Introduction
Optimal cytoreduction achieving residual tumour ≤1
cm followed by a platinum-based chemotherapy has best prognostic
impact in advanced epithelial ovarian cancer (1). Optimal time of surgery (up-front vs.
interval debulking) is still discussed (2). A study demonstrated that patients with
primarily inoperable advanced ovarian cancer who received
neoadjuvant chemotherapy followed by interval debulking surgery
have no inferior benefit compared to patients with primary
cytoreductive surgery (2). Most
important was complete cytoreduction with residual tumour=0 mm
(2).
CA125 level (Cancer antigen 125) has revealed an
important role in monitoring patients with ovarian cancer and its
importance at initial diagnosis of ovarian cancer is still
controversial (3,4). Measurement of CA125 level is performed
preoperatively in patients who are suspected of having or are
diagnosed with ovarian cancer. At initial diagnosis CA125 levels
(normal range <35 U/ml) are elevated in about 80% of epithelial
ovarian cancer patients and correlate well with response to
therapy, disease progression or recurrence (3-9).
Known is the greater CA125 levels the more advanced stages
(10).
A previous study demonstrated that a CA125 level
>550 U/ml before neoadjuvant chemotherapy is an independent risk
factor of suboptimal debulking at interval debulking surgery
(11). Selection criteria
identifying patients who will benefit from interval debulking
surgery are required. Thus, the question arises if there is a CA125
level an optimal cytoreduction cannot be achieved while primary
surgery in advanced stages and these patients may benefit from
interval debulking surgery.
Some studies assessed the CA125 level to predict the
extent of cytoreduction (12-14).
A previous study demonstrated a significant positive correlation
between optimal cytoreduction and CA125 level; patients with
optimal cytoreduction had a significant lower CA125 level than
patients after suboptimal cytoreduction (12). They concluded CA125 level is ‘a weak
positive and negative factor for predicting cytoreductive surgery’
(12). Another study demonstrated a
significant higher CA125 level in suboptimal cytoreduced (mean:
1,067 U/ml) vs. optimal cytoreduced patients, but concluded CA125
level is not a reliable predictor of optimal cytoreduction
(13). Brockbank et al
(14) recommended pretreatment
CA125 level as a ‘reliable component of preoperative assessment’;
CA125 level was described as ‘best predictor of disease
suboptimally cytoreduced’ and an optimal cut-off for serum CA125 as
a predictor of suboptimal debulking was defined as 586 U/ml
(14). A retrospective analysis
comparing interval debulking surgery and up-front surgery showed
improved survival after normalization of CA125 level during
chemotherapy prior to interval debulking surgery compared to
patients with suboptimally cytoreduction in primary surgery
(15). So CA125 seems to be a good
marker controlling therapy effort. A previous study demonstrated
CA125 level ≥500 U/ml is most often correlated with advanced FIGO
stages in ovarian cancer (10,13)
and is described as a factor optimal cytoreduction can be achieved
in approximately 20% of cases (10). They concluded CA125 levels seem to
be helpful in predicting suboptimal cytoreductive surgery for
patients with primary ovarian cancer, but it should be interpreted
together with clinical and radiologic findings (16). They defined 11 parameters (CA125
U/ml included) and evaluated a predictive score (16). They detected 3 clinical (age/CA125
≥600 U/ml/ASA 3-4) and 8 radiological criteria that were
significantly associated with presence of residual disease in their
prospective study (16). They
concluded this may improve defining optimal treatment strategy in
these patients (16). Achieving
suboptimal cytoreduction by laparotomy doesn't offer a survival
advantage, demonstrated by Chi et al (10) with the recommendation of modifying
primary surgical approach or alter the standard sequence of
cytoreductive surgery and chemotherapy.
Objective was to analyze relevance of a pretreatment
serum CA125 ≥500 U/ml as a non-invasive factor for estimating
optimal cytoreduction (≤1 cm) in primary serous ovarian cancer and
its prognostic relevance. We tested cut-off CA125 values to detect
subgroups affecting prognosis.
Materials and methods
Description of collective
A total of 136 consecutive patients with primary
epithelial ovarian cancer and existing pretreatment CA125 levels
≥500 U/ml and CA125 <500 U/ml at time of surgery treated at the
department of obstetrics and gynecology treated from January 2005
through December 2012, at University Tübingen, Germany were
enrolled in the study and retrospectively analyzed. Every patient
underwent surgical staging or cytoreductive surgery as clinically
indicated. Primary surgical treatment of epithelial ovarian cancer
consisted of hysterectomy, bilateral salpingo-oophorectomy,
retroperitoneal (pelvic and para-aortic) lymphadenectomy,
omentectomy and resection of any metastatic lesion if possible, as
well. Postoperatively, patients got platinum-based chemotherapy (6
cycles) every three weeks. Patients with neoadjuvant chemotherapy
(NACT) for ovarian cancer and unlikely to be completely cytoreduced
to no residual disease and even patients not capable of surgery
were excluded from study. A total of 74 patients with serous
cancers and CA125 level ≥500 U/ml who underwent primary
cytoreductive surgery met inclusion criteria for the subgroup of
patients with CA125 level beyond 500 and were enrolled. A
preoperative CA125 level ≥500 U/ml was identified as the value with
most predictive power achieving optimal cytoreduction according to
Chi et al (10).
Procedure of evaluation
Objective of our manuscript was to analyze the
relevance of a pretreatment serum CA125 ≥500 U/ml as a non-invasive
factor for estimating optimal cytoreduction (≤1 cm) in primary
serous ovarian cancer and its prognostic relevance. We tested
cut-off CA125 values to detect subgroups affecting prognosis. The
preoperative CA125 level ≥500 U/ml we applied was identified as the
value with most predictive power achieving optimal cytoreduction
according to Chi et al (10). The lowest CA125 level in this
subgroup was 500 U/ml. The range of CA125 level in the cohort was
500-48,470 U/ml. As described in materials and methods objective
was to analyze the highest CA125 level optimal cytoreduction could
be achieved. To determine the ability of the preoperative serum
CA125 level to predict optimal cytoreduction, the true-positive
rate was defined as percentage of suboptimally cytoreduced patients
who had a preoperative serum CA-125 level above the median CA125
level 1,404.5 U/ml (range 501-48,470 U/ml). Based on our results
(comparison of subgroups with CA125 >500 U/ml with median 1,404
U/ml without significant impact based on CA125 level) aim was
evaluate the impact of CA125 level on OS and PFS in ovarian cancer
patients with lower CA125 levels and comparing results with each
other.
Therefore, we additionally identified patients with
same characteristics and CA125 <500 U/ml to compare groups and
impact of CA125 on OS and PFS with CA125 level above and below 500
U/ml.
Age was 62 years in median (range: 38-81 years).
Patients' individual records were reviewed and the following
information abstracted: Age at time of diagnosis, pretreatment
CA125 level, residual disease at completion of procedure, final
histopathologic diagnosis, node involvement, histologic grade,
prognostic impact (PFS, OS). The value CA125 >35 U/ml was
defined as an elevated level. Tumour stage was classified according
to FIGO classification (17). All
surgical pathologic samples were examined by a gynecological
pathologist. The histological diagnosis was classified according to
FIGO-classification (17). Extent
of residual tumour mass was taken from surgical reports and was
subdivided in following groups: R=0 mm; R>0 cm to 1 cm; R>1
cm. Optimal cytoreduction was defined as being attained when
diameter of largest residual tumor nodule remaining at end of
procedure measured less than or equal to 1 cm. Median follow up was
55 months.
Control group
Out of the above mentioned cohort of 136 patients
with advanced FIGO stages a control group was defined with same
inclusion criteria and CA125 level <500 U/ml. Therefore, 62
patients were included and had similar distribution of parameters
like FIGO, node involvement, histological grade, residual tumour
mass (Table I).
 | Table IPatients' characteristics
(n=136). |
Table I
Patients' characteristics
(n=136).
| Parameter | CA125 ≥500
U/ml | CA125 <500
U/ml | P-value |
|---|
| FIGO stage, n
(%) | | | 0.045 |
|
IIIA/B | 8 (10.8) | 15 (24.2) | |
|
IIIC | 49 (66.2) | 40 (64.5) | |
|
IV | 17 (23.0) | 7 (11.3) | |
| Histologic grade, n
(%) | | | 0.222 |
|
G1/2 | 30 (40.5) | 31(50) | |
|
G3 | 44 (59.5) | 31(50) | |
| Histological
subtype, n (%) | | | |
|
Serous | 74(100) | 62(100) | |
| R-status, n
(%) | | | 0.343 |
|
R=0 mm | 16 (21.6) | 21 (33.9) | |
|
R>0 mm-≤1
cm | 39 (52.7) | 25 (40.3) | |
|
R>1
cm | 19 (25.7) | 16 (25.8) | |
| N-status, n
(%) | | | 0.191 |
|
N0 | 13 (17.5) | 18 (29.0) | |
|
N+ | 46 (62.2) | 36 (58.1) | |
|
Nx | 15 (20.3) | 8 (12.9) | |
| CA125-level,
U/ml | 74(100) | 62(100) | |
Statistical analysis
Univariate analyses were performed using PASW
(Version 26 SPSS Inc.). Results are reported as medians with the 5
and 95% upper and lower quartiles (CI). The results are expressed
as means, standard deviations, minimums, maximums and percentages.
Survival curves were calculated using Kaplan-Meier analyses for OS
to calculate hazard ratio (HR) and 95% CI. The log-rank test was
used to test for significant differences between the groups.
P-values <0.05 were considered statistically significant. Data
were compared with above mentioned control group after evaluation
in same way.
Results
Evaluation of collective
All enrolled patients with CA125 level ≥500 U/ml had
advanced FIGO stages (Table I).
89.2% of the patients were classified to FIGO IIIC/IV; the
remaining 10.8% of patients were classified to FIGO IIIA/B
(Table I). Patients'
characteristics are given in Table
I. Data of control group CA125 <500 U/ml are given in
Table I, as well. 75.8% of control
group had FIGO IIIC/IV (Table I).
There was a comparable distribution of FIGO, node involvement,
histological grade, residual tumour mass in study and control group
(P=n.s., Table I). Group CA125 ≥500
U/ml: Most often histologic grade 3 (59.5%, Table I) and node involvement (62.2%,
Table I) were detected,
respectively. In patients with pretreatment CA125 level ≥500 U/ml
optimal cytoreduction (R≤1 cm) could be achieved in 74.3% of all
patients and suboptimal debulking in 25.7% (Table I), respectively. In control group
optimal cytoreduction was achieved in 74.2% (Table I), respectively. Rate of suboptimal
cytoreduction was similar in group CA125 <500 U/ml and CA125
≥500 U/ml (25.8 vs. 25.7%; Table
I).
Correlation CA125 level and residual
tumour
To determine the ability of the preoperative serum
CA125 level to predict optimal cytoreduction, the true-positive
rate (sensitivity) was defined as percentage of suboptimally
cytoreduced patients who had a preoperative serum CA125 level above
the median CA125 level 1,404.5 U/ml (range 501-48,470 U/ml).
The range of CA125 level ≥500 U/ml in serous cancers
was 501-48,470 U/ml with a median of 1,404.5 U/ml. The range of
CA125 level associated with optimal cytoreduction was 501-16,062
U/ml (Table II). The correlation
of CA125 level and residual tumour mass showed highest median CA125
level in suboptimally cytoreduced patients (2,347 U/ml) with a
range of 502-48,470 U/ml compared to patients with complete
cytoreduction (R=0 mm) and R>0 mm-≤1 cm (1,332.5 vs. 1,250 U/ml;
Table II) without a significant
difference (Table II),
respectively. In median CA125 level of patients with R>0 mm-≤1
cm compared to patients after complete cytoreduction was lower
(Table II), respectively. The
maximum CA125 level in complete cytoreduced patients was lower
compared to other groups (7,249 U/ml, Table II). The highest CA125 level was
48,470 U/ml in suboptimally debulked patients (Table II). The higher the CA125 level the
higher the rate of suboptimal debulking, respectively (Table II).
 | Table IIPretreatment CA125 level ≥500 U/ml in
association with residual tumor mass of 74 patients with primary
serous ovarian cancer. |
Table II
Pretreatment CA125 level ≥500 U/ml in
association with residual tumor mass of 74 patients with primary
serous ovarian cancer.
| Resection
status | CA125 level U/ml
[median (range)] | P-value |
|---|
| R=0 mm | 1,332.5
(543-7,249) | 0.061 |
| R>0 mm-≤1
cm | 1,250
(501-16,062) | |
| R>1 cm | 2,347
(502-8,470) | |
CA125 level >/<500 U/ml and rate
of cytoreduction
To identify a CA125 level optimal debulking could
not be achieved, we evaluated the following after subdivision of
groups with CA125 level ≥500 U/ml due to median of CA125 level: The
correlation with residual tumour mass leads to following results
(Table III). The group with CA125
level below median 1,404.5 had a higher rate of optimal
cytoreduction (81.1%) compared to CA125 >1,404.5 U/ml (67.6%;
Table III), respectively. The
group with CA125 level >1,404.5 U/ml had a rate of suboptimal
debulking of 32.4% (Table III)
without significant difference compared to CA125 level <1,404.5
(Table III).
 | Table IIIComparison of pretreatment CA125
level after division into two groups (≥500-1,404 U/ml vs. >1,404
U/ml) and evaluation of residual tumor mass in 74 patients with
primary serous ovarian cancer. |
Table III
Comparison of pretreatment CA125
level after division into two groups (≥500-1,404 U/ml vs. >1,404
U/ml) and evaluation of residual tumor mass in 74 patients with
primary serous ovarian cancer.
| Resection
status | CA125 level
≥500-1,404 U/ml [n (%)] | CA125 level
>1,404 U/ml [n (%)] | P-value |
|---|
| R=0 mm-≤1 cm | 30 (81.1) | 25 (67.6) | 0.183 |
| R>1 cm | 7 (18.9) | 12 (32.4) | |
Prognostic impact of CA125 level
The evaluation of prognostic impact on PFS and OS of
CA125 level ≥500 U/ml leads to the following results (Table IV): CA125 level below 1,404.5 U/ml
compared to CA125 level >1,404.5 U/ml leads to longer PFS and OS
without significant difference (P=n.s., Table IV). There was no significant
difference (P=n.s., Table IV.
[median (range; 95% CI)], respectively. Therefore, no significant
difference is demonstrated due to the calculation of CA125 level
with median 1,404.5 U/ml regarding prognostic impact (Table IV).
 | Table IVPrognostic impact of CA125 level
≥500-1,404 U/ml vs. >1,404 U/ml in 74 patients with serous
ovarian cancer on OS and PFS (months; median, 95% CI). |
Table IV
Prognostic impact of CA125 level
≥500-1,404 U/ml vs. >1,404 U/ml in 74 patients with serous
ovarian cancer on OS and PFS (months; median, 95% CI).
| Parameter | CA125 ≥500-1,404
U/ml | CA125 >1,404
U/ml | P-value |
|---|
| PFS months (median;
95% CI) | 15.2
(12.18-18.215) | 12.6
(10.23-14.97) | 0.259 |
| OS months (median;
95% CI) | 33.7
(2.4-64.9) | 23.4
(18.7-28.08) | 0.536 |
Table V demonstrates
the evaluation of CA125 level ≥500 U/ml subdivided in 2 groups
</>1,404.5 U/ml regarding prognostic impact on PFS and OS
correlated with residual tumor mass (Table V).
 | Table VPrognostic impact of CA125 level
≥500-1,404 U/ml vs. >1,404 U/ml on OS and PFS (months; median,
95% CI) in 74 patients with serous ovarian cancer regarding
residual tumor mass. |
Table V
Prognostic impact of CA125 level
≥500-1,404 U/ml vs. >1,404 U/ml on OS and PFS (months; median,
95% CI) in 74 patients with serous ovarian cancer regarding
residual tumor mass.
|
| PFS, months
(median; 95% CI) | | OS, months (median;
95% CI) | |
|---|
| Resection
status | CA125 level
≥500-1,404 U/ml | CA125 level
>1,404 U/ml | P-value | CA125 level
≥500-1,404 U/ml | CA125 level
>1,404 U/ml | P-value |
|---|
| R=0 mm-≤1 cm | 13.73
(12.7-16.8) | 13.47
(9.7-17.2) | 0.475 | 22.3
(12.85-31.75) | 24.47
(22.3-26.64) | 0.466 |
| R>1 cm | 33.5
(23.04-43.96) | 12.1
(10.02-14.25) | | 43.7
(10.5-65.3) | 17.57
(13.96-21.2) | |
There was no significant prognostic impact on PFS
and OS regarding the different CA125 levels and residual tumour
mass after subdivision of groups (Table
V).
Complete cytoreduction has significant best impact
on PFS (median 22 months; P=0.021; Table VI) compared to R>0 mm-≤1 cm and
R>1 cm in patients with CA125 level ≥500 U/ml, respectively.
 | Table VIImpact of residual tumor mass on PFS
(median, 95% CI) in 74 patients with serous ovarian cancer with
CA125 level ≥500 U/ml. |
Table VI
Impact of residual tumor mass on PFS
(median, 95% CI) in 74 patients with serous ovarian cancer with
CA125 level ≥500 U/ml.
| Parameter | PFS, months
(median; 95% CI) | P-value |
|---|
| R=0 mm | 22.0 (0-56.0) | 0.021 |
| R>0-≤1 cm | 11.6
(9.3-13.9) | |
| R>1 cm | 16.0
(10.9-21.06) | |
The comparison of study and control group leads to
following results: Optimal cytoreduction in correlation with CA125
<500 U/ml has significant better prognostic impact on OS and
even PFS than patients with R>1 cm and CA125 ≥500 U/ml (OS:
P=0.046; PFS: P=0.011; Table VII
[median (range; 95% CI)], respectively. Longest OS was detected in
optimally cytoreduced patients with CA125 <500 U/ml (Table VII) and has significant better
prognostic impact on OS (P=0.023) and PFS (P=0.011) in optimally
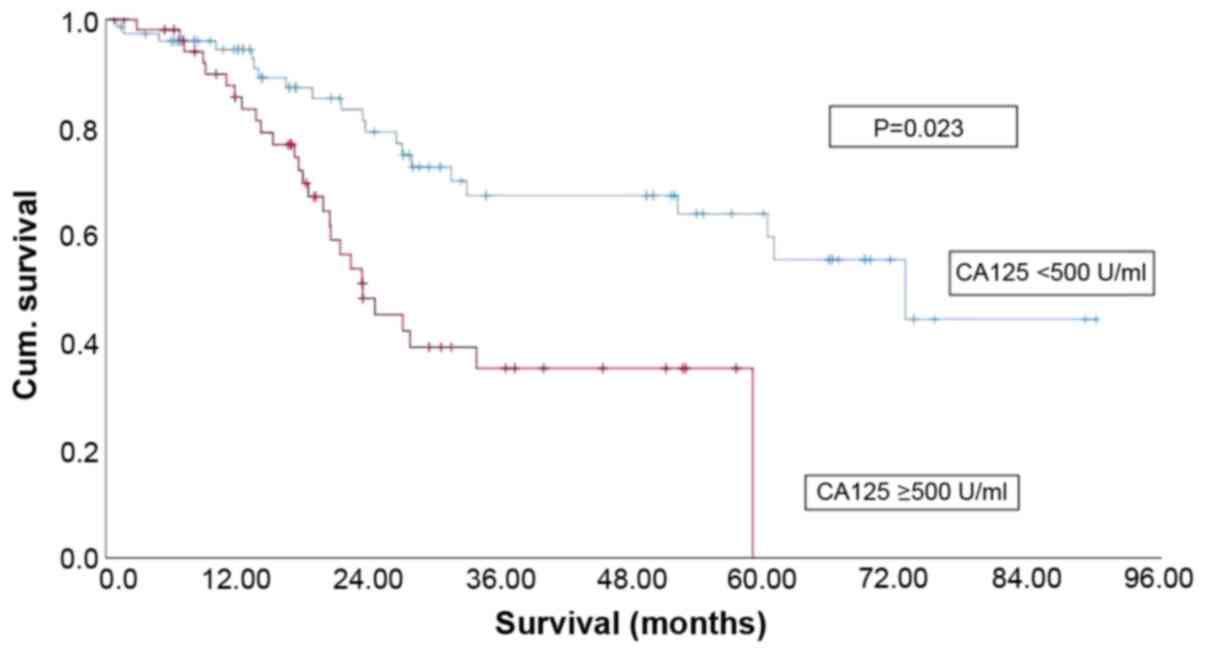
cytoreduced patients compared to CA125 ≥500 U/ml (Table VII; Fig. 1). No significant impact on OS was
detected in suboptimally cytoreduced patients regarding CA125 level
(<500 U/ml vs. ≥500 U/ml; P=n.s., Table VII). Evaluation of PFS:
Significant better impact on PFS was seen in optimally cytoreduced
patients and CA125 <500 U/ml (P=0.011; Table VII) compared to CA125 ≥500 U/ml,
respectively. Similar results were detected for PFS in suboptimally
cytoreduced patients with CA125 level <500 U/ml and CA125 ≥500
U/ml (Table VII) without
significant difference.
 | Table VIIPrognostic impact of CA125 level on
OS and PFS regarding residual tumor mass. |
Table VII
Prognostic impact of CA125 level on
OS and PFS regarding residual tumor mass.
| | OS | | PFS | |
|---|
|
| CA125 level <500
U/ml | CA125 ≥500
U/ml | | CA125 level <500
U/ml | CA125 ≥500
U/ml | |
|---|
| Resection
status | No. | Months (median; 95%
CI) | No. | Months (median; 95%
CI) | P-value | No. | Months (median; 95%
CI) | No. | Months (median; 95%
CI) | P-value |
|---|
| R≤1 cm | 46 | 60.8
(50.2-80.3) | 54 | 23.4
(17.23-29.57) | 0.023 | 46 | 29.43
(19.49-39.38) | 54 | 13.73
(11.22-16.25) | 0.011 |
| R>1 cm | 16 | 49.37
(14.22-84.52) | 19 | 47.97
(4.03-91.9) | 0.716 | 16 | 17.4
(14.15-20.65) | 19 | 16.0
(10.94-21.1) | - |
Kaplan-Meier curves (Figs. 1 and 2) underline the impact on OS after
subdivision of patients due to CA125 level. Fig. 1 demonstrates Kaplan-Meier curves for
OS in optimally cytoreduced patients correlated with CA125 level
(Fig. 1) with significant
difference regarding CA125 level <500 U/ml compared to CA125
≥500 U/ml (P=0.023, Fig. 1).
Optimally cytoreduced patients with CA125 <500 U/ml had
significant better OS than patients with CA125 level ≥500 U/ml and
optimal cytoreduction (P=0.023, Fig.
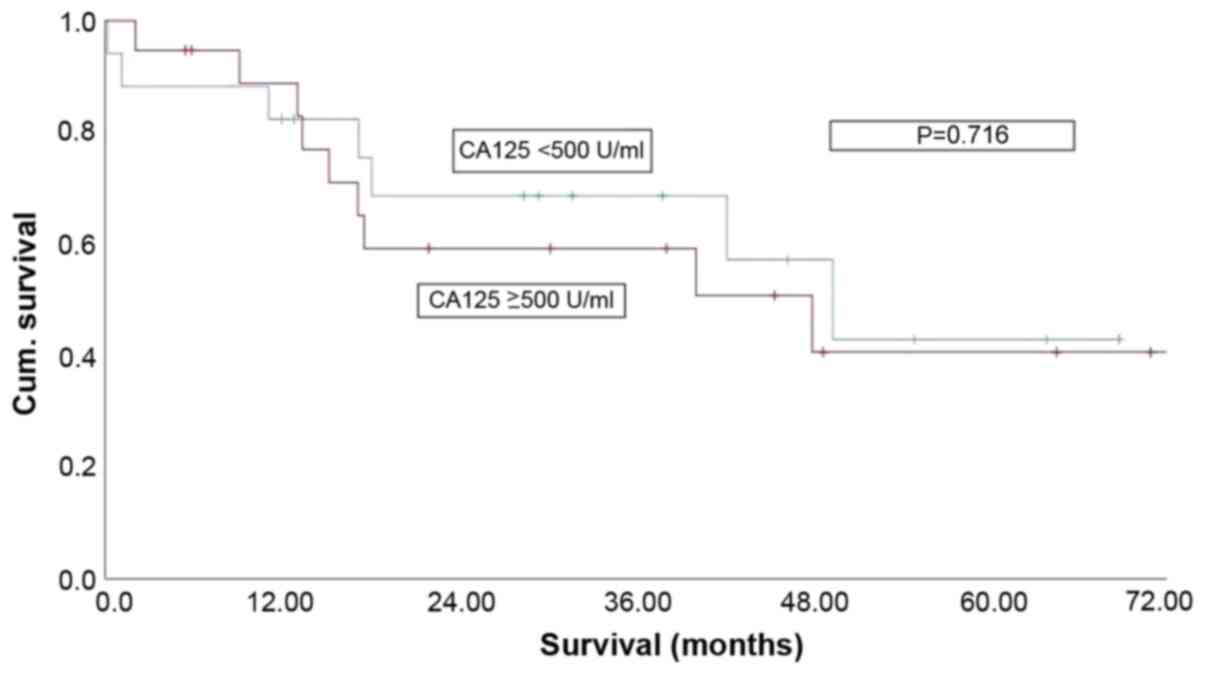
1). Patients with R>1 cm subdivided regarding Ca125 level
≥500 U/ml vs. CA125 <500 U/ml had no significant impact (P=n.s.,
Fig. 2) on OS.
Discussion
Best prognostic impact has optimal cytoreduction in
patients with advanced ovarian cancer as shown in many previous
studies (1,2,18).
Time of surgery is still discussed (2,15).
Important is the fact that suboptimally cytoreduced patients with
(R>1 cm) have similar OS regardless of residual tumor size
(19). Previous studies reported
rates of optimal cytoreduction with wide variation from 15 to 85%
(19). Therefore, the question is
if there are non-invasive factors predicting complete cytoreduction
to stratify patients for primary surgery vs. neoadjuvant
chemotherapy.
Currently, pretreatment CA125 is the only
serological biomarker routinely used in management of epithelial
ovarian cancer patients (4) as a
baseline value to monitor success of ovarian cancer treatment
(3). CA125 level is useful in
treatment monitoring in ovarian cancer and its importance at
initial diagnosis of ovarian cancer is still controversial
(3,4). Normal CA125 level is <35 U/ml.
Approximately 80% of epithelial ovarian cancer patients have
elevated CA125 level (>35 U/ml), with elevations in 50 to 60% of
patients to greater than 90% in advanced FIGO stages (4). This fact corresponds to our data
(Table I). CA125 level ≥500 U/ml
showed mostly advanced FIGO stages (IIIC/IV); early FIGO stages
were not detected in study (Table
I). A previous study demonstrated that pretreatment CA125 level
was significantly lower in patients with grade 1 serous ovarian
cancer compared to those with high-grade serous ovarian cancer
(6).
Known is the increasing CA125 level in advanced FIGO
stages according to other studies as even shown in our analysis
(Table I) (20,21).
Previous studies demonstrated serous ovarian cancers have highest
CA125 level followed by endometrioid and clear cell types (4,21,22);
one study showed CA125 is not expressed in pure mucinous tumors of
ovarian cancer (4). Other studies
demonstrated that besides tumor characteristics CA125 levels are
also influenced by several epidemiologic factors (age, parity oral
contraceptives) and suggest that high BMI may influence CA125
levels independent of tumor characteristics (22). This should be mentioned in CA125
levels.
A study described a subgroup of FIGO III high-grade
serous ovarian cancer patients had an enhanced survival rate with
CA125 level ≥500 U/ml compared to lower levels (21). Against it, this correlation was not
detected in our evaluation; our analysis showed the higher the
CA125 level the worse the progression free survival without
significant difference (Table IV).
Additionally, our analysis described a significant better PFS and
OS in advanced FIGO stages and CA125 level <500 U/ml compared to
CA125 ≥500 U/ml (Table VII).
Especially significant best prognostic impact on PFS and OS was
seen in optimally cytoreduced patients and CA125 level <500 U/ml
compared to CA125 ≥500 U/ml (Table
VII). To this topic a previous study demonstrated that
pretreatment CA125 level was an independent predictor of PFS in
patients with advanced epithelial ovarian cancer who received a
standard chemotherapy regimen, particularly in patients with
debulking to a microscopic residual and in serous or endometrioid
subtypes (23). Optimal
cytoreduction has best prognostic impact (1) so currently a prospective study (TRUST
study, AGO Germany) investigates the optimal timing of
cytoreduction (up-front vs. interval-debulking) to enhance the rate
of optimal cytoreduction. Selection criteria identifying patients
who will benefit from interval debulking surgery are required.
Pretreatment CA125 level is correlated with tumourload as
demonstrated in many previous studies (5), while other studies found no
association (5); achieving complete
cytoreduction is correlated with it as well (5,10).
Many previous studies demonstrated the preoperative CA125 level can
serve as a predictor of optimal tumor cytoreduction in advanced
ovarian cancer. In these studies high preoperative CA125 levels
were associated with decreased likelihood of achieving optimal
cytoreduction, while the cutoff levels differed in them (5). Therefore, other studies demonstrated
that preoperative CA125 level is no reliable predictor of optimal
cytoreduction (5). A previous study
demonstrated a correlation of normal CA125 level with lower
recurrence rate in FIGO III/IV patients compared to FIGO III/IV
patients with higher CA125 levels (5). The question arises if there is a
pretreatment CA125 level predicting the achievement of optimal
cytoreduction is not obtainable and rather interval debulking
should be performed. A previous study found that normal
pretreatment CA125 levels in ovarian cancer patients are
significantly associated with lower stage/grade, optimal
cytoreduction and platinum sensitivity (5). They demonstrated that patients with
normal pretreatment CA125 levels had better PFS and OS compared to
those with elevated CA125 levels (5) and a normal CA125 level at diagnosis of
ovarian cancer is also of prognostic significance, as well
(5). Our analysis demonstrated a
significant better OS and PFS in optimally cytoreduced patients and
CA125 level <500 U/ml compared to higher levels (Table VII).
A previous study found in advanced ovarian cancers
‘no threshold CA125 level that accurately predicted cytoreductive
outcome’ (24). Our analysis
couldn't find a cut-off level of suboptimal debulking, but we found
a association of CA125 levels and suboptimal cytoreduction
(Table III). The group of CA125
1,404 U/ml had higher suboptimal cytoreduction rate than patients
with CA125 ≥500-1,404 U/ml (Table
III), without significant difference. Additionally, there was
no prognostic impact on PFS and OS depending on CA125 levels ≥500
U/ml after subdivision of groups in ≥500-1,404 U/ml and CA125
>1,404 U/ml (Table IV). They
concluded with a preoperative CA-125 >500 U/ml, extensive upper
abdominal procedures were necessary in 50% of cases to achieve
residual disease ≤1 cm compared to those with CA-125 <500 U/ml
(P=0.001) (24). A previous study
evaluated a predictive score to anticipate optimal cytoreduction at
interval debulking surgery (11).
In that study three criteria predicted independently R>0 mm
significantly at interval debulking surgery: CA125 before
neoadjuvant chemotherapy >550 U/ml (11); the other criteria were age ≥ 60
years and Peritoneal Cancer Index (PCI) >16(11).
Previous studies demonstrated various CA125 levels
at which optimal cytoreduction could not be achieved. Therefore,
the analysis of Barlow et al (20) described that optimal cytoreduction
was achieved in ≤40% of patients with CA125 level ≥4,500 in
advanced ovarian cancer patients. They concluded that preoperative
serum CA125 did not reliably predict optimal cytoreduction in
advanced ovarian cancer patients (20).
Our analysis demonstrated with a cut-off value of
CA125 level >1,404 U/ml optimal cytoreduction was achieved in a
lower rate (67.6%; Table III).
This underlines the higher CA125 level the worse optimal
cytoreduction rate.
Another study described CA125 level ≥500 U/ml in
stage III ovarian cancer patients is most often correlated with
advanced FIGO stages and is described as a factor in which
cytoreduction can be achieved in approximately 20% of cases
(10). In our study the rate of
optimally cytoreduced patients was higher in the group of CA125
level ≥500 U/ml in advanced stages (74.3%, Table I). Therefore, the retrospective
character of our analysis and absence of some pretreatment CA125
levels should be noted. Cooper et al (13) demonstrated a ‘preoperative CA 125
values less than 500 U/ml had a positive predictive value for
optimal cytoreduction of 82%, but a poor negative predictive value
of 48%’. They concluded that ‘preoperative CA125 is an independent
risk factor for death due to disease in ovarian cancer, but not a
reliable predictor of optimal cytoreduction’ (13). Our analysis concluded a CA125 level
<500 U/ml has similar rate of optimal cytoreduction compared to
CA125 ≥500 U/ml (Table I), so
pretreatment CA125 level cannot predict optimal cytoreduction, but
has significant prognostic impact on OS and PFS in fact of optimal
cytoreduction and CA125 level <500 U/ml compared to CA125 ≥500
U/ml (Table VII; Figs. 1 and 2). A previous study demonstrated that
pretreatment CA125 level was not useful for predicting clinical
outcome in ovarian clear cell cancer (25). Even the normalization of CA125 level
after chemotherapy cycle 1 leads to decreased survival (25). Another study demonstrated similar to
other previous studies pretreatment CA125 was not associated with
OS, and in contrast to other studies serial CA125 measurements
during chemotherapy were prognostic, with normalization before
second chemotherapy cycle associated with a decreased risk of death
(6). A correlation of reduction of
CA125 level over the initial two cycles of platinum-based
chemotherapy as a powerful independent predictor of survival for
patients was detected in patients with suboptimal stage III or IV
ovarian cancer as well (7). Without
significant declines in CA125 after 2 cycles of platinum-based
chemotherapy they have poor prognosis (7). Another previous study demonstrated
that CA125 levels after the first chemotherapy cycle and even the
time to normalization were the most significant prognostic factors
for both OS and PFS (26).
Therefore, CA125 level seems to be powerful as prognostic marker
during chemotherapy.
Further, our analysis demonstrated in patients with
CA125 level ≥500 U/ml the higher the CA125 level the lower optimal
cytoreduction rate (Table III)
without significant difference. Best improvement on PFS was
detected in this group with complete cytoreduction independent to
level of CA125 level ≥500 U/ml (Table
VI).
Our analysis demonstrated no significant difference
on PFS and OS in patients with CA125 level ≥500 U/ml after
subdivision of group according to CA125 level 1,404.5 U/ml
(Table IV).
Additionally, there was no significant difference of
PFS and OS in patients with CA125 level ≥500 U/ml after subdivision
of group according to CA125 level 1,404.5 U/ml regarding residual
tumour mass (Table V).
There is no predictive biomarker in ovarian cancer
identifying patients who will benefit from treatment or factors
which have predictive value for an optimal cytoreduction. So other
groups investigated other markers and clinical parameter to predict
optimal cytoreduction. Pretreatment HE4 (human epididymis protein
4) seems to be a better marker to predict optimal cytoreduction in
primary surgery than CA125, but should be used in combination with
clinical parameter like ascites <500 ml (27). They found a level of HE4 with a high
rate of predicting optimal cytoreduction in a study with primary
laparoscopic approach followed by primary debulking surgery or NACT
(27). Another study described that
the combination of HE4 and CA125 expression in plasma might predict
the surgical outcome in advanced ovarian cancer patients and found
higher HE4 levels and CA125 levels in patients with suboptimal
cytoreduction (28). A previous
study described pretreatment analysis of HE4 as a superior factor
than CA125 in differential diagnosis of ovarian cancer type I and
II (29). HE4 is superior to CA125
in detecting ovarian cancers type II, but none of these factors
seems to be an effective diagnostic tool for type I ovarian cancers
(29). Further studies are
required. Due to the retrospective character of our study
unfortunately HE4 was not determined in our group. HE4 seems to be
an important factor in preoperative evaluation of cytoreduction,
but should be combined with other factors (27).
Standard of care in ovarian cancer is primary
debulking surgery followed by adjuvant platinum-based chemotherapy
(1,2). A previous study demonstrated that in
patients with primarily inoperable advanced ovarian cancer who got
neoadjuvant chemotherapy followed by interval debulking surgery
have no inferior benefit compared to patients with primary
debulking surgery (1,2,30). The
most important factor is complete cytoreduction (1,2,30) and
optimal treatment is still discussed. Yet, there is no reliable
non-invasive assessment to predict optimal cytoreduction and to
avoid suboptimal debulking in advanced ovarian cancer. Previous
studies evaluated CA125 level, radiologic techniques (CT/PET-CT) to
optimize predicting optimal cytoreduction to stratify patients to
interval debulking surgery vs. neoadjuvant chemotherapy and
evaluated the role of laparoscopy-based scores in management of
advanced ovarian cancer (31). More
recently, laparoscopy has been proposed as a reliable predictor of
complete cytoreduction (31). They
concluded that laparoscopy offers assessment for optimal surgery in
advanced ovarian cancer and presented Fagotti laparoscopy-based
score as a useful predictor of optimal cytoreduction better than
other clinical parameters (31).
A prospective study identified 3 clinical and 6
radiologic criteria significantly associated with suboptimal
debulking (19). One criterium was
CA125 ≥500 U/ml (19). The
combination of these criteria in a prognostic model had a
predictive accuracy of 0.758(19)
and might be useful for treatment planning. Other groups described
that the evaluation of their CONATS index (a novel index for
preoperative, non-invasive prediction of complete primary
cytoreduction in advanced ovarian cancer) combined with
radiological (CT-scan, ultrasound) and/or laparoscopic findings may
improve the assessment of optimal treatment strategy in patients
with advanced epithelial ovarian cancer to predict optimal
cytoreduction (32). This new index
of non-invasive parameters should be used combined with clinical
and radiologic findings as recommended in our analysis.
The Fagotti-score is a laparoscopic model for
prediction of incomplete cytoreduction in advanced epithelial
ovarian cancer (33). The updated
score showed a lower rate of inappropriate laparotomic explorations
(33), so laparoscopy improves
further management in these patients. A previous study demonstrated
that CT may be a useful preoperative predictor of suboptimal
debulking in advanced ovarian cancer (34). Yet, there is still no final approach
to predict optimal cytoreduction.
A previous study evaluates a non-invasive test to
predict complete cytoreduction. The serum analysis demonstrates
that the combination of serum miR-34a-5p and CA125 could predict
surgical outcome in high grade serous ovarian cancer (35). They conclude that ‘a molecular test
incorporating circulating miRNA to predict completeness of surgical
resection has the potential to contribute planning for optimal
management (35). Further results
are required.
Our data showed a correlation of CA125 level ≥500
U/ml and residual tumour mass (Table
III) according to other studies (10). The range of CA125 level associated
with optimal cytoreduction was 501-16,062 U/ml (Table II). The correlation of CA125 level
and residual tumour mass showed highest median CA125 level in
suboptimally debulked patients (2,347 U/ml) with a range of
502-48,470 U/ml compared to patients with complete cytoreduction
(R=0 mm) and R>0 mm-≤1 cm (1,332.5 vs. 1,250 U/ml; Table II). So optimal cytoreduction can be
achieved in individual patients despite very high CA125 levels
(Table II). In our study, we
demonstrate a significant better prognostic impact on OS and PFS in
optimally cytoreduced patients as well (OS: P=0.023; PFS: P=0.011;
Table VII; Fig. 1) in the group of CA125 <500 U/ml
compared to CA125 ≥500 U/ml. Therefore, we could demonstrate that
CA125 ≥500 U/ml has relevance for frequency of complete
cytoreduction and prognosis. These patients might benefit from an
initial laparoscopic approach to achieve a histological evidence of
disease and to specify resectability to determine how to proceed
(primary debulking surgery vs. neoadjuvant chemotherapy) taking
into account radiologic findings. Previous studies have shown that
CA125 is insufficient as a single predictor for optimal debulking,
as seen in our analysis, and several more complex models have since
been suggested (14). Another study
described CA125 level as a reliable component in preoperative
management as a predictor of suboptimal debulking (14). That study demonstrates an optimal
cut-off for serum CA125 as a predictor of suboptimal debulking of
586 U/ml (36), in contrast our
cut-off CA125 level was higher in suboptimal debulking (Table III). Arits et al (36) could not find a clear correlation
between preoperative serum CA125 levels and suboptimal debulking.
They consider the radiologic amount of ascites and considerable
weight loss instead of preoperative CA125 values as independent
prognostic predictors for suboptimal cytoreduction in patients with
advanced ovarian cancer and recommend the discussion of neoadjuvant
chemotherapy in these cases (36).
A further analysis described that CA125 ≥500 U/ml demonstrated
strong association with a risk of suboptimal cytoreduction
(37), as seen in our analysis
(Table III) and it may be applied
in preoperative counseling and treatment planning (37), but an accurate possibility to
predict optimal cytoreduction won't work with CA125 level alone
(37).
Another study demonstrated preoperative CA125 is a
poor, but statistically significant predictive factor for complete
cytoreduction after primary cytoreductive surgery, but has no
predictive impact on PFS nor OS in a group of high-grade vs.
low-grade ovarian cancer patients (38). Our study demonstrated a significant
prognostic impact of CA125 level on OS and PFS with CA125 <500
U/ml in optimally cytoreduced patients compared to CA125 ≥500 U/ml
and demonstrated a higher rate of complete cytoreduction in lower
CA125 levels (Table VI),
respectively. The comparison of patients with CA125 <500 U/ml
vs. CA125 ≥500 U/ml leads to the finding of a comparable rate of
suboptimally cytoreduced patients (25.7% vs. 25.8%; Table I), but we could demonstrate a
significant better impact on OS and PFS in the group of CA125
<500 U/ml leading to a better prognosis (Table VII).
Our analysis demonstrates with CA125 level ≥500 U/ml
the higher the CA125 level the higher the suboptimal cytoreduction
rate (Table III). The prognostic
impact on PFS and OS is worse the higher the CA125 level
(>1,404.5 U/ml), but there is no significant difference
(Table IV). Additionally, in the
group with CA125 level ≥500 U/ml we could demonstrate that patients
with higher CA125 level (>1,404.5 U/ml) have most often shorter
or similar OS and PFS, respectively, regarding the residual tumour
mass without significant difference (Table V). To improve rate of optimal
debulking and knowing higher suboptimal cytoreduction rate with
higher CA125 level, as demonstrated in our analysis (Table III), laparoscopic approach should
be mentioned in this context next to clinical and radiological
findings.
Another very interesting aspect to CA125 levels and
metastases is demonstrated in a previous study. They demonstrated
that CA125 stimulates ovarian cancer cell migration via the Wnt
signaling pathway (8).
Additionally, they evaluated a new cut-off value for serum CA125
(82.9 U/ml) that may be predictive of metastasis in patients with
ovarian cancer (8). This result
might be correlated with the known fact that the higher CA125 level
more often advanced FIGO stages are found, but further results are
still missing.
With discordant data on the impact of CA125 level
predicting optimal cytoreduction pretreatment CA125 level should be
used in combination with other parameters (clinical and radiologic
findings) in defining treatment strategy. This may improve defining
optimal treatment strategy in these patients to resolve this
clinical dilemma and predict preoperatively which patients with
ovarian cancer could be cytoreduced optimally. The current TRUST
study (AGO-Ovar) will answer the question of resectability with a
two-armed study comparing upfront primary debulking surgery vs.
neoadjuvant chemotherapy followed by debulking surgery and will
hopefully improve the individualized management and optimize time
of surgery. Hopefully, that study will also identify predictive and
prognostic biomarkers of resectability/survival and parameters
toward a more individualized surgical treatment.
Considering the results of our analysis and those of
previous studies, CA125 levels seem to be helpful in predicting
suboptimal cytoreductive surgery for patients with primary ovarian
cancer, but should be interpreted in association with clinical and
radiologic (CT-scan/ultrasound) findings. The higher CA125 levels
the lower the rate of optimal cytoreduction in our analysis. In
optimally cytoreduced patients pretreatment CA125 ≥500 U/ml has
significant worse prognostic impact on OS (P=0.023) and PFS
(P=0.011) compared to CA125 <500 U/ml. The evaluation of
clinical and radiological parameter may improve defining the
optimal treatment strategy in these patients. At present, there is
no sole non-invasive parameter predicting optimal cytoreduction.
Perhaps these patients can be chosen by pretreatment CA125 levels.
Patients with CA125 ≥500 U/ml may be candidates for initial
laparoscopic approach to obtain histologic diagnosis, to evaluate
resectability and to determine how to proceed (primary debulking
surgery vs. neoadjuvant chemotherapy) taking into account of
radiologic findings.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
RB made substantial contributions to conception and
design, acquisition of data, analysis and interpretation, and was
involved in drafting the manuscript and revising it critically for
important intellectual content. SB was involved in analysis and
interpretation of data, and drafting the manuscript and revising it
critically for important intellectual content. AS performed
histopathologic analysis of specimen and made substantial
contributions to acquisition of data, and was involved in revising
the manuscript critically for important intellectual content. BK,
RL, AK and DW were involved in analysis and interpretation of data,
and drafting the manuscript and revising it critically for
important intellectual content. CB made substantial contributions
to conception and design, data acquisition, analysis and
interpretation of data, and was involved in drafting the manuscript
and revising it critically for important intellectual content. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
All patients provided written informed consent prior
to inclusion in the evaluation. This investigation was approved by
the appropriate ethics committee of University Hospital Tuebingen
(May 6, 2015; approval no. 244/2015R) and has therefore been
performed in accordance with the ethical standards laid down in the
1964 Declaration of Helsinki.
Patient consent for publication
All patients provided written informed consent for
publication of data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
du Bois A, Reuss A, Pujade-Lauraine E,
Harter P, Ray-Coquard I and Pfisterer J: Role of surgical outcome
as prognostic factor in advanced epithelial ovarian cancer: A
combined exploratory analysis of 3 prospectively randomized phase 3
multicenter trials: By the arbeitsgemeinschaft gynaekologische
onkologie studiengruppe ovarialkarzinom (AGO-OVAR) and the groupe
d'investigateurs nationaux pour les etudes des cancers de l'Ovaire
(GINECO). Cancer. 115:1234–1244. 2009.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Vergote I, Tropé CG, Amant F, Kristensen
GB, Ehlen T, Johnson N, Verheijen RH, van der Burg ME, Lacave AJ,
Panici PB, et al: Neoadjuvant chemotherapy or primary surgery in
stage IIIC or IV ovarian cancer. N Engl J Med. 363:943–953.
2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bottoni P and Scatena R: The role of CA
125 as tumor marker: biochemical and clinical aspects. Adv Exp Med
Biol. 867:229–244. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Sölétormos G, Duffy MJ, Othman Abu Hassan
S, Verheijen RH, Tholander B, Bast RC Jr, Gaarenstroom KN, Sturgeon
CM, Bonfrer JM, Petersen PH, et al: Clinical use of cancer
biomarkers in epithelial ovarian cancer updated guidelines from the
European group on tumor markers. Int J Gynecol Cancer. 26:43–51.
2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Menczer J, Ben-Shem E, Golan A and Levy T:
The significance of normal pretreatment levels of CA125 (<35
U/ml) in epithelial ovarian carcinoma. Rambam Maimonides Med J.
6(e0005)2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Fader AN, Java J, Krivak TC, Bristow RE,
Tergas AI, Bookman MA, Armstrong DK, Tanner EJ and Gershenson DM:
The prognostic significance of pre- and post-treatment CA-125 in
grade 1 serous ovarian carcinoma: A gynecologic Oncology Group
study. Gynecol Oncol. 132:560–565. 2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Markman M, Federico M, Liu PY, Hannigan E
and Alberts D: Significance of early changes in the serum CA-125
antigen level on overall survival in advanced ovarian cancer.
Gynecol Oncol. 103:195–198. 2006.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yuan Q, Song J, Yang W, Wang H, Huo Q,
Yang J, Yu X, Liu Y, Xu C and Bao H: The effect of CA125 on
metastasis of ovarian cancer: Old marker new function. Oncotarget.
8:50015–50022. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Randall LM, Sill MW, Burger RA, Monk BJ,
Buening B and Sorosky JI: Predictive value of serum CA-125 levels
in patients with persistent or recurrent epithelial ovarian cancer
or peritoneal cancer treated with bevacizumab on a Gynecologic
Oncology Group phase II trial. Gynecol Oncol. 124:563–568.
2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chi DS, Venkatraman ES, Masson V and
Hoskins WJ: The ability of preoperative serum CA-125 to predict
optimal primary tumor cytoreduction in stage III epithelial ovarian
carcinoma. Gynecol Oncol. 77:227–231. 2000.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ghisoni E, Katsaros D, Maggiorotto F,
Aglietta M, Vaira M, De Simone M, Mittica G, Giannone G, Robella M,
Genta S, et al: A predictive score for optimal cytoreduction at
interval debulking surgery in epithelial ovarian cancer: A two-
centers experience. J Ovarian Res. 11(42)2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Memarzadeh S, Lee SB, Berek JS and
Farias-Eisner R: CA-125 levels are a weak predictor of optimally
cytoreductive surgery in patients with advance epithelial ovarian
cancer. Int J Gynecol Cancer. 13:120–124. 2003.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Cooper BC, Sood AK, Davis CS, Ritchie JM,
Sorosky JI, Anderson B and Buller RE: Preoperative CA 125 levels:
An independent prognostic factor for epithelial ovarian cancer.
Obstet Gynecol. 100:59–64. 2002.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Brockbank EC, Ind TE, Barton DP, Shepherd
JH, Gore ME, A'Hern R and Bridges JE: Preoperative predictors of
suboptimal primary surgical cytoreduction in women with clinical
evidence of advanced primary epithelial ovarian cancer. Int J
Gynecol Cancer. 14:42–50. 2004.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Gill SE, McGree ME, Weaver AL, Cliby WA
and Langstraat CL: Optimizing the treatment of ovarian cancer:
Neoadjuvant chemotherapy and interval debulking versus primary
debulking surgery for epithelial ovarian cancers likely to have
suboptimal resection. Gynecol Oncol. 144:266–273. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Suidan RS, Ramirez PT, Sarasohn DM,
Teitcher JB, Iyer RB, Zhou Q, Iasonos A, Denesopolis J, Zivanovic
O, Long Roche KC, et al: A multicenter assessment of the ability of
preoperative computed tomography scan and CA-125 to predict gross
residual disease at primary debulking for advanced epithelial
ovarian cancer. Gynecol Oncol. 145:27–31. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Prat J: FIGO Committee on Gynecologic
Oncology. Staging classification for cancer of the ovary, fallopian
tube, and peritoneum. Int J Gynaecol Obstet. 124:1–5.
2014.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Wimberger P, Lehmann N, Kimmig R, Burges
A, Meier W and Du Bois A: Arbeitsgemeinschaft Gynaekologische
Onkologie Ovarian Cancer Study Group. Prognostic factors for
complete debulking in advanced ovarian cancer and its impact on
survival. An exploratory analysis of a prospectively randomized
phase III study of the Arbeitsgemeinschaft gynaekologische
onkologie ovarian cancer study group (AGOOVAR). Gynecol Oncol.
106:69–74. 2007.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Suidan RS, Ramirez PT, Sarasohn DM,
Teitcher JB, Mironov S, Iyer RB, Zhou Q, Iasonos A, Paul H, Hosaka
M, et al: A multicenter prospective trial evaluating the ability of
preoperative computed tomography scan and serum CA-125 to predict
suboptimal cytoreduction at primary debulking surgery for advanced
ovarian, fallopian tube, and peritoneal cancer. Gynecol Oncol.
134:455–461. 2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Barlow TS, Przybylski M, Schilder JM,
Moore DH and Look KY: The utility of presurgical CA125 to predict
optimal tumor cytoreduction of epithelial ovarian cancer. Int J
Gynecol Cancer. 16:496–500. 2006.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Morales-Vásquez F, Pedernera E,
Reynaga-Obregón J, López-Basave HN, Gómora MJ, Carlón E, Cárdenas
S, Silva-Ayala R, Almaraz M and Méndez C: High levels of
pretreatment CA125 are associated to improved survival in high
grade serous ovarian carcinoma. J Ovarian Res. 9(41)2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Babic A, Cramer DW, Kelemen LE, Köbel M,
Steed H, Webb PM, Johnatty SE, deFazio A, Lambrechts D, Goodman MT,
et al: Predictors of pretreatment CA125 at ovarian cancer
diagnosis: A pooled analysis in the ovarian cancer association
consortium. Cancer Causes Control. 28:459–468. 2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zorn KK, Tian C, McGuire WP, Hoskins WJ,
Markman M, Muggia FM, Rose PG, Ozols RF, Spriggs D and Armstrong
DK: The prognostic value of pretreatment CA 125 in patients with
advanced ovarian carcinoma: A Gynecologic Oncology Group study.
Cancer. 115:1028–1035. 2009.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Chi DS, Zivanovic O, Palayekar MJ,
Eisenhauer EL, Abu-Rustum NR, Sonoda Y, Levine DA, Leitao MM, Brown
CL and Barakat RR: A contemporary analysis of the ability of
preoperative serum CA-125 to predict primary cytoreductive outcome
in patients with advanced ovarian, tubal and peritoneal carcinoma.
Gynecol Oncol. 112:6–10. 2009.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Bai H, Sha G, Xiao M, Gao H, Cao D, Yang
J, Chen J, Wang Y, Zhang Z and Shen K: The prognostic value of
pretreatment CA-125 levels and CA-125 normalization in ovarian
clear cell carcinoma: A two-academic-institute study. Oncotarget.
7:15566–15576. 2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Lee M, Chang MY, Yoo H, Lee KE, Chay DB,
Cho H, Kim S, Kim YT and Kim JH: Clinical significance of CA125
level after the first cycle of chemotherapy on survival of patients
with advanced ovarian cancer. Yonsei Med J. 57:580–587.
2016.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Angioli R, Plotti F, Capriglione S, Aloisi
A, Montera R, Luvero D, Miranda A, Cafà EV, Damiani P and
Benedetti-Panici P: Can the preoperative HE4 level predict optimal
cytoreduction in patients with advanced ovarian carcinoma? Gynecol
Oncol. 128:579–583. 2013.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Braicu EI, Fotopoulou C, Van Gorp T,
Richter R, Chekerov R, Hall C, Butz H, Castillo-Tong DC, Mahner S,
Zeillinger R, et al: Preoperative HE4 expression in plasma predicts
surgical outcome in primary ovarian cancer patients: Results from
the OVCAD study. Gynecol Oncol. 128:245–251. 2013.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Gąsiorowska E, Michalak M, Warchoł W,
Lemańska A, Jasiński P, Spaczyński M and Nowak-Markwitz E: Clinical
application of HE4 and CA125 in ovarian cancer type I and type II
detection and differential diagnosis. Ginekol Pol. 86:88–93.
2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Sioulas VD, Schiavone MB, Kadouri D,
Zivanovic O, Roche KL, O'Cearbhaill R, Abu-Rustum NR, Levine DA,
Sonoda Y, Gardner GJ, et al: Optimal primary management of bulky
stage IIIC ovarian, fallopian tube and peritoneal carcinoma: Are
the only options complete gross resection at primary debulking
surgery or neoadjuvant chemotherapy? Gynecol Oncol. 145:15–20.
2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Gómez-Hidalgo NR, Martinez-Cannon BA, Nick
AM, Lu KH, Sood AK, Coleman RL and Ramirez PT: Predictors of
optimal cytoreduction in patients with newly diagnosed
advanced-stage epithelial ovarian cancer: Time to incorporate
laparoscopic assessment into the standard of care. Gynecol Oncol.
137:553–558. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Karlsen MA, Fagö-Olsen C, Høgdall E,
Schnack TH, Christensen IJ, Nedergaard L, Lundvall L, Lydolph MC,
Engelholm SA and Høgdall C: A novel index for preoperative,
non-invasive prediction of macro-radical primary surgery in
patients with stage IIIC-IV ovarian cancer-a part of the Danish
prospective pelvic mass study. Tumour Biol. 37:12619–12626.
2016.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Petrillo M, Vizzielli G, Fanfani F,
Gallotta V, Cosentino F, Chiantera V, Legge F, Carbone V, Scambia G
and Fagotti A: Definition of a dynamic laparoscopic model for the
prediction of incomplete cytoreduction in advanced epithelial
ovarian cancer: Proof of a concept. Gynecol Oncol. 139:5–9.
2015.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Son HM, Kim SH, Kwon BR, Kim MJ, Kim CS
and Cho SH: Preoperative prediction of suboptimal resection in
advanced ovarian cancer based on clinical and CT parameters. Acta
Radiol. 58:498–504. 2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Shah JS, Gard GB, Yang J, Maidens J,
Valmadre S, Soon PS and Marsh DJ: Combining serum microRNA and
CA-125 as prognostic indicators of preoperative surgical outcome in
women with high-grade serous ovarian cancer. Gynecol Oncol.
148:181–188. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Arits AH, Stoot JE, Botterweck AA, Roumen
FJ and Voogd AC: Preoperative serum CA125 levels do not predict
suboptimal cytoreductive surgery in epithelial ovarian cancer. Int
J Gynecol Cancer. 18:621–628. 2008.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Kang S, Kim TJ, Nam BH, Seo SS, Kim BG,
Bae DS and Park SY: Preoperative serum CA-125 levels and risk of
suboptimal cytoreduction in ovarian cancer: A meta-analysis. J Surg
Oncol. 101:13–17. 2010.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Muallem MZ, Parashkevova A, Almuheimid J,
Richter R, Diab Y, Braicu EI and Sehouli J: Preoperative CA-125
values as a predictive factor for the postoperative outcome in
primary serous ovarian cancer. Anticancer Res. 37:3157–3161.
2017.PubMed/NCBI View Article : Google Scholar
|