1. Introduction
Approximately 1.9 million new cases of colorectal
cancer (CRC) are diagnosed worldwide each year, and 935,000 CRC
patients died of the disease in 2020. In Asia, based on the Global
Cancer Incidence, Mortality, and Prevalence (GLOBOCAN) estimates of
cancer incidence in 2020, there were 0.96 million cases of CRC,
ranked second among all types of cancer, and mortality reached 0.46
million, ranked fourth among all cancer types (1). CRC has a poor prognosis that depends
on the stage of the tumor. Data from the US Surveillance,
Epidemiology, and End Results (SEER) Program revealed that the
5-year relative survival rate for stage I colon cancer is 92%. It
decreases in stage IV CRC, to only 12%. Meanwhile, the 5-year
relative survival rate for rectal cancer is likely lower, 88% for
stage I and 13% for stage IV (2).
The prognosis of CRC is related to its invasion, progression, or
treatment effects (3).
Pathological examination plays a vital role in therapeutic
decision-making and disease prognosis.
The Tumor-Node-Metastasis (TNM) staging system,
developed by the Union for International Cancer Control (UICC) and
American Joint Committee on Cancer (AJCC), is one of the most
common staging systems used in clinical practice (4,5). The
system is also used as a reference by some pathology reporting
standards, such as the International Collaboration on Cancer
Reporting (ICCR) (6) and the
Korean Society of Pathologists (7). However, this method remains
problematic since patients with the same tumor stage could have
significant variations in histopathological features, such as tumor
budding, invasion of the vascular and perineural tissue, tumor
grade, and regression levels (5).
Controversies in CRC pathology reporting also exist, including the
subjective nature of some elements assessed, low reporting accuracy
and reproducibility, and the lack of standard protocols (5,8). In
addition, there are no established biomarkers available on
conventional histopathological prognosis of CRC that can predict
the risk of cancer recurrence, metastasis, resistance to
chemotherapy, prompt targeted therapy, and survival (8).
The best biomarkers should support determining CRC
staging for clinical use. Biomarkers are biological entities that
detect the existence or progression of certain diseases or the
effects of treatments. Biomarkers should have several important
characteristics, such as high diagnostic accuracy, safety, easy
measurements, value to establish an accurate diagnosis, and
capability to narrow down treatment options (9). One of the advantages of using
biomarkers is that tests become more accessible and less invasive
and can be more accepted as part of a routine clinical examination
(10). Thus, identifying new
prognostic biomarkers in CRC is essential to identify changes that
allow us to predict the prognosis of individual tumors to develop
targeted treatments for better clinical outcomes.
DEK is a gene found in the structure of human
genetic material and described as a transcription factor that is
overexpressed in several neoplasms, including CRC. Functionally,
DEK is involved in DNA repair, suppressing cellular aging,
inhibiting apoptosis, and encouraging differentiation, which is
involved in chronic inflammatory pathways and tumorigenesis
(11). Related to its role in
tumorigenesis and its correlation with some pathologic features of
CRC, DEK is considered to have the potential for predicting
CRC prognosis. This review aims to summarize current knowledge and
pieces of evidence supporting the potential of DEK
proto-oncogene as a prognostic biomarker of colorectal cancer.
2. Methods
In writing this evidence-based review, we developed
a searching strategy using the PIO approach (Population: CRC
patients or experimental cell models; Importance/intervention:
pathological examination using the novel DEK biomarker; and
Outcome: prognostic factor) to obtain studies examining the
potential of DEK as a prognostic factor of CRC (12,13).
We did not use comparison as there was no study with a direct
comparison between DEK and previously established biomarkers for
CRC. We used scientific databases such as PubMed, Pubmed Central
(PMC), Proquest, EBSCOHost, Scopus, and Cochrane to obtain evidence
with combined consecutively ordered keywords according to the
disease-determinant-outcome (DDO) approach,
‘colorectal/colon/rectal cancer’ as the disease, ‘DEK’ and
‘biomarker’ as the determinant, and ‘prognosis’ as the outcome.
Articles included in this review are in the level of
evidence 1 to 5 according to the Oxford Centre for Evidence-Based
Medicine (CEBM) guidelines (14),
written in English, and published in the last 10 years. We excluded
non-English studies that were not accessible in full text, did not
match the relevant study design criteria and did not follow with
clinical questions. In the final review, we included meta-analysis,
systematic review, and cohort studies published after January 2012
(a 10-year study period). We also included non-clinical studies
highlighting the role of DEK in tumorigenesis and CRC
prognosis.
3. Results
We obtained seven main articles, comprising a
meta-analysis of cohort studies (level of evidence 1) (15), two cohort studies (level of
evidence 2) (16,17), one cohort study combined with a
non-clinical study (level of evidence 2) (18), and three non-clinical studies using
CRC cell lines as bench research for DEK (level of evidence 5)
(19-21).
One of these studies, a cell line study, was discovered through
hand-searching. As summarized in Table
I (15-21),
generally, existing evidence suggests that DEK is linked with worse
clinicopathological characteristics and survival rates, especially
in patients with specific genotypes [i.e., wild-type Kirsten rat
sarcoma viral oncogene (KRAS oncogene) genotype]. Cell line
studies indicate that high DEK expression is linked to the ability
of cancer cells to avoid apoptosis, and DEK degradation might be
decreased due to mutations leading to tumorigenesis. Lower
expression of DEK is also related to a lack of
epithelial-mesenchymal transition (EMT) and more infiltrative
cancer. Lower DEK might suggest better therapy response with
irinotecan-based chemotherapy regimens (with a biomarker, annexin
A5, being increased). In contrast, in patients with stage II-III
rectal adenocarcinoma, increased DEK is linked to better treatment
response when fluoropyrimidine-based (FOLFIRI or 5-FU) chemotherapy
regimens are used, due to a link with the pro-apoptotic factor
p38.
 | Table ISummary of the findings of DEK as a
prognostic factor for CRC (15–21). |
Table I
Summary of the findings of DEK as a
prognostic factor for CRC (15–21).
| No. | Authors (ref),
year | Design | LoE | Sample | Classification of
positive DEK expression | Findings |
|---|
| 1. | Liu et al
(15), 2017 | Meta-analysis | 1 | 14 cohort studies,
8 of which cover cancers of the digestive system | N/A
(meta-analysis) | DEK overexpression
was significantly attributed to worse overall cancer survival, with
low heterogeneity overall: |
| | | | | | | i) All type of
cancer: HR 1.70, 95% CI: 1.48-1.96, P<0.001 (I2=9%,
P=0.36) |
| | | | | | | ii) Cancers of the
digestive system : HR 1.83, 95% CI: 1.52-2.19, P<0.001
(I2=18%, P=0.30) |
| 2. | Martinez-Useros
et al (17), 2018 | Cohort study | 2 | 74 stage II-III
rectal adenocarcinoma patients undergoing neoadjuvant
chemoradiotherapy using FOLFOX (n=14) or 5-FU (n=60) | Based on expression
rating methods used for DEK in the Human Protein Atlas | High expression of
DEK was associated with the possi bility of better neoadjuvant
fluoropyrimidine-based chemotherapy response (P=0.023) |
| 3. | Martinez-Useros
et al (18), 2014 | Cohort study | 2 | 67 stage IV CRC
patients treated with FOLFIRI regimen of chemotherapy | DEK intensity is
based on a HistoScore calculating cellular antigen intensity and
the number of positive cells. The intensity cutoff was not
described | i) Progression-free
survival was shorter in patients with higher DEK expression.
However, the correlation between DEK and progression-free survival
was only found in the KRASwt patient group
(P<0.05) |
| | | | | | | ii) The risk of
progression was higher in KRASwt patients with
increased expression of DEK (HR 2.4, 95% CI: 1.04-5.58,
P=0.04) |
| | | Experimental
study | 5 | 9 human-derived CRC
cell lines, especially DLD-1 and SW620 | | iii) There was a
higher DEK expression in CRC cell lines than in normal cells,
especially in metastasis-derived cell lines |
| | | | | | | iv) DEK
silencing was correlated with a decrease in, viability delay of
cell repair, a reduction in migrating ability, and better response
to irinotecan-based chemotherapy (P<0.05) |
| | | | | | | v) The difference
in DEK expression did not correlate with a better response to 5-FU
or oxaliplatin |
| 4. | Lin et al
(16), 2013 | Cohort study | 2 | 109 CRC
patients | Positive for DEK:
5-25% positive cells Strongly positive for DEK: >25% positive
cells | i) The proportion
of positive and strongly positive DEK expression was significantly
higher in cancer tissue of CRC patients than in normal tissue
surrounding cancer in those same patients or patients with adenoma
(P<0.01). (Positivity rate: 95.41 vs. 33.03 vs. 32.69%, strong
positivity rate: 48.62 vs. 9.17 vs. 13.46%) |
| | | | | | | ii) Proportion of
patients with DEK overexpression was higher in group of patients
with larger tumor size [OR 2.353 (95% CI: 1.086–5.101), P=0.029],
moderate to poor differentiation [OR 2.824 (95% CI: 1.291–6.177),
P=0.009], lymph node metastasis [OR 2.975 (95% CI: 1.360–6.509),
P=0.006], invasion of serosal layer [OR 2.353 (95% CI:
1.072–5.163), P=0.031], and worse staging [OR 2.744 (95% CI:
1.261–5.971), P=0.010] |
| | | | | | | iii) Proportion of
patients with DEK overexpression was higher but not statistically
significant in patients with male sex [OR 2.461 (0.924–6.556),
P=0.067], older age ≥49 years [OR 1.298 (0.611–2.756), P=0.497],
location on rectal compared to colon/ileocaecal [OR 0.900
(0.424–1.910), P=0.784], and increased CEA [OR 0.610 (0.273–1.362),
P=0.228] |
| | | | | | | iv) Patients with
DEK overexpression had lower 5-year survival and lower disease-free
survival (P<0.001) |
| | | | | | | v) The lower
survival rate was also found in patients with DEK overexpression
coexisting with the appearance of one of four characteristics of
aggressive CRC: serosal invasion, metastasis of the lymph nodes,
worse staging, or higher CEA levels (P<0.001) |
| | | | | | | vi) Overall, the
median survival of patients in this study was 56 months; however,
there was no specific information regarding the comparison of
median survival in each subpopulation |
| | | | | | | vii) DEK
overexpression was an independent prognostic factor of CRC (HR
1.805, 95% CI 1.208–2.699, P=0.004) |
| 5. | Lin et al
(21), 2014 | Experimental
study | 5 | 55 CRC patient
tissue specimens, 22 standard colon mucosal specimens, 18
colorectal adenoma specimens | Positive for DEK:
5-25% positive cells Strongly positive for DEK: >25% positive
cells | i) DEK protein
expression was positively correlated to the Ki-67 index (P=0.030)
but negatively correlated to the apoptosis index (P=0.010) |
| | | | | | | ii) DEK
silencing inhibited tumor cell growth and its ability to form
colonies while contributing to a higher apoptosis rate |
| | | | | | | iii) DEK
silencing stimulated the p53/MDM2 pathway and Bcl-2/Bax
pathways, and caspase-dependent pathways. All three contributed to
a pro-apoptotic cell milieu |
| 6. | Babaei-Jadidi et
al (20), 2011 | Experimental
study | 5 | Human and mouse
cell lines | An ordinal score
scale of 1 (low) to 4 (extremely intense) was used to classify IHC
for DEK | i) High expression
of DEK was found in gut-specific Fbxw7- deleted intestine
mouse tissue. The increased expression of DEK was also correlated
with cells with mutated Fbxw7 (P=0.001) |
| | | | | | | ii)
DEK-silenced cells were found to have more cells in the
G0/G1 phase of the cell cycle and fewer cells in the S or M stage.
In contrast, cells with high expression of DEK had more cells in S,
G2, or M phase (P<0.05) |
| | | | | | | iii) Fbxw7
deactivated DEK by E3 ligase activity in the pres ence of
GSK-3β |
| 7. | You et al
(19), 2017 | Experimental
study | 5 | Human-derived CRC
cell lines, especially SW620 and SW480 | DEK positivity was
analyzed through western blot analysis as ‘positive’ or
‘negative’ | i) DEK
silencing was significantly associated (P<0.05) with decreased
expression of IMP3, increased E-cadherin, and decreased vimentin
and MMP-9 |
| | | | | | | ii) DEK
silencing was significantly associated (P<0.05) with a drastic
decrease in cell viability, the elevation of apop tosis rate, and
decreased ability of cell invasion (accompanied by cellular
transformation from interstitial- like spindle cells into
epithelioid cells) |
| | | | | | | iii) DEK
silencing was not only associated with increased apoptosis (related
to p53 activity) but also EMT |
The role of DEK as a biomarker for
colorectal cancer prognosis: Non-clinical studies
Martinez-Useros et al (18) recorded that all CRC cell lines used
in the research as samples were found to have overexpression of DEK
protein. On the other hand, they also observed that when DEK
gene expression was suppressed, especially in the representative
cell lines DLD-1 and SW620, the ability of CRC cells to survive or
migrate was significantly decreased. The suppression of DEK
gene expression was found to cause slightly increased expression of
annexin A5, a protein associated with cell apoptosis, and a
significant simultaneous decrease in cell viability. However, a
cell culture given 7-ethyl-10-hydroxycamptothecin (SN38), an active
component of irinotecan, showed a significant (P<0.05) increase
in expression of annexin A5 after experiencing suppression of DEK.
These findings in both cell lines (DLD-1 and SW620) indicate the
potential of DEK as a response marker to irinotecan-based
chemotherapy in patients of CRC. This effect was not observed in
cell cultures given 5-FU or LOHP (an active component of
oxaliplatin). Cells with low DEK expression also were shown to have
decreased Ki-67 index levels alongside increased production of
cleaved caspase-3(18).
A study by Lin et al (21) in 2014 discovered a significant
positive relationship between the expression of the DEK protein and
Ki-67, a protein encoded by the MKI67 gene associated with
cell proliferation. The reverse correlation was found between DEK
expression and apoptosis (lower DEK expression means higher
apoptosis count, and vice versa). Transfection of silencer RNA for
DEK (siDEK) also significantly decreased cell growth of the
SW620 CRC cell line due to increased early apoptosis. In addition,
DEK suppression also decreased mutant p53, MDM2, and Bcl-2
expression while upregulating Bax expression. Caspase-dependent
apoptosis pathways were also found to be upregulated in cells with
low DEK, as suggested by lowered expression levels of cleaved
caspase-3 and caspase-9, but unaltered levels of caspase-8 and
increased levels of cleaved poly-ADP ribose polymerase (PARP).
These findings indicate that DEK suppression also suppresses
pathways related to apoptosis, such as p53/MDM2, Bcl-2/Bax, and
caspase-dependent pathways of apoptosis.
Babaei-Jadidi et al (20) noted that in the intestines of mice
with mutations of the Fbxw7 gene locus, a known
tumor-suppressor locus, tumorigenesis was observed accompanied by
changes in the expression of several proteins and genes, including
DEK and RNA tropomyosin. Additionally, some data showed an
association between DEK accumulation and the oncogenicity of the
Fbxw7 mutation, both in human and murine intestines. This
association might elucidate the mechanism allowing DEK to cause
tumorigenesis in CRC. Although the DEK transcription level did not
change, mutations related to CRC tumorigenesis affected the DEK
degradation process.
A study by You et al (19) using the SW480 and SW620 CRC cell
lines testing the impacts of DEK knockdown with a
DEK-interfering lentivirus showed decreased expression of DEK and
insulin-like growth factor II mRNA binding protein 3 (IMP3), as
well as changes in proteins associated with epithelial-mesenchymal
transition (EMT); E-cadherin was significantly increased, along
with a significant decrease in vimentin and matrix metalloprotein-9
(MMP-9). DEK downregulation was also associated with decreased cell
viability, promotion of apoptosis, and decrease of cell invasion,
which was related to the enhancement of E-cadherin and
downregulation of vimentin and MMP-9.
The potential of DEK as a biomarker
for colorectal cancer prognosis: Clinical studies
A meta-analysis by Liu et al (15) examined 14 cohort studies of DEK in
cancers of various origins, eight of which were digestive system
cancers. The study found that DEK overexpression was significantly
attributed to worse survival of all types of cancer, including
cancer of the digestive system. In cancers of all origins, overall
survival of cases with DEK overexpression was lower compared to
cases without DEK overexpression, either in univariate [n=13,
hazard ratio (HR) 1.83, 95% confidence interval (CI): 1.64-2.05,
P<0.001 (I2=0%, P=0.71)] or multivariate analysis
[n=9, HR 1.70, 95% CI: 1.48-1.96, P<0.001 (I2=9%,
P=0.36)]. The same finding was also reported in the subpopulation
with cancers of the digestive system, either in univariate [n=8, HR
1.87, 95% CI: 1.62-2.15, P<0.001 (I2=0%, P=0.69)] or
multivariate analysis [n=6, HR 1.83, 95% CI: 1.52-2.19, P<0.001
(I2=18%, P=0.30)]. All results of this meta-analysis
have low to no heterogeneity.
A cohort study by Martinez-Useros et al
(18) reported findings in a 67
stage IV CRC cohort receiving FOLFIRI, a chemotherapy regimen
comprising folinic acid, 5-FU, and irinotecan. They revealed that
progression-free survival was shorter in patients with higher DEK
expression. By univariate Cox analysis, they reported significantly
lower progression-free survival of CRC based on DEK status (HR
2.825, 95% CI: 1.238-6.449; P=0.014), while the progression-free
survival of CRC based on BRAF (HR 1.119; 95% CI: 0.410-3.055;
P=0.828) and topoisomerase-I status (HR 1.017; 95% CI: 0.364-2.845,
P=0.974) was found to be insignificant. However, the correlation
between DEK and progression-free survival was only found in the
KRAS-wild-type (KRASwt) patient group (P<0.05). In
contrast, this correlation was not observed in KRAS-mutated
(KRASmut) patients. They also documented that the risk
of progression was quantitatively higher in KRASwt
patients with increased DEK expression (HR 2.4, 95% CI: 1.04-5.58,
P=0.04). Therefore, according to this study, CRC patients with
KRASwt and higher DEK expression have a poorer
prognosis.
The same authors (17) also conducted another study in a
cohort of 74 stages II-III rectal adenocarcinoma patients
undergoing neoadjuvant chemoradiotherapy using FOLFOX, a
chemotherapeutic regiment comprising folinic acid, 5-FU, and
oxaliplatin (n=14), or 5-FU only (n=60). They observed that high
expression of DEK was associated with the possibility of better
neoadjuvant fluoropyrimidine-based chemotherapy response. In
patients with an increased expression of DEK, 19% were found to
have a complete reaction to neoadjuvant chemoradiotherapy. In
contrast, no patient with low expression of DEK reached complete
response (P=0.023). Although this appears to contradict the
previous study, the authors argue that this occurs because the
characteristics of the patients (including tumor type and stage)
are different; thus, the potential for DEK as a cancer biomarker
could be varied. In the previous study, annexin A5 expression was
unchanged precisely in cell lines given 5-FU or the oxaliplatin
active component LOPD (18). They
explained that, in this situation, the association of DEK with the
p38 pro-apoptotic factor might contribute to a better therapeutic
response in patients (17). In
addition, the high expression of DEK was possibly correlated to
lower residual tumor cell burden after neoadjuvant
chemoradiotherapy (22).
Lin et al (16) documented significant overexpression
of DEK protein in CRC tissues compared to colorectal adenoma tissue
or normal tissue. Positive expression for DEK was noted in 104 of
109 samples (95.41%) of CRC tissue specimens in the cohort,
compared to 36 in adjacent normal tissue mucosa (33.03%) or 17 in
52 specimens of colorectal adenomas (32.69%). A strong expression
was also found in favor of CRC specimens (52/109, 48.62%) compared
to adjacent normal colon mucosa (10/109, 9.17%) or colorectal
adenomas (7/52, 13.46%). All the results were statistically
significant (P<0.05).
The same study (16) also documented a significant
association between several clinicopathological characteristics
related to worse CRC and overexpression of DEK, such as tumor size,
lymph node metastasis, grades of differentiation, clinical cancer
stage, and serous layer invasion. However, other characteristics
showed no relationship with DEK overexpression, such as age, sex,
tumor location, and carcinoembryonic antigen (CEA) level. It was
also noted that CRC patients with serosal invasion, lymph node
metastasis, increased CEA levels, and late-stage tumors with DEK
overexpression respectively had significantly (P<0.01) lower
5-year survival rates than their counterparts without DEK
overexpression. Multivariate analysis using the Cox proportional
hazards model discovered serosal invasion (HR 1.708, 95% CI:
1.414-2.555, P=0.009), late-stage disease (HR 1.663, 95% CI:
1.081-2.558, P=0.021), and DEK overexpression (HR 1.805, 95% CI:
1.208-2.699, P=0.004) as independent predictors of poor survival in
CRC. Another known prognostic biomarker of CRC, CEA, was
insignificant (HR 1.415; 95% CI: 0.904-2.214, P=0.129).
4. Discussion
The DEK proto-oncogene
The DEK proto-oncogene is a gene found in the
structure of human genetic material. This gene is located at the
chromosomal locus 6p22.3. It encodes a protein not currently
categorized in any protein family and comprises 375 amino acids
with an estimated weight of 43 kilodaltons (kDa) (16,23,24).
The DEK protein has two DNA binding domains, namely the
SLAM-associated protein (SAP) domain, found in several other
proteins, and other DNA binding structures located in its
carboxy-terminal region (24-26).
Although its exact role is still being explored today, this protein
is strongly suspected of being a regulator of the structure of
genetic material, rather than of genetic sequences, where the SAP
domain plays a role in triggering positive supercoiling of
reversible DNA. The other DNA binding structures found in this
protein can regulate the affinity of this protein for DNA, which
can influence the transcription of genetic material (24-27).
Moreover, the DEK protein was also documented to have a role in DNA
replication and RNA splicing (28-30).
The DEK gene was initially investigated in
acute myeloid leukemia (AML) patients, where it fused with the CAN
protein/nucleoporin 214 (CAN/NUP214) gene on t(6; 9)(p23; q24)
translocation. This gene translocation is even considered a basis
for the stratification of AML patients (16,28).
Although chromosomal changes in the DEK locus are not
typical features of various malignant cases, there is a higher
expression of DEK protein in different malignancies. Some types of
malignancies that show increased expression of DEK include AML,
hepatocellular carcinoma, glioblastoma, cervical cancer, ovarian
cancer, melanoma, and others, including CRC (20,28,31-36).
The role of DEK in tumorigenesis and
pathogenesis of colorectal cancer
Several studies on DEK have unraveled clues on how
DEK plays a role in tumorigenesis in general and the pathogenesis
of CRC. DEK can control several signaling pathways associated with
cell proliferation, apoptosis, and inflammation, as shown in
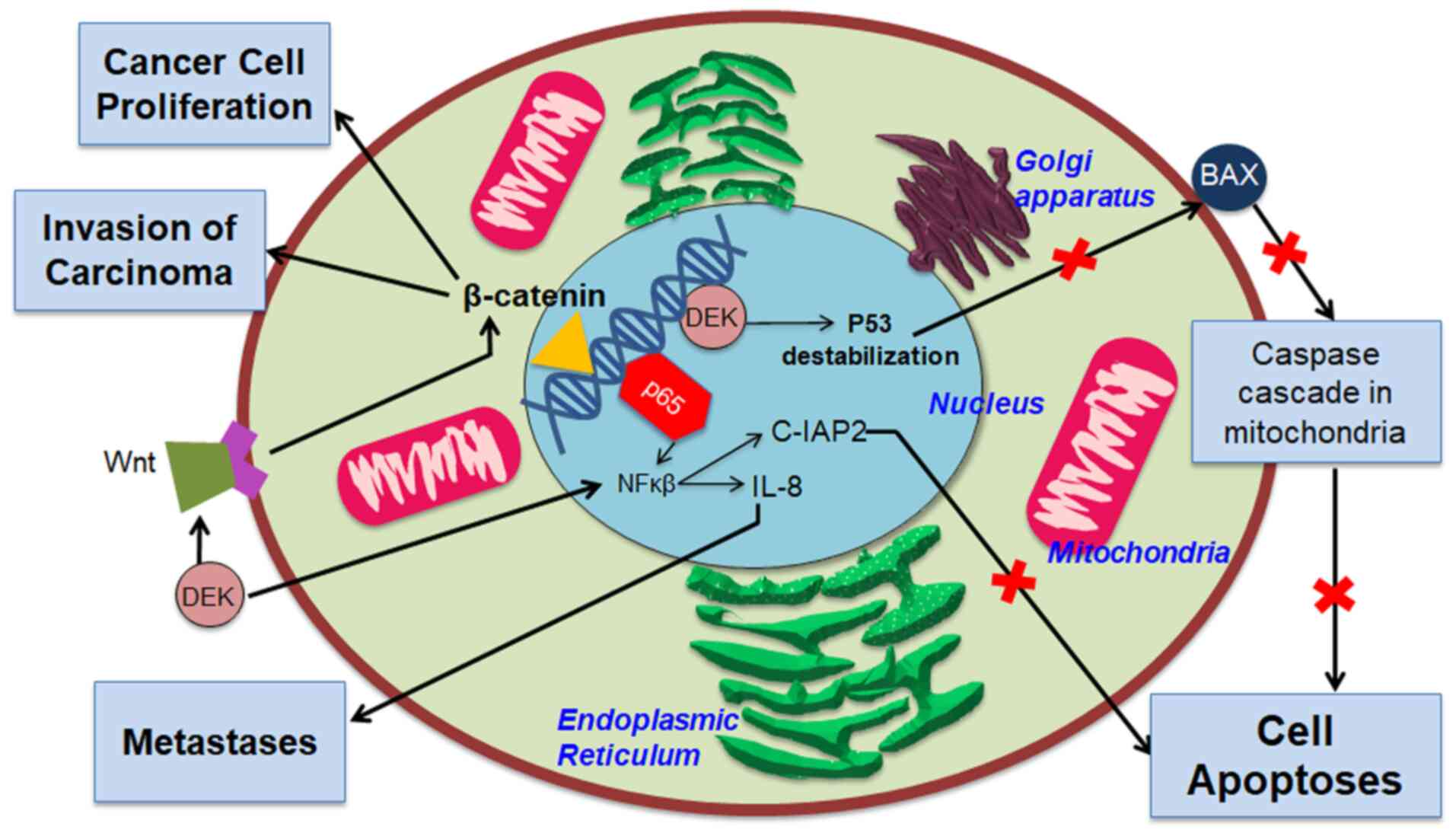
Fig. 1 (11,21).
DEK can influence cell proliferation by affecting
the expression of several molecular signaling pathways, such as
Wingless-related integration site/β-catenin-1 (Wnt/β-catenin). DEK
can adjust Wnt molecular signaling pathways, including Wnt4, Wnt7b,
and Wnt10b. These three Wnt pathways, known to affect cell
proliferation and oncogenic cell phenotype, trigger β-catenin
activation. Activation of β-catenin supports carcinoma
proliferation, invasion, and metastasis (37,38).
Some studies have also demonstrated the ability of
DEK to prevent apoptosis. One mechanism that allows this to happen
is the destabilization of the gene TP53 producing the tumor
suppressor protein p53, which plays a role in responding to stress
experienced by cells. Destabilization of p53 causes its function as
a tumor suppressor to cease, triggering tumorigenesis. In the
condition that DEK is suppressed, the role of p53 as a tumor
suppressor can be carried out through the activation of p53 target
genes, one of which produces the Bcl2-associated X protein (Bax).
Activation of Bax, a pro-apoptotic factor commonly expressed on
cell membranes, causes Bax to beome an integral protein in the
mitochondrial membrane. Bax then triggers the release of apoptotic
factors from the mitochondria, which further triggers cytochrome
c and initiates a cascade reaction from caspase enzymes.
This chain reaction triggers apoptosis (21,39).
Some literature suggests the relationship between
inflammatory processes and tumorigenesis (40,41),
and DEK was shown to have a role in bridging these two events. It
is postulated that different pathways induce DEK overexpression in
inflammatory and proliferative situations. Molecular pathways that
trigger DEK overexpression in inflammatory conditions include AP-1,
Ets-1, NFB, NF-AT, STAT4, and C/EBP-β. In addition, the interleukin
(IL)-8 cytokine is also known to trigger the secretion of
phosphorylated DEK under inflammatory conditions. Meanwhile,
several pathways that trigger DEK overexpression in proliferative
conditions include E2F, ERα, NF-Y, and YY1. However, a study on
these various molecular pathways would be more fitted to help
determine the etiology of CRC and therefore is outside the scope of
this review (11).
Related to its role in bridging the
inflammatory-tumorigenesis process, although DEK is a core protein
expressed by cells into the cytoplasm and the cell nucleus strictly
regulates its secretion, extracellular secretion of DEK also
fulfills several roles affecting both inflammation and
tumorigenesis. DEK is a chemoattractant attracting leukocytes to
specific locations to trigger autoimmune reactions by reacting with
anti-DEK antibodies. DEK secretions can also initiate chromatin
re-formation and other activities that impair normal cell functions
and trigger pathogenetic reactions to produce transformation,
chemoresistance, inflammation, and tumor development of surrounding
cells (24,42,43).
The expression of DEK has also been found to affect various
inflammatory signaling pathways, one of which is NF-κB, a factor
that plays a role in chronic inflammation and tumorigenesis. The
increase in DEK expression was noted to trigger changes in NF-κB
transcription activity through colocalization with the
transcription factor p65, which would further trigger activation of
tumorigenesis-supporting genes, such as cellular inhibitors of
apoptosis proteins 2 (c-IAP2), a part of inhibitors of apoptosis
proteins (IAP) family, and IL-8 which is pro-metastatic (11,41).
The applicability of DEK as a
prognostic biomarker for colorectal cancer
The expression of DEK can be detected by
immunohistochemistry, using 3,3’-diaminobenzidine as the chromogen
and hematoxylin as the counterstain (16). Therefore, the practicality of DEK
as a prognostic biomarker varies according to whether the health
facility has pathology installations able to conduct
immunohistochemistry examinations. The evaluation approach is
mainly semi-quantitative, using either positively stained cell
count, referencing other literature such as the Human Protein Atlas
for cut-off points, or the HistoScore approach that counts the
number of cells based on staining intensity (16-18).
Lin et al used a defined cut-off to define the positive
expression of DEK if 5-25% of cells were stained and robust
expression of DEK if >25% of cells were stained (16,21).
Other methods used include ordinal scoring systems and positive or
negative expression measures using western blot analysis instead of
immunohistochemistry (19,20). A semi-quantitative cut-off
measuring the area of stained cells in tissue samples might
decrease the possibility of bias. Regarding accuracy, despite the
studies reviewed, no actual clinical data have confirmed the
accuracy of DEK in projecting CRC prognosis and comparing it to
other options existing in current clinical settings. However, since
many previous studies have reported the association between DEK and
CRC prognosis, its potential as a prognostic biomarker for CRC
should be pursued through further research.
Additionally, in the clinical setting, given that
CRC is a complex disease with multiple carcinogenic pathways and
several cases found to be associated with inflammation and
autoimmune disorders, we suggest that testing of several biomarkers
involved with DEK may be conducted only to distinguish the role of
DEK in CRC etiology, whether it is dominated by inflammatory or
tumorigenesis and proliferative process (11). The testing of molecules from other
pathways may be more useful in the case of pre-cancer conditions,
such as adenoma or polyps of the colon and rectum, to better
understand the involved pathways of progression from these tumors
to malignancy, whether it be chronic inflammation or proliferation
that trigger DEK overexpression. However, in the clinical context
of determining CRC prognosis, these tests need not be conducted
because there is no further implication of these biomarkers on the
overexpression of DEK and its impact on the (generally worse)
prognosis of CRC.
In the setting of further research, however,
conduction of testing for DEK overexpression along with other
biomarkers related to it and CRC prognosis would be essential to
validate and compare the prognostic accuracy of this biomarker
compared with other, more established biomarkers, such as
microsatellite instability (MSI) status (indicative of good
prognosis), KRAS/neuroblastoma RAS viral oncogene
(NRAS) mutation (poor prognosis), and v-raf murine sarcoma
viral oncogene homolog B1 (BRAF) mutation (poor prognosis,
primarily related to metastasis), for which comparative studies
with DEK and each other are still unknown (44). This knowledge gap can be a great
opportunity for researchers to better understand the role of DEK in
CRC prognosis. Our review also indicates a dearth of literature
regarding comparisons between DEK and other prognostic biomarkers
of CRC or differences in prognosis between CRCs caused by different
pathogenetic pathways. This is a significant knowledge gap that may
provide for excellent research topics in the future.
Based on the present review, the authors found that
DEK has promising potential as a biomarker for CRC prognosis for
several reasons. First, DEK is expressed by cells from all human
body tissues, but its overexpression is linked with cell
proliferation conditions, especially in carcinogenesis (26,41).
Therefore, overexpression of DEK is one of the potential biomarkers
of carcinoma progression in various types of cancer, as the authors
have stated at the outset, including in the colon and rectum.
Second, the involvement of DEK in tumorigenesis and CRC
pathogenesis is quite extensive, where DEK can affect tumorigenesis
from various mechanisms. DEK is also involved in several
inflammatory pathways; thus, it can be used as a biomarker in CRC
cases, given that several CRC cases are associated with
inflammatory bowel diseases (IBD), such as Crohn’s disease and
ulcerative colitis (41,45). Third, as noted by Martinez-Useros
et al (17,18), DEK can be a marker of CRC tissue
response to irinotecan-based and fluoropyrimidine-based CRC
chemotherapy.
5. Conclusions
Summarizing the current literature, several pieces
of evidence show that DEK is a promising prognostic biomarker of
CRC. Overexpression of DEK is related to apoptosis avoidance,
epithelial-mesenchymal transition, and more infiltrative cancer,
and is clinically significantly associated with several clinical
and pathological features of CRC, such as tumor size, lymphatic
node metastasis, serous tissue invasion, and therapy response to
specific chemotherapeutic regimens, generally predicting worse CRC.
It was also proven to predict worse survival. The clear
pathogenesis and clinical association between DEK and worse
features of CRC makes it capable of being used as a biomarker to
predict CRC prognosis in a clinical setting by histopathological
analysis through immunohistochemistry, then quantitative and
semiquantitative analysis of its expression. The role of DEK in the
multiple pathogeneses of CRC and comparison to other prognostic
biomarkers are prime subjects for further research.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable. All cited articles are accessible
through various online research paper databases.
Authors' contributions
MH and MPW conceptualized the review and created the
study methodology. MH, MPW, and SS curated the data, investigated
the studies used in the review, and prepared the original draft of
this review. MH created the visualization aids used in this review.
MH and NR confirmed the validity of the raw data and findings cited
in this review. NR supervised and validated the operation of this
review and approved the final version of the manuscript. All
authors reviewed and edited the draft and read and approved the
final manuscript for publication.
Ethics approval and consent to
participate
Not applicable; the article is a literature
review.
Patient consent for publication
Not applicable; the article is a literature
review.
Competing interests
The authors do not have any conflicts of
interest.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Surveillance Epidemiology, End Results
(SEER) Program: SEER*Stat Database: Incidence - SEER 18 Regs
Research Data.Nov 2015 Sub (1973-2013) - Linked To County
Attributes - Total U.S., 1969-2014 Counties. National Cancer
Institute, DCCPS, Surveillance Research Program, Surveillance
Systems Branch, released April. 2016, based on the November 2015
submission. National Cancer Institute, Maryland, 2016.
|
|
3
|
Barnett A, Cedar A, Siddiqui F, Herzig D,
Fowlkes E and Thomas CR Jr: Colorectal cancer emergencies. J
Gastrointest Cancer. 44:132–142. 2013.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Glynne-Jones R, Brown G, Chau I and Moran
BJ: Colon and rectum. In: UICC Manual of Clinical Oncology. 9th
edition. O'Sullivan B, Brierley J, D'Cruz AK, Fey MF, Pollock R,
Vermorken JB and Huang SH (eds.) John Wiley and Sons, Ltd.,
Chichester West Sussex, UK Hoboken, NJ, pp308-326, 2015.
|
|
5
|
Jessup JM, Goldberg RM, Asare EA, Benson
AB, Brierley JD, Chang GJ, Chen V, Compton CC, De Nardi P, Goodman
KA, et al: Colon and Rectum. In: AJCC Cancer Staging Manual 8th
Edition. Amin MB, Edge SB, Greene FL, Byrd DR, Brookland RK,
Washington MK, Gershenwald JE, Compton CC, Hess KR, Sullivan DC,
et al (eds.) Springer International Publishing, 251-274,
2017.
|
|
6
|
Loughrey MB, Webster F, Arends MJ, Brown
I, Burgart LJ, Cunningham C, Flejou JF, Kakar S, Kirsch R, Kojima
M, et al: Dataset for Pathology Reporting of Colorectal Cancer:
Recommendations From the International Collaboration on Cancer
Reporting (ICCR). Ann Surg. 275:e549–e561. 2022.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kim BH, Kim JM, Kang GH, Chang HJ, Kang
DW, Kim JH, Bae JM, Seo AN, Park HS, Kang YK, et al: Standardized
pathology report for colorectal cancer, 2nd edition. J Pathol
Transl Med. 54:1–19. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Maguire A and Sheahan K: Controversies in
the pathological assessment of colorectal cancer. World J
Gastroenterol. 20:9850–9861. 2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Diamandis EP: Cancer biomarkers: Can we
turn recent failures into success? J Natl Cancer Inst.
102:1462–1467. 2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Duffy MJ, van Dalen A, Haglund C, Hansson
L, Holinski-Feder E, Klapdor R, Lamerz R, Peltomaki P, Sturgeon C
and Topolcan O: Tumour markers in colorectal cancer: European Group
on tumour markers (EGTM) guidelines for clinical use. Eur J Cancer.
43:1348–1360. 2007.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Pease NA, Wise-Draper T and Privette
Vinnedge L: Dissecting the potential interplay of DEK functions in
inflammation and cancer. J Oncol. 2015(106517)2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Holloway S and Peart J: Evidence-based
reviews: Principles and methodological considerations. Wounds UK.
14:26–32. 2018.
|
|
13
|
Siwek J, Gourlay ML, Slawson DC and
Shaughnessy AF: How to write an evidence-based clinical review
article. Am Fam Physician. 65:251–258. 2002.PubMed/NCBI
|
|
14
|
Howick J, Chalmers I, Glasziou P,
Greenhalgh T, Heneghan C, Liberati A, Moschetti I, Phillips B,
Thornton H, Goddard O, et al: The Oxford 2011levels of evidence.
2011. https://www.cebm.net/wp-content/uploads/2014/06/CEBM-Levels-of-Evidence-2.1.pdf.
|
|
15
|
Liu G, Xiong D, Zeng J, Xu G, Xiao R, Chen
B and Huang Z: Prognostic role of DEK in human solid tumors: A
meta-analysis. Oncotarget. 8:98985–98992. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Lin L, Piao J, Gao W, Piao Y, Jin G, Ma Y,
Li J and Lin Z: DEK over expression as an independent biomarker for
poor prognosis in colorectal cancer. BMC Cancer.
13(366)2013.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Martinez-Useros J, Moreno I,
Fernandez-Aceñero MJ, Rodriguez-Remirez M, Borrero-Palacios A,
Cebrian A, Gomez Del Pulgar T, Del Puerto-Nevado L, Li W,
Puime-Otin A, et al: The potential predictive value of DEK
expression for neoadjuvant chemoradiotherapy response in locally
advanced rectal cancer. BMC Cancer. 18(144)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Martinez-Useros J, Rodriguez-Remirez M,
Borrero-Palacios A, Moreno I, Cebrian A, Gomez del Pulgar T, del
Puerto-Nevado L, Vega-Bravo R, Puime-Otin A, Perez N, et al: DEK is
a potential marker for aggressive phenotype and irinotecan-based
therapy response in metastatic colorectal cancer. BMC Cancer.
14(965)2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
You S, Guan Y and Li W:
Epithelial-mesenchymal transition in colorectal carcinoma cells is
mediated by DEK/IMP3. Mol Med Rep. 17:1065–1070. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Babaei-Jadidi R, Li N, Saadeddin A,
Spencer-Dene B, Jandke A, Muhammad B, Ibrahim EE, Muraleedharan R,
Abuzinadah M, Davis H, et al: FBXW7 influences murine intestinal
homeostasis and cancer, targeting Notch, Jun, and DEK for
degradation. J Exp Med. 208:295–312. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lin L, Piao J, Ma Y, Jin T, Quan C, Kong
J, Li Y and Lin Z: Mechanisms underlying cancer growth and
apoptosis by DEK overexpression in colorectal cancer. PLoS One.
9(e111260)2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Witkiewicz AK, Balaji U and Knudsen ES:
Systematically defining single-gene determinants of response to
neoadjuvant chemotherapy reveals specific biomarkers. Clin Cancer
Res. 20:4837–4848. 2014.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Shibata T, Kokubu A, Miyamoto M, Hosoda F,
Gotoh M, Tsuta K, Asamura H, Matsuno Y, Kondo T, Imoto I, et al:
DEK oncoprotein regulates transcriptional modifiers and sustains
tumor initiation activity in high-grade neuroendocrine carcinoma of
the lung. Oncogene. 29:4671–4681. 2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Hu H, Scholten I, Gruss C and Knippers R:
The distribution of the DEK protein in mammalian chromatin. Biochem
Biophys Res Commun. 358:1008–1014. 2007.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Kappes F, Scholten I, Richter N, Gruss C
and Waldmann T: Functional domains of the ubiquitous chromatin
protein DEK. Mol Cell Biol. 24:6000–6010. 2004.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Sandén C and Gullberg U: The DEK
oncoprotein and its emerging roles in gene regulation. Leukemia.
29:1632–1636. 2015.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Waldmann T, Eckerich C, Baack M and Gruss
C: The ubiquitous chromatin protein DEK alters the structure of DNA
by introducing positive supercoils. J Biol Chem. 277:24988–24994.
2002.PubMed/NCBI View Article : Google Scholar
|
|
28
|
von Lindern M, Fornerod M, van Baal S,
Jaegle M, de Wit T, Buijs A and Grosveld G: The translocation
(6;9), associated with a specific subtype of acute myeloid
leukemia, results in the fusion of two genes, dek and can, and the
expression of a chimeric, leukemia-specific dek-can mRNA. Mol Cell
Biol. 12:1687–1697. 1992.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Sitwala KV, Adams K and Markovitz DM: YY1
and NF-Y binding sites regulate the transcriptional activity of the
dek and dek-can promoter. Oncogene. 21:8862–8870. 2002.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lamond AI and Spector DL: Nuclear
speckles: A model for nuclear organelles. Nat Rev Mol Cell Biol.
4:605–612. 2003.PubMed/NCBI View
Article : Google Scholar
|
|
31
|
Kappes F, Khodadoust MS, Yu L, Kim DS,
Fullen DR, Markovitz DM and Ma L: DEK expression in melanocytic
lesions. Hum Pathol. 42:932–938. 2011.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Carro MS, Spiga FM, Quarto M, Ninni VD,
Volorio S, Alcalay M and Müller H: DEK expression is controlled by
E2F and deregulated in diverse tumor type. Cell Cycle. 5:1202–1207.
2006.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Kondoh N, Wakatsuki T, Ryo A, Hada A,
Aihara T, Horiuchi S, Goseki N, Matsubara O and Takenaka K:
Identification and characterization of genes associated with human
hepatocellular carcinogenesis. Cancer Res. 59:4990–4996.
1999.PubMed/NCBI
|
|
34
|
Kroes RA, Jastrow A, McLone MG, Yamamoto
H, Colley P, Kersey DS, Yong VW, Mkrdichian E, Cerullo L, Leestma J
and Moskal JR: The identification of novel therapeutic targets for
the treatment of malignant brain tumors. Cancer Lett. 156:191–198.
2000.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Casas S, Nagy B, Elonen E, Aventín A,
Larramendy ML, Sierra J, Ruutu T and Knuutila S: Aberrant
expression of HOXA9, DEK, CBL and CSF1R in acute myeloid leukemia.
Leuk Lymphoma. 44:1935–1941. 2003.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Han S, Xuan Y, Liu S, Zhang M, Jin D, Jin
R and Lin Z: Clinicopathological significance of DEK overexpression
in serous ovarian tumors. Pathol Int. 59:443–447. 2009.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Privette Vinnedge LM, McClaine R, Wagh PK,
Wikenheiser-Brokamp KA, Waltz SE and Wells SI: The human DEK
oncogene stimulates β-catenin signaling, invasion and mammosphere
formation in breast cancer. Oncogene. 30:2741–2752. 2011.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Privette Vinnedge LM, Benight NM, Wagh PK,
Pease NA, Nashu MA, Serrano-Lopez J, Adams AK, Cancelas JA, Waltz
SE and Wells SI: The DEK oncogene promotes cellular proliferation
through paracrine Wnt signaling in Ron receptor-positive breast
cancers. Oncogene. 34:2325–2336. 2015.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Khodadoust MS, Verhaegen M, Kappes F,
Riveiro-Falkenbach E, Cigudosa JC, Kim DS, Chinnaiyan AM, Markovitz
DM and Soengas MS: Melanoma proliferation and chemoresistance
controlled by the DEK oncogene. Cancer Res. 69:6405–6413.
2009.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Balkwill F and Mantovani A: Inflammation
and cancer: Back to Virchow? Lancet. 357:539–545. 2001.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Karin M and Greten FR: NF-kappaB: Linking
inflammation and immunity to cancer development and progression.
Nat Rev Immunol. 5:749–759. 2005.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Dong X, Wang J, Kabir FN, Shaw M, Reed AM,
Stein L, Andrade LE, Trevisani VF, Miller ML, Fujii T, et al:
Autoantibodies to DEK oncoprotein in human inflammatory disease.
Arthritis Rheum. 43:85–93. 2000.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Saha AK, Kappes F, Mundade A, Deutzmann A,
Rosmarin DM, Legendre M, Chatain N, Al-Obaidi Z, Adams BS, Ploegh
HL, et al: Intercellular trafficking of the nuclear oncoprotein
DEK. Proc Natl Acad Sci USA. 110:6847–6852. 2013.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Koncina E, Haan S, Rauh S and Letellier E:
Prognostic and predictive molecular biomarkers for colorectal
cancer: Updates and challenges. Cancers (Basel).
12(319)2020.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Jawad N, Direkze N and Leedham SJ:
Inflammatory Bowel disease and colon cancer. In: Inflammation and
Gastrointestinal Cancers. Jankowski JAZ (ed.) Springer, Berlin,
Heidelberg, pp99-115, 2011.
|