Introduction
Dermatofibrosarcoma protuberans (DFSP) is a rare
slow growing sarcoma of the dermis, with a low propensity for
distant metastases but high likelihood of local invasion into
subcutaneous tissues. As a result, this tumor is treated with wide
local excision, ensuring negative resection margins. Recurrence is
a feature of DFSP when excision is incomplete. DFSP is most
commonly seen in young adults on the trunk and limbs.
Fibrosarcomatous transformation sometimes occurs in
a subset of DFSP tumours, referred to as FS-DFSP. Accurate
differentiation of this variant is associated with a worse
prognosis due to the incidence of distant metastases as compared to
ordinary DFSP. Although the aetiology remains unclear, antecedent
trauma has been proposed as a predisposing factor in the
development of DFSP (1,2).
Case report
A 22-year old female presented with a history of
sharp sensations, described as ‘sticking’ in nature over the
mid-back region. Two years later the patient had a fall and
sustained blunt trauma to her back. She then noted that the area
became swollen approximately 4 cm and was painful. Swelling of the
area gradually decreased, however the sharp pain persisted. There
was no further medical intervention at this time. Four years later,
the area in question began to increase in size once again, this
time without any precipitating factors. Within two months the
lesion grew to approximately 4 cm and was accompanied by return of
excruciating pain with spontaneous resolution of swelling within
one month. One-year later the lesion returned however, now
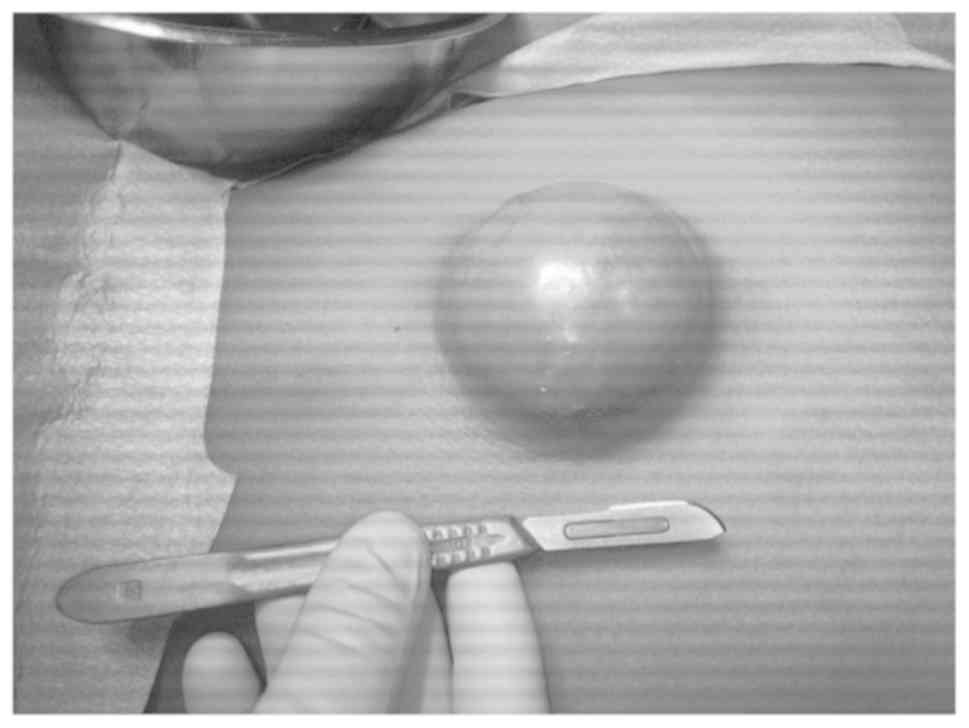
painless, and grew to 8 cm within 4 months (Fig. 1). No Erythema nor discharge were
noted at this time. The area became pruritic and developed
overlying telangiectasia. The social history revealed occasional
alcohol use with no smoking.
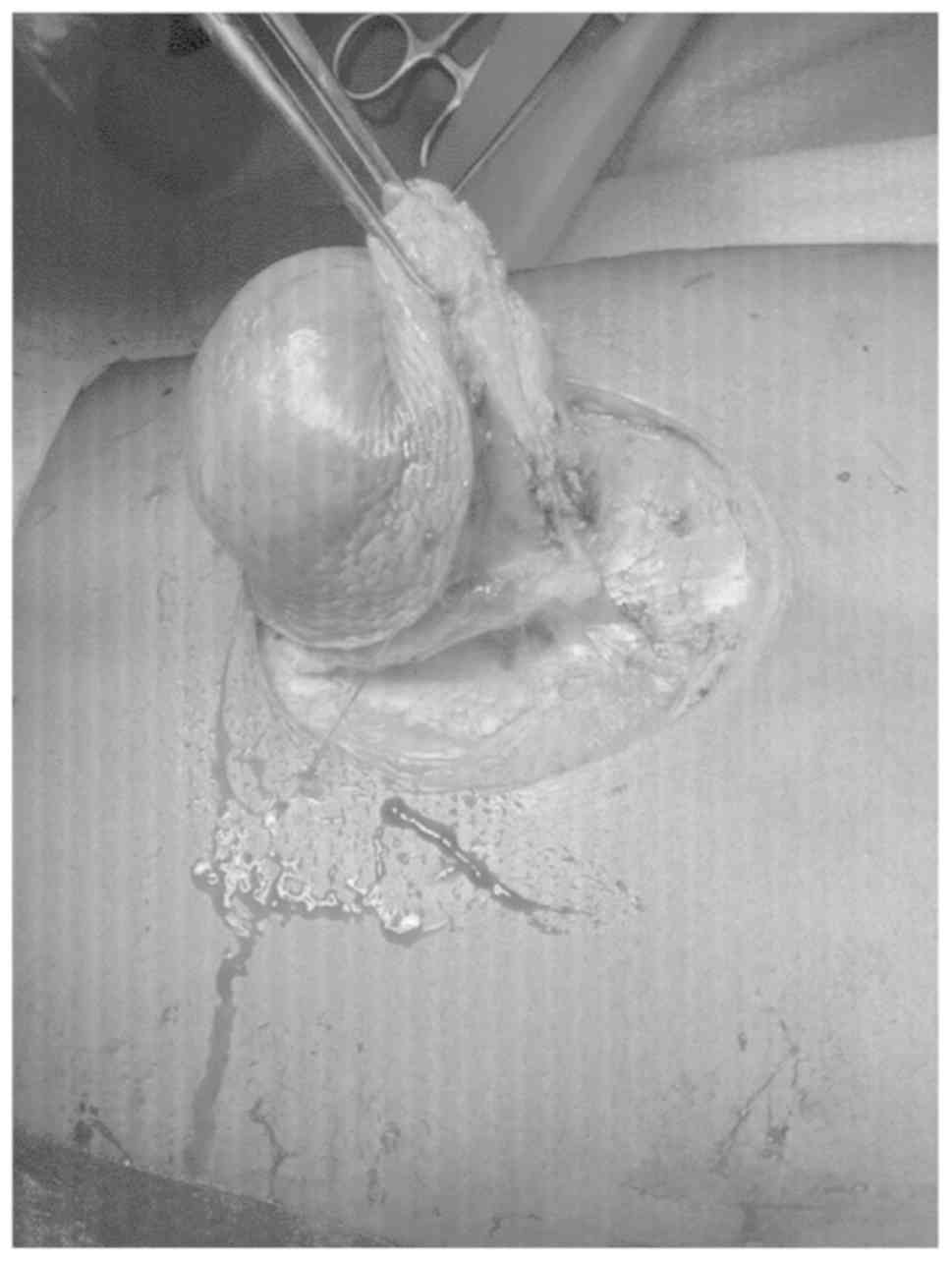
The mass was completely excised with a wide margin
and a rotational skin flap done to cover the large defect (Figs. 2 and 3). Histological evaluation
revealed a well-circumscribed non-encapsulated highly cellular
fibroblastic proliferation. Cells displayed scant cytoplasm,
tapering elongated nuclei with granular chromatin. Abnormal mitoses
were seen at a rate of 10 per HPF. Immunohistochemical analysis
showed the tumor to be SMA, Desmin, CD 34, and S100 negative with
Ki-67: 30% proliferation. No vascular or local invasion was
present.
Discussion
DFSP is a rare tumor of the dermis with unknown
etiology. The first case of DFSP was described by Taylor in
1890(3). It was first described as
a distinct cutaneous disease entity called progressive and
recurring dermatofibroma by Darier and Ferrand in 1924. DFSP is a
slow growing sarcoma with a low likelihood of distant metastases,
but can often result in local invasion of neighboring tissues. As a
result, wide and deep local surgical excision has been the mainstay
method of treatment. However, even 3-cm-wide local excision margins
have resulted in a local recurrence rate of 11% and as such, new
methods have been employed in management. In recent years, reports
of DFSPs successfully treated with Mohs micrographic surgery have
been appearing in the literature (4).
DFSP most commonly presents on the trunk (50%) and
extremities (30-40%) with 10-15% of cases involving the head and
neck (5). The overall annual
incidence has been found to be 4.2 per million per year worldwide
(6). According to Scott et
al (7) the incidence in the
Afro-Caribbean population is almost double that of the Caucasian
(6.5 and 3.9 per million respectively). The most common age of
presentation is between the ages of 30 and 50 and throughout our
search of the literature, it was found that the prevalence or DFSP
in the male gender outweighed that of the female. Our index case
fell outside of these margins with a 22-year old female. One study
by Criscione and Weinstock, contradictorily found that females were
more frequently affected by DFSP (6).
DFSP is associated with translocation of genetic
material between chromosomes 17 and 22. This translocation (17;22)
fuses the COL1A1 gene on chromosome 17 with part of PDGFB gene on
chromosome 22. These mutations which occur after birth, are not
inherited and are only found in tumor cells (2). Two types of DFSP have been described;
85% of these display a more indolent course and have low potential
for metastases, representing the classical DFSP. The remaining 15%
are described as fibrosarcomatous ‘high grade’ type (FS-DFSP), and
these can present more aggressively (1).
Benign fibrohistocytomas (BFH) are slow growing,
asymptomatic neoplasms of the dermis which usually measure less
than 1cm in diameter (8). Like
DFSP, these lesions show spindle shaped cells during microscopic
evaluation. BFH is usually differentiated from DFSP by the positive
finding of CD34 in the latter. However, not all DFSP are CD34
positive. A very small percentage may be negative for this
immunohistochemical marker as documented by Fernandez-Flores and
Manjon (9). Despite negative
staining of our index case for CD34, we still lean toward the
diagnoses of DFSP versus BFH due to the presence of features such
as overlying venous dilatation, large size and rapid increase in
size over short period of time, more in keeping with a clinical
diagnosis of DFSP as opposed to BFH. Although other
Immunohistochemical markers used to differentiate between these two
neoplasms are CD10 Apo D FXIIIa and NGFR, due to lack of resources,
we were unable to check for the presence of the above mentioned in
this case.
In the case described above, the area of skin at
which DFSP subsequently developed, displayed altered sensation
prior to sustaining blunt trauma. Considering this sensory change,
we hypothesize that a pathological process possibly involving
incidental translocation (17;22), had begun at the site prior to
injury. With subsequent trauma and release of inflammatory
mediators, propagation of the malignant process may have occurred,
thus adding insult to injury and resulting finally in development
of a rapidly growing DFSP. A similar inference has been made by
Boukovalas et al, who suggest that trauma and dermal debris
causing chronic low grade infection and inflammation, may trigger
the molecular changes that lead to development of DFSP. They
explained that the exact mechanism by which trauma may predispose
to development of DFSP is unknown, but it seems intuitive that
chronic inflammation and stimulation of the immune system at a
local level may trigger the immunopathologic changes that could
lead to the malignant transformation of dermal cells. A similar
mechanism has been described in the pathogenesis of Marjolin ulcers
after trauma or skin injury (1). Of
note, no research linking inflammatory mediators or other molecular
effects of trauma on tissues, to development of malignant
processes, was found during our literature search.
Throughout the literature, however, DFSP has been
reported to arise in areas with history of prior trauma, including
tattoos, vaccination sites, burn scars, surgical scars and
radiation treatment, trauma scars, radiodermatitis, sites of
central venous lines and insect bites (1,10).
According to Monnier et al, in a study conducted, 21% of
patients had a history of prior trauma at the site where DFSP
developed (11). Of note, all cases
linked to trauma found in the literature involved some form of
breech of skin or penetrating injury. This is the first case, to
our knowledge, documented to show development of DFSP after blunt
trauma.
Traditionally, DFSP has been described as a slow
growing tumor, but our case was an exception to this description.
The tumor grew from 0 to 8 cm within 4 months. Few similar cases
were described in the literature.
Limitations in this case included the lack of IHC
markers needed for complete diagnosis of the tumor excised. This
lack of resources poses a hindrance to diagnosis of many tumors in
our setting.
Here we have described a case of suspected DFSP of
the trunk with a history of trauma, which presented on an area of
skin with previous altered sensation, in a young female.
Based on the literature search and our current
findings, the authors conclude that both non congenital mutation as
well as trauma play a role in the development of this dermal
neoplasm. Future research into the relationship between trauma and
DFSP on a cellular level is suggested, as there seems to be direct
link.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
RC was involved in conception and design,
acquisition of data, drafting the manuscript and revising it
critically for important intellectual content. MW was involved in
substantial contributions to conception and design and drafting the
manuscript. MJR was the operating surgeon, was responsible for the
conception of this paper and revised it critically for important
intellectual content. All authors have read and given final
approval of the version to be published.
Ethics approval and consent to
participate
Both verbal and retrospective written consent were
provided by the patient whose information was used for the writing
of this case. In addition, consent for journal publication was also
obtained from the patient.
Patient consent for publication
Consent was received from the patients owing to the
publication of data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Boukovalas S, Castillo AC, Andry D,
Lombana N, Qiu S and Murphy KD: Dermatofibrosarcoma Protuberans:
Trauma and genetics. Ann Plast Reconstr Surg. 1(1001)2017.
|
|
2
|
Patel KU, Szabo SS, Hernandez VS, Prieto
VG, Abruzzo LV, Lazar AJ and López-Terrada D: Dermatofibrosarcoma
protuberans COL1A1-PDGFB fusion is identified in virtually all
dermatofibrosarcoma protuberans cases when investigated by newly
developed multiplex reverse transcription polymerase chain reaction
and fluorescence in situ hybridization assays. Hum Pathol.
39:184–193. 2008.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Garg MK, Yadav MK, Gupta S, Kumar N and
Khandelwal N: Dermatofibrosarcoma protuberans with contiguous
infiltration of the underlying bone. Cancer Imaging. 9:63–66.
2009.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Haycox CL, Odland PB, Olbricht SM and
Casey B: Dermatofibrosarcoma protuberans (DFSP): growth
characteristics based on tumor modeling and a review of cases
treated with mohs micrographic surgery. Ann Plast Surg. 38:246–251.
1997.PubMed/NCBI
|
|
5
|
Stamatakos M, Fyllos A, Siafogianni A,
Ntzeros K, Tasiopoulou G, Rozis M and Kontzoglou K:
Dermatofibrosarcoma protuberans: A rare entity and review of the
literature. J BUON. 19:34–41. 2014.PubMed/NCBI
|
|
6
|
Criscione VD and Weinstock MA: Descriptive
epidemiology of dermatofibrosarcoma protuberans in the United
States, 1973 to 2002. J Am Acad Dermatol. 56:968–973.
2007.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Scott N, Causey C, Hodder SC and Kittur
MA: ‘It Started as a Spot’ … Dermatofibrosarcoma Protuberans. J
Cytol Histol. S5:1:2016.
|
|
8
|
Vanni R: Skin: Cutaneous benign fibrous
histiocytomas. Atlas Genet Cytogenet Oncol Haematol. 5:213–214.
2001.
|
|
9
|
Fernandez-Flores A and Manjon JA: Mitosis
in dermatofibroma: A worrisome histopathologic sign that does not
necessarily equal recurrence. J Cutan Pathol. 35:839–842.
2008.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Stivala A, Lombardo GA, Pompili G, Tarica
MS, Fraggetta F and Perrotta RA: Dermatofibrosarcoma protuberans:
Our experience of 59 cases. Oncol Lett. 4:1047–1055.
2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Monnier D, Vidal C, Martin L, Danzon A,
Pelletier F, Puzenat E, Algros MP, Blanc D, Laurent R, Humbert PH
and Aubin F: Dermatofibrosarcoma protuberans: A population-based
cancer registry descriptive study of 66 consecutive cases diagnosed
between 1982 and 2002. J EurAcad Dermatol Venereol. 20:1237–1242.
2006.PubMed/NCBI View Article : Google Scholar
|