Introduction
Angiosarcoma is a rare aggressive malignant tumor of
vascular endothelial cells accounting for ~2% of soft tissue
sarcomas (1,2). Two-thirds of angiosarcomas occur in
cutaneous tissues (1), insidiously
presenting as a bruise-like lesion or a purplish papule and
exhibiting a tendency to spread hematogenously. One-third of
cutaneous angiosarcomas develop in the scalp of elderly individuals
(3).
The survival rate of angiosarcoma of the scalp and
face has been reported to be as low as 15–45% at 5 years (4–6). It has
been previously reported that a clear surgical margin is a major
factor affecting survival (6).
However, angiosarcoma invades extensively and is usually found
beyond the surgical margin (5,6). Thus,
adjuvant postoperative radiotherapy has been employed to improve
the outcome (4–6).
Various techniques of external beam radiotherapy
have been applied for angiosarcoma of the scalp and face. However,
despite the advances in external radiotherapy, due to the complex
shape of the head, the dose distribution is usually inadequate. The
build-up effect of high-energy photons or electrons reduces the
radiation dose delivered to the scalp surface. In brachytherapy,
the radiation dose is inversely proportional to the distance from
the radiation source; thus, theoretically higher doses are
delivered to the scalp surface and lower doses to the deep-seated
area compared with external beam radiotherapy. This means that
brachytherapy may be an optimal treatment modality for angiosarcoma
of the scalp. Thus, Imai et al introduced brachytherapy with
192Ir for angiosarcoma of the scalp, and reported that 3
patients were successfully treated using this method (7). However, there is little information on
clinical outcomes and precise dose-volume evaluation of
brachytherapy for angiosarcoma of the scalp. Thus, the aim of the
present study was to evaluate the local control and overall
survival of patients with angiosarcoma of the scalp and face who
underwent brachytherapy, and determine the optimal dose volume of
brachytherapy.
Patients and methods
Patients and methods
Between November, 2009 and January, 2015, 9
consecutive patients with angiosarcoma of the scalp and/or face who
received image-guided brachytherapy at the Tokyo Medical University
Hospital (Tokyo, Japan) were retrospectively evaluated. The
characteristics of the patients are summarized in Table I. All the patients had histologically
proven angiosarcoma. The median age was 83.4 years (range,
67.7–91.9 years) and the main tumor locations were as follows:
Parietal area in 7 patients, temporal area in 1 and occipital area
in 1 patient; 3 patients had tumors in multiple sites, and 6 had
tumors in a single location on the scalp area. Of the 9 patients, 8
had tumors in the scalp area and 1 had a tumor in the facial area;
1 patient also had metastasis to the cervical lymph nodes.
 | Table I.Patient characteristics and treatment
methods. |
Table I.
Patient characteristics and treatment
methods.
|
|
|
|
|
|
|
|
| Brachytherapy |
|
|
|
|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
|---|
| Patient no. | Gender | Age (years) | Main primary
site | No. of lesions | Tumor size (small,
<30 mm; large, >50 mm) | Other metastatic
sites | Limited surgery | Dose, Gy | Fractions, no. | Electron boost | IL-2 | Chemotherapy |
|---|
| 1 | F | 69.8 | Occipital | Multiple | Small | None | None | 60 | 20 |
| Local+arterial | None |
| 2 | F | 67.7 | Parietal | Single | Middle | None | Done | 48 | 16 | 12 Gr/6
fractions | None | None |
| 3 | F | 83.4 | Parietal | Single | Small | None | None | 60 | 20 |
| None | None |
| 4 | M | 91.9 | Temporal | Multiple | Small | None | None | 45 | 15 |
| Local | None |
| 5 | M | 81.3 | Parietal | Multiple | Small | Cervical lymph
nodes | None | 60 | 20 |
| Local | None |
| 6 | M | 91.7 | Parietal | Single | Middle | None | None | 60 | 20 |
| Local | None |
| 7 | M | 84.2 | Parietal | Single | Middle | None | None | 45 | 15 |
| Local | None |
| 8 | M | 88.4 | Parietal | Single | Large | None | None | 45 | 15 |
| Local | Paclitaxel |
| 9 | M | 80.9 | Parietal | Single | Small | None | Done | 45 | 15 |
| Local | None |
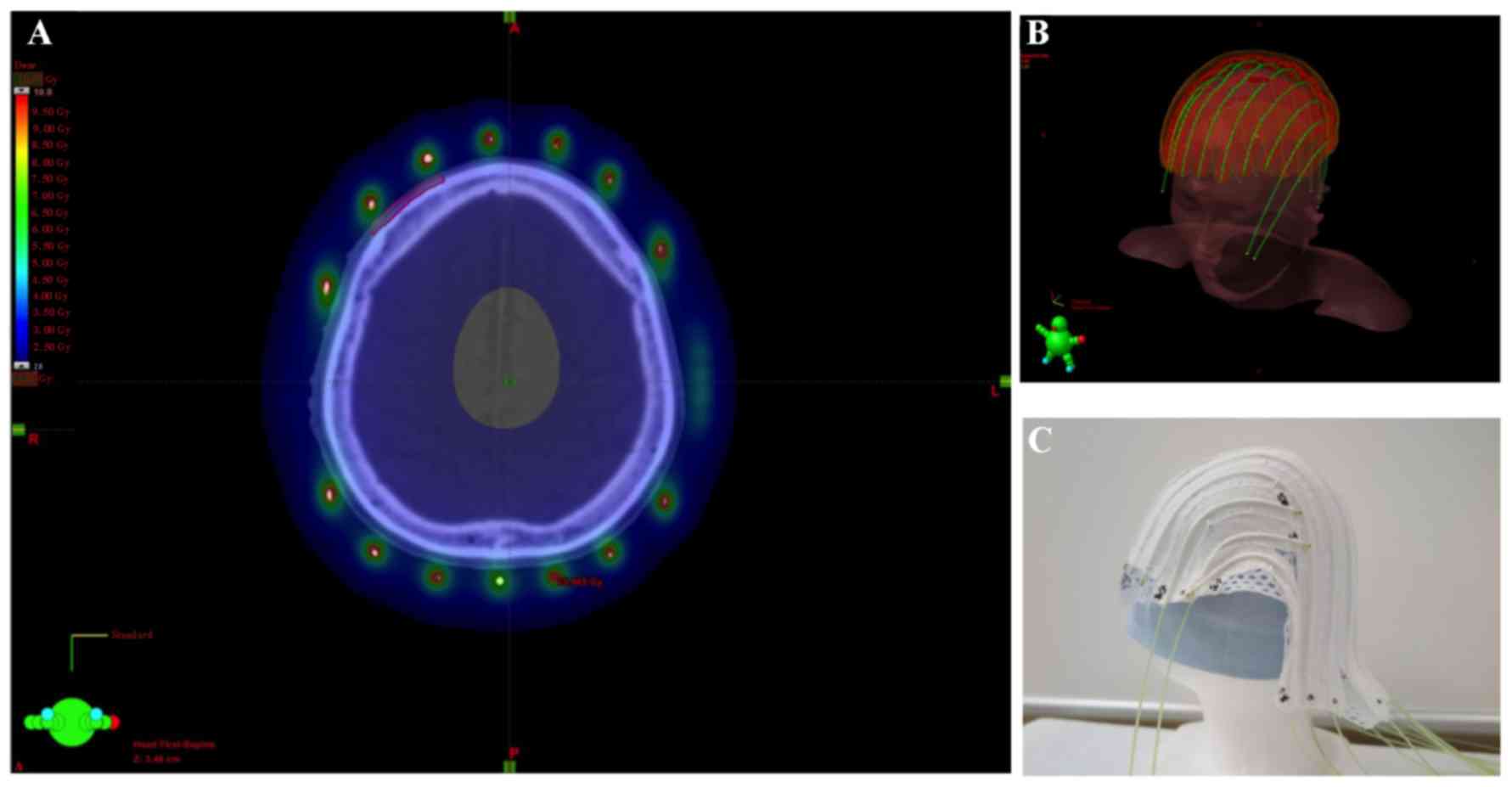
The patients were immobilized with a custom-made
thermoplastic mask. Applicators were fixed to avoid displacement of
the mask using cotton fabric tapes at intervals of 2 cm on the
outer side (Fig. 1), except for the
applicators of the first patient, which were fixed on the inner
side of the mask.
A visible lesion of angiosarcoma was defined as the
gross tumor volume (GTV); the clinical target volume (CTV) was
determined as the GTV plus an adequate margin along the skin and a
1–2-mm margin perpendicular to its axis. The planning target volume
(PTV) was defined as the CTV with a 10-mm margin along the skin and
a 1–2-mm margin perpendicular to its axis. The skull, cerebrum and
cerebellum were also delineated. All the patients underwent
image-guided brachytherapy based on computed tomography (CT)
images. The arrangement of radiation sources of 192Ir
was adequately placed to cover the entire GTV within the line of 3
Gy, and PTV within the line of 2 Gy. Heterogeneity of dose
distribution to the scalp was geometrically optimized using a
brachytherapy 3-dimensional planning system (BrachyVision; Varian
Medical Systems Inc., Palo Alto, CA, USA). Radiation sources of
192Ir were placed through the applicator using the
VariSource afterloading system (Varian Medical Systems).
The patients were irradiated with a dose of 3 Gy
three times per week for varying lengths of time. A total of 4
patients received a total dose of 60 Gy, 1 received 48 Gy and 4
patients received 45 Gy. The patient who received 48 Gy also
underwent additional electron therapy of 16 Gy in 8 fractions. The
equivalent dose delivered at 2.0 Gy per fraction was calculated by
the linear-quadratic model with α/β ratios of 10 (8). The cervical lymph node metastasis of
the patient was additionally treated by external radiotherapy at a
dose of 50 Gy in 25 fractions.
Two patients underwent limited surgery prior to
radiotherapy and 7 patients underwent recombinant interleukin-2
(rIL-2) immunotherapy; of those 7 patients, 6 received a local
injection and 1 was treated with both local and arterial
injections. One patient received sequential chemotherapy with
paclitaxel following completion of radiotherapy: Paclitaxel at a
dose of 100 mg/m2 was administered once per week;
however, after two administrations, paclitaxel was discontinued due
to cholangitis.
Follow-up was principally performed by
dermatologists every 3 months. The adverse events were assessed
according to the Common Terminology Criteria for Adverse Events,
version 4.0 (https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE
_4.03_2010-06-14_QuickReference_5×7.pdf).
Dose evaluation
Doses of 100, 95, 90, 85, 80, 70, 60, 50, 10 and 5%
of the PTV and the maximal doses were evaluated in the skull,
cerebrum and cerebellum using the BrachyVision brachytherapy
3-dimensional planning system (Varian Medical Systems Inc.). The
data were used for necessary dose analysis.
Statistical analysis
The overall survival time was calculated from the
first day of brachytherapy to the date of death from any cause.
Hospital visits provided data on surviving patients. The
progression-free survival time was calculated from the first day of
brachytherapy to the date of the first relapse at any site or the
date of death. The local progression-free rate was calculated
according to to the date of relapse in irradiated sites. Survival
time was calculated using the Kaplan-Meier method and the
difference was compared using the log-rank test. Paired variables
were compared by the Wilcoxon matched-paired signed-rank test.
Stata statistical software, version 13 (StataCorp., College
Station, TX USA) was used. A probability value of <0.05 was
considered to be statistically significant.
Results
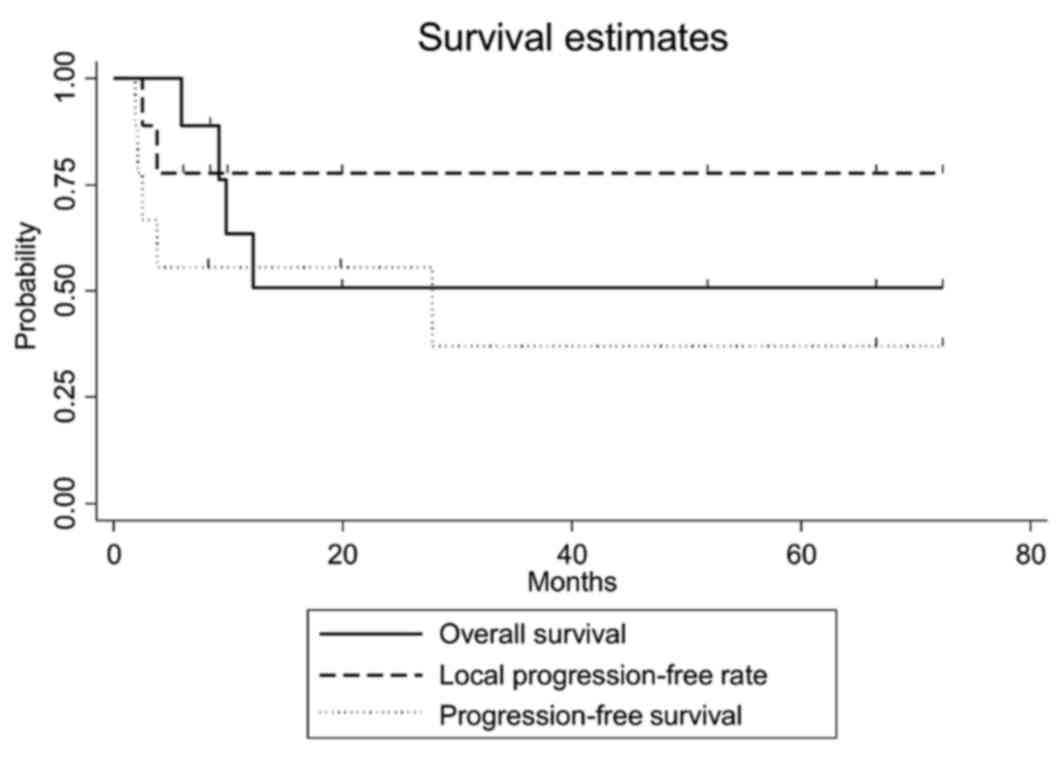
The clinical outcomes are presented in Table II. The median observation period was
12.2 months (range, 5.9–72.2 months). The overall survival,
progression-free survival and local progression-free rates at 3
years were 50.8% [95% confidence interval (CI): 15.6–78.1%], 37.0%
(95% CI: 6.8–69.3%), and 77.8% (95% CI: 36.5–93.9%), respectively
(Fig. 2). The equivalent
fractionated and total doses according to the biological
effectiveness for GTV and PTV are shown in Table III. Of the 9 patients, 2 developed
local infield recurrence within 4 months following completion of
brachytherapy with a radiation dose of 45 Gy in 15 fractions. Three
patients who were treated with a dose of ≥60 Gy had no local
recurrence and survived without late complications. The patient
with the cervical lymph node metastasis who underwent radiotherapy
of ≥60 Gy developed lung metastasis 2 months after the completion
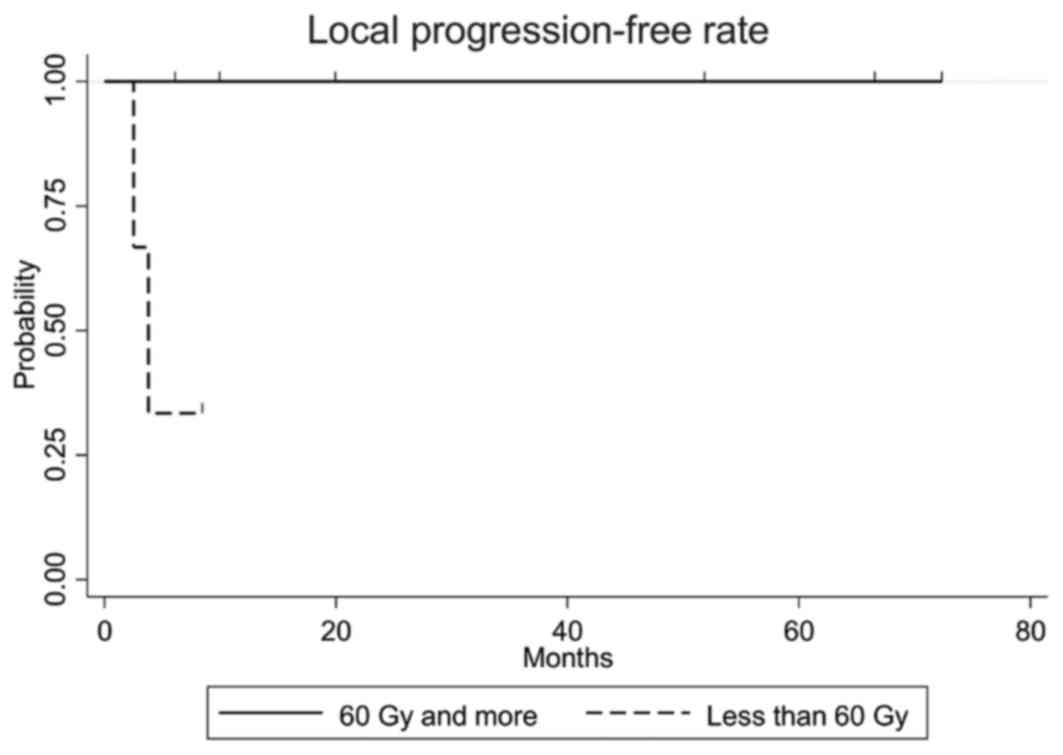
of radiotherapy. The local progression-free rate in patients who
received a dose of ≥60 Gy was statistically significantly higher
compared with that in patients who received a dose of <60 Gy
(P=0.027, Fig. 3). Of the 2 patients
who experienced recurrence in the forehead outside the irradiated
area, 1 was successfully treated by local injection of rIL-2, but
the other patient developed bone metastasis. Distant metastases
were observed in the bone, lung, or liver, and the patient with
lung metastasis had developed cervical lymph node metastasis prior
to brachytherapy. The dose-volume for organs at risk is shown in
Table IV. The maximum doses to the
skull, cerebrum and cerebellum were statistically significantly
lower compared with GTV (P<0.01).
 | Table II.Clinical outcomes. |
Table II.
Clinical outcomes.
|
|
|
|
|
|
|
|
| Adverse events,
no. |
|---|
| Patient no. | Local PFS
(months) | Local recurrence
site | PFS (months) | Regional recurrence
site | Distant metastatic
site | OS (months) | Final status | Dermatitis | Alopecia |
|---|
| 1 | 72.2 | None | 72.2 | None | None | 72.2 | Alive | 2 | 2 |
| 2 | 66.4 | None | 66.4 | None | None | 66.4 | Alive | 3 | 2 |
| 3 | 51.7 | None | 27.8 | Forehead | None | 51.7 | Alive | 2 | 2 |
| 4 | 8.3 | None | 8.3 | None | None | 8.3 | Alive | 2 | 0 |
| 5 | 5.9 | None | 1.9 | None | Lung | 5.9 | Deceased | 3 | 2 |
| 6 | 19.8 | None | 19.8 | None | None | 19.8 | Alive | 2 | 2 |
| 7 | 2.5 | Parietal | 2.5 | Forehead | Bone | 12.2 | Deceased | 2 | 2 |
| 8 | 9.8 | None | 2.1 | None | Liver | 9.8 | Deceased | 2 | 2 |
| 9 | 3.8 | Parietal | 9.2 | None | None | 9.2 | Deceased | 2 | 2 |
 | Table III.Doses of target volume. |
Table III.
Doses of target volume.
|
|
|
| GTV |
|
|
|
| PTV |
|
|
|---|
|
|
|
|
|
|
|
|
|
|
|
|
|---|
| Patient no. | D100 | D95 | D90 | D85 | D80 | D70 | D60 | D50 | D10 | D5 | D100 | D95 | D90 | D85 | D80 | D70 | D60 | D50 | D10 | D5 |
|---|
| 1 | 2.7/58.2 | 3.0/64.2 | 3.1/66.6 | 3.1/68.0 | 3.2/70.1 | 3.3/73.2 | 3.4/76.5 | 3.6/82.2 | 5.2/132.1 | 5.9/155.3 | 0.7/12.5 | 2.2/44.7 | 2.5/52.1 | 2.7/57.2 | 2.8/59.3 | 3.0/65.0 | 3.1/67.7 | 3.3/73.2 | 4.9/121.7 | 5.8/152.7 |
| 2 | 2.7/58.3 | 2.8/59.2 | 2.8/59.6 | 2.8/60.0 | 2.8/60.2 | 2.9/60.8 | 2.9/61.3 | 2.9/61.7 | 3.0/63.8 | 3.0/64.6 | 1.6/36.8 | 2.4/51.7 | 2.6/55.7 | 2.8/59.8 | 2.8/59.8 | 3.0/64.0 | 3.1/66.2 | 3.2/66.3 | 4.0/86.7 | 4.5/99.0 |
| 3 | 2.6/54.6 | 2.7/56.1 | 2.7/56.6 | 2.7/56.9 | 2.7/57.4 | 2.7/57.9 | 2.8/58.4 | 2.8/59.0 | 2.9/62.6 | 2.9/63.4 | 1.4/26.6 | 2.2/44.7 | 2.4/49.6 | 2.5/52.1 | 2.6/54.6 | 2.7/57.2 | 2.8/59.7 | 2.9/62.4 | 3.6/81.6 | 4.0/93.3 |
| 4 | 2.3/34.8 | 2.5/39.1 | 2.6/40.4 | 2.6/41.5 | 2.7/42.7 | 2.8/44.4 | 2.9/45.8 | 2.9/47.4 | 3.3/55.3 | 3.4/57.6 | 0.3/3.9 | 1.8/26.6 | 1.9/28.3 | 2.0/30.0 | 2.1/31.8 | 2.3/35.4 | 2.5/39.1 | 2.7/42.9 | 3.8/65.6 | 4.3/76.9 |
| 5 | 2.6/54.1 | 2.7/56.1 | 2.7/56.9 | 2.7/57.4 | 2.7/57.9 | 2.8/59.0 | 2.8/59.7 | 2.8/60.5 | 3.0/64.5 | 3.0/65.5 | 1.2/22.4 | 2.2/44.7 | 2.5/52.1 | 2.6/54.6 | 2.7/57.2 | 2.9/62.4 | 3.0/65.0 | 3.1/67.7 | 3.9/60.4 | 4.3/102.5 |
| 6 | 2.8/60.3 | 3.0/64.2 | 3.0/65.5 | 3.1/66.6 | 3.1/67.7 | 3.2/69.6 | 3.3/72.1 | 3.4/74.5 | 3.8/87.7 | 4.0/92.7 | 1.4/26.6 | 2.1/42.4 | 2.4/49.6 | 2.5/52.1 | 2.7/57.2 | 3.0/65.0 | 3.1/67.7 | 3.3/73.2 | 4.1/96.4 | 4.6/111.9 |
| 7 | 2.2/33.4 | 2.5/38.1 | 2.5/39.6 | 2.6/40.6 | 2.6/41.3 | 2.7/42.5 | 2.7/43.6 | 2.8/44.8 | 3.3/54.0 | 3.5/58.2 | 0.9/12.3 | 2.1/31.8 | 2.3/35.4 | 2.5/39.1 | 2.6/41.0 | 2.7/42.9 | 2.8/44.8 | 2.9/46.8 | 3.7/63.4 | 4.2/74.6 |
| 8 | 2.9/46.8 | 3.1/50.0 | 3.1/50.8 | 3.1/51.6 | 3.2/52.4 | 3.3/53.8 | 3.3/55.5 | 3.4/57.0 | 4.0/68.9 | 4.1/73.2 | 1.3/18.4 | 2.0/30.0 | 2.2/33.6 | 2.4/37.2 | 2.5/39.1 | 2.7/42.9 | 2.8/44.8 | 2.9/46.8 | 3.8/65.6 | 4.3/76.9 |
| 9 | 2.6/40.4 | 2.6/41.3 | 2.6/41.7 | 2.7/41.9 | 2.7/42.1 | 2.7/42.7 | 2.7/43.4 | 2.8/44.0 | 2.9/47.6 | 2.9/47.0 | 1.2/16.8 | 2.2/33.6 | 2.5/39.1 | 2.6/41.0 | 2.7/42.9 | 2.8/44.8 | 2.9/46.8 | 3.0/48.8 | 3.8/65.6 | 4.3/76.9 |
 | Table IV.Dosimetric comparison between PTV and
organ at risk. |
Table IV.
Dosimetric comparison between PTV and
organ at risk.
| Gy (mean ± SD) | D95 | D90 | D85 | D80 | D70 | D60 | D50 | D10 | D5 | Max |
|---|
| PTV | 2.13±0.17 | 2.37±0.21 | 2.51±0.23 | 2.61±0.21 | 2.79±0.23 | 2.90±0.20 | 3.03±0.21 | 3.96±0.38 | 4.48±0.52 | 28.3±11.2 |
| Cerebrum |
0.77±0.37a |
0.84±0.40a |
0.92±0.44b |
1.01±0.45a |
1.14±0.48a |
1.27±0.49a |
1.39±0.49a |
2.07±0.37a |
2.21±0.31a |
2.81±0.25a |
| Cerebellum |
0.88±0.49a |
0.91±0.51a |
0.93±0.52a |
0.98±0.53a |
1.02±0.56a |
1.06±0.57a |
1.11±0.58a |
1.44±0.69a |
1.50±0.72a |
1.79±0.83a |
| Skull |
1.79±0.27a |
2.02±0.23b |
2.14±0.20b |
2.23±0.21b |
2.39±0.20b |
2.47±0.17b |
2.57±0.19b |
2.99±0.26a |
3.12±0.29a |
4.77±1.63a |
As regards adverse events, all the patients had
grade 2 alopecia; 7 and 2 patients had grade 2 and 3 radiation
dermatitis, respectively; none experienced grade ≥4 adverse
events.
Discussion
Angiosarcoma arising from endothelial cells is known
to have a propensity to spread hematologenously. A report of 95
autopsies in Japanese patients demonstrated that the most common
sites of metastasis were the lung, bone and liver (9). In this study, 3 patients experienced
hematologenous metastasis. By contrast, 4 of the 9 patients treated
by brachytherapy exhibited prolonged survival without local or
distant metastasis at a median of 38.4 months. Guadagnolo et
al reported that approximately two-thirds of the patients
experienced local recurrence in an analysis of 70 patients with
angiosarcoma, and survival time was short following relapse. The
authors denoted that local recurrence was associated with a high
mortality rate and that local disease control was crucial (4).
Reports on angiosarcoma of the scalp and face
included a variety of treatments. Several reports demonstrated that
the addition of radiotherapy following surgery was the optimal
approach to achieving local control and improving survival
(4–6). Patel et al demonstrated that
surgery followed by radiotherapy improved local control in a series
of 55 patients (5). Pawlik et
al also reported that the addition of radiotherapy was a
prognostic factor for overall survival, along with age and T stage
in an analysis of 29 patients (6);
they demonstrated that radiotherapy significantly decreased the
mortality risk (hazard ratio=0.16; P<0.01) using a multivariate
analysis.
As regards other prognostic factors, Abraham et
al reported that surgical margin negativity was a significant
prognostic factor (3), whereas
others were not (4–6). Pawlik et al employed
radiotherapy, although no tumors were detected beyond the surgical
margin (6). As for reports from the
United States, surgery was the first option and radiotherapy was
secondary; whereas in a Japanese study, 42–100% of the patients
underwent radiotherapy alone (10–14), in
the United States study only 7–39% underwent radiotherapy alone
(3–6). Regarding the local control rates,
44–67% was reported by Japanese studies (10,12–14) and
it was comparable to 47–52% reported by studies from the United
States (5,6,12). These
results suggested that surgery may not be required for angiosarcoma
of the face and scalp.
In the present study, a dose of ≥60 Gy achieved
significantly better local control compared with a dose of <60
Gy. Of the 9 patients who underwent brachytherapy of ≥60 Gy at the
50% dose (D50%), 3 survived. Hata et al also reported
results with definitive external radiotherapy for 17 patients
(10). All the tumors regressed with
radiotherapy; however, 2 patients had a local relapse and 5
relapsed at other sites of the scalp. The authors reported that a
dose of 50–60 Gy at 2 Gy per fraction attained local control when
the tumor did not invade deeper tissues. Ogawa et al
reported clinical results from 14 patients treated with definitive
radiotherapy: The 10 patients who received a dose of ≥70 Gy did not
develop local relapse, but 4 patients who received a dose of <70
Gy relapsed. Of the 11 patients treated with surgery followed by
radiotherapy, none relapsed with a dose of ≥60 Gy, but 2 relapsed
when a dose of <60 Gy was administered (12). Based on those results and ours
combined, a brachytherapy dose of 60 Gy may be adequate to obtain
good local control for angiosarcoma.
Various external radiotherapy techniques have
applied homogenous irradiation to the surface of the scalp. Able
et al reported a technique of six-field electron therapy to
the entire scalp (15). Akazawa
et al introduced a parallel-opposed lateral field method
using both photons and electrons (16), which was verified by Tung et
al (17). Kinard et al
reported a technique using four-arc photons (18). Those reports were technical, or
included only a paucity of clinical data. Ostheimer et al
described the doses to the target and organs at risk in clinical
cases for intensity-modulated radiotherapy for the scalp (19); they reported that the mean and
maximum brain doses ranged from 43 to 51% and from 101 to 109%,
respectively, compared with the prescribed dose. These doses were
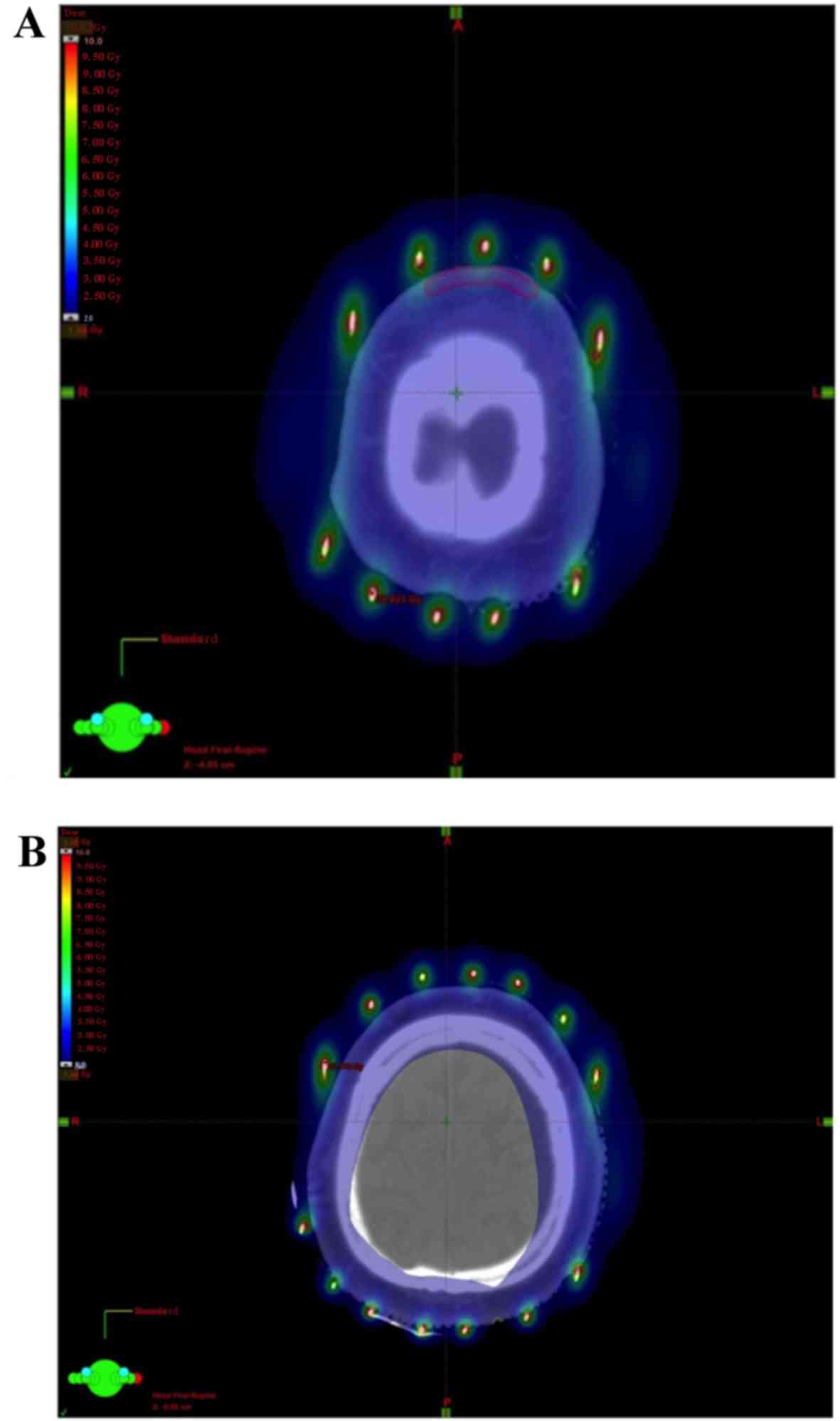
higher compared with those in our methods. In external photon
therapy, an adequate bolus may be required to increase the dose to
the skin surface due to the build-up phenomenon. In our
image-guided brachytherapy method, an adequate dose to the scalp
was feasible and the dose to the cerebrum was significantly reduced
(Fig. 4).
In this study, 1 patient with cervical lymph node
metastasis developed distant metastasis. Other reports also
demonstrated that patients with cervical lymph node metastasis are
prone to develop distant metastases (10,13).
Paclitaxel and doxorubicin have been used in such patients with a
curative intent (20). Sasaki et
al reported that the combination of rIL-2 and radiotherapy led
to prolonged survival (14). In our
study, recurrent lesions in the forehead were successfully treated
by local rIL-2 injections. The evidence base for the regimen of
chemotherapy is limited (2);
however, adding chemotherapy to radiotherapy may improve local
control and survival. The small number of patients in the present
study did not allow for clarification of prognostic factors, such
as combined chemotherapy, age and tumor size.
A frequent scalp relapse outside the irradiated
fields was reported, but a precise recurrence site has not been
described (5,10–12). Our
study demonstrated that 2 of the 9 patients relapsed on the
cutaneous area of the forehead, suggesting that the entire scalp
and forehead should be included in the irradiated volume.
In conclusion, brachytherapy of 60 Gy at the GTV
covering PTV within the 40-Gy line was an effective and less toxic
treatment method for angiosarcoma.
References
|
1
|
Penel N, Marréaud S, Robin YM and
Hohenberger P: Angiosarcoma: State of the art and perspectives.
Crit Rev Oncol Hematol. 80:257–263. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Young RJ, Brown NJ, Reed MW, Hughes D and
Woll PJ: Angiosarcoma. Lancet Oncol. 11:983–991. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Abraham JA, Hornicek FJ, Kaufman AM,
Harmon DC, Springfield DS, Raskin KA, Mankin HJ, Kirsch DG,
Rosenberg AE, Nielsen GP, et al: Treatment and outcome of 82
patients with angiosarcoma. Ann Surg Oncol. 14:1953–1967. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Guadagnolo BA, Zagars GK, Araujo D, Ravi
V, Shellenberger TD and Sturgis EM: Outcomes after definitive
treatment for cutaneous angiosarcoma of the face and scalp. Head
Neck. 33:661–667. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Patel SH, Hayden RE, Hinni ML, Wong WW,
Foote RL, Milani S, Wu Q, Ko SJ and Halyard MY: Angiosarcoma of the
scalp and face: The Mayo Clinic experience. JAMA Otolaryngol Head
Neck Surg. 141:335–340. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pawlik TM, Paulino AF, McGinn CJ, Baker
LH, Cohen DS, Morris JS, Rees R and Sondak VK: Cutaneous
angiosarcoma of the scalp: A multidisciplinary approach. Cancer.
98:1716–1726. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Imai M, Nishimura T, Nozue M, Suzuki K,
Kaneko M and Nimi M: The 192Ir surface-mold technique
for a whole scalp irradiation. J Jpn Soc Thre Radiol Oncol.
11:27–31. 1999.
|
|
8
|
Fowler JF: The linear-quadratic formula
and progress in fractionated radiotherapy. Br J Radiol. 62:679–694.
1989. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kitagawa M, Tanaka I, Takemura T,
Matsubara O and Kasuga T: Angiosarcoma of the scalp: Report of two
cases with fatal pulmonary complications and a review of Japanese
autopsy registry data. Virchows Arch A Pathol Anat Histopathol.
412:83–87. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hata M, Wada H, Ogino I, Omura M, Koike I,
Tayama Y, Odagiri K, Kasuya T and Inoue T: Radiation therapy for
angiosarcoma of the scalp: Treatment outcomes of total scalp
irradiation with X-rays and electrons. Strahlenther Onkol.
190:899–904. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Miki Y, Tada T, Kamo R, Hosono MN, Tamiya
H, Shimatani Y, Tsutsumi S, Ogino R and Miki Y: Single
institutional experience of the treatment of angiosarcoma of the
face and scalp. Br J Radiol. 86:201304392013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ogawa K, Takahashi K, Asato Y, Yamamoto Y,
Taira K, Matori S, Iraha S, Yagi N, Yogi A and Haranaga S:
Treatment and prognosis of angiosarcoma of the scalp and face: A
retrospective analysis of 48 patients. Br J Radiol. 85:e1127–e1133.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ohguri T, Imada H, Nomoto S, Yahara K,
Hisaoka M, Hashimoto H, Tokura Y, Nakamura K, Shioyama Y and Honda
H: Angiosarcoma of the scalp treated with curative radiotherapy
plus recombinant interleukin-2 immunotherapy. Int J Radiat Oncol
Biol Phys. 61:1446–1453. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sasaki R, Soejima T, Kishi K, Imajo Y,
Hirota S, Kamikonya N, Murakami M, Kawabe T, Ejima Y, Matsumoto A
and Sugimura K: Angiosarcoma treated with radiotherapy: Impact of
tumor type and size on outcome. Int J Radiat Oncol Biol Phys.
52:1032–1040. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Able CM, Mills MD, McNeese MD and Hogstrom
KR: Evaluation of a total scalp electron irradiation technique. Int
J Radiat Oncol Biol Phys. 21:1063–1072. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Akazawa C: Treatment of the scalp using
photon and electron beams. Med Dosim. 14:129–131. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tung SS, Shiu AS, Starkschall G, Morrison
WH and Hogstrom KR: Dosimetric evaluation of total scalp
irradiation using a lateral electron-photon technique. Int J Radiat
Oncol Biol Phys. 27:153–160. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kinard JD, Zwicker RD, Schmidt-Ullrich RK,
Kaufman N and Pieters R: Short communication: Total craniofacial
photon shell technique for radiotherapy of extensive angiosarcomas
of the head. Br J Radiol. 69:351–355. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ostheimer C, Janich M, Hübsch P, Gerlach R
and Vordermark D: The treatment of extensive scalp lesions using
coplanar and non-coplanar photon IMRT: A single institution
experience. Radiat Oncol. 9:822014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Italiano A, Cioffi A, Penel N, Levra MG,
Delcambre C, Kalbacher E, Chevreau C, Bertucci F, Isambert N, Blay
JY, et al: Comparison of doxorubicin and weekly paclitaxel efficacy
in metastatic angiosarcomas. Cancer. 118:3330–3336. 2012.
View Article : Google Scholar : PubMed/NCBI
|