Introduction
Epithelial-myoepithelial carcinoma (EMC) is a rare
malignant tumor that typically arises in the salivary glands. This
tumor displays a typical biphasic pattern, with a central ductular
structure surrounded by clear cells of myoepithelial origin. EMC
accounts for ~1% of all salivary gland tumors (1,2). EMC may
also arise in other locations, albeit less often, such as the minor
salivary glands or the upper and lower respiratory tract (3). EMC of the lung is an extremely rare
histological form that originates in the bronchial glands. Although
EMC of the salivary gland is considered to originate from the
intercalated duct (4), pulmonary EMC
appears to originate from the ductal structure of the bronchial
gland, which is one of the lung counterparts to the intercalated
duct (5), and accounts for ~0.1% of
all primary lung carcinomas (6). To
the best of our knowledge, a case of pulmonary EMC with a cavitary
lesion has never been reported to date. We herein report the case
of a patient with cavity-forming pulmonary EMC treated by
thoracoscopic surgery.
Case report
A 72-year-old man was referred to the Department of
Thoracic Surgery, Shin-Kokura Hospital (Kitakyushu, Japan) in March
2014 due to thickening of the cystic wall in the left upper lung
field. The cystic lesion had first been identified on routine
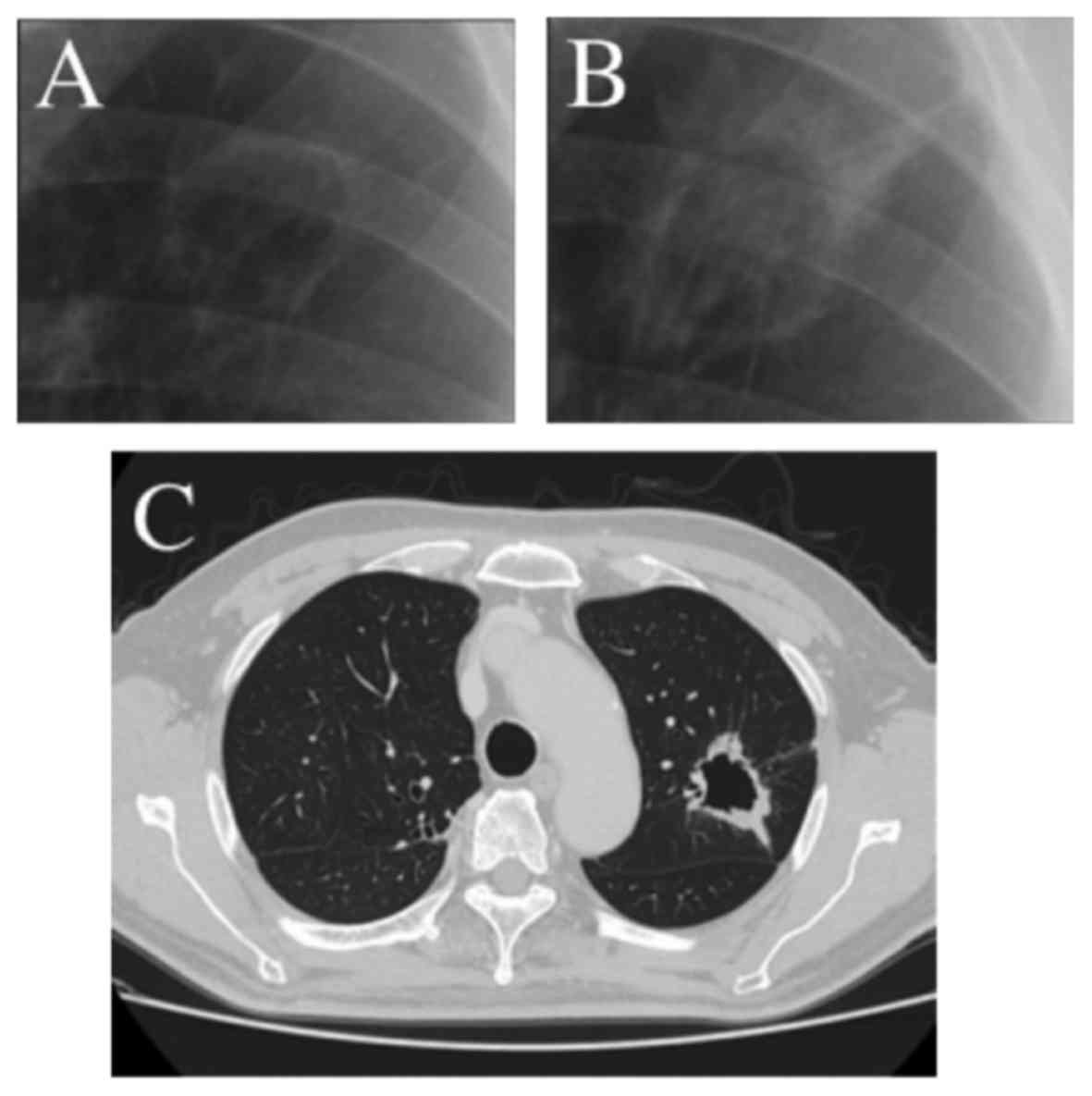
medical checkup (annual chest X-ray) 3 years earlier (Fig. 1A and B). The patient did not have any
respiratory symptoms, such as cough, hemoptysis or dyspnea. The
laboratory findings were unremarkable. A computed tomography (CT)
scan of the chest revealed an irregularly shaped lung tumor, 35 mm
in greatest diameter, with a cavitary lesion (Fig. 1C). There was no associated
enlargement of the hilar or mediastinal lymph nodes. Brain magnetic
resonance imaging examination, bone scintigraphy and body CT scan
with contrast enhancement detected no distant metastasis or
lymphadenopathy. The diagnosis following transbronchial lung biopsy
was non-small-cell lung carcinoma; thus, thoracoscopic left upper
lobectomy was performed.
Macroscopically, the tumor was gray-white in color,
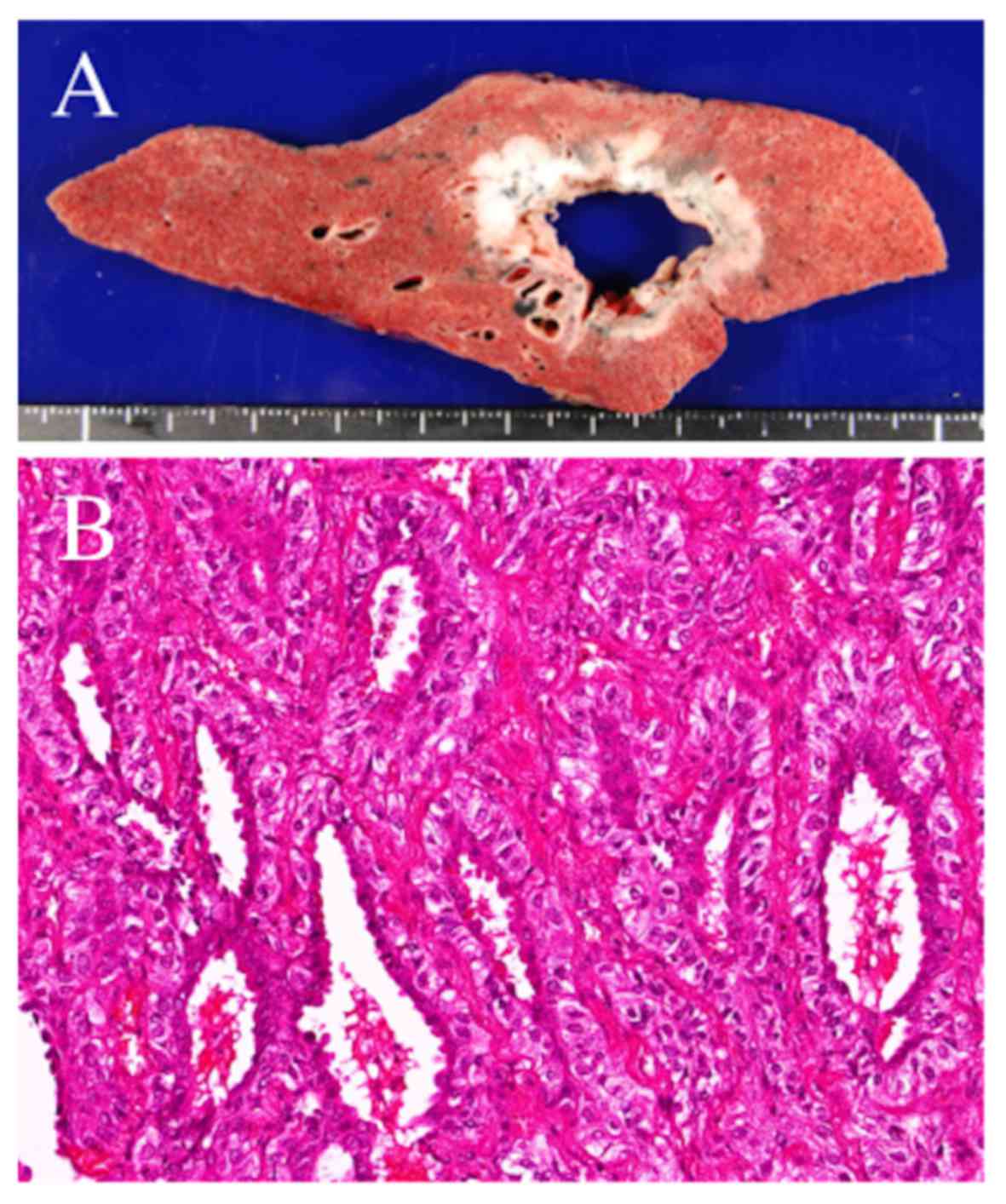
30×25×20 mm in size, with a central cavitary lesion (Fig. 2A). Microscopic examination
demonstrated a biphasic architecture, with glands surrounded by
myoepithelial cells (Fig. 2B). The
inner layers were composed of ductal cells with eosinophilic
cytoplasm, whereas the outer layers were composed of myoepithelial
cells with clear cytoplasm. The pathological diagnosis was EMC. The
dissected hilar and mediastinal lymph nodes were free of metastatic
disease. The postoperative course was uneventful. Although the
patient received no adjuvant therapy and the EMC did not recur;
however, rectal cancer was subsequently diagnosed and the patient
succumbed to mortality in February, 2018.
Discussion
EMC is known as a salivary gland-type tumor and
displays a typical biphasic pattern: A central ductular structure
surrounded by clear cells of myoepithelial origin. EMC accounts for
~1% of all salivary gland tumors (1,2). Despite
its predilection for the parotid gland, EMC also arises in other
locations, such as the minor salivary glands or the upper and lower
respiratory tract, albeit less often (3). EMC of the salivary gland is considered
to originate from the intercalated duct (4). Pulmonary EMC is considered to originate
from the ductal structure of the bronchial gland, which is one of
the pulmonary counterparts to the intercalated duct (5). The salivary gland-type tumor of the
lung accounts for 0.1% of all primary lung carcinomas (6), among which mucoepidermoid carcinoma is
the most frequently observed histological subtype, adenoid cystic
carcinoma is the second, and EMC is the third. The frequency of
each subtype is reported to be 70, 23 and 7%, respectively
(7,8).
EMC tends to be located in the central region of the
lung, rather than the periphery (8).
Almost all the bronchial glands are located in the central airway
(9), which may explain the tumor's
propensity to arise in central lung regions. The shape of EMC is
mostly round to oval or lobulated (8). The tumor in the present case was
located in the peripheral region of the lung, and appeared as a
lobulated mass accompanied by a cavitary lesion. To the best of our
knowledge, a case of EMC with a cavitary lesion has never been
reported to date.
The reported mechanisms of cavitary formation are as
follows: i) Central necrosis due to the rapid tumor growth, with
nutritional needs exceeding the blood supply; ii) bronchial or
alveolar expansion as a result of the ectatic changes of the
peripheral part following tumor invasion to more central parts; and
iii) infectious diseases, such as abscesses, fungal infections and
tuberculosis (10). In the present
case, however, the microscopic findings revealed neither necrotic
tissues nor expanding bronchi or alveoli in the inner part of the
tumor. The bacteriological examination revealed no signs of
infection. Therefore, it was hypothesized that a ductal structure
of the bronchial gland in the primary cystic lesion that had been
detected 3 years earlier was the site of origin of the EMC and grew
to become a cavitary lesion.
We herein described a rare case of primary EMC of
the lung with a cavitary lesion. Although the mechanism of the
cavity formation remains to be elucidated, EMCs of the lung can
include cavitary lesions.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
MM and MY conceived and designed this case report.
SA collected and interpreted the data. MM and TH wrote the initial
draft of the report. TH, RN and FT critically reviewed the
manuscript. The final version of the manuscript has been read and
approved by all authors.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient provided consent to the publication of
the case details and associated images.
Competing interests
The authors declare that they have no competing
interests to disclose.
References
|
1
|
Batsakis JG, El-Naggar AK and Luna MA:
Epithelial-myoepithelial carcinoma of salivary glands. Ann Otol
Rhinol Laryngol. 101:540–542. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Morrow TA, Chun T and Mirani N: Epithelial
myoepithelial carcinoma of the parotid gland. Ear Nose Throat J.
69:646–648. 1990.PubMed/NCBI
|
|
3
|
Fonseca I and Soares J:
Epithelial-myoepithelial carcinoma. World Health Organization
Classication of Tumours. Pathology and Genetics of Head and Neck
Tumours. 3rd edition. Barnes L, Eveson JW, Reichart P and Sidransky
D: IARC Press; Lyon, France: pp. 225–226. 2005
|
|
4
|
Corio RL, Sciubba JJ, Brannon RB and
Batsakis JG: Epithelial-myoepithelial carcinoma of intercalated
duct origin. A clinicopathologic and ultrastructural assessment of
sixteen cases. Oral Surg Oral Med Oral Pathol. 53:280–287. 1982.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Moran CA: Primary salivary gland-type
tumors of the lung. Semin Diagn Pathol. 12:106–122. 1995.PubMed/NCBI
|
|
6
|
Masuda M, Kuwano H, Okumura M, Arai H,
Endo S, Doki Y, Kobayashi J, Motomura N, Nishida H, Saiki Y, et al
Committee for Scientific Affairs, The Japanese Association for
Thoracic Surgery, : Thoracic and cardiovascular surgery in Japan
during 2013: Annual report by The Japanese Association for Thoracic
Surgery. Gen Thorac Cardiovasc Surg. 63:670–701. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kang DY, Yoon YS, Kim HK, Choi YS, Kim K,
Shim YM and Kim J: Primary salivary gland-type lung cancer:
Surgical outcomes. Lung Cancer. 72:250–254. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhu F, Liu Z, Hou Y, He D, Ge X, Bai C,
Jiang L and Li S: Primary salivary gland-type lung cancer:
Clinicopathological analysis of 88 cases from China. J Thorac
Oncol. 8:1578–1584. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mitani S: Studies on distribution and
histological observation of mucus gland in the bronchial tree of
adult human lungs. Jpn J Lung Cancer. 14:21–29. 1974. View Article : Google Scholar
|
|
10
|
Miura H, Taira O, Hiraguri S, Hagiwara M
and Kato H: Cavitating adenocarcinoma of the lung. Ann Thorac
Cardiovasc Surg. 4:154–158. 1998.PubMed/NCBI
|