Introduction
Cancer immunotherapy has been actively explored in
the treatment of various malignant neoplasms of the
gastrointestinal tract (1,2). Recently, nanotechnology-mediated
delivery approaches have attracted considerable attention in
colorectal cancer (CRC) immunotherapy. Nanoparticle-based
immunotherapy exhibits higher specificity and efficacy in
comparison with conventional immunotherapy (3). Those immune-related transmitters can
locally deliver immune components to the antigen presenting cells
with high efficiency and modulate the tumor immune
microenvironment. In addition, they act as an adjuvant that can
boost immune reactions to antigens (4). CRC is a heterogeneous group of
malignant lesions with different types of immune responses
(5). Previous studies have focused
on the morphological assessment and molecular changes that occur
during CRC, which includes mutational status and chromosomal
instability, somatic copy number variation, immune infiltration and
metabolic regulation. Based on this evidence CRC can be divided
into five groups of tumors as follows: Microsatellite
instability-associated, immune-associated, canonical, metabolic and
mesenchymal (6). The proposed
division of cancerous tumors is closely associated with tumor stage
and survival time. Recent studies have shown that patients with a
high status of microsatellite instability respond better to
immunotherapy (7). Therefore,
systemic immune response is associated with infiltration of immune
cells in CRC tumors, which appear to be separated from the tumor
microenvironment (8). The immune
cells infiltrating CRC tumors are macrophages, dendritic cells,
neutrophils and lymphocytes. Immunotherapy is focused on the
ability of lymphocytes to infiltrate the cancerous tissue. It has
been shown that lymphocytes infiltrates exhibit considerable
variation with regard to their biological characteristics in the
different parts of the tumor (9).
Laghi et al (9) demonstrated
that the infiltrate of T cells in the invasive margin of CRC tumors
could improve disease prognosis compared with cells examined at the
center of the tumor. In addition, Bindea et al (10) indicated that the amount of T cells
present in the tumor was decreased over time, while the number of B
cells and innate immune cells was increased. Moreover, local immune
response in the tumor tissue was closely associated with systemic
exposure to circulating cancer cells in the bloodstream. Therefore,
the analysis of hematological parameters appears to be an important
indicator of immune response to cancer. Currently, several studies
using hematological factors, such as absolute lymphocyte count
(ALC), absolute neutrophil count, neutrophil-to-lymphocyte ratio
(NLR) and platelet-to-lymphocyte ratio (PLR) have been conducted
(11-13).
Tanio et al (11) indicated
that the combined absolute number of lymphocytes, monocytes and
neutrophils in the preoperative whole blood can be a useful
prognostic factor in CRC patients (11). Kozak et al (12) proposed that the elevated NLR
independently predicts worse overall survival in patients with CRC.
In addition, Yang et al (13) demonstrated that an increased PLR
value was associated with poor overall survival. In light of these
reports, the present study aimed to investigate the combination of
the ALC, tumor-infiltrating lymphocyte (TIL) percentage and tumor
progression status in patients with CRC who underwent surgery.
Patients and methods
Patients and tissue specimens
A total of 160 patients who were diagnosed with CRC
were retrospectively reviewed (96 male, 64 female; median age, 72
years). In the present study, the paraffin-embedded tissues were
obtained from the Comprehensive Cancer Centre between April 2014
and December 2016. The patients who were diagnosed with CRC
underwent surgery in the Division of Surgical Oncology. All of the
tissue samples were processed by an identical dissection protocol.
The tissues obtained from surgery were fixed in 4% buffered
formalin and subsequently small sections of tissues were embedded
in paraffin. The sections (4 µm-thick) were cut from paraffin
blocks and stained with haematoxylin and eosin (H&E). The
slides were deparaffinised in an oven at 60˚C for 5 min.
Subsequently, the slides were rehydrated in xylene (three washes)
and graded ethanol (100, 95, 85 and 75%). Histological assessment
was performed by two pathologists, which were blinded to the study
protocol. The following parameters were recorded from each patient:
Type of tumour growth, Tumour-Node-Metastasis (TNM) stage, tumour
size, histological type, percentage of mucinous components and
grade of malignancy. The clinical stage of CRC was evaluated
according to the TNM classification (14). Venous, perineural and lymphatic
invasion (the number of resected and invaded lymph nodes, the
presence of micro- and macro-metastases, invasion of the pouch
lymph node, presence of distant metastases and the size of
metastases) were also assessed. The deposits of cancer (their
number and size) were also included (15).
The patients enrolled in the present study presented
similar symptoms. The most common symptoms were abdominal pain,
vomiting, rectal bleeding, constipation or diarrhoea. The medical
history demonstrated that some patients suffered from hypertension,
diabetes (type II), coronary heart disease or osteoarthritis.
However, none of the patients had received anti-inflammatory or
immunosuppressive therapy. All of the patients underwent routine
diagnostic tests prior to surgery (blood tests, electrocardiography
and spirometry). The diagnostic examinations, such as a chest
computerized tomography and chest X-ray were utilized to identify
CRC distal metastasis. The patients with neoplasm in the rectum
received preoperative therapy (n=53): Radiotherapy (n=39),
chemotherapy (n=7) and radio-chemotherapy (n=7). The radiation dose
used was 25 Gy, in fractions of 5 Gy during one week in the pelvic
area. According to the Response Evaluation Criteria in Tumours
(16), the response to preoperative
therapy was evaluated. A total of 26 patients were recruited with a
stable disease and 27 patients exhibited partial response.
The inclusion criteria were as follows: i)
Pathologically confirmed CRC; ii) treatment with radical resection;
and iii) lack of anti-inflammatory therapy. The exclusion criteria
were the following: i) Incomplete clinicopathological and follow-up
data; and ii) presence of haematological disorders, such as
anaemia.
The present study was performed in conformity with
the Declaration of Helsinki for Human Experimentation and the
protocol was approved by the Bioethics Committee of the Medical
University of Bialystok (no. R-I-002/353/2016).
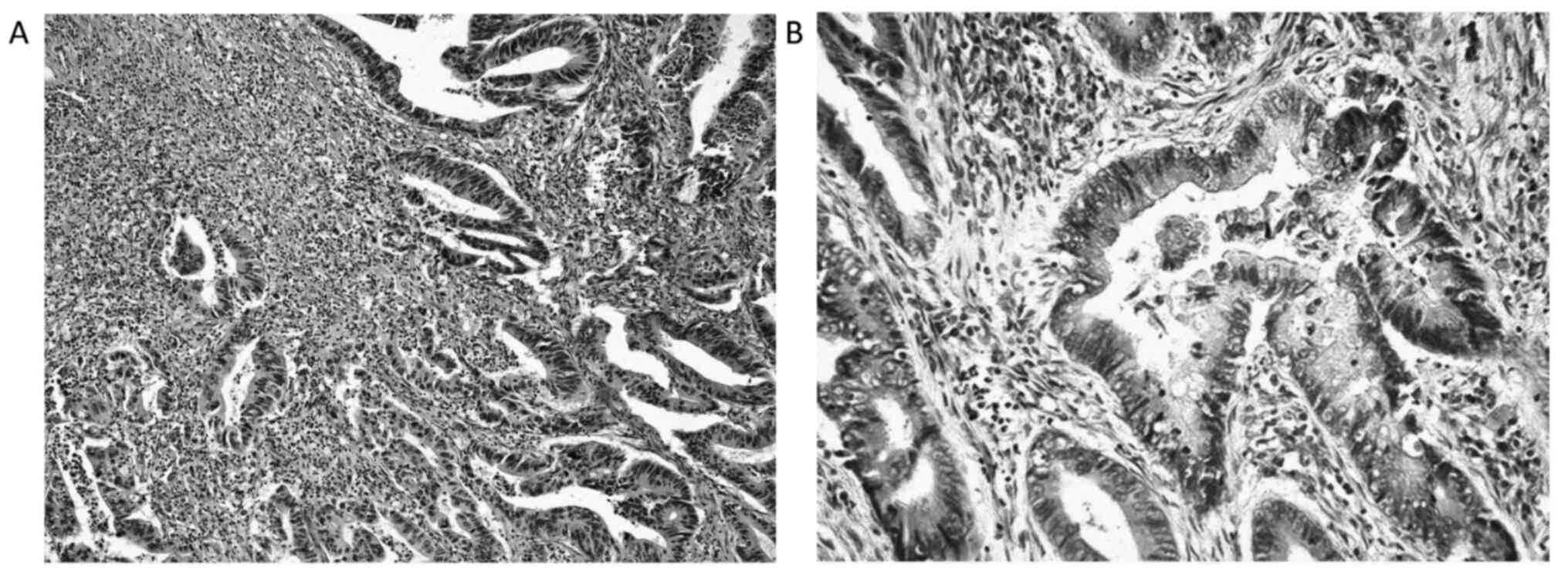
Assessment of the percentage of TILs
in CRC tissues
The analysis of TILs was described in our previous
study (17). Briefly, tissue
material obtained from routine histopathological diagnosis was
stained with H&E and used to assess the TIL populations located
in the tumor stroma at the invasive front and centre of the tumour
by light microscopy (magnification, x200-400; Leica DM6 B; Leica
Microsystems, Inc.). The analysis was evaluated by two independent
pathologists who were blinded to the clinical information of the
study. TILs in the stroma were identified according to
recommendations by the International TILs Working Group,
2014(18). They were determined as
a percentage of mononuclear inflammatory cells in the total
intratumoral or stromal area as counted in 5 high power fields
(HPF; magnification, x200-400), at the invasive front and in the
centre of the tumour, with the exception of tumour areas with crush
artefacts, necrosis or regressive hyalinization. The invasive front
of the tumour was defined as the percentage of most progressed
cancer cells on the advanced edge of the tumour. For statistical
analysis, the percentage of stromal TILs was scored as follows: 1,
weak (0-10% stromal TILs); 2, moderate (20-40% stromal TILs); and
3, strong (50-90% stromal TILs). The study population was divided
into two groups based on the stromal TIL score as follows: 1, score
1; and 2, score 2 or 3. Representative images of TILs are shown in
Fig. 1.
Blood sample examination
Blood samples were obtained within 3 days prior to
and 7 days following surgical treatment. Venous blood samples were
also obtained from 42 healthy control subjects (female-21, male-21;
mean age 45 years old; min-max 25-65 years old). The differential
white blood cell count was counted using an XN-1000 automated
haematology analyzer (Sysmex America, Inc.). Normal total
lymphocyte count ranges were between 0.90 and 4.00x10³ cells/µl
(mean 2.25x10³ cells/µl). The total lymphocyte count in CRC
patients ranged from 0.47 to 4.06x10³ cells/µl (mean 1.05x10³
cells/µl).
Combination of TIL percentage and
ALC
The percentage of TILs was examined in the CRC
tissue at the invasive front and centre of the primary tumour mass
in combination with the ALC and in whole blood samples obtained
prior to and following surgery from patients with CRC. The
classification of the immune response system included: i) TILs at
the invasive front and ALC prior to surgery; ii) TILs in the centre
of the tumour mass and ALC prior to surgery; iii) TILs at the
invasive front and ALC following surgery, iv) TILs in the centre of
the tumour mass and ALC following surgery. All groups included 4
subgroups: i) High TILs (moderate or strong stromal TILs) and high
ALC (≥4.0x10³ cells/µl); ii) high TILs (moderate or strong stromal
TILs) and low ALC (≤0.9x10³ cells/µl); iii) low TILs (weak stromal
TILs) and high ALC (≥4.0x10³ cells/µl); iv) low TILs (weak stromal
TILs) and low ALC (≤0.9x10³ cells/µl).
In the present study, the combination of TIL
percentage and ALC was also examined with regard to the tumor
progression status (TILs/ALC/tumour progression status). The tumour
progression status was determined on the basis of 3 parameters as
follows: i) Lymphatic vessel invasion; ii) lymph node involvement
and iii) distant metastasis. Based on the tumour progression status
the study population was divided into 3 groups as follows: i)
patients with local disease; ii) patients with lymphatic vessel
invasion and positive lymph node status without distant metastasis;
iii) patients that exhibited cancer cell invasion to all the
aforementioned structures. The local and systemic response of
lymphocytes prior to and following surgical treatment was
investigated. Taken together, the patients were divided into 4
groups as follows: i) Negative status of tumour progression with
high percentage of TILs at the invasive front and in the centre of
the primary tumour mass and high ALC, ii) negative status of tumour
progression with high percentage of TILs as the invasive front and
in the centre of the primary tumour mass and low ALC, iii) positive
status of tumour progression (group 2 or 3) with high percentage of
TILs at the invasive front and in the centre of the primary tumour
mass and high ALC and iv) positive status of tumour progression
(group 2 or 3) with high percentage of TILs at the invasive front
and in the centre of the primary tumour mass and high ALC.
Statistical analysis
All calculations were performed using the
statistical software STATISTICA 13.0 (StatSoft, Inc.). The
enumeration data of examined parameters were analysed using the
χ2 test. The comparisons among multiple groups (examined
parameters and clinicopathological features) were analysed using
one-way ANOVA. Fisher's Least Significant Difference post hoc test
was used to analyze multiple comparisons of TILs, ALC, progression
status and histopathological factors following the ANOVA test (data
not shown). Tukey's test was used to compare more than three
groups. P<0.05 was considered to indicate a statistically
significant difference.
Results
Patient characteristics and
distribution of the combination index including TIL percentage and
ALC in CRC patients
The median age was 67.5 years (range, 32 to 88
years) and 60% of the patients were male. The rectum was the most
common tumour location (51.25%). The pathological staging was the
following: Stage I in 1.88% of patients, stage II in 38.75% of
patients, stage III in 56.88% of patients and stage IV in 2.5% of
patients. The patients developed adenocarcinoma (81.25%) more
frequently than mucinous CRC (18.75%). The distribution of TILs was
evaluated based on several factors, such as invasion of cancer
cells, distant metastasis or status of tumour progression. The data
indicated that the distribution of the combination index including
TIL percentage and ALC differed significantly among the groups
(Table I). However, statistical
significance was not obtained for the distribution of the
TILs/ALC/progression tumour status (Table II).
 | Table IDistribution of the combined
parameters of TILs and ALC. |
Table I
Distribution of the combined
parameters of TILs and ALC.
| | Groups | |
|---|
| Parameter | High TILs/high ALC,
n | High TILs/low ALC,
n | Low TILs/high ALC,
n | Low TILs/low ALC,
n | P-value |
|---|
| TILs at the invasive
front and ALC before surgery | 39 | 116 | 2 | 9 | <0.001 |
| TILs at the invasive
front and ALC after surgery | 75 | 72 | 4 | 1 | |
| TILs in the main mass
of primary tumor and ALC before surgery | 39 | 121 | 2 | 7 | <0.001 |
| TILs in the main mass
of primary tumor and ALC after surgery | 74 | 69 | 3 | 4 | |
 | Table IIDistribution of the TILs/ALC/tumor
progression status. |
Table II
Distribution of the TILs/ALC/tumor
progression status.
| | Groups | |
|---|
| Parameter | 1, n | 2, n | 3, n | 4, n | P-value |
|---|
| TILs at the invasive
front, ALC and tumor progression status | 12 | 48 | 28 | 72 | 0.164 |
| TILs in the main mass
of the primary tumor, ALC and tumor progression status | 29 | 30 | 47 | 54 | |
Correlation of the combination index
including TIL percentage and ALC with the clinicopathological
parameters of CRC patients
The percentage of TILs and the ALC prior to and
following surgery were significantly associated with the tumour
growth in the main mass (Tables
III and IV, P=0.001, P=0.007).
A significant correlation was observed in the percentage of TILs
with the tumour size (P=0.031), pT stage (P=0.049) and necrosis
percentage (P=0.037) following surgery. The histological type
correlated with the combination index prior to surgery (P=0.046).
Lymph node pouch invasion was associated with the percentage of
TILs at the invasive front of the tumour and with ALC prior to and
following surgery (P=0.006, P=0.037). Moreover, the percentage of
TILs exanimated at the invasive front and centre of the tumour, and
the ALC prior to and following surgery correlated with the
treatment status (P=0.032, P=0.018, P≤0.001, P≤0.001). The patients
with neoadjuvant therapy (status treatment group 1 and 2) exhibited
better local and systemic chronic inflammatory response.
Furthermore, no significant difference was observed in the
remaining evaluated parameters (Tables III and IV).
 | Table IIIAssociations between combined
parameters of TILs and absolute lymphocyte count obtained before
surgery, and clinicopathological features of patients with CRC
(n=160). |
Table III
Associations between combined
parameters of TILs and absolute lymphocyte count obtained before
surgery, and clinicopathological features of patients with CRC
(n=160).
| |
TILs and
absolute lymphocyte count before surgery |
|---|
| | Invasive front | Main mass |
|---|
| Parameter | No. | 1, n | 2, n | 3, n | 4, n | P-value | 1, n | 2, n | 3, n | 4, n | P-value |
|---|
| Age, years | | | | | | | | | | | |
|
<60 | 40 | 5 | 30 | 0 | 5 | 0.420 | 5 | 32 | 0 | 3 | 0.420 |
|
≥60 | 120 | 29 | 69 | 2 | 20 | | 29 | 70 | 2 | 19 | |
| Sex | | | | | | | | | | | |
|
Female | 64 | 12 | 41 | 0 | 11 | 0.696 | 12 | 41 | 0 | 11 | 0.696 |
|
Male | 96 | 22 | 58 | 2 | 14 | | 22 | 61 | 2 | 11 | |
| Localization | | | | | | | | | | | |
|
Right-side | 20 | 8 | 11 | 1 | 0 | 0.407 | 4 | 12 | 1 | 3 | 0.561 |
|
Transverse | 14 | 4 | 4 | 0 | 6 | | 1 | 4 | 0 | 9 | |
|
Left-side | 15 | 1 | 5 | 0 | 9 | | 1 | 5 | 0 | 10 | |
|
Sigmoid | 29 | 1 | 11 | 0 | 7 | | 8 | 8 | 0 | 13 | |
|
Rectum | 82 | 17 | 54 | 1 | 10 | | 16 | 54 | 0 | 12 | |
| Tumor growth | | | | | | | | | | | |
|
Expanding | 133 | 28 | 82 | 2 | 21 | 0.109 | 28 | 82 | 2 | 21 | 0.001 |
|
Infiltrate | 27 | 6 | 17 | 0 | 3 | | 6 | 20 | 0 | 1 | |
| Tumor size, cm | | | | | | | | | | | |
|
<2.5 | 27 | 6 | 15 | 1 | 5 | 0.412 | 6 | 17 | 1 | 3 | 0.181 |
|
2.5-5.0 | 106 | 26 | 63 | 0 | 17 | | 25 | 64 | 1 | 16 | |
|
>5.0 | 27 | 2 | 21 | 1 | 3 | | 3 | 21 | 0 | 3 | |
| Histological
type | | | | | | | | | | | |
|
Muc | 30 | 4 | 25 | 0 | 1 | 0.322 | 8 | 22 | 0 | 0 | 0.046 |
|
Adca | 130 | 30 | 74 | 2 | 24 | | 30 | 79 | 2 | 19 | |
| Mucinous component,
% | | | | | | | | | | | |
|
10-30 | 15 | 0 | 10 | 0 | 5 | 0.392 | 0 | 10 | 0 | 5 | 0.148 |
|
31-50 | 15 | 4 | 11 | 0 | 0 | | 4 | 11 | 0 | 0 | |
| TNM stage | | | | | | | | | | | |
|
1+2 | 73 | 10 | 49 | 1 | 13 | 0.518 | 11 | 53 | 0 | 9 | 0.417 |
|
3+4 | 87 | 24 | 50 | 1 | 12 | | 23 | 49 | 2 | 13 | |
| Grade of
malignancies | | | | | | | | | | | |
|
2 | 148 | 32 | 93 | 2 | 21 | 0.812 | 32 | 96 | 2 | 18 | 0.482 |
|
3 | 12 | 2 | 6 | 0 | 4 | | 2 | 6 | 0 | 4 | |
| pT stage | | | | | | | | | | | |
|
1+2 | 65 | 13 | 39 | 1 | 12 | 0.159 | 13 | 41 | 1 | 10 | 0.154 |
|
3+4 | 95 | 21 | 60 | 1 | 13 | | 21 | 61 | 1 | 12 | |
| Venous
invasion | | | | | | | | | | | |
|
Absent | 113 | 22 | 70 | 1 | 20 | 0.642 | 23 | 73 | 0 | 17 | 0.083 |
|
Present | 46 | 12 | 29 | 1 | 4 | | 11 | 29 | 2 | 4 | |
| Lymphatic
invasion | | | | | | | | | | | |
|
Absent | 121 | 24 | 75 | 1 | 21 | 0.952 | 24 | 78 | 0 | 19 | 0.961 |
|
Present | 38 | 10 | 24 | 1 | 3 | | 10 | 24 | 2 | 2 | |
| Perineural
invasion | | | | | | | | | | | |
|
Absent | 143 | 30 | 89 | 2 | 22 | 0.415 | 30 | 92 | 2 | 19 | 0.478 |
|
Present | 17 | 4 | 10 | 0 | 3 | | 4 | 10 | 0 | 3 | |
| Lymph node
metastasis | | | | | | | | | | | |
|
Absent | 81 | 16 | 60 | 1 | 4 | 0.350 | 16 | 62 | 1 | 2 | |
|
Present | 79 | 18 | 39 | 1 | 21 | | 18 | 40 | 1 | 20 | 0.405 |
| Lymph node pouch
invasion | | | | | | | | | | | |
|
Absent | 41 | 17 | 9 | 2 | 13 | 0.006 | 10 | 5 | 13 | 13 | |
|
Present | 39 | 17 | 3 | 0 | 19 | | 14 | 2 | 6 | 17 | 0.775 |
| Distant
metastasis | | | | | | | | | | | |
|
Absent | 143 | 28 | 91 | 2 | 22 | 0.916 | 29 | 95 | 1 | 18 | 0.873 |
|
Present | 17 | 6 | 8 | 0 | 3 | | 5 | 7 | 1 | 4 | |
| Tumor deposits | | | | | | | | | | | |
|
Absent | 133 | 24 | 84 | 2 | 23 | 0.565 | 25 | 87 | 1 | 20 | 0.474 |
|
Present | 27 | 10 | 14 | 0 | 3 | | 9 | 14 | 1 | 2 | |
| Tumor budding | | | | | | | | | | | |
|
Absent | 94 | 20 | 57 | 1 | 16 | 0.436 | 20 | 59 | 1 | 14 | 0.945 |
|
Present | 66 | 14 | 42 | 1 | 9 | | 14 | 43 | 1 | 8 | |
| Necrosis | | | | | | | | | | | |
|
Absent | 45 | 6 | 29 | 0 | 10 | 0.632 | 5 | 31 | 1 | 8 | 0.538 |
|
Focal | 61 | 13 | 41 | 0 | 7 | | 13 | 39 | 0 | 9 | |
|
Moderate | 36 | 9 | 20 | 2 | 5 | | 11 | 24 | 1 | 0 | |
|
Extensive | 18 | 6 | 9 | 0 | 3 | | 5 | 8 | 0 | 5 | |
| Fibrosis | | | | | | | | | | | |
|
Absent | 11 | 6 | 5 | 0 | 0 | 0.603 | 6 | 4 | 1 | 0 | 0.917 |
|
Focal | 72 | 14 | 48 | 1 | 9 | | 13 | 49 | 0 | 10 | |
|
Moderate | 43 | 7 | 26 | 1 | 9 | | 8 | 28 | 1 | 6 | |
|
Extensive | 34 | 7 | 20 | 0 | 7 | | 7 | 20 | 0 | 7 | |
| Treatment
statusa | | | | | | | | | | | |
|
1 | 36 | 14 | 11 | 5 | 6 | 0.032 | 14 | 20 | 1 | 1 | 0.018 |
|
2 | 22 | 5 | 14 | 1 | 2 | | 11 | 11 | 0 | 0 | |
|
3 | 71 | 36 | 22 | 12 | 1 | | 7 | 59 | 1 | 5 | |
|
4 | 31 | 4 | 12 | 2 | 13 | | 7 | 21 | 0 | 3 | |
 | Table IVCorrelations between combined
parameters of tumor-infiltrating lymphocytes and absolute
lymphocyte count obtained after surgery, and clinicopathological
features of patients with colorectal cancer (n=160). |
Table IV
Correlations between combined
parameters of tumor-infiltrating lymphocytes and absolute
lymphocyte count obtained after surgery, and clinicopathological
features of patients with colorectal cancer (n=160).
| |
TILs and
absolute lymphocyte count |
|---|
| |
After
surgery |
|---|
| | Invasive front | Main mass |
|---|
| Parameter | No. | 1, n | 2, n | 3, n | 4, n | P-value | 1, n | 2, n | 3, n | 4, n | P-value |
|---|
| Age, years | | | | | | | | | | | |
|
<60 | 40 | 22 | 12 | 0 | 6 | 0.245 | 21 | 13 | 1 | 5 | 0.545 |
|
≥60 | 120 | 47 | 47 | 1 | 25 | | 47 | 44 | 2 | 27 | |
| Sex | | | | | | | | | | | |
|
Female | 64 | 30 | 20 | 0 | 14 | 0.984 | 31 | 20 | 0 | 13 | 0.719 |
|
Male | 96 | 39 | 39 | 1 | 17 | | 37 | 37 | 3 | 19 | |
| Localization | | | | | | | | | | | |
|
Right-side | 20 | 8 | 7 | 0 | 5 | 0.320 | 10 | 6 | 1 | 3 | 0.438 |
|
Transverse | 14 | 2 | 2 | 1 | 9 | | 2 | 2 | 0 | 10 | |
|
Left-side | 15 | 3 | 3 | 0 | 9 | | 3 | 2 | 0 | 10 | |
|
Sigmoid | 29 | 12 | 7 | 0 | 10 | | 2 | 7 | 0 | 20 | |
|
Rectum | 82 | 37 | 30 | 0 | 20 | | 34 | 28 | 0 | 20 | |
| Tumor growth | | | | | | | | | | | |
|
Expanding | 133 | 54 | 51 | 1 | 27 | 0.489 | 53 | 49 | 3 | 28 | 0.007 |
|
Infiltrate | 27 | 15 | 8 | 0 | 4 | | 15 | 8 | 0 | 4 | |
| Tumor size, cm | | | | | | | | | | | |
|
<2.5 | 27 | 9 | 12 | 0 | 6 | 0.426 | 8 | 14 | 1 | 4 | 0.031 |
|
2.5-5.0 | 106 | 48 | 38 | 0 | 19 | | 48 | 35 | 2 | 21 | |
|
>5.0 | 27 | 12 | 9 | 1 | 6 | | 12 | 8 | 0 | 7 | |
| Histological
type | | | | | | | | | | | |
|
Muc | 30 | 16 | 12 | 1 | 1 | 0.079 | 16 | 7 | 0 | 7 | 0.056 |
|
Adca | 130 | 53 | 47 | 0 | 30 | | 52 | 50 | 3 | 25 | |
| Mucinous component,
% | | | | | | | | | | | |
|
10-30 | 15 | 5 | 5 | 1 | 4 | 0.148 | 5 | 4 | 0 | 6 | 0.081 |
|
31-50 | 15 | 11 | 2 | 0 | 2 | | 11 | 3 | 0 | 2 | |
| TNM stage | | | | | | | | | | | |
|
1+2 | 73 | 33 | 24 | 0 | 16 | 0.913 | 33 | 27 | 1 | 12 | 0.651 |
|
3+4 | 87 | 36 | 35 | 1 | 3 | | 35 | 30 | 2 | 20 | |
| Grade of
malignancies | | | | | | | | | | | |
|
2 | 148 | 66 | 54 | 1 | 27 | 0.618 | 65 | 52 | 3 | 28 | 0.806 |
|
3 | 12 | 3 | 5 | 0 | 4 | | 3 | 5 | 0 | 4 | |
| pT stage | | | | | | | | | | | |
|
1+2 | 65 | 28 | 22 | 0 | 15 | 0.124 | 27 | 24 | 2 | 12 | 0.049 |
|
3+4 | 95 | 41 | 37 | 1 | 16 | | 41 | 33 | 1 | 20 | |
| Venous
invasion | | | | | | | | | | | |
|
Absent | 113 | 48 | 40 | 0 | 25 | 0.861 | 48 | 40 | 3 | 22 | 0.300 |
|
Present | 46 | 21 | 19 | 1 | 5 | | 20 | 17 | 0 | 9 | |
| Lymphatic
invasion | | | | | | | | | | | |
|
Absent | 121 | 51 | 44 | 0 | 26 | 0.935 | 51 | 43 | 1 | 26 | 0.879 |
|
Present | 38 | 18 | 15 | 1 | 4 | | 17 | 14 | 2 | 5 | |
| Perineural
invasion | | | | | | | | | | | |
|
Absent | 143 | 61 | 53 | 1 | 28 | 0.803 | 60 | 52 | 3 | 28 | 0.903 |
|
Present | 17 | 8 | 6 | 0 | 3 | | 8 | 5 | 0 | 4 | |
| Lymph node
metastasis | | | | | | | | | | | |
|
Absent | 81 | 42 | 31 | 0 | 8 | 0.788 | 41 | 32 | 2 | 6 | 0.623 |
|
Present | 79 | 27 | 28 | 1 | 23 | | 27 | 25 | 1 | 26 | |
| Lymph node pouch
invasion | | | | | | | | | | | |
|
Absent | 41 | 12 | 10 | 1 | 18 | 0.037 | 23 | 15 | 0 | 3 | 0.888 |
|
Present | 39 | 5 | 23 | 0 | 10 | | 12 | 7 | 2 | 18 | |
| Distant
metastasis | | | | | | | | | | | |
|
Absent | 143 | 63 | 52 | 1 | 27 | 0.526 | 63 | 51 | 2 | 27 | 0.539 |
|
Present | 17 | 6 | 7 | 0 | 3 | | 5 | 6 | 1 | 5 | |
| Tumor deposits | | | | | | | | | | | |
|
Absent | 133 | 52 | 51 | 1 | 29 | 0.953 | 52 | 50 | 2 | 29 | 0546 |
|
Present | 27 | 16 | 8 | 0 | 3 | | 15 | 7 | 1 | 4 | |
| Tumor budding | | | | | | | | | | | |
|
Absent | 94 | 40 | 33 | 1 | 20 | 0.230 | 39 | 34 | 2 | 19 | 0.090 |
|
Present | 66 | 29 | 26 | 0 | 11 | | 29 | 23 | 1 | 13 | |
| Necrosis | | | | | | | | | | | |
|
Absent | 45 | 17 | 15 | 1 | 12 | 0.201 | 16 | 16 | 1 | 12 | 0.037 |
|
Focal | 61 | 26 | 27 | 0 | 8 | | 25 | 24 | 1 | 11 | |
|
Moderate | 36 | 15 | 14 | 0 | 7 | | 17 | 15 | 0 | 4 | |
|
Extensive | 18 | 11 | 3 | 0 | 4 | | 10 | 2 | 1 | 5 | |
| Fibrosis | | | | | | | | | | | |
|
Absent | 11 | 7 | 4 | 0 | 0 | 0.687 | 7 | 3 | 1 | 0 | 0.455 |
|
Focal | 72 | 32 | 27 | 1 | 12 | | 32 | 24 | 0 | 16 | |
|
Moderate | 43 | 15 | 16 | 0 | 12 | | 16 | 17 | 2 | 8 | |
|
Extensive | 34 | 15 | 12 | 0 | 7 | | 13 | 12 | 0 | 19 | |
| Treatment
statusa | | | | | | | | | | | |
|
1 | 36 | 27 | 3 | 0 | 6 | <0.001 | 26 | 3 | 2 | 5 | <0.001 |
|
2 | 22 | 17 | 5 | 0 | 0 | | 17 | 5 | 0 | 0 | |
|
3 | 71 | 19 | 41 | 1 | 11 | | 19 | 37 | 1 | 14 | |
|
4 | 31 | 12 | 19 | 0 | 0 | | 12 | 19 | 0 | 0 | |
Subsequent correlation analysis was performed
between different clinical parameters. Specifically, the percentage
of TILs, the ALC, the tumour progression status and the
clinicopathological features were compared (Table V). The histopathological type,
percentage of mucinous component and lymph node pouch invasion
correlated significantly with the evaluated combined parameters
prior to and following surgery. The growth of the tumour was
associated with the percentage of TILs, the ALC and the tumour
progression status prior to surgery (P=0.037). A significant
correlation was observed between eight features with evaluated
combined parameters following surgery. These were the tumour size
(P=0.021), TNM stage (P<0.001), tumour deposits (P=0.001),
necrosis (P=0.042) and lymph node metastasis (P<0.001).
Moreover, the percentage of invasion of venous (P<0.001),
lymphatic (P<0.001) and perineural (P<0.001) sites exhibited
a significant correlation with the combination index including the
percentage of TILs and the ALC. Moreover, no additional
correlations were noted between the percentage of TILs and the ALC,
the tumour progression status and the remaining clinicopathological
parameters. Furthermore, multiparametric analysis was associated
with the treatment status for patients who had received
preoperative treatment. Moreover, TILs/ALC/tumour progression
status and recurrence/prognosis were not statistically significant
(data not shown).
 | Table VAssociations between combined
parameters of tumor-infiltrating lymphocytes, absolute lymphocyte
count and tumor progression status, and clinicopathological
features of patients with colorectal cancer (n=160). |
Table V
Associations between combined
parameters of tumor-infiltrating lymphocytes, absolute lymphocyte
count and tumor progression status, and clinicopathological
features of patients with colorectal cancer (n=160).
| |
TILs,
absolute lymphocyte count and tumor progression status |
|---|
| | Before surgery | After surgery |
|---|
| Parameter | No. | 1, n | 2, n | 3, n | 4, n | P-value | 1, n | 2, n | 3, n | 4, n | P-value |
|---|
| Age, years | | | | | | | | | | | |
|
<60 | 40 | 2 | 12 | 3 | 21 | 0.112 | 9 | 6 | 13 | 10 | 0.688 |
|
≥60 | 120 | 11 | 34 | 22 | 37 | | 25 | 21 | 28 | 32 | |
| Sex | | | | | | | | | | | |
|
Female | 64 | 15 | 17 | 8 | 25 | 0.353 | 15 | 8 | 17 | 16 | 0.966 |
|
Male | 96 | 3 | 29 | 17 | 33 | | 19 | 19 | 24 | 26 | |
| Localization | | | | | | | | | | | |
|
Right-side | 20 | 3 | 6 | 5 | 8 | 0.287 | 5 | 5 | 7 | 5 | 0.864 |
|
Transverse | 14 | 1 | 1 | 4 | 4 | | 5 | 14 | 5 | 3 | |
|
Left-side | 15 | 1 | 9 | 1 | 3 | | 2 | 4 | 2 | 2 | |
|
Sigmoid | 29 | 0 | 3 | 1 | 4 | | 0 | 1 | 4 | 6 | |
|
Rectum | 82 | 7 | 26 | 11 | 30 | | 20 | 3 | 20 | 22 | |
| Tumor growth | | | | | | | | | | | |
|
Expanding | 133 | 10 | 36 | 21 | 49 | 0.037 | 25 | 23 | 33 | 37 | 0.605 |
|
Infiltrate | 27 | 3 | 10 | 4 | 9 | | 9 | 4 | 8 | 5 | |
| Tumor size, cm | | | | | | | | | | | |
|
<2.5 | 27 | 5 | 3 | 4 | 12 | 0.599 | 4 | 4 | 6 | 10 | 0.021 |
|
2.5-5.0 | 106 | 7 | 35 | 19 | 34 | | 23 | 20 | 29 | 24 | |
|
>5.0 | 27 | 1 | 9 | 2 | 12 | | 7 | 3 | 6 | 8 | |
| Histological
type | | | | | | | | | | | |
|
Mucinous | 30 | 1 | 11 | 3 | 14 | 0.001 | 8 | 4 | 9 | 8 | 0.002 |
|
Adenocarcinoma | 130 | 12 | 35 | 22 | 44 | | 26 | 23 | 32 | 34 | |
| Mucinous component,
% | | | | | | | | | | | |
|
10-30 | 15 | 12 | 6 | 0 | 4 | 0.008 | 3 | 3 | 2 | 2 | 0.013 |
|
31-50 | 15 | 1 | 29 | 3 | 10 | | 5 | 1 | 7 | 6 | |
| TNM stage | | | | | | | | | | | |
|
1 | 42 | 4 | 15 | 5 | 14 | 0.085 | 13 | 8 | 10 | 8 | <0.001 |
|
2 | 31 | 2 | 11 | 1 | 14 | | 7 | 5 | 7 | 9 | |
|
3 | 69 | 5 | 17 | 4 | 26 | | 13 | 12 | 18 | 20 | |
|
4 | 18 | 2 | 3 | 15 | 4 | | 1 | 2 | 6 | 5 | |
| Grade of
malignancies | | | | | | | | | | | |
|
2 | 148 | 13 | 44 | 23 | 54 | 0.961 | 33 | 26 | 39 | 38 | 0.659 |
|
3 | 12 | 0 | 2 | 2 | 4 | | 1 | 1 | 2 | 4 | |
| pT stage | | | | | | | | | | | |
|
1 | 3 | 0 | 2 | 0 | 1 | 0.770 | 0 | 2 | 1 | 0 | 0.096 |
|
2 | 62 | 6 | 16 | 10 | 22 | | 15 | 9 | 16 | 15 | |
|
3 | 91 | 6 | 27 | 15 | 34 | | 19 | 14 | 24 | 26 | |
|
4 | 4 | 1 | 1 | 0 | 1 | | 0 | 2 | 0 | 1 | |
| Venous
invasion | | | | | | | | | | | |
|
Absent | 113 | 10 | 35 | 14 | 39 | 0.518 | 24 | 21 | 27 | 26 | <0.001 |
|
Present | 46 | 3 | 11 | 11 | 19 | | 10 | 6 | 10 | 16 | |
| Lymphatic
invasion | | | | | | | | | | | |
|
Absent | 121 | 11 | 37 | 15 | 43 | 0.397 | 27 | 21 | 28 | 30 | <0.001 |
|
Present | 38 | 2 | 9 | 10 | 15 | | 7 | 6 | 13 | 12 | |
| Perineural
invasion | | | | | | | | | | | |
|
Absent | 143 | 12 | 41 | 22 | 53 | 0.661 | 31 | 24 | 36 | 39 | 0.001 |
|
Present | 17 | 1 | 5 | 3 | 5 | | 3 | 3 | 5 | 3 | |
| Lymph node
metastasis | | | | | | | | | | | |
|
Absent | 81 | 8 | 31 | 10 | 34 | 0.196 | 23 | 17 | 23 | 21 | <0.001 |
|
Present | 79 | 5 | 46 | 15 | 24 | | 11 | 10 | 18 | 21 | |
| Lymph node pouch
invasion | | | | | | | | | | | |
|
Absent | 41 | 10 | 33 | 11 | 40 | <0.001 | 25 | 19 | 25 | 26 | 0.006 |
|
Present | 39 | 3 | 13 | 14 | 18 | | 9 | 8 | 16 | 16 | |
| Distant
metastasis | | | | | | | | | | | |
|
Absent | 143 | 11 | 41 | 20 | 56 | 0.851 | 34 | 19 | 35 | 41 | 0.218 |
|
Present | 17 | 2 | 5 | 5 | 2 | | 0 | 5 | 6 | 1 | |
| Tumor deposits | | | | | | | | | | | |
|
Absent | 133 | 9 | 39 | 18 | 51 | 0.654 | 26 | 23 | 32 | 37 | 0.001 |
|
Present | 27 | 4 | 6 | 7 | 7 | | 7 | 4 | 9 | 4 | |
| Tumor budding | | | | | | | | | | | |
|
Absent | 94 | 8 | 24 | 15 | 35 | 0.459 | 15 | 19 | 29 | 21 | 0.941 |
|
Present | 66 | 5 | 22 | 10 | 23 | | 19 | 8 | 12 | 21 | |
| Necrosis | | | | | | | | | | | |
|
Absent | 45 | 3 | 13 | 5 | 16 | 0.401 | 11 | 7 | 10 | 11 | 0.042 |
|
Focal | 61 | 7 | 18 | 6 | 24 | | 12 | 13 | 14 | 16 | |
|
Moderate | 36 | 1 | 11 | 10 | 13 | | 6 | 6 | 11 | 12 | |
|
Extensive | 18 | 2 | 4 | 4 | 5 | | 5 | 1 | 6 | 3 | |
| Fibrosis | | | | | | | | | | | |
|
Absent | 11 | 2 | 24 | 4 | 5 | 0.365 | 2 | 1 | 5 | 3 | 0.294 |
|
Focal | 72 | 5 | 11 | 9 | 27 | | 19 | 10 | 17 | 20 | |
|
Moderate | 43 | 5 | 11 | 6 | 17 | | 6 | 9 | 11 | 12 | |
|
Extensive | 34 | 3 | 0 | 6 | 9 | | 7 | 7 | 8 | 7 | |
| Treatment
statusa | | | | | | | | | | | |
|
1 | 36 | 12 | 6 | 16 | 2 | 0.396 | 2 | 12 | 5 | 17 | 0.007 |
|
2 | 22 | 5 | 12 | 5 | 0 | | 14 | 2 | 2 | 4 | |
|
3 | 71 | 45 | 20 | 5 | 1 | | 25 | 20 | 11 | 15 | |
|
4 | 31 | 23 | 2 | 2 | 4 | | 4 | 12 | 3 | 12 | |
Discussion
Multiple studies have emphasized on the importance
of the host immunity during the development of CRC. The local
inflammation has been previously measured in the tumour mass of CRC
patients following surgical resection (19,20).
More specifically, these studies were based on the measurement of
CD3+ T cells, CD8+ T cells, and/or
CD45RO+ memory T cells that were localized in the
invasive front and in the centre of the primary tumour mass. The
main finding was that the immune score could be used as a superior
predictor of CRC recurrence in everyday practice (19,20).
Furthermore, in response to the predictive value of the intensive
local inflammation caused by high density TILs, the systemic
inflammation is also frequently investigated. It has been shown
that systemic inflammatory response is associated with a pro-tumour
response and poor prognosis for CRC patients (21).
Recent data have suggested that local and systemic
inflammatory response in CRC should be assessed in combination.
This type of assessment may affect patient prognosis. Park et
al (22) demonstrated a
significant role of the density of T lymphocytes within the cancer
cell areas. Moreover, an increased level of modified Glasgow
Prognostic Score (mGPS) was noted in patients with mismatch
repair-deficient (dMMR) CRC (22).
Another study indicated that the combination of the number of
intratumoral CD8-positive T cells with a high level of C-reactive
protein (CRP) may possess predictive value for recurrence in CRC
patients receiving chemotherapy treatment (23). The present study indicated that
local and systemic chronic inflammation was simultaneously observed
in 24% of cases. The majority of the cases (~73%) exhibited a
predominantly local chronic response (high percentage of TILs and
low ALC). Moreover, Turner et al (24) demonstrated the presence of the local
chronic response consisting of lymphocytes, plasma cells,
macrophages within tumour areas (24). In addition, systemic chronic
inflammation was assessed by the expression of
neutrophil-to-lymphocyte ratio (NLR) in preoperative blood samples
in only 10% of stage II colon cancer cases (24). The authors of that study further
indicated that patients with predominantly local response exhibited
the best outcomes, while those with systemic inflammatory response
exhibited reduced survival and poor prognosis (24).
The present study demonstrated the association
between the combination index of TIL percentage and ALC with
clinicopathological features. Patients with low local and systemic
inflammatory response exhibited higher tumour growth and larger
tumour mass associated with necrosis. Moreover, the patients from
group 4 who presented with lymph node metastasis indicated higher
percentage of cancer cell invasion beyond the lymph node pouch to
the local fat tissue. Wu et al (25) demonstrated that the percentage of
TILs and the ALC correlated with local recurrence. Moreover, Xiao
et al (26) postulated that
the density of CD8+ lymphocytes in the biopsy materials
could be combined with the circulating lymphocyte ratio in order to
predict complete response following neoadjuvant chemoradiotherapy
in rectal cancer. Similar observations were found in studies
conducted on breast cancer (27,28).
The present study further indicated that patients with neoadjuvant
therapy (status treatment group 1 and 2) exhibited optimal local
and systemic chronic inflammatory response. Unfortunately, the
present study contains certain limitations. Initially, the sample
size that corresponded to the patients who responded to
preoperative treatment was relatively small. In addition, our
analysis did not provide complete data regarding the type of
chemotherapeutic drugs used and the number of chemotherapy cycles.
Despite these limitations, the present study indicated that the
measured parameters were associated with tumour progression and
neoadjuvant therapy.
The previous study conducted by our group
demonstrated that the percentage of stromal TILs present at the
invasive tumour front correlated with the histopathological
features of tumour progression (17). In the present study, the data
demonstrated that approximately half of all patients exhibited a
locally advanced tumour with lymph node involvement, whereas the
incidence of distant metastases was less common. Therefore, the
study group was divided into 3 subgroups including patients without
invasion of cancer cells, patients with invasion of cancer cells to
lymphatic vessels and local lymph nodes, patients with lack of
distant metastasis and patients with invasion of cancer cells to
all aforementioned structures. In addition, the current study
attempted to select CRC patients whose TILs and lymphocyte count
may have an optimal prognostic value. In the current study, the
combination index of different parameters (TILs/lymphocyte
count/status progression) exhibited the highest diagnostic value in
patients of group 4 (high TILS/high lymphocyte count/presence of
local or distant metastases). The aforementioned combination of
parameters was significantly associated with numerous pathological
and clinical variables, such as tumour growth, tumour size,
histological type, percentage of mucinous components, TNM stage,
venous, lymphatic and perineural invasions, lymph node metastasis,
cancer cell invasion beyond the lymph node pouch, tumour deposits
and incidence of necrosis. It is important to note that the
findings regarding the examined parameters obtained following
surgery exhibited the highest diagnostic value.
In light of these observations, the present study
demonstrated that the investigation of local and systemic chronic
inflammation was associated with tumour progression of CRC
patients. Due to this fact, the diagnosis and prognosis of CRC
patients should be undertaken on the basis of numerous local and
systemic factors.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analysed during this study are
included in this published article.
Authors' contributions
KJ collected data, performed analysis, wrote the
paper, reviewed the literature, acquired the data and contributed
to manuscript drafting. MK analysed and interpreted the
pathological examination and approved the final version of the
article. WF and KL collected and analysed data, and approved the
final version of the article. MG wrote the paper, reviewed the
literature, acquired the data and contributed to manuscript
drafting. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The study was performed in conformity with the
Declaration of Helsinki for Human Experimentation. Written informed
consent was not required, and informed consent included an opt-out
clause approved by the Medical Ethics Committee. The protocol was
approved by the Bioethics Committee of the Medical University of
Bialystok (NoR-I-002/353/2016).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ando K, Hamada K, Watanabe M, Ohkuma R,
Shida M, Onoue R, Kubota Y, Matsui H, Ishiguro T, Hirasawa Y, et
al: Plasma levels of soluble PD-L1 correlate with tumor regression
in patients with lung and gastric cancer treated with immune
checkpoint inhibitors. Anticancer Res. 39:5195–5201.
2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Badran YR, Cohen JV, Brastianos PK, Parikh
AR, Hong TS and Dougan M: Concurrent therapy with immune checkpoint
inhibitors and TNFα blockade in patients with gastrointestinal
immune-related adverse events. J Immunother Cancer.
7(226)2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Fan Y and Moon JJ: Nanoparticle drug
delivery systems designed to improve cancer vaccines and
immunotherapy. Vaccines (Basel). 3:662–685. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Xiong Y, Wang Y and Tiruthani K: Tumor
immune microenvironment and nano-immunotherapeutics in colorectal
cancer. Nanomedicine. 21(102034)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Compton CC: Colorectal carcinoma:
Diagnostic, prognostic, and molecular features. Mod Pathol.
16:376–388. 2003.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Guinney J, Dienstmann R, Wang X, de
Reyniès A, Schlicker A, Soneson C, Marisa L, Roepman P, Nyamundanda
G, Angelino P, et al: The consensus molecular subtypes of
colorectal cancer. Nat Med. 21:1350–1356. 2015.PubMed/NCBI View
Article : Google Scholar
|
|
7
|
Le DT, Uram JN, Wang H, Bartlett BR,
Kemberling H, Eyring AD, Skora AD, Luber BS, Azad NS, Laheru D, et
al: PD-1 blockade in tumors with mismatch-repair deficiency. N Engl
J Med. 372:2509–2520. 2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Norton SE, Ward-Hartstonge KA, Taylor ES
and Kemp RA: Immune cell interplay in colorectal cancer prognosis.
World J Gastrointest Oncol. 7:221–232. 2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Laghi L, Bianchi P, Miranda E, Balladore
E, Pacetti V, Grizzi F, Allavena P, Torri V, Repici A, Santoro A,
et al: CD3+ cells at the invasive margin of deeply
invading (pT3-T4) colorectal cancer and risk of post-surgical
metastasis: A longitudinl study. Lancet Oncol. 10:877–884.
2009.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Bindea G, Mlecnik B, Tosolini M,
Kirilovsky A, Waldner M, Obenauf AC, Angell H, Fredriksen T,
Lafontaine L, Berger A, et al: Spatiotemporal dynamics of
intratumoral immune cells reveal the immune landscape in human
cancer. Immunity. 39:782–795. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Tanio A, Saito H, Uejima C, Takaya S,
Yamamoto M, Tokuyasu N, Sakamoto T, Honjo S, Ashida K and Fujiwara
Y: A prognostic index for colorectal cancer based on preoperative
absolute lymphocyte, monocyte, and neutrophil counts. Surg Today.
49:245–253. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Kozak MM, von Eyben R, Pai JS, Anderson
EM, Welton ML, Shelton AA, Kin C, Koong AC and Chang DT: The
prognostic significance of pretreatment hematologic parameters in
patients undergoing resection for colorectal cancer. Am J Clin
Oncol. 4:405–412. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Yang L, He W, Kong P, Jiang C, Yang Q, Xie
Q and Xia LP: Clinical baseline and prognostic difference of
platelet lymphocyte ratio (PLR) in right-sided and let-sided colon
cancers. BMC Cancer. 17(873)2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hamilton SR and Aaltonen LA: Tumors of the
colon and rectum. In: World Health Organization Classification of
Tumors. Pathology and Genetics of Tumours of the Digestive System.
Lyon, IARC Press, pp103-104, 2000.
|
|
15
|
Lin Q, Wei Y, Ren L, Zhong Y, Qin C, Zheng
P, Xu P, Zhu D, Ji M and Xu J: Tumor deposit is a poor prognostic
indicator in patients who underwent simultaneous resection for
synchronous colorectal liver metastases. Onco Targets Ther.
8:233–240. 2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Therasse P, Arbuck SG, Eisenhauer EA,
Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van
Oosterom AT, Christian MC and Gwyther SG: New guidelines to
evaluate the response to treatment in solid tumors. J Natl Cancer
Inst. 92:205–216. 2000.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Jakubowska K, Kisielewski W, Kańczuga-Koda
L, Koda M and Famulski W: Stromal and intraepithelial
tumour-infiltrating lymphocytes in colorectal carcinoma. Oncol
Lett. 14:6421–6432. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Salgado R, Denkert C, Demaria S, Sirtaine
N, Klauschen F, Pruneri G, Wienert S, Van den Eynden G, Baehner FL,
Penault-Llorca F, et al: The evaluation of tumour-infiltrating
lymphocytes (TILs) in breast cancer: Recommendations by an
International TILs working group 2014. Ann Oncol. 26:259–271.
2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Galon J, Pagès F, Marincola FM, Angell HK,
Thurin M, Lugli A, Zlobec I, Berger A, Bifulco C, Botti G, et al:
Cancer classification using the Immunoscore: A worldwide task
force. J Transl Med. 10(205)2012.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Mlecnik B, Tosolini M, Kirilovsky A,
Berger A, Bindea G, Meatchi T, Bruneval P, Trajanoski Z, Fridman
WH, Pagès F and Galon J: Histopathologic-based prognostic factors
of colorectal cancers are associated with the state of the local
immune reaction. J Clin Oncol. 29:610–618. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lam M, Tie J, Lee B, Desai J, Gibbs P and
Tran B: Systemic inflammation-impact on tumor biology and outcomes
in colorectal cancer. J Clin Cell Immunol. 6(377)2015.
|
|
22
|
Park JH, Powell AG, Roxburgh CS, Horgan
PG, McMillan DC and Edwards J: Mismatch repair status in patients
with primary operable colorectal cancer: Associations with the
local and systemic tumour environment. Br J Cancer. 114:562–570.
2016.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Mori K, Toiyama Y, Saigusa S, Fujikawa H,
Hiro J, Kobayashi M, Ohi M, Araki T, Inoue Y, Tanaka K, et al:
Systemic analysis of predictive biomarkers for recurrence in
colorectal cancer patients treated with curative surgery. Dig Dis
Sci. 60:2477–2487. 2015.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Turner N, Wong HL, Templeton A, Tripathy
S, Whiti Rogers T, Croxford M, Jones I, Sinnathamby M, Desai J, Tie
J, et al: Analysis of local chronic inflammatory cell infiltrate
combined with systemic inflammation improves prognostication in
stage II colon cancer independent of standard clinicopathologic
criteria. Int J Cancer. 138:671–678. 2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wu B, Zhao S, Sheng Y, Ren L and Song G:
The prognostic value of combining the CD8+ lymphocyte
density and the circulating lymphocyte ratio in circumferential
resection margin biopsy in rectal cancer. Medicine (Baltimore).
97(e11972)2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Xiao B, Peng J, Zhang R, Xu J, Wang Y,
Fang Y, Lin J, Pan Z and Wu X: Density of CD8+ lymphocytes in
biopsy samples combined with the circulating lymphocyte ratio
predicts pathologic complete response to chemoradiotherapy for
rectal cancer. Cancer Manag Res. 9:701–708. 2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Afghahi A, Purington N, Han SS, Desai M,
Pierson E, Mathur MB, Seto T, Thompson CA, Rigdon J, Telli ML, et
al: Higher absolute lymphocyte counts predict lower mortality from
early-stage triple-negative breast cancer. Clin Cancer Res.
24:2851–2858. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Lee KH, Kim EY, Yun JS, Park YL, Do SI,
Chae SW and Park CH: The prognostic and predictive value of
tumor-infiltrating lymphocytes and hematologic parameters in
patients with breast cancer. BMC Cancer. 18(938)2018.PubMed/NCBI View Article : Google Scholar
|