Introduction
As the population ages, the incidence of head and
neck cancer in older people has increased. Aged patients will have
many complications, but aortic valve stenosis [or aortic stenosis,
(AS)] may be found in preoperative examination for non-cardiac
surgery such as cancer surgery.
The European Heart Association guidelines recommend
surgical aortic replacement (SAVR) for patients with severe
symptomatic AS before non-cardiac surgery. SAVR is recommended for
patients with severe asymptomatic AS only when the risk of
non-cardiac surgery is high and when the risk of SAVR is low
(1).
Therefore, cancer patients who are contraindicated
for SAVR may not be able to receive sufficient cancer treatment.
Even if SAVR can be performed, the burden of surgery may be great,
and the timing of cancer-bearing surgery may be delayed.
In this study, we performed low-invasive
transcatheter aortic valve implantation (TAVI) before carrying out
oral cancer surgery at an early stage for a patient with advanced
AS and tongue cancer. To our knowledge, there are no reports of
oral cancer surgery after TAVI. Therefore, we also provide a
summary and literature review to help evaluate the safety and
efficacy of oral cancer surgery after TAVI.
Case report
The patient was a 79-year-old woman who had a
history of hypertension, diabetes, angina, cerebral infarction, and
hysteromyoma after artificial joint replacement. Her family history
was unremarkable. Around January 2019, a mass was noticed on the
left tongue margin, but it was judged to be stomatitis and left
untreated. In May 2019, she visited a dental clinic for denture
fabrication. She was subsequently referred to our department
because of the suspicion of tongue cancer. The patient's physique
was moderate and her nutritional status was good. Her face was
symmetrical, and several elastic, mobile, bean-sized lymph nodes
were palpated. No tenderness was observed in the submandibular
region. However, left lower body paralysis due to cerebral
infarction was observed.
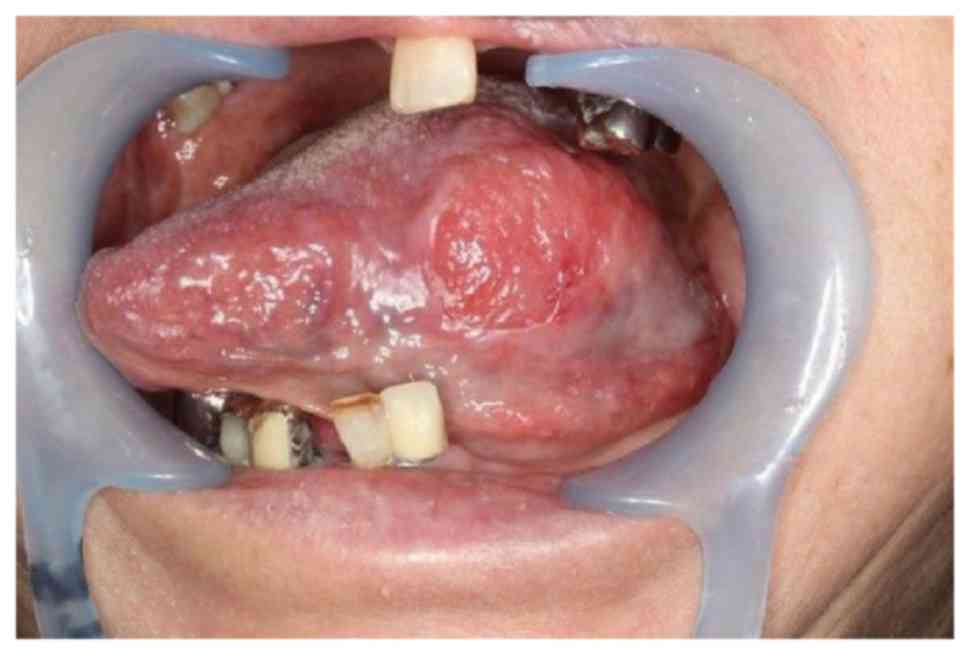
Intraoral findings showed a mass consolidation
measuring approximately 25x15 mm with no spontaneous pain or
tenderness in the left tongue margin (Fig. 1). Hemorrhage, ulceration, tongue
movement disorder, and sensory disturbance were not observed.
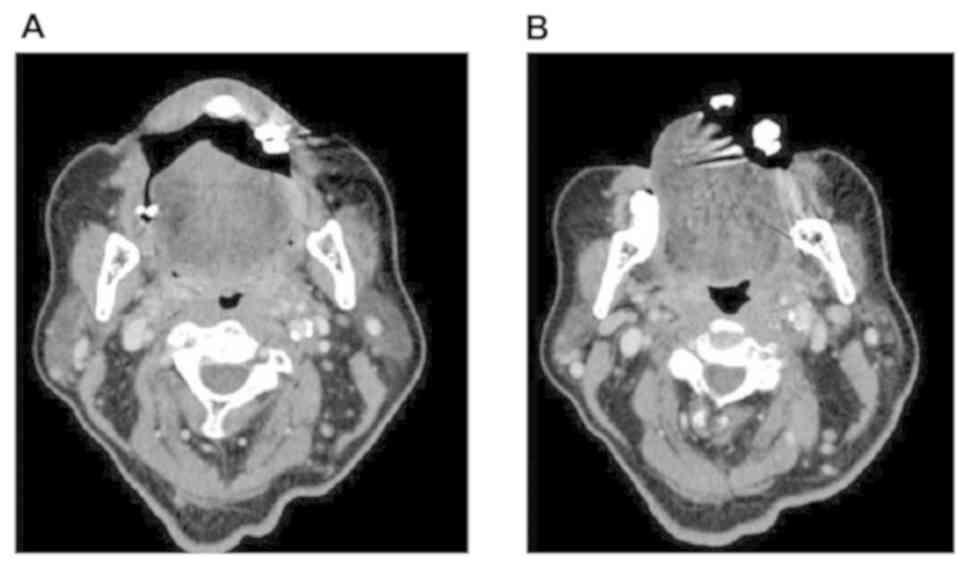
Contrast-enhanced computed tomography revealed a
tumor with an unclear border showing a contrast effect and
measuring 10x23 mm on the left lingual margin (Fig. 2A). The left upper internal deep
cervical lymph node showed an enlarged, uniformly circular lymph
node measuring 9x15 mm (Fig. 2B).
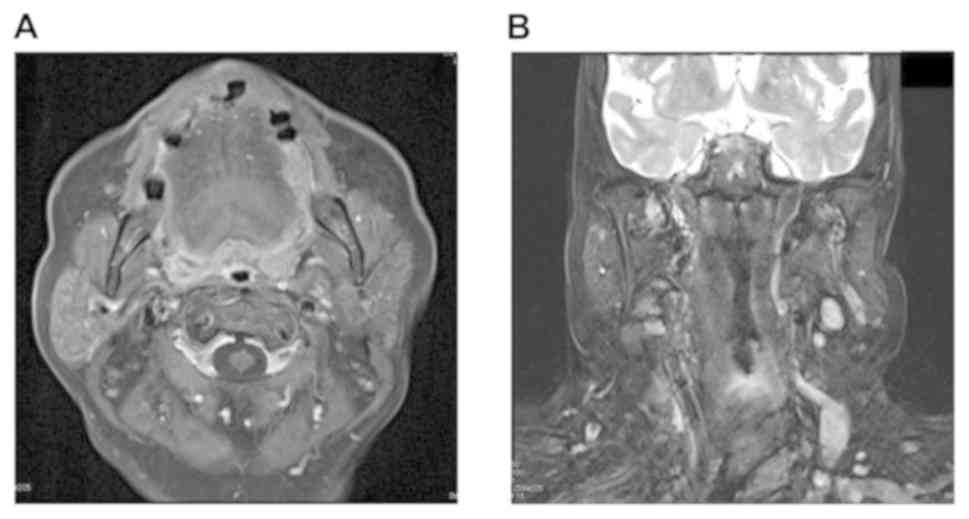
Contrast-enhanced MRI showed a high-signal region with a uniform
internal contrast area measuring 10x23 mm on the left side of the
tongue (Fig. 3A) and a high-signal
region measuring 9x15 mm in the left upper internal deep cervical
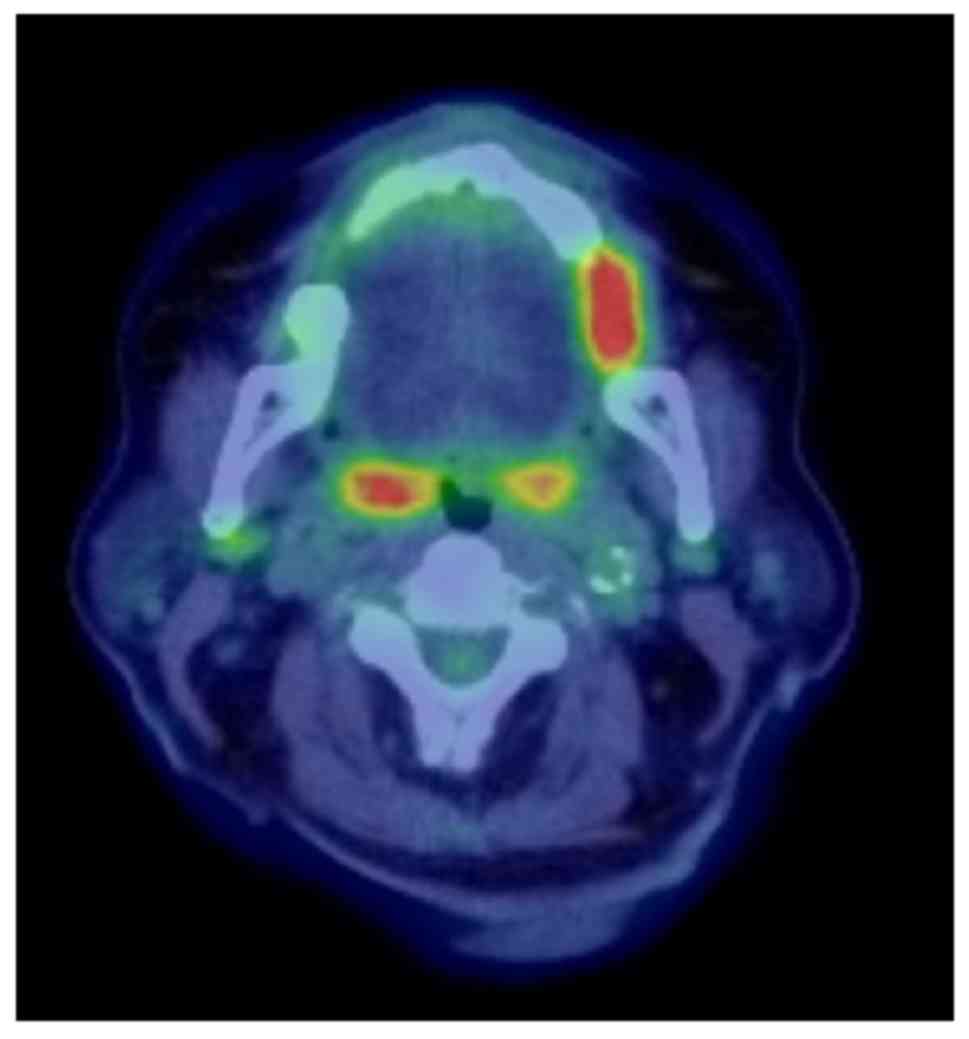
lymph node (Fig. 3B). Preoperative
use of 2-[18F]-fluoro-2-deoxy-D-glucose positron
emission tomography/computed tomography (FDG-PET/CT) showed
abnormal FDG accumulation in the left lingual margin (SUVmax: 9.14)
and bilateral superior internal cervical lymph nodes (right:
SUVmax: 2.65, left: SUVmax: 3.93) (Fig.
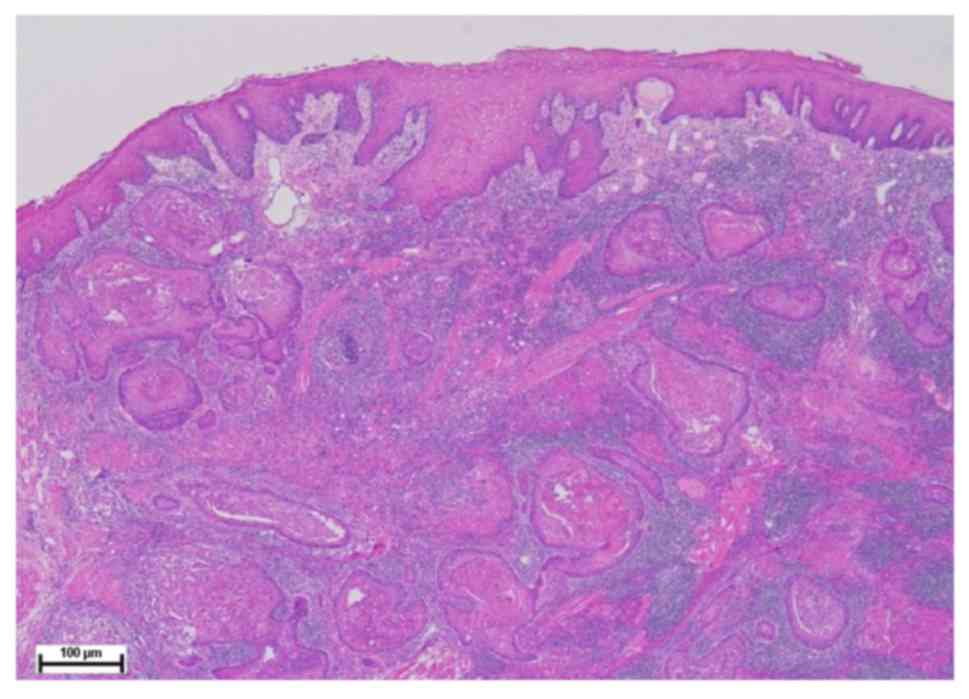
4). Biopsy findings indicated squamous cell carcinoma (Fig. 5). Ultrasonography showed that the
internal structure of the lymph node was uniform and the morphology
of the lymph node was round. The lymph nodes examined by palpation
were mobile and not indurated. Under the diagnosis of tongue cancer
(cT2N0M0), partial tongue excision and neck dissection were
scheduled. We thus staged the tumor as N0 clinically by
comprehensively physical assessments and various imaging findings.
However, the possibility of metastasis could not be ruled out, and
it was anticipated that postoperative follow-up would be difficult
because the patient lives on a remote island. Therefore, we
performed preventive neck dissection because the detection of
cervical metastases could be delayed.
In May 2019, a biopsy was performed under local
anesthesia. Biopsy findings at this time indicated squamous cell
carcinoma. Therefore, partial tongue excision and left functional
neck dissection were scheduled under general anesthesia, and a
preoperative examination was performed. AS was confirmed by
echocardiography. Cardiac ultrasonography revealed an aortic valve
maximum blood flow of 5.0 m/s, aortic valve mean pressure
difference of 58 mmHg, aortic valve opening area of 0.5
cm2, severe AS, and left ventricular wall thickening. EF
of 63.4% and left ventricular wall motion were within normal
limits. Based on these findings, AS treatment was considered to be
necessary before tongue cancer surgery. However, after consultation
with the heart team, the patient was judged to be a high-risk case
for open-heart surgery according to her advanced age and history of
diabetes, cerebral infarction, and angina. We therefore decided to
perform TAVI with the femoral artery approach.
Based on findings from coronary computed tomography
angiography during preoperative examination of TAVI, we suspected
coronary artery stenosis, but coronary blood flow was maintained by
coronary angiography. In July 2019, a guide wire was passed from
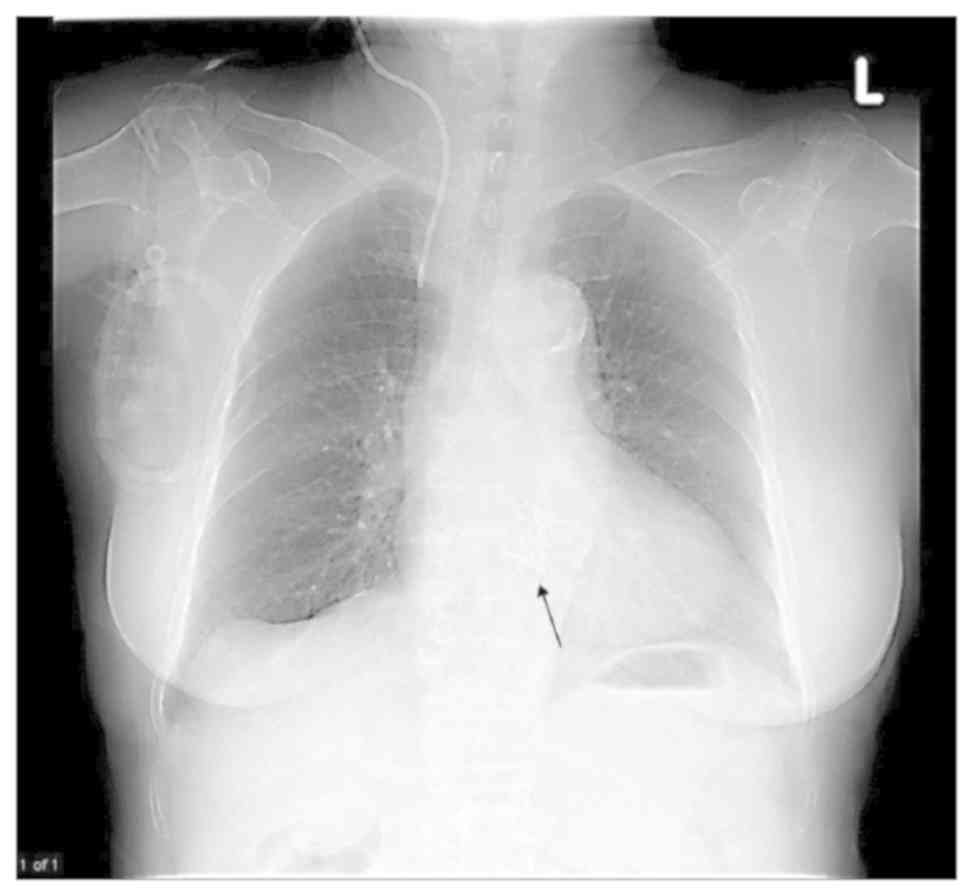
the left femoral artery to the aortic valve, and a 23 mm SAPIEN 3
valve (Edwards Lifesciences Corp.) was placed under rapid pacing
(180 times/min) (Fig. 6). Only a
small amount of periarticular regurgitation was present, and the
coronary artery was well visualized. The patient was extubated in
the operating room and left the bed the next day. Blood tests
confirmed improvement of inflammatory responses and no platelet
count decrease, and partial tongue excision, free skin grafting,
and neck dissection were performed under general anesthesia 21 days
after TAVI surgery. Histopathological findings showed that tissue
was squamous cell carcinoma with prominent keratinization
proliferated in solid alveolar form. Keratin pearls were
occasionally found and nuclear atypia was relatively mild. The
tumor follicle measured 30x22x12 mm and the depth of penetration
was 11 mm. There were no metastases in cervical lymph nodes. The
patient was discharged without complications 22 days after tongue
cancer surgery.
Discussion
AS is a narrowing of the opening of the aortic
valve, leading to obstruction of left ventricular outflow,
subsequent left ventricular systolic dysfunction, and ultimately
long-term heart failure. In patients undergoing non-cardiac
surgery, severe AS is a known high-risk factor for mortality and
morbidity because it reduces coronary blood flow due to surgical
stress and hemodynamic changes. The European Heart Association
guidelines recommend SAVR for patients with severe symptomatic AS
before non-cardiac surgery. However, for patients with severe
asymptomatic AS, SAVR is recommended only when the risk of SAVR is
low and when the risk of non-cardiac surgery is high (1). The severity and symptoms of AS, which
may increase the risk of oral cancer surgery in such older cancer
patients, may not be readily apparent. We prioritized TAVI for AS
treatment in the present case because of her older age and multiple
complications, which were contraindications for major invasive
procedures such as thoracotomy, extracorporeal circulation, and
cardiac arrest (2).
TAVI is a minimally invasive approach to replace an
aortic valve that is different from SAVR in that it does not
require cardiac arrest, extracorporeal circulation, and
thoracotomy. In high-risk or ineligible patients, PARTNER trials
have shown that treatment outcomes such as mortality, LV recovery,
and morbidity after TAVI are comparable or superior to conventional
treatment (3-5).
Thus, it has been reported that TAVI can be a standard alternative
treatment for high-risk surgical patients. In addition, certain
benefits of TAVI have been reported in cancer patients because it
does not require cardiopulmonary bypass. These include reducing the
risk associated with tumor bleeding with anticoagulant disorders
and administration of anticoagulants, and reducing tumor
dissemination with immunosuppressive and inflammatory effects of
cardiopulmonary bypass (6,7). In severe AS patients in need of cancer
treatment, TAVI may not only allow a smooth transition to the next
treatment strategy due to minimal invasiveness, but also yield
oncological benefits.
As far as we are aware, there have been no reports
of TAVI being performed before surgery for head and neck cancers.
There were a few reports found of TAVI procedures performed before
surgery for cancers in other parts of the body (8-12).
The interval between AS treatment and cancer treatment should be
short to prevent malignant tumor progression. However, when SAVR
and cancer-bearing surgery are performed in two stages, a short
interval between operations may not be possible because of surgical
tolerance. In addition, surgery decreases the body's natural
defenses; thus, insufficient time between operations increases the
risk of postoperative infection. To our knowledge, there is no
report on the appropriate interval between TAVI and surgery,
however, an interval of about 2-3 weeks between cancer resection
surgery and TAVI was applied in colon cancer and lung cancer
patients (8-12).
We scheduled a similar time interval in the present case.
Due to factors such as age, heart failure, and
cancer, 30-50% of patients with symptomatic AS are considered
ineligible for SAVR (3-5).
Additionally, Bach et al reported that almost the same
number of patients with SAVR had refused surgery (13). Previously, older cancer patients
with severe AS, such as the present case, may not have been able to
undergo severe AS or cancer treatment. However, TAVI is potentially
useful for such patients. While there are several reports of cancer
surgery after TAVI (8-12),
to our knowledge, this is the first report of TAVI for a patient
undergoing oral cancer surgery. Moreover, there is little data on
the feasibility of TAVI for severe AS patients scheduled for cancer
surgery and the impact on subsequent surgery. A greater
accumulation of similar cases and further investigation of multiple
factors, such as the appropriate treatment interval between TAVI
and cancer surgery, perioperative antiplatelet drugs, and surgical
indications, are needed.
In conclusion, we performed oral cancer surgery
early after TAVI without serious complications. TAVI could be a
useful option for severe AS patients with high surgical risk prior
to cancer resection surgery. However, a greater accumulation of
cases is needed to confirm the safety and effectiveness of
TAVI-containing treatment strategies.
Acknowledgements
The authors would like to thank Dr Takaaki Nagano
and Dr Shotaro Higa (both, Department of Thoracic and
Cardiovascular Surgery, Graduate School of Medicine, University of
the Ryukyus, Okinawa, Japan) and Dr Masashi Iwabuchi and Dr
Hidekazu Ikemiyagi, (Department of Cardiovascular Medicine,
Nephrology and Neurology, University of the Ryukyus, Okinawa,
Japan) who contributed to patient treatment and care.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
TK acquired patient data, performed the literature
review and edited the manuscript. TN conceived and designed the
present study. TN acquired the data, provided clinical advice and
revised the manuscript. AM and TN evaluated specimens and gave
histopathological advice. TK wrote the manuscript. All authors read
and approved the final manuscript.
Ethics approval and consent to
participate
The present report was submitted for ethical review
to the Ethics Committee of the University of the Ryukyus (Okinawa,
Japan), which waived the requirement for review per institutional
protocol due to the study not containing content that requires
ethical approval. The Ethics Committee approved the submission and
publication of the manuscript. Written informed consent was
obtained from the patient for the publication of this case report
and accompanying images.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of this case report and the
accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kristensen SD, Knuuti J, Saraste A, Anker
S, Bøtker HE, Hert SD, Ford I, Gonzalez-Juanatey JR, Gorenek B,
Heyndrickx GR, et al: 2014 ESC/ESA guidelines on non-cardiac
surgery: Cardiovascular assessment and management: The joint task
force on non-cardiac surgery: Cardiovascular assessment and
management of the European Society of Cardiology (ESC) and the
European Society of Anaesthesiology (ESA). Eur Heart J.
35:2383–2431. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Cribier A, Eltchaninoff H, Bash A,
Borenstein N, Tron C, Bauer F, Derumeaux G, Anselme F, Laborde F
and Leon MB: Percutaneous transcatheter implantation of an aortic
valve prosthesis for calcific aortic stenosis: First human case
description. Circulation. 106:3006–3008. 2002.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Elmariah S, Palacios IF, McAndrew T,
Hueter I, Inglessis I, Baker JN, Kodali S, Leon MB, Svensson L,
Pibarot P, et al: Outcomes of transcatheter and surgical aortic
valve replacement in high-risk patients with aortic stenosis and
left ventricular dysfunction: Results from the Placement of Aortic
Transcatheter Valves (PARTNER) trial (cohort A). Circ Cardiovasc
Interv. 6:604–614. 2013.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Mack MJ, Leon MB, Smith CR, Miller DC,
Moses JW, Tuzcu EM, Webb JG, Douglas PS, Anderson WN, Blackstone
EH, et al: 5-year outcomes of transcatheter aortic valve
replacement or surgical aortic valve replacement for high surgical
risk patients with aortic stenosis (PARTNER1): A randomised
controlled trial. Lancet. 385:2477–2484. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kapadia SR, Leon MB, Makkar RR, Tuzcu EM,
Svensson LG, Kodali S, Webb JG, Mack MJ, Douglas PS, Thourani VH,
et al: 5-year outcomes of transcatheter aortic valve replacement
compared with standard treatment for patients with inoperable
aortic stenosis (PARTNER 1): A randomised controlled trial. Lancet.
385:2485–2491. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Scott BH, Seifert FC, Glass PS and Grimson
R: Blood use in patients undergoing coronary artery bypass surgery:
Impact of cardiopulmonary bypass pump, hematocrit, gender, age, and
body weight. Anesth Analg. 97:958–963. 2003.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Pinto CA, Marcella S, August DA, Holland
B, Kostis JB and Demissie K: Cardiopulmonary bypass has a modest
association with cancer progression: A retrospective cohort study.
BMC Cancer. 13(519)2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Tamai K, Okamura S, Kitahara T, Minoji T,
Takabatake H, Watanabe N, Yamamura N, Fukuchi N, Ebisui C, Yokouchi
H and et al: Laparoscopic colectomy after transcatheter aortic
valve implantation in an elderly patient with obstructive
descending colon cancer and severe aortic stenosis: A case report.
Surg Case Rep. 5(102)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sakai T, Yahagi K, Miura S, Hoshino T,
Yokota T, Tanabe K and Ikeda S: Transcatheter aortic valve
implantation for patients with lung cancer and aortic valve
stenosis. J Thorac Dis. 10:E387–E390. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Tanaka T, Yahagi K, Okuno T, Horiuchi Y,
Kusuhara T, Yokozuka M, Miura S and Tanabe K, Miura S and Tanabe K
and Tanabe K: Transcatheter aortic valve implantation in a patient
with severe aortic valve stenosis, colon cancer, and obstructive
ileus: A case report. J Cardiol Cases. 17:163–166. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Okuno T, Yahagi K, Horiuchi Y, Sato Y,
Tanaka T, Koseki K, Osanai A, Yokozuka M, Miura S and Tanabe K: The
role of transcatheter aortic valve replacement in the patients with
severe aortic stenosis requiring major non-cardiac surgery.
Cardiovasc Interv Ther. 13:345–351. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Nagata H, Kanzaki R, Kanou T, Ose N,
Funaki S, Shintani Y, Minami M, Mizote I, Sakata Y, Maeda K, et al:
Two cases of lobectomy for lung cancer after transcatheter aortic
valve implantation. Surg Case Rep. 4(139)2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Bach DS, Cimino N and Deeb GM: Unopoerated
patients with severe aortic stenosis. J Am Coll Cardiol.
50:2018–2019. 2007.PubMed/NCBI View Article : Google Scholar
|