Introduction
Mucinous tubular and spindle cell carcinoma (MTSCC)
is a rare subtype of renal cell carcinoma (RCC) introduced in the
World Health Organization (WHO) classification system in 2004
(1) and included in the recent
version of 2016 (2). Based on
clinical characteristics, MTSCC predominantly occurs in middle-aged
to elderly women (1:4 male-to-female ratio) (3,4). At
present, fewer than 100 cases of MTSCC have been reported according
to a recent review (5). MTSCC
characteristically comprises elongated tubules lined by low
cuboidal epithelium and fascicles of spindle cells with a stroma
containing mucinous substance (4).
According to previous reports, MTSCC is subclassified into 2
histological types: classic and mucin-poor (6,7).
Initially, MTSCC was considered to be derived from the distal
nephron (8,9). However, recent examinations revealed
significant morphological and immunohistochemical overlaps between
MTSCC and papillary renal cell carcinoma (PRCC); the latter
possesses characteristics similar to proximal tubular epithelia.
The cellular origin of MTSCC, thus, remains to be elucidated
(10).
Although MTSCC has been described as a low-grade and
relatively indolent tumor in the WHO2004 classification, there are
several case reports on aggressive MTSCC, showing high-grade
transformation (11–14). In the WHO 2016 classification, the
description, presented in 2004, that the MTSCC version is indolent
has been eliminated. In the present study, we report 2 cases of
aggressive MTSCCs. To understand the exact clinicopathological
characteristics of MTSCC, accumulation and comparison of aggressive
and indolent cases are required.
Case reports
Clinical results
Case 1
A 71-year-old male patient who presented with
hematuria and pyuria was admitted to our hospital. A computed
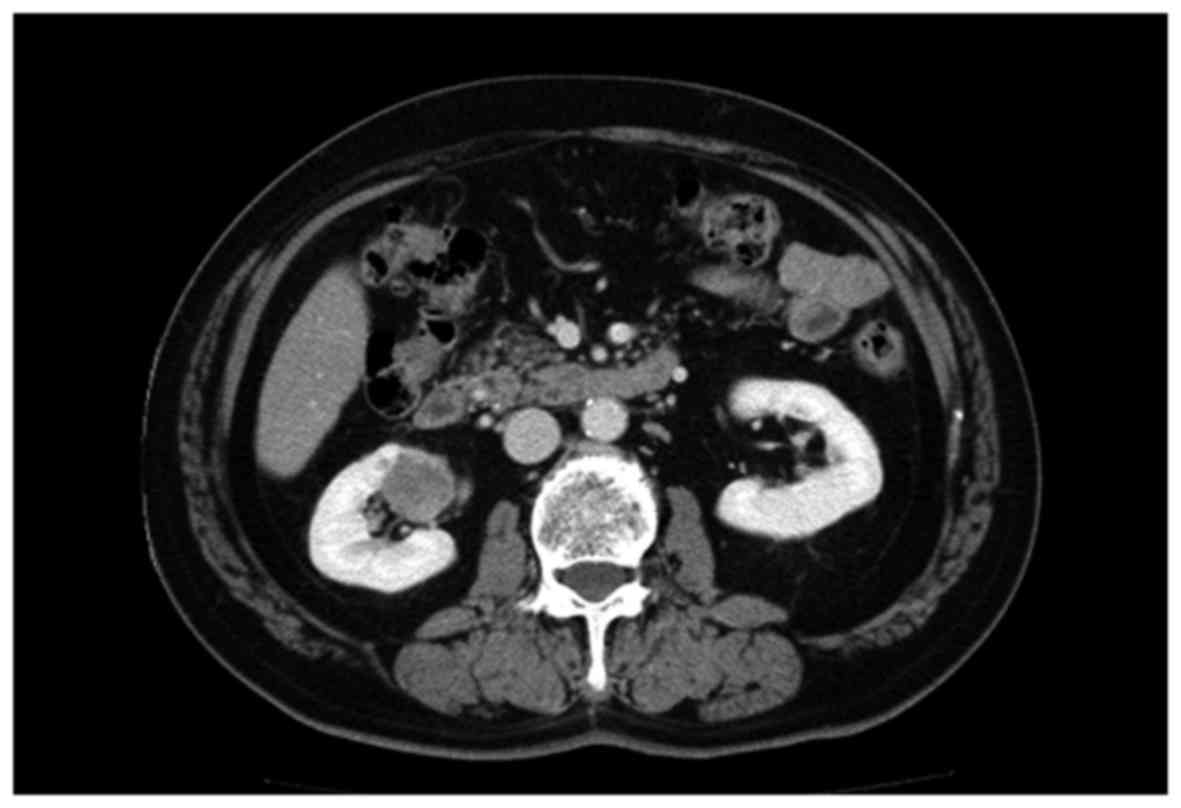
tomography (CT) scan revealed a mass lesion, measuring 30 mm in
diameter, in the lower pole of the right kidney (Fig. 1). Accordingly, partial nephrectomy
was performed. One month later, follow-up imaging revealed multiple
metastatic lesions in the abdominal paraaortic lymph nodes, pleura,
and bones (ribs and pubic bone); the left lung; and in segment 4 of
the liver. Although sunitinib, temsirolimus, and axitinib were
serially administrated along with palliative radiotherapy to the
left rib for pain control, therapeutic effects were limited and an
adverse effect, interstitial pneumonia, developed. Finally, the
patient died of respiratory failure due to the progression of the
disease, 2 years after the surgery. Autopsy was not performed.
Case 2
A 64-year-old male patient incidentally presented a
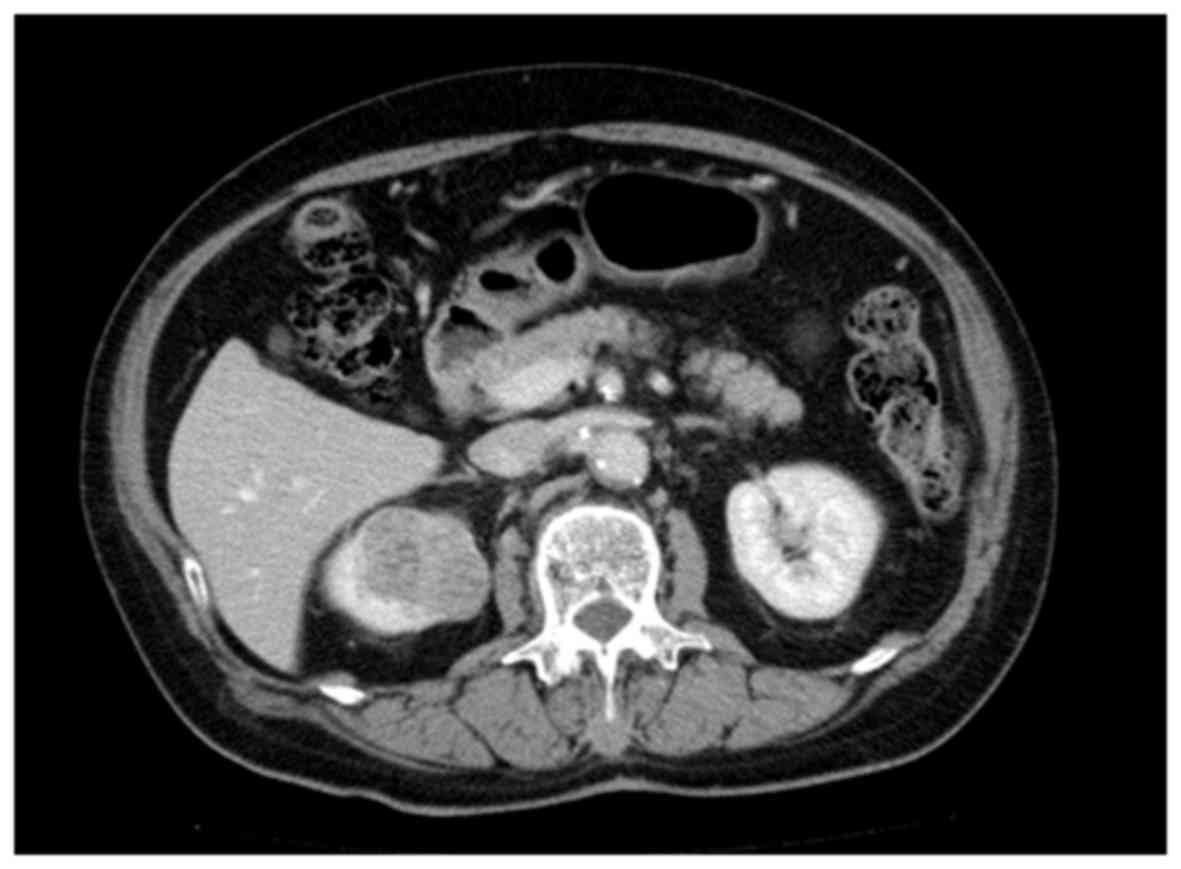
mass lesion in the upper pole of the right kidney, detected by an
abdominal CT scan performed during a routine health checkup
(Fig. 2). The patient underwent
radical nephrectomy with a clinical diagnosis of RCC. Six months
later, the tumor metastasized to the lungs and pleurae with
effusion. Despite additional chemotherapy, 9 months after the
surgery, the patient died of progressive disease and acute
respiratory failure. Autopsy was not performed.
General. Surgically resected specimens of the 2
cases were immediately fixed in 10% buffered formalin and were
embedded in paraffin. Paraffin sections (4 µm) were subjected to
histopathological staining. In addition, immunohistochemistry was
performed by standard methods using the antibodies listed in
Table I.
 | Table I.Immunohistochemistry results. |
Table I.
Immunohistochemistry results.
|
|
|
|
|
|
| Case 1 | Case 2 |
|---|
|
|
|
|
|
|
|
|
|
|---|
| Antibodies | Source | Clone | Dilution | Pretreatment | Normal kidney
positive ina | Conventional
area | High-grade area | Conventional
area | High-grade area |
|---|
| α-methylacyl- CoA
racemase | Dako | P504S | Prediluted | Heat treatment | PT | Positive | Positive | Positive | Positive |
| CD10 | Ventana | SP67 | Prediluted | Heat treatment | PT | Focally positive | Focally positive | Focally positive | Focally positive |
| CD15 | Ventana | MMA | Prediluted | Heat treatment | PT | Focally positive | Focally positive | Focally positive | Focally positive |
| CK, High molecular
weight | Dako | 34βE12 | 1:50 | Heat treatment | CD, PM | Negative | Negative | Negative | Negative |
| EMA | Dako | E29 | 1:200 | Heat treatment | DT, CD, DM | Focally positive | Negative | Focally positive | Focally positive |
| CK7 | Dako | OV-TL12/30 | 1:100 | Heat treatment | DT, CD, DM | Negative | Negative | Focally positive | Negative |
| E-cadherin | Ventana | 36 | Prediluted | Heat treatment | DT, CD, DM | Positive | Positive | Positive | Positive |
| Vimentin | Dako | vim3B4 | 1:200 | Heat treatment | Pod, PT | Negative | Negative | Negative | Negative |
| c-kit | Dako | polyclonal | 1:100 | Heat treatment |
| Negative | Negative | Negative | Negative |
| Ki67b | Dako | MIB1 | 1:50 | Heat treatment | Less than 1% | 4% | 34% | 7% | 34% |
| p53b | Dako | DO-7 | 1:100 | Heat treatment | Negative | 6% | 30% | 7% | 40% |
Pathological findings and
immunohistochemistry
Case 1
The resected kidney contained a poorly circumscribed
tumor extending into the medulla. The tumor was tan-yellow to white
and firm in cut surface, measuring 3.2×2.2×2.2 cm in size.
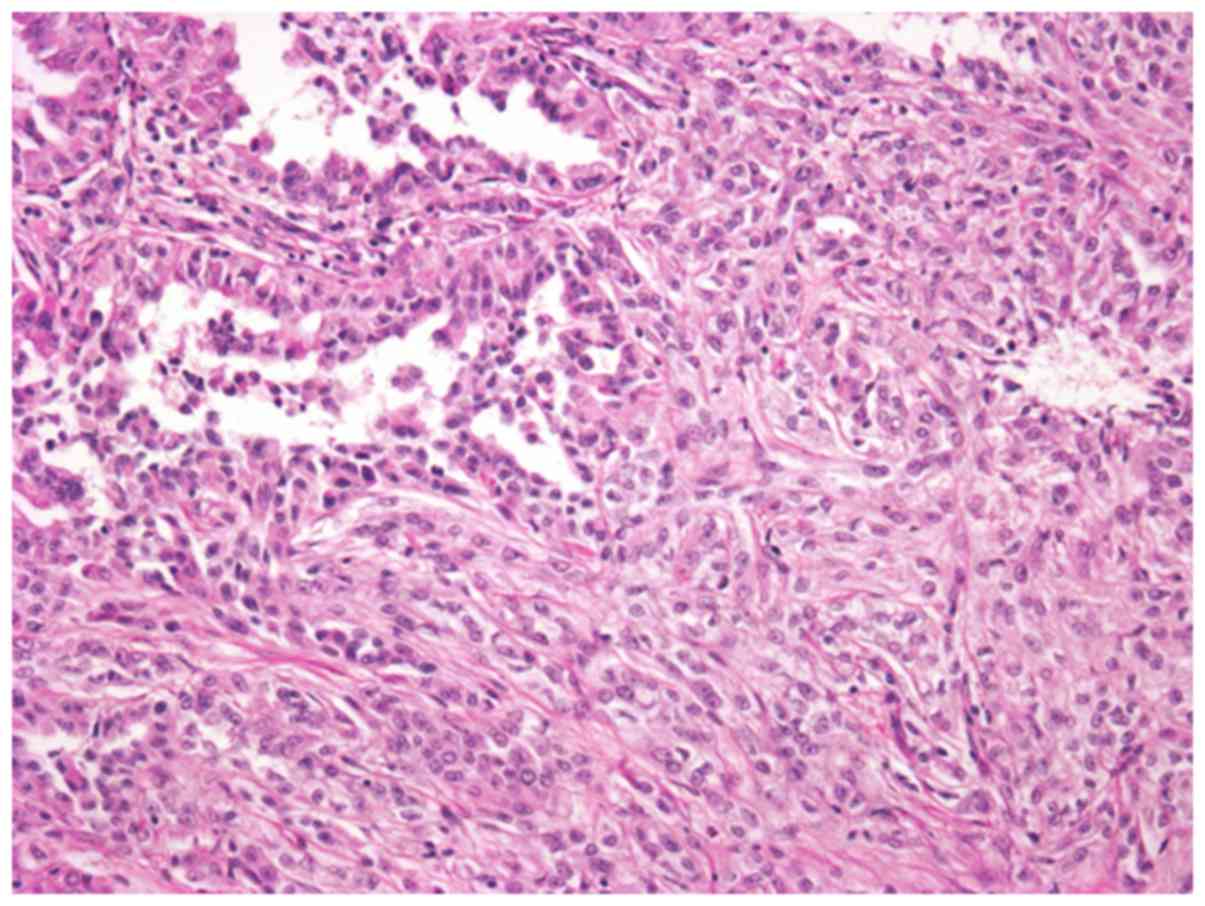
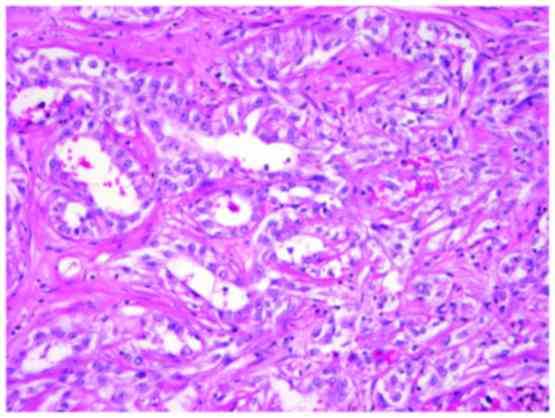
Microscopically, the tumor comprised slender and
elongated tubulopapillary structure lined by cuboidal epithelial
cells and fascicles of spindle cells. The tumor cells were small
and uniform with round and spindle-shaped nuclei, containing fine
chromatin and inconspicuous nucleoli (Fig. 3). The cytoplasm was pale to weakly
eosinophilic. Depletion in extracellular mucinous material was
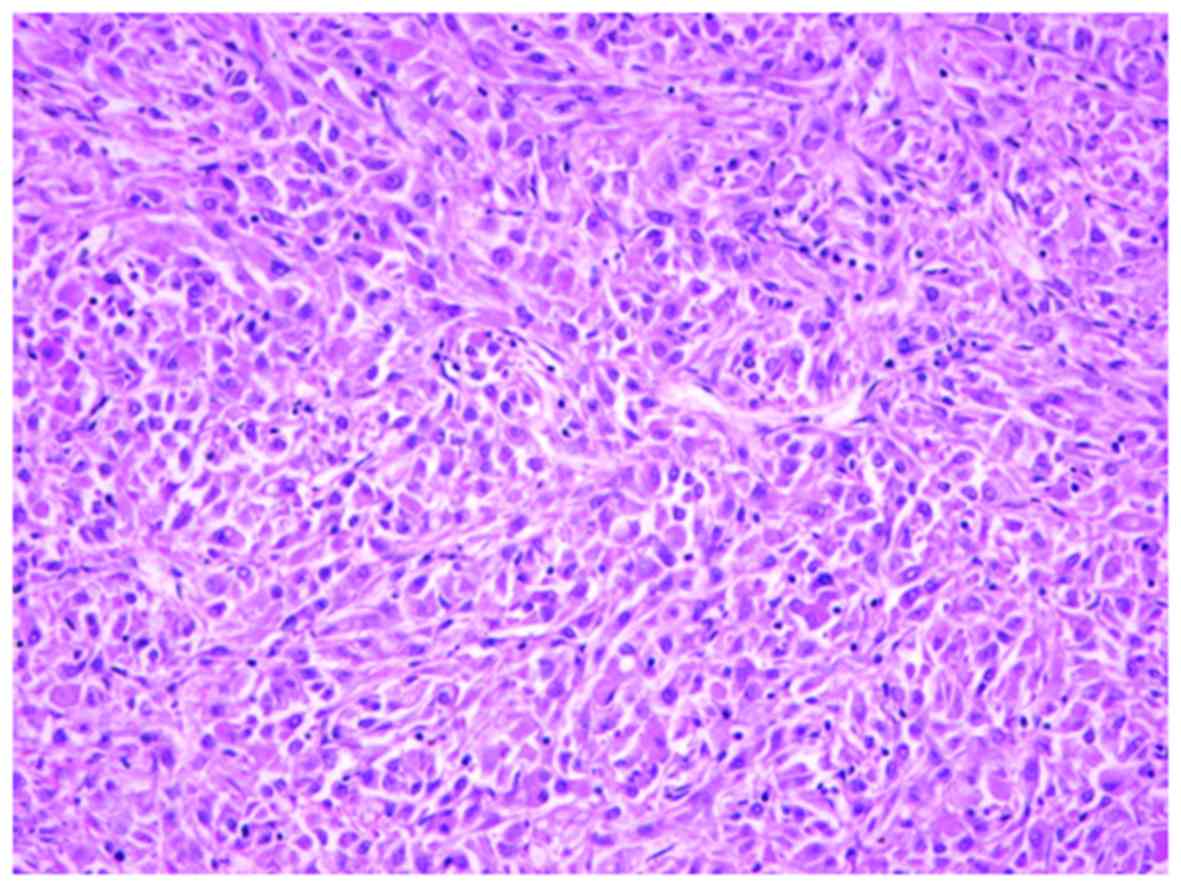
identified. In addition, the tumor exhibited occasional foci of
high-grade transformation, solid nest growth, and sheet-like growth
pattern (Fig. 4). Nuclei with a
higher grade contained prominent nucleoli. Precisely, the nuclear
grade of the tumor was classified as Fuhrman grade 3. The tumor
showed vascular invasion, but the surgical margin was negative.
However, necrosis and hemorrhage areas were not recognized.
By immunohistochemistry, the tumor cells were
positive for α-methyl acyl CoA racemase (AMACR), E-cadherin, and
CD10, CD15 and EMA were focally positive. However, the tumor cells
were negative for high molecular weight CK (34bE12), CK7, Vimenin,
and c-kit. In the conventional area, the Ki-67 labeling index was
7%, and the nuclear accumulation of p53 was focally observed. In
the high-grade area, the tumor cells demonstrated an identical
immunohistochemical reaction. However, the labeling Ki-67 labeling
index was 34%, and the p53 nuclear accumulation was markedly
increased.
Case 2
The resected kidney contained a tumor in the upper
pole, measuring 8.0×8.0×2.5 cm. The tumor showed an ill-defined
boundary with an invasive growth. The cut surface was solid,
tan-yellow to white with hemorrhage and necrosis, and firm in
consistence.
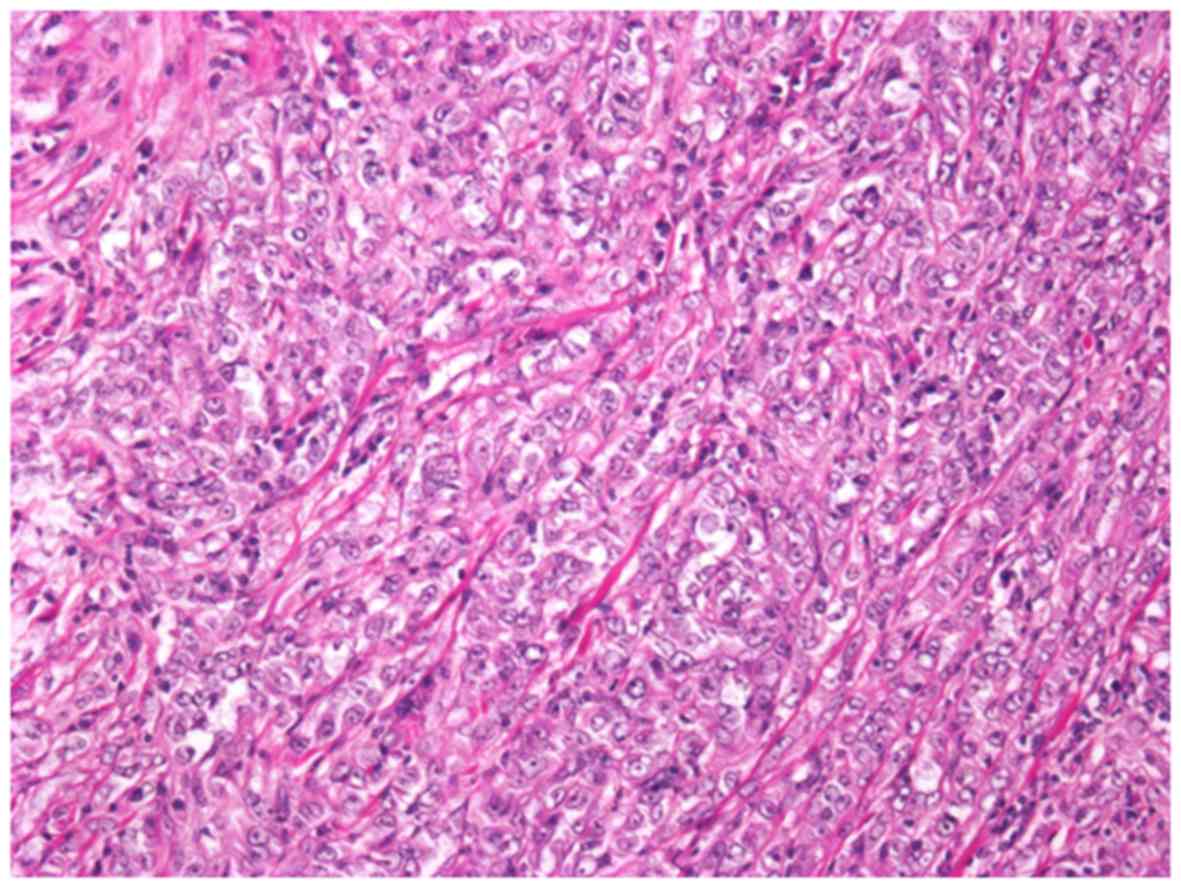
Although the predominant part of the tumor showed
high-grade morphology, conventional MTSCC histology was
occasionally identified (Fig. 5).
The tumor lacked mucinous substance in the stroma. The majority of
the tumor presented high nuclear grade, classified as Fuhrman grade
3, showing solid growth nests, trabecular pattern, and sheet-like
growth pattern (Fig. 6). An
extensive hemorrhagic necrotic area and vascular invasion were
identified.
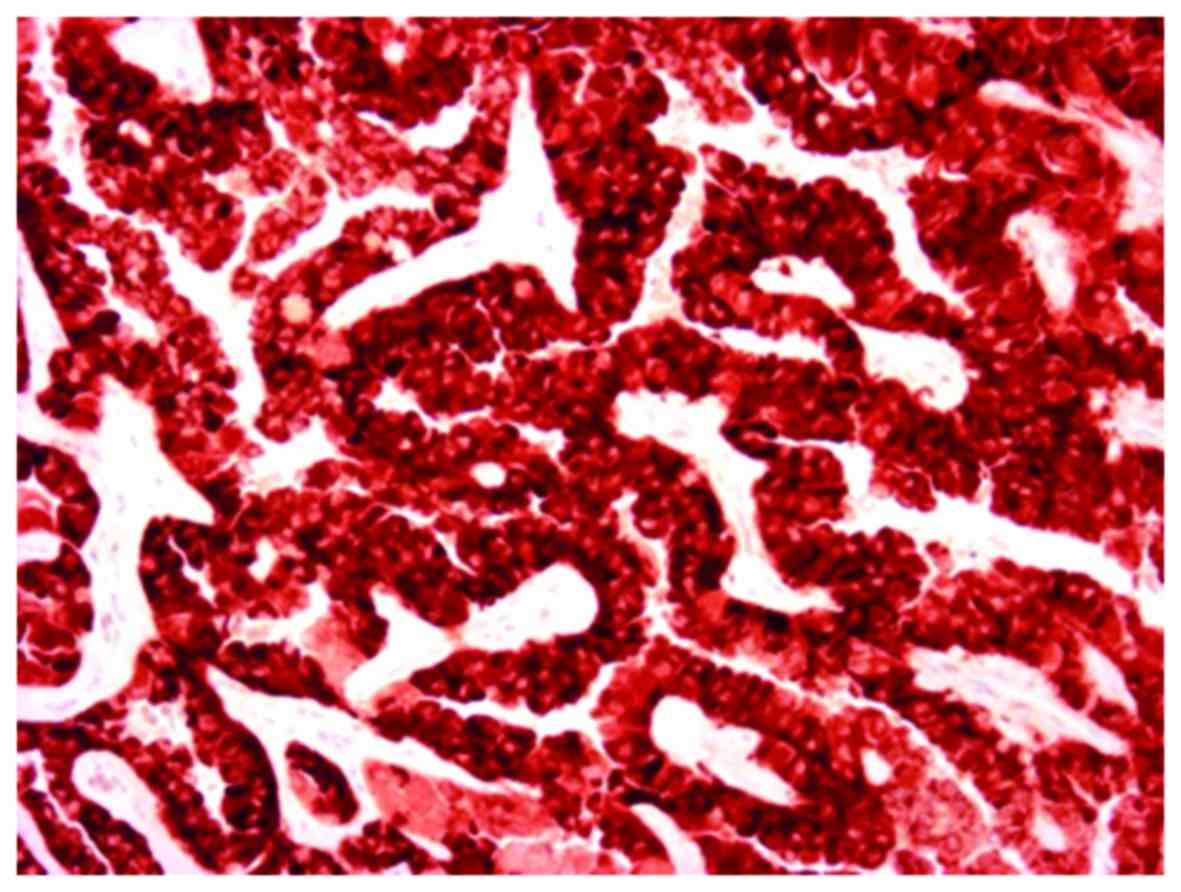
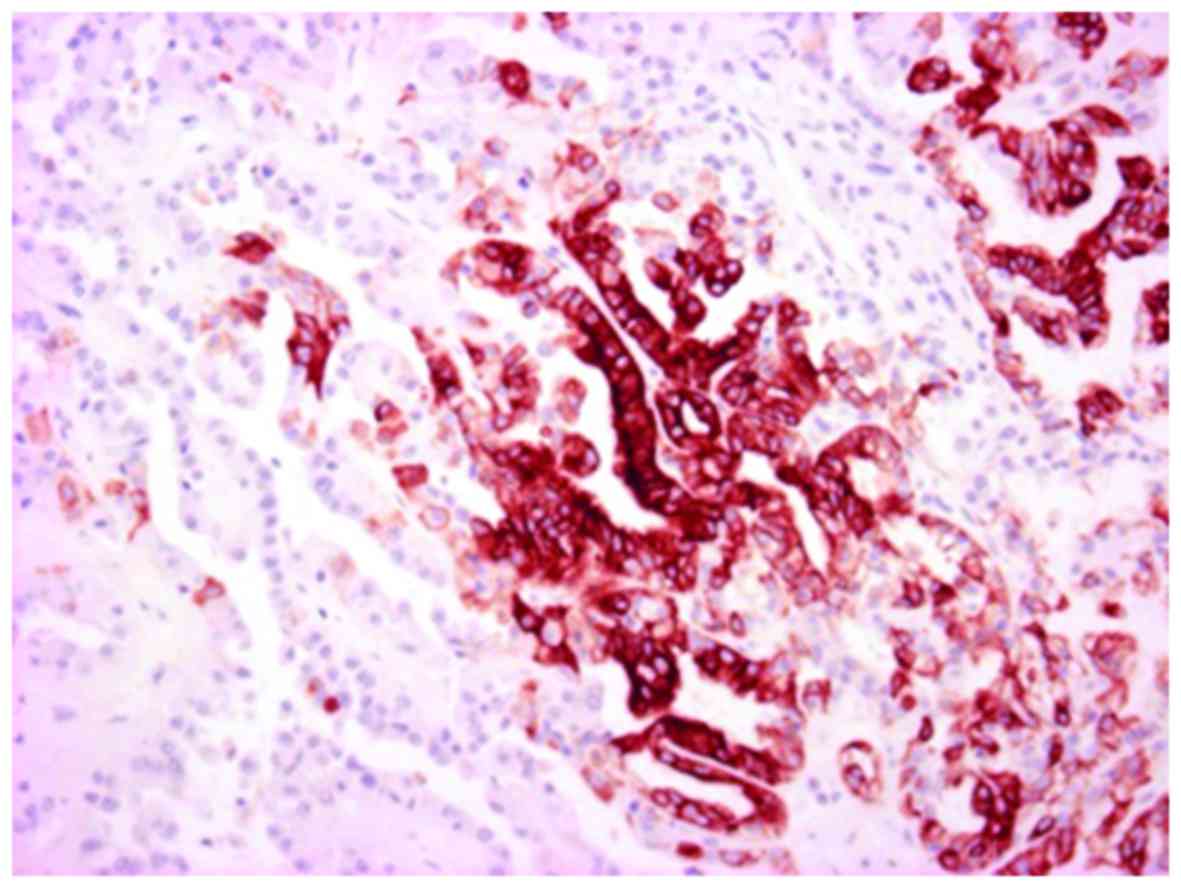
Immunohistochemistry results showed that the tumor
cells were positive for AMACR (Fig.
7) and focally positive for E-cadherin, CD10, CD15, EMA, and
CK7 (Fig. 8). However, the tumor
cells were negative for high molecular weight CK (clone 34βE12),
Vimentin and c-kit. In the conventional area, the Ki-67 labeling
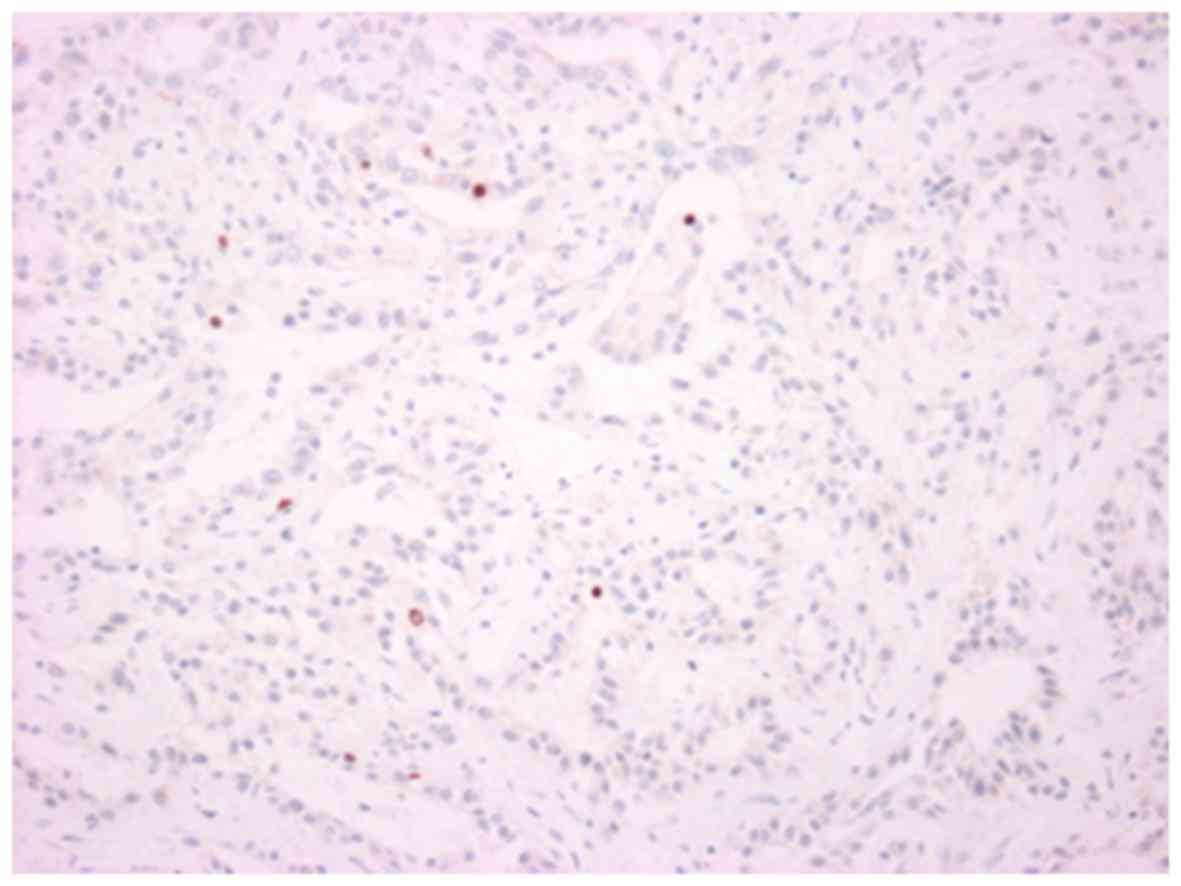
index was 7% (Fig. 9), and the level
of nuclear accumulation of p53 was low (Fig. 10). In the high-grade area, the Ki-67
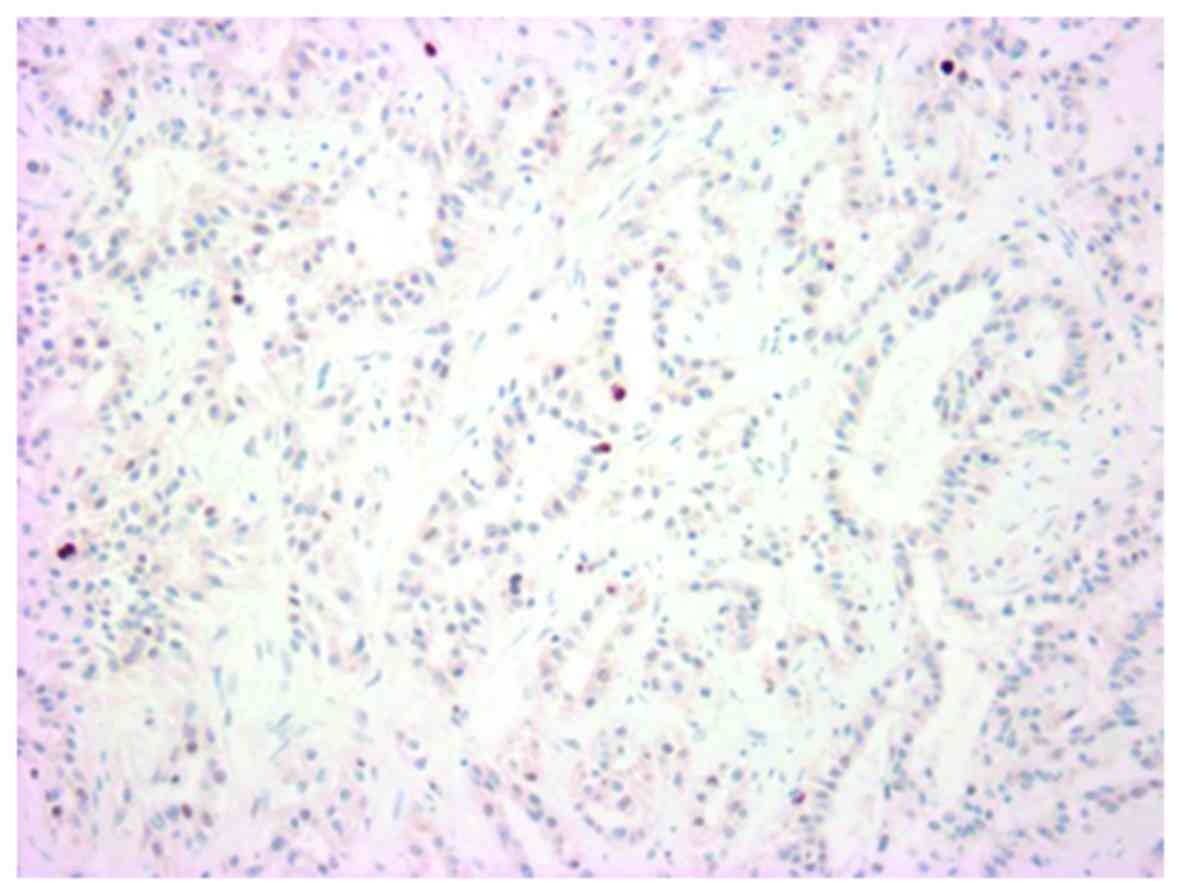
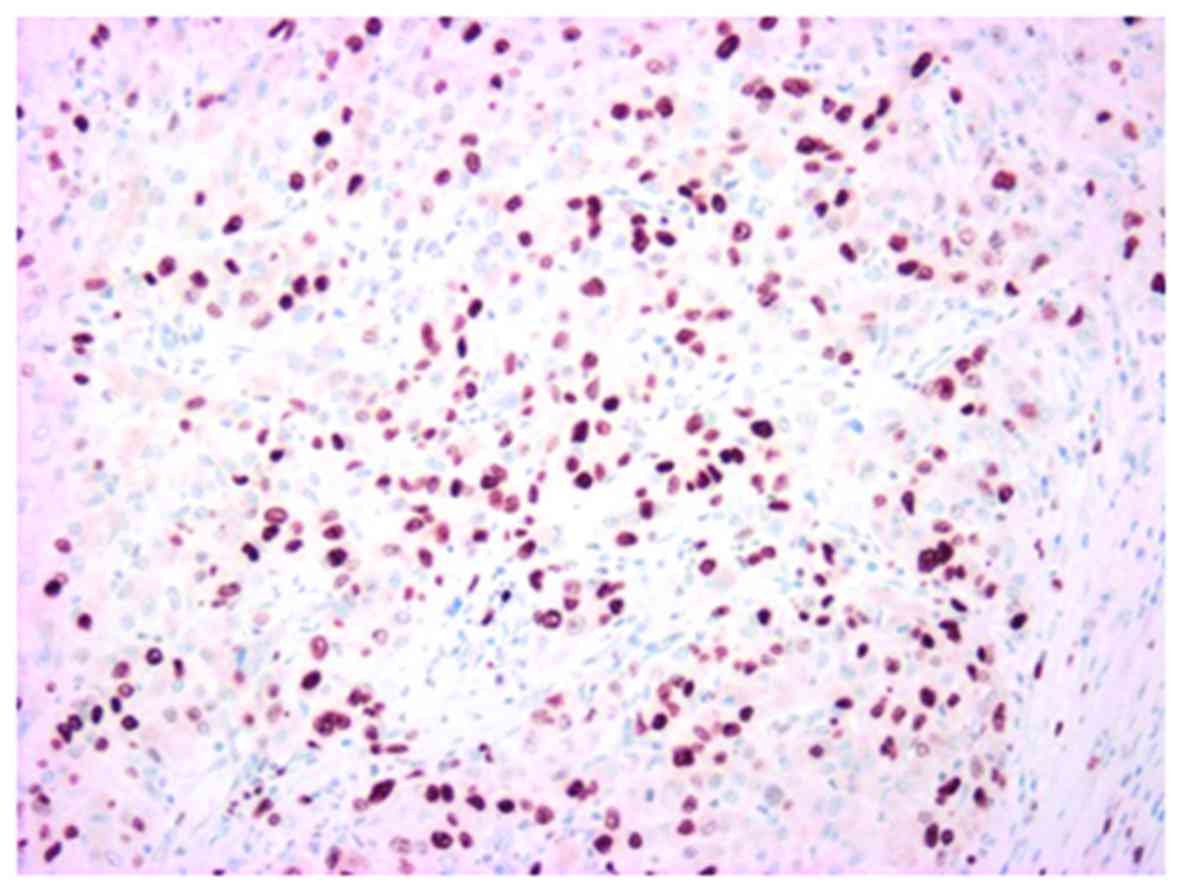
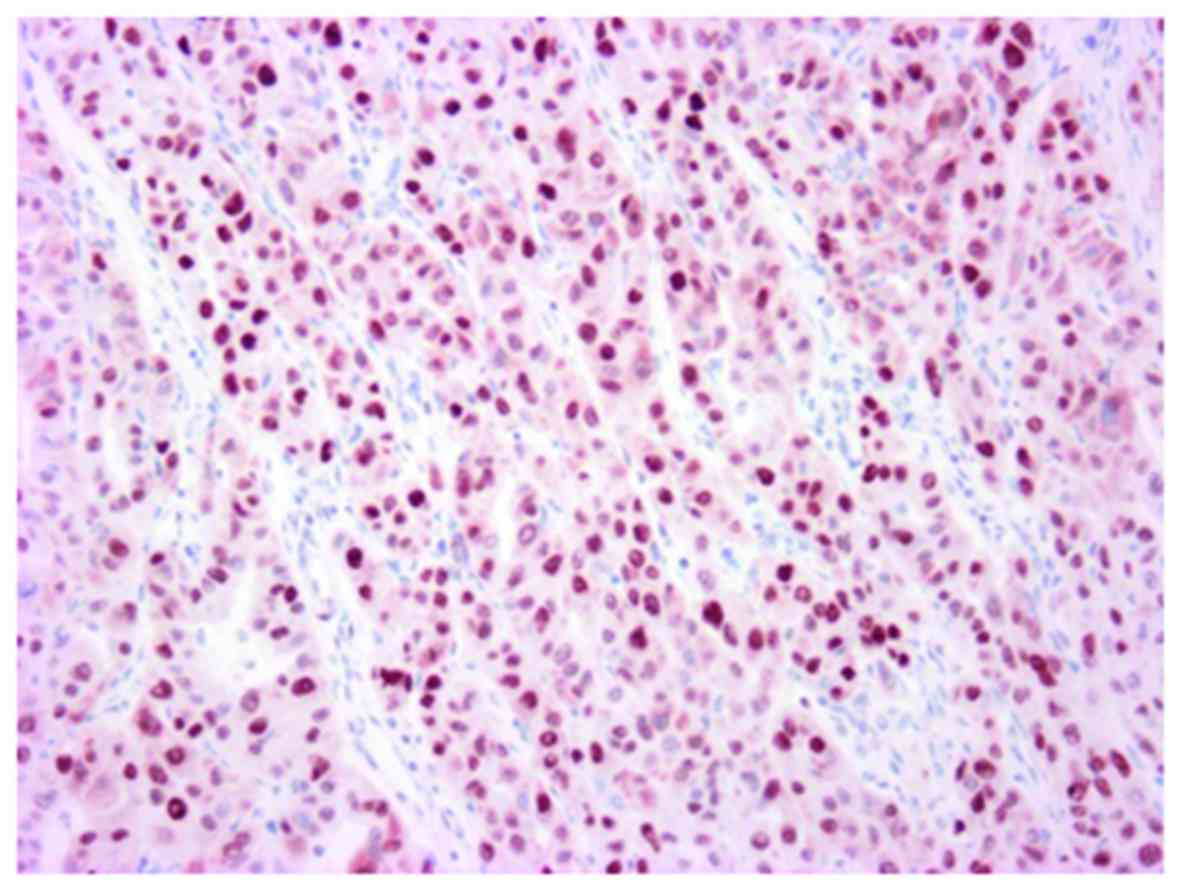
labeling index was 32% (Fig. 11),
and a marked p53 nuclear accumulation (Fig. 12) was observed.
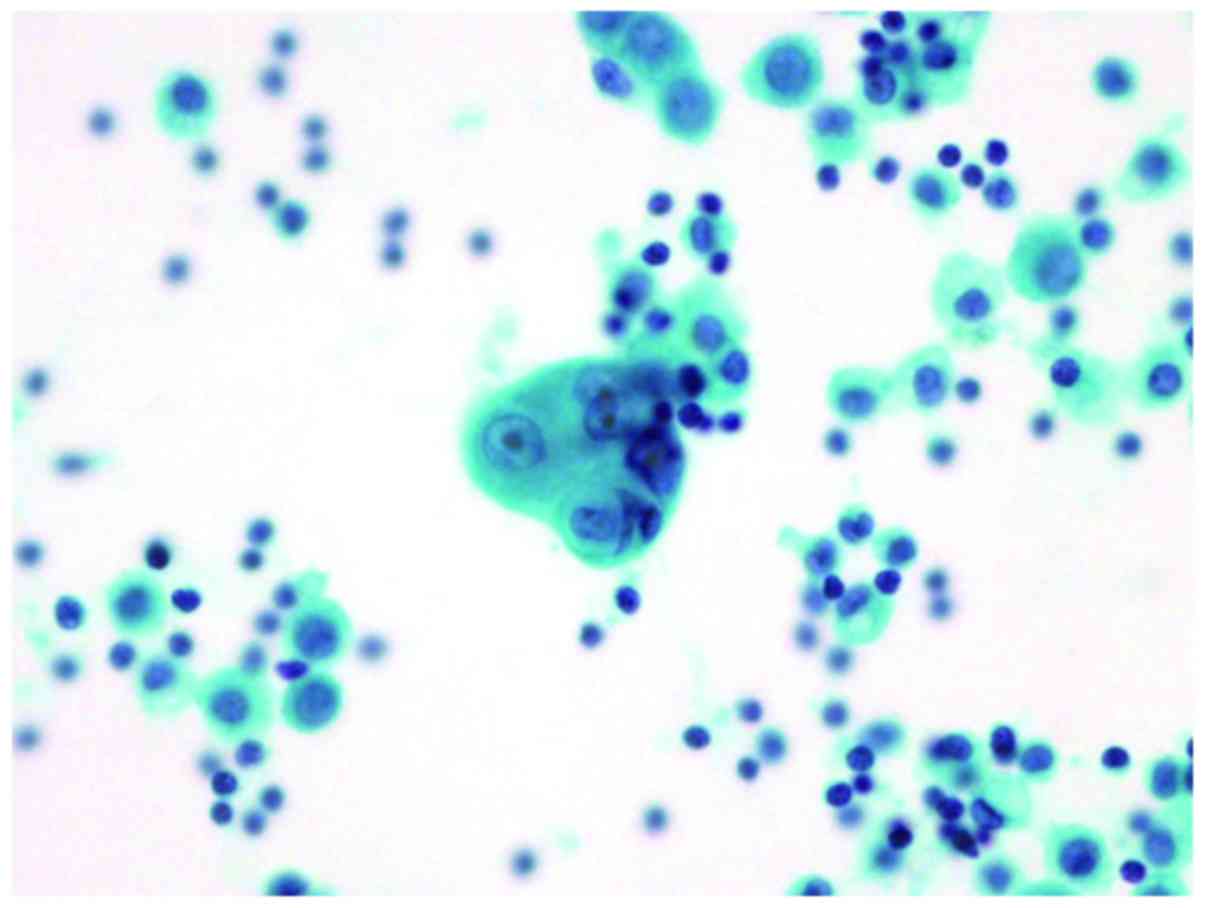
In the cytological specimens sampled from the
pleural effusion, small clusters of atypical cells were observed,
which showed high nucleocytoplasmic ratio and hyperchromatic nuclei
with nucleoli (Fig. 13).
Discussion
In 2004, MTSCC was introduced in the WHO
classification as a relatively uncommon histological type of RCC.
Initially, MTSCC was recognized as a low-grade collecting duct
carcinoma (8); however, because of
its significantly favorable prognosis, unlike that of collecting
duct carcinoma, it was later recognized as an independent
histological type (1). Recently,
fatal cases of MTSCC with nodal and distant metastases have been
reported (11–14). Therefore, in the 2016 WHO
classification, description that MTSCC is of low grade has been
deleted (2). A representative MTSCC
is characterized by elongated tubular and spindle cell components
with a mucinous stroma. Nevertheless, several MTSCCs have
demonstrated unusual mucin-poor features (6) in addition to secondary progression,
which is characterized by marked cytologic atypia, increased
mitotic activity of tumor necrosis, and sarcomatoid changes
(11–14). The cases presented in this report
were considered ‘mucin-poor’ MTSCC with high-grade transformation,
and caused distant metastasis and tumor-associated death.
MTSCC has been reported to possess similar
morphological and immunohistochemical characteristics of PRCC along
with AMACR expression (6), which
suggests that MTSCC may be a subtype of PRCC (15). Although AMACR expression hypothesizes
that MTSCC demonstrates proximal nephron differentiation (10), to the best of our knowledge,
histogenesis of MTSCC remains debatable. Our immunohistochemical
study demonstrated that the tumor cells are positive for both a
proximal nephron marker (AMACR) and distal nephron markers
(E-cadherin and CK7). These results are similar to previous reports
(6,16). In addition, our immunohistochemical
study demonstrated similar ecpression patterns between conventional
areas and high-grade areas for AMACR, E-cadherin, CD10, CD15, EMA
and CK7 and that the high-grade areas exhibited a high Ki-67
labeling index and p53 nuclear accumulation, which may be markers
of high-grade transformation and predictors of poor prognoses in
MTSCC. It is suggested that two hypotheses exist for high-grade
transformation. While one transformation is thought to occur from
low-grade MTSCC, the second possibility is ‘de novo’
development of a high-grade tumor (14). In our cases, low-grade MTSCC and
high-grade area were transient. In brief, we observed that the
high-grade area occurred by secondary progression of low-grade
MTSCC. Thus, it is necessary to clarify the mechanism related to
secondary high-grade progression. In case 2, the cytological
examination of pleural effusion revealed pleuritis carcinomatosa,
and the patient died 9 months after surgery. Only one report has
reported on pleural invasion; in that report, the patient died 1
month after surgery for acute respiratory failure (14).
Apparently, most MTSCCs are successfully treated
with radical surgical resection. However, for metastatic MTSCC, no
authorized guideline is available at present (5). In a previous study, one case of
metastatic MTSCC showing a response to sunitinib was identified
(17). Although case 1 in the
present study was treated with a molecular targeted therapy,
including sunitinib, its effect was limited. Further studies are
necessary to establish the systemic therapy guideline for
metastatic MTSCC.
In conclusion, MTSCC sometimes displays high-grade
transformation, including sarcomatoid change. Therefore, it is
necessary to perform a careful postoperative investigation of MTSCC
with high-grade transformation. Our study revealed that large tumor
size, tumor necrosis, vascular invasion, immunohistochemical
findings of p53 abnormal accumulation, and high Ki-67 labeling
index provide predict for poor prognosis. Therefore, clinicians
should pay attention to distantmetastasis and tumor-associated
death. Indeed, MTSCC is now considered a broad-spectrum tumor group
because it exhibits various histological patterns,
immunophenotypes, and clinical prognoses. In addition, further
studies are necessary to elucidate the biological behavior,
malignant potential, and histogenesis of MTSCC.
References
|
1
|
Srigley J: Mucinous tubular and spindle
cell carcinomaPathology and genetics of tumours of the urinary
system and male genital organs. Eble JN, Sauter G and Epstein JI:
IARC press; Lyon, France: pp. 402004
|
|
2
|
Kuroda N, Paner G and Srigley JR: Mucinous
tubular and spindle cell carcinoma. In: Pathology and genetics of
tumours of the urinary system and male genital organsHolger Moch.
Humphrey PA and Ulbright TM: IARC Press; Lyon, France: pp.
372016
|
|
3
|
Wu XR, Chen YH, Sha JJ, Zhao L, Huang JW,
Bo JJ, Liu DM and Huang YR: Renal mucinous tubular and spindle cell
carcinoma: A report of 8 cases and review of the literature. Diagn
Pathol. 8:2062013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Srigley JR, Delahunt B, Eble JN, Egevad L,
Epstein JI, Grignon D, Hes O, Moch H, Montironi R, Tickoo SK, et
al: The international society of urological pathology (ISUP)
vancouver classification of renal neoplasia. Am J Surg Pathol.
37:1469–1489. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhao M, He XL and Teng XD: Mucinous
tubular and spindle cell renal cell carcinoma: A review of
clinicopathologic aspects. Diagn Pathol. 10:1682015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fine SW, Argani P, DeMarzo AM, Delahunt B,
Sebo TJ, Reuter VE and Epstein JI: Expanding the histologic
spectrum of mucinous tubular and spindle cell carcinoma of the
kidney. Am J Surg Pathol. 30:1554–1560. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Farghaly H: Mucin poor mucinous tubular
and spindle cell carcinoma of the kidney, with nonclassic
morphologic variant of spindle cell predominance and psammomatous
calcification. Ann Diagn Pathol. 16:59–62. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
MacLennan GT, Farrow GM and Bostwick DG:
Low-grade collecting duct carcinoma of the kidney: Report of 13
cases of low-grade mucinous tubulocystic renal carcinoma of
possible collecting duct origin. Urology. 50:679–684. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Parwani AV, Husain AN, Epstein JI,
Beckwith JB and Argani P: Low-grade myxoid renal epithelial
neoplasms with distal nephron differentiation. Hum Pathol.
32:506–512. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Paner GP, Srigley JR, Radhakrishnan A,
Cohen C, Skinnider BF, Tickoo SK, Young AN and Amin MB:
Immunohistochemical analysis of mucinous tubular and spindle cell
carcinoma and papillary renal cell carcinoma of the kidney:
Significant immunophenotypic overlap warrants diagnostic caution.
Am J Surg Pathol. 30:13–19. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dhillon J, Amin MB, Selbs E, Turi GK,
Paner GP and Reuter VE: Mucinous tubular and spindle cell carcinoma
of the kidney with sarcomatoid change. Am J Surg Pathol. 33:44–49.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Simon RA, di Sant'agnese PA, Palapattu GS,
Singer EA, Candelario GD, Huang J and Yao JL: Mucinous tubular and
spindle cell carcinoma of the kidney with sarcomatoid
differentiation. Int J Clin Exp Pathol. 1:180–184. 2008.PubMed/NCBI
|
|
13
|
Pillay N, Ramdial PK, Cooper K and Batuule
D: Mucinous tubular and spindle cell carcinoma with aggressive
histomorphology-a sarcomatoid variant. Hum Pathol. 39:966–969.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bulimbasic S, Ljubanovic D, Sima R, Michal
M, Hes O, Kuroda N and Persec Z: Aggressive high-grade mucinous
tubular and spindle cell carcinoma. Hum Pathol. 40:906–907. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shen SS, Ro JY, Tamboli P, Truong LD, Zhai
Q, Jung SJ, Tibbs RG, Ordonez NG and Ayala AG: Mucinous tubular and
spindle cell carcinoma of kidney is probably a variant of papillary
renal cell carcinoma with spindle cell features. Ann Diagn Pathol.
11:13–21. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ferlicot S, Allory Y, Compérat E,
Mege-Lechevalier F, Dimet S, Sibony M, Couturier J and Vieillefond
A: Mucinous tubular and spindle cell carcinoma: A report of 15
cases and a review of the literature. Virchows Arch. 447:978–983.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Larkin J, Fisher R, Pickering L, Thway K,
Livni N, Fisher C and Gore M: Metastatic mucinous tubular and
spindle cell carcinoma of the kidney responding to sunitinib. J
Clin Oncol. 28:e539–e540. 2010. View Article : Google Scholar : PubMed/NCBI
|