Introduction
Neuroendocrine cell tumor is considered a carcinoid
and recognized as a benign tumor (1). However, as a result of subsequent
research, it has come to light that neuroendocrine tumors (NETs) of
the digestive system are malignant (2). Gallbladder NETs (GB-NETs) are rare,
accounting for only 0.5% of all tumors (3). It is usually difficult to diagnose a
GB-NET based on ultrasound (US) examination, abdominal computed
tomography (CT), and abdominal magnetic resonance imaging (MRI)
(4). GB-NETs are often detected
during pathological examination after cholecystectomy performed for
other conditions, such as cholelithiasis or gallbladder benign
polyps (5,6). In addition, it is difficult to diagnose
GB-NET by only conducting preoperative examination.
The current WHO classification of NETs divides them
into subgroups: Low-grade tumors are classified as NET G1, which
exhibit benign behavior; medium-grade tumors as NET G2; high-grade
tumors as neuroendocrine carcinoma (NEC); and tumors comprising
normal adenocarcinoma and NET components as mixed adenoendocrine
carcinoma (7). A majority of GB-NETs
are poorly differentiated and exhibit increased mitotic activity
and clinically aggressive course (3). However, NET G1, which has a benign
behavior, is extremely rare in the gallbladder. Herein, we report
the case of a patient with polypoid GB-NET G1 of the gallbladder
and discuss the development of therapy for this tumor.
Case report
A 50-year-old male was admitted to Naito Hospital
with diarrhea as the main complaint. No abnormalities were found,
particularly in the thoracoabdominal region. Laboratory examination
revealed elevated white blood cell counts (11,760/µl; normal range:
3,500-9,000/µl). CEA and CA19-9 levels were within the normal range
(<37 and <5.0 U/ml, respectively). Abdominal US examination
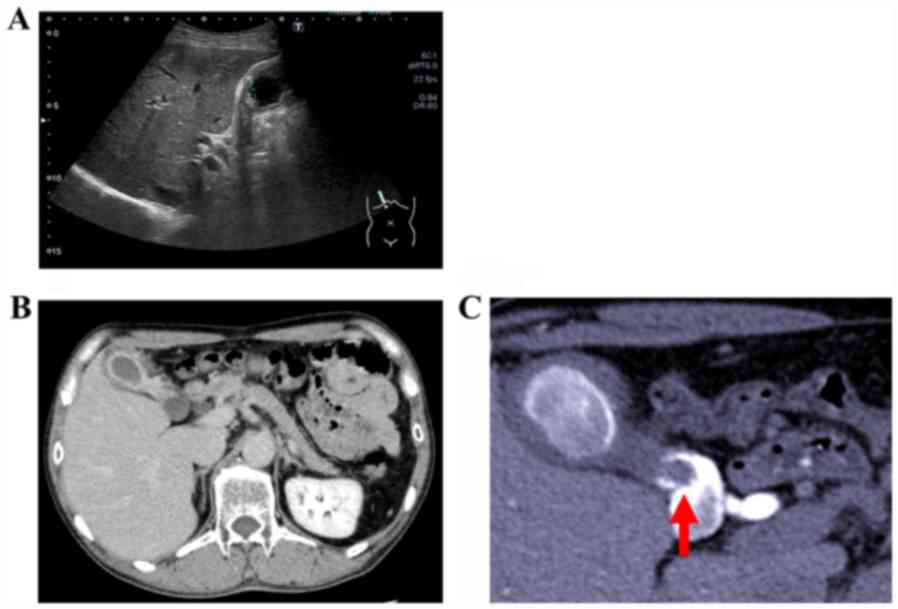
revealed edematous wall thickening in the body and fundus of the
gallbladder (Fig. 1A), raising
suspicion of a cystic lesion. Rokitansky-Ashoff sinus (RAS) was
observed in the thickened area. Some small stones were detected in
the lumen of the RAS; the neck of gallbladder could not be clearly
observed because of extensive wall thickening. A contrast-enhanced
CT scan as well as abdominal US confirmed the presence of RAS and
stones (Fig. 1B). Drip infusion
cholecystocholangiography-CT (DIC-CT) revealed a pedunculated 8-mm
polyp in the gallbladder neck, with multiple stones visible in the
gallbladder (Fig. 1C).
As the patient had not undergone routine abdominal
examinations, we could not determine whether the polyp was growing.
Generally, pedunculated polyps <1 cm are considered to have low
possibility of being cancerous. Thus, the patient was diagnosed as
having a benign small gallbladder polyp and adenomyomatosis, and
laparoscopic cholecystectomy was performed.
Pathological findings
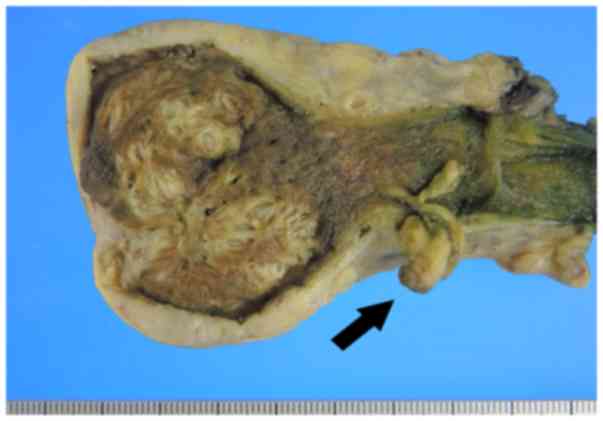
Macroscopically, the gallbladder wall was thickened,
and RAS could be observed clustered in the wall. These observations
supported the diagnosis of diffuse gallbladder adenomyomatosis. In
addition, a pedunculated polyp (10x12 mm) was observed in the neck
of the gallbladder (Fig. 2).
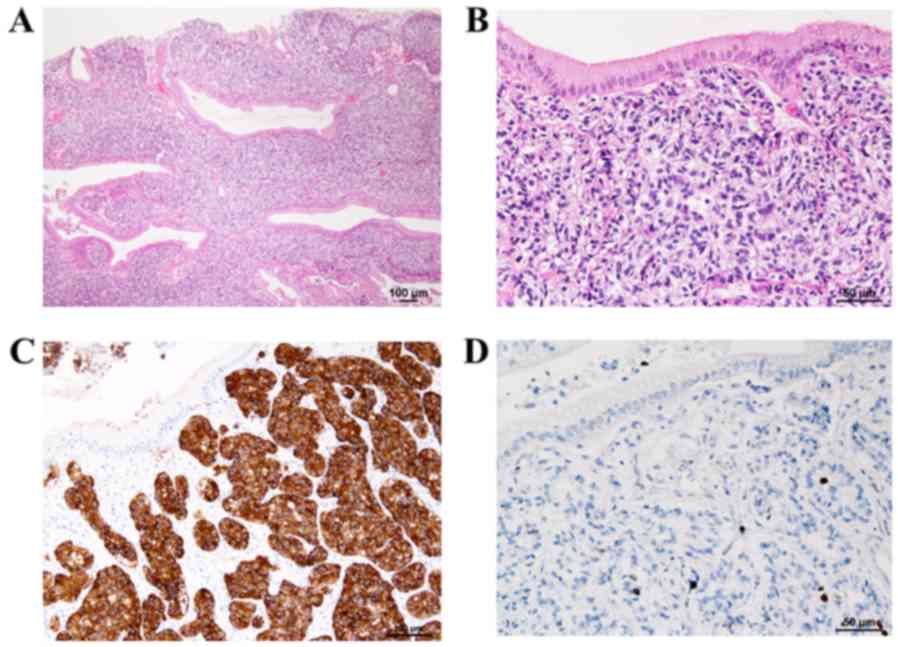
Histopathological examination revealed that the
polypoid lesion contained NET cells in a cord- or ribbon-like
arrangement (Fig. 3A). The tumor
cell morphology revealed isolated as well as disseminated small
oval cells with high nucleus-to-cytoplasm ratios exhibiting a fine
granular chromatin pattern (salt-and-pepper appearance) with slight
aggregations, confirming the presence of small nucleoli (Fig. 3B). No necrosis or mitotic figures
were observed in the background. Immunohistochemical staining with
synaptophysin (Fig. 3C) and
chromogranin A were confined to the tumor, and Ki-67 labeling
showed a labeling index of <1% in the hot spot (Fig. 3D). Based on these findings, the
patient was diagnosed as having polypoid GB-NET in the gallbladder
neck area along with adenomyomatosis and cholecystitis. Tumor
infiltration was limited to the submucosal layer, and there was no
vascular invasion. We determined that because R0 resection was
performed by cholecystectomy, no additional treatment was
administered. The patient is alive and well without recurrence 1
year after the surgery.
Discussion
NETs originate from endocrine cells, and
approximately 60% of the tumors occur in the gastrointestinal
tract, with the frequency of occurrence being highest in the
pancreas and rectum. Primary GB-NETs are rare, representing only
0.5% of all NETs (3). Therefore,
little is known about their biological behavior (3). Neuroendocrine cells do not exist within
the normal gall bladder or are present in small amounts in the
epithelium. Various mechanisms have been proposed to explain the
pathogenesis of GB-NET, and coexistence of cholelithiasis and
gallbladder stones could be one such important factor (3). The proportion of neuroendocrine cells
increases during chronic inflammation in the affected metaplastic
epithelium, which may become the site of development of NETs
(6). Generally, individuals with
GB-NET do not exhibit any specific symptoms (8,9), and the
proportion of hormone-producing GB-NETs is unknown because of the
small number of reported cases. According to previous findings from
Japan, the percentage of pedunculated polyps in gall bladder NETs
is higher than that in gastrointestinal NETs (10). However, it is difficult to
distinguish GB-NETs from benign polyps because of the absence of
specific findings on CT or MRI (4).
Therefore, GB-NET is often misdiagnosed as benign prior to initial
surgery (5,6). In the present case, diagnosis was
complicated owing to the presence of another gallbladder disease.
Polypoid lesion was detected in the neck of the gallbladder on
DIC-CT; however, the polyp was small (8 mm), and there were no
other specific findings. Therefore, we could not establish the
presence of a GB-NET using other physical and imaging techniques,
including contrast-enhanced CT. In case of gastrointestinal NETs,
other than those of the small bowel and pancreas, small tumors
(<2 cm) and G1 tumors are believed to be successfully treated
with local excision, even endoscopically, without lymph node
dissection (11). The extent of
surgery usually depends on the presence (or possibility) of lymph
node metastasis. However, as the number of reported GB-NET cases is
relatively small, risk factors for lymph node metastasis in GB-NET
remain to be clarified. Yokoyama et al reported seven cases,
two (28.6%) of which had carcinoid tumors measuring <1 cm, with
metastasis at presentation, whereas the remaining five cases had
tumors measuring ≥3 cm, with all five cases (100%) developing
metastases (12). These observations
indicate that the risk of metastasis increases with increase in
tumor size. Conversely, Hirose et al reported that even G1
tumors of extremely small size (≤1 cm) metastasize to the
gallbladder lymph nodes, and extended additional surgery is
required in such cases (13). In
general, in cases of gastrointestinal and pancreatic NETs, high
mitotic counts, high Ki-67 proliferation index values, large tumor
diameters, and high invasion depths are considered risk factors for
the existence of lymph node or distant metastasis (14-16).
However, little is known about the risk factors for lymph node or
distant metastasis in GB-NET cases. Furthermore, there is no
evidence regarding the efficacy of extended surgery or adjuvant
chemotherapy in GB-NET G1 and G2 cases (17). If metastasis is observed during the
histopathological examination of the cystic duct node, it should be
determined whether a radical second resection with regional
lymphadenectomy is the appropriate treatment of choice (13). In cases with cystic duct lymph node
metastasis, radical second resection with lymph node dissection
should be considered as an option for accurate nodal evaluation,
based on the reported outcomes of incidental gallbladder cancer
(18). Our patient had NET-G1 with
limited infiltration depth without lymph vascular invasion and
lymph node metastasis. Therefore, he was closely followed up
without adjuvant therapy. Regarding the prognosis, Eltawil et
al reported that 5-year survival rates for tumors classified as
carcinoids-neuroendocrine or small-cell carcinoma, are 36.9 and 0%,
respectively (5). By contrast,
Koizumi et al reported that a tumor did not recur in 68.8%
cases in which GB-NET was resected and that the average
disease-free period of surviving cases was 28.7 months (range:
6-180 months). The substantial difference between the two
aforementioned reports may be attributed to the difference in the
pathology or stage of the tumor. The report by Eltawi et al
could have included some case series with no stratification of the
type of gallbladder NET and NEC. Some researchers advocate that in
case of GB-NET, facilitated invasion and lymphatic metastasis to
adjacent organs contribute to the high grade of malignancy
(10). Further research is necessary
to evaluate imaging findings prior to surgery, to determine the
appropriate surgical procedure, and to identify the potential
prognostic factors.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during the present
study are included in this article.
Authors' contributions
SN collected the patient data and was a major
contributor to writing the manuscript. MN contributed to analysis
and interpretation of data, and assisted in the preparation of the
manuscript. YN and YK performed the histological examination. SHo,
NY, and TK were in charge of the operation of the patient in the
hospital. HN, and SHa reviewed and contributed to the discussion
and critical review of draft versions of the manuscript. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient provided informed consent for
publication of the data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
La Rosa S, Bongiovanni M and Uccella S:
Pathology of neuroendocrine neoplasms: Morphological,
immunophenotypical, and circulating molecular markers. In: Atlas of
thyroid and neuroendocrine tumor markers. Giovanella L. (eds).
Springer, Cham, pp13-38, 2018.
|
|
2
|
Klöppel G, Perren A and Heitz PU: The
gastroenteropancreatic neuroendocrine cell system and its tumors:
The WHO classification. Ann N Y Acad Sci. 1014:13–27.
2004.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Eltawil KM, Gustafsson BI, Kidd M and
Modlin IM: Neuroendocrine tumors of the gallbladder: An evaluation
and reassessment of management strategy. J Clin Gastroenterol.
44:687–695. 2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Zou YP, Li WM, Liu HR and Li N: Primary
carcinoid tumor of the gallbladder: A case report and brief review
of the literature. World J Surg Oncol. 8(12)2010.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Mezi S, Petrozza V, Schillaci O, La Torre
V, Cimadon B, Leopizzi M, Orsi E and La Torre F: Neuroendocrine
tumors of the gallbladder: A case report and review of the
literature. J Med Case Rep. 5(334)2011.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Jun SR, Lee JM, Han JK and Choi BI:
High-grade neuroendocrine carcinomas of the gallbladder and bile
duct: Report of four cases with pathological correlation. J Comput
Assist Tomogr. 30:604–609. 2006.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Komminoth P, Arnold R, Capella C, Klimstra
DS and Kloppel G: Neuroendocrine neoplasms of the gallbladder and
extrahepatic bile ducts. In: Bosman FT Carneiro F Hruban RH Theise
HD, editors. WHO classification of tumours of the digestive system.
Lyon: International Agency for Research on Cancer, pp. 274-276,
2010.
|
|
8
|
Monier A, Saloum N, Szmigielski W,
Alrashid A and Napaki SM: Neuroendocrine tumor of the gallbladder.
Pol J Radiol. 80:228–231. 2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Porter JM, Kalloo AN, Abernathy EC and Yeo
CJ: Carcinoid tumor of the gallbladder: Laparoscopic resection and
review of the literature. Surgery. 112:100–105. 1992.PubMed/NCBI
|
|
10
|
Yotsumoto H, Godai T, Suematsu H, Yamauchi
M, Fujikawa H, Fukano F, Tamura I, Rino Y, Suzuki S and Masuda M: A
neuroendocrine tumor of the gallbladder. Jpn J Gastroenterol Surg.
51:263–270. 2018. View Article : Google Scholar
|
|
11
|
Kulke MH, Benson AB III, Bergsland E,
Berlin JD, Blaszkowsky LS, Choti MA, Clark OH, Doherty GM, Eason J,
Emerson L, et al: Neuroendocrine tumors. J Natl Compr Canc Netw.
1:724–764. 2012.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yokoyama Y, Fujioka S, Kato K, Tomono H,
Yoshida K and Nimura Y: Primary carcinoid tumor of the gallbladder:
Resection of a case metastasizing to the liver and analysis of
outcomes. Hepatogastroenterology. 47:135–139. 2000.PubMed/NCBI
|
|
13
|
Hirose Y, Sakata J, Endo K, Takahashi M,
Saito R, Imano H, Kido T, Yoshino K, Sasaki T and Wakai T: A 0.8-cm
clear cell neuroendocrine tumor G1 of the gallbladder with lymph
node metastasis: A case report. World J Surg Oncol.
16(150)2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Saund MS, Al Natour RH, Sharma AM, Huang
Q, Boosalis VA and Gold JS: Tumor size and depth predict rate of
lymph node metastasis and utilization of lymph node sampling in
surgically managed gastric carcinoids. Ann Surg Oncol.
18:2826–2832. 2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Sato Y, Hashimoto S, Mizuno K, Takeuchi M
and Terai S: Management of gastric and duodenal neuroendocrine
tumors. World J Gastroenterol. 22:6817–6828. 2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Chatzipantelis P, Konstantinou P,
Kaklamanos M, Apostolou G and Salla C: The role of cytomorphology
and proliferative activity in predicting biologic behavior of
pancreatic neuroendocrine tumors: A study by endoscopic
ultrasound-guided fine-needle aspiration cytology. Cancer.
17:211–216. 2009.PubMed/NCBI View Article : Google Scholar
|
|
17
|
National Comprehensive Cancer Network:
NCCN Clinical Practice Guidelines in Oncology. Neuroendocrine
tumors. version 1, 2011. http://www.lecba-rakoviny.cz/dokumenty/NCCN_Guidelines_neuroendocrine_2011.pdf.
Accessed December 18, 2019.
|
|
18
|
Wakai T, Shirai Y and Hatakeyama K:
Radical second resection provides survival benefit for patients
with T2 gallbladder carcinoma first discovered after laparoscopic
cholecystectomy. World J Surg. 26:867–871. 2002.PubMed/NCBI View Article : Google Scholar
|