Introduction
The current World Health Organization (WHO)
classification of soft tissue and bone tumors includes 13 types of
benign adipocytic tumors, of which lipoma is the most common
(1). The mesenchymoma was
originally defined by Stout in 1948 to describe a tumor containing
at least two mesenchymal tissues, not usually found together, in
addition to fibrous elements (2),
and lipomas occasionally contain other mesenchymal elements, for
example, osseous tissue, fibrous tissue, or smooth muscle, and are
classified as osteolipoma (3-5),
fibrolipoma (6-8)
or myolipoma (9,10), respectively. Lipomas can contain
cartilage metaplasia, which is referred to as chondrolipoma, a rare
form of benign mesenchymoma. Sporadic case reports of chondrolipoma
in adults exist; however, pediatric presentation is extremely rare
(11). Chondrolipomas frequently
present in the breast, pharynx, and tongue (12-18),
and there is no previous report of a chondrolipoma arising in the
finger.
We present a rare case of a chondrolipoma of the
finger in an 11-year-old girl, with a review of the corresponding
magnetic resonance imaging (MRI) and pathological findings. To our
knowledge, this is the first pediatric case report of chondrolipoma
arising in the finger. The patient and her family were informed
that data from the case would be submitted for publication and gave
us consent for the academic use of clinical information.
Case report
An 11-year-old girl was referred to the Kobe
University hospital on March 2017, with a painless mass on the
dorsal aspect of the proximal phalanx of her left middle finger,
which she had noticed a year ago after catching the finger in a
closing door. Clinical examination revealed a well-defined elastic
soft mass, measuring 2.5x2 cm, with no associated redness, local
heat nor tenderness, and the lesion was mobile without fixation to
overlying skin or deeper structures. The functions of the finger
including range of motion and sensation were not impaired. A plain
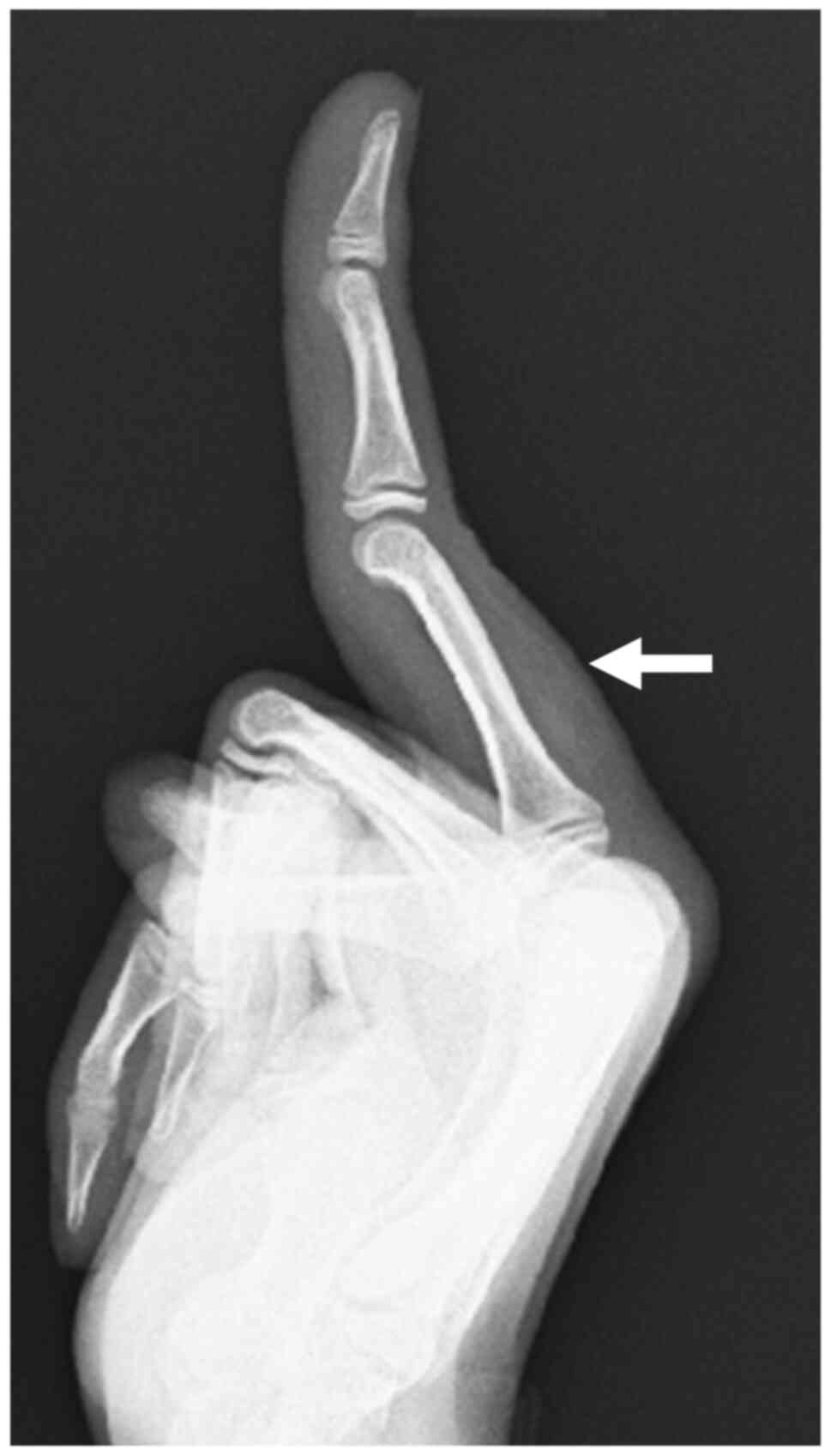
radiograph of the left middle finger revealed a soft tissue mass
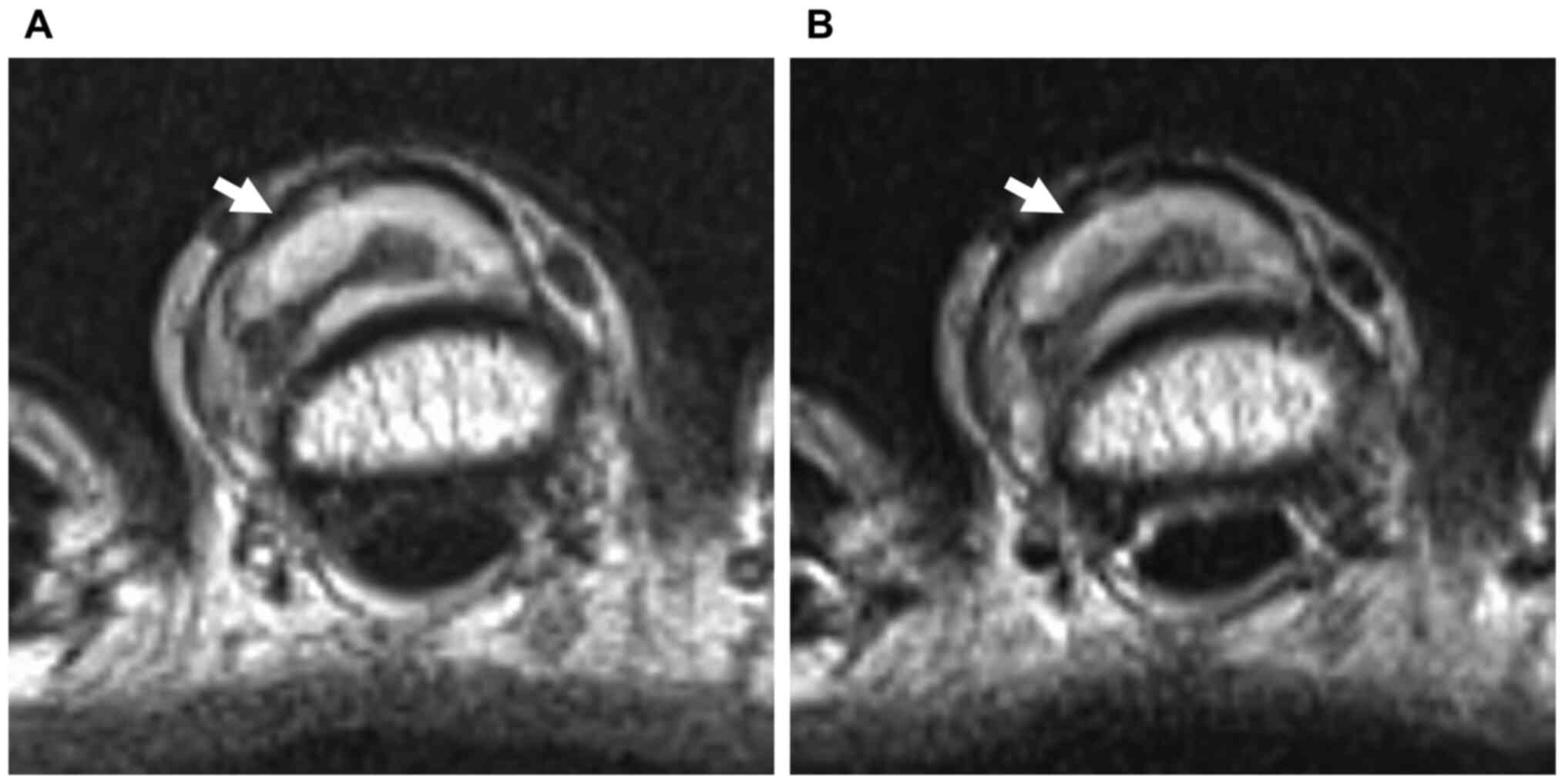
without evidence of a calcification and a bone erosion (Fig. 1). MRI revealed a well-circumscribed
2.5x2x1 cm-sized lesion with heterogeneous signal intensity. On
both T1- and T2-weighted images, the lesion showed a predominantly
marked hyperintense signal containing linear hypointense regions
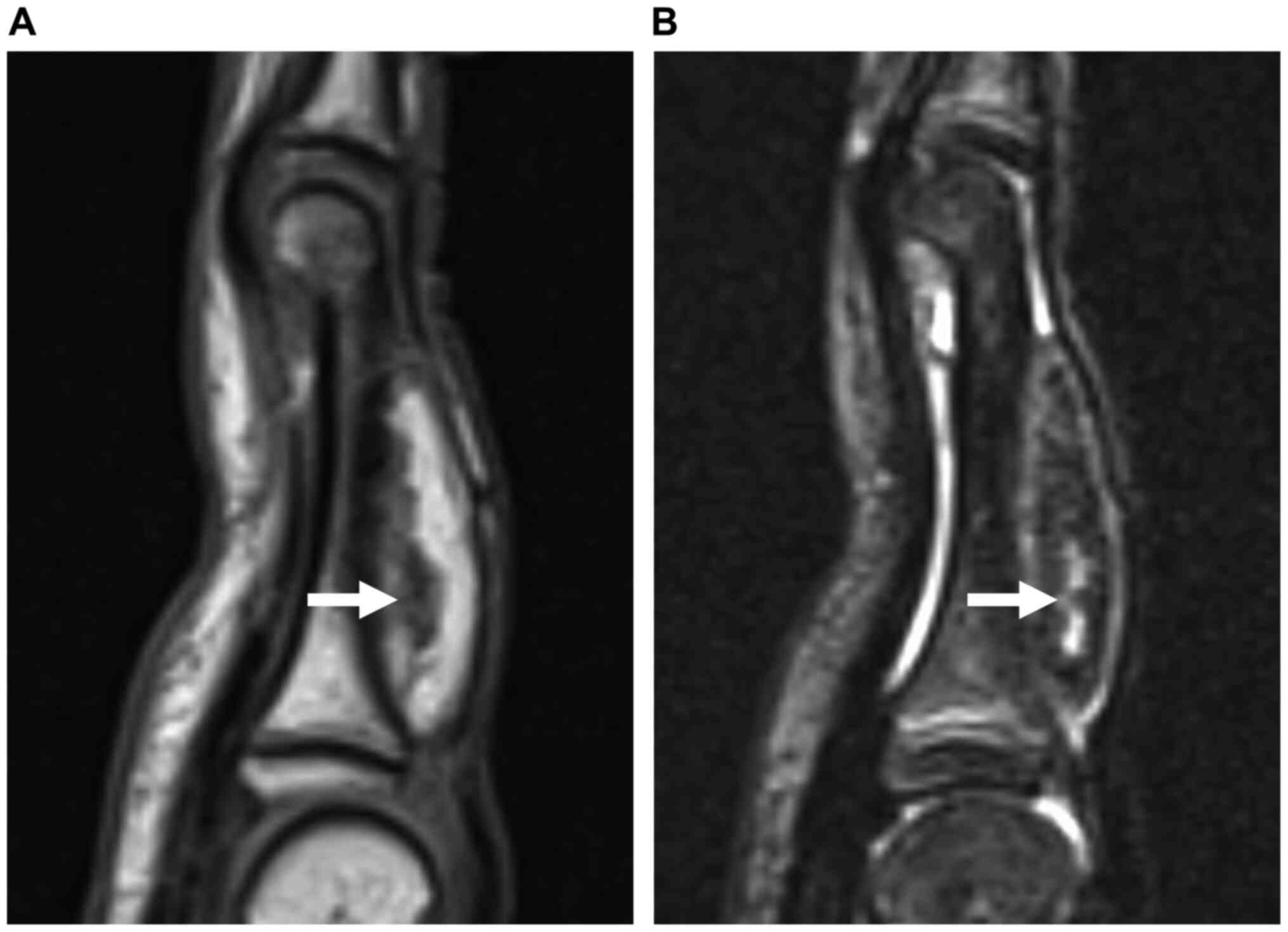
(Figs. 2 and 3A), and on fat-suppressed short-tau
inversion recovery (STIR) sequences, the lesion showed a
predominant hypointensity, with linear regions displaying
hyperintensity (Fig. 3B). MRI
findings suggested a diagnosis of lipoma with non-adipose elements,
such as cartilage matrix.
During marginal resection, a well-circumscribed
tumor was found on the extensor tendon without adhesion to
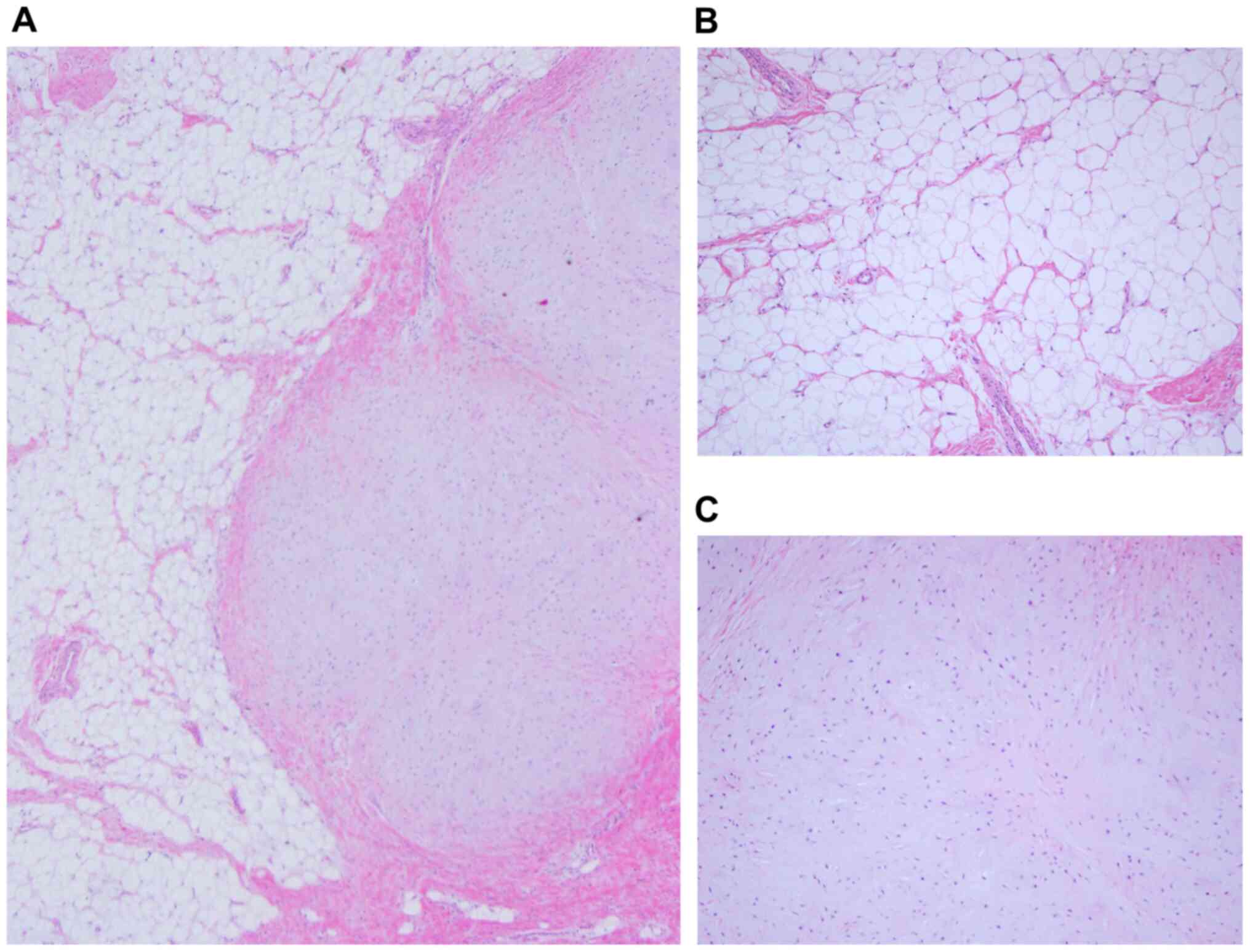
surrounding structures. Histologically, the major component of the
tumor was mature adipose tissue, with a limited area of hyaline
cartilage matrix which included the flat/asteroid form cell
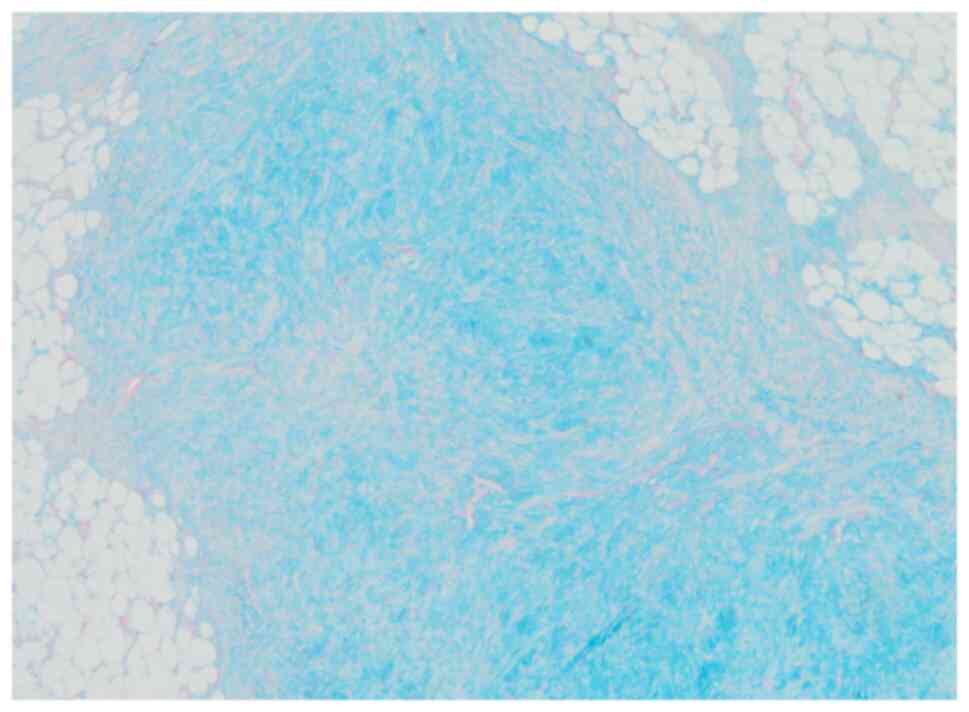
(Fig. 4). Alcian blue staining was
moderately positive in the cartilage matrix (Fig. 5). There were no lipoblasts or
histological features of malignancy, and the histopathological
diagnosis was a chondrolipoma. The patient experienced excellent
recovery, without any functional impairment or local recurrence
three years after surgery.
Discussion
Chondrolipoma is often confused with chondroid
lipoma, a subtype of benign adipocytic tumors, because of the
terminological similarity. Histologically, chondroid lipomas have
tumor cells with central irregular nuclei surrounded by clear
vacuolated cytoplasm resembling lipoblasts without mature hyaline
cartilages (1,16-21).
However, in the present case, characteristic features of coexisting
areas with lipoma and true hyaline cartilage, with no evidence of
lipoblasts, resulted in diagnosis of chondrolipoma.
Controversy exists regarding chondrolipomas'
etiology, with two predominant theories regarding its developmental
mechanisms. One posits that pluripotent mesenchymal cells
differentiate into both adipose and cartilage cells, and another
postulates that lipomas obtain chondrometaplasia following
traumatic injury (19). In this
case, the tumor development was probably triggered when the finger
caught in the door. Signorini et al. discussed the pathogenetic
mechanisms of post-traumatic lipomas, suggesting the
differentiation of mesenchymal precursors (preadipocytes) to mature
adipocytes by a trauma as a possible mechanism (22). Copcu and Sivrioglu reported fat
necrosis as the major etiological factor in post-traumatic
subcutaneous lipomas (23).
Furthermore, Copcu and Sivrioglu mentioned that soft tissue trauma
could result in hematoma formation, subsequent lymphatic effusion,
and the formation of lipomatous lesions (23). Since the finger has limited space,
this may explain how a post-traumatic hematoma caused pluripotent
mesenchymal cells to differentiate into both adipose and cartilage
cells simultaneously, then eventually forming a chondrolipoma.
Therefore, obtaining a history of trauma is invaluable in
diagnosing this neoplasm.
The characteristic histopathological feature of
chondrolipoma is coexisting mature adipose tissue and hyaline
cartilage (1,11-18,24).
Lipomas typically show high-signal intensity on both T1-and
T2-weighted MRI, compared to iso-signal intensity on the
fat-suppressed sequence. In contrast, hyaline cartilage reveals
low-signal intensity on T1-weighted images and higher-signal
intensity on STIR sequences, which reflect high ratios of the water
content within the cartilage component. Pathological and MRI
evidence pointed to the diagnosis of this rare variant of lipoma;
that is, there were contrasting signal intensities between
cartilage and adipose components on T1-weighted and STIR images,
respectively. The STIR sequence of MRI examination, which has
recently been widely used in musculoskeletal imaging, is a valuable
diagnostic tool for chondrolipoma. Despite the difficulty in
diagnosis, local recurrence or malignant transformation following
resection has never been reported in the literature (11-18,24),
suggesting that the therapeutic strategy for chondrolipoma should
be consistent with that of lipoma. Given that, a wait-and-see
policy for chondrolipomas should be acceptable, depending on the
size and location.
In conclusion, we describe an unprecedented case of
pediatric chondrolipoma arising in the finger, and identified the
characteristic MRI appearances in T1- and T2-weighted, and STIR
images. Clinicians should consider chondrolipoma as a differential
diagnosis when they come across such characteristic MRI findings in
patients with history of trauma, as this may be beneficial for
treatment strategies.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author or reasonable
request.
Author's contributions
KM and TK designed the experiments and wrote the
initial draft of the manuscript. MK, TK and HH performed the
surgery of the lesion. NF, YK, MM, TT, SF, KK, SY and TM provided
medical care for the patients and collected the data. MK and NJ
performed histological examinations of the tumor tissue. RK and TA
were responsible for the design and interpretation of the study as
well as revisions and approval of the final draft of the
manuscript. All the authors have read and approved the final
version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient and her family were informed that data
from the case would be submitted for publication and gave us
consent for the academic use of clinical information.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Fletcher CD, Bridge JA, Hogendoorn P and
Mertens F: WHO Classification of Tumours of Soft Tissue and Bone.
4th edition. IARC Press, Lyon, pp19-30, 2013.
|
|
2
|
Stout AP: Mesenchymoma, the mixed tumor of
mesenchymal derivatives. Ann Surg. 127:278–290. 1948.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kwan Ip NS, Lau HW, Wong WY and Yuen MK:
Osteolipoma in the forearm. J Clin Imaging Sci.
8(20)2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Demiralp B, Alderete JF, Kose O, Ozcan A,
Cicek I and Basbozkurt M: Osteolipoma independent of bone tissue: A
case report. Cases J. 2(8711)2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Fritchie KJ, Renner JB, Rao KW and Esther
RJ: Osteolipoma: Radiological, pathological, and cytogenetic
analysis of three cases. Skeletal Radiol. 41:237–244.
2012.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Lee JH and Oh DH: Fibrolipoma in an
unusual location: The nasopharynx. Ear Nose Throat J. 98:66–67.
2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Sethia R, Rawlins KW, Aljasser A, Nogan S,
Elmaraghy CA and Wiet GJ: Pediatric nasopharyngeal fibrolipoma: A
case report and review of the literature. Int J Pediatr
Otorhinolaryngol. 125:103–106. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Shin SJ: Subcutaneous fibrolipoma on the
back. J Craniofac Surg. 24:1051–1053. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Meis JM and Enzinger FM: Myolipoma of soft
tissue. Am J Surg Pathol. 15:121–125. 1991.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Fukushima M, Schaefer IM and Fletcher CD:
Myolipoma of soft tissue: Clinicopathologic analysis of 34 cases.
Am J Surg Pathol. 41:153–160. 2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ishibashi T, Nishio J, Kobayashi S,
Shiramizu K and Yamamoto T: Chondrolipoma of the ankle in a child:
A case report. J Foot Ankle Surg. 56:1284–1287. 2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Boltze C, Hribaschek A, Lippert H and
Roessner A: Intermuscular chondrolipoma of the thigh: The
diagnostic way of a rare entity. Pathol Res Pract. 199:503–507.
2003.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Fushimi H, Kotoh K, Nishihara K, Fujinaka
H and Takao T: Chondrolipoma of the breast: A case report with
cytological and histological examination. Histopathology.
35:478–479. 1999.PubMed/NCBI
|
|
14
|
Halaas YP, Mra Z and Edelman M:
Chondrolipoma of the oropharynx. Ear Nose Throat J. 80:146–147.
2001.PubMed/NCBI
|
|
15
|
Tomonaga M and Kudawara I: Ossifying
chondrolipoma of the thigh: Radiographic pathologic correlation.
Curr Orthop Pract. 25:493–496. 2014.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Raj V, Dwivedi N, Sah K and Chandra S:
Chondrolipoma: Report of a rare intra oral variant with review of
histiogenetic concepts. J Oral Maxillofac Pathol. 18:276–280.
2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Özer F and Bal N: A rare diagnosis in the
neck during childhood: Congenital chondrolipoma. Turk Patoloji
Derg. 33:161–163. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Nonaka CF, Miguel MC, de Souza LB and
Pinto LP: Chondrolipoma of the tongue: A case report. J Oral Sci.
51:313–316. 2009.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Katzer B: Histopathology of rare
chondroosteoblastic metaplasia in benign lipomas. Pathol Res Pract.
184:437–445. 1989.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Goldblum JR, Weiss SW and Folpe AL:
Enzinger and Weiss's Soft Tissue Tumors. 6th edition. Mosby, St.
Louis, MO, pp452-455 2014.
|
|
21
|
Meis JM and Enzinger FM: Chondroid lipoma.
A unique tumor simulating liposarcoma and myxoid chondrosarcoma. Am
J Surg Pathol. 17:1103–1112. 1993.PubMed/NCBI
|
|
22
|
Signorini M and Campiglio GL:
Posttraumatic lipomas: Where do they really come from? Plast
Reconstr Surg. 101:699–705. 1998.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Copcu E and Sivrioglu NS: Posttraumatic
lipoma: Analysis of 10 cases and explanation of possible
mechanisms. Dermatol Surg. 29:215–220. 2003.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Nakano M, Arai E, Nakajima Y, Nakamura H,
Miyazono K and Hirose T: Immunohistochemical study of
chondrolipoma: Possible importance of transforming growth factor
(TGF)-betas, latent TGF-beta binding protein-1 (LTBP-1), and bone
morphogenetic protein (BMP) for chondrogenesis in lipoma. J
Dermatol. 30:189–195. 2003.PubMed/NCBI View Article : Google Scholar
|