Introduction
Calcifying fibrous pseudotumor (CFP) are rare benign
lesions first reported by Rosenthal and Abdul-Karim in
1988(1). Pelvic CFP was first
reported by Saglam et al in 2005(2). Recently, we found an interesting CFP in
the pelvic cavity. It was the first case of pelvic CFP in China.
Because of the low morbidity and non-significant clinical symptoms,
preoperative diagnosis of CFP is difficult. Here we provide the
clinical and pathological features of this tumor, and we review CFP
cases reported over the past ten years. We also compared the
characteristics of this disease between China and other countries,
hoping to provide new concepts for clinical diagnosis and
treatment.
Case report
A 67-year-old man presented with mild abdominal
discomfort on 1 month duration. The patient had no history of
trauma or surgery. Physical examination suggested a 1.5x2.0 cm lump
in the left lower abdomen, characterized as hard, painless, and
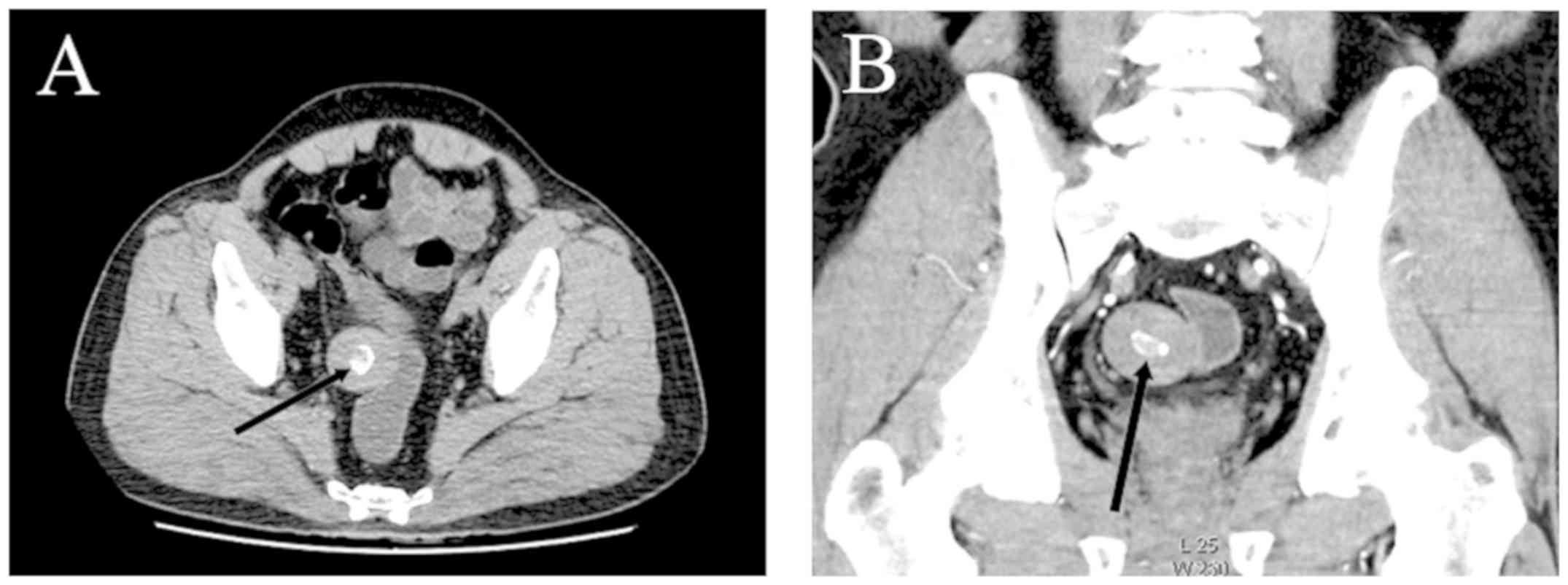
movable. An enhanced abdominal CT revealed a 3.76x3.44 cm mass near
the sigmoid colon, with a smooth edge and a hyper-dense center
(Fig. 1). No obvious enhancement
appeared after contrast injection. Exploratory laparotomy and
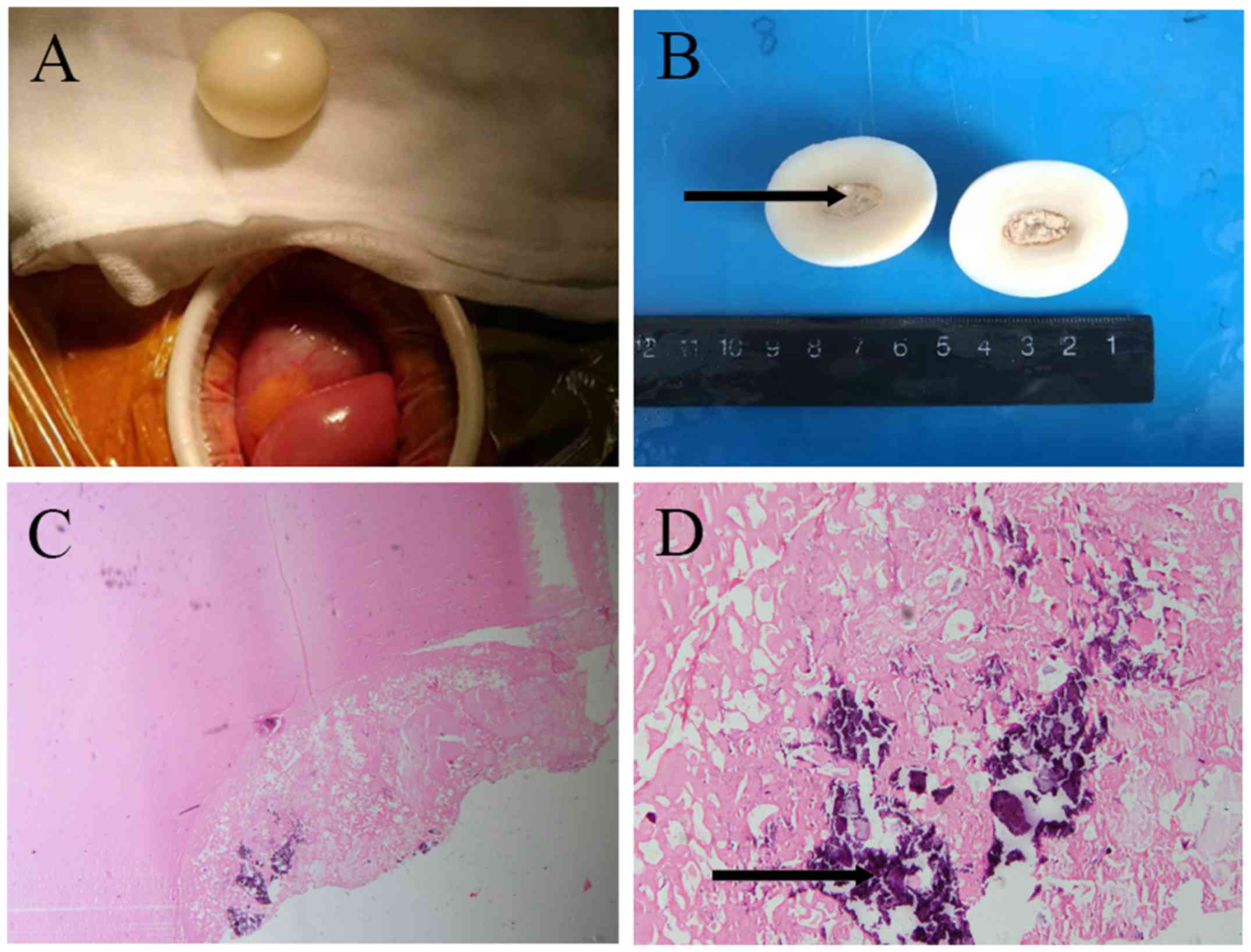
lumpectomy was performed under general anesthesia. Intraoperative
exploration revealed a solitary, well-circumscribed, tough mass
near the sigmoid colon. Lymph nodes in the mesentery were normal.
The shape of the tumor was unusual (Fig.
2A and B). There was an
extremely smooth and regular surface. The cross-section of the
tumor was uniformly greyish white. A large calcification lesion
with a diameter of 1 cm was located in the central part of the
tumor, surrounded by abundant fibrous tissues. Histological
examination revealed that the tumor was mainly composed by hyaline
degeneration tissues and multifocal calcification. Few cells were
detected except for some infiltrating lymphocytes and foamy cells
(Fig. 2C and D). Immunohistochemistry showed vimentin(+),
smooth muscle actin(+), CD34(+), CD117(-), and S-100 protein(-).
The mass was surgically removed without complication. At 1-year
follow-up, the patient showed no recurrence and was physically
intact.
This study was approved by the Institutional Review
Board of Ethics Committee of Changhai Hospital (Shanghai, China).
We retrospectively analyzed 64 patients with CFPs over the past 10
years. One patient was admitted to our hospital at October 3, 2018.
The patient gave written informed consent to appear in study. The
other 63 patients were obtained by searching PubMed. Fifteen were
reported by Chinese authors and 48 were from other countries. The
search terms ‘calcifying fibrous pseudotumor’ and ‘calcifying
fibrous tumor’ were used in our literature search. Definite
diagnoses of CFP in these patients were obtained. All eligible
studies with full-text articles were included. Data were managed
with microsoft excel.
A total of 64 patients were retrospectively
analyzed. There were more male than female patients in China, but
no significant differences overall. The mean age of the patients
was 39.2 (range 8-77 years). Over 80% of patients were from 18 to
60 years old. The disease has no characteristic symptoms; 48.4%
patients discovered the tumors incidentally without obvious
symptoms; 31.3% of patients presented with abdominal discomfort,
and 20.3% of patients suffered from other symptoms (Table I). Abdominal discomfort was most
common in Chinese patients, accounting for 43.6%. The majority of
patients had single lumps, and the mean diameter was 3.7+2.3, with
regular or irregular shape (Table
II). The tumors were most seen in stomach (19 patients, 29.7%),
intestine (13 patients, 20.3%) and thorax (12 patients, 18.7%).
Among the 16 patients from China, 7 (41.2%) presented with stomach
CFPs. Immunohistochemically, only some articles reported
immunohistochemistry results and the measured parameters also
varied. CFP cells were diffusely positive for vimentin and factor
XIII (30 patients for vimentin and 15 for factor XIII. Tumors in 20
of 42 (47.6%) patients stained focally with CD34. Tumors in 11 of
27 (40.7%) cases were partially positive for smooth muscle actin
(SMA). Tumors in 6 of 29 (20.7%) patients were variably positive
for desmin. Almost all tumors were negative for CD117 and S-100
protein. In terms of treatment, all patients included were treated
with surgical removal. During follow-up, only one patient showed
recurrence of the tumor.
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| Variables | China (%) | Other countries
(%) | Total (%) |
|---|
| Sex | | | |
|
Male | 10 (62.5) | 24 (50.0) | 34 (53.1) |
|
Female | 6 (37.5) | 24 (50.0) | 30 (46.9) |
| Age | | | |
|
0-17 | 1 (6.3) | 2 (4.2) | 3 (4.7) |
|
18-60 | 13 (81.2) | 43 (89.5) | 56 (87.5) |
|
>60 | 2 (12.5) | 3 (6.3) | 5 (7.8) |
| Symptoms | | | |
|
No obvious
symptoms | 3 (18.8) | 28 (58.3) | 31 (48.4) |
|
Abdominal
discomfort | 7 (43.6) | 13 (27.1) | 20 (31.3) |
|
Painless
nodules | 3 (18.8) | 2 (4.2) | 5 (7.8) |
|
Shortness of
breath | 1 (6.3) | 1 (2.1) | 2 (3.1) |
|
Chest
pain | 1 (6.3) | 3 (6.2) | 4 (6.3) |
|
Other
symptoms | 1 (6.3) | 1 (2.1) | 2 (3.1) |
 | Table IITumor characteristics. |
Table II
Tumor characteristics.
| Characteristic | China (%) | Other countries
(%) | Total (%) |
|---|
| Tumor size, cm | | | |
|
Min | 1 | 0.5 | 0.5 |
|
Max | 7.8 | 10 | 10 |
|
Average
(mean ± SD) | 2.8±1.7 | 4.0±2.5 | 3.7±2.3 |
| Location | | | |
|
Thorax | 2 (12.5) | 10 (20.8) | 12 (18.7) |
|
Stomach | 7 (41.2) | 12 (25.0) | 19 (29.7) |
|
Intestine | 1 (6.3) | 12 (25.0) | 13 (20.3) |
|
Abdominal
cavity | 2 (12.5) | 6 (12.5) | 8 (12.5) |
|
Pelvic or
perineum | 3 (18.8) | 3 (7.3) | 6 (9.4) |
|
Other | 1 (6.3) | 5 (10.4) | 6 (9.4) |
| Immunoreactive | | | |
|
Vimentin | 14/14 (100.0) | 16/16 (100.0) | 30/30 (100.0) |
|
Factor
XIIIa | 2/2 (100.0) | 13/13 (100.0) | 15/15 (100.0) |
|
SMA | 8/10 (80.0) | 3/17 (17.6) | 11/27 (40.7) |
|
CD34 | 11/14 (78.6) | 9/28 (32.1) | 20/42 (47.6) |
|
CD117 | 0/10 (0.0) | 1/19 (5.3) | 1/29 (3.4) |
|
Desmin | 6/10 (60.0) | 0/19 (0.0) | 6/29 (20.7) |
|
S-100
protein | 1/4 (25.0) | 0/28 (0.0) | 1/32 (3.1) |
Discussion
CFP is a rare benign tumor identified by the World
Health Organization in 2002(1). The
tumor is common in children and adolescents (3), and is mostly found in skin, neck,
pleura (4), abdominal cavity, as
well as in internal organs such as the gastrointestinal tract
(5), adrenal gland (6), and lung (7). Patients typically have no obvious
symptoms. The tumors are often detected incidentally during imaging
examinations or surgery.
The pathogenesis of CFP is not clear. Tumors in
patients with a history of trauma and surgery are considered as a
result of excessive postoperative inflammation responses (8). Larson et al suggested that CFP
could be an unrecognized lesion of IgG4-related disease (9); however, Shimizu et al reported a
patient with no plasma cells containing IgG4 in the tumor (10). Mehrad et al found copy number
losses on chromosome 8 and deleterious mutations in ZN717, FRG1,
and CDC27 genes, all of which are a novel findings for studies of
CFTP tumorigenesis (11).
We analyzed 64 cases and found that the disease may
be more common in young and middle aged (18-60-year-old) males.
Symptoms are atypical and most likely induced by the tumor
compressing surrounding tissues. CFPs in the abdominal cavity and
gastrointestinal tract might lead to indigestion, bloating and
abdominal pain, while those in thorax could induce pleural
effusion. Abdominal discomfort was more common in China (43.6%)
than other countries (27.1%), while 41.2% patients in China
presented with stomach masses and in other countries, the
proportion was 25%. It has been suggested that abdominal discomfort
is possibly related to stomach lesions. Dietary differences between
China and other countries might explain the differences, and more
studies are needed to confirm this. Among the included cases, the
mean diameter of the tumor was 3.7 cm. The shapes of the tumors
were considered to be related to their position. We found that the
CFPs from the gastrointestinal tract and abdominal cavity were
solitary, regular, with typical appearance on CT. Most of the
tumors in the thoracic cavity and arms were small, scattered, and
irregular.
Imaging is important for primary diagnosis of CFP.
In most cases, CT reveals that CFPs appear regular isodense masses
with hyper-dense cores of calcification. Diffuse hyper-dense
signals could also be detected in tumors with scattered
calcification lesions (12). On MRI,
the lesion had a hypointense signal on T1 and T2 weighted imaging,
but an isodensity signal on gadolinium-enhanced T1 weighted imaging
(13). In addition, tumors from
gastrointestinal wall could be detected directly using barium
gastroenterography and gastrointestinal endoscopy.
On gross inspection, CFPs are characterized as
homogenous and hypovascular masses. These masses were composed of
dense and hyalinized stroma, with scattered strips of
calcification. Under microscopic observation, hypocellular
sclerosis and coarse collagen were mostly seen. There were also
scattered inflammatory infiltrates, plasma cells, lymphocytes and
mast cells. Spindle cells were dispersed among thick collagen
bundles (14).
Immunohistochemically, CFP cells were diffusely positive for
vimentin and factor XIIIa, while focally positive for SMA, CD34 and
desmin. Antibodies against CD117 and S100 were negative. Compared
with other countries, tumors of China patients showed higher
staining positivity for SMA and CD34. However, only two patients
were tested for factor XIIIa. Our statistics suggest that vimentin
and factor XIIIa are valuable immunohistochemical mark of CFP.
Improving rates of examination for vimentin and factor XIIIa might
be valuable for the diagnosis of CFP.
The differential diagnosis of CFP is difficult. We
listed the widely used immunostains for CFP and other potential
differential diagnoses (Table
III). For patients presenting with abdominal discomfort,
gastrointestinal stromal tumors (GISTs) and inflammatory
myofibroblastic tumors (IMTs) should be considered first. GIST
originating from the interstitial cells of Cajal are the most
common single type of sarcoma in China. They are mostly seen in
stomach (50-60%) and small intestine (30-35%). More than 80% of the
patients were over 50 years old. The clinical symptoms were mild
(15). Their characteristics include
lack of hyalinized collagen, calcification lesions and staining
positive for CD117, CD34 and anoctamin 1 were the main differences
from CFP. IMT was possibly related to genetic predisposition and
infection (16). The tumors were
abundant in fibrous tissue, consisting of variable quantities of
plasma cells, lymphocytes, eosinophils, foamy histiocytes, and mast
cells. IMT could distinguished from CFP by expression of anaplastic
lymphoma kinase and rearrangement of chromosome 2p23(6). Moreover, CFP from pleura had to be
differentiated from other pleural lesions such as solitary fibrous
tumors (SFTs). SFTs were often positive for CD34 and had no
dystrophic calcifications (17).
Leiomyoma and calcifying aponeurotic fibroma should also be
considered in the differential diagnoses of tumors in the
musculoskeletal system. Leiomyoma has calcifications and
ossifications, and its cells are positive for desmin, SMA,
caldesmon, and factor XIIIa. Calcifying aponeurotic fibromas are
less circumscribed and usually occur in the hands and feet.
Metaplastic cartilage and multinuclear giant cells are typically
(18).
 | Table IIITumor immunohistochemistry. |
Table III
Tumor immunohistochemistry.
| Immunostain | CFP | GIST | IMT | SFT | Leiomyoma |
|---|
| Factor XIIIa | ﹢ | N | ﹢ | ﹢ | ﹣ |
| Vimentin | ﹢ | ﹢ | ﹢ | ﹢ | ﹢ |
| SMA | ± | ± | ﹢ | ± | ﹢ |
| CD34 | ± | ± | ± | ﹢ | ﹣ |
| CD117 | ﹣ | ﹢ | ﹣ | ﹣ | ﹣ |
| Desmin | ± | ﹣ | ﹣ | ﹣ | ﹢ |
| S-100 protein | ﹣ | ﹣ | ﹣ | ﹣ | ﹣ |
In conclusion, CFPs are rare benign tumors that are
difficult to diagnose preoperatively. We found that tumors were
more common in patients aged from 18 to 60 years. They were
immunoreactive for vimentin and factor XIIIa. In China, the most
common location of CFP was the stomach. Perhaps this is related to
the eating habits of the Chinese. Nevertheless, the number of
reported cases is low. Clinical features and pathogenesis of this
disease need to be characterized in further studies.
Acknowledgements
Not applicable.
Funding
The present study was supported by The National
Natural Science Foundation of China (grant no. 30972877), The
Science and Technology Commission of Shanghai Municipality (grant
no. 16ZR1400800) and The Cohort Study of Colorectal Cancer (grant
no. 2017YFC0908200).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
HYM was responsible for the analysis of data and the
writing of the manuscript. MTF contributed to acquisition of the
materials and the design of the study. YGH was responsible for the
conception of the manuscript, design of the study and checking of
the primary data. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by The Institutional
Review Board of Ethics Committee of Changhai Hospital (Shanghai,
China). The patient gave consent to be included in the present
study.
Patient consent for publication
The patient gave written consent for image and data
publication.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Rosenthal NS and Abdul-Karim FW: Childhood
fibrous tumor with psammoma bodies. Clinicopathologic features in
two cases. Arch Pathol Lab Med. 112:798–800. 1988.PubMed/NCBI
|
|
2
|
Saglam EA, Usubütün A, Kart C, Ayhan A and
Küçükali T: Reactive nodular fibrous pseudotumor involving the
pelvic and abdominal cavity: A case report and review of
literature. Virchows Arch. 447:879–882. 2005.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Hill KA, Gonzalez-Crussi F, Omeroglu A and
Chou PM: Calcifying fibrous pseudotumor involving the neck of a
five-week-old infant. Presence of factor XIIIa in the lesional
cells. Pathol Res Pract. 196:527–531. 2000.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Xu L, Zhu H, Yang Y and Bao J: Imaging
features of calcifying fibrous tumor. Chin J Med Imaging.
24:298–302. 2016.
|
|
5
|
Prucker J, Salaheddin-Nassr Y and Leidl S:
Calcifying fibrous tumor of the terminal ileum mesentery: Case
report. Medicine (Baltimore). 97(e13351)2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Wu T, Zhu P, Duan X, Yang X and Lu D:
Calcifying fibrous pseudotumor of the adrenal gland: A rare case
report. Mol Clin Oncol. 5:252–254. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Özkan S, Demırağ F, Yekeler E and
Karaoğlanoğlu N: Calcifying fibrous pseudotumor of lungs. Turk J
Med Sci. 44:901–903. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Kang W, Cui Z, Li X, Sun P and Jin X:
Calcifying Fibrous Tumor of the Tunica Vaginalis Testis: A report
of 2 cases. Urology. 100:e9–e13. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Larson BK, Balzer B, Goldwasser J and
Dhall D: Calcifying fibrous tumor: An unrecognized IgG4-Related
disease. APMIS. 123:72–76. 2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Shimizu S, Funakoshi Y, Yoon HE, Okuma T,
Utsumi T, Ito N, Sakaguchi M, Taniguchi K, Eimoto T and Matsumura
A: Small calcifying fibrous pseudotumor of the heart confined to
the epicardium. Cardiovasc Pathol. 24:191–193. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mehrad M, LaFramboise WA, Lyons MA, Trejo
Bittar HE and Yousem SA: Whole-exome sequencing identifies unique
mutations and copy number losses in calcifying fibrous tumor of the
pleura: Report of 3 cases and review of the literature. Hum Pathol.
78:36–43. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wesecki M, Radziuk D, Niemiec S, Waniczek
D and Lorenc Z: Calcifying fibrous tumor of the small bowel
mesentery in a 27-year old male patient-case report. Pol Przegl
Chir. 86:436–439. 2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Fan SF, Yang H, Li Z and Teng GJ: Gastric
calcifying fibrous pseudotumour associated with an ulcer: Report of
one case with a literature review. Br J Radiol. 83:e188–e191.
2010.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Li BJ, Yang XD, Chen WX, Shi YH, Nie ZH
and Wu J: Calcifying fibrous tumor of stomach: A case report.
Medicine (Baltimore). 96(e8882)2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Joensuu H, Hohenberger P and Corless CL:
Gastrointestinal stromal tumour. Lancet. 382:973–983.
2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Liu HK, Lin YC, Yeh ML, Chen YS, Su YT and
Tsai CC: Inflammatory myofibroblastic tumors of the pancreas in
children: A case report and literature review. Medicine
(Baltimore). 96(e5870)2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ağaçkıran Y, Fındık G, Aydoğdu K, Günay E,
Günay S and Kaya S: An extremely rare case of multiple calcifying
tumor of the pleura. Tuberk Toraks. 60:385–388. 2012.PubMed/NCBI View
Article : Google Scholar
|
|
18
|
Hoffmann H, Beaver ME and Maillard AA:
Calcifying fibrous pseudotumor of the neck. Arch Pathol Lab Med.
124:435–437. 2000.PubMed/NCBI View Article : Google Scholar
|