Introduction
Glucocorticoids (GCs) are essential for survival and
serve a major role in embryonic development, tissue homeostasis and
in the regulation of the inflammatory response (1,2). In
breast, the functions of GCs are complex and depend, in part, if
they are linked to their receptor (GR) and consist of the control
of milk secretion, differentiation and apoptosis (3). Morphologically, previous studies have
demonstrated that GR nuclear expression is observed both in normal
breast, in situ carcinoma and less frequently in invasive
carcinoma (4–7). In invasive tumors, their expression is
limited in tumors with a small size, low grade, good prognosis and
expressing estrogen receptor (ER) (4,7). In
addition, our previous study clearly demonstrated that
dexamethasone has an antiproliferative effect in the MCF-7 breast
cells that express GR (8).
Over previous years, the tumor-associated stroma
and, in particular, the cancer associated fibroblasts (CAFs) have
been demonstrated to serve a crucial role in cancer pathogenesis
(9,10).
Our previous study clearly demonstrated that the
majority of these CAFs were smooth muscle actin (SMA)-positive with
a myofibroblastic-like phenotype and that the presence of these
peritumoral myofibroblasts (PMY) is important both in situ
and in invasive breast carcinoma of no special type (11). This is also important in metastatic
disease by promoting tumor invasion, growth and angiogenesis
through paracrine factors and/or direct cell-cell crosstalk
(11–13).
Our previous study demonstrated the presence of ER
or progesterone receptors (PR); however, the presence and potential
role of GR is poorly understood in breast carcinoma PMYs (4).
Therefore, the present study aimed to assess, by
immunohistochemistry, the presence or absence of GR in breast CAFs
and in CAFs smooth muscle positive/PMY in correlation with
clinicopathological variables. Investigating this may assist with
elucidating the role of GR in breast carcinoma.
Materials and methods
Patient selection
Breast tissue samples were retrieved from the
Departments of Pathology at the Erasme Hospital and IRIS South
Hospital (Brussels, Belgium), and consisted of 56 cases of invasive
carcinoma. The present study was approved by the Ethics Committee
from Erasme University Hospital (no. P2014/418).
Immunohistochemistry
The immunohistochemical assessment of ER, PR, Ki-67
and human epidermal growth factor receptor (HER)2 was routinely
performed using an antigen retrieval method using the Leica
BOND-III fully automated system (Leica Microsystems, Ltd.,
Newcastle, UK), as previously described (4). According to these parameters, carcinoma
were divided into five groups, as previously described (14): Luminal A (n=19), Luminal B (n=12),
HER2+/ER+ (n=7), HER2+/ER- (n=9) and triple negative (n=9). In
addition, the following parameters were also included for each
patient: Age, stage, tumoral size and lymph node status. All
parameters are shown in the Table
I.
 | Table I.Association of clinicopathological
characteristics with immunohistochemical levels of GR in the
peritumoral stroma. |
Table I.
Association of clinicopathological
characteristics with immunohistochemical levels of GR in the
peritumoral stroma.
|
|
| GT expression in the
stroma |
|
|---|
|
|
|
|
|
|---|
| Characteristic | No. cases (%) | Strong | Weak | Negative | P-value |
|---|
| Age, years |
|
|
|
| 0.56 |
| ≤50 | 24 (43) | 18 | 5 | 1 |
|
|
>50 | 32 (57) | 23 | 5 | 4 |
|
| Tumor size, mm |
|
|
|
| 1 |
|
<20 | 28 (50) | 21 | 5 | 2 |
|
| ≥20 | 28 (50) | 20 | 5 | 3 |
|
| Stage |
|
|
|
| 0.81 |
| T1 | 28 (50) | 21 | 5 | 2 |
|
| T2 | 21 (37.5) | 15 | 3 | 3 |
|
| T3 | 7 (12.5) | 5 | 2 | 0 |
|
| Tumor grade |
|
|
|
| 0.03 |
| Grade
1 | 10 (17.9) | 10 | 0 | 0 |
|
| Grade
2 | 22 (39.2) | 12 | 8 | 2 |
|
| Grade
3 | 24 (42.9) | 19 | 2 | 3 |
|
| Lymph node
status |
|
|
|
| 0.19 |
|
Negative | 29 (51.8) | 24 | 4 | 1 |
|
|
Positive | 27 (48.2) | 17 | 6 | 4 |
|
| ER status |
|
|
|
| 0.64 |
|
Negative | 18 (32.1) | 14 | 2 | 2 |
|
|
Positive | 38 (67.9) | 27 | 8 | 3 |
|
| PR status |
|
|
|
| 0.39 |
|
Negative | 23 (41.1) | 15 | 6 | 2 |
|
|
Positive | 33 (58.9) | 26 | 4 | 3 |
|
| Ki-67 index, % |
|
|
|
| 0.003 |
|
≤14 | 19 (33.9) | 19 | 0 | 0 |
|
|
>14 | 37 (66.1) | 22 | 10 | 5 |
|
| HER 2 status |
|
|
|
| 0.2 |
|
Negative | 40 (71.4) | 31 | 7 | 2 |
|
|
Positive | 16 (28.6) | 10 | 3 | 3 |
|
| Intrinsic
subtype |
|
|
|
| 0.189 |
| Luminal
A | 19 (33.9) | 19 | 0 | 0 |
|
| Luminal
B/HER2− | 12 (21.4) | 5 | 5 | 2 |
|
| Luminal
B/HER2+ | 7 (12.5) | 4 | 2 | 1 |
|
|
HER2+ | 9 (16.1) | 6 | 1 | 2 |
|
| Triple
negative | 9 (16.1) | 7 | 2 | 0 |
|
| GR status in
glands |
|
|
|
| 0.01 |
|
Negative | 26 (46.4) | 15 | 6 | 5 |
|
|
Positive | 30 (53.6) | 26 | 4 | 0 |
|
For the demonstration of GR, a manual technique was
applied. Tissue sections (4 µm) were cut sequentially and mounted
onto superfrost-treated slides (Menzel-Gläser, Braunschweig,
Germany). The slides were dried overnight at 37°C prior to
deparaffinization in xylene and rehydration through graded
ethanols. For epitope retrieval, the slides were immersed in a
waterbath at 95–99°C for 90 min with an ethylenediamine tetraacetic
acid buffer (pH 9.0; S236; Dako Corp., Glostrup, Denmark). The
slides were subsequently cooled in the buffer for 20 min at room
temperature. H2O2 (0.3%) was subsequently
added to the slides and incubated for 30 min. The tissues were then
incubated for 1 h with a monoclonal antibody against the N-terminus
of the GR (clone 4H2; cat. no. NCL-GCR; 1:25; Novocastra
Laboratories, Newcastle, UK) (4).
Double immunostaining
In addition, for the specific visualization of the
expression of ER/PR and GR in SMA-positive CAFs, a double stain was
also performed by using the EnVision G/2 double stain system (Dako
Corp.), as previously described (15). The same monoclonal antibodies (ER, PR
and GR) described above were applied to rehydrated paraffin tissue
sections and allowed to incubate for 1 h at room temperature.
Endogenous peroxidase was inhibited and 3,3′-diaminobenzidine was
used to visualize the binding of these primary antibodies. The
sections were subsequently incubated for 1 h with a secondary
antibody against SMA (clone αSM-1; 1:50; Novocastra Laboratories,
Newcastle, UK). Alkaline phosphatase-conjugated secondary antibody
and fuchsin as substrate chromogen system were used to complete the
secondary immunostain. Negative controls used the replacement of
the different primary antibodies with the corresponding isotypes.
In addition, to ensure the absence of PMY in the normal breast, 10
cases of normal breast tissue from patients who underwent plastic
surgery were also included.
Immunohistochemical evaluation
All the slides were examined by two independent
observers (Xavier Catteau and Jean-Christophe Noël) and the
evaluation of ER, PR and GR was made independently by the two
pathologists using the Allred score (16), and estimated the proportion of
positive CAFs (0, no positive cells; 1, ≤1; 2, 1–10; 3, 11–33; 4,
34–66; 5, 67–100% positive cells) and the average staining
intensity (0, negative; 1, weak; 2, Intermediate; 3, strong). The
proportion score and the intensity score were added to obtain a
total score ranging from 0–8. Subsequently, three grades of
immunoreactivity were established: Score 0–2, negative; score 3–4,
weak positivity; score 5–8, strong positivity.
Statistical analysis
The correlation analysis was performed.
χ2-test and Fisher's exact test were used. All
statistical analyses were performed using XLSTAT software
(Addinsoft, Paris, France). P<0.05 was considered to indicate a
statistically significant difference.
Results
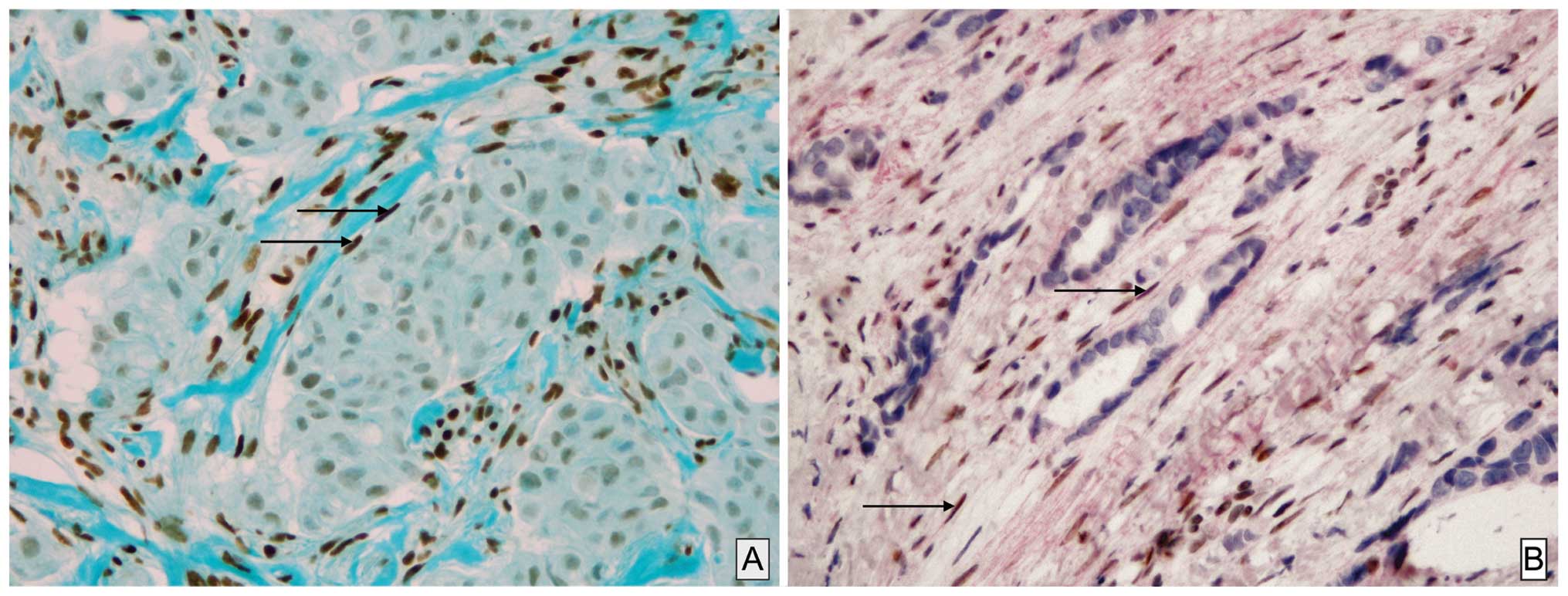
Weak or strong expression of GR in CAFs was observed
in 10 cases (18%) and 41 cases (73%), respectively. A total of 5
cases were negative (5%; Table I).
The stromal expression was frequent in luminal A tumor (100% of
cases; Fig. 1A) and is statistically
correlated with the tumor grade (P=0.03), the Ki-67 index (P=0.003)
and GR status in glandular/carcinomatous component (P=0.01);
however, was not correlated with age, tumor size, lymph node status
and the expression of ER or PR, at least with a positive status for
the latest as ≥1% in accordance with the World Health Organization
recommendations (Table I). The double
stain immunohistochemistry confirming unequivocally that among
these CAFs, SMA-positive PMY clearly showed a nuclear staining of
GR (Fig. 1B).
Discussion
For numerous years, the majority of studies in
breast carcinoma have been focused predominantly on the epithelial
component; however, recently CAFs and in particular CAFs
SMA-positive PMY have been demonstrated to serve an important role
in cancer pathogenesis as a result of paracrine cross-interaction
between these and epithelial cancer cells. Indeed CAFs/PMY are able
to secrete various factors implicated in invasion, matrix
remodeling, cell proliferation, differentiation and apoptosis. In
breast carcinoma, the hormonal regulation of epithelial cells is
well documented and is the result of interaction between estrogen
and progesterone, and their respective receptors, at least in
hormone-dependent tumor types (16,17). The
role of GCs in normal breast is more controversial and likely
depends on the balance between targets of linked and non-linked GR
with opposing functions: linked GR being involved in maintaining
functional differentiation and non-linked GR appearing to be
proapoptotic (3,6). In breast carcinoma, the GCs acting
through their nuclear receptors are considered as a potential tumor
suppressor promoting accurate chromosome segregation during mitosis
occurring tumoral cell division (6,18,19). Indeed, according to these data, our
previous study demonstrated an antiproliferative effect of
dexamethosone in the MCF-7 breast cancer cells line that contains
nuclear GR (8). The underlying
mechanisms of hormonal regulation of CAFs/PMY remain to be
determined; however, our previous study and other previous studies
have clearly demonstrated that ER and PR were not present in these
cells.
By contrast, the present study clearly demonstrated
the presence of a marked GC nuclear immunoreactivity of CAFs in 73%
of cases. In addition, this strong immunoreactivity was
demonstrated by double labeling in CAFs SMA-positive PMY (Fig. 1) for the first time, to the best of
our knowledge. This strong CAFs immunoreactivity was typically more
frequent in luminal A (100%) compared with in other subtypes. In
addition, it appeared to be correlated with different conventional
clinicopathological parameters, including the grade (P=0.03) and
Ki-67 index (P=0.003). The CR expression in CAFs was also more
frequent when these receptors were present in the carcinomatous
counterpart (P=0.01). These data suggested that, as previously
shown for matrix metalloproteinase-2, the characteristics and
properties of CAFs present in breast carcinoma microenvironment are
probably complex and different from one subtype to another
(10,20–22).
GCs are considered as agents capable of regulating
the proliferation of myofibroblasts in different pathologies where
they serve a major role as wound healing or asthma (23,24).
Previously, in a myofibroblast cell line associated with colonic
carcinoma, dexamethasone inhibited the expression of different
classical procarcinogenic factors, including tenascin C, hepatocyte
growth factor and transforming growth factor-β, in a
receptor-dependent manner (25).
The present data are interesting since over the last
few years it appears that in addition to the development concerning
the classical therapies (hormone therapies, chemotherapies and
immunotherapies), the peritumoral stroma served as potential target
therapy in various carcinomas (9,24,25). Finally, even if it remains
hypothetical, it has been postulated that the stress through GCs
can be a promoting agent in breast cancer (26,27). From
this point of view, demonstrating the presence of GR in the CAFs
may be important.
The demonstration of a frequent expression of GR in
breast CAFs may serve as an interesting target for future therapy
in the regulation of the tumoral breast microenvironment.
Naturally, future research is required, firstly to establish with
larger cohorts the assocaition between the presence of GR in CAFs
and the overall survival, and to understand how a therapy may
influence the CAF associated with breast carcinoma. Such
investigations are in progress.
Acknowledgements
The authors would like to thank Mrs. Isabelle Fayt
and Mrs. Nadège De Kindt for their excellent technical work. The
present study was supported by the Institut de Recherche
Scientifique de Pathologie et de Génétique.
References
|
1
|
Hollenberg SM, Weinberger C, Ong ES,
Cerelli G, Oro A, Lebo R, Thompson EB, Rosenfeld MG and Evans RM:
Primary structure and expression of a functional human
glucocorticoid receptor cDNA. Nature. 318:635–641. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
ArangoLievano M, Lambert WM and Jeanneteau
F: Molecular biology of glucocorticoid signaling. Adv Exp Med Biol.
872:33–57. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ritter HD and Mueller CR: Expression
microarray identifies the unliganded glucocorticoid receptor as a
regulator of gene expression in mammary epithelial cells. BMC
Cancer. 14:2752014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Buxant F, EngohanAloghe C and Noël JC:
Estrogen receptor, progesterone receptor, and glucocorticoid
receptor expression in normal breast tissue, breast in situ
carcinoma, and invasive breast cancer. Appl Immunohistochem Mol
Morphol. 18:254–257. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lien HC, Lu YS, Cheng AL, Chang WC, Jeng
YM, Kuo YH, Huang CS, Chang KJ and Yao YT: Differential expression
of glucocorticoid receptor in human breast tissues and related
neoplasms. J Pathol. 209:317–327. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Vilasco M, Communal L, Mourra N, Courtin
A, Forgez P and Gompel A: Glucocorticoid receptor and breast
cancer. Breast Cancer Res Treat. 130:1–10. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Abduljabbar R, Negm OH, Lai CF, Jerjees
DA, AlKaabi M, Hamed MR, Tighe PJ, Buluwela L, Mukherjee A, Green
AR, Ali S, et al: Clinical and biological significance of
glucocorticoid receptor (GR) expression in breast cancer. Breast
Cancer Res Treat. 150:335–346. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Buxant F, Kindt N, Laurent G, Noël JC and
Saussez S: Antiproliferative effect of dexamethasone in the MCF-7
breast cancer cell line. Mol Med Rep. 12:4051–4054. 2015.PubMed/NCBI
|
|
9
|
Otranto M, Sarrazy V, Bonté F, Hinz B,
Gabbiani G and Desmoulière A: The role of the myofibroblast in
tumor stroma remodeling. Cell Adh Migr. 6:203–219. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gandellini P, Andriani F, Merlino G,
D'Aiuto F, Roz L and Callari M: Complexity in the tumour
microenvironment: Cancer associated fibroblast gene expression
patterns identify both common and unique features of tumour-stroma
crosstalk across cancer types. Semin Cancer Biol. 35:96–106. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Catteau X, Simon P, Vanhaeverbeek M and
Noël JC: Variable stromal periductular expression of CD34 and
smooth muscle actin (SMA) in intraductal carcinoma of the breast.
PLoS One. 8:e577732013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Catteau X, Simon P and Noël JC:
Myofibroblastic stromal reaction and lymph node status in invasive
breast carcinoma: Possible role of the TGF-β1/TGF-βR1 pathway. BMC
Cancer. 14:4992014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Catteau X, Simon P and Noël JC:
Myofibroblastic reaction is a common event in metastatic disease of
breast carcinoma: A descriptive study. Diagn Pathol. 9:1962014.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Preat F, Simon P and Noel JC: Differences
in breast carcinoma immunohistochemical subtypes between immigrant
Arab and European women. Diagn Pathol. 9:262014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Noel JC, Fayt I and Buxant F:
Proliferating activity in paget disease of the nipple. Pathol Oncol
Res. 16:7–10. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Allred DC, Harvey JM, Berardo M and Clark
GM: Prognostic and predictive factors in breast cancer by
immunohistochemical analysis. Mod Pathol. 11:155–168.
1998.PubMed/NCBI
|
|
17
|
Senkus E, Kyriakides S, Ohno S,
PenaultLlorca F, Poortmans P, Rutgers E, Zackrisson S and Cardoso
F: ESMO Guidelines Committee: Primary breast cancer: ESMO clinical
practice guidelines for diagnosis, treatment and follow-up. Ann
Oncol. 26(Suppl 5): v8–v30. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
MitreAguilar IB, CabreraQuintero AJ and
Zentella-Dehesa A: Genomic and non-genomic effects of
glucocorticoids: Implications for breast cancer. Int J Clin Exp
Pathol. 8:1–10. 2015.PubMed/NCBI
|
|
19
|
Matthews LC, Berry AA, Morgan DJ, Poolman
TM, Bauer K, Kramer F, Spiller DG, Richardson RV, Chapman KE,
Farrow SN, et al: Glucocorticoid receptor regulates accurate
chromosome segregation and is associated with malignancy. Proc Natl
Acad Sci USA. 112:5479–5484. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rønnov-Jessen L and Bissell MJ: Breast
cancer by proxy: Can the microenvironment be both the cause and
consequence? Trends Mol Med. 15:5–13. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Karagiannis GS, Poutahidis T, Erdman SE,
Kirsch R, Riddell RH and Diamandis EP: Cancer-associated
fibroblasts drive the progression of metastasis through both
paracrine and mechanical pressure on cancer tissue. Mol Cancer Res.
10:1403–1418. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Powell DW: Myofibroblasts: Paracrine cells
important in health and disease. Trans Am Clin Climatol Assoc.
111:271–292; discussion 292-293. 2000.PubMed/NCBI
|
|
23
|
Hinz B, Phan SH, Thannickal VJ, Prunotto
M, Desmoulière A, Varga J, De Wever O, Mareel M and Gabbiani G:
Recent developments in myofibroblast biology: Paradigms for
connective tissue remodeling. Am J Pathol. 180:1340–1355. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Grose R and Werner S, Kessler D,
Tuckermann J, Huggel K, Durka S, Reichardt HM and Werner S: A role
for endogenous glucocorticoids in wound repair. EMBO Rep.
3:575–582. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Drebert Z, Bracke M and Beck IM:
Glucocorticoids and the non-steroidal selective glucocorticoid
receptor modulator, compound A, differentially affect colon
cancer-derived myofibroblasts. J Steroid Biochem Mol Biol.
149:92–105. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Michael YL, Carlson NE, Chlebowski RT,
Aickin M, Weihs KL, Ockene JK, Bowen DJ and Ritenbaugh C: Influence
of stressors on breast cancer incidence in the Women's Health
Initiative. Health Psychol. 28:137–146. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Antonova L, Aronson K and Mueller CR:
Stress and breast cancer: From epidemiology to molecular biology.
Breast Cancer Res. 13:2082011. View
Article : Google Scholar : PubMed/NCBI
|