gene signatures
were compared between CRC and normal tissue. The results provide
evidence that high levels of PD1, PD-L1/L2,
CTLA-4 and IDO-1 are associated with the TIL load, a
high mutation rate and the overall survival of colon cancer
patients.
Materials and methods
Data extraction and analysis
Next generation sequencing (NGS) and
clinicopathological data for 453 colorectal adenocarcinoma patients
were extracted from the Cancer Genome Atlas (TCGA-COAD and
TCGA-READ datasets, containing colon and rectum adenocarcinomas,
respectively) and the data were computationally examined.
The expression of a list of immune checkpoint
molecules and other, prospective checkpoint molecules, including
programmed cell death 1 (PD-1; PDCD1), PD-L1
(CD274), PD-L2 (PDCD1LG2), CTLA-4,
T-cell immunoreceptor with Ig and ITIM domains (TIGIT),
IDO1, IDO2, lymphocyte activating 3 (LAG3),
V-set domain-containing T-cell activation inhibitor 1
(VTCN1), V-domain Ig suppressor of T cell activation
(VISTA), Ig-like transcript (ILT)2,
ILT4 and human leukocyte antigen G (HLA-G) were
analyzed using RNA-seq data of 275 COAD and 92 READ tumors, and
these were compared to the gene expression data of a total of 349
normal colon and 318 normal rectum samples, which were extracted
from the TCGA and GTEx projects. The expression levels of each gene
were calculated in transcripts per million mapped reads (TPM),
adding an offset of 0.1, as previously described (16-18).
One-way analysis of variance (ANOVA) was performed using the
disease state (tumor or normal) as a variable to determine the
statistically significant differentially expressed genes. The
expression data were first log-transformed for differential
analysis and the fold change (log2FC) was defined as the
difference of the median value of the tumor samples from the median
value of the normal samples. Genes with |log2FC>1|
and P<0.01 were considered as differentially expressed. The
corresponding percentage (%) of TIL
('percent_lymphocyte_infiltration') and tumor-associated
neutrophilic (TAN) load ('percent_neutrophil_infiltration') of the
patients with CRC were extracted from TCGA using the Genomic Data
Commons (GDC) Data Portal (https://portal.gdc.cancer.gov/), as previously
described (17) (Table SI).
Patient survival analysis
Survival analysis was based on the expression status
of adenosine A2A receptor (ADORA2A), CD8,
CTLA-4, hepatitis A virus cellular receptor 2
(HAVCR2), IDO1, IDO2, LAG3, PD1,
PD-L1, PD-L2, TIGIT, VISTA and
VTCN1 or the multi-gene signatures. The overall patient
survival was plotted on Kaplan-Meier curves using the Gene
Expression Profiling Interactive Analysis (GEPIA2) web server
(19). Differences in overall
survival between high- and low gene-expressing patients were scored
using the log-rank test. Spearman's correlation analysis was used
to examine the correlation between the TIL load with the expression
of each immune checkpoint molecule.
Immune-related gene signatures in
CRC
The following immune-related gene signatures from
GEPIA2 (19) were compared between
the CRC tumor and normal samples, within each TCGA dataset: Naive
T-cell [C-C motif chemokine receptor (CCR)7, lymphoid
enhancer-binding factor 1 (LEF1), transcription factor 7
(TCF7) and L-selectin (SELL)]; effector T-cell [CX3C
chemokine receptor 1 (CX3CR1), fibroblast growth factor
binding protein 2 (FGFBP2) and Fc fragment of IgG receptor
IIIa (FCGR3A)]; effector memory T-cell [PDCD1, dual
specificity protein phosphatase 4 (DUSP4), granzyme
(GZM)K, GZMA and interferon gamma
(IFNG)]; central memory T-cell [CCR7, SELL and
interleukin (IL)7R]; resident memory T-cell
[CD69, integrin, alpha E (ITGAE), C-X-C chemokine
receptor type 6 (CXCR6) and myeloid-associated
differentiation marker (MYADM)]; exhausted T-cell
(HAVCR2, TIGIT, LAG3, PDCD1,
CXCL13 and LAYN); resting Treg T-cell [forkhead box
P3 (FOXP3), IL2RA); effector Treg T-cell
[FOXP3, CTLA-4, CCR8 and tumor necrosis factor
(TFN) receptor superfamily member 9 (TNFRSF9)]; and Th1-like
[CXCL13, HAVCR2, IFNG, CXCR3, basic
helix-loop-helix family member e40 (BHLHE40) and
CD4]. For the analysis of gene signatures, the mean value of
the log2(TPM+1) was used as the signature score and the
CRC samples were compared against matched normal data from both the
TCGA. The gene signatures with |log2FC>1| and
P<0.01 (ANOVA) were considered as significantly different
between tumor and normal tissues.
Association between the mutation rate and
the expression of immune checkpoint molecules in COAD
iCoMut Beta for FireBrowse was used to categorize
COAD tumors into those having a low (<34 mutations per Mb) or
high (>34 mutations per Mb) mutation rate. The expression of 5
widely-established immune checkpoint molecules (PD-1,
PD-L1, PD-L2, CTLA-4 and IDO1) was then
compared between COAD with a 'high' and 'low' mutation rate. The
data were analyzed using the R environment.
Cell-type fractions within microsatellite
stable (MSS) and instable (MSI) colon adenocarcinomas
The Cancer Immunome Database (TCIA) (20) was used to gain insight into the
cell type fractions within MSS, MSI-low or MSI-high patients within
the TCGA-COAD database. The MSI status was defined according to the
Cancer Genome Atlas Network (21).
Detection of single nucleotide variants
(SNVs) in immune checkpoint genes and association with their
expression
The gene expression and mutations across the
GDC-TCGA-COAD and READ datasets were explored through the UCSC Xena
platform (22). Gene expression
(RNAseq) was evaluated using the normalized HTSeq-Fragments Per
Kilobase of transcript per million mapped reads (FPKM). Somatic
SNVs and insertions/deletions (indels) (deleterious, splice,
missense/inframe, silent and complex or unannotated mutations)
across immune checkpoint genes were called using MuTect2 (v.4.1)
variant aggregation and masking. The GRCh37 build of the human
reference genome was used for analysis.
Results
Elevated levels of CTLA-4, HAVCR2,
IDO1, PD-1, PD-L1, PD-L2, TIGIT,
VTCN1 and HLA-G were detected in COAD and READ tumors
against their corresponding normal tissues. However, the difference
did not reach statistical significance. Among these checkpoint
molecules, IDO1 was exceptionally upregulated both in COAD
and READ. On the other hand, ILT2, ADORA2A,
LAG3 and VISTA exhibited a lower expression in CRC
compared to normal tissue. In the case of LAG3 and
VISTA, the difference reached statistical significance
(P<0.01) (Fig. 1).
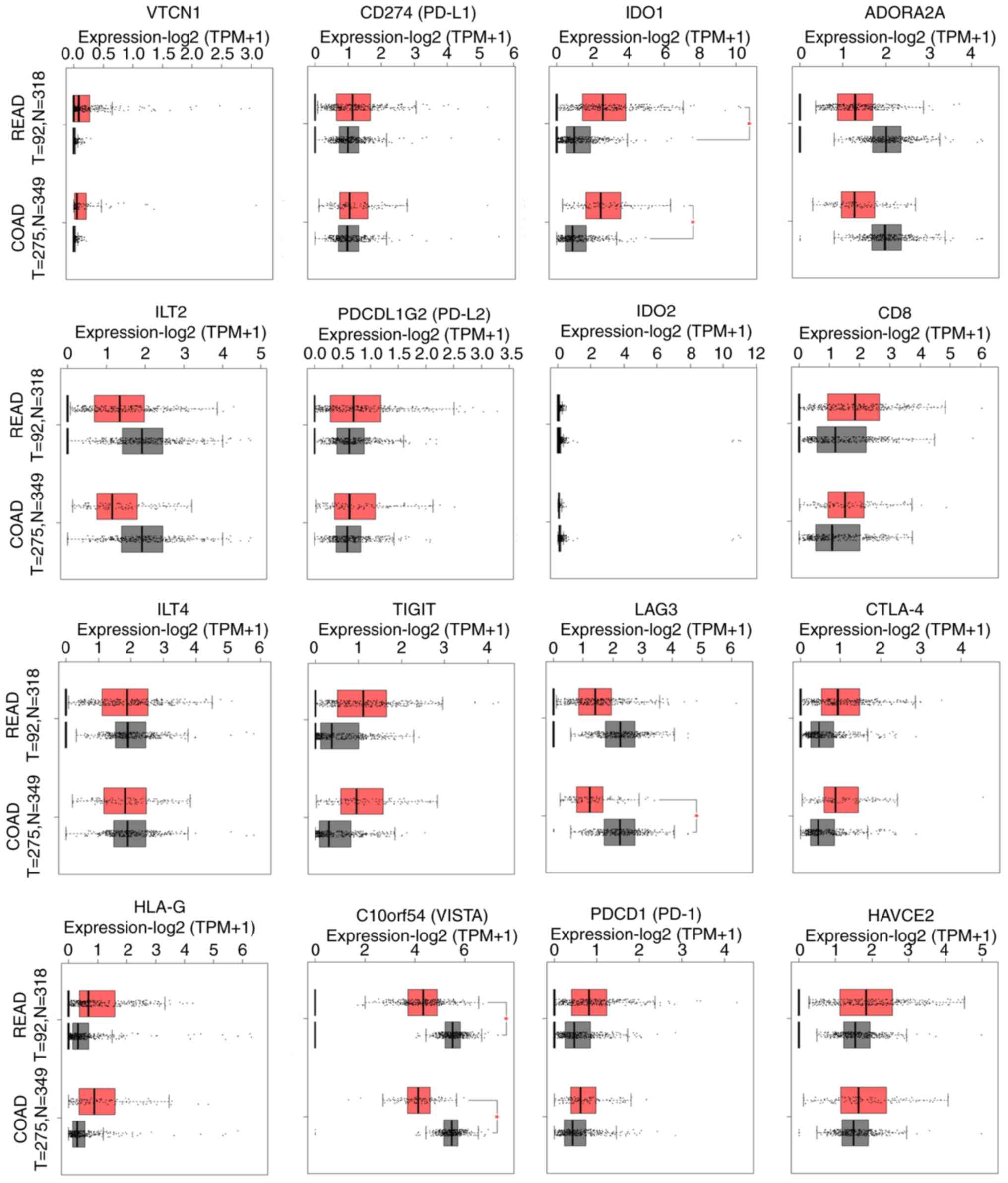 | Figure 1Among the immune checkpoints
analyzed, IDO1 was significantly upregulated in CRC,
whereas, LAG3 and VISTA were significantly
downregulated. The higher levels of CTLA-4, HAVCR2,
IDO1, PD-1, PD-L1, PD-L2, TIGIT,
VTCN1 and HLA-G in CRC did not reach statistical
significance. Equally, the lower levels of ILT2 and
ADORA2A in CRC did not reach statistical significance. Red
stars denote statistically significant differences (P<0.01)
between COAD (or READ tumors) and the normal tissue from the TCGA
and GTEx projects. CRC, colorectal cancer; COAD, colon
adenocarcinomas; READ, rectal adenocarcinomas. |
Patients with CRC expressing high levels of
CD8A, CTLA-4, IDO1, PD-1 and
TIGIT, exhibited a better overall survival compared to
patients expressing with low levels of these molecules (Fig. 2). This overexpression correlated
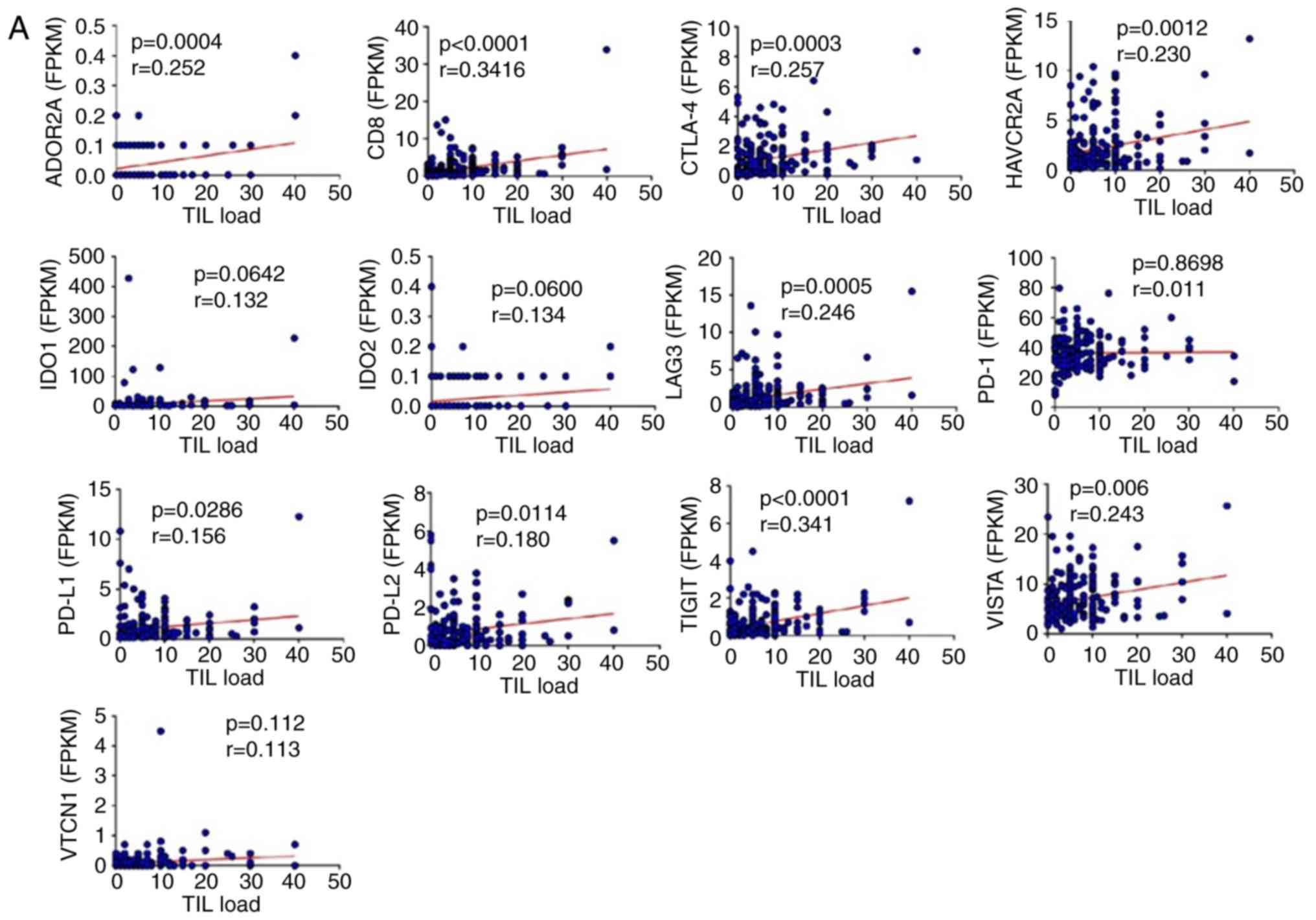
with the TIL load in both datasets. Specifically, among the COAD
tumors, the TIL load positively correlated with the expression of
CD8, as well as that of ADORA2A, CTLA-4,
HAVCR2, LAG3, PD-L1, PD-L2,
TIGIT and VISTA (P<0.005, Spearman's correlation
analysis) (Fig. 3A). On the other
hand, among the READ tumors, such positive correlations between the
TIL load and the expression of immune checkpoint molecules (or the
TCR co-receptor marker CD8A, which acts on the recognition
of antigens displayed by an antigen presenting cell in the context
of MHC-I molecules), were scored only for LAG3 and
PD-L2 (Fig. 3B). Of note,
the expression of CTLA-4 significantly correlated with that
of the remaining immune checkpoint molecules in COAD, indicating
that immune response in colon tumors elicits multiple host and
tumor mechanisms of immune suppression in the tumor
microenvironment, other than the PD1/PD-L1 axis. Therefore, this
observation supports the hypothesis that a combinatorial targeting
of multiple immune checkpoint pathways may expand the clinical
benefit for these patients (17)
(Fig. 3C).
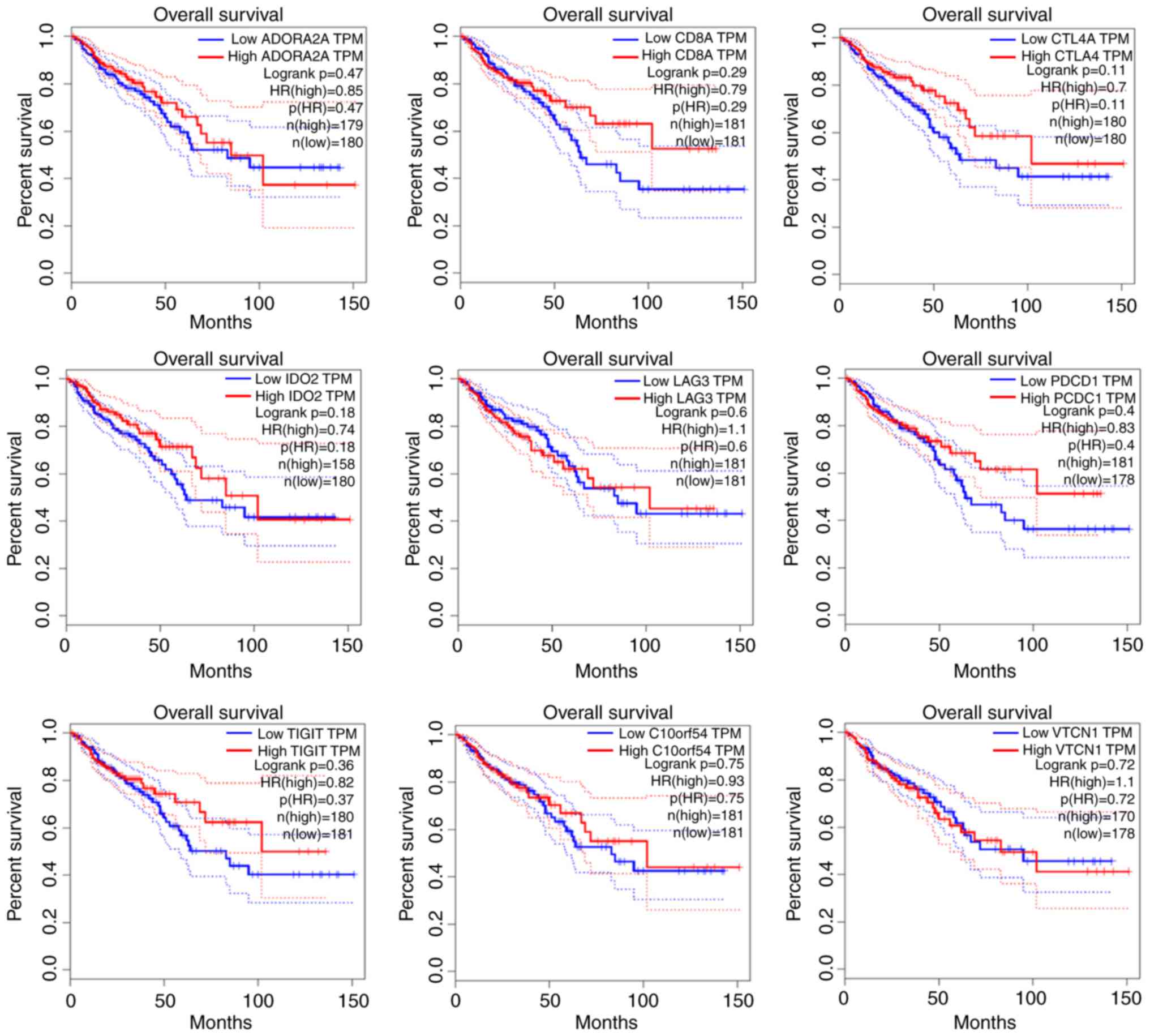 | Figure 2Overall survival curves of patients
with CRC, expressing high or low levels of the immune checkpoint
molecules ADORA2A, CTLA-4, HAVCR2,
IDO1/2, LAG3, PD-1 (PDCD1),
PD-L1 (CD274), PD-L2 (PDCD1LG2),
TIGIT, VISTA (C10orf54), HLA-G,
ILT2 and VTCN1, or the TCR co-receptor marker
CD8. The patients' high CD8, CTLA-4,
IDO1, PD1 and TIGIT expression levels,
exhibited a tendency for improved overall survival, compared to
those with low levels of the corresponding genes (log rank,
P>0.05). CRC, colorectal cancer. |
Although the outcome of patients with CRC has
improved significantly with the recent implementation of annual
screening programs, reliable prognostic biomarkers are still
required due to the heterogeneity of the disease. Cumulative
evidence indicates an association between immune signature and
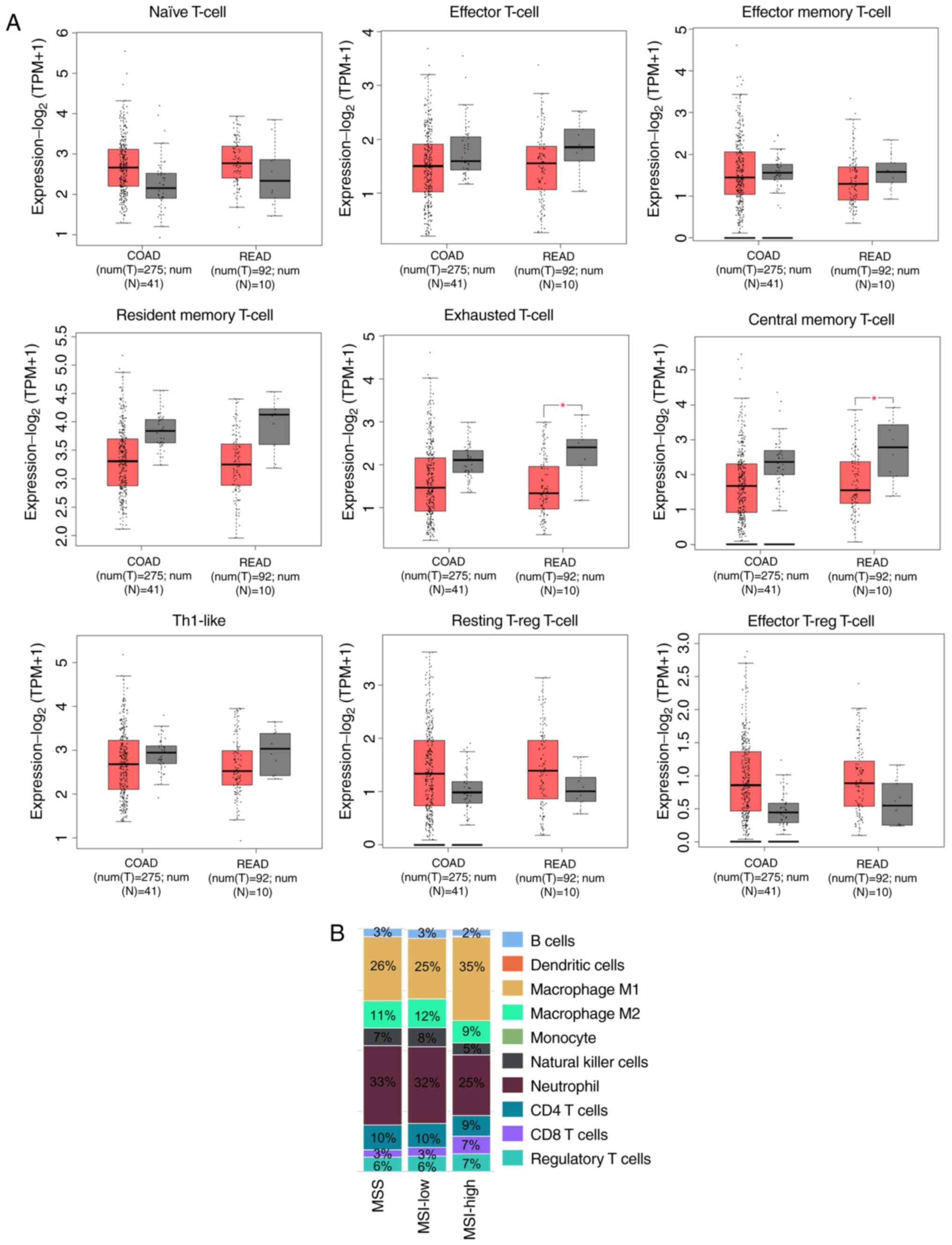
prognosis of the disease. Therefore, the present study explored 9
immune-related gene signatures in CRC and compared them to normal
tissue from the TCGA and GTEx projects. A significantly lower
expression of the 'central memory T-cell' and 'exhausted
T-cell'- related gene signatures, was found in READ tumors. The
'resting and effector Treg T-cell', 'naïve T-cell'
and 'Th1-like' gene signatures were enriched among both CRC
subtypes (COAD and READ), although without reaching statistical
significance. On the other hand, the 'resident memory
T-cell' signature revealed lower levels in the CRC samples
compared to the normal tissue. Overall, these findings reveal
significant differences in the immune-related gene signatures
between colorectal tumors and normal tissue, reflecting their
association with the prognosis of the disease (Fig. 4A).
The second subtype of CRC (CMS2) contains
hypermutated, microsatellite instable (MSI+) tumors, with a strong
immune activation. MSI occurs due to a defective DNA mismatch
repair (dMMR), which accumulates a high number of mutations
(23). A higher mutational load
(and hence a higher neoepitope load) is positively associated with
overall TIL infiltration, memory T cells, and CRC-specific survival
(24). Herein, differences were
found in the percentage (%) of different cell types between tumors,
based on their MSI status. Specifically, MSI-high tumors contained
a higher percentage of M1 macrophages (35%) and CD8+ T
cells (7%) compared to MSS (26% M1 macrophages and 3%
CD8+ T cells) or MSI-low CRCs (25% M1 macrophages and 3%
CD8+ T cells). On the other hand, MSI-high CRCs
contained less neutrophils (25%) compared to MSS (33%) or MSI-low
(32%) tumors (Fig. 4B).
In addition, the CRC tumors were stratified based on
their mutation rate and the association of the expression of immune
checkpoint molecules with the corresponding TIL load (%) was
investigated in each tumor. Tumors with a high mutational rate
(>34 mutations/Mb) exhibited the same mutational signature
profile, i.e., a preference for *CpG>T mutations, with those
having a low mutational rate (<34 mutations/Mb) (Fig. 5A). Overall, 25 genes were
recurrently mutated in CRC, exhibiting elevated mutation rates
among hypermutated tumors. The significantly mutated genes in the
hypermutated cancers included APC, TP53,
PIK3CA, PTEN, KRAS, ATM, SYNE1,
SMAD4, FBXW7, KIT, BRAF, PTCH1,
CSMD3, NF1, RB1, among others at a lower
frequency (<100 mutations) (Fig.
5A). A significantly higher PD-1, PD-L1,
PD-L2, CTLA-4 and IDO1 expression was found
among the hypermutated colon adenocarcinomas, compared to those
with a lower mutation rate (Fig.
5B). These data suggest that a high (synonymous and
non-synonymous) tumor mutation rate seems to be associated with
clinical benefit in patients who receive anti-PD1, anti-PD-L1 or
anti-CTLA-4 therapy. Of major interest, it was found that the TIL
load (%) was also significantly higher (P=0.021) among the
hypermutated tumors, suggesting that part of these mutations,
belonging to cancer neoepitopes, might be recognized by TILs that
are in immediate contact with the tumor cells (Fig. 5B).
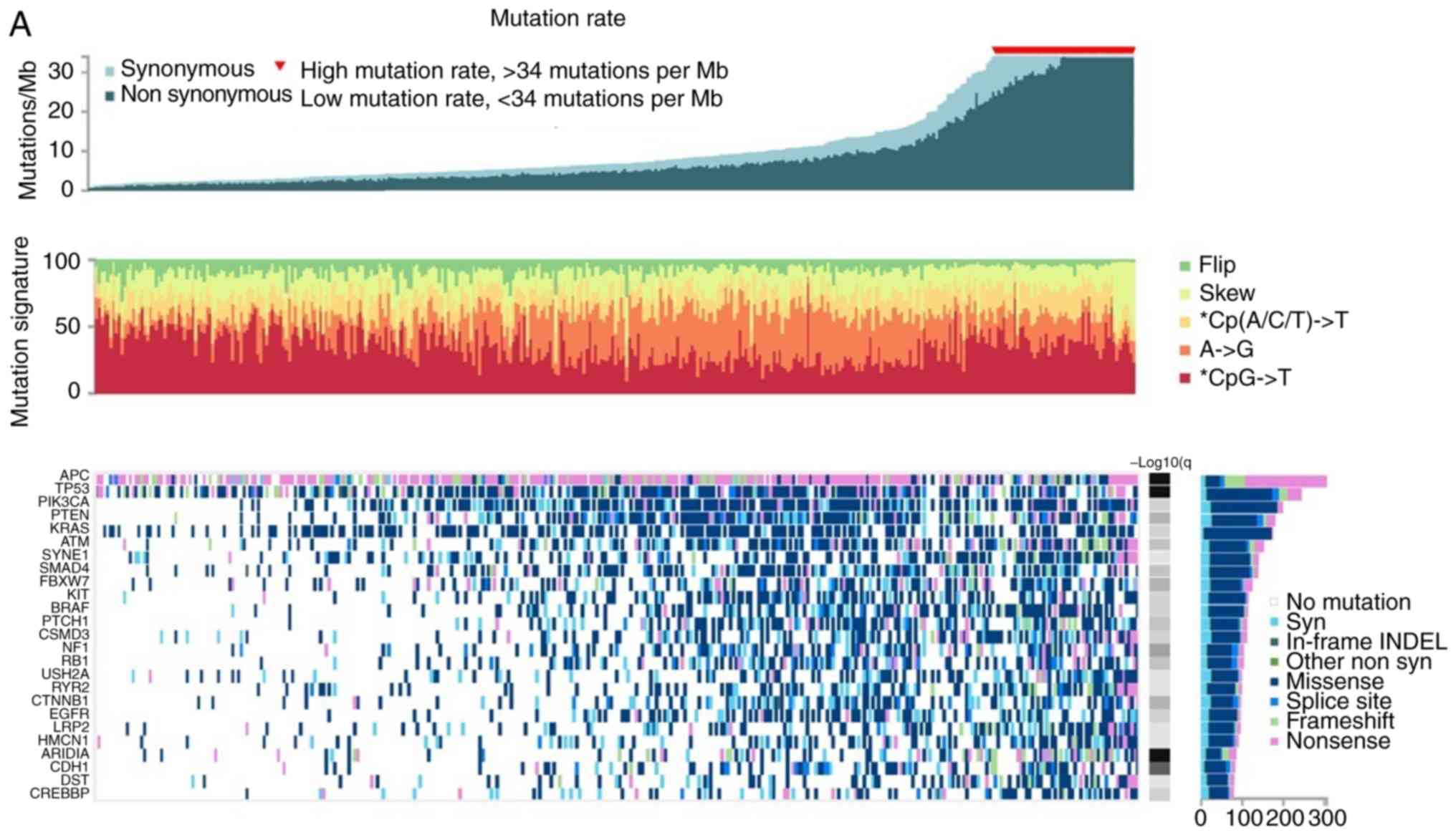 | Figure 5(A) COAD tumors were stratified to
those having a high (>34 mutations/Mb) or a low (<34
mutations/Mb) mutation rate. Mutational signatures did not differ
between hyper-mutated and non-hypermutated CRC tumors, both having
a preference for *CpG>T mutations. The long tail graph shows the
25 significantly mutated genes in hypermutated and non-hypermutated
tumors. The significantly mutated genes among hypermutated tumors
included APC, TP53, PIK3CA, PTEN,
KRAS, ATM, SYNE1, SMAD4, FBXW7,
KIT, BRAF, PTCH1, CSMD3, NF1,
RB1, among others. Both mutational signatures and
significantly mutated genes were assessed using iCoMut Beta for
FireBrowse. (B) The expression levels of PD-1, PD-L1,
PD-L2, CTLA-4 and IDO1 were significantly
higher among colon adenocarcinomas with a high mutation rate per
Mb. The TIL (%) load was significantly higher among tumors with a
high mutation rate (P=0.021). COAD, colon adenocarcinomas; READ,
rectal adenocarcinomas. |
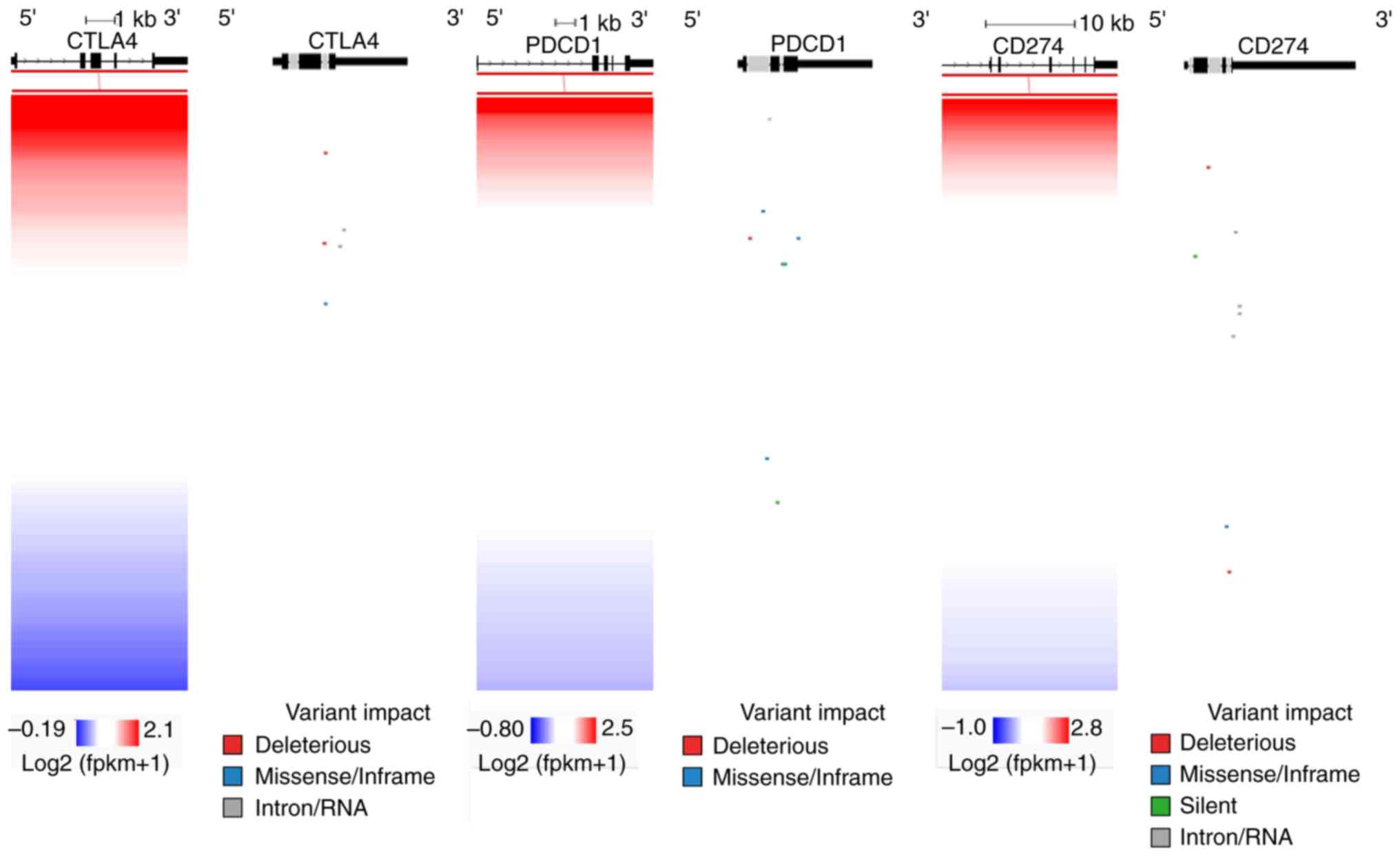
In addition, the existence of somatic mutations in
PD-1 (PDCD1), PD-L1 (CD274),
CTLA-4 and other checkpoint genes was investigated. A total
of 100 SNVs and Indels were detected, containing missense/inframe,
deleterious, silent, or intron/RNA somatic point mutations within
the 12 immune checkpoint molecules of interest, across the COAD and
READ tumors (Table SII). All
variants were randomly distributed and did not seem to associate
with the corresponding expression levels of each gene (Figs. 6 and S1). Therefore, these results indicate
that gene expression is not driven by mutations in these checkpoint
genes.
Discussion
Immunoediting has turned out to be critical in
appreciating the immune system's ability to harness tumor growth
and spread in several types of cancer (25,26).
New immune-based therapies have been recently proposed as treatment
against primary and metastatic CRC, using either PD-1, PD-L1 and
CTLA-4 inhibitors, or a combination of them in refractory (MSI-H
and MSS) colorectal tumors (13,27).
In addition, adoptive cell therapy, using TILs from patients or
donors, or differentiated from stem cells, is a highly promising
immunotherapeutic strategy for CRC patients (28). These immune cells are then
activated and expanded in vitro, and subjected to gene
modification, before finally being infused back into the patients
(28). Recently, Baek and Kim
(29) obtained TILs from patients
with CRC and evaluated their potential as an immunotherapeutic
modality. They demonstrated that the ex vivo expanded TILs
contained mostly effector memory T-cells and they were found to
elicit an anti-tumor response. However, within the tumor
microenvironment, the expression of co-inhibitory immune
checkpoints can lead to the inactivation of such TILs (30).
In the present study, the expression of several
immune checkpoints between CRC and normal tissue was compared,
using data extracted from the TCGA and GTEx platforms. In addition,
their association with patient survival, TIL load and the mutation
rate of each tumor and was evaluated. Furthermore, the expression
of different immune-related gene signatures in CRC compared to the
normal tissue was investigated. A higher percentage of the
so-called 'tumor preventing' M1 macrophages and CD8+
T-cells was found among MSI-high tumors, compared to the MSI-low or
MSS ones. In addition, the percentage of the 'tumor promoting' M2
macrophages, as well as that of neutrophils was lower in MSI-high
tumors compared to the other two microsatellite groups, in
accordance with reports that have previously associated these with
an improved survival of patients with MSI-high CRC (31-33).
A lower percentage of neutrophils was also found among MSI-high
tumors. This is in agreement with a previous report by the authors
demonstrating that patients with CRC with a low TAN percentage have
an improved survival compared to those with a higher TAN load
(17).
By stratifying patients with colon cancer based on
their mutation rate (mutations per Mb), it was found that those
having a high mutation rate expressed significantly higher levels
of PD-1, PD-L1/L2, IDO1 and CTLA-4.
These observations are in agreement with those of previous reports
(34-39), indicating that these patients may
benefit more from a corresponding immune checkpoint blockade
therapy. Therefore, the quantification of the mutational burden in
these patients may be used as a predictive biomarker of
immunotherapy via checkpoint inhibition. The expression of PD-L1
and TMB was recently found to have non-overlapping effects on the
response rate to PD-1/PD-L1 inhibitors and was proposed that it can
be used to categorize the immunologic subtypes of different tumor
types, including CRC (37). In
addition, the authors of the present study previously demonstrated
that the protein levels of PD-1, PD-L1, PD-L2 and CTLA-4, similar
to the CD8 marker, were significantly higher in dMMR/MSI-H CRCs,
compared to dMMR/MSI-L and pMMR-MSS tumors. These observations
indicate the influence that these immune checkpoint-expressing
cells have on the tumor microenvironment by regulating immune
responses (17).
The data of the present study revealed an enrichment
of IDO1 in CRC, highlighting its prominent role in the tumor
microenvironment. Along with IDO1, CRC tumors expressed high
levels of further immune checkpoint molecules, including
CTLA-4 and PD-1. On the other hand, a low expression
of ADORA2A, LAG3 and VISTA was found in CRC.
This may be in contrast to the recent study by Xie et al
(40), who found that VISTA
protein was highly expressed in CRC; but this was mainly due to
TILs. Therefore, it seems that VISTA (C10orf54) is
indeed, downregulated in CRC, compared to CTLA-4 and
PD-1. In accordance with the data of the present study, Lee
et al (41) found a low
percentage (23.6%) of CRCs expressing LAG3. The blockade of LAG3
was also found to enhance tumor-infiltrating T-cell responses of
mismatch repair-proficient (pMMR) liver metastasis of CRC, and was
suggested as a new promising immunotherapeutic target for these
tumors (42).
An increased mutational load in CRC was previously
associated with other metrics, including high cytolytic activity,
the count of MHC-I cancer neoepitopes, high micro-satellite
instability and deregulated expression of several immune
checkpoints (17). The tumor's
mutational burden was recently suggested to be predictive of the
patients' response to immune checkpoint inhibition in MSI-high
metastatic CRC (43). Herein,
higher levels of PD-1, PD-L1, PD-L2,
CTLA-4 and IDO1 were also found among hypermutated
colorectal tumors, indicating an association with the clinical
benefit in patients who receive anti-PD1, anti-PD-L1 or anti-CTLA-4
therapy. Importantly, it was found that the TIL load was
significantly higher among tumors with a high mutation rate.
Overall, the lymphocytic score was previously associated with the
better survival of patients with CRC (44). In a similar study, Giannakis et
al found an association between a higher neoantigen load and
increased overall lymphocytic score in CRC (24). These observations date back even
earlier, when Jass et al demonstrated that a high TIL load
was an independent factor for the survival of patients with rectal
cancer (45), and later on, Ogino
et al demonstrated that higher levels of lymphocytic
reactions and TILs were associated with patient prognosis (44). All these observations confirm that
the presence of a high level of lymphoid reaction in the CRC tissue
is associated with an improved prognosis.
The molecular landscape of CRC was previously
characterized by the Cancer Genome Atlas Network (21), predicting the significantly mutated
genes in CRC. In addition to their role in affecting normal cell
function, tumor somatic mutations can generate neoantigens, which
can be recognized by the host immune system (46). It was found that a high mutation
rate was significantly associated with a high TIL load in these CRC
tumors. This result is consistent with previous reports, and shows
that patients with a big number of immunogenic mutations have an
increased survival (47).
Moreover, the corresponding tumors had higher cytotoxic T-cell
(CTL) content, inferred from the expression of CD8A.
Overall, the findings of the present study
highlight the association of immune checkpoints with the TIL load,
patient survival and high mutation rate in CRC. The data
corroborate that patients with colon cancer with a higher
PD1, PD-L1/2, CTLA-4 and IDO-1
expression, and a high mutation rate, are the ones who will benefit
more from the respective immune checkpoint inhibition
therapies.
Supplementary Data
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study
are included in this published article [and its supplementary
information files].
Authors' contributions
MK acquired and analyzed the data. GDA was involved
in the conception and design of the study and critically reviewed
the manuscript. AZ developed the methodology, and analyzed and
interpreted the data; AZ also wrote the manuscript and supervised
the study. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
The authors would like to acknowledge the platforms
TCGA, HPA, Broad Institute GDAC and The Cancer Immunome Atlas for
downloading genetic and clinical data of the CRC patients.
References
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and Major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar
|
|
2
|
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ,
Meester RGS, Barzi A and Jemal A: Colorectal cancer statistics,
2017. CA Cancer J Clin. 67:177–193. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Poulogiannis G, Ichimura K, Hamoudi RA,
Luo F, Leung SY, Yuen ST, Harrison DJ, Wyllie AH and Arends MJ:
Prognostic relevance of DNA copy number changes in colorectal
cancer. J Pathol. 220:338–347. 2010. View Article : Google Scholar
|
|
4
|
Arends MJ: Pathways of colorectal
carcinogenesis. Appl Immunohistochem Mol Morphol. 21:97–102.
2013.PubMed/NCBI
|
|
5
|
Ibrahim AE, Arends MJ, Silva AL, Wyllie
AH, Greger L, Ito Y, Vowler SL, Huang TH, Tavaré S, Murrell A and
Brenton JD: Sequential DNA methylation changes are associated with
DNMT3B overexpression in colorectal neoplastic progression. Gut.
60:499–508. 2011. View Article : Google Scholar
|
|
6
|
Falzone L, Salomone S and Libra M:
Evolution of cancer pharmacological treatments at the turn of the
third millennium. Front Pharmacol. 9:13002018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ganesh K, Stadler ZK, Cercek A, Mendelsohn
RB, Shia J, Segal NH and Diaz LA Jr: Immunotherapy in colorectal
cancer: Rationale, challenges and potential. Nat Rev Gastroenterol
Hepatol. 16:361–375. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kalyan A, Kircher S, Shah H, Mulcahy M and
Benson A: Updates on immunotherapy for colorectal cancer. J
Gastrointest Oncol. 9:160–169. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Copur MS: Immunotherapy in colorectal
cancer. Oncology (Williston Park). 33:6865062019.
|
|
10
|
Abakushina EV, Gelm YV, Pasova IA and
Bazhin AV: Immunotherapeutic approaches for the treatment of
colorectal cancer. Biochemistry (Mosc). 84:720–728. 2019.
View Article : Google Scholar
|
|
11
|
Dawood S: The evolving role of immune
oncology in colorectal cancer. Chin Clin Oncol. 7:172018.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Christofi T, Baritaki S, Falzone L, Libra
M and Zaravinos A: Current perspectives in cancer immunotherapy.
Cancers (Basel). 11:E14722019. View Article : Google Scholar
|
|
13
|
Koi M and Carethers JM: The colorectal
cancer immune micro-environment and approach to immunotherapies.
Futur Oncol. 13:1633–1647. 2017. View Article : Google Scholar
|
|
14
|
Chen DS and Mellman I: Elements of cancer
immunity and the cancer-immune set point. Nature. 541:321–330.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen DS and Mellman I: Oncology meets
immunology: The cancer-immunity cycle. Immunity. 39:1–10. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ayiomamitis GD, Notas G, Vasilakaki T,
Tsavari A, Vederaki S, Theodosopoulos T, Kouroumalis E and
Zaravinos A: Understanding the interplay between COX-2 and hTERT in
colorectal cancer using a multi-omics analysis. Cancers (Basel).
11:E15362019. View Article : Google Scholar
|
|
17
|
Zaravinos A, Roufas C, Nagara M, de Lucas
Moreno B, Oblovatskaya M, Efstathiades C, Dimopoulos C and
Ayiomamitis GD: Cytolytic activity correlates with the mutational
burden and deregulated expression of immune checkpoints in
colorectal cancer. J Exp Clin Cancer Res. 38:3642019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Roufas C, Chasiotis D, Makris A,
Efstathiades C, Dimopoulos C and Zaravinos A: The expression and
prognostic impact of immune cytolytic activity-related markers in
human malignancies: A comprehensive meta-analysis. Front Oncol.
8:272018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tang Z, Kang B, Li C, Chen T and Zhang Z:
GEPIA2: An enhanced web server for large-scale expression profiling
and interactive analysis. Nucleic Acids Res. 47:W556–W560. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Charoentong P, Finotello F, Angelova M,
Mayer C, Efremova M, Rieder D, Hackl H and Trajanoski Z: Pan-cancer
immunogenomic analyses reveal genotype-immunophenotype
relationships and predictors of response to checkpoint blockade.
Cell Rep. 18:248–262. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cancer Genome Atlas Network: Comprehensive
molecular characterization of human colon and rectal cancer.
Nature. 487:330–337. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Goldman M, Craft B, Hastie M, Repečka K,
Kamath A, McDade F, Rogers D, Brooks AN, Zhu J and Haussler D: The
UCSC Xena platform for public and private cancer genomics data
visualization and interpretation. bioRxiv. 3264702019.
|
|
23
|
Guinney J, Dienstmann R, Wang X, de
Reyniès A, Schlicker A, Soneson C, Marisa L, Roepman P, Nyamundanda
G, Angelino P, et al: The consensus molecular subtypes of
colorectal cancer. Nat Med. 21:1350–1356. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Giannakis M, Mu XJ, Shukla SA, Qian ZR,
Cohen O, Nishihara R, Bahl S, Cao Y, Amin-Mansour A, Yamauchi M, et
al: Genomic correlates of immune-cell infiltrates in colorectal
carcinoma. Cell Rep. 15:857–865. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Swann JB and Smyth MJ: Immune surveillance
of tumors. J Clin Invest. 117:1137–1146. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ribatti D: The concept of immune
surveillance against tumors. The first theories. Oncotarget.
8:7175–7180. 2017. View Article : Google Scholar
|
|
27
|
Boland PM and Ma WW: Immunotherapy for
colorectal cancer. Cancers (Basel). 9:E502017. View Article : Google Scholar
|
|
28
|
Fan J, Shang D, Han B, Song J, Chen H and
Yang JM: Adoptive cell transfer: Is it a promising immunotherapy
for colorectal cancer? Theranostics. 8:5784–5800. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Baek JH and Kim KJ: Expansion of
tumor-infiltrating lymphocytes and their potential for application
to adoptive cell therapy in patients with colorectal cancer. Eur J
Surg Oncol. 45:e58–e59. 2019. View Article : Google Scholar
|
|
30
|
Kodumudi KN, Siegel J, Weber AM, Scott E,
Sarnaik AA and Pilon-Thomas S: Immune checkpoint blockade to
improve tumor infiltrating lymphocytes for adoptive cell therapy.
PLoS One. 11:e01530532016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Edin S, Wikberg ML, Dahlin AM, Rutegård J,
Öberg Å, Oldenborg PA and Palmqvist R: The distribution of
macrophages with a M1 or M2 phenotype in relation to prognosis and
the molecular characteristics of colorectal cancer. PLoS One.
7:e470452012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Narayanan S, Kawaguchi T, Peng X, Qi Q,
Liu S, Yan L and Takabe K: Tumor infiltrating lymphocytes and
macrophages improve survival in microsatellite unstable colorectal
cancer. Sci Rep. 9:134552019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhao Y, Ge X, Xu X, Yu S, Wang J and Sun
L: Prognostic value and clinicopathological roles of phenotypes of
tumour-associated macrophages in colorectal cancer. J Cancer Res
Clin Oncol. 145:3005–3019. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rosenbaum MW, Bledsoe JR, Morales-Oyarvide
V, Huynh TG and Mino-Kenudson M: PD-L1 expression in colorectal
cancer is associated with microsatellite instability, BRAF
mutation, medullary morphology and cytotoxic tumor-infiltrating
lymphocytes. Mod Pathol. 29:1104–1112. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Fabrizio DA, George TJ Jr, Dunne RF,
Frampton G, Sun J, Gowen K, Kennedy M, Greenbowe J, Schrock AB,
Hezel AF, et al: Beyond microsatellite testing: Assessment of tumor
mutational burden identifies subsets of colorectal cancer who may
respond to immune checkpoint inhibition. J Gastrointest Oncol.
9:610–617. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chan TA, Yarchoan M, Jaffee E, Swanton C,
Quezada SA, Stenzinger A and Peters S: Development of tumor
mutation burden as an immunotherapy biomarker: Utility for the
oncology clinic. Ann Oncol. 30:44–56. 2019. View Article : Google Scholar :
|
|
37
|
Yarchoan M, Albacker LA, Hopkins AC,
Montesion M, Murugesan K, Vithayathil TT, Zaidi N, Azad NS, Laheru
DA, Frampton GM and Jaffee EM: PD-L1 expression and tumor
mutational burden are independent biomarkers in most cancers. JCI
Insight. 4:1269082019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lal N, Beggs AD, Willcox BE and Middleton
GW: An immunogenomic stratification of colorectal cancer:
Implications for development of targeted immunotherapy.
Oncoimmunology. 4:e9760522015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hamada T, Soong TR, Masugi Y, Kosumi K,
Nowak JA, da Silva A, Mu XJ, Twombly TS, Koh H, Yang J, et al: TIME
(Tumor Immunity in the MicroEnvironment) classification based on
tumor CD274 (PD-L1) expression status and tumor-infiltrating
lymphocytes in colorectal carcinomas. Oncoimmunology.
7:e14429992018. View Article : Google Scholar :
|
|
40
|
Xie S, Huang J, Qiao Q, Zang W, Hong S,
Tan H, Dong C, Yang Z and Ni L: Expression of the inhibitory B7
family molecule VISTA in human colorectal carcinoma tumors. Cancer
Immunol Immunother. 67:1685–1694. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Lee SJ, Byeon SJ, Lee J, Park SH, Park JO,
Park YS, Kang WK, Lim HY, Kim KM and Kim ST: LAG3 in solid tumors
as a potential novel immunotherapy target. J Immunother.
42:279–283. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhou G, Noordam L, Sprengers D, Doukas M,
Boor PPC, van Beek AA, Erkens R, Mancham S, Grünhagen D, Menon AG,
et al: Blockade of LAG3 enhances responses of tumor-infiltrating T
cells in mismatch repair-proficient liver metastases of colorectal
cancer. Oncoimmunology. 7:e14483322018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Schrock AB, Ouyang C, Sandhu J, Sokol E,
Jin D, Ross JS, Miller VA, Lim D, Amanam I, Chao J, et al: Tumor
mutational burden is predictive of response to immune checkpoint
inhibitors in MSI-high metastatic colorectal cancer. Ann Oncol.
30:1096–1103. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ogino S, Nosho K, Irahara N, Meyerhardt
JA, Baba Y, Shima K, Glickman JN, Ferrone CR, Mino-Kenudson M,
Tanaka N, et al: Lymphocytic reaction to colorectal cancer is
associated with longer survival, independent of lymph node count,
microsatellite instability, and CpG island methylator phenotype.
Clin Cancer Res. 15:6412–6420. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Jass JR: Lymphocytic infiltration and
survival in rectal cancer. J Clin Pathol. 39:585–589. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Schumacher TN and Schreiber RD:
Neoantigens in cancer immunotherapy. Science. 348:69–74. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Brown SD, Warren RL, Gibb EA, Martin SD,
Spinelli JJ, Nelson BH and Holt RA: Neo-antigens predicted by tumor
genome meta-analysis correlate with increased patient survival.
Genome Res. 24:743–750. 2014. View Article : Google Scholar : PubMed/NCBI
|