Introduction
The case reports on carcinosarcoma of the kidney are
extremely rare in the literature. Sarcomatoid kidney carcinoma was
first described by Farrow et al (1) in 1968. However, carcinosarcoma of the
urinary tract was first described by Robson (2) in 1935. Although this type of tumor
accounts for <1% of all malignant renal tumors, it requires
strict follow-up upon establishing the diagnosis due to its
aggressive nature and high metastatic potential. The presence of
the sarcomatoid component is an indication of an aggressive tumor
nature (3). Carcinosarcoma of the
kidney is a biphasic tumor and the biphasic nature of the tumor
must be confirmed using immunohistochemical methods while
establishing the pathological diagnosis (4). Tumor location in the renal pelvis and
calyceal epithelial components together with mesenchymal malignant
components have been considered to promote early metastasis
(3).
Case report
Clinical characteristics
A 56-year-old male patient presented with left flank
pain persisting over the previous 6 months. The patient's history
included diabetes mellitus and heavy smoking. The liver function
tests were normal. The blood biochemistry results were as follows:
Glucose, 153 mg/dl; creatinine, 1.5 mg/dl; urea, 56 mg/dl; white
blood cell count, 7.11×103/µl; hemoglobin, 12.3 g/dl;
platelet count, 308,000 mm3; sodium, 133 mmol/l;
potassium, 4.9 mmol/l; chloride, 103 mEq/l; calcium, 8.6 mg/dl; and
erythrocyte sedimentation rate, 42 mm/h. On physical examination,
there was tenderness on palpation in the left lumbar region.
Ultrasonography revealed left-sided grade IV hydronephrosis and the
computed tomography (CT) revealed left ureterohydronephrosis and a
urinary stone in the left distal ureter measuring 25 mm in
diameter. Renal scan with dimercaptosuccinic acid and
diethylenetriamine pentaacetate revealed a non-functional left
kidney and the patient underwent a nephroureterectomy.
Immunohistopathological
characteristics
On macroscopic examination, the nephroureterectomy
specimen included the left kidney, measuring 18×13×8 mm, a ureteral
segment 190 mm in length and a ureteral calculus measuring 25 mm in
diameter. The thickness of the renal cortical parenchyma was
reduced to 1 mm. Three nodular lesions with irregular margins,
brown to dark yellow in color were identified in the kidney, with
the largest of the lesions measuring 30 mm in diameter. The total
diameter of the nodular lesions was 70 mm. The microscopic
examination of the lesions revealed tumor cells with fusiform
nuclei and a pink cytoplasm, exhibiting diffuse pleomorphism and
areas of necrosis. The mitotic count was 19–20/10 high-power
fields. Islands of carcinomatous cells were identified, embedded in
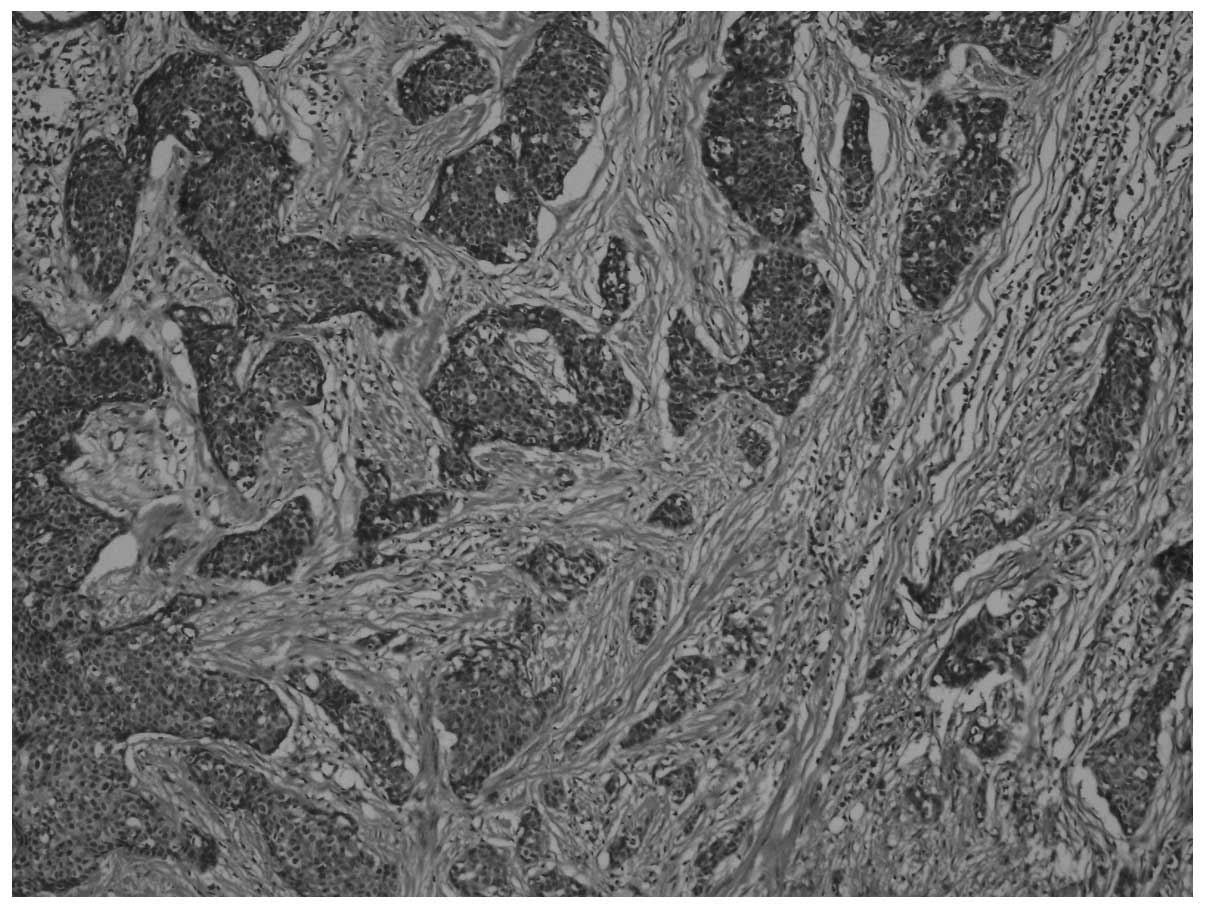
a desmoplastic stroma [hematoxylin and eosin (H&E) staining;
magnification, x40; Fig. 1].
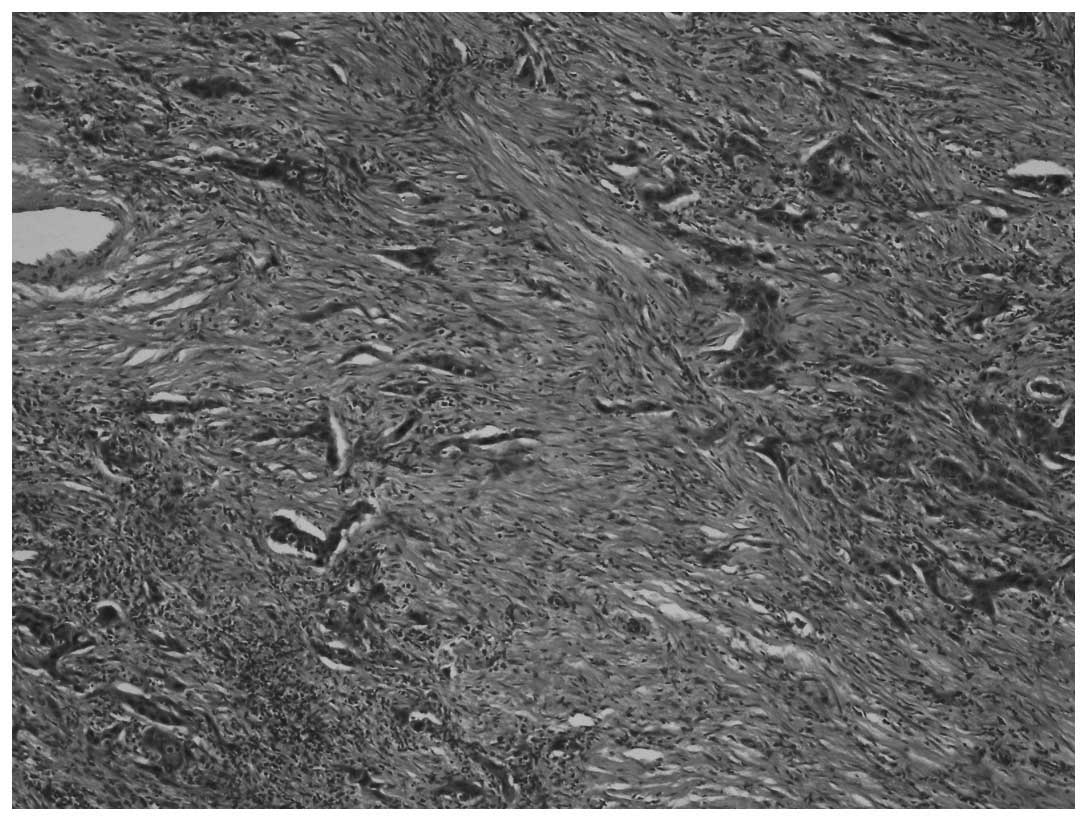
Sarcomatous areas, composed of pleomorphic fusiform cells with
marked atypia were also identified (Fig. 2) (H&E staining, magnification,
x40).
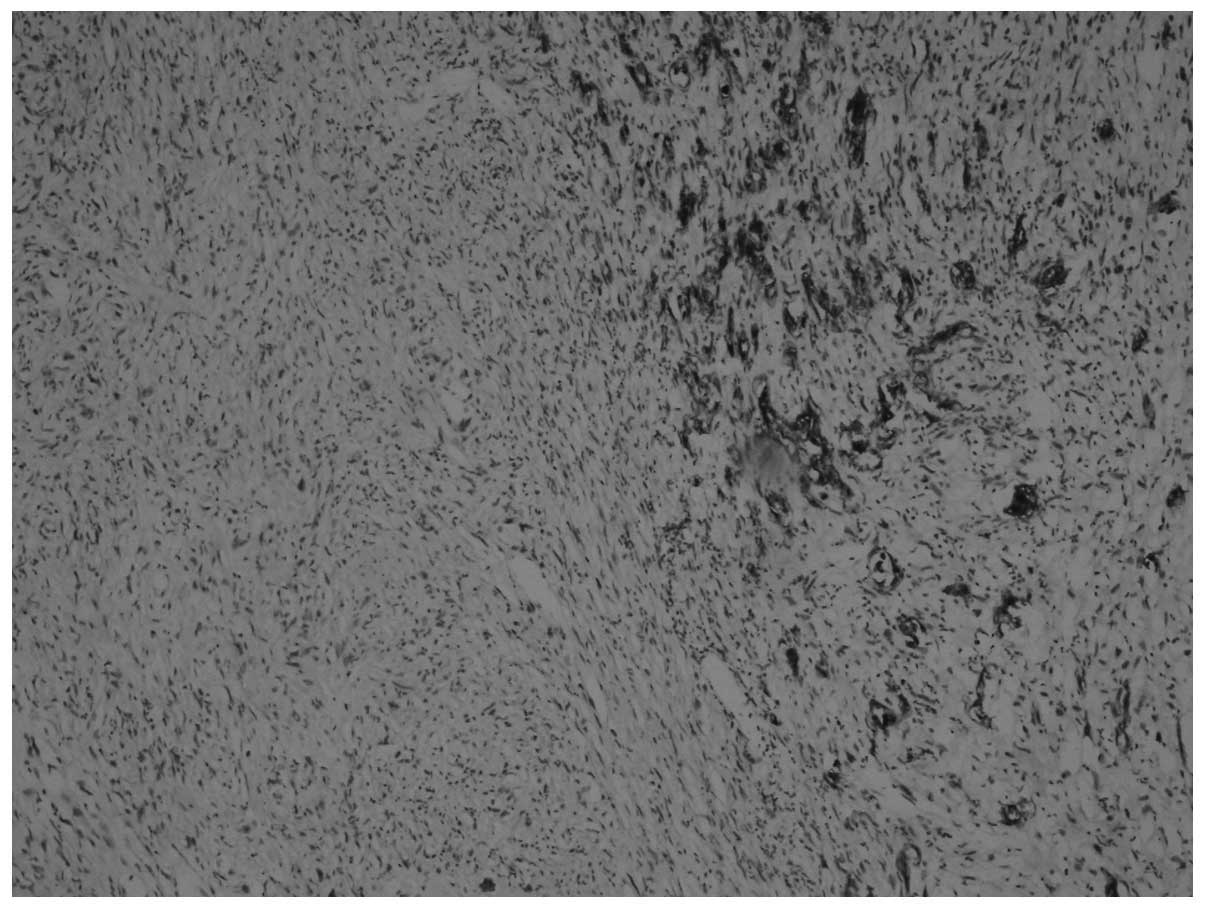
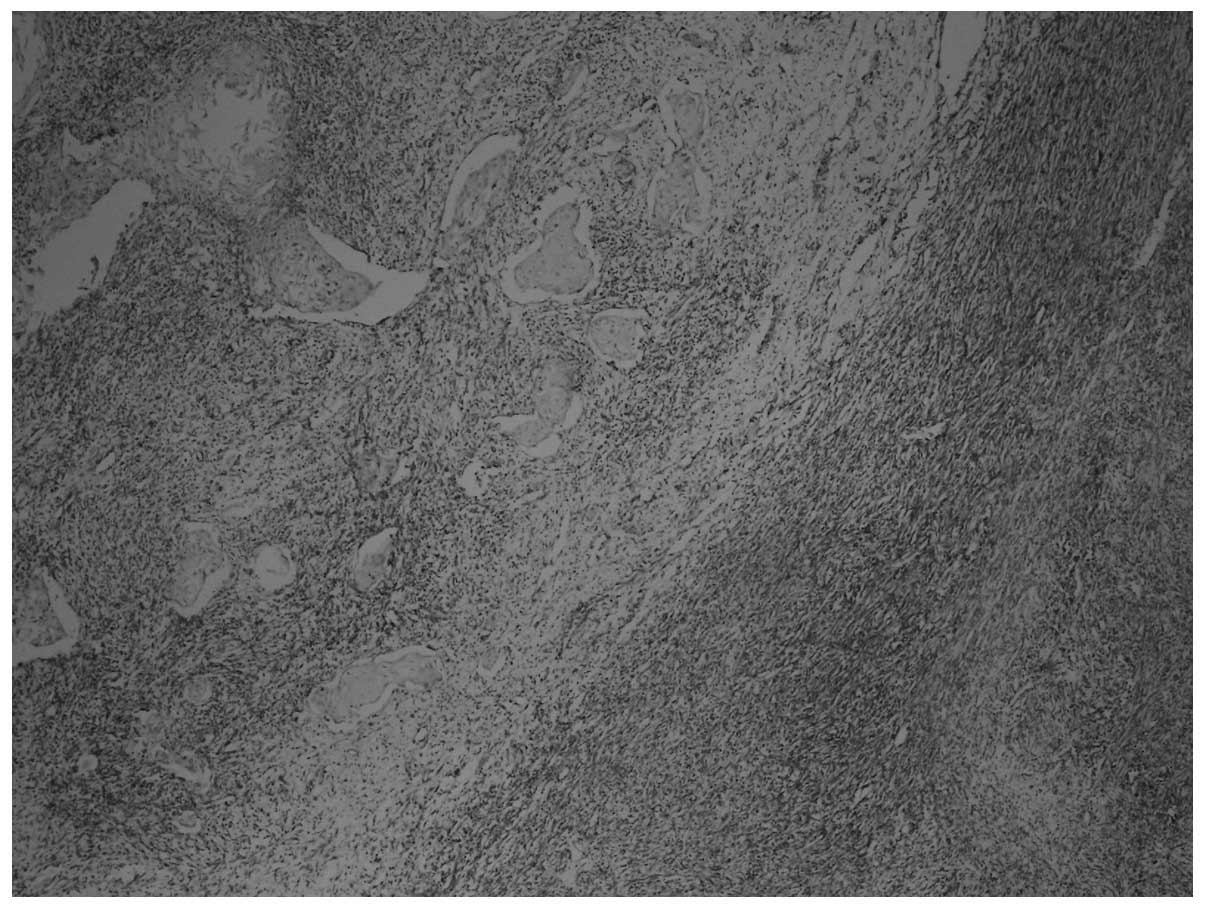
On immunohistochemical examination, the tumor cells
were pan-cytokeratin+, DKA+,
desmin+, vimentin+, CD117−,
CD34− and S-100− (Figs. 3 and 4). The Ki-67 proliferation index was 70%.
Sarcomatous components were identified, together with carcinomatous
components and transitional zones between the two. The transitional
zones between sarcomatous and carcinomatous areas are demonstrated
in Figs. 3 and 4, using pan-cytokeratin and vimentin
immunostaining, respectively.
Staging, treatment and outcome
According to Union for International Cancer Control
and the tumour-node-metastasis staging system of the European
Association of Urology, the case was diagnosed as high-grade, stage
T3aN0M0 cystic carcinosarcoma of the kidney (5). On conventional cystoscopy, there was
no concomitant bladder tumor. Distant metastasis was not observed
at the time of the diagnosis and during nephroureretectomy.
However, widespread lung and liver metastases were identified on
18F-fluorodeoxyglucose positron-emission tomography/CT
at 6 months follow-up and systemic adjuvant chemotherapy (CTx) was
administered. However, there was no response to CTx. The patient
was succumbed to the disease in the first year of follow-up.
Ethics statement and informed
consent
All the procedures were in accordance with the
ethical standards of the responsible Committee on Human
Experimentation and with the Helsinki Declaration of 1975, as
revised in 2000. Written informed consent was obtained from the
patient.
Discussion
According to the World Health Organization
classification of urothelial cancers in 2014, it is recommended to
use the term ‘sarcomatoid carcinoma’ for all biphasic malignant
tumors in which epithelial and mesenchymal differentiation is
confirmed using morphological and/or immunohistochemical criteria
(6). However, both definitions
were used in this study. Sarcomatoid carcinomas may occur in any
location in the urinary tract. Sarcomatoid carcinoma of the bladder
is the most common occurrence reported in the literature.
Sarcomatoid carcinoma of the kidney is less common, but it is an
aggressive tumor with a high metastatic potential. Sarcomatoid
carcinoma of the kidney most commonly occurs at ages ≥60 years and
the majority of the patients are men. Sarcomatoid carcinomas are
solitary tumors and rarely occur in multiple locations (7). Contrary to the information reported
in the literature, our case presented with mass lesions in 3
different locations in a cystic hydronephrotic kidney.
There are also other case reports on the multifocal
occurrence of these lesions. Ishumura et al (8) reported a case with multiple
carcinosarcomatous lesions located concurrently in the kidney and
in different locations in the ipsilateral ureter. Sarcomas occur as
solitary tumors; however, the identification of tumor lesions with
similar histopathological characteristics in different locations of
the kidney in the present case suggests that they may be affected
by similar carcinogenic factors. The underlying chronic urinary
tract obstruction secondary to ureteral lithiasis may have
triggered the development of the disease in multiple locations
independently from one another. The factors triggering the
development of this disease have not yet been clearly determined.
Sarcomatoid carcinomas are biphasic tumors with carcinomatous and
sarcomatous components that are considered to arise from primitive
pluripotent stem cells (9).
However, the precise histogenesis of this type of cancer has not
been fully elucidated (10). Wang
et al (11) identified
duplications of chromosomes 3, 7 and 17 and deletion of chromosome
9p21 as the common genetic alterations among sarcomatoid carcinomas
of the upper urinary tract.
The biphasic components of the primary
carcinosarcoma of the kidney include epithelial and mesenchymal
components. The most common epithelial components are urothelial
carcinoma, carcinoma in situ, adenocarcinoma, squamous cell
carcinoma and small-cell carcinoma. The most common mesenchymal
component is leiomyosarcoma and less common components include
rhabdomyosarcoma, liposarcoma, osteosarcoma, angiosarcoma,
fibrosarcoma, chondrosarcoma, malignant Schwannoma and Ewing's
sarcoma (12). The present case
was diagnosed with high-grade transitional cell carcinoma together
with high-grade leiomyosarcoma. The mesenchymal components of
sarcomatoid carcinomas must be confirmed by pathological
examination. Osseous and chondroid metaplasia and carcinomas with
pseudosarcomatous stroma must be excluded, as metaplastic changes
may have malignant potential, but they should not be considered as
malignant lesions. One of the most significant histopathological
parameters that supports the diagnosis of sarcomatoid carcinoma is
the identification of transitional zones between epithelial and
mesenchymal cells (10).
The epithelial and mesenchymal components must be
distinguished using immunohistochemical methods. The presence of
diffuse and marked mitotic activity and atypical mitoses support
the diagnosis of sarcomatoid carcinoma (12). Carcinosarcomas are malignant and
highly aggressive tumors with a high metastatic potential. Radical
nephrectomy is the primary treatment method for renal
carcinosarcoma. No beneficial effect of adjuvant radiotherapy and
CTx has been demonstrated thus far (9). We did not achieve any response to
adjuvant CTx in the present case. The most commonly used
chemotherapeutic agents include cisplatin, dacarbazine, docetaxel,
gemcitabine, methotrexate, oxaliplatin, doxorubicin, paclitaxel,
vincristine and vinorelbine. However, no treatment protocol has
been established for adjuvant CTx.
Carcinosarcomas may be asymptomatic, but they may
also present as palpable lumbar or abdominal masses, accompanied by
lumbar or abdominal pain and hematuria. The patients may also
exhibit systemic symptoms, including anorexia, weight loss,
malaise, fatigue, fever, night sweats or cough (13).
The etiology of sarcomas remains unknown and there
is no sufficient data on their biological behavior. In the
literature, there are reports of cases who sustained spontaneous
intraperitoneal rupture and life-threatening hemorrhage due to the
aggressive nature of carcinosarcomas (14). This type of biphasic tumor is
associated with poor prognosis and such tumors are devoid of
natural barriers due to the mesenchymal tissue arising from the
sarcomatoid component, whereas the tumors typically carry a
pseudocapsule. These tumors have high metastatic potential and
multifocal occurrence is an indicator of poor prognosis (15), with a 5-year survival rate of
<10% in T3 tumors (16). In the
present case, multifocal involvement and with advanced stage
reduced the life expectancy of the patient.
In conclusion, primary carcinosarcoma of the kidney
is a rare tumor with aggressive bilogical behavior and a high
metastatic potential. The patients should be closely monitored due
to the unknown or unpredictable biological behavior of this tumor.
The scarcity of the described cases in the literature does not
allow for efficient histopathological classification and staging,
further complicating the selection of the appropriate treatment
modality with respect to disease stage. Our basic knowledge on
carcinosarcomas of the kidney dictates the need for a
multidisciplinary approach, including radical surgical excision,
radiotherapy and adjuvant CTx.
References
|
1
|
Farow GM, Harrison EG Jr and Utz DC:
Sarcomas and sarcomatoid and mixed malignant tumors of the kidney
in adults. 3. Cancer. 22:556–563. 1968. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Robson SM: Atypical carcinoma of the
urinary bladder simulating myosarcoma. Report of two cases and
review of the literature. J Urol. 34:638–669. 1935.
|
|
3
|
Cheville JC, Lohse CM, Zincke H, Weaver
AL, Leibovich BC, Frank I and Blute ML: Sarcomatoid renal cell
carcinoma: an examination of underlying histologic subtype and an
analysis of associations with patient outcome. Am J Surg Pathol.
28:435–441. 2004. View Article : Google Scholar
|
|
4
|
Petersen RO: Urologic Pathology. 2nd. JB
Lippincott; Philadelphia, PA: pp. 128–132. 1992
|
|
5
|
Sobin L, Gospodarowicz M and Wittekind C:
TNM Classification of Malignant Tumours. Urological Tumours. Renal
Pelvis and Ureter. 7th. Wiley-Blackwell, UICC; pp. 258–261.
2009
|
|
6
|
Eble JN, Sauter G, Epstein JI and
Sesterhenn IA: Infiltrating urothelial carcinoma. World Health
Organization Classification of TumoursPathology and Genetics of
Tumours of the Urinary System and Male Genital Organs. 1st. IARC
Press; Lyon: pp. 1022004
|
|
7
|
Perimenis P, Athanasopoulos A, Gerathy J
and Speakman M: Carcinosarcoma of the ureter: a rare, pleomorphic,
aggressive malignancy. Int Urol Nephrol. 35:491–493. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ishimura H, Momose A, Narita S and
Kurotaki H: Synchronous multiple carcinosarcoma of the renal pelvis
and ureter: a case report. Acta Urol Jap. 56:381–384. 2010.(In
Japanese).
|
|
9
|
Volker HU, Zettl A, Georg S, et al:
Molecular findings in two cases of sarcomatoid carcinoma of the
ureter: evidence for evolution from a common pluripotent progenitor
cell? Virchows Arch. 452:457–463. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Darko A, Das K, Bhalla RS and Heller D:
Carcinosarcoma of the ureter: report of a case with unusual
histology and review of the literature. Int J Urol. 13:1528–1531.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang X, MacLennan GT, Zhang S, et al:
Sarcomatoid carcinoma of the upper urinary tract: clinical outcome
and molecular characterization. Hum Pathol. 40:211–217. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Maeda D, Fujii A, Yamaguchi K, et al:
Sarcomatoid carcinoma with a predominant basaloid squamous
carcinoma component: the first report of an unusual biphasic tumor
of the ureter. Jpn J Clin Oncol. 37:878–883. 2007. View Article : Google Scholar
|
|
13
|
Raman JD, Shariat SF, Karakiewicz PI, et
al: Does preoperative symptom classification impact prognosis in
patients with clinically localized upper-tract urothelial carcinoma
managed by radical nephroureterectomy? Urol Oncol. 29:716–723.
2011. View Article : Google Scholar
|
|
14
|
Quaresima S, Manzelli A, Ricciardi E, et
al: Spontaneous intraperitoneal rupture of pyonephrosis in a
patient with unknown kidney carcinosarcoma: a case report. World J
Surg Oncol. 9:392011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chromecki TF, Cha EK, Fajkovic H, et al:
The impact of tumor multifocality on outcomes in patients treated
with radical nephroureterectomy. Eur Urol. 61:245–253. 2012.
View Article : Google Scholar
|
|
16
|
Jeldres C, Sun M, Isbarn H, et al: A
population-based assessment of perioperative mortality after
nephroureterectomy for upper-tract urothelial carcinoma. Urology.
75:315–320. 2010. View Article : Google Scholar : PubMed/NCBI
|