Introduction
Radiofrequency ablation (RFA) has been accepted as a
minimally invasive treatment for liver tumors (1) due to its tolerable side effects
(2,3).
Although RFA is a major technique for the treatment of liver
tumors, there is an important limitation of image-guided RFA,
including higher local recurrence rate compared with that of
surgical resection. It has been reported that the main cause of the
higher local recurrence rate associated with RFA is inadequate
volumes of tumor ablation (4–8). In order to improve these results,
significant efforts have been focused on enhancing the therapeutic
effects of RFA by combining it with other modalities, such as
transcatheter arterial chemoembolization (9,10), saline
injection (11,12), and percutaneous ethanol injection
(PEI) (13). However, the local
recurrence rate remains high.
D-sorbitol has been used as an irrigating fluid
during transurethral prostate resection (14) and considered to be a dielectric fluid
(15). During transurethral resection
of the prostate, D-sorbitol is used as lavage for removing blood
and tissue fragments. If absorbed, D-sorbitol is metabolized to
carbon dioxide and water via the fructose pathway, or excreted by
the kidneys. To the best of our knowledge, thus far no experimental
trial has demonstrated the efficacy of combination therapy using
D-sorbitol and RFA.
In the present study, we attempted to demonstrate
the efficacy of RFA with D-sorbitol in increasing the total volume
of coagulation necrosis and analyzed the total delivered energy to
elucidate the underlying mechanism.
Materials and methods
Animals and ethics
All animal experiments were performed in the
Preclinical Animal Laboratory of Kagawa University (Kagawa, Japan).
The protocol of this study was approved by the Institutional Animal
Care and Use Committee of Kagawa University. Adult pig livers were
purchased from Tokyo Shibaura Zoki K.K. (Tokyo, Japan). The
13-month-old dogs were purchased from Hokuzan Rabesu (Nagano,
Japan) and maintained in a pathogen-free facility under controlled
conditions with a 12-h light-dark cycle.
Anesthesia and procedure
RFA and D-sorbitol RFA were performed under general
anesthesia with intramuscular injection of ketamine (10 mg/kg) and
xylazine hydrochloride (4 mg/kg). Intubation was performed 10–15
min later and the administration of 5% sevoflurane mixed with
oxygen was maintained at 1–2 l/min. RFA was initiated after stable
anesthesia was attained.
Equipment and D-sorbitol RFA
procedure
D-sorbitol was purchased from Baxter Ltd. (Tokyo,
Japan). D-sorbitol RFA was performed under real-time
ultrasonographic guidance with a 3.5 MHz sector probe (Power Vision
5000; Toshiba Medical, Tokyo, Japan) using the Cool-tip RF ablation
system (Radionics, Burlington, MA, USA). For the D-sorbitol RFA
treatment, a 16-gauge RFA needle was first inserted into the center
of the tumor and a 18-gauge PEI needle was then introduced through
the same hole of the attachment beside the echo probe. D-sorbitol
3% was slowly injected into the lesion through the side hole of the
handpiece. The maximum total injected volume of D-sorbitol was 20
ml and RFA was terminated if the threshold of impedance was
exceeded. RFA was performed for 20 W in 2 min, 30 W in 2 min and 50
W until the end. The maximum allowable output power was 50 W under
the impedance control injecting D-sorbitol.
Calculation of energy requirement for
ablation and assessment of coagulation necrosis
The energy required for ablation was calculated as
follows: Energy (J) = Watt (W) × duration of ablation (sec). Animal
livers containing RFA-induced coagulation were sliced in the
transverse plane perpendicular to the electrode tracks. The
approximation volume of the whole coagulated necrosis area in
cm3 was calculated as follows:
Whole coagulation volume = 4/3π × r1 × r2 × r3,
where r1, longest diameter/2; r2, shortest
diameter/2; and r3, height/2, all measured in cm. Two hepatologists
with 10 years of clinical experience, who were blinded to the
information regarding the RFA techniques used in the study,
measured these parameters.
Stastistical analysis
All data are presented as means ± standard
deviation. Statistical significance was set at P<0.05 for
Student's unpaired t-tests.
Results
Comparison of coagulation between RFA
and D-sorbitol-RFA in pig liver
RFA and D-sorbitol RFA were performed in 5 different
parts of pig livers. All RFA techniques were performed using the
Cool-tip RF system and 20 ml of D-sorbitol was injected into the
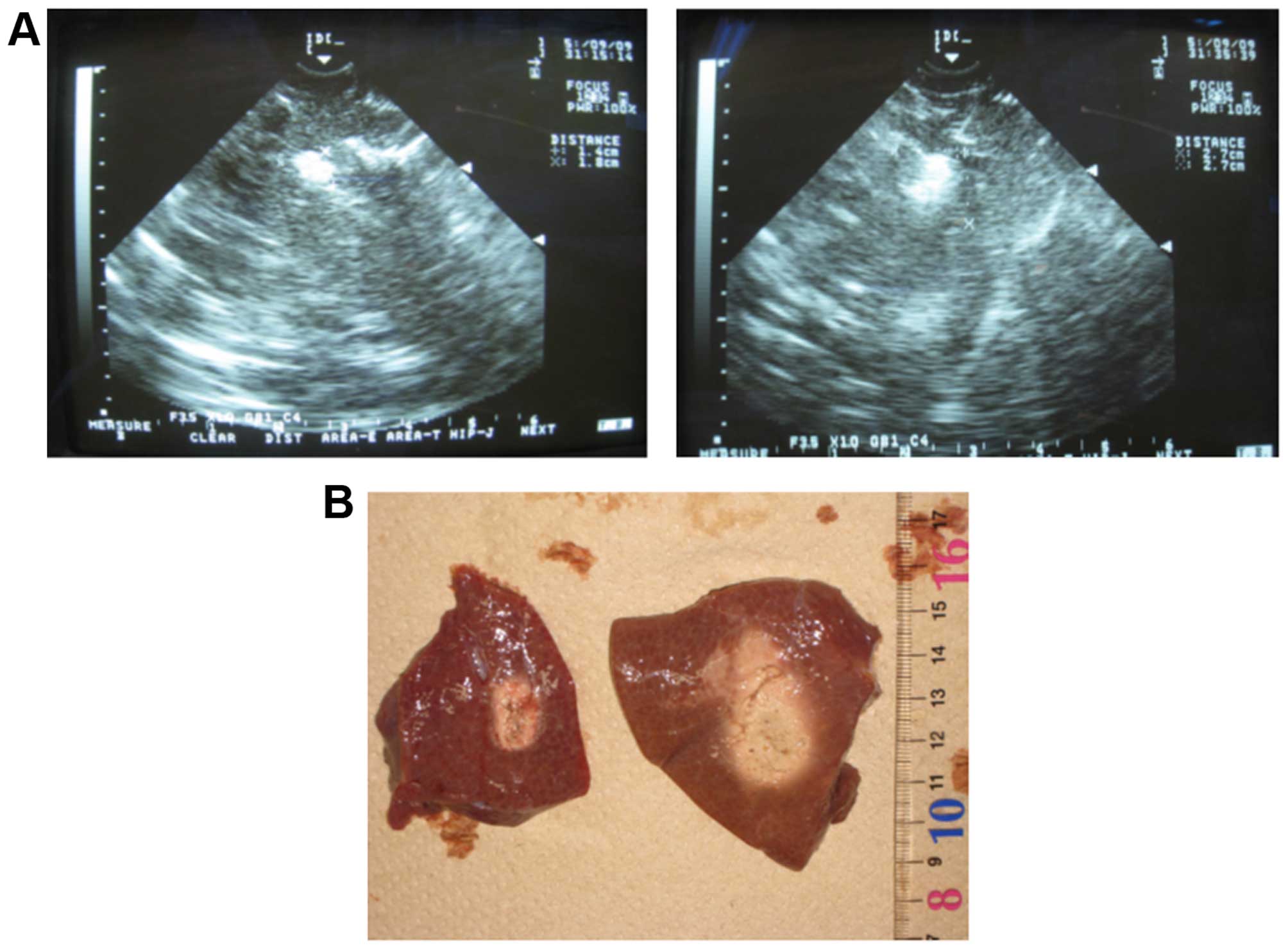
lesion through a 18-gauge PEI needle during RFA (Fig. 1A and B, right). The results of the
comparison of the volume of coagulation necrosis and the total
delivered energy for inducing coagulation between the two groups
are summarized in Table I. The
volumes of coagulation necrosis in the D-sorbitol RFA group
(Fig. 1A and B, right) were
significantly higher compared with those in the RFA group (Fig. 1A and B, left). Of note, the total
delivered energy in the D-sorbitol RFA group was also higher
compared with that in the RFA group (Table I).
 | Table I.Comparison of mean parameter values
between the two groups in pig liver. |
Table I.
Comparison of mean parameter values
between the two groups in pig liver.
| Coagulation
necrosis | RFA | D-sorbitol RFA | P-value |
|---|
| Dmax (mm) |
14.4±1.14 |
28.2±7.79 | 0.0044 |
| Dmin (mm) |
11.4±0.89 |
16±2.92 | 0.0097 |
| Dv (mm) |
10.8±0.84 |
16.4±2.51 | 0.0015 |
| Volume
(mm3) |
2,351.5±520.1 | 10,821.1±6,580.3 | 0.0209 |
| Dmax/Dmin |
1.33±0.05 |
1.70±0.23 | 0.0084 |
| Total delivered
energy (J) |
6,340±313.0 |
14,800±3,898.7 | 0.0013 |
Comparison of coagulation between RFA
and D-sorbitol RFA in a dog model in vivo
The effect of D-sorbitol RFA was also examined in
vivo using a dog model. The volumes of coagulation necrosis in
the D-sorbitol RFA group were clearly higher compared with those in
the RFA group in the dog model in vivo (Table II). In addition, the total delivered
energy in the D-sorbitol RFA group was also higher compared with
that in the RFA group (Table II). No
significant complications, such as bleeding or damage, were
observed during the procedures and 3 days after D-sorbitol RFA.
 | Table II.Comparison of mean parameter values
between the two groups in dog liver. |
Table II.
Comparison of mean parameter values
between the two groups in dog liver.
| Coagulation
necrosis | RFA | D-sorbitol RFA | P-value |
|---|
| Dmax (mm) |
18.2±0.31 |
32.1±2.97 |
0.0013 |
| Dmin (mm) |
14.9±1.37 |
24.1±0.36 |
0.0004 |
| Dv (mm) | 14.7±1.3 |
24.7±0.76 |
0.0003 |
| Volume
(mm3) | 5,318.5±917.3 | 25,418.2±1,545.6 | <0.0001 |
| Dmax/Dmin |
1.23±0.12 |
1.33±0.13 |
0.3213 |
| Total delivered
energy (J) | 7,083.3±381.9 |
17,333±3,214.6 |
0.0054 |
Discussion
RFA remains a promising technique for the local
therapy of liver cancers, such as hepatocellular carcinoma and
metastatic liver cancer (6,16,17).
However, the efficacy of this technique is limited by the size of
coagulation necrosis, and only tumors sized <3 cm are
recommended for complete ablation, due to increased impedance. In
order to control impedance during the ablation, D-sorbitol was used
in combination with RFA therapy. During the D-sorbitol RFA
procedure, impedance was regulated by the injection of D-sorbitol
and the total operative time was longer compared with that of RFA
alone. Therefore, the size of the areas of induced coagulation
necrosis was increased using D-sorbitol in animal livers. This
suggests that the total volume of coagulation necrosis may be
controlled by the injected volume of D-sorbitol suppressing
impedance elevation in the ablated livers. It has also been
reported that combination of RFA with other modalities enhances the
therapeutic effects of RFA (10,11,18).
Kurokohchi et al demonstrated that ethanol-combined RFA
increased the coagulated volumes 4-fold compared with those treated
with RFA alone. In addition, the amount of injected ethanol and
total energy were both required to increase coagulation necrosis
(19). These studies support our
finding that impedance control is key to the regulation of total
energy, and D-sorbitol may increase the effectiveness of RFA
treatment.
In conclusion, RFA combined with D-sorbitol
increases the total volume of coagulation necrosis through
controlling impedance in the ablated liver and, therefore,
D-sorbitol may be useful for the treatment of liver cancers.
Glossary
Abbreviations
Abbreviations:
|
RFA
|
radiofrequency ablation
|
|
PEI
|
percutaneous ethanol injection
|
References
|
1
|
Decadt B and Siriwardena AK:
Radiofrequency ablation of liver tumours: Systematic review. Lancet
Oncol. 5:550–560. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Solbiati L, Goldberg SN, Ierace T,
Livraghi T, Meloni F, Dellanoce M, Sironi S and Gazelle GS: Hepatic
metastases: Percutaneous radio-frequency ablation with cooled-tip
electrodes. Radiology. 205:367–373. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Solbiati L, Ierace T, Goldberg SN, Sironi
S, Livraghi T, Fiocca R, Servadio G, Rizzatto G, Mueller PR, Del
Maschio A, et al: Percutaneous US-guided radio-frequency tissue
ablation of liver metastases: Treatment and follow-up in 16
patients. Radiology. 202:195–203. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shiina S, Tateishi R, Arano T, Uchino K,
Enooku K, Nakagawa H, Asaoka Y, Sato T, Masuzaki R, Kondo Y, et al:
Radiofrequency ablation for hepatocellular carcinoma: 10-year
outcome and prognostic factors. Am J Gastroenterol. 107:569–577.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shiina S, Teratani T, Obi S, Sato S,
Tateishi R, Fujishima T, Ishikawa T, Koike Y, Yoshida H, Kawabe T,
et al: A randomized controlled trial of radiofrequency ablation
with ethanol injection for small hepatocellular carcinoma.
Gastroenterology. 129:122–130. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Curley SA, Izzo F, Delrio P, Ellis LM,
Granchi J, Vallone P, Fiore F, Pignata S, Daniele B and Cremona F:
Radiofrequency ablation of unresectable primary and metastatic
hepatic malignancies: Results in 123 patients. Ann Surg. 230:1–8.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lin SM, Lin CJ, Lin CC, Hsu CW and Chen
YC: Radiofrequency ablation improves prognosis compared with
ethanol injection for hepatocellular carcinoma < or =4 cm.
Gastroenterology. 127:1714–1723. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lencioni RA, Allgaier HP, Cioni D,
Olschewski M, Deibert P, Crocetti L, Frings H, Laubenberger J,
Zuber I, Blum HE, et al: Small hepatocellular carcinoma in
cirrhosis: Randomized comparison of radio-frequency thermal
ablation versus percutaneous ethanol injection. Radiology.
228:235–240. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Buscarini L, Buscarini E, Di Stasi M,
Quaretti P and Zangrandi A: Percutaneous radiofrequency thermal
ablation combined with transcatheter arterial embolization in the
treatment of large hepatocellular carcinoma. Ultraschall Med.
20:47–53. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yamasaki T, Kurokawa F, Shirahashi H,
Kusano N, Hironaka K and Okita K: Percutaneous radiofrequency
ablation therapy with combined angiography and computed tomography
assistance for patients with hepatocellular carcinoma. Cancer.
91:1342–1348. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Livraghi T, Goldberg SN, Monti F, Bizzini
A, Lazzaroni S, Meloni F, Pellicanò S, Solbiati L and Gazelle GS:
Saline-enhanced radio-frequency tissue ablation in the treatment of
liver metastases. Radiology. 202:205–210. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Honda N, Guo Q, Uchida H, Ohishi H and
Hiasa Y: Percutaneous hot saline injection therapy for hepatic
tumors: An alternative to percutaneous ethanol injection therapy.
Radiology. 190:53–57. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kurokohchi K, Watanabe S, Masaki T, Hosomi
N, Funaki T, Arima K, Yoshida S, Miyauchi Y and Kuriyama S:
Combined use of percutaneous ethanol injection and radiofrequency
ablation for the effective treatment of hepatocelluar carcinoma.
Int J Oncol. 21:841–846. 2002.PubMed/NCBI
|
|
14
|
Norlén H, Allgén LG, Bendz R and Wiklund
A: Influence of various irrigation fluids on serum enzyme patterns
following transurethral resection of the prostate. Scand J Urol
Nephrol. 20:127–136. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Norlén H, Allgén LG and Wicksell B:
Sorbitol concentrations in plasma in connection with transurethral
resection of the prostate using sorbitol solution as an irrigating
fluid. Scand J Urol Nephrol. 20:9–17. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Goldberg SN, Gazelle GS, Compton CC,
Mueller PR and Tanabe KK: Treatment of intrahepatic malignancy with
radiofrequency ablation: Radiologic-pathologic correlation. Cancer.
88:2452–2463. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jiang HC, Liu LX, Piao DX, Xu J, Zheng M,
Zhu AL, Qi SY, Zhang WH and Wu LF: Clinical short-term results of
radiofrequency ablation in liver cancers. World J Gastroenterol.
8:624–630. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kitamoto M, Imagawa M, Yamada H, Watanabe
C, Sumioka M, Satoh O, Shimamoto M, Kodama M, Kimura S, Kishimoto
K, et al: Radiofrequency ablation in the treatment of small
hepatocellular carcinomas: Comparison of the radiofrequency effect
with and without chemoembolization. AJR Am J Roentgenol.
181:997–1003. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kurokohchi K, Watanabe S, Masaki T, Hosomi
N, Miyauchi Y, Himoto T, Kimura Y, Nakai S, Deguchi A, Yoneyama H,
et al: Comparison between combination therapy of percutaneous
ethanol injection and radiofrequency ablation and radiofrequency
ablation alone for patients with hepatocellular carcinoma. World J
Gastroenterol. 11:1426–1432. 2005. View Article : Google Scholar : PubMed/NCBI
|