Introduction
Ovarian borderline tumors (OBT) have firstly been
reported as the entity of ovarian tumors by Taylor in 1929(1). After then, OBT was recognized by the
International Federation of Gynecology and Obstetrics (FIGO) in
1971 and the World Health Organization (WHO) in 1973 (2,3).
Briefly, histological feature of OBT was the presence of slight
nuclear atypia or cellular proliferation, with or without
microinvasion defined as small foci of stromal invasion measuring
<5 mm in greatest linear extent (4). Its 10-year survival was 99% for FIGO
stage I, 98% for stage II, 96% for stage III, and 77% for stage IV,
respectively (5). The term of OBT
has been changed during decades, but the concept has been adopted
to 2020 WHO criteria (6).
In histological subtypes of OBT, the major types
were serous (50%) and mucinous (45%) borderline tumor.
Endometrioid, clear cell, and seromucinous borderline tumor, and
borderline Brenner tumor were the minor types (4,7).
According to 2020 WHO criteria, the frequency of clear cell
borderline tumor (CCBT) was <1% of OBT and was defined as an
adenofibromatous clear cell tumors with glandular crowding and
low-grade nuclear atypia without stromal invasion (6). Hence, Suzuki et al reported
CCBT without fibromatous component (8). However, due to the rarity, the
clinical outcome was unclear because CCBT, particularly, without
fibromatous component was rare.
Herein, the aim of our study was to explore CCBT
through pathological review for cases diagnosed with CCBT and clear
cell carcinoma (CCC) and review literature about CCBT with and
without fibromatous component.
Materials and methods
Patients, pathological review and
definition
Patients with ovarian CCBT or CCC treated with
surgery at our hospital between 1984 and 2015 were identified. We
excluded patients which had no medical records and hematoxylin and
eosin (HE) slide. Pathological review for all patients using the
definition of 2020 WHO criteria and the previous report by
Lokuhetty et al and Suzuki et al (6,8).
Briefly, the definition of CCBT was that tumors characterized by
glands lined by cubic or flat cells with enlarged nuclei, clear or
eosinophilic cytoplasm, low mitotic activity, and sometimes
nucleoli, with or without fibromatous component. Also, the
definition of CCC was that tumors composed of clear, eosinophilic,
and hobnail cells, with tubulocystic, papillary, and solid
architecture.
Immunohistochemistry (IHC) staining
and interpretation of IHC staining
For IHC staining, we used rabbit monoclonal antibody
for hepatocyte nuclear factor 1 beta (HNF1-β) (EPR18644-13;
dilution 1:2,000; Abcam), mouse monoclonal antibody for p53 (DO7,
dilution 1:50; Dako), mouse monoclonal antibody for Wilms' tumor 1
(WT1) (6F-H2; dilution 1:50; Dako), and mouse monoclonal antibody
for Ki-67 (M7240; dilution 1:50; Dako). All specimens were cut into
4 µm thick slices to make tissue sections for IHC staining. The
tissue sections were deparaffinized in xylene and hydrated with
alcohol. Endogenous peroxidase activity was blocked using methanol
added to 0.3% hydrogen peroxidase. The tissue sections were boiled
at 98˚C for 40 min in Tris/EDTA buffer (pH 9.0) using HNF-1β and in
an autoclave at 121˚C for 15 min in citrate buffer (pH 6.0) using
p53, WT-1, and Ki-67, and were then allowed to cool at room
temperature. The slides were incubated at 4˚C overnight with
primary antibodies. Following incubation, the samples were reacted
with the DAKO EnVision + system-HRP labeled polymer as secondary
antibody for 30 min at room temperature. Specific antigen-antibody
reactions were visualized with 0.2% diaminobenzidine
tetrahydrochloride and hydrogen peroxide, and counterstained with
Mayer's hematoxylin. As negative controls, tissue sections without
the primary antibody were used. For the evaluation of IHC
activities of HNF1-β, p53, WT1, and Ki-67, the presence of nuclear
immunoreaction in >10% of all tumor cells was defined as
positive.
Medical and surgical data, stage and
ethics approval
Medical and surgical data were obtained from the
medical and surgical records. All cases were staged according to
the 2014 International Federation of Gynecology and Obstetrics
(FIGO) staging system (9). This
study was approved by the Ethics Committee of the National Defense
Medical College, Tokorozawa, Japan.
Results
Results of pathological review
During study period, 136 cases with CCC and 2 cases
with CCBT were identified. Through pathological review, Among 136
cases with CCC, 126 cases were diagnosed with CCC, 10 cases with
other histological subtypes, and there were no cases with CCBT
(Table I). Among 126 cases with
CCC, median age was 53.4 and 76 cases (60.3%) were diagnosed with
FIGO stage I, 17 cases (13.5%) with FIGO stage II, 30 cases (23.8%)
with FIGO stage III, and 3 cases (2.4%) with FIGO stage IV.
Endometriosis was observed in 58 cases (46.0%). Seventy-three cases
(57.9%) had Positive peritoneal cytology, and 28 cases (22.2%) had
residual tumor at primary surgery. Conventional chemotherapy was
performed for 120 cases (95.3%). Among cases with evaluable
diseases, 12 cases (42.9%) were complete response or partial
response to conventional chemotherapy. Two cases were diagnosed
with CCBT. The initial diagnosis of these 2 cases were CCBT.
 | Table ICharacteristics of two cases with
clear cell borderline tumor and 126 cases with clear cell
carcinoma. |
Table I
Characteristics of two cases with
clear cell borderline tumor and 126 cases with clear cell
carcinoma.
| Variables | Clear cell borderline
tumor (n=2) | Clear cell carcinoma
(n=126) |
|---|
| Age (years) | | |
|
Median ±
SD | - | 53.4±9.4 |
| FIGO stage (%) | | |
|
I | 2 (100.0) | 76 (60.3) |
|
II | 0 (0.0) | 17 (13.5) |
|
III | 0 (0.0) | 30 (23.8) |
|
IV | 0 (0.0) | 3 (2.4) |
| Endometriosis
(%) | | |
|
Yes | 2 (100.0) | 58 (46.0) |
|
No | 0 (0.0) | 68 (54.0) |
| Peritoneal cytology
(%) | | |
|
Positive | 0 (0.0) | 73 (57.9) |
|
Negative | 2 (100.0) | 53 (42.1) |
| Residual tumor at
primary | | |
| surgery (%) | | |
|
Yes | 0 (0.0) | 28 (22.2) |
|
No | 2 (100.0) | 98 (77.8) |
| Adjuvant chemotherapy
(%) | | |
|
Taxane-platinum
therapy | 0 (0.0) | 33 (26.2) |
|
Platinum-based
therapy | 0 (0.0) | 87 (69.1) |
|
Not
administered | 2 (100.0) | 6 (4.7) |
| Response rate
(%) | | |
|
CR/PR | N/A | 12 (42.9) |
|
SD/PD | N/A | 16 (57.1) |
Case 1
A 43-year-old woman, gravida 0, para 0, with no
symptom was referred to our hospital for ovarian tumor. She had no
surgical, medical, and specific family history. Serum tumor markers
did not elevate: 9.2 U/ml of carbohydrate antigen 25 (CA125) and
6.8 U/ml of carbohydrate antigen 9-9 (CA19-9). Magnetic resonance
imaging (MRI) showed that multilocular ovarian cyst with a size of
86x50x65 mm had a solid part with a weak Gadolinium enhancement in
low signal area in T2-weighted images. Computer tomography (CT)
images did not reveal no metastasis. The endometrial and cervical
cytology could not detect malignant cell. The preoperative
diagnosis suspected OBT. She received total hysterectomy, bilateral
salpingo-oophorectomy and partial omentectomy because operative
rapid pathological diagnosis showed borderline ovarian tumor. In
the pelvic cavity, the uterus and bilateral ovaries did not adhere
to the other pelvic organs. The ovarian tumor was unruptured.
Ascites was not observed. Macroscopically, the left ovary had
multiple cysts with a size of 20x40 mm. Pathologically, cysts were
lined by cuboidal and hobnail cells with clear and eosinophilic
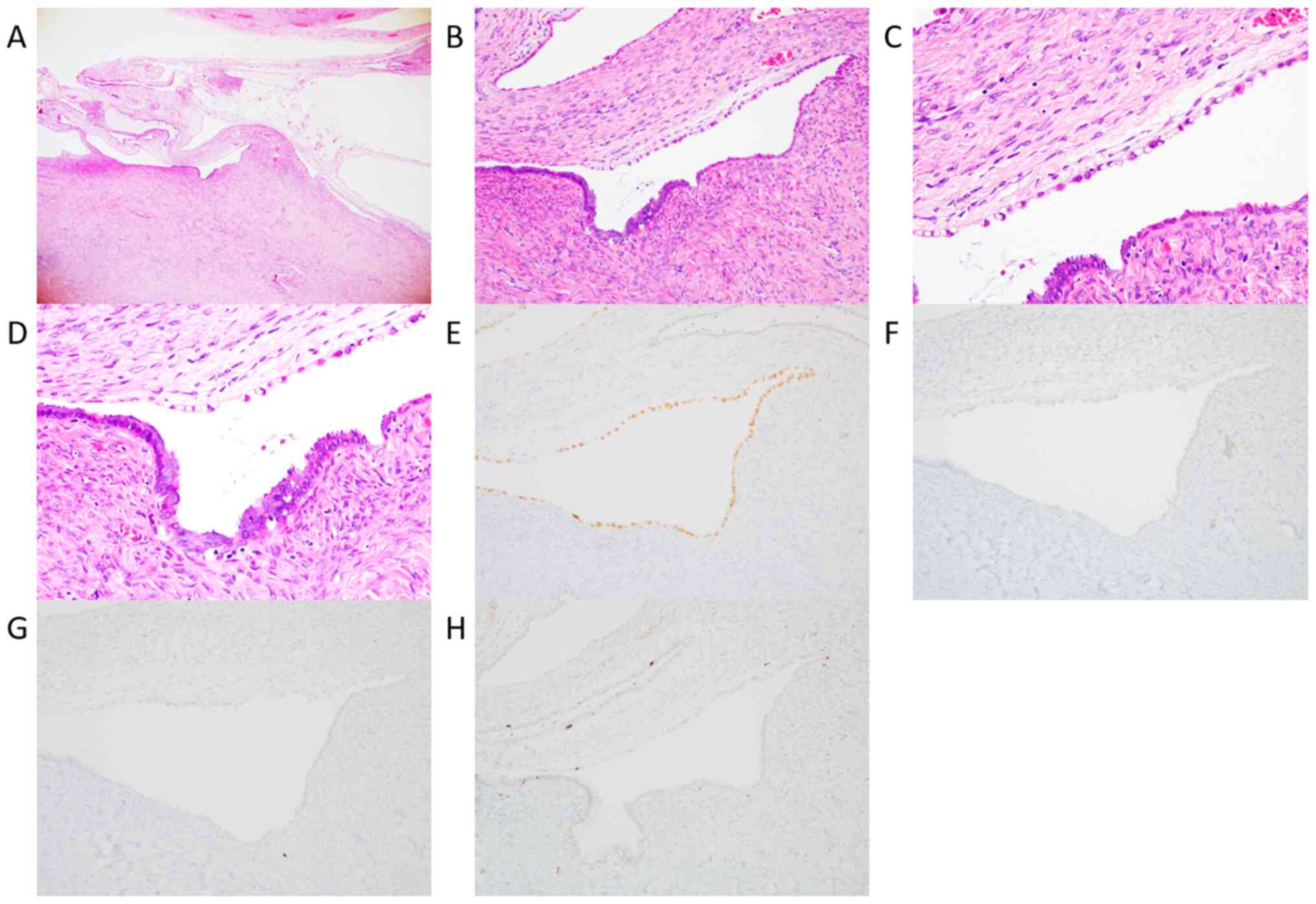
cytoplasm (Fig. 1A-D). The lining
cells showed moderate nuclear atypia without microinvasion and
fibromatous component. CCBT was adjacent to atypical endometriosis.
In IHC analysis, almost all CCBT cells was positive for HNF-1β
(Fig. 1E), and negative for p53
(Fig. 1F) and WT-1 (Fig. 1G). They were partially positive for
Ki-67 (<10%, Fig. 1H). She was
diagnosed with CCBT without fibromatous component. She did not
receive any adjuvant therapy and lived with no evidence of disease
for 37 months from surgery.
Case 2
A 42-year-old woman, gravida 3, para 3, presented
with no symptom. She had no surgical, medical, and specific family
history. Serum tumor markers did not elevate: 26.2 U/ml of CA125
and 33.7 U/ml of CA19-9. Transvaginal ultrasonography revealed
unilocular cyst with a size of ~8 cm. MRI demonstrated that
unilocular ovarian cyst with the size of 80 mm had scattered solid
part with a weak Gadolinium enhancement in low signal area in
T2-weighted images. CT images did not reveal no metastasis. The
endometrial and cervical cytology did not enable us to detect
malignant cells. The preoperative diagnosis suspected OBT. The
patient underwent left salpingo-oophorectomy, partial omentectomy,
and pelvic lymphadenectomy because operative rapid pathological
diagnosis revealed serous or mucinous borderline tumor. In the
pelvic cavity, the left ovary adhered to the uterus. The ovarian
tumor was unruptured. There was a little amount of ascites.
Macroscopically, the left ovary was a cyst with the size of
76x67x42 mm and nodule component was observed in the cyst.
Pathologically, nodule component was lined by increasing calcified
spindle cells without atypia. The tumor cells were lined by hobnail
cells with mild nuclear atypia and eosinophilic cytoplasm without
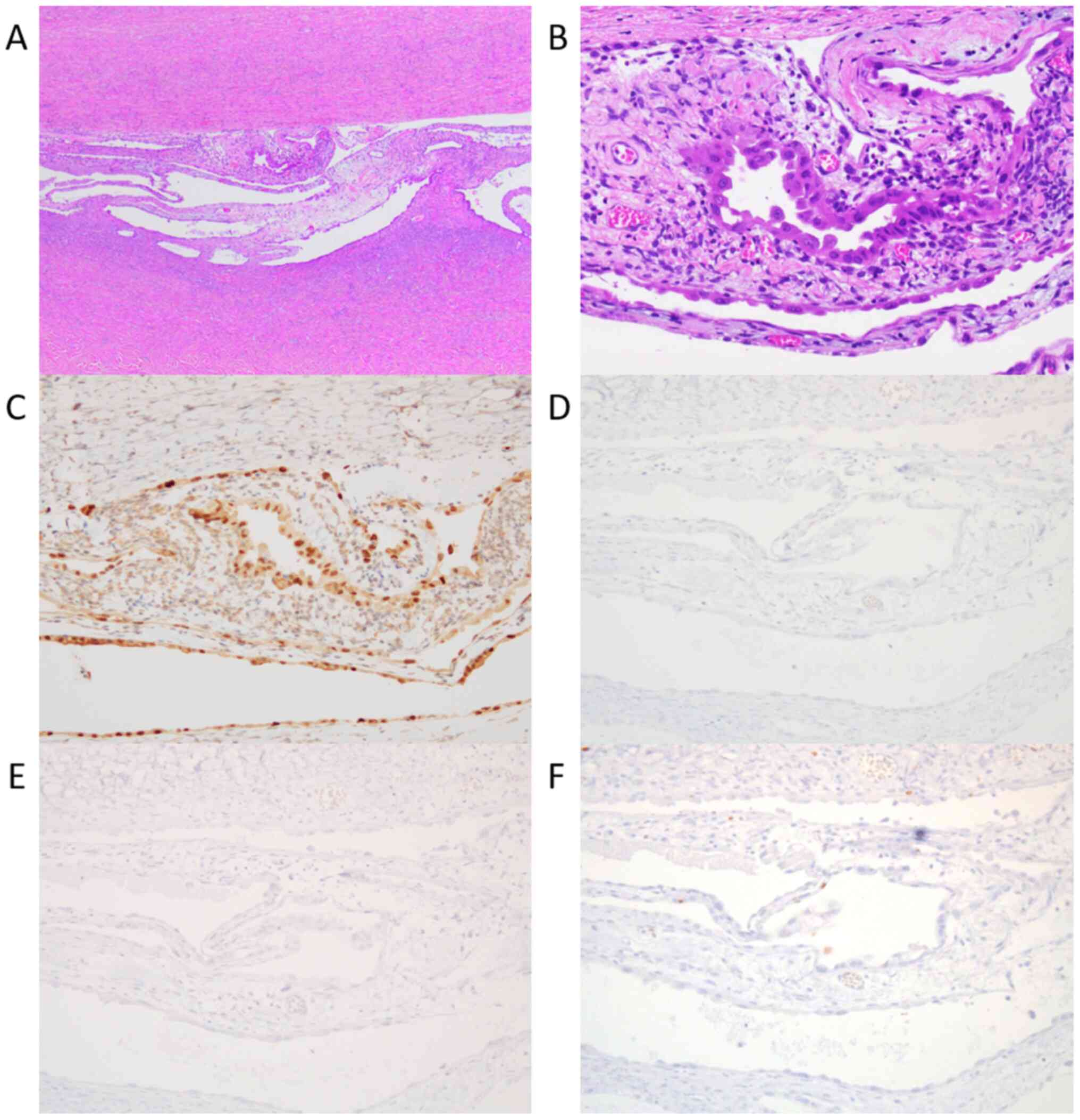
microinvasion, fibromatous component, and endometriosis (Fig. 2A and B). In IHC analysis, almost all CCBT cells
were positive for HNF-1β (Fig. 2C),
negative for p53 (Fig. 2D), WT-1
(Fig. 2E), and Ki-67 (Fig. 2F). She was diagnosed with CCBT
without fibromatous component and lived with no evidence of disease
after the surgery for 20 months.
Discussion
In our study, we reviewed 136 cases with CCC and 2
cases with CCBT, and found 2 cases with CCBT through review. There
was no fibromatous component in both cases with CCBT, and 1 of them
had endometriosis component which showed a continuum of
differentiation to CCBT component.
CCBT was reported to be rare and difficult to
diagnose (10). The discriminating
histological findings between CCBT and CCC was nuclear grading.
CCBT was characterized by an intermediate nuclear grade, while CCC
was characterized by at least focal high-grade nuclear atypia with
prominent nucleoli. However, nuclear grading was highly subjective
and that should err on the side of malignancy (11). Also, when CCBT was complicated with
serous borderline tumor or seromucinous borderline tumors, the
diagnosis was difficult. This complication was frequently observed
(12,13). Then, immunochemical study was
helpful and CCBT was negative for WT-1 and p53 and positive for
HNF-1β (14,15). In our study, no case with CCC was
diagnosed with CCBT and the diagnosis between CCBT and CCC was not
difficult. Also, immunochemical analysis confirmed 2 cases with
CCBT were immunohistochemically positive for HNF-1β, and negative
for p53 and WT-1. Therefore, our cases were not complicated with
other OBT and was diagnosed with pure-type CCBT.
A review of literature of CCBT with and without
fibromatous component including our study were demonstrated in
Table II (8,16-26).
There were 81 cases with CCBT; 77 cases (95.1%) with fibromatous
component and 4 cases (4.9%) without fibromatous component. About
cases with CCBT with fibromatous component, age ranged from 30 to
86 years, all cases were diagnosed with FIGO stage I, and 4 cases
(5.2%) had tumors in bilateral ovaries. 2 cases (2.6%) suffered
from recurrence, but no case died of the disease. On contrast,
about cases with CCBT without fibromatous component, age ranged
from 42 to 76 years, all cases were diagnosed with FIGO stage I,
and no cases had tumors in bilateral ovaries. No case recurred or
died of disease. Comparing these two groups, the presence or
absence of fibromatous component in CCBT might not be related to
their clinicopathological features.
 | Table IILiterature review about clear cell
borderline tumor without and with fibromatous component. |
Table II
Literature review about clear cell
borderline tumor without and with fibromatous component.
| A, Clear cell
borderline with fibromatous component |
|---|
| Authors year | Total no of
cases | Mean age (Range) | FIGO Stage | No of cases with
tumors in bilateral ovaries | Surgical form | No. of
recurrence | Mean follow-up period
(Range) | Refs. |
|---|
| Kao and
Norris, 1979 | 3 | 59 (54-62) | Stage I: 3 cases | 1 | 3 cases, TAH+BSO | 0 | 32 months
(28-36) | (16) |
| Roth et al,
1984 | 4 | 67 (66-68) 1 case:
LFU | Stage I: 4 cases | 0 | 2 cases, TAH+BSO; 2
cases, LFU | 1 | 60 months (12-72) 1
case: LFU | (17) |
| Bell and
Scully, 1985 | 11 | 61 (30-86) | Stage I: 11
cases | 1 | 8 cases, TAH+BSO; 2
cases, BSO; 1 case, USO | 1 | 70 months
(22-156) | (18) |
| Katsube et al,
1989 | 1 | 59 | Stage I | 0 | RSO | 0 | 78 months | (19) |
| Liu JLet al,
2010 | 1 | 52 | Stage I | 0 |
LSO+omentectomy+lymphadenectomy | 0 | 10 months | (20) |
| Momotani et
al, 2011 | 1 | 79 | Stage I | 0 | TAH+BSO | 0 | 8 months | (21) |
| Zhao et al,
2011 | 41 | 59 (54-62) | Stage I: 41
cases | 1 | LFU | 0 | LFU | (22) |
| Cakir et al,
2012 | 1 | 53 | Stage I | 0 | TAH+BSO | 0 | 6 months | (23) |
| Vasilakaki et
al, 2012 | 1 | 34 | Stage I | 0 | RSO | 0 | 48 months | (24) |
| Uzan et al,
2012 | 12 | 68 (36-83) | Stage I: 12
cases | 1 | 8 cases, TAH+BSO; 2
cases, USO; 1 case, BSO; 1 case, LFU | 0 | 28 months
(2-129) | (25) |
| Kleebkaow et
al, 2017 | 1 | 58 | Stage I | 0 | TAH+BSO | 0 | 36 months | (26) |
| B, Clear cell
borderline without fibromatous component |
| Authors year | Total no of
cases | Mean age
(Range) | FIGO Stage | No of cases with
tumors in bilateral ovaries | Surgical form | No. of
recurrence | Mean follow-up
period (Range) | Refs. |
| Suzuki et
al, 2006 | 2 | 52, 76 | Stage I: 2
cases | 0 | 2 cases,
TAH+BSO | 0 | 16 and 24
months | (8) |
| The present
study | 2 | 42, 43 | Stage I: 2
cases | 0 | 1 case, TAH+BSO
+omentectomy; 1 case, LSO+omentectomy+pelvic lymphadenectomy | 0 | 20 and 37
months | |
Past report proposed the pathogenesis of CCBT and
CCC with 2 developing pathways; endometriotic cystic pathway and
adenofibromatous pathway (22). In
the endometriotic cystic pathway, endometriosis formed an
endometriotic cyst, through atypical endometriosis, developed CCC.
In the adenofibromatous pathway, endometriosis accompanied by a
fibromatous reaction, and subsequently progressed to CCBT and then
to CCC. Thus, CCBT was regarded as deriving from only clear cell
adenofibroma in the adenofibromatous pathway. In previous reports,
95.1% of cases with CCBT coexisted fibromatous component (16-26).
However, in case 1 in our case, CCBT was considered to develop from
endometriosis because CCBT was adjacent to atypical endometriosis
and endometriosis. This finding might indicate the new development
of CCBT from endometriosis. Also, 2 cases reported by Suzuki et
al. and case 2 in our study, CCBT was without endometriosis and
fibromatous component (8). This
finding might suggest CCBT derived from neither endometriosis or
adenofibroma.
The limitations of this study included a small
sample size at a single-institution, and retrospective analysis.
Further studies with a large sample size are needed to confirm
clinical significance of CCBT without fibromatous component. In
addition, our study could not discover the origin except
adenofibroma and endometriosis. This problem is the future
challenge.
In conclusion, through pathological review, 2 cases
of CCBT without fibromatous component was reported. CCBT with
fibromatous component was rare and needed to be examined by future
study.
Acknowledgements
The authors would like to thank Mrs. Ayako Suzuki
for collecting samples.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
TH designed the study, performed pathological and
immunochemical analysis, collected the data and prepared and
revised the manuscript. MM designed the study, performed
pathological and immunochemical analysis, and prepared and revised
the manuscript. HI, HM, TS, SK, HI, and RS collected the data. HT
performed pathological and immunochemical analysis. TH and MM
confirmed the authenticity of the raw data. MT designed the study,
and prepared and revised the manuscript. All authors read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
All procedures performed in studies involving human
participants were in accordance with the ethical standards of the
National Defense Medical Collage hospital and with the 1964
Helsinki declaration and its later amendments or comparable ethical
standards. For this type of study formal consent is not
required.
Patient consent for publication
Not applicable.
Competing interests
All authors declare that they have no competing
interests.
References
|
1
|
Taylor HC Jr: Malignant and semimalignant
tumors of the ovary. Surg Gynecol. 48:204–230. 1929.
|
|
2
|
Classification and staging of malignant
tumours in the female pelvis. Acta Obstet Gynecol Scand 50: 1-7,
1971.
|
|
3
|
Serov SS, Scully RE and Sobin LH (eds);
World Health Organization: Histological Typing of Ovarian Tumors.
Springer, Berlin, Heidelberg, New York, NY, Geneva, 1973.
|
|
4
|
Silverberg SG, Bell DA, Kurman RJ, Seidman
JD, Prat J, Ronnett BM, Copeland L, Silva E, Gorstein F and Young
RH: Borderline ovarian tumors: Key points and workshop summary. Hum
Pathol. 35:910–917. 2004.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Trimble CL, Kosary C and Trimble EL:
Long-term survival and patterns of care in women with ovarian
tumors of low malignant potential. Gynecol Oncol. 86:34–37.
2002.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Lokuhetty D, White VA and Cree IA (eds):
WHO Classification of Tumours of Female Reproductive Organs. Lyon,
IARC, Lyon, 2020.
|
|
7
|
Seidman JD, Soslow RA, Vang R, Berman JJ,
Stoler MH, Sherman ME, Oliva E, Kajdacsy-Balla A, Berman DM and
Copeland LJ: Borderline ovarian tumors: Diverse contemporary
viewpoints on terminology and diagnostic criteria with illustrative
images. Hum Pathol. 35:918–933. 2004.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Suzuki A, Shiozawa T, Mori A, Kimura K and
Konishi I: Cystic clear cell tumor of borderline malignancy of the
ovary lacking fibromatous components: Report of two cases and a
possible new histological subtype. Gynecol Oncol. 101:540–544.
2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Pereira A, Pérez-Medina T, Magrina JF,
Magtibay PM, Rodríguez-Tapia A, Peregrin I, Mendizabal E and
Ortiz-Quintana L: International Federation of Gynecology and
Obstetrics staging classification for cancer of the ovary,
fallopian tube, and peritoneum: Estimation of survival in patients
with node-positive epithelial ovarian cancer. Int J Gynecol Cancer.
25:49–54. 2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Seidman JD, Soslow RA, Vang R, Berman JJ,
Stoler MH, Sherman ME, Oliva E, Kajdacsy-Balla A, Berman DM and
Copeland LJ: Borderline ovarian tumors: Diverse contemporary
viewpoints on terminology and diagnostic criteria with illustrative
images. Hum Pathol. 35:918–933. 2004.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Hauptmann S, Friedrich K, Redline R and
Avril S: Ovarian borderline tumors in the 2014 WHO classification:
Evolving concepts and diagnostic criteria. Virchows Arch.
470:125–142. 2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Ohishi Y, Oda Y, Kurihara S, Kaku T,
Yasunaga M, Nishimura I, Okuma E, Kobayashi H, Wake N and
Tsuneyoshi M: Hobnail-like cells in serous borderline tumor do not
represent concomitant incipient clear cell neoplasms. Hum Pathol.
40:1168–1175. 2009.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ohishi Y, Kurihara S, Aman M, Takeuchi T,
Imamura H, Kaku T, Kobayashi H, Wake N and Oda Y: ‘Piling up’ clear
cells in müllerian-type mucinous and mixed cell-type borderline
tumor do not represent concomitant clear cell neoplasms. Hum
Pathol. 43:1618–1626. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Köbel M, Kalloger SE, Boyd N, McKinney S,
Mehl E, Palmer C, Leung S, Bowen NJ, Ionescu DN, Rajput A, et al:
Ovarian carcinoma subtypes are different diseases: Implications for
biomarker studies. PLoS Med. 5(e232)2008.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kato N, Sasou S and Motoyama T: Expression
of hepatocyte nuclear factor-1beta (HNF-1beta) in clear cell tumors
and endometriosis of the ovary. Mod Pathol. 19:83–89.
2006.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kao GF and Norris HJ: Unusual
cytadenofibromas: Endometrioid, mucinous, and clear cell types.
Obstet Gynecol. 54:729–736. 1979.PubMed/NCBI
|
|
17
|
Roth LM, Langley FA, Fox H, Wheeler JE and
Czernobilsky B: Ovarian clear cell adenofibromatous tumors. Benign,
of low malignant potential, and associated with invasive clear cell
carcinoma. Cancer. 53:1156–1163. 1984.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bell DA and Scully RE: Benign and
borderline clear cell adenofibromas of the ovary. Cancer.
56:2922–2931. 1985.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Katsube Y, Fujiwara H, Tanioka Y, Imajo M
and Fujiwara A: Ovarian clear cell adenofibroma of borderline
malignancy. A case report. Hiroshima J Med Sci. 38:87–90.
1989.PubMed/NCBI
|
|
20
|
Liu JL, Chu PY, Yeh KT and Huang RH:
Borderline clear cell adenofibroma with extensive hemorrhagic
necrosis. Hematol Oncol Stem Ther. 3:158–160. 2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Momotani K, Tnaka T, Iwai E, Kanda T,
Munakata S and Ohmichi M: Ovarian clear cell adenofibromatous tumor
of borderline malignancy associated with high levels of
carbohydrate antigen 9-9. J Obstet Gynaecol Res. 37:472–477.
2011.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zhao C, Wu LS and Barner R: Pathogenesis
of ovarian clear cell adenofibroma, atypical proliferative
(borderline) tumor, and carcinoma: Clinicopathologic features of
tumors with endometriosis or adenofibromatous components support
two related pathways of tumor development. J Cancer. 2:94–103.
2011.PubMed/NCBI View
Article : Google Scholar
|
|
23
|
Cakir E, Aydin E, Durmus NI, Samdanci E,
Sahin N and Nurkabul Z: Primary ovarian clear cell adenofibroma of
borderline malignancy. Oman Med J. 27(e031)2012.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Vasilakaki T, Skafida E, Arkoumani E,
Grammatoglou X, Firfiris N and Manoloudaki K: Borderline clear cell
adenofibroma of the ovary associated with ovarian endometriosis: A
case report. Eur J Gynarcol Oncol. 33:230–232. 2012.PubMed/NCBI
|
|
25
|
Uzan C, Dufeu-Lefebvre M, Fauvet R, Gouy
S, Duvillard P, Darai E and Morice P: Management and prognosis of
clear cell borderline ovarian tumor. Int J Gynecol Cancer.
22:993–999. 2012.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Kleebkaow P, Aue-Aungkul A,
Temtanakitpaisan A and Kietpeerakool C: Borderline clear cell
adenofibroma of the ovary. Case Rep Pathol.
2017(3860107)2017.PubMed/NCBI View Article : Google Scholar
|