Introduction
Amyloidosis is a rare progressive disorder that
includes a group of diseases characterized by accumulation of
pathologic deposits of amyloids in the tissues. The amyloids are
protein polymers formed of identical monomer units. Pathological
amyloids are usually formed from misfolded proteins. The deposits
of amyloids situated either intracellularly or extracellularly
alter the normal function of organs. There are over 23 proteins
that can form amyloid fibers in vivo (1).
There are two main types of amyloidosis, namely
systemic and localized. Localized amyloidosis is rare, and it
usually occurs in the head and neck region, mostly in the larynx
and trachea (2,3). Tongue involvement is common in systemic
amyloidosis and can be diffuse as macroglossia or localized
(4-7).
Localized tongue amyloidosis is extremely rare. This study presents
a rare case of localized amyloidosis at the level of the tongue
base and also reviews the important aspects of the literature for
localized amyloidosis.
Case report
A 36-year-old female with a history of dysmenorrhea
and menorrhagia due to uterine fibroids was initially admitted for
hysterectomy in the ‘Queen Mary’ Hospital of Bucharest, Romania.
Her complaint was dysmenorrhea and menometrorrhagia. Uterine
ultrasound was consistent with the diagnosis of uterine fibroid
dimensions 45/6/44 mm. The gynecologists did not perform the
surgery. The intervention was terminated due to difficult
intubation. A biopsy of the uterine lesions was not available.
During the intubation attempt, a right-sided tongue tumor was
noted, thus rendering intubation difficult and the hysterectomy was
thus aborted. Subsequently, the patient was referred to the Ear
Nose Throat (ENT) Department of ‘Queen Mary’ Hospital of Bucharest
for further evaluation.
At presentation, the patient did not report dyspnea,
dysphonia, dysphagia, hemoptysis, or dysarthria. The patient did
not report weight loss, fever, chills, joint pain, skin changes,
rashes, numbness or paresthesia. An analysis of her past medical
history revealed pulmonary tuberculosis treated 10 years prior (she
had completed anti-TB treatment and had a complete recovery),
hypercholesterolemia, gastric ulcer and uterine fibroid. A physical
examination revealed a yellowish mass on the tongue base, on the
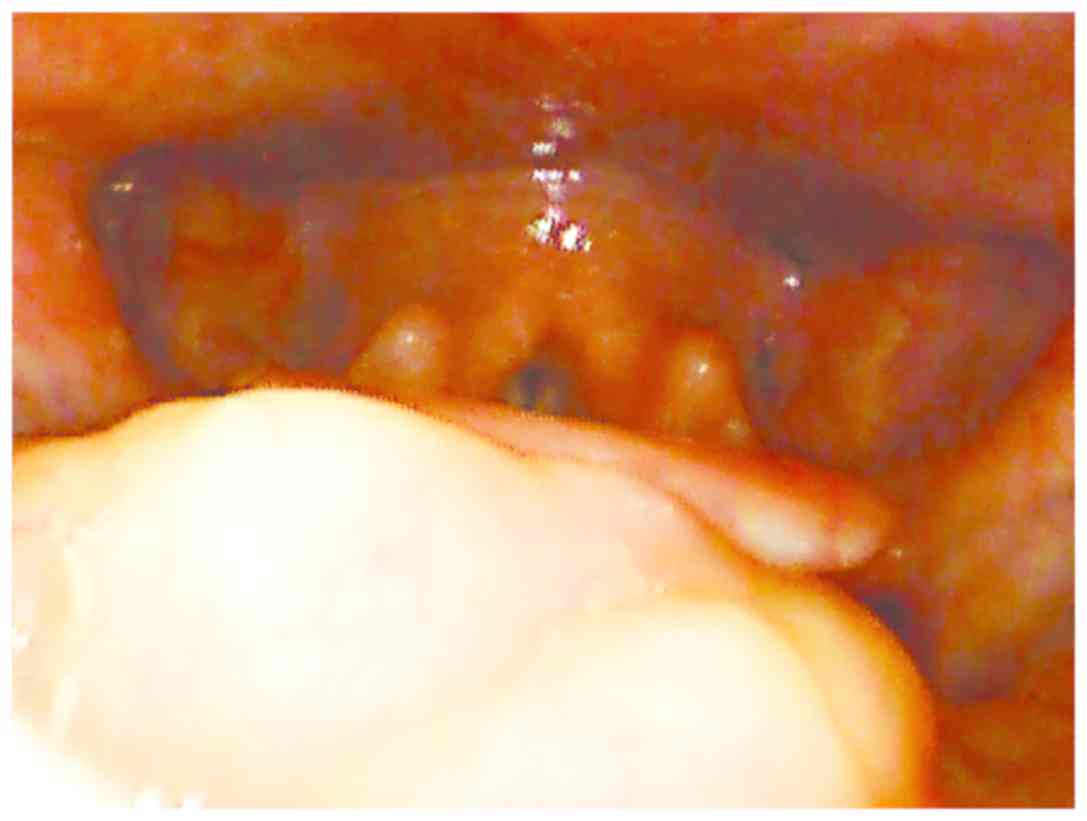
right side. The mass was non-friable and soft in nature. A flexible
endoscopy was performed, revealing a diameter of approximately 2-3
cm and a thickness of approximately 1 cm that occupied the right
vallecula (Fig. 1).
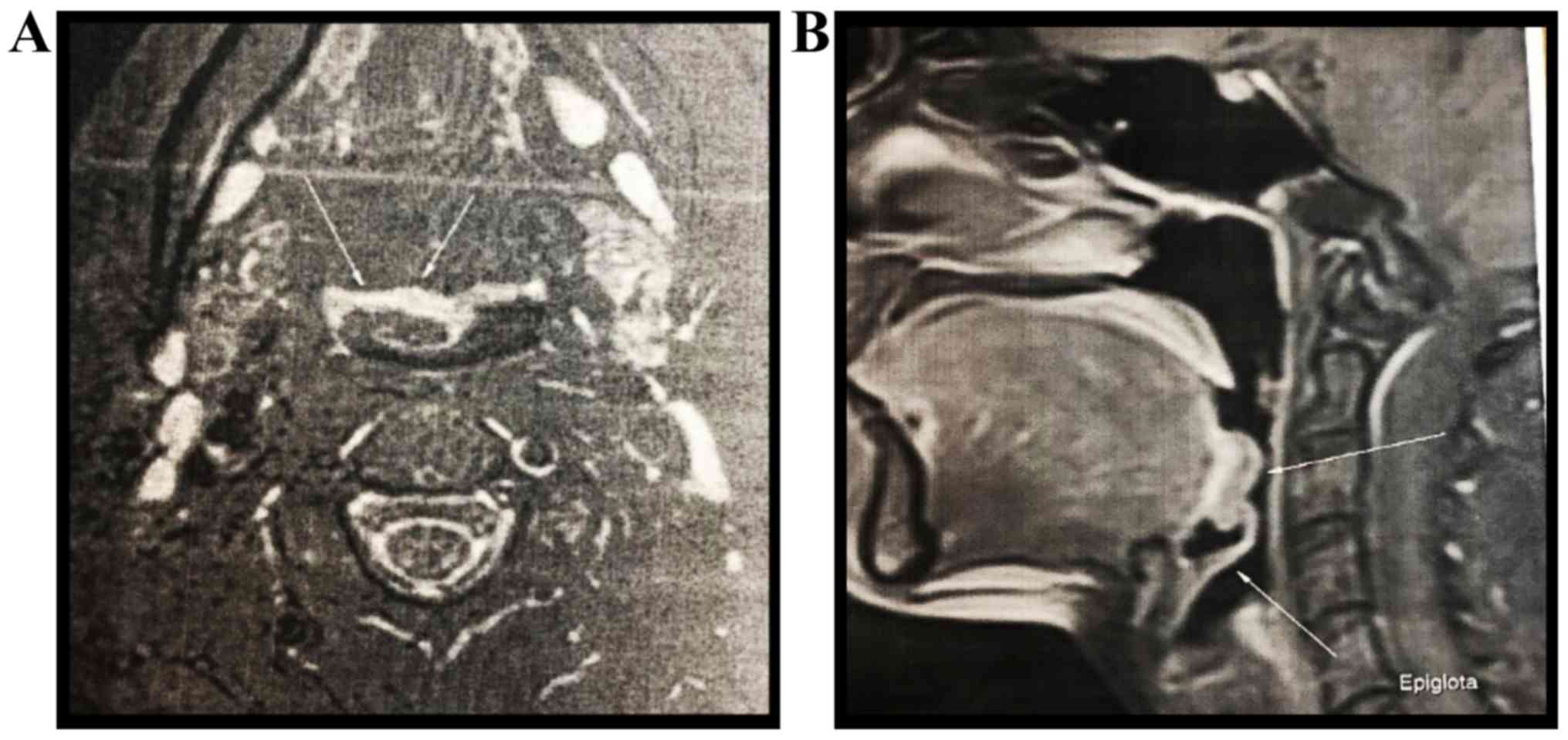
A head and neck MRI with and without contrast
revealed a polypoid protrusive mass with a length/width/depth of
20/7/17.8 mm at the base of the tongue, on the right, partially
occupying the right vallecula without evident signs of invasion
(Fig. 2). A biopsy of the tumor was
performed using a biopsy clamp and several bioptic fragments from
the tumor were obtained.
A gross examination revealed multiple tissue
fragments with variable dimensions ranging from 0.5/0.5/0.3 cm and
0.5/0.5/0.5 cm, with a waxy, starch-like, translucent and firm
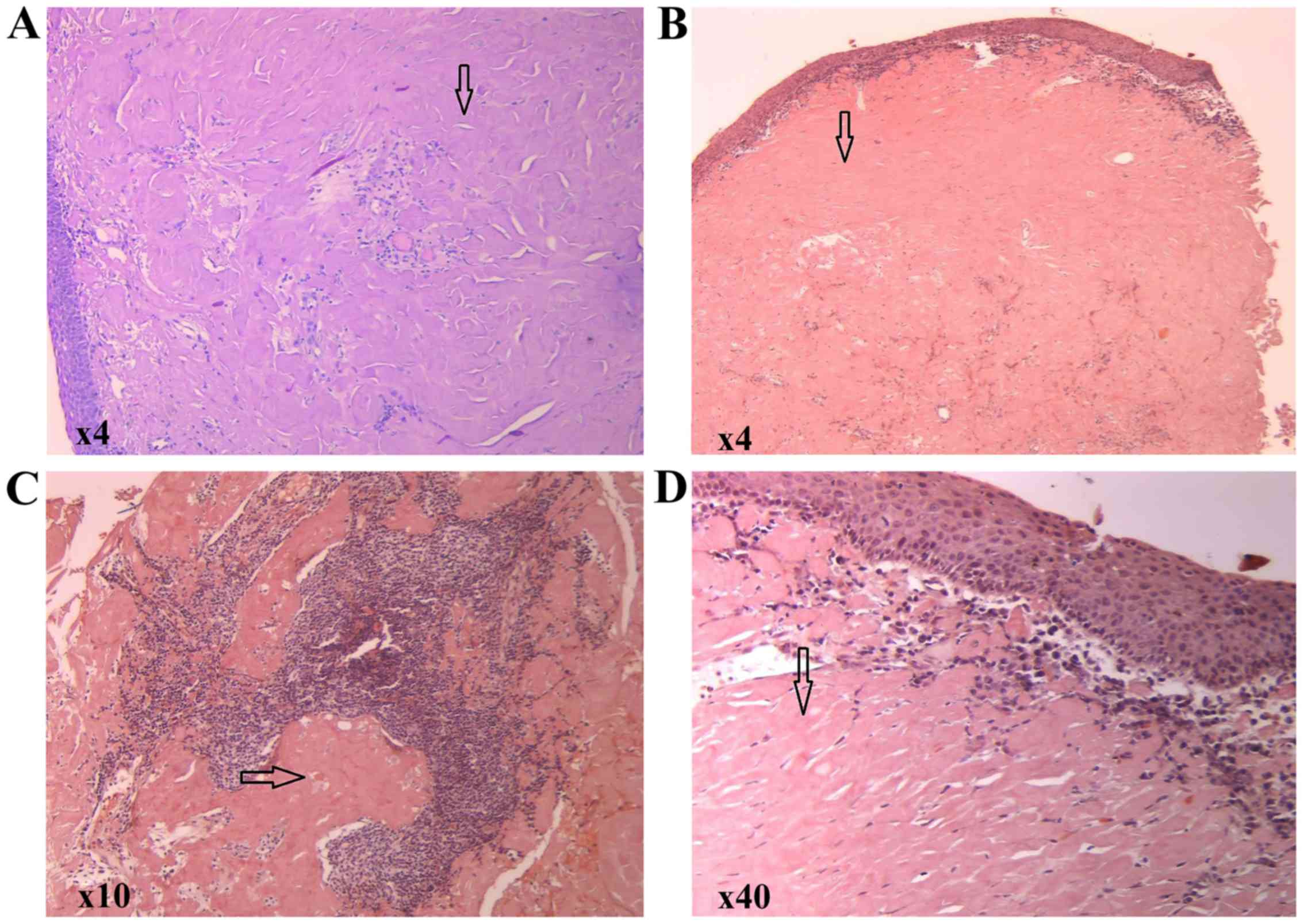
consistency. A microscopic examination following hematoxylin and
eosin staining revealed subepithelial deposits of an acellular,
extracellular, eosinophilic and homogenous matrix material; a
sparse inflammatory infiltrate composed of lymphocytes and plasma
cells was identified. Congo red staining was performed, revealing
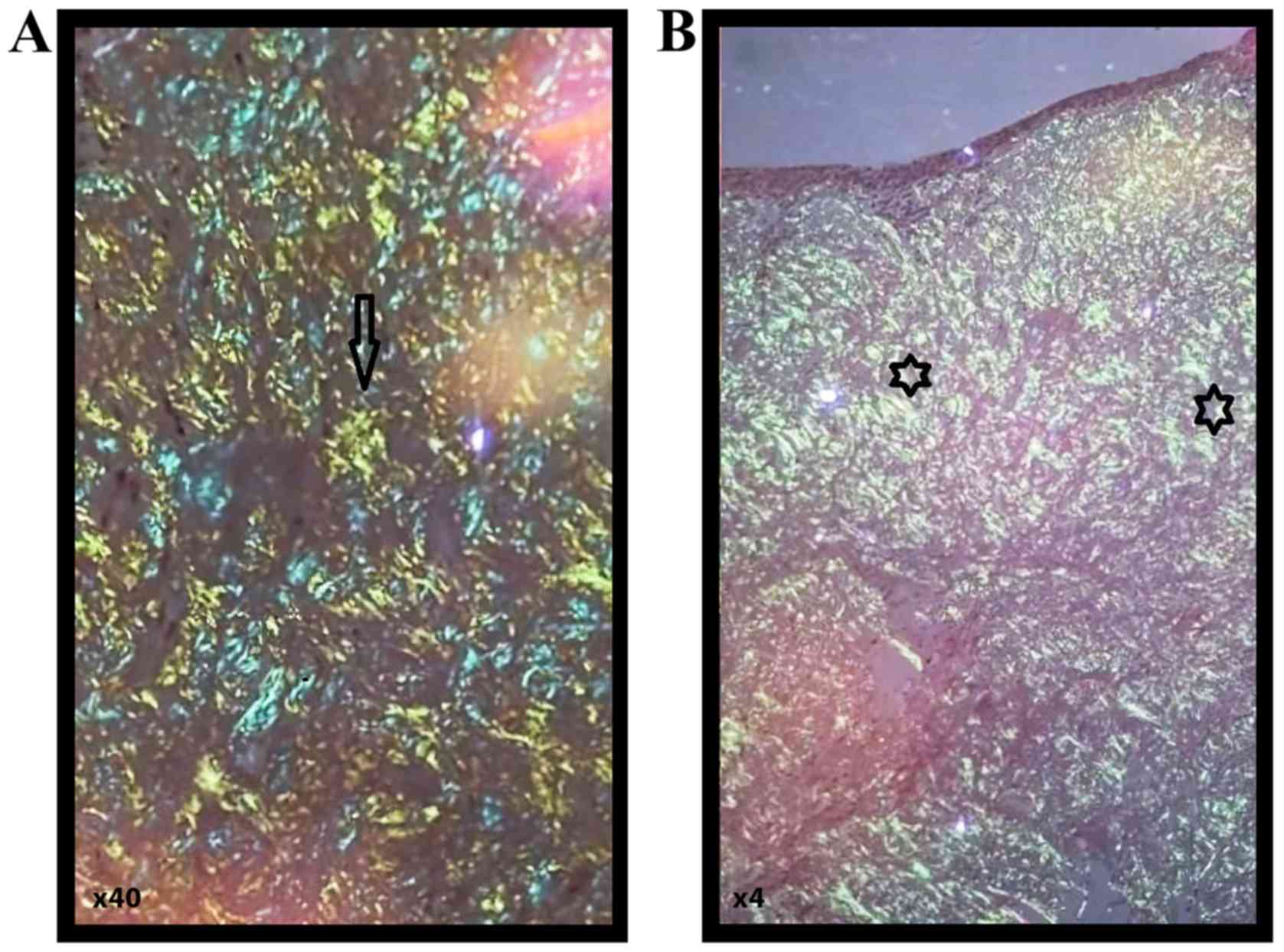
red homogenous subepithelial deposits (Fig. 3). Red homogenous subepithelial
deposits turn into an apple-green birefringence under polarized
light (magnification, x40 and x4) (Fig.
4). Immunohistochemical staining was positive for CD20,
indicating B lymphocytes and light-chain lambda amyloid deposits,
and was negative for amyloid deposits.
The consulting hematologist performed a thorough
evaluation of the amyloidosis, including a chest X-ray, 2D
echocardiogram, NT-pro-brain natriuretic peptide, liver fibroscan,
immunoelectrophoresis and the Schirmer test. The results of all
these tests were within normal ranges, without any signs to suggest
systemic disease. The patient refused to undergo fat tissue
biopsy.
Discussion
The classification of amyloidosis is often based on
clinicopathological criteria. In this scheme i) primary amyloidosis
is systemic, but has no identifiable cause (Table I); ii) secondary amyloidosis is
systemic and is often caused by a chronic disease, such as
tuberculosis or rheumatoid arthritis; iii) amyloidosis associated
with multiple myeloma is multisystemic; and iv) localized
amyloidosis is characterized by a lack of evidence of systemic
amyloidosis or underlying chronic disease. It is noteworthy that a
number of cases with localized amyloidosis have been associated
with Sjögren's syndrome (8,9).
 | Table ITests used to rule out systemic
amyloidosis. |
Table I
Tests used to rule out systemic
amyloidosis.
| Tests | Results |
|---|
| Chest radiograph | Normal |
| Echocardiography | Normal |
| NT pro BNP | In normal range |
| Liver fibroscan | Within normal
limits |
| Basic laboratory
investigation | Unremarkable |
|
Unoelectrophoresis | Within normal
limits |
| Schirmer | Normal |
The modern classification of amyloidosis is chemical
and is based on the type of protein that is deposited and
characterized as AA or AL amyloidosis. ‘A’ stands for amyloid,
followed by the fibril protein abbreviation. For example,
light-chain fibril proteins are abbreviated as ‘L’; thus, AL
amyloidosis indicates light-chain amyloidosis (10). AL amyloidosis occurs when the bone
marrow produces abnormal antibodies that cannot be broken down. The
antibodies are deposited in the tissues as amyloid, interfering
with normal functions. AA indicates serum A amyloidosis.
Systemic amyloidosis is determined by different
types of amyloid deposits, as follows: i) AA amyloidosis, which has
as a precursor, the serum amyloid A, is the most frequent systemic
amyloidosis and it usually involves the liver, kidneys and spleen
(11). It is implicated in various
rheumatological disorders, hematological malignancies, autoimmune
disorders, hematological malignancies (Hodgkin's disease and
non-Hodgkin's lymphoma and multiple myeloma), rheumatologic
diseases (rheumatoid arthritis, ankylosing spondylitis and juvenile
idiopathic arthritis), autoimmune disorders (psoriasis and
psoriatic arthritis). Previously-treated tuberculosis and chronic
inflammation due to recurrent respiratory tract infections were
considered to be responsible for AA amyloidosis (12). ii) AL (light-chain) amyloidosis is
another type of systemicamyloidosis, in which the precursor protein
is a clonal immunoglobulin light-chain or light-chain fragment. The
organs involved are usually the heart, kidneys, gastrointestinal
tract, respiratory tract and peripheral nervous system. iii) AH
amyloidosis is produced by the fibrils of heavy chain sequences of
immunoglobulin. There are also transthyretin amyloidosis (ATTR),
beta 2 microglobulin amyloidosis (Abeta2M) and other types of
systemic amyloidosis.
Localized amyloidosis is formed by the local
production and deposit of fibers in different tissues. In localized
amyloidosis, plasma cells secrete light-chain immunoglobulins that
misfold and are deposited locally (13-15).
Localized AL amyloidosis (‘amyloidoma’) represents a true plasma
cell neoplasm and not a pseudotumor. Specifically, although
localized AL amyloidosis appears as a tumor according to a
classical definition, the basic lesion is a neoplasm in the form of
a clonal expansion of one plasma cell (3). The sites of development of localized
amyloidosis include the bladder, trachea, bronchi, larynx,
gastrointestinal tract, orbit, tonsils, lymph nodes and skin. One
of the most well-studied localized types of amyloidosis is the one
localized in the brain tissue typical in Alzheimer's disease
(16).
The exact etiology of localized amyloidosis is not
yet known. Given that the amyloidosis is usually formed in mucous
membranes, it has been suggested that the prolonged reaction of
tissue plasma cells to environmental antigen is the first step
(17). These reactive cells exhibit
an increase production of immunoglobulin light-chains that are
amyloidogenic. The transformation of these immunoglobulin light
chains to insoluble fibrils by macrophages seems to be a key step
in the formation of amyloidoma. This theory is supported by the
fact that giant cells are a common finding in cases of localized
amyloidoma (3).
The larynx is the most involved organ in head and
neck localized amyloidosis. In contrast to systemic amyloidosis in
which tongue involvement is common, leading to extensive
enlargement of the tongue known as macroglossia (5), the tongue is seldom a place of
appearance for the localized amyloidosis and a small number of
cases has been reported (7) and can
be expressed as nodules of local accumulation of protein deposits
(18). The most extensive case
series has been published by the Mayo Clinic in 2013; they reported
6 cases of localized tongue amyloidosis in a 42-year period of time
(7).
The clinical suspicion of amyloidosis requires
histological confirmation (19). The
majority of localized oral amyloidoses are easily biopsied under
local anesthesia. Tissue biopsy and microscopic examination is
usually sufficient to establish a diagnosis. The amyloid stained
with hematoxylin and eosin staining in light microscopy, appears as
a homogenous eosinophilic, amorphous substance. However, following
staining with Congo red and placed under polarized light, it
reveals an apple-green birefringence. If the result is positive,
the next step would be to establish the subtype of amyloidosis.
This is usually tested using serum or urine immunofixation
electrophoresis to search for a clonal disorder. In the case of a
negative result, immunohistochemistry is useful in finding K or
lamda light-chains to confirm a clonal disorder - AL and amyloid A
to confirm AA. Nevertheless, this type of testing is neither
specific nor precise. Future studies should be performed on a
larger cohort of patients for more reliable results (20).
Oral localized amyloidosis can easily be found on
the tongue, which can be enlarged diffusely due to macroglossia or
as nodular deposits (21). If the
disorder is extensive, the tongue usually loses its elasticity and
may be firm, fissured, ulcerated and occasionally red and painful.
Function impairment is present, interfering with speech, chewing
and swallowing. Macroglossia is rare in type AA and more frequent
in type AL, although associating the type of amyloid to the type of
clinical presentation is not recommended. Tissue biopsy is
mandatory for histological confirmation.
Current recommendations regarding amyloidosis
diagnosis are the following: i) Congo red stain is currently the
gold standard for amyloid detection; ii) the type of amyloid must
be identified microscopically or immunohistochemically, not solely
on clinical or DNA studies; iii) immunohistochemistry has to be
performed cautiously and amyloidosis suspicion must be completed
using more sophisticated methods in referral centers (22).
The prognosis for patients with AL amyloidosis is
linked to the extent of the disease. Localized amyloidosis has an
excellent prognosis and rarely progresses to systemic disease. A
study on 606 cases of localized amyloidosis over a period of 30
years revealed that only one case progressed to a systemic AL
(4,23). Therefore, patients with localized
form usually do not require treatment with systemic therapy.
Management can be supportive or localized. The most common
approaches include local excision, ablation or clinical
observation. Surgical excision or ablation of the amyloidoma is
usually the treatment of choice and is indicated for symptomatic
management to ensure the airway patency; however, recurrence is
frequent and revisions may be required (24).
Tongue amyloidoma is a rare presentation of
localized amyloidosis. A limited number of cases of localized
amyloidosis have been previously reported to involve the tongue
(2,3), in contrast to systemic amyloidosis that
can commonly involve the tongue (4-7).
This study presents a rare localized amyloidoma of
the tongue. Taking the history into consideration, physical
findings, head and neck MRI findings and histopathological results,
using Congo red stain, the diagnosis of localized lambda
light-chain amyloidosis of the tongue base was made. Although,
active TB is associated with systemic amyloidosis (2,12), as
the patient had a complete recovery, and investigations did not
reveal any signs of systemic amyloidosis, the amyloidosis was
considered localized.
Usually, localized amyloidosis does not progress to
systemic involvement and the prognosis is excellent. Surgical
resection is the treatment of choice; however, clinical follow-up
can also be performed if the airway is not compromised. In the
current case, the therapeutic plan presented herein was initially
the removal of the tumor by laser or electrocautery, in suspension
laryngoscopy; however, the patient refused. The pros and cons of
the procedure were extensively discussed with the patient. The
patient refused treatment at this point and follow-up for further
clinical monitoring was arranged. The 2-year follow up of the
patient did not reveal any change in the size of the tumor, or
further symptomatology.
In summary, i) the cases of localized amyloidosis of
the tongue remain rare; ii) the diagnosis of systemic examination
should be evidence based and necessary investigations to rule out
systemic causes of amyloidosis; iii) although the localized
amyloidomas may have a localized mass effect, prognosis is overall
good; and iv) the etiopathogenesis of localized amyloidosis is not
yet clear and further investigations are required.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during the current
study are included in this published article or are available from
the corresponding author upon reasonable request.
Authors' contributions
All the authors (GM, AE, DE, AOD, SGD, DPV, CN, DAS
and MM) were involved in conceiving and designing the study. GM, AE
and MM contributed to sample collection. DC, AOD, SGD and DPV
performed the statistical analysis. DC, AOD, GM, SD, DPV and MM
drafted and wrote the manuscript. AOD, DC, DAS and CN gave advice
on the experimental design, interpreted the results and critically
revised the manuscript. All authors have read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
The access of the database for the purpose of this
study was approved by the Ethics Committee of ‘Saint Mary’ Clinic
Hospital, Bucharest, Romania. As the hospital to which the patient
was admitted is a teaching hospital, all patients admitted to our
hospital signed a written consent by which they agree that their
medical data can be used in scientific studies.
Patient consent for publication
The participation in the study was approved by the
patient and she gave her approval for the patient information
presented herein to be published.
Competing interests
DAS is the Editor-in-Chief for the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision, for this article.
All the other authors declare that they have no competing
interests.
References
|
1
|
Buxbaum JN: The systemic amyloidoses. Curr
Opin Rheumatol. 16:67–75. 2004.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kerner MM, Wang MB, Angier G, Calcaterra
TC and Ward PH: Amyloidosis of the head and neck. A
clinicopathologic study of the UCLA experience, 1955-1991. Arch
Otolaryngol Head Neck Surg. 121:778–782. 1995.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Westermark P: Localized AL amyloidosis: A
suicidal neoplasm? Ups J Med Sci. 117:244–250. 2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Mahmood S, Sachchithanantham S, Bridoux F,
Lane T, Rannigan L, Foard D, Sayed R, Patel K, Fontanna M, Whelan
C, et al: Risk of Progression of localised amyloidosis to systemic
disease in 606 patients over 30 years. Blood. 122(3143)2013.
View Article : Google Scholar
|
|
5
|
van der Wal N, Henzen-Logmans S, van der
Kwast WA and van der Waal I: Amyloidosis of the tongue: A clinical
and postmortem study. J Oral Pathol. 13:632–639. 1984.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Pentenero M, Davico Bonino L, Tomasini C,
Conrotto D and Gandolfo S: Localised oral amyloidosis of the
palate. Amyloid. 13:42–46. 2006.PubMed/NCBI
|
|
7
|
O'Reilly A, D'Souza A, Lust J and Price D:
Localized tongue amyloidosis: A single institutional case series.
Otolaryngol Head Neck Surg. 149:240–244. 2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Moon AO, Calamia KT and Walsh JS: Nodular
amyloidosis: Review and long-term follow-up of 16 cases. Arch
Dermatol. 139:1157–1159. 2003.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Biewend ML, Menke DM and Calamia KT: The
spectrum of localized amyloidosis: A case series of 20 patients and
review of the literature. Amyloid. 13:135–142. 2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Westermark P, Benson MD, Buxbaum JN, Cohen
AS, Frangione B, Ikeda SI, Masters CL, Merlini G, Saraiva MJ and
Sipe JD: A primer of amyloid nomenclature. Amyloid. 14:179–183.
2007.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Hazenberg BP: Amyloidosis: A clinical
overview. Rheum Dis Clin North Am. 39:323–345. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Balwani MR, Kute VB, Shah PR, Wakhare P
and Trivedi HL: Secondary renal amyloidosis in a patient of
pulmonary tuberculosis and common variable immunodeficiency. J
Nephropharmacol. 4:69–71. 2015.PubMed/NCBI
|
|
13
|
Berk JL, O'Regan A and Skinner M:
Pulmonary and tracheobronchial amyloidosis. Semin Respir Crit Care
Med. 23:155–165. 2002.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Charlot M, Seldin DC, O'Hara C, Skinner M
and Sanchorawala V: Localised amyloidosis of the breast: a case
series. Amyloid. 18:72–75. 2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
O'Regan A, Fenlon HM, Beamis JF Jr, Steele
MP, Skinner M and Berk JL: Tracheobronchial amyloidosis. The Boston
University experience from 1984 to 1999. Medicine (Baltimore).
79:69–79. 2000.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Hardy J and Selkoe DJ: The amyloid
hypothesis of Alzheimer's disease: Progress and problems on the
road to therapeutics. Science. 297:353–356. 2002.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Krishnan J, Chu WS, Elrod JP and Frizzera
G: Tumoral presentation of amyloidosis (amyloidomas) in soft
tissues. A report of 14 cases. Am J Clin Pathol. 100:135–144.
1993.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kubota K, Ito R, Furudate K, Kon T,
Nakagawa H and Kobayashi W: Localized AL amyloidosis of the tongue:
A case report and literature review. J Oral Maxillofac Surg Med
Pathol. 29:142–145. 2017. View Article : Google Scholar
|
|
19
|
Thompson L and Bishop J: Head and Neck
Pathology. 3rd edition. A Volume in the Series: Foundations in
Diagnostic Pathology. Elsevier, 2019.
|
|
20
|
Binmadi N, Intapa C, Chaisuparat R, Akeel
S, Sindi A and Meiller T: Immunophenotyping Oral Amyloidosis for
the Precise Identification of the Biochemical Forms: A
Retrospective Study. Open Dent J. 12:1036–1042. 2018. View Article : Google Scholar
|
|
21
|
Picken MM: Amyloidosis-where are we now
and where are we heading? Arch Pathol Lab Med. 134:545–551.
2010.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yamagata K and Bukawa H: Oral localised
amyloidosis. In: Amyloidosis - An Insight to Disease of Systems and
Novel Therapies. IntechOpen, London, 2011. https://www.intechopen.com/books/amyloidosis-an-insight-to-disease-of-systems-and-novel-therapies/oral-localized-amyloidosis.
Accessed November 16, 2011.
|
|
23
|
Girnius S: Overview of systemic and
localised amyloidosis. Rev Health Care. 4:231–247. 2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Wald MH: Clinical studies of secondary
amyloidosis in tuberculosis. Ann Intern Med. 43:383–395.
1955.PubMed/NCBI View Article : Google Scholar
|