Introduction
Dural lymphoma is a rare subtype of primary central
nervous system lymphoma (PCNSL) found in the dura mater that
differs biologically from types of PCNSLs that are usually
high-grade, diffuse large B-cell lymphomas (1,2). Dural
lymphoma is usually a low-grade marginal zone B-cell lymphoma of
mucosa-associated lymphoid tissue (MALT). MALT lymphoma responds
well to local therapies such as surgery and radiation therapy. Most
patients recover completely and have good local disease control in
response to this treatment, but the risk of systemic relapse
appears to be high, as the dura mater is outside of the blood-brain
barrier (BBB) (2). Previous reports
showed that systemic chemotherapy was mainly high-dosage
methotrexate treatment (HD-MTX) together with radiation therapy or
surgery (3). Because the dura mater
is outside of the BBB, HD-MTX may not be useful for treating dural
MALT lymphomas. In this report, we cover two dural MALT lymphoma
patients: One with systemic lymphoid swelling and one who
experienced a third relapse after local radiation therapy and
surgery. These two patients went into complete remission after
treatment with rituximab and bendamustine.
Case report
Case 1
A 65-year-old man was urgently transported to a
neurosurgical hospital on May 19, 2019 with a generalized seizure.
A space-occupying lesion with midline deviation was found in the
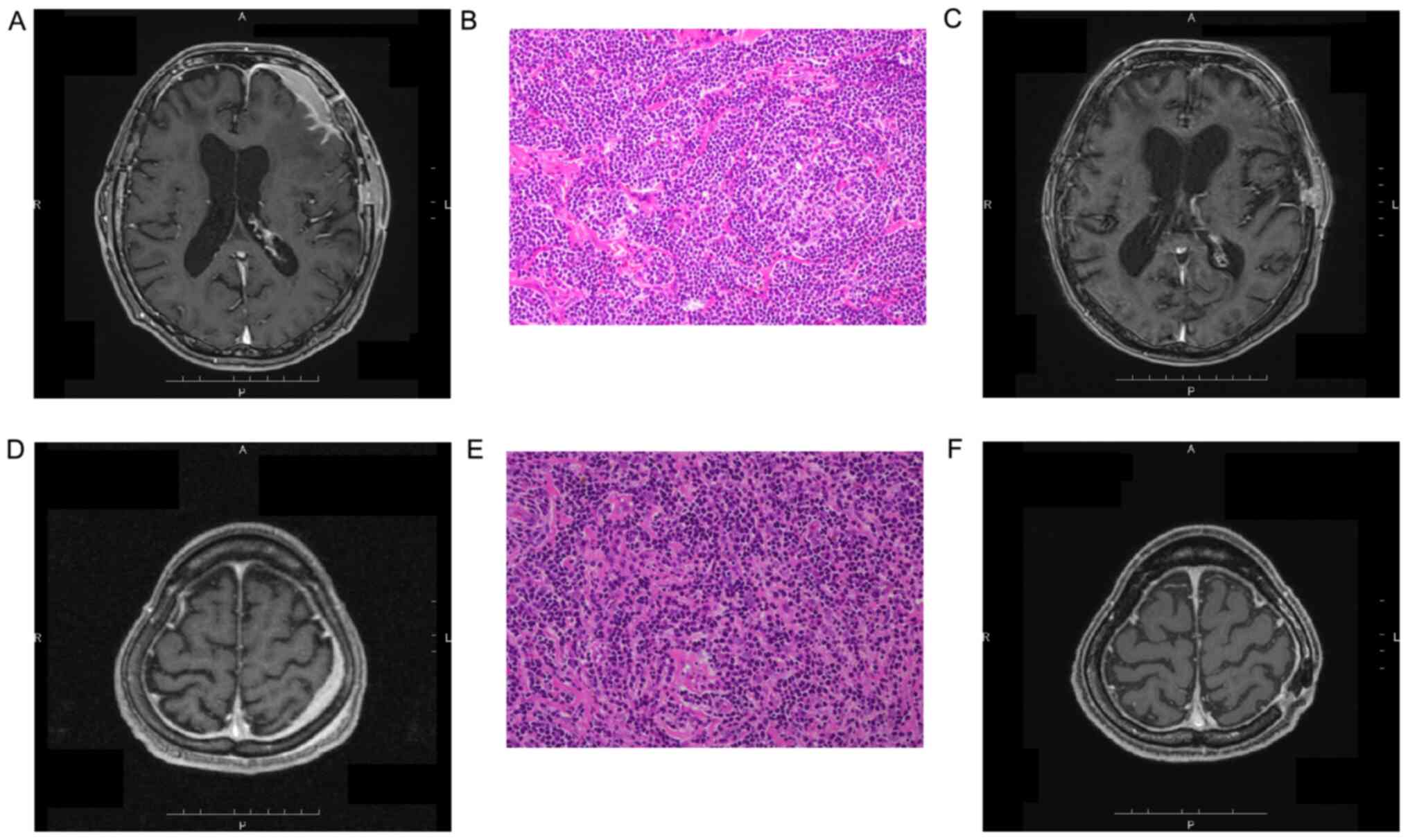
left frontal region of his brain (Fig.
1A), and a craniotomy tumor removal was performed on May 21.
The tumor was distributed in layers in the subcranial subcutaneous
tissue with a hematoma inside and outside the dura mater.
Hematoxylin and eosin staining was was performed according to
routine hospital procedures. The pathological diagnosis of the
tumor was MALT lymphoma (Fig. 1B).
Although flow cytometry was positive for CD20, CD19, cell surface
kappa, and HLA-DR, and G-band pattern showed poor proliferation,
fluorescence in situ hybridization (FISH) was negative for
BIRC3-MALT1 translocation. Moreover, the patient was in the MALT
Lymphoma Clinical Stage IVA IPI High-Intermediate risk group.
Rituximab was used in combination for systemic therapy, and on July
17, 2019, treatment was started with rituximab 375 mg/m2
(Day 1) and bendamustine 90 mg/m2 (Days 2 and 3:
R-Bendamustine). Methotrexate 12 mg and prednisolone 10 mg were
only administered intrathecally the first time (because the
cytodiagnosis was Class I). Six courses of the same regimen were
administered thereafter. Symptoms such as headaches and seizures
quickly improved with the first treatment, and by the time
treatment was completed, his disability had also improved. The
lesions shrunk (Fig. 1C) and are
currently being monitored.
Case 2
A 55-year-old man underwent a medical checkup in
June 2010, and a computed tomography (CT) scan showed a parasternal
tumor and axillary lymphadenopathy. In July of the same year, a
sternum tumor and right axillary lymph node biopsy was performed,
and marginal zone B-cell lymphoma was diagnosed. At the first
visit, his immunoglobulin M (IgM) was 1,981 mg/dl (normal 400
mg/dl), and IgM-κ type M protein was detected by
immunoelectrophoresis. Since Helicobacter pylori (H.
pylori) was detected in his stomach in August 2010, H.
pylori was eradicated with clarithromycin 400 mg, amoxicillin
1500 mg, and a proton pump inhibitor in anticipation of lymphoma
reduction without any effect. Thus, on Day 1, rituximab 375
mg/m2 was administered; on Day 2, cyclophosphamide 750
mg/m2, doxorubicin 50 mg/m2, and vincristine
1.4 mg/m2 were administered; on Days 2-6, prednisolone
60 mg/day was administered. However, no symptoms such as fever or
general fatigue were observed. IgM did not decrease at all and was
determined to be unaffected. In September 2010, the symptoms
improved from daily administration of thalidomide 100 mg and
prednisolone 10 mg, but the sternum tumor did not shrink. In
November, radiation therapy was performed at 40 Gy/20 Fr on the
sternum tumor and bilateral axilla. The tumor shrunk, and the
thalidomide was discontinued. In October 2014, a CT scan revealed a
soft mass in the left heart diaphragm angle. In December 2014, a
thoracoscopic longitudinal tumor resection was performed, and a
recurrence of a marginal zone lymphoma was diagnosed. Flow
cytometry was positive for CD20, CD19, cell surface kappa, and
HLA-DR. G-band pattern showed no chromosomal abnormalities, and
FISH was negative for BIRC3-MALT1 translocation. Since then, we
have been following the patient after re-administration with
prednisolone to maintain remission. In July 2016, interleukin-2
receptor (IL-2R) and IgM increased again, but the chest and
abdominal CT scan showed no new lesions. However, an image with
contrast enhancement showed thickening on the left parietal lobe in
the head MRI performed in August 2016 (Fig. 1D). A dural biopsy was performed in
September 2016. Hematoxylin and eosin staining was performed
according to routine hospital procedures. Small- and medium-sized
lymphocytes were diffused in the dura mater tissue (Fig. 1E), and it was determined that a
marginal zone lymphoma had recurred. Since the patient did not
desire a cerebrospinal fluid (CSF) injection, rituximab was used in
combination for systemic therapy. Four courses of R-Bendamustine
were administered, and the dural lesion disappeared (Fig. 1F). In February 2019, positron
emission tomography was performed and detected multiple bone
metastases. R-Bendamustine was used, but the patient stopped using
it because he was allergic to it. After that, four courses of FCM
therapy (Day 1: Mitoxantrone 8 mg/m2; Days 1-3:
Fludarabine 25 mg/m2 and Cyclophosphamide 200
mg/m2) were performed, and remission was achieved. The
patient is currently undergoing follow-up, and the dural lesion has
not recurred during the course.
Discussion
Although dura mater is outside of the BBB, the
status of dural lymphomas is confused with that of PCNSL. Almost
all dural lymphomas are MALT lymphomas, so almost all treatments
involve surgery and radiotherapy (4). Chemotherapy is used for CNS lymphomas,
so treatment almost always involves regimens with HD-MTX (4). Bustoros et al analyzed previous
reports to determine prognosis for dural MALT lymphoma and reported
that the five-year overall survival rate was 96.7% and the
progression-free survival (PFS) rate was 81.2%, while de la Fuente
et al reported that the three-year and five-year PFS rates
were 89 and 76%, respectively (4,3).
However, from a developmental point of view, the CNS originates at
the neural tube or ectoderm, while the dura mater originates at the
neural crest or mesoderm, wherein the dura mater is not considered
to be nerve tissue or a central nerve. In fact, de la Fuente et
al reported that almost all patients received surgery and
radiotherapy (3). Thus, HD-MTX may
be an excessive treatment against dural MALT lymphoma.
In this report, two cases of dural MALT lymphoma
were treated with rituximab and bendamustine. One patient relapsed
three times and received refractory treatment of R-CHOP,
bortezomib, and thalidomide therapy. A dural tumor with systemic
MALT lymphoma was discovered. Both patients had systemic MALT, so
focal radiation therapy and surgery were insufficient. Our
rituximab and bendamustine therapy was effective on both patients
and prevented CNS metastasis or CSF invasion of MALT from occurring
afterwards. Both cases were Class I in early CSF cytology with no
meningeal dissemination, which may be why treatment with rituximab
and bendamustine therapy was effective and administration of CSF
methotrexate or high-dose methotrexate is possibly unnecessary. On
the other hand, the possibility of dural lesions directly
infiltrating the CNS cannot be ruled out, therefore, mass
methotrexate therapy cannot be ruled out for cases with meningeal
invasion (2).
Rituximab and bendamustine therapy is an effective
treatment for MALT lymphoma (5).
There was an 88% possibility of event-free survival being four
years in this study. Kim et al reported the effectiveness of
rituximab, bendamustine, vincristine, cytarabine, and dexamethasone
(R-BOAD) against PCNSL. Although the CSF concentration of
bendamustine was only 0.025%, they reported tumor regression
(6). Furthermore, Li et al
analyzed the CNS penetration of bendamustine and found that
bendamustine penetrated 35% of the CNS in a mouse model (7). These reports suggest that bendamustine
was effective against both systemic and CNS MALT lymphomas. Some
reports showed the effectiveness of bendamustine in treatments
during the refractory period or after recurrence of the PCNSL with
patients who were treated with HD-MTX (8-10).
In conclusion, although there have only been a few
cases thus far, rituximab and bendamustine treatment appears to be
effective against dural MALT lymphomas and may prevent invasion of
the CNS. Further study is necessary to clarify its clinical
efficacy against dural MALT lymphoma.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
YT, SI and JN were clinical investigators for the
case. YT drafted the manuscript. TTa contributed to acquisition of
data, and TTe contributed to analysis and interpretation of data,
and both participated in writing or technical editing of the
manuscript. YT and SI confirm the authenticity of all the raw data.
All authors read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The study was approved by the local Ethics Committee
of Hakodate Municipal Hospital Institutional Review Board (approval
no. 2019-47; Hakodate, Japan).
Patient consent for publication
Written informed consent was obtained from the
patients described in the report. A copy of the consent may be
requested from the corresponding author.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ayanambakkam A, Ibrahimi S, Bilal K and
Cherry MA: Extranodal marginal zone lymphoma of central nervous
system. Clin Lymphoma Myeloma Leuk. 18:34–37.e8. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Iwamoto FM and Abrey LE: Primary dural
lymphoma: A review. Neurosurg Focus. 21(E5)2006.PubMed/NCBI View Article : Google Scholar
|
|
3
|
de la Fuente MI, Haggiagi A, Moul A, Young
RJ, Sidani C, Markoe A, Vega F, DeAngelis LM and Lossos IS:
Marginal zone dural lymphoma: The memorial sloan kettering cancer
center and university of miami experience. Leuk Lymphoma.
58:882–888. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Bustoros M, Liechty B, Zagzag D, Liu C,
Shepherd T, Gruber D, Raphael B and Placantonakis DG: A rare case
of composite dural extranodal marginal zone lymphoma and chronic
lymphocytic leukemia/small lymphocytic lymphoma. Front Neurol.
9(267)2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Salar A, Domingo-Domenech E, Panizo C,
Nicolas C, Bargay J, Muntanola A, Canales M, Bello JL, Sancho JM,
Tomas JF, et al: First-line response-adapted treatment with the
combination of bendamustine and rituximab in patients with
mucosa-associated lymphoid tissue lymphoma (MALT2008-01): A
multicentre, single-arm, phase 2 trial. Lancet Haematol.
1:e104–e111. 2014.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Kim T, Choi HY, Lee HS, Jung SH, Ahn JS,
Kim HJ, Lee JJ, Yoo HD and Yang DH: Clinical response and
pharmacokinetics of bendamustine as a component of salvage R-B(O)AD
therapy for the treatment of primary central nervous system
lymphoma (PCNSL). BMC Cancer. 18(729)2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Li Z, Caulfield T, Qiu Y, Copland JA and
Tun HW: Pharmacokinetics of bendamustine in the central nervous
system: Chemoinformatic screening followed by validation in a
murine model. Med Chem Commun. 3:1526–1530. 2012.
|
|
8
|
Lim T, Kim SJ, Kim K, Lee JI, Lim DH, Lee
DJ, Baek K, Lee HY, Han B, Uhm JE, et al: Primary CNS lymphoma
other than DLBCL: A descriptive analysis of clinical features and
treatment outcomes. Ann Hematol. 90:1391–1398. 2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chamberlain MC: Salvage therapy with
bendamustine for methotrexate refractory recurrent primary CNS
lymphoma: A retrospective case series. J Neurooncol. 118:155–162.
2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Renfrow JJ, Detroye A, Chan M, Tatter S,
Ellis T, McMullen K, Johnson A, Mott R and Lesser GJ: Initial
experience with bendamustine in patients with recurrent primary
central nervous system lymphoma: A case report. J Neurooncol.
107:659–663. 2012.PubMed/NCBI View Article : Google Scholar
|