Introduction
Extragonadal germ cells tumors are uncommon in
adults and only 2-5% of teratomas can develop in extragonadal sites
(1,2). The most frequent extragonadal teratoma
locations are anterior mediastinum and retroperitoneum in almost,
occurring 60 and 40% of cases, respectively (1). However, the proportion of head and
neck teratomas is approximately 6% (3-5).
Primary Thyroid Teratomas (PTTs) are rare thyroid
neoplasms of germ cells derivation that are characterized by
features of trilineage differentiation. PTTs are commonly benign,
especially, in infants and children; however, they are mostly
malignant in adults (6). Females
are more frequently diagnosed with PTT than men. Typical clinical
manifestations of PTTs are neck mass and neck swelling accompanied
by lymphadenopathy, while malignant PTTs can develop distant
metastases; the most common locations of metastatic disease are
lungs and bones (7,8). According to the grading criteria of
gonadal teratomas, PTTs are categorized in three groups: Benign
(grade 0), immature (grade 1 and 2) and malignant (immature grade
3) teratomas (2). Malignant
teratomas affect almost only adults and can occur with a
significantly more aggressive clinical behavior compared with
benign and immature teratomas (9).
In the majority of cases, surgical resection is the
primary treatment of choice for PTT. However, the introduction of
chemotherapy as a postoperative adjuvant therapy has produced some
revolutionary outcomes in prognosis and overall survival (10). Although benign PTTs have an
excellent long-term prognosis, malignant PTTs are characterized by
overall survival rates even less than 12 months (6,7).
Recurrence of disease, either in the thyroid bed or regional neck
lymph nodes, and the development of metastatic disease are commonly
associated with decreased survival rates and higher rates of
cancer-related deaths (7).
To that end, in the present study, an individual
case report of a patient with PTT and a systematic review of the
literature. We analyzed the clinical characteristics, perioperative
management and the long-term postoperative and oncological outcomes
after surgical resection in patients diagnosed with PTT.
Case report
A 65-year-old female presented to our clinic with a
one-month history of a neck mass. No pertinent past medical illness
or family history was present. On physical examination, a large
hard and mobile mass was palpated in the right thyroid lobe. The
laboratory findings were unremarkable, and her thyroid function was
normal The Ultrasound (US) scan of neck revealed a 5.5x3.5 cm solid
nodule in the right lobe of thyroid. Fine needle aspiration (FNA)
of thyroid nodule was performed by the Endocrine surgeons of our
Unit under US guidance, and the diagnosis of papillary thyroid
carcinoma was then suggested (Bethesda VI). As a next diagnostic
step, a detailed US neck of central and lateral compartments was
performed, and a 3 cm lymph node was detected in the right cervical
compartment. Lymph's node FNA was negative for malignancy. The
patient was scheduled for total thyroidectomy with central neck
dissection. Intraoperatively, the tumor was penetrating the thyroid
capsule and was invading the adjacent tissue involving the right
recurrent laryngeal nerve, thus, a small part of thyroid tissue
surrounding the right recurrent laryngeal nerve was not excised.
Frozen section was not performed. The postoperative period was
uneventful, except for a diagnosis of asymptomatic hypocalcemia.
The patient was discharged on 1st postoperative day with oral
calcium supplementation. Histopathological examination of the
surgical specimen revealed a 5.5 cm malignant PTT (grade 3) of the
right thyroid lobe with heterogenous characteristics and a
synchronous 0.5 cm PTC of left thyroid lobe. Immunohistochemical
studies showed positive staining of PTT's cells CD56, Glypican3 and
TTF-1 and negative staining for Synaptophysin, Chromogranin, CD45,
CD30 and S-100. Regarding the enlarged cervical lymph node, the
histology was negative for malignancy. Four weeks after the
patient's first operation, she presented at our emergency
department with acute airway obstruction. She urgently underwent
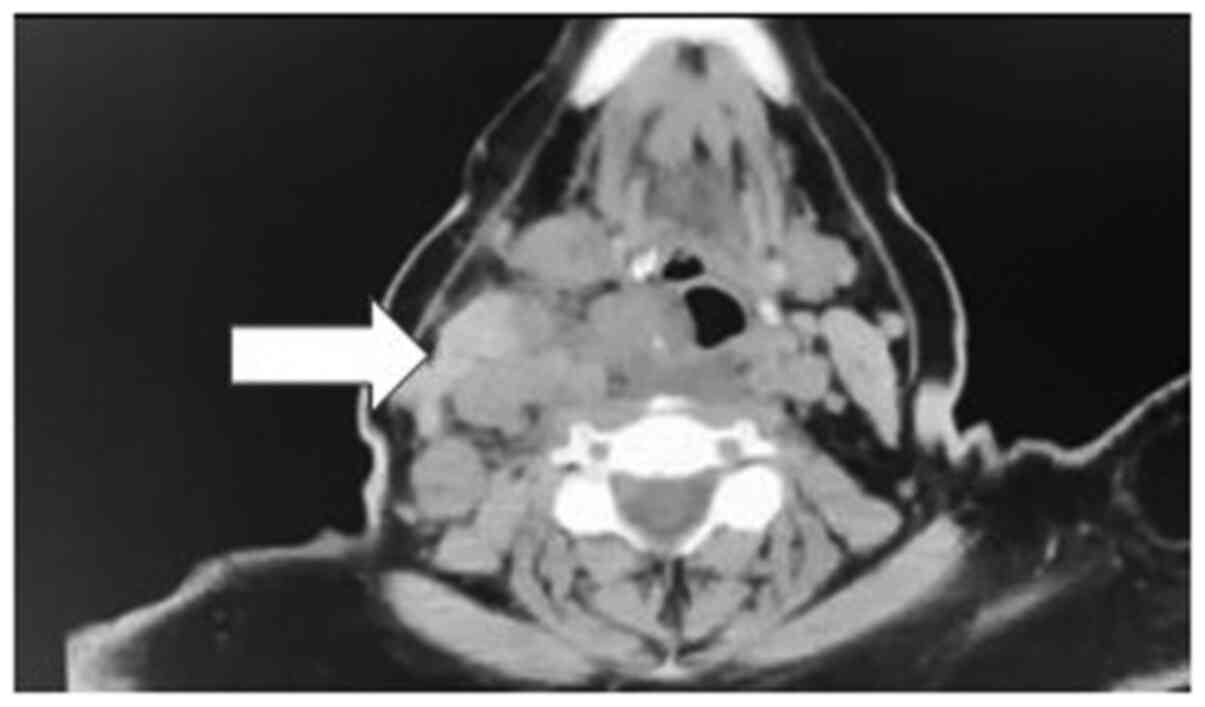
surgery for tracheostomy. A computed tomography scan after the
tracheostomy showed a considerable progression of disease in neck
(Fig. 1). The patient is still
alive and has been admitted in the Oncology Unit of our hospital,
where she received a cis-platinum based chemotherapy.
Literature review
We systematically searched the literature for
articles published from January 2000 up to September 2020 using
PubMed (1966-2019), Scopus (2004-2019) and Google Scholar
(2004-2019) databases along with the references of the articles,
which were retrieved in full text. The following key words were
used for the search: ‘Thyroid teratoma’ ‘primary thyroid teratoma’
‘thyroidectomy’ and ‘malignant thyroid teratoma’. A minimum number
of key words were utilized in order to assess an eligible number
that could be easily searched while simultaneously minimizing the
potential loss of articles. Articles that fulfilled or were deemed
to fulfil the inclusion criteria were retrieved. All articles
published from January 2000 which reported cases of patients aged
>16 years who underwent thyroidectomy and/or neck dissection for
primary thyroid teratoma were included. Data on patient
characteristics included age and sex, while disease characteristics
included primary symptoms and diagnosis, preoperative imaging
studies, size of tumor and type of surgery. Concerning the main
findings of the study, histopathological outcomes, survival rates
regarding incidence of recurrence and cancer-related deaths were
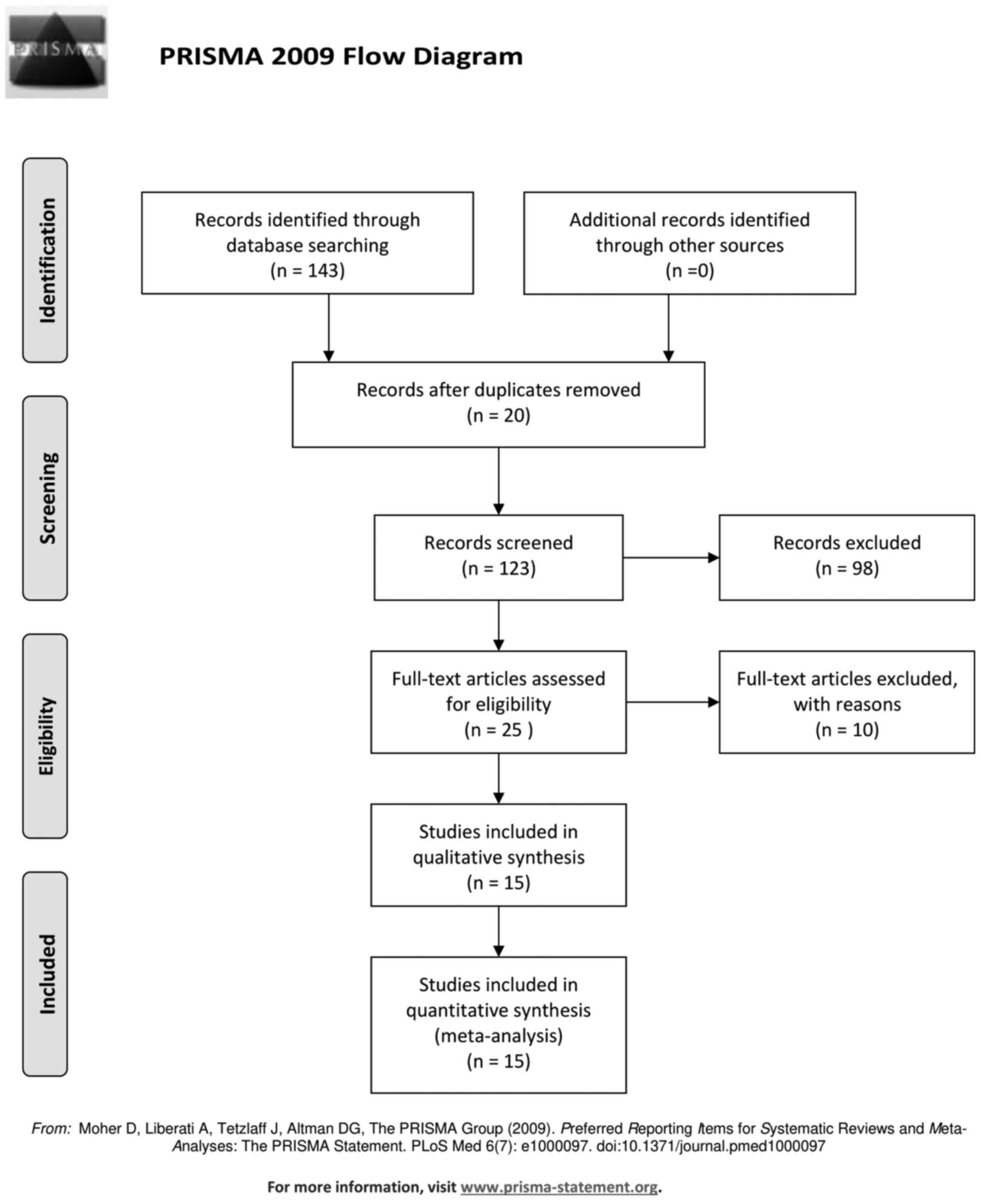
also assessed. The Preferred Reporting Items for Systematic Reviews
and Meta-analyses (PRISMA) flow diagram schematically presents the
stages of article selection (Fig.
2).
A total of 15 studies (11 case reports and 4 case
series) described outcomes for 27 patients who were diagnosed with
PTT and underwent thyroidectomy and in some cases an additional
neck dissection (1,2,4,8,9,11-20).
One study was excluded from the present systematic review (6). The study conducted by Thompson et
al was excluded due to insufficient specific data concerning
patients' main characteristics and survival outcomes (6).
Table I illustrates
the main characteristics of the 27 patients reviewed. The
proportion of men was 18.5% in the current review, while the ages
ranged from 17 to 65 years. As shown in Table I, primary symptoms and signs were
described in all included cases. A growing mass or neck swelling
were the primary sign in 14 (51.8%) patients, while odynophagia was
observed in 4 (14.8%) patients. Four (14.8%) patients presented
with other symptoms (4,11,17,18).
Of these, one (3.7%) presented with severe airway obstruction, one
(3.7%) with dysphonia, one (3.7%) with vocal cord paralysis and one
(3.7%) case with hoarseness of voice. Preoperative imaging studies
were reported in 77.7% of cases (n=21 out of 27 patients), included
a combination of neck Ultrasound (US), Sestamibi scan, Computed
Tomography (CT), Positron Emission Tomography (PET) and Fine-needle
aspiration (FNA) biopsy are presented in Table I. More specifically, the percentage
of patients who had a preoperative neck US and CT for assessing
nodules of thyroid gland were 66.6 and 57.1%, respectively. PET
scan performed preoperatively in 3 out of 21 patients (14.2%),
whereas only 2 cases had a Sestamibi scan (9.5%) (2,8,13,16,18).
Moreover, FNA biopsy of suspicious lesions of thyroid gland was
used in approximately 90.4% (n=19 out of 21 patients) of included
patients.
 | Table IMain characteristics and oncological
outcomes of included patients. |
Table I
Main characteristics and oncological
outcomes of included patients.
| Author (year) | Age (years)/sex | Primary symptoms | Preoperative
studies | Preoperative
diagnosis | Type of surgery | Site of
recurrence/distant metastasis | Time of recurrence or
disease regression (months) | Follow-up
(months) | Overall survival
(months) | Cancer related
deaths | (Refs.) |
|---|
| Jayaram (2000) | 32/F | Neck swelling | US, CT and FNA | Neuroepithelial
tumor | TT and left cervical
and mediastinal lymph nodes | No/Bone, liver
vertebral, paraortic lymph nodes | 6 | 16 | 16 | Yes | (20) |
| Djalilian (2000) | 33/F | Airway
obstruction | FNA | MTC | TT and central neck
dissection | Left side neck
mass/lung meta | 9 | 21 | 21 | Yes | (11) |
| Tsang (2003) | 37/F | Neck swelling | US, MIBI and FNA | Folicular
neoplasm | Subtotal
thyroidectomy | No/no | No | 120 | 120 | No | (8) |
| Martins (2006) | 37/F | Odynophagia | US and FNA | MTC | ΤΤ and Centra neck
dissection | No/no | No | 15 | 15 | NA | (12) |
| Kim (2007) | 31/F | No | CT, PET and
FNA | MTC | TT and bilateral
modified radical neck dissection | No/no | No | 22 | 22 | No | (13) |
| Perez-Mies
(2010) | 38/F | Odynophagia and
dyspnea | US | None | TT | Central compartment
Lymph nodes/no | 2 | NA | NA | NA | (14) |
| Villalonga
(2013) | 64/M | Pain, dysphonia and
dyskinesia | US, CT and FNA | Colloid nodule with
cellular atypia | TT | Thyroid bed/lung
and bone meta | 2 | 5 | 5 | Yes | (4) |
| Oak (2013) | 54/F | Growing mass | US, CT and FNA | Follicular cystic
tumor | Left lobectomy | No | No | NA | NA | No | (15) |
| Pichler (2014) | 43/M | Growing mass | US, MIBI and
FNA | Malignant
tumor | Subtotal
thyroidectomy | Mediastinal mass
and enlarged lymph nodes/no | 24 | 144 | 144 | No | (16) |
| Rabinowits
(2016) | 59/F | Growing mass,
hoarseness | FNA and CT | Undifferentiated
carcinoma | TT and bilateral
modified radical neck dissection | No/no | No | 48 | 48 | No | (17) |
| Starling
(2019) | 17/M | Νο | US, CT and FNA | Poorly
differentiated malignant neoplasm | TT | Thyroid bed/No | 3 | 16 | 16 | No | (1) |
| Ting (2019) Case
1 | 25/F | Pain and
odynophagia | CT, FNA and
PET | Malignant
Teratoma | TT and central neck
dissection, left lateral neck dissection and superior mediastinal
dissection (sternotomy) | No/no | No | 52 | 52 | No | (2) |
| Ting (2019) Case
2 | 30/F | No | US and FNA | Hurthle cell
neoplasm | Left lobectomy
& isthmectomy | No/no | No | 69 | 69 | No | (2) |
| Ting (2019) Case
3 | 36/F | No | US and FNA | No | Right
lobectomy | Thyroid bed,
lateral lymph nodes/no | 3 weeks | 282 | 282 | No | (2) |
| Ting (2019) Case
4 | 32/F | Growing mass | NA | NA | Subtotal
thyroidectomy and tracheostomy | No/no | No | 275 | 275 | No | (2) |
| Miller (2020) Case
1 | 60/M | No | US and FNA | Malignant
neoplasm | NA | Centra compartment
lymph nodes/bone meta | 6 | 13 | 13 | Yes | (18) |
| Miller (2020) Case
2 | 42/F | Growing mass | CT and FNA | Suspicious for
malignancy | NA | No | No | | | No | (18) |
| Miller (2020) Case
3 | 22/F | Growing mass | US, CT, PET and
FNA | Undifferentiated
neoplasm | NA | No/lung meta | No | 12 | 12 | Yes | (18) |
| Miller (2020) Case
4 | 29/F | Vocal cord
paralysis | US, CT and FNA | Poorly
differentiated caricnoma | NA | No/paraaortic and
clavicle lymph nodes | 17 | 51 | 51 | Yes | (18) |
| Miller (2020) Case
5 | 65/F | Growing mass | CT | Poorly
differentiated caricnoma | NA | No | No | 52 | 52 | No | (18) |
| Agaimy (2020) Case
1 | 17/M | Growing mass | NA | NA | Subtotal
thyroidectomy | Thyroid bed/no | 1 | 8 | 8 | No | (9) |
| Agaimy (2020) Case
2 | 17/F | Growing mass | US and FNA | MTC | TT | Mediastinum/no | 1 | 12 | 12 | Yes | (9) |
| Agaimy (2020) Case
3 | 45/F | Pain and
dyphagia | CT and FNA | Unclassified
high-grade sarcoma | TT | NA | NA | NA | NA | NA | (9) |
| Rooper (2020) Case
1 | 65/F | Growing mass | NA | NA | NA | No/no | No | 125 | 125 | No | (19) |
| Rooper (2020) (case
2) | 29/F | Growing mass | NA | NA | NA | No/paraaortic lymph
nodes and bone metastasis | 17 | 53 | 53 | Yes | (19) |
| Rooper (2020) Case
3 | 42/F | Growing mass | NA | NA | NA | No/no | No | 64 | 64 | No | (19) |
| Rooper (2020) Case
4 | 60/F | No | NA | NA | NA | Central compartment
lymph nodes/bone metastasis | 6 | 10 | 10 | Yes | (19) |
A specific preoperative diagnosis was recorded in 18
(66.6%) cases. Among them, 4 (22.2%) patients diagnosed with
medullary thyroid carcinoma, whereas poorly differentiated thyroid
carcinoma and undifferentiated thyroid carcinoma was reported in 3
(16.6%) and 2 (11.1%) patients, respectively. Additionally,
follicular thyroid neoplasm was diagnosed in 3 (16.6%) cases, while
one (5.5%) patient was diagnosed with hurthle cell neoplasm and one
(5.5%) diagnosed with high-grade sarcoma, preoperatively. As a
preoperative diagnosis, FNA biopsy reported malignant tumor and
colloid nodule with cellular atypia in 1 (5.5%) case, respectively
(4,16). One case (5.5%) had a preoperative
diagnosis of a tumor with neuroepithelial differentiation (20). Only one (5.5%) patient was diagnosed
with malignant thyroid teratoma, preoperatively (2). The median size of thyroid lesions from
preoperative studies was 6.1 cm.
Operative outcomes
All patients underwent surgery for PTT. The specific
technique was described in 18 (66.6%) patients. Among them, 5
(27.7%) underwent total thyroidectomy, 4 (22.2%) subtotal
thyroidectomy and 3 (16.6%) lobectomies. Additionally, 6 (33.3%)
patients underwent a more complicated surgical approach due to a
considerably advanced disease (2,11-13,17,20).
Of these cases, 2 (33.3%) underwent total thyroidectomy and central
neck dissection, 2 (33.3%) had total thyroidectomy combined with
bilateral modified neck dissection, one (16.6%) underwent total
thyroidectomy with additional central and left lateral neck
dissection, and one (16.6%) had total thyroidectomy plus left
cervical and mediastinal lymph node dissection. Only one (16.6%)
patient needed additional tracheostomy during subtotal
thyroidectomy due to advanced disease (2). Histopathological examination was
reported in all cases. Only one (3.7%) patient was diagnosed with a
primary benign thyroid teratoma (15), whereas 26 cases were diagnosed with
primary malignant thyroid teratoma. Metastatic disease in cervical
lymph nodes was reported in approximately 44.4% of the included
patients (n=12 out of 27 cases).
Postoperative and oncological
outcomes
Twelve (44.4%) patients received a combination of
adjuvant chemoradiation postoperatively. Eight (29.6%) patients
received only adjuvant chemotherapy. After surgery, 4 (14.8%) cases
received no adjuvant therapy, whereas interestingly 2 (7.4%)
patients had cycles of neoadjuvant chemotherapy preoperatively
before undergoing their primary surgery for PTT. Recurrence of the
disease after their first surgical treatment was reported in ten
(45.4%) patients. More specifically, the location of recurrence in
these 11 patients was the thyroid bed (n=4), the central
compartment lymph nodes (n=3), the lateral lymph nodes (n=2) and
the mediastinum (n=2). The proportion of patients who needed
reoperation after their first surgery due to recurrence, either in
the thyroid bed or in regional lymph nodes was 22.7% (n=5 out of 22
cases) (1,2,4,14,16).
Distant metastases occurred in 8 (29.6%) patients; 5 (62.5%) had
bone metastases, 3 (37.5%) had lung metastases and 3 (37.5%) were
diagnosed with metastases in paraaortic and clavicular lymph nodes
(4,11,18-20).
The average time of recurrence or disease progression after the
first surgery was 3.6±6.25 months, while the average time of
follow-up was 66,7±77 months. Nine (39.1%) patients died due to
progression of the disease (4,9,11,18-20).
Discussion
In our study, we highlighted and evaluated the
long-term outcomes of patients who were diagnosed with PTT and
underwent surgical resection. Despite the fact that a plethora of
preoperative imaging studies were selected, including US and FNA,
only one patient was diagnosed with PTT preoperatively. The
relatively high recurrence rates and the low average time of
recurrence after the first surgery mirror the considerable
aggressiveness of these rare tumors. Additionally, cancer-related
death presented in almost one third of the included cases.
PTTs represent <0.1% of all primary thyroid gland
neoplasms (6). In 1908, Lurje
(21) was the first author who
described a case of PTT in the literature. Bale reported the first
case series of PTT in the English literature in 1950(22). One of the following criteria must be
met to establish the diagnosis of PTT: i) the lesion occupies a
segment of the thyroid gland, ii) the thyroid gland is totally
replaced by the tumor without any evidence of thyroid gland during
the surgery or in preoperative imaging modalities or iii) there is
a direct connection between the thyroid gland and the tumor
(6). Although PTTs are a very rare
entity, they should be considered as a potential diagnosis in every
young patient presenting with a high-grade thyroid tumor with a
rapidly growing neck mass. The asymptomatic gradual enlargement of
the neck is the most frequent clinical manifestation. Depending on
the size and site of PTTs, patients can also present with
significant airway obstruction (23).
Preoperative diagnosis of primary malignant thyroid
teratoma is very challenging. Preoperative US usually shows a
well-defined heterogenous lesion with cystic components;
sensitivity and specificity of 41 and 92%, respectively (23). CT and MRI of the neck could be
additional preoperative diagnostic tools, presenting more favorable
imaging outcomes. The typical appearance of a PTT is a
well-circumscribed mass with both solid and cystic components
(24,25). Although, the vast majority of PTTs
receive a preoperative evaluation through FNA biopsy, they are not
characterized by specific and unique cytologic features. Few
studies reported a neuroendocrine origin in cytologic findings of
MTC (11-13).
In the present systematic review, 4 patients were diagnosed with
MTC and only one patient with PTT preoperatively, using a
preoperative FNA biopsy (2).
Moreover, regarding the 27 included patients there was no case of
synchronous papillary thyroid carcinoma. Our patient had a
synchronous papillary tumor, but the whole postoperative management
organized according to the dominant diagnosis of primary malignant
thyroid teratoma. In most cases, the final diagnosis and grading of
PTT are given following a meticulous histopathological evaluation
of the surgical specimen. Immature neuroepithelium is the main
feature that defines malignancy in surgical specimens.
Immunochemical studies can be useful to confirm neuronal
differentiation and to detect ectodermal or mesodermal
characteristics (1,20,26).
More specifically, Starling et al used several
immunochemical stains, both in preoperative FNA and in final
specimens (1). Immunochemical
studies can also be helpful to exclude some diagnosis from our
differential diagnostic algorithm. The majority of MTCs have
positive calcitonin in immunochemistry, while neuroendocrine tumors
are commonly keratin positive (1).
With regards to molecular characteristics of PTT,
several studies have been published highlighting mutations of genes
which were associated with these rare tumors (9,17,19).
Many authors have already reported, a DICER1 somatic mutation which
potentially has a crucial role in malignant transformation in
patients diagnosed with PTT. More precisely, Rabinowits et
al were the first to report the presence of a pathogenic DICER1
mutation in a case of a malignant thyroid teratoma in a 59-year-old
female (17). The authors linked
the DICER1 mutation to the PNET-like transformation (17). In addition, Rooper et al
describing pathogenicDICER1 mutations in 4 of 4 malignant PTTs, but
none of 4 mature or immature PTTs (19). Although DICER1 mutations have been
associated with multiple malignancies, including thyroid neoplasia
(multinodular goiter, adenomas or differentiated thyroid gland
neoplasia), and ovarian sex cord-stromal tumors, they have not been
demonstrated in gonadal or extragonadal teratomas (9-11).
The presence of the DICER1 mutation might considerably change the
treatment strategy of patients with PTT, in terms of extension of
surgery and neo- or adjuvant chemotherapy.
PTTs described prior to effective therapy were
generally associated with an overall poor outcome and a median
survival of about 8 months (1).
Although, benign PTTs have an excellent prognosis after radical
surgical resection, malignant PTTs have significantly poor survival
even less than a year. Malignant PTTs are relatively insensitive to
radiation and chemotherapy, and radical surgical approach is
considered the standard of care. However, chemotherapy has been
commonly used for patients with immature PTTs, producing better
long-term outcomes (17). Several
studies described significant long-term survival outcomes with the
aggressive use of adjuvant chemotherapy, especially cis-platinum
based regimens (2,8,10).
Although radiation therapy has often been applied in addition to
surgery and chemotherapy, its role is controversial.
Non-seminomatous germ-cell tumors are, generally, not sensitive in
radiotherapy, and, as a result, is not clear if it is beneficial
for patients diagnosed with PTT (2,7,10).
However, if there is an indication of invasion of the oesophagus,
trachea or larynx, postoperative radiotherapy might be an
advantageous option for these patients (10). Interestingly, neoadjuvant
chemotherapy has been described in few cases in the current
literature and may be have promising oncological outcomes in the
management of patients with PTT. However, specific indications of
its usage, such as extrathyroidal extension of the lesion, cervical
lymph nodes metastases or distant metastases should be demonstrated
(2,17).
To our best of our knowledge, this present study is,
the only systematic review in the literature, which describes
characteristics, perioperative management and oncological outcomes
of patients who were diagnosed with PTT and underwent surgical
resection. Of note, the results of our systematic review should be
interpreted in the context of its limitations. The total number of
patients in our study is small and were meticulously selected by
surgical teams, thus, objective results. Further, the true
prevalence of PTT cannot be determined precisely, and data
regarding their characteristics and treatment strategies is limited
to case reports and small case series. Thus, further evaluation and
research is precluded due to their rare entity. None of the
included studies blinded or randomized the participants or
assessors. The significant heterogeneity among the included studies
and the lack of mention of certain parameters by some authors were
additional limitations. Therefore, a potential selection bias must
influence our results.
In conclusion, PTTs are a rare and malignant PTTs
are a very aggressive tumor with a challenging preoperative
diagnosis. Although radical surgery and adjuvant chemotherapy with
several regimens seems to be beneficial for patients, malignant
PTTs have considerably poor survival outcomes. A better and more
accurate preoperative diagnosis in these rare tumors might indicate
a more aggressive surgical approach should be adopted by
experienced endocrine surgeons with a more extensive lymph node
dissection in order to achieve better long-term outcomes.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
CN and APa conceived and designed the study. MS, IM,
KP, APs, GT and EP acquired, analyzed and interpreted the data. APa
and APs drafted and revised the manuscript. CN and EP confirm the
authenticity of all the raw data. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient for publication of this case report and any accompanying
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Starling CE, Sabra J, Brady B, Horton M
and Traweek ST: Malignant teratoma of the thyroid: A difficult
diagnosis by fine-needle aspiration. Dia Cytopathol. 47:930–934.
2019.PubMed/NCBI View
Article : Google Scholar
|
|
2
|
Ting J, Bell D, Ahmed S, Ying A,
Waguespack SG, Tu SM, Weber R and Zafereo M: Primary Malignant
thyroid teratoma: An institutional experience. Thyroid. 29:229–236.
2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Rothschild MA, Catalano P, Urken M,
Brandwein M, Som P, Norton K and Biller HF: Evaluation and
management of congenital cervical teratoma: Case report and review.
Arch Otolaryngol Neck Surg. 120:444–448. 1994.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Vilallonga R, Zafon C, Ruiz-Marcellan C,
Obiols G, Fort JM, Baena JA, Villanueva B, Garcia A and
Sobrinho-Simões M: Malignant thyroid teratoma: Report of an
aggressive tumor in a 64-year-old man. Endocr Pathol. 24:132–135.
2013.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Tonni G, De Felice C, Centini G and
Ginanneschi C: Cervical and oral teratoma in the fetus: A
systematic review of etiology, pathology, diagnosis, treatment and
prognosis. Arch Gynecol Obstet. 282:355–361. 2010.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Thompson LD, Rosai J and Heffess CS:
Primary thyroid teratomas: A clinicopathologic study of 30 cases.
Cancer. 88:1149–1158. 2000.PubMed/NCBI
|
|
7
|
Ueno NT, Amato RJ, Ro JJ and Weber RS:
Primary malignant teratoma of the thyroid gland: Report and
discussion of two cases. Head Neck. 20:649–653. 1998.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Tsang RW, Brierley JD, Asa SL and Sturgeon
JF: Malignant teratoma of the thyroid: Aggressive chemoradiation
therapy is required after surgery. Thyroid. 13:401–404.
2003.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Agaimy A, Witkowski L, Stoehr R, Cuenca
JC, González-Muller CA, Brütting A, Bährle M, Mantsopoulos K, Amin
RM, Hartmann A, et al: Malignant teratoid tumor of the thyroid
gland: An aggressive primitive multiphenotypic malignancy showing
organotypical elements and frequent DICER1 alterations-is the term
‘thyroblastoma’ more appropriate? Virchows Arch. 477:787–798.
2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chen JS, Lai GM and Hsueh S: Malignant
thyroid teratoma of an adult: A long-term survival after
chemotherapy. Am J Clin Oncol. 21:212–214. 1998.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Djalilian HR, Linzie B and Maisel RH:
Malignant teratoma of the thyroid: Review of literature and report
of a case. Am J Otolaryngol. 21:112–115. 2000.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Martins T, Carrilho F, Gomes L, Mesquita
C, Martins MJ and Carvalheiro M: Malignant teratoma of the thyroid:
Case report. Thyroid. 16:1311–1313. 2006.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kim E, Tae SB, Kwon Y, Kim TH, Chung KW,
Kim SW, Ro J and Lee ES: Primary malignant teratoma with a
primitive neuroectodermal tumor component in thyroid gland: A case
report. J Korean Med Sci. 22:568–571. 2007.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Pérez-Mies B, Regojo Zapata RM,
García-Fernández E and Serrano MN: Malignant teratoma of the
thyroid in a pregnant woman. Ann Diagn Pathol. 14:264–267.
2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Oak CY, Kim HK, Yoon TM, Lim SC, Park HB,
Park HC, Han MG and Kang HC: Benign teratoma of the thyroid gland.
Endocrinol Metab (Seoul). 28:144–148. 2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Pichler R, Heidegger I, Brunner A and
Steiner H: Long-term follow-up of a primary teratoma with
somatic-type malignancy within the thyroid gland mimicking thyroid
carcinoma. Clin Genitourin Cancer. 12:e121–e124. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Rabinowits G, Barletta J, Sholl LM, Reche
E, Lorch J and Goguen L: Successful management of a patient with
malignant thyroid teratoma. Thyroid. 27:125–128. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Miller DL, Thompson LD, Bishop JA, Rooper
LM and Ali SZ: Malignant teratomas of the thyroid gland:
Clinico-radiologic and cytomorphologic features of a rare entity. J
Am Soc Cytopathol. 9:221–231. 2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Rooper LM, Bynum JP, Miller KP, Lin MT,
Gagan J, Thompson LD and Bishop JA: Recurrent DICER1 hotspot
mutations in malignant thyroid gland teratomas: Molecular
characterization and proposal for a separate classification. Am J
Surg Pathol. 44:826–833. 2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Jayaram G, Cheah PL and Yip CH: Malignant
teratoma of the thyroid with predominantly neuroepithelial
differentiation: Fine needle aspiration cytologic, histologic and
immunocytochemical features of a case. Acta Cytol. 44:375–379.
2000.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lurje M: Über ein Teratom der Schilddrüse.
Inaugural Disseration, J. J. Meier, Zürich, 1908.
|
|
22
|
Bale GF: Teratoma of the neck in the
region of the thyroid gland; a review of the literature and report
of 4 cases. Am J Pathol. 26:565–579. 1950.PubMed/NCBI
|
|
23
|
Lv Z, Bai X, Sheng Q, Liu J and Wu Y: A
case report of a giant mature teratoma of the thyroid gland in a
young girl. Medicine (Baltimore). 98(e14703)2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Henrichsen TL and Reading CC: Thyroid
ultrasonography. Part 2: Nodules. Radiol Clin North Am. 49:417–424.
2011.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wolter NE, Siegele B and Cunningham MJ:
Cystic cervical teratoma: A diagnostic and management challenge.
Int J Pediatr Otorhinolaryngol. 95:97–100. 2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Craver RD, Lipscomb JT, Suskind D and
Velez MC: Malignant teratoma of the thyroid with primitive
neuroepithelial and mesenchymal sarcomatous components. Ann Diagn
Pathol. 5:285–292. 2001.PubMed/NCBI View Article : Google Scholar
|