Introduction
The incidence and mortality of lung cancer is on the
rise worldwide (1) and is the
leading cause of death among men and the second leading cause of
cancer death among women all over the world (2). In China, it is estimated that more than
4 million cases of cancer and nearly 3 million cancer deaths occur
(3). Most lung cancer patients were
diagnosed as at advanced stages and were clinically inoperable.
Conventional treatments including surgery, chemotherapy and
radiotherapy for lung cancer have proven to be ineffective for late
stages of lung cancer. The 5-year survival rate of lung cancer is
poor (4). Early diagnosis and
accurate prognosis analysis are important to improve the survival
rate of lung cancer patients. Discovery of microRNAs (miRNAs/miRs)
has opened up a new horizon for predicting the prognosis of lung
cancer. Many cancer-associated miRNAs can predict the prognosis of
lung cancer and can be targeted for treatment (5), but the results from individual
experiments of the miRNAs are still inconsistent.
miRNAs are short noncoding RNAs that consist of
21–23 nucleotides in length, which participate in the process of
translational repression or degradation of mRNA (6). Emerging studies showed that the
expression levels of miRNAs in cancer were different from that in
normal tissues. Besides, the specific expression signatures were
correlated with prognosis of cancer, such as breast cancer,
colorectal cancer and gastric cancer. According to the existing
research results, a growing number of researchers found that
different genes were related to the prognosis of lung cancer.
miRNAs may play a key role in the pathogenesis of lung cancer and
can be targets for potential therapeutics for the disease (7,8). For
example, it was reported that high levels of miR-211 were
associated with poor survival in human non-small cell lung cancer
(NSCLC) patients. miR-211 promotes tumor proliferation and
invasion via regulating MxA expression in NSCLC, which suggested
that manipulation of the level of miR-211 may provide a
novel therapy for NSCLC patients in the future (9). miR-197 expression was associated
with tumor size and identified as a novel independent predictor of
unfavorable prognosis for NSCLC patients (10). miR-638 levels were associated
with the survival of NSCLC patients and may be also considered a
potential independent predictor for NSCLC prognosis (11). The associations between miRNAs and
lung cancers were reported in many meta-analyses (12–14), but
the evaluation of the prognostic role of miRNAs in lung cancer has
not been fully analyzed. In this report, we systematically
performed a meta-analysis of miRNAs profiling with cancer stage,
LNM and OS rate of the lung cancer patient.
Materials and methods
Study selection and inclusion
criteria
Literature in PubMed database was comprehensively
searched (last updated search being February 1st, 2018). The
publications were identified with a combination of the key words:
miRNA, miRNAs, Micro RNA, RNA Micro, miRNA, Primary MicroRNA,
Primary miRNA, pri-miRNA, pri miRNA, small temporal RNA, small
stRNA, small temporal RNA, pre-miRNA, pre miRNA, lung neoplasms,
lung neoplasia, lung neoplasias, lung tumors, lung benign
neoplasms, lung benign neoplasm, lung benign malignancy, lung
malignancies, lung cancer and lung cancers.
To be included in the meta-analysis, studies had to
meet the following criteria: i) The subjects of studies were lung
cancer patients diagnosed through pathology or cytology; ii) the
studies aimed to investigate the relationship between miRNAs and
lung cancer patients; iii) patients were grouped according to
expression levels of miRNAs, which were measured in primary tumor
tissues or adjacent normal tissue; iv) related clinical
pathological characteristics were shown, such as tumor stages of
cancers (T), lymph node metastasis (LNM), or distant metastasis
(DM); v) available data contained information about the prognostic
value of patients with survival outcomes, such as overall survival
(OS)/recurrence-free survival (RFS)/event-free
survival(EFS)/distant metastases-free survival
(DMFS)/progression-free survival (PFS); vi) data contained hazard
ratio (HR) or relative risk (RR) and corresponding 95% CI, directly
or indirectly; and vii) full-text paper was available.
Studies were excluded if one of the following
existed: i) Duplicate publications; ii) non-human study or
non-clinical study or animal study or non-English study; iii)
reviews, case reports, letters, editorials, and expert opinions;
and iv) studies without available data or no complete text.
Data extraction
For the eligible studies, data was extracted
independently by two investigators (DY and SY). Disagreements were
resolved by discussion with another investigator (WX or YZ). The
following data was extracted: i) Publication information: The first
author's name, the year of publication and the country of origin;
ii) patients' characteristic information: Number of participants,
type of lung cancer, type of miRNAs, clinical tumor stage, and
follow-up duration; iii) miRNA detection information: Tissue
sample, method and cut-off values; iv) prognosis information: The
number of patients with lymph nodes metastasis and different tumor
stage; and v) survival analysis and multivariate analysis: ORs of
miRNAs for LNM, HRs of miRNAs for OS and corresponding 95% Cl and
P-values. This data was either obtained from the original article
directly with sufficient data or via Engauge Digitizer version 4.1
to extract when only Kaplan-Meier curves were available.
Two investigators (YZ and WX) used the
Newcastle-Ottawa Scale (NOS) to independently assess the quality of
all the included studies. There were three major sections
(selection, comparability and outcome) in NOS. The selection
consisted of adequacy information of case definition, number of
case in the research and representative miRNAs. The comparability
contained miRNA exposure, detection method and cut-off values. The
outcome included assessment results and adequate follow-up time.
The highest score of NOS criteria was 8 with the lowest score being
0. The higher the score on this test is indicative of a better
quality and a study with an NOS score equal to or more than 5 is
considered to be of good quality. Finally, we used Begg's test to
evaluate potential publication bias. The result pattern was not
significantly impacted by removing a single study each time.
Statistical analysis
The STATA software version 12.0 (StataCorp LP,
College Station, TX, USA) was used for the following statistical
analysis and to generate a forest plot to show the content of this
statistical analysis. I2 statistics and P-values were
used for investigating the heterogeneity among combined studies.
The heterogeneity was regarded as significant when the
I2 value >50% or a P-value <0.05 for Q test, while
I2 values <50% or P-value >0.05 indicated that
there was no significant heterogeneity, indicating that a
random-effects model had been used to test heterogeneity.
When evaluating the association between miRNAs,
prognosis, LNM and T, we used both HR/RR and 95% CI. Sensitivity
analyses were used to test the effect of each study on pooled
results. The potential publication bias was performed by Begg's
test and funnel plots were used to show the results. P<0.05 was
considered to indicate a statistically significant difference. The
biased risks of all the included studies were according to the
basis for assessing the internal validity of the prognostic study
and the recommendations on the biomarker research report (15–17).
Results
Characteristics of eligible studies,
publication bias and sensitivity analysis
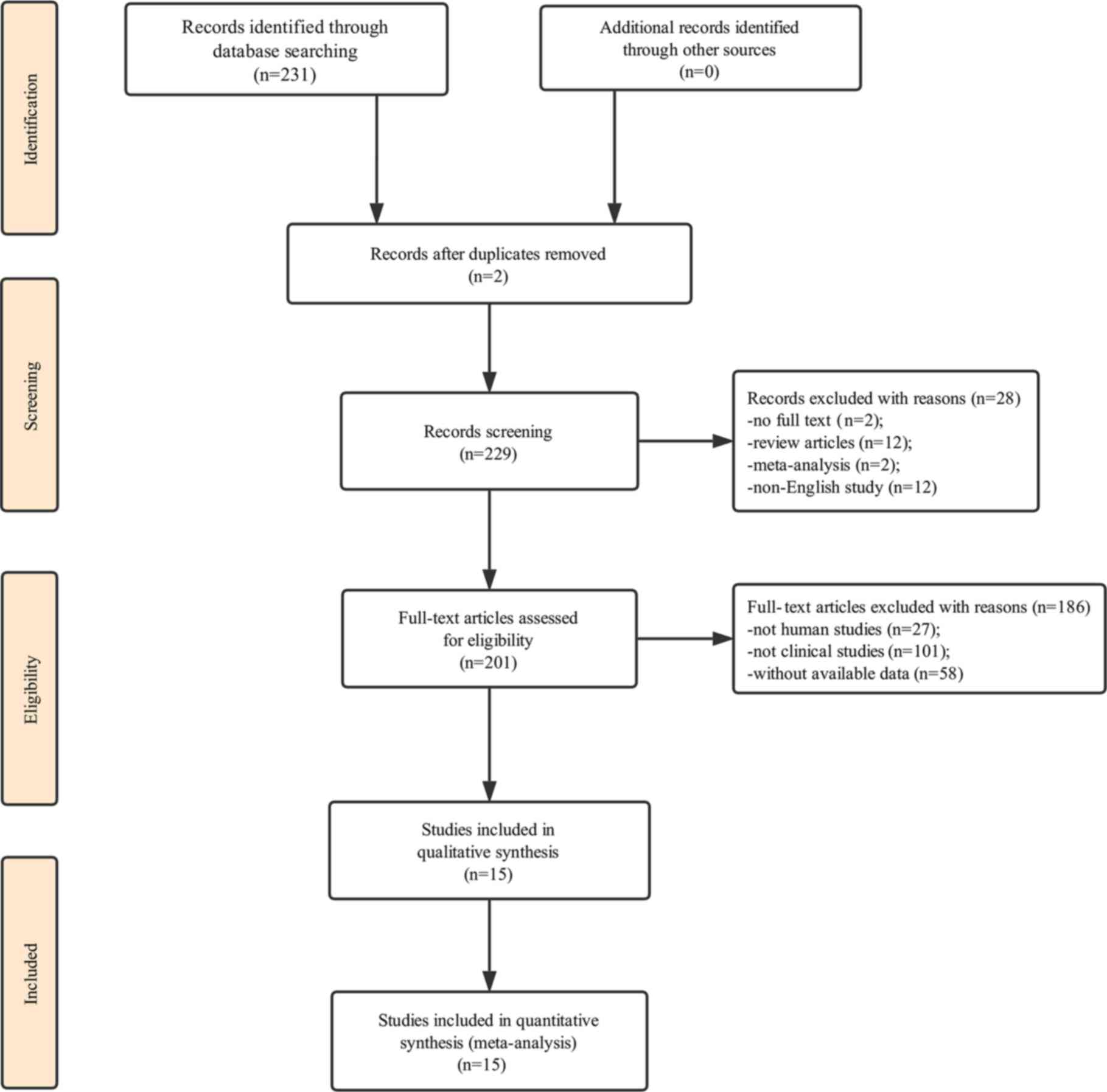
Our initial search of the database identified 231
publications. Two duplicate studies were excluded. We first
carefully reviewed the titles and abstracts of each article, a
total of 28 of them were excluded due to not containing full text,
review articles, meta-analysis or not being in English. We then
performed a detailed evaluation of each full text and 186 of them
were further excluded due to being animal experiments, irrelevant
clinical studies or without data available; therefore, only 15
articles were eligible for the final analysis (10,11,18–30). The
process of study selection is presented in Fig. 1 and the main characteristics of the
included articles are summarized in Table I.
 | Table I.Characteristics of studies in the
present meta-analysis. |
Table I.
Characteristics of studies in the
present meta-analysis.
| A, Study
information |
|---|
|
|---|
|
|
|
|
|
|
|
|
|
| miRNA
expression |
|
|---|
|
|
|
|
|
|
|
|
|
|
|
|
|---|
| Author, year | Country | miRNA | Total no. | Sample tissue | Detection
method | Cut-off | Expression
status | High
expression | Low expression | Refs. |
|---|
| Mavridis et
al, 2015 | Greece | 197 | 124 | Frozen tissue | qPCR | X-tile
algorithm | UR | 49 | 75 | (10) |
| Wang et al,
2015 | P.R. China | 638 | 189 | Serum | qPCR | X-tile
algorithm | DR | 112 | 77 | (11) |
| Chen et al,
2013 | P.R. China | 146α | 101 | Tissue | qPCR | Median | DR | 0 | 101 | (18) |
| Chen et al,
2017 | P.R. China | 148α | 159 | Tissue | qPCR | Median | DR | 73 | 86 | (19) |
| Cui et al,
2013 | P.R. China | 125b | 260 | Serum | qPCR | Median | UR | 260 | 0 | (20) |
| Guo et al,
2015 | P.R. China | 204 | 126 | Plasma | qPCR | Median | DR | 66 | 60 | (21) |
| Li et al,
2015 | P.R. China | 148α | 73 | Frozen tissue | qPCR | Mean | DR | 31 | 42 | (22) |
| Liu et al,
2012 | P.R. China | 21 | 70 | Frozen tissue | qPCR | Mean | UR | 48 | 22 | (23) |
|
|
| 141 | 70 |
|
|
|
| 49 | 21 |
|
|
|
| 200c | 70 |
|
|
|
| 38 | 32 |
|
| Nadal et al,
2014 | USA | 411 | 24 | Frozen tissue | qPCR | Median | UR | 24 | 0 | (24) |
|
|
| 370 | 33 | Frozen tissue | qPCR | Median | UR | 33 | 0 |
|
|
|
|
376α | 34 | Frozen tissue | qPCR | Median | UR | 34 | 0 |
|
| Skrzypski et
al, 2014 | Poland | 192 | 223 | Frozen tissue | qPCR | Median | UR | 90 | 133 | (25) |
|
|
| 662 | 223 | Frozen tissue | qPCR | Median | UR | 90 | 133 |
|
| Sun et al,
2013 | P.R. China | 150 | 90 | Frozen tissue | qPCR | Median | UR | 0 | 90 | (26) |
|
|
| 3940-5p | 90 | Frozen tissue | qPCR | Median |
| 0 | 90 |
|
| Võsa et al,
2011 | Estonia | 374α | 36 | Tissue | qPCR | Median | DR | 18 | 18 | (27) |
| Wang et al,
2016 | P.R. China | 148b | 39 | Frozen tissue | qPCR | Median | DR | 18 | 21 | (28) |
| Jiang et al,
2016 | P.R. China | 26b | 154 | Frozen tissue | qPCR | Median | DR | 77 | 77 | (29) |
| Rothschild et
al, 2012 | Bern | 381 | 18 | Tissue | qPCR | Median | DR | 3 | 15 | (30) |
|
|---|
| B,
Statistics |
|---|
|
|---|
|
| miRNA
expression |
|
|
|
|
|
|
|---|
|
|
|
|
|
|
|
|
|
|---|
| Author,
year | High with
T2-4/3-4 | Low with
T2-4/3-4 | High with
LNM | Low with
LNM | Survival
analysis | Multivariate
analysis | HR
statistic | HR (95%
CI) | Follow-up,
months | Refs. |
|
| Mavridis et
al, 2015 | 30 | 38 |
|
| OS | YES | SC | 1.91
(1.75–3.19) | 84 | (10) |
| Wang et al,
2015 | 73 | 51 | 87 | 70 | OS | YES | SC | 0.61
(0.35–1.06) | 60 | (11) |
| Chen et al,
2013 | 0 | 73 | 0 | 66 | OS | Unreported | SC | 0.56
(0.21–1.51) | 60 | (18) |
| Chen et al,
2017 | 58 | 70 | 27 | 48 | OS | YES | SC | 0.85
(0.54–1.35) | 120 | (19) |
| Cui et al,
2013 | 260 | 0 |
|
| OS | YES | Rep | 2.03
(1.14–3.05) | 25 | (20) |
| Guo et al,
2015 |
|
| 0 | 49 | OS | YES | SC | 0.54
(0.31–0.94) | 60 | (21) |
|
|
|
|
|
| DFS |
|
| 0.78
(0.44–1.36) |
|
|
| Li et al,
2015 | 0 | 37 | 9 | 30 | NA | Unreported |
|
| NA | (22) |
| Liu et al,
2012 | 34 | 0 | 32 | 0 | OS | YES | SC | 1.31
(0.42–3.03) | 24 | (23) |
|
| 34 | 0 | 32 | 0 |
|
|
| 1.02
(0.37–2.82) |
|
|
|
| 34 | 0 | 32 | 0 |
|
|
| 1.28
(0.04–2.53) |
|
|
| Nadal et al,
2014 | 5 | 0 |
|
| OS | YES | Rep | 3.37
(1.42–8.0) | 60 | (24) |
|
|
|
|
|
| DFS |
|
| 3.32
(1.61–6.88) |
|
|
|
| 16 | 0 |
|
| OS | YES | Rep | 3.37
(1.42–8.0) | 60 |
|
|
|
|
|
|
| DFS |
|
| 3.32
(1.61–6.88) |
|
|
|
| 22 | 0 |
|
| OS | YES | Rep | 3.37
(1.42–8.0) | 60 |
|
|
|
|
|
|
| DFS |
|
| 3.32
(1.61–6.88) |
|
|
| Skrzypski et
al, 2014 | 17 | 24 |
|
| OS | YES | SC | 1.24
(1.06–1.45) | 72 | (25) |
|
|
|
|
|
| DMFS |
|
| 1.12
(1.01–1.24) |
|
|
|
| 17 | 24 |
|
| OS | YES | SC | 2.58
(1.52–4.37) | 72 |
|
|
|
|
|
|
| DMFS |
|
| 1.23
(1.09–1.38) |
|
|
| Sun et al,
2013 | 0 | 52 | 0 | 34 | NA | YES |
|
| NA | (26) |
|
| 0 | 52 | 0 | 34 |
|
|
|
|
|
|
| Võsa et al,
2011 |
|
|
|
| OS | YES | SC | 0.98
(0.16–5.88) | 84 | (27) |
| Wang et al,
2016 | 8 | 19 | 9 | 7 | OS | YES | SC | 0.86
(0.31–2.38) | 60 | (28) |
| Jiang et al,
2016 | 27 | 43 | 18 | 51 | OS | YES | SC | 0.61
(0.26–1.44) | 60 | (29) |
| Rothschild et
al, 2012 |
|
|
|
| OS | Unreported | SC | 0.37
(0.25–0.66) | 35 | (30) |
|
|
|
|
|
| EFS |
|
| 0.48
(0.31–0.93) |
|
|
All of the included studies are considered as high
quality, as their NOS score was equal to or more than 5, while five
of them scored 8 in the quality assessment. Quality assessment of
eligible studies is shown in Table
II.
 | Table II.Quality assessment of eligible
studies (NOS). |
Table II.
Quality assessment of eligible
studies (NOS).
|
| Selection | Comparability | Outcome |
|
|
|---|
|
|
|
|
|
|
|
|---|
| Author, year | Adequacy of case
definition | No. of cases | Representativeness
of the cases | Ascertainment of
exposure | Ascertainment of
detection method | Ascertainment of
cut-off | Assessment of
outcome | Adequate
follow-up | Total | Refs. |
|---|
| Mavridis et
al, 2015 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 | (10) |
| Wang et al,
2015 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 | (11) |
| Chen et al,
2013 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 7 | (18) |
| Chen et al,
2017 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 | (19) |
| Cui et al,
2013 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 7 | (20) |
| Guo et al,
2015 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 5 | (21) |
| Li et al,
2015 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 5 | (22) |
| Liu et al,
2012 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 7 | (23) |
| Nadal et al,
2014 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 7 | (24) |
| Skrzypski et
al, 2014 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 | (25) |
| Sun et al,
2013 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 6 | (26) |
| Võsa et al,
2011 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 7 | (27) |
| Wang et al,
2016 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 7 | (28) |
| Jiang et al,
2016 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 | (29) |
| Rothschild et
al, 2012 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 6 | (30) |
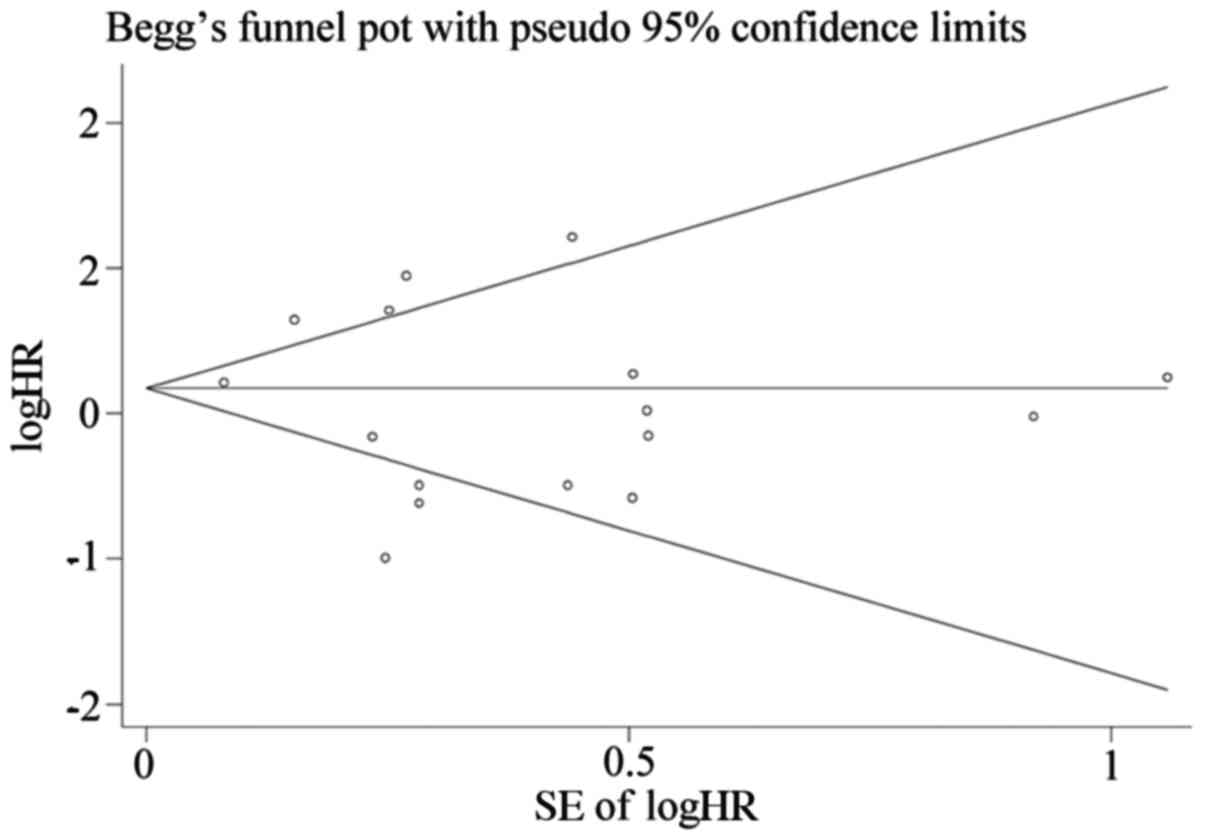
We used Begg's test and funnel plots to evaluate
potential publication bias. The shape of the funnel plot of the OS
group was symmetrical (Begg's test, t=−0.78, P=0.447), indicating
that no significant publication bias was observed by the Begg's
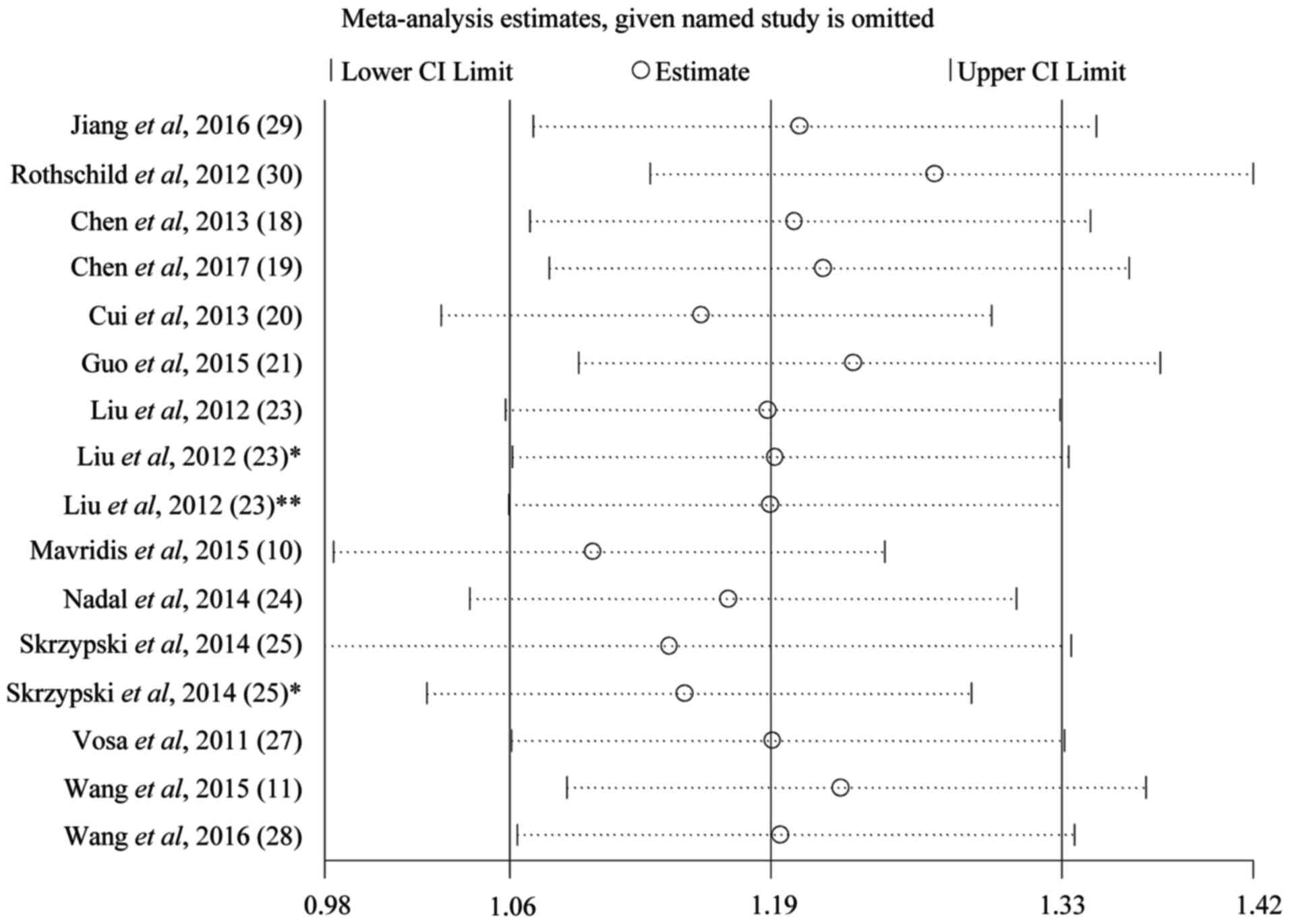
test (Fig. 2). Sensitivity analysis
of the publications in the OS group is presented in Fig. 3. The results pattern was not
significantly impacted by removing a single study each time.
Association between expression levels
of miRNAs and LNM in lung cancer
Six studies with 740 lung cancer patients were
included to assess the association between the expression levels of
different miRNAs and LNM. Five miRNAs were evaluated in this group,
including miR-26b, miR-148α, miR-204, miR-638 and
miR-148b. There was significant heterogeneity between the
studies included (P<1×10−4, I2=93.8%). The
random-effects model was applied and miR-148α was
investigated in two studies (19,22).
There were 150 patients who became LNM among 377 patients with high
miRNA expression, with a percentage of 39.8%; but patients with low
miRNA expression were more likely to become LNM as the percentage
was 71.8%. Analysis showed that the pooled RR was 0.53 (95% CI:
0.46–0.61, P<1×10−4), which indicated that low
expression of miRNAs was predictive of LNM (Fig. 4A).
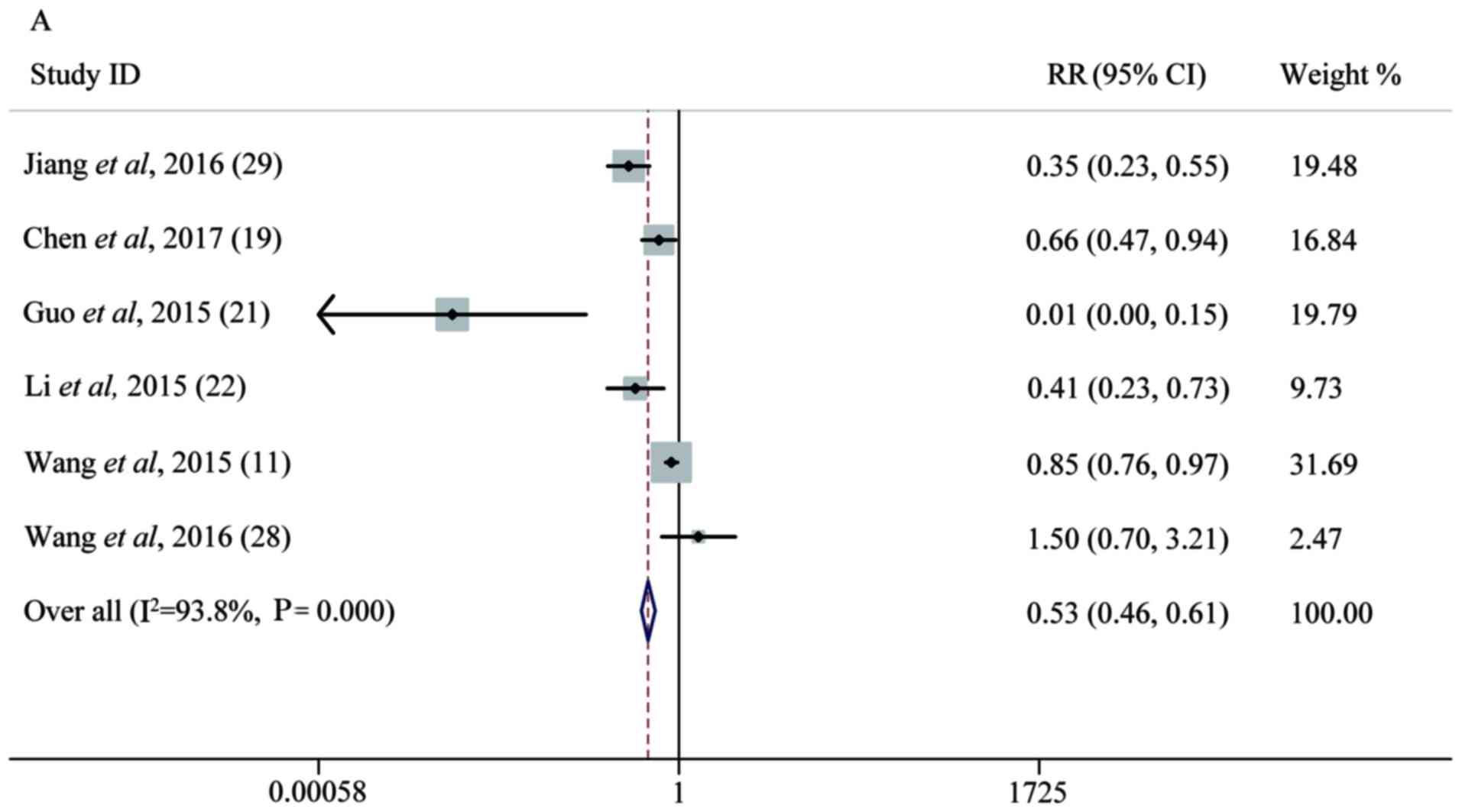 | Figure 4.Forest plot analysis. (A) Forest plot
of the LNM group. (B) Forest plot of the T group. (C) Forest plot
of the OS group. Each plot A-C corresponds to the meta-analysis of
a different group: (A) LNM, (B) T, and (C) OS. The effect size for
the estimate of each study was presented as a grey square
proportional in size to the weight of that study. The confidence
interval around that effect size was presented as a horizontal
line. The vertical line across these estimates represents HR=1 and
any horizontal line crossing this vertical line represents a
non-statistically significant result. The summary effect size was
presented as a rhombus, the center of which represents the summary
effect size and the width of which represents its CI. All
meta-analyzed groups, apart from the T group (plot B), were
statistically significantly associated with microRNAs. LNM, lymph
node metastasis; RR, relative risk; CI, confidence interval; T,
tumor stages of cancer; OS, overall survival; DR, downregulated;
UR, upregulated. |
Association between miRNA levels and
lung cancer T stages
Seven studies containing 1,031 patients were
included to evaluate the relationship between miRNA level and tumor
T stage. We divided T1 into a low T stage group and T2-4 into high
a T stage group, according to T stage level (T1/2/3/4). Significant
heterogeneity was found among the studies (P<1×10−4,
I2=79%) and a random-effects model study showed a pooled
RR of 1.07 with a 95% CI of 0.94–1.22 (P=0.23), indicating that the
expression of miRNAs might not be a direct predictor of T stage
(Fig. 4B).
Association between the expression of
miRNAs and OS of lung cancer patients
Thirteen studies of 1,536 lung cancer patients were
included to assess the correlation between different miRNA
expression levels and OS. Subgroup analysis was performed on the
basis of the expression of miRNAs. Eight studies of 768 lung cancer
patients reported the relationship between downregulated expression
of different miRNAs and OS, including miRNA-26b, miRNA-381,
miRNA-146α, miRNA-148α, miRNA-204, miRNA-374α, miRNA-638 and
miRNA-148b. There was no significant heterogeneity among the
combined studies (P=0.434, I2=0%) by applying the random
effects model (31). We found that
low expression of these miRNAs was associated with shorter OS of
lung cancer patients, according to the pooled HR of 0.59 with a 95%
CI of 0.47–0.75, (P<1×10−4). High expression of 8
miRNAs mentioned above increased the likelihood of survival. Five
studies of 768 lung cancer patients reported the relationship
between upregulated expression of different miRNAs and OS,
including miRNA-125b, miRNA-21, miRNA-141, miRNA-200c,
miRNA-197, miRNA-41, miRNA-370, miRNA-376α, miRNA-192 and
miRNA-662. There was significant heterogeneity among the
combined studies (P=0.014, I2=60.2%). High expression of
these 10 miRNAs was associated with poorer OS of lung cancer
patients, according to the pooled HR of 1.76 with a 95% CI of
1.31–2.35 (P<1×10−4) (Fig.
4C). Only 2 studies included, had studied the association
between the expression of miRNAs and disease-free survival (DFS) of
lung cancers, while only 1 study accessed the association between
the expression of miRNAs and DMFS of lung cancer patients. Since
there were less than three of each type of study there was no test
for association. All meta-analysis results are shown in Table III.
 | Table III.Results of meta-analysis. |
Table III.
Results of meta-analysis.
| Outcomes | No. of studies | No. of
patients | HR/OR/RR (95%
CI) | P-value | Heterogeneity
I2 (%) | Publication bias
P-value |
|---|
| LNM | 6 | 740 | 0.53
(0.46–0.61) |
<1×10−4 | 93.8 | 0.145 |
| T | 7 | 1,031 | 1.07
(0.94–1.22) | 0.23 | 79.0 | 0.527 |
| OS (DR) | 8 | 768 | 0.59
(0.47–0.75) |
<1×10−4 | 0.0 | 0.447 |
| OS (UR) | 5 | 768 | 1.76
(1.31–2.35) |
<1×10−4 | 60.2 | 0.447 |
Discussion
Lung cancer is the most commonly diagnosed cancers
among men and women. It is also the most commonly diagnosed cancer
and the leading cause of cancer death in men aged 75 years or older
(3). Although early diagnosis,
targeting therapy and immunotherapy treatment of lung cancer have
developed rapidly in recent years, a vast majority of lung cancers
are still diagnosed at a late stage (32) and have poor prognosis. The 5-year
survival rate is only approximately 10% and is one of the lowest
among cancer patients (33). Finding
new biomarkers can not only improve early diagnosis but also
provide new targeting means after prognosis analysis. miRNAs have
emerged as new regulators of cancer genomes and accumulating
studies have found that many miRNAs are associated with the
prognosis of lung cancer, especially NSCLC (34,35).
On the one hand, there are few studies focus on the
relationship between the same miRNA and lung cancer, so it is
impossible for us to discuss. A meta-analysis is a statistical
analysis that combines the results of multiple scientific studies.
In our meta-analysis, we examined all evidences on the potential
role of various miRNAs in lung cancer in order to systemically
analyze the available clinical data. On the other hand, the targets
and mechanism of miRNAs in lung cancer are still unclear,
summarizing all the similar researches can provide us a direction
in clinical work and improve our comprehension of potential
intricate biological mechanism. We analyzed and evaluated the
association between expression levels of multiple miRNAs and
clinical prognosis for lung cancer patients, which laid a
foundation for the diagnosis, prognosis evaluation and targeted
therapy of lung cancer.
We found that high expression of miR-125b,
miR-21, miR-141, miR-200c, miR-197, miR-41, miR-370, miR-376α,
miR-192 and miR-662 resulted in a lower level of
survival among patients. We also found that low levels of
expression of miR-26b, miR-381, miR-146α, miR-148α, miR-204,
miR-374α, miR-638 and miR-148b also resulted in a lower
level of survival among patients. Our analysis showed that high
expression of miRNAs was correlated with LNM in lung cancer. We
could not observe the relationships between the expression of
various miRNAs and tumor stages, DMS or DMFS, which may be due to
the samples size of this study.
miRNAs can regulate growth factors, target tumor
suppression genes and other transcriptional factors to promote or
inhibit the proliferation of cancer in humans. miR-125b may
be associated with TGF-β to stimulate cancer growth and the
potential activation of TGF-β with the possibility of playing a
role in promoting cancer (36).
miR-21 is one of the most commonly observed aberrant miRNAs
in human cancers and a large scale miRNA analysis of 540 samples
from six different types of solid tumors showed that miR-21
was the only miRNA that was upregulated in all cancer types
(37). miR-21 is believed to
target many tumor suppressors to regulate epithelial-mesenchymal
transition, invasion and metastasis in lung cancer (38). There is evidence that miR-141
promoted the proliferation of non-small cell lung cancer cells by
regulating the expression of PH domain leucine-rich-repeats protein
phosphatase 1 and 2 (PHLPP1 and PHLPP2) (39). miR-200c was found to exert
tumor-suppressive effects for NSCLC through the suppression of
USP25 expression (40).
miRNA-197 acts as an oncogene downregulating p53 and FUS1
tumor suppressor gene expression (41,42). It
was also shown to be associated with brain metastasis in
EGFR-mutant lung cancer (43).
miR-370 was reported to inhibit the progression of non-small
cell lung cancer by downregulating the oncogene TRAF4 (44). miR-376α could suppress the
proliferation and invasion of non-small-cell lung cancer by
targeting c-Myc (45).
miR-192 was found to regulate chemo-resistance of lung
adenocarcinoma for gemcitabine and cisplatin combined therapy by
targeting Bcl-2 (46).
miR-662 may provide an alternative mechanism of
downregulation of a tumor suppressor gene GDF10 that belongs to the
TGF-β family ligands (25).
miR-26b was shown to function as a critical regulator of
tumor progression and carcinogenesis because it acts as either a
tumor suppressor or an oncogene in various cancers (29). miR-381 targeted IGF-1R to
deactivate the protein kinase B (AKT) and extracellular
signal-regulated kinase (ERK) signaling pathways (47). Overexpression of miR-146α
significantly enhanced cell apoptosis, inhibiting cell viability
and motility in vitro and in vivo and miR-146α
could specially degrade the mRNA of cyclin J in the development of
acquired drug resistance to DDP-based chemotherapy in NSCLC cells
(48). Overexpression of
miRNA-148α inhibited Wnt-1 protein expression in cancer
cells. Knocking down Wnt-1 by siRNA had a similar effect to that of
miRNA-148α overexpression on cell migration and invasion in
lung cancer cells (19).
miR-148b was reported to regulate radio-resistance of lung
cancer cells by modulating the level of MLH1 expression (49). miRNA-204 suppressed human
non-small cell lung cancer by targeting activating transcription
factor 2 (ATF2) (50).
miR-374α had very significantly affected pathways of cell
migration (27). Downregulation of
miRNA-638 promotes angiogenesis and growth of hepatocellular
carcinoma by targeting VEGF (51).
Though the sample types were different from each
other in the publications included in our analysis, we put them
together for some reasons. Firstly, all the studies included in the
paper used qPCR, allowing comparisons to be made between them.
Secondly, it was reported that serum miRNAs have potential clinical
value as tumor markers for screening NSCLC. Serum expression levels
of miR-125b and miR-22 in NSCLC patients were
significantly higher than those with benign lung diseases and those
in the healthy controls (52). It
also reported that serum and plasma could be better mediums to
measure miRNA compared to sputum and whole blood. When combining CT
scanning with miRNA measured in serum or plasma, the sensitivity
value increased (35). Last but not
least, there is a continued need for the development of
minimally-invasive, cost-effective and easy methods to diagnose
lung cancer at an earlier, curable stage. The miRNA levels in
plasma, serum and other biological fluids could be optimistic and
promising markers in the future.
Several high-quality studies arouse us great
interest before we drafted our study. Similar publications have
been published looking into the value of miRs in lung cancer
(53–55). Since the topic has been hot in recent
research, we performed this analysis. Different from previous
studies, we further evaluated the relationship between expression
levels of multiple miRNAs and cancer stage and LNM for lung cancer
patients. In our meta-analysis, we summarized and reviewed all the
similar researches and identified that a few miRNAs could serve as
a novel predictor for the prognosis of NSCLC. It also provided very
useful resources for new therapeutic targets for the management of
lung cancer in the future. However, we also understood that there
had a number of limitations in our research. First, it was only
based on the results of one database (PubMed) and some relevant
studies might have been missed. Second, only 15 articles were
included in this analysis and all of the cancer types were NSCLC.
Third, though the detection method of miRNAs in all studies
analyzed were reverse transcription qPCR, the kits, analytical
methods and positive results evaluation criteria might have had
variations which may have led to different experimental results.
Fourth, the search language in this study was limited to English
and other languages such as Chinese were not included, which might
lead to an inevitable bias. Fifth, although no significant
publication bias was found in the included studies, it still could
not be completely avoided as positive results are more likely to be
accepted while negative results are more likely to be rejected or
not published. Sixth, the sample types were different from each
other in the publications included in the analysis. These results
were combined and therefore the overall determined conclusion of
the study might not be widely applicable due to the inclusion of
these different samples.
All in all, we have examined a substantial number of
prognostic data about the association of various miRNAs and the
survival of lung cancer patients. Our meta-analysis identified that
a few miRNAs could serve as a novel predictor of prognosis in
NSCLC. Larger studies are required to further confirm our
observation and understanding of the roles of these miRNAs and
their targets in cancer cells that can bring new therapeutic
methods to NSCLC lung cancer treatment.
Acknowledgements
The authors would like thank their colleagues at the
Department of Thoracic Medicine of Shenzhen Second People's
Hospital (Guangdong, China) for their assistance during data
extraction and analysis.
Funding
The present study was supported by a grant from the
Shenzhen Municipal Science and Technology Innovation Committee
(grant no. JCY20170412170814624).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
DY and SY extracted and assessed that data from
eligible studies. WX and YZ interpreted the data for miRNA
expression levels and prognosis in NSCLC. LW and MZ assessed and
revised the data and manuscript. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
Statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Torre LA, Siegel RL and Jemal A: Lung
cancer statistics. Adv Exp Med Biol. 893:1–19. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li D, Song L, Wen Z, Li X, Jie J, Wang Y
and Peng L: Strong evidence for LncRNA ZNRD1-AS1, and its
functional Cis-eQTL locus contributing more to the susceptibility
of lung cancer. Oncotarget. 7:35813–35817. 2016.PubMed/NCBI
|
|
5
|
Castro D, Moreira M, Gouveia AM, Pozza DH
and De Mello RA: microRNAs in lung cancer. Oncotarget.
8:81679–81685. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Aghanoori MR, Mirzaei B and Tavallaei M:
miRNA molecular profiles in human medical conditions: Connecting
lung cancer and lung development phenomena. Asian Pac J Cancer
Prev. 15:9557–9565. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kozomara A and Griffiths-Jones S: miR
base: Annotating high confidence microRNAs using deep sequencing
data. Nucleic Acids Res. 42:D68–D73. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhong Y, Chen Z, Guo S, Liao X, Xie H,
Zheng Y, Cai B, Huang P, Liu Y, Zhou Q, et al: TUG1, SPRY4-IT1, and
HULC as valuable prognostic biomarkers of survival in cancer: A
PRISMA-compliant meta-analysis. Medicine (Baltimore). 96:e85832017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kang M, Shi J, Peng N and He S:
MicroRNA-211 promotes non-small-cell lung cancer proliferation and
invasion by targeting MxA. OncoTargets Ther. 10:5667–5675. 2017.
View Article : Google Scholar
|
|
10
|
Mavridis K, Gueugnon F, Petit-Courty A,
Courty Y, Barascu A, Guyetant S and Scorilas A: The oncomiR miR-197
is a novel prognostic indicator for non-small cell lung cancer
patients. Br J Cancer. 112:1527–1535. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang F, Lou JF, Cao Y, Shi XH, Wang P, Xu
J, Xie EF, Xu T, Sun RH, Rao JY, et al: miR-638 is a new biomarker
for outcome prediction of non-small cell lung cancer patients
receiving chemotherapy. Exp Mol Med. 47:e1622015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Võsa U, Vooder T, Kolde R, Vilo J,
Metspalu A and Annilo T: Meta-analysis of microRNA expression in
lung cancer. Int J Cancer. 132:2884–2893. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Guan P, Yin Z, Li X, Wu W and Zhou B:
Meta-analysis of human lung cancer microRNA expression profiling
studies comparing cancer tissues with normal tissues. J Exp Clin
Cancer Res. 31:542012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liang Y: An expression meta-analysis of
predicted microRNA targets identifies a diagnostic signature for
lung cancer. BMC Med Genomics. 1:612008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lin Y, Ge Y, Wang Y, Ma G, Wang X, Liu H,
Wang M, Zhang Z and Chu H: The association of rs710886 in lncRNA
PCAT1 with bladder cancer risk in a Chinese population. Gene.
627:226–232. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Altman DG, McShane LM, Sauerbrei W and
Taube SE: Reporting recommendations for tumor marker prognostic
studies (REMARK): Explanation and elaboration. PLoS Med.
9:e10012162012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bouwmeester W, Zuithoff NP, Mallett S,
Geerlings MI, Vergouwe Y, Steyerberg EW, Altman DG and Moons KG:
Reporting and methods in clinical prediction research: A systematic
review. PLoS Med. 9:1–12. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chen G, Umelo IA, Lv S, Teugels E, Fostier
K, Kronenberger P, Dewaele A, Sadones J, Geers C and De Grève J:
miR-146α inhibits cell growth, cell migration and induces apoptosis
in non-small cell lung cancer cells. PLoS One. 8:e603172013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen Y, Min L, Ren C, Xu X, Yang J, Sun X,
Wang T, Wang F, Sun C and Zhang X: miRNA-148α serves as a
prognostic factor and suppresses migration and invasion through
Wnt1 in non-small cell lung cancer. PLoS One. 12:e01717512017.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cui EH, Li HJ, Hua F, Wang B, Mao W, Feng
XR, Li JY and Wang X: Serum microRNA 125b as a diagnostic or
prognostic biomarker for advanced NSCLC patients receiving
cisplatin-based chemotherapy. Acta Pharmacol Sin. 34:309–313. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Guo W, Zhang Y, Zhang Y, Shi Y, Xi J, Fan
H and Xu S: Decreased expression of miR-204 in plasma is associated
with a poor prognosis in patients with non-small cell lung cancer.
Int J Mol Med. 36:1720–1726. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li J, Yu T, Cao J, Liu L, Liu Y, Kong HW,
Zhu MX, Lin HC, Chu DD, Yao M, et al: MicroRNA-148α suppresses
invasion and metastasis of human non-small-cell lung cancer. Cell
Physiol Biochem. 37:1847–1856. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu XG, Zhu WY, Huang YY, Ma LN, Zhou SQ,
Wang YK, Zeng F, Zhou JH and Zhang YK: High expression of serum
miR-21 and tumor miR-200c associated with poor prognosis in
patients with lung cancer. Med Oncol. 29:618–626. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nadal E, Zhong J, Lin J, Reddy RM, Ramnath
N, Orringer MB, Chang AC, Beer DG and Chen G: A microRNA cluster at
14q32 drives aggressive lung adenocarcinoma. Clin Cancer Res.
20:3107–3117. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Skrzypski M, Czapiewski P, Goryca K,
Jassem E, Wyrwicz L, Pawłowski R, Rzyman W, Biernat W and Jassem J:
Prognostic value of microRNA expression in operable non-small cell
lung cancer patients. Br J Cancer. 110:991–1000. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sun Y, Su B, Zhang P, Xie H, Zheng H, Xu
Y, Du Q, Zeng H, Zhou X, Chen C, et al: Expression of miR-150 and
miR-3940-5p is reduced in non-small cell lung carcinoma and
correlates with clinicopathological features. Oncol Rep.
29:704–712. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Võsa U, Vooder T, Kolde R, Fischer K, Välk
K, Tõnisson N, Roosipuu R, Vilo J, Metspalu A and Annilo T:
Identification of miR-374α as a prognostic marker for survival in
patients with early-stage nonsmall cell lung cancer. Genes
Chromosomes Cancer. 50:812–822. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang R, Ye F, Zhen Q, Song T, Tan G, Chu
W, Zhang Y, Lv B, Zhao X and Liu J: MicroRNA-148b is a potential
prognostic biomarker and predictor of response to radiotherapy in
non-small-cell lung cancer. J Physiol Biochem. 72:337–343. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jiang LP, Zhu ZT and He CY: Expression of
miRNA-26b in the diagnosis and prognosis of patients with
non-small-cell lung cancer. Future Oncol. 12:1105–1115. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Rothschild SI, Tschan MP, Jaggi R, Fey MF,
Gugger M and Gautschi O: microRNA-381 represses ID1 and is
deregulated in lung adenocarcinoma. J Thorac Oncol. 7:1069–1077.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Al Khalaf MM, Thalib L and Doi SA:
Combining heterogenous studies using the random-effects model is a
mistake and leads to inconclusive meta-analyses. J Clin Epidemiol.
64:119–123. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sittka A and Schmeck B: MicroRNAs in the
lung. Adv Exp Med Biol. 774:121–134. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang T, Nelson RA, Bogardus A and Grannis
FW Jr: Five-year lung cancer survival: Which advanced stage
nonsmall cell lung cancer patients attain long-term survival?
Cancer. 116:1518–1525. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mizuno K, Mataki H, Seki N, Kumamoto T,
Kamikawaji K and Inoue H: MicroRNAs in non-small cell lung cancer
and idiopathic pulmonary fibrosis. J Hum Genet. 62:57–65. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gyoba J, Shan S, Roa W and Bédard EL:
Diagnosing lung cancers through examination of micro-RNA biomarkers
in blood, plasma, serum and sputum: A review and summary of current
literature. Int J Mol Sci. 17:4942016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang X, Zhang Y, Fu Y, Zhang J, Yin L, Pu
Y and Liang G: MicroRNA-125b may function as an oncogene in lung
cancer cells. Mol Med Rep. 11:3880–3887. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Volinia S, Calin GA, Liu CG, Ambs S,
Cimmino A, Petrocca F, Visone R, Iorio M, Roldo C, Ferracin M, et
al: A microRNA expression signature of human solid tumors defines
cancer gene targets. Proc Natl Acad Sci USA. 103:2257–2261. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Markou A, Zavridou M and Lianidou ES:
miRNA-21 as a novel therapeutic target in lung cancer. Lung Cancer
(Auckl). 7:19–27. 2016.PubMed/NCBI
|
|
39
|
Mei Z, He Y, Feng J, Shi J, Du Y, Qian L,
Huang Q and Jie Z: MicroRNA-141 promotes the proliferation of
non-small cell lung cancer cells by regulating expression of PHLPP1
and PHLPP2. FEBS Lett. 588:3055–3061. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li J, Tan Q, Yan M, Liu L, Lin H, Zhao F,
Bao G, Kong H, Ge C, Zhang F, et al: miRNA-200c inhibits invasion
and metastasis of human non-small cell lung cancer by directly
targeting ubiquitin specific peptidase 25. Mol Cancer. 13:1662014.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Fiori ME, Barbini C, Haas TL, Marroncelli
N, Patrizii M, Biffoni M and De Maria R: Antitumor effect of
miR-197 targeting in p53 wild-type lung cancer. Cell Death Differ.
21:774–782. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Du L, Schageman JJ, Subauste MC, Saber B,
Hammond SM, Prudkin L, Wistuba II, Ji L, Roth JA, Minna JD, et al:
miR-93, miR-98, and miR-197 regulate expression of tumor suppressor
gene FUS1. Mol Cancer Res. 7:1234–1243. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Remon J, Alvarez-Berdugo D, Majem M, Moran
T, Reguart N and Lianes P: miRNA-197 and miRNA-184 are associated
with brain metastasis in EGFR-mutant lung cancers. Clin Trans
Oncol. 18:153–159. 2016. View Article : Google Scholar
|
|
44
|
Chen T, Gao F, Feng S, Yang T and Chen M:
MicroRNA-370 inhibits the progression of non-small cell lung cancer
by downregulating oncogene TRAF4. Oncol Rep. 34:461–468. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wang Y, Cong W, Wu G, Ju X, Li Z, Duan X,
Wang X and Gao H: miR-376α suppresses the proliferation and
invasion of non-small-cell lung cancer by targeting c-Myc. Cell
Biol Int. 42:25–33. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Cao J, He Y, Liu HQ, Wang SB, Zhao BC and
Cheng YS: MicroRNA 192 regulates chemo-resistance of lung
adenocarcinoma for gemcitabine and cisplatin combined therapy by
targeting Bcl-2. Int J Clin Exp Med. 8:12397–12403. 2015.PubMed/NCBI
|
|
47
|
Tu C, Wang F and Wan J: MicroRNA-381
inhibits cell proliferation and invasion in endometrial carcinoma
by targeting the IGF-1R. Mol Med Rep. 17:4090–4098. 2018.PubMed/NCBI
|
|
48
|
Shi L, Xu Z, Wu G, Chen X, Huang Y, Wang
Y, Jiang W and Ke B: Up-regulation of miR-146α increases the
sensitivity of non-small cell lung cancer to DDP by downregulating
cyclin J. BMC Cancer. 17:1382017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhai G, Li G, Xu B, Jia T, Sun Y, Zheng J
and Li J: miRNA-148b regulates radioresistance in non-small lung
cancer cells via regulation of MutL homologue 1. Biosci Rep.
36:e003542016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Zhang S, Gao L, Thakur A, Shi P, Liu F,
Feng J, Wang T, Liang Y, Liu JJ, Chen M, et al: miRNA-204
suppresses human non-small cell lung cancer by targeting ATF2.
Tumour Biol. 37:11177–11186. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Cheng J, Chen Y, Zhao P, Liu X, Dong J, Li
J, Huang C, Wu R and Lv Y: Downregulation of miRNA-638 promotes
angiogenesis and growth of hepatocellular carcinoma by targeting
VEGF. Oncotarget. 7:30702–30711. 2016.PubMed/NCBI
|
|
52
|
Shi GL, Chen Y, Sun Y, Yin YJ and Song CX:
Significance of serum microRNAs in the auxiliary diagnosis of
non-small cell lung cancer. Clin Lab. 63:133–140. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Zhan B, Lu D, Luo P and Wang B: Prognostic
value of expression of microRNAs in non-small cell lung cancer: A
systematic review and meta-analysis. Clin Lab. 62:2203–2211. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Yu N, Zhang Q, Liu Q, Yang J and Zhang S:
A meta-analysis: microRNAs' prognostic function in patients with
nonsmall cell lung cancer. Cancer Med. 6:2098–2105. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Jiang M, Li X, Quan X, Li Xi and Zhou B:
Clinically correlated microRNAs in the diagnosis of non-small cell
lung cancer: A systematic review and meta-analysis. Biomed Res Int.
2018:59309512018. View Article : Google Scholar : PubMed/NCBI
|