Introduction
Immune checkpoint inhibitors (ICIs) are used to
treat various malignant tumors, including lung cancer (1). However, the widespread use of ICIs has
also led to reports regarding various immune-related adverse events
(irAEs). Aseptic meningitis is a rare type of irAE (2) that typically responds well to steroid
treatment. Thus, early diagnosis and treatment are important. Given
the rarity of aseptic meningitis, our experience with a patient who
developed this irAE during ICI treatment for non-small cell lung
cancer (NSCLC) is reported herein and the related literature was
reviewed.
Case report
In August 2018, a 67-year-old Japanese man with a
history of asthma and chronic obstructive pulmonary disease was
referred to our hospital due to chest pain. Chest radiography
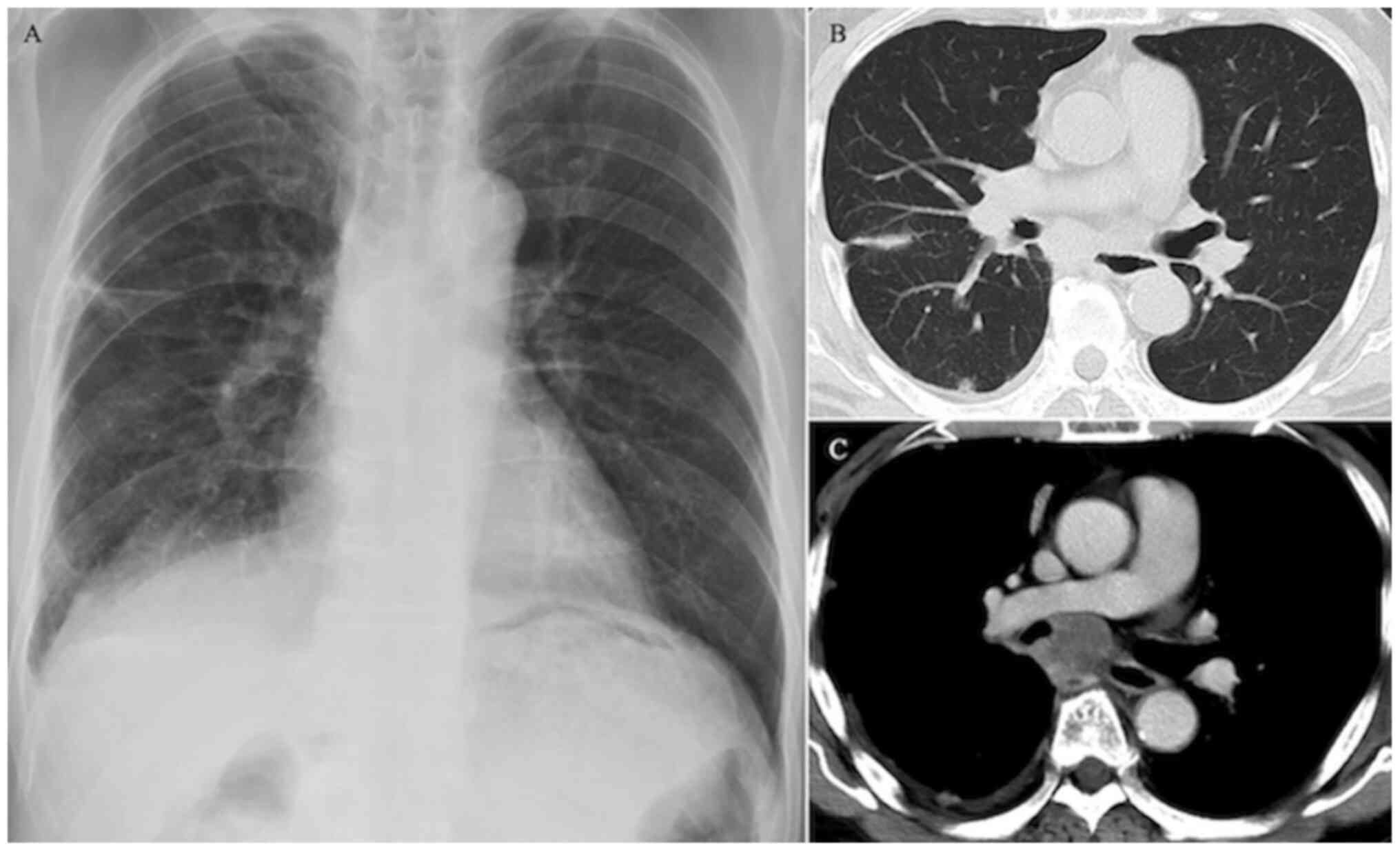
revealed linear opacity in the right middle lung field (Fig. 1A). A chest CT scan revealed a 27-mm
tumor in the subcarinal space, pleural invasion in the right upper
and middle lung segments (Fig. 1B)
and a hilar mass shadow with contrast enhancement along the right
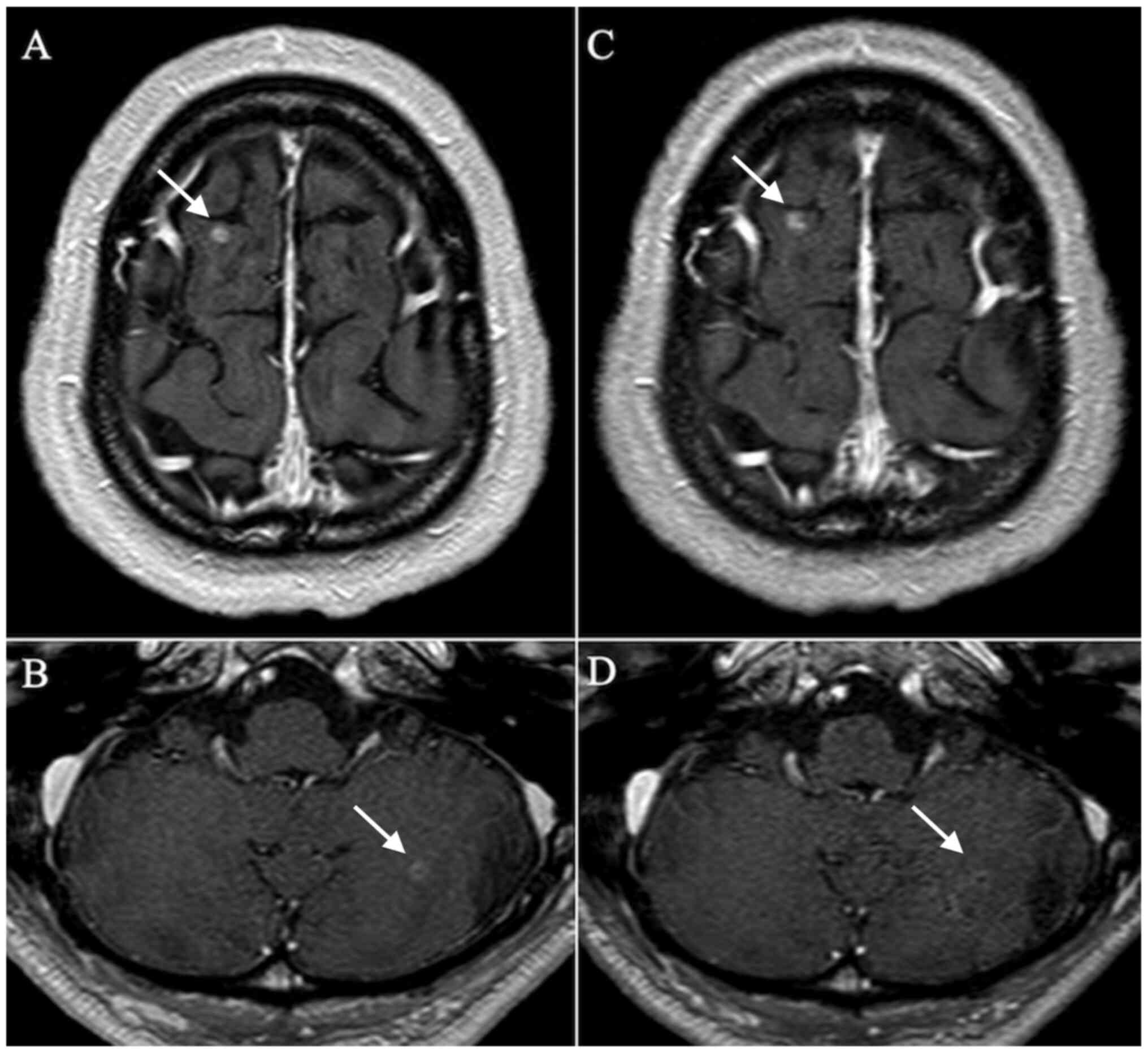
main bronchus from below the tracheal bifurcation (Fig. 1C). A brain contrast-enhanced MRI
revealed two small nodules in the cerebellum and cerebrum (Fig. 2A and B). Fluorodeoxyglucose (FDG) positron
emission tomography with CT revealed intense FDG accumulation in
the subcarinal space and pleural nodule. Histological examination
of the tumor in the transbronchial lung biopsy specimens from the
subcarinal space revealed adenocarcinoma, with a tumor proportion
score of 100% for programmed death-ligand (PD-L1) using a PD-L1
antibody (clone 22C3; Agilent Technologies, Inc.) and no expression
of epidermal growth factor receptor (EGFR) mutation, anaplastic
lymphoma kinase (ALK) fusion, ROS proto-oncogene 1, receptor
tyrosine kinase (ROS1) fusion and B-Raf proto-oncogene,
serine/threonine kinase (BRAF) mutation. Based on these findings,
the patient was diagnosed with stage IVB lung adenocarcinoma
(TxN2M1c, brain pleura).
Oxycodone hydrochloride hydrate tablet (10 mg/day)
was administered for the chest pain that was caused by the pleural
lesions, and γ-knife irradiation was performed for the two
metastatic brain lesions. Six days later, the patient started
treatment using pembrolizumab (200 mg, once every 3 weeks).
However, on day 2 after starting pembrolizumab treatment, the
patient developed persistent nausea and was admitted to our
department on day 5 to identify the cause of the nausea. A physical
examination showed no fever, Kernig's sign, Brudzinski's sign and a
stiff neck, other than tachycardia, and no altered mental status.
Blood tests revealed a slight increase in C-reactive protein (CRP)
concentration (0.74 mg/dl), but no other abnormalities in
electrolyte concentrations or endocrine function were observed.
Contrast-enhanced abdominal CT showed only a slight thickening of
the stomach wall and contrast effects, without other new findings,
and upper gastrointestinal endoscopy showed no obstructive or
bleeding lesions, only atrophic gastritis, which was insufficient
to identify the cause of the nausea. The nausea was therefore
attributed to the opioid treatment, and opioid rotation (from
oxycodone hydrochloride hydrate tablet 30 mg/day to fentanyl
continuous infusion 0.6 mg/day) was performed. The nausea appeared
to improve by day 9 after starting pembrolizumab treatment, but
subsequently worsened on day 15. Brain contrast-enhanced MRI
revealed no tumor progression, no occurrence of new tumors and no
subcranial enhancement, suggesting meningitis (Fig. 2C and D). Lumbar puncture was thus performed
(Table I). Laboratory test results,
including PCR findings, revealed a normal glucose concentration and
negative results for the cerebrospinal fluid (CSF) smear, bacteria,
fungi, mycobacteria and tuberculosis. However, the total cell count
was slightly elevated, with an increased subset of lymphocytes.
Pathological findings revealed lymphocytic inflammation and an
elevated level of adenosine deaminase (ADA), which suggested the
presence of lymphocyte proliferation and differentiation.
Furthermore, at the same time as the nausea recurrence, the patient
had an elevated IgG index and various symptoms that supported
suspicion of an irAE, including a rash, liver enzyme elevation and
destructive thyroiditis. Aseptic meningitis was therefore
considered as an irAE for differential diagnosis. However, despite
the negative brain MRI findings, meningeal carcinomatosis was also
considered for differential diagnosis, due to the patient's history
of metastatic brain tumors. Therefore, treatment was started using
betamethasone (4 mg) to potentially manage meningitis as an irAE,
and as a palliative treatment for meningeal carcinomatosis. The
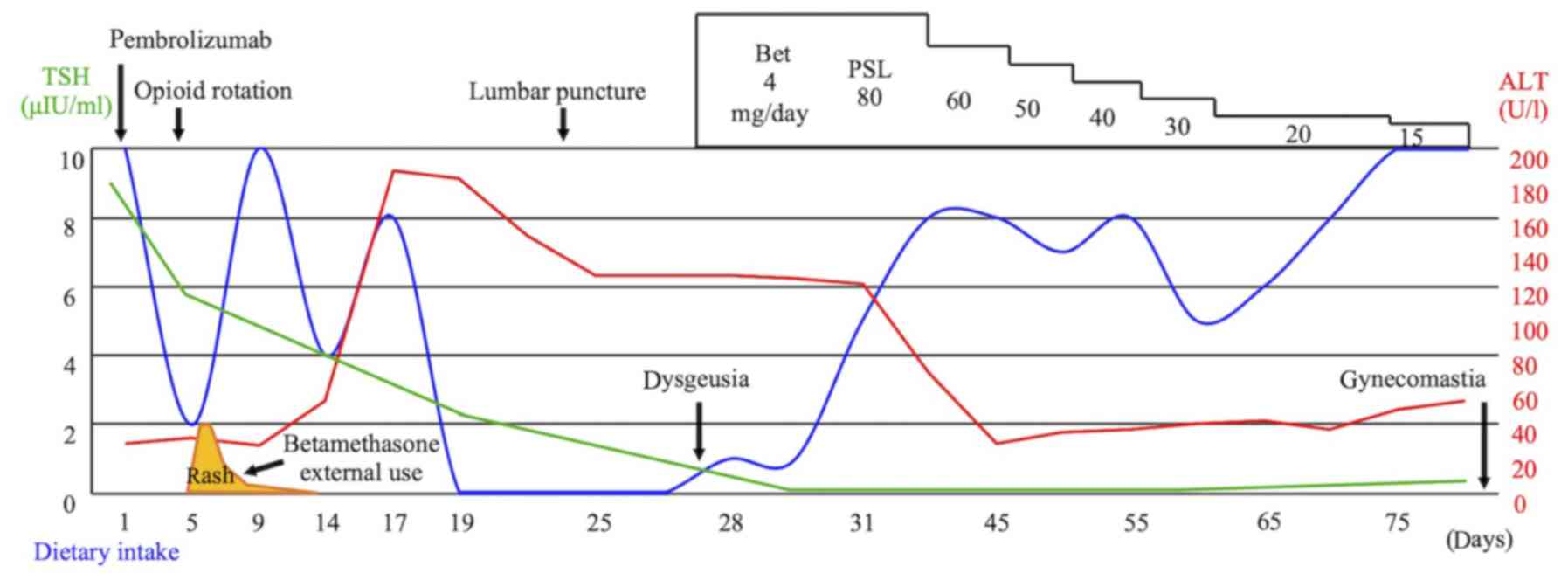
patient experienced immediate improvement of the nausea after
starting steroid treatment (Fig.
3). The cytology findings were also negative for malignant
cells, so the case was diagnosed as pembrolizumab-induced aseptic
meningitis (grade 3). The betamethasone treatment was changed to
prednisolone (80 mg at 1 mg/kg) for long-term treatment, with the
dose tapering from 80 to 60 mg, then from 60 to 20 mg in 10-mg
increments every 5 days, and finally from 20 to 10 mg in 5-mg
increments every 2 weeks. The patient was discharged on day 75 of
hospitalization, and steroid treatment was terminated on day 83
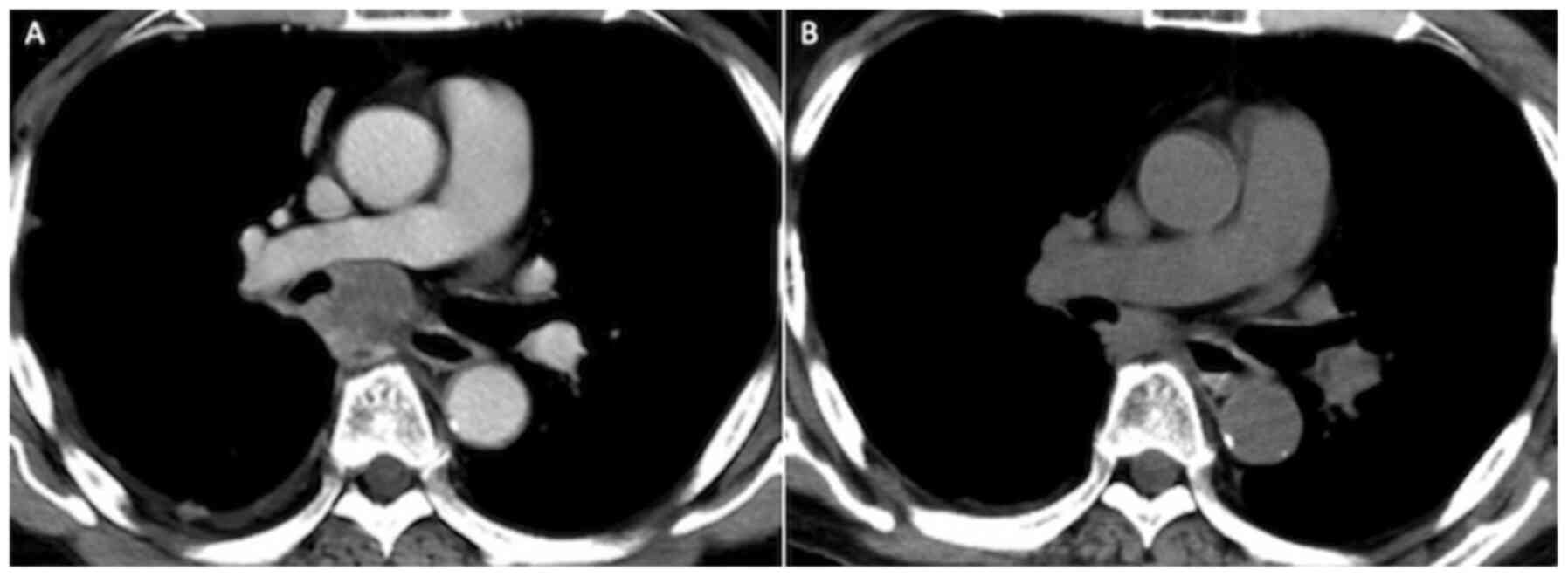
after steroid treatment initiation. The tumor response was judged
to be a partial response (Fig. 4)
at the patient's discharge, although ICI re-challenge was not
attempted based on the potential risk of other irAEs. The patient
experienced disease progression 4 months later, which presented as
an enlargement of the tumor in the subcarinal space and was treated
using second-line carboplatin plus pemetrexed.
 | Table ICerebrospinal fluid analysis. |
Table I
Cerebrospinal fluid analysis.
| Test | Result | Test | Result | Test | Result |
|---|
| Color | Colorless | Total protein
(mg/dl) | 80 | Bacteria | Negative |
| Turbidity | Clear | Albumin
(mg/dl) | 41 | Fungi | Negative |
| Total cell count
(/µl) | 12 | LDH (U/l) | 22 | Mycobacteria | Negative |
| Polynuclear
(%) | 8 | Glucose
(mg/dl) | 61 | Tb-PCR | Negative |
| Mononuclear
(%) | 92 | CRP (µg/dl) | <1 | MAC-PCR | Negative |
| Open pressure
(cmH2O) | 10 | CEA (ng/dl) | <0.8 | Pathology | No malignant
cells |
| | | ADA | 3.1 | | Lymphocyte
infiltration |
| | | IgG (mg/dl) | 11.5 | | |
| | | IgG index | 0.64 | | |
Discussion
Treatment with immune checkpoint inhibitors (ICIs)
can be used for various types of cancer, including renal cell
carcinoma, melanoma, head and neck cancers, urothelial carcinoma
and Hodgkin lymphoma (3).
Furthermore, ICIs are an effective option for non-small cell lung
cancer (NSCLC) treatment, along with chemotherapy and targeted
therapy (1). Certain patients can
experience long-term response to ICI treatment, which has less
toxicity than chemotherapy (4).
Recent studies have also combined ICIs with chemotherapy (5,6),
utilized ICIs in maintenance therapy following chemoradiotherapy
(7) or have used ICIs for small
cell lung cancer treatment (8,9).
However, while ICIs are effective, safe and increasingly used, they
are also associated with a risk of immune-related adverse events
(irAEs), which can cause treatment interruption and reduce the
quality of life of these patients. The reported frequencies of
irAEs grade ≥3 are 8% for nivolumab, 5-10% for pembrolizumab, 5-7%
for atezolizumab, 2% for durvalumab and 15-42% for ipilimumab
(3). Grade 3-4 neurological irAEs
are uncommon (<1%), but include inflammatory myopathies,
myasthenia gravis, neuropathies, multiple sclerosis, autoimmune
encephalitis and aseptic meningitis.
Head jolt sign, Kernig's sign, Brudzinski's sign and
stiff neck are well-known signs of meningitis, although only 29% of
cases of aseptic meningitis involve these symptoms (10). Numerous other cases are associated
with non-specific symptoms, such as headache, nausea, and vomiting
(10). The frequency of aseptic
meningitis as an irAE is thought to be 0.1-0.2% (2), although the underdiagnosis of this
rare irAE may be associated with the manifestation of flu-like
symptoms, headaches (11) or other
mild symptoms. Head contrast-enhanced MRI rarely produces
significant findings, and cerebrospinal fluid (CSF) examination is
therefore necessary. Moreover, the diagnosis of irAE must exclude
other diseases, such as bacterial, fungal, mycobacterial and viral
infections, as well as cancerous meningitis. The pathophysiological
development of irAEs is associated with the ICI-induced activation
of CD8 T-cells (12), and an
increased proportion of lymphocytes in the CSF is useful for
diagnosing irAEs (2). Table II shows that an elevation in CSF
lymphocytes was observed in 12 cases. In addition, adenosine
deaminase (ADA) is a well-known marker for tuberculous meningitis
(CSF ADA levels of >8 U/l provide a sensitivity of <59% and
specificity of >96%) (13), as
well as a marker of cellular immunity, based on its relationship
with lymphocyte proliferation and differentiation (12). Therefore, elevated ADA levels may
reflect a CD8 T-cell-related irAE, based on the elevated levels
being detected in patients who received ICIs and developed
meningoencephalitis (14) and
autoimmune encephalitis (12).
Therefore, lymphocyte-related inflammatory findings (for example,
elevated lymphocytes or ADA in the CSF) may be useful for
diagnosing aseptic meningitis as an irAE. In the setting of
elevated ADA, tuberculous cultures, PCR findings or CSF/serum
glucose ratio (<0.5 in 95% of tuberculous meningitis cases) are
useful for distinguishing aseptic meningitis from tuberculous
meningitis (15). The present
patient exhibited an elevated lymphocyte percentage and
pathological findings suggestive of lymphocytic inflammation, but
no marked increase in ADA level. This may be associated with the
relatively mild clinical symptoms and the unremarkable increase in
the CSF total cell count. In this setting, the IgG index
[(CSF-IgG/CSF-albumin)/(serum-IgG/serum-albumin)] may reflect
increased IgG synthesis in the central nervous system, which could
indicate the presence of multiple sclerosis or central nervous
system infections, such as encephalitis or meningitis (16). A previous report described an
increased IgG index in nivolumab-induced encephalopathy, which
might have been caused by nivolumab promoting IgG release as B
cells were converted to plasma cells (17). Thus, ICI treatment may lead to an
increased IgG index in cases involving central nervous system
disorders.
 | Table IIReported cases of meningitis as an
immune-related adverse event after immune checkpoint inhibitor
treatment. |
Table II
Reported cases of meningitis as an
immune-related adverse event after immune checkpoint inhibitor
treatment.
| Patient no./
Investigators (Refs.) | Age (years) | Sex | Cancer | ICIs | Cycle | Initial
symptoms | CSF lymph
ocytes | MRI abnormal | Other irAEs | Treatment | Time to
resolution | Tumor Response | Re-challenge
ICIs |
|---|
| #1/Spain et
al (19) | N/A | N/A | Mela | I+N | 1 | Headache,
nausea | + | - | Hepatitis | - | 7 weeks | PR | After 4 weeks |
| #2/Spain et
al (19) | N/A | N/A | Mela | I | 2 | Headache,
drowsiness, nausea, vomiting | + | - | - | - | 10 days | SD | - |
| #3/Spain et
al (19) | N/A | N/A | Mela | I | 2 | Delirium | - | - | - | PSL p.o | 8 weeks | PD | - |
| #4/Bot et al
(20) | 51 | F | Mela | I | 1 | Headache,
fever | + | - | N/A | Dex 8 mg p.o | 2 days | N/A | N/A |
| #5/Voskens et
al (21) | 52 | F | Mela | I | 1 | Nausea, vomiting,
chills, rash | + | - | N/A | DEX | N/A | PD | - |
| #6/Bompaire et
al (22) | 56 | M | Mela | I | 4 | Vertigo, dizziness,
cervicalgia, headache | + | Brain-/
spinal+ | Radiculo
neuritis | mPSL 1g IV +
IVIg | 2 years | CR | - |
| #7/Yang et
al (23) | N/A | N/A | Renal | I | 4 | Headache,
photophobia, cranial nerve disorder | + | - | N/A | DEX | <1 month | N/A | N/A |
| #8/Takamatsu et
al (24) | 70 | F | Renal | I+N | 2 | Headache, nausea,
dizziness | + | - | Isolated ACTH
deficiency | PSL 1 mg/kg IV | A few days | CR | 50th day with PSL
10 mg/day |
| #9/Takamatsu et
al (24) | 70 | F | Renal | I+N | 3 | Headache,
anorexia | + | N/A | Liver
dysfunction | PSL 1 mg/kg IV | N/A | CR | - |
| #10/Cordes et
al (25) | 58 | M | UC | N | 12 | Fever, chills,
malaise, dry cough, headache, bilateral eye pain, right ear
pain | + | + | N/A | mPSL 1 mg/kg
IV | <3 days | PR | - |
| #11/Toyozawa et
al (26) | 71 | F | NSCLC | A | 1 | Fever,
consciousness disorder | - | - | N/A | mPSL 1 mg/kg
IV | 1 day | N/A | - |
| #12/Toyozawa et
al (26) | 55 | M | NSCLC Ade | A | 1 | Fever,
consciousness disorder | - | - | N/A | mPSL 1 mg/kg
IV | 4 days | N/A | - |
| #13/Toyozawa et
al (26) | 50 | M | NSCLC Ade | A | 1 | Fever, neck and
legs pain, consciousnesss disorder | + | + | N/A | mPSL 1 mg/kg
IV | 2 days | N/A | - |
| #14/Lima et
al (27) | 55 | M | NSCLC Ade | P | 11 | Bilateral
throbbing, frontal headache | + | - | Hepatitis | Dex 10 mg IV | 1 day | CR | - |
| #15/Present
case | 67 | M | NSCLC Ade | P | 1 | Nausea,
vomiting | + | - | Hepatitis,
dysgeusia, gynecomastia thyroid dysfunction | PSL 1 mg/kg IV | 3 days | PR | - |
To distinguish between cancerous and aseptic
meningitis as an irAE is challenging. The sensitivity of CSF
puncture for cancerous meningitis is 50-60% for a single dose and
80% for multiple doses (18); thus
we cannot completely deny cancerous meningitis. Actually, the
patient's general condition was not good, thus we judged that
repeat lumbar puncture was risky. We should have examined after the
recovery condition. In the present case, aseptic meningitis was
diagnosed as an irAE based on the following findings: no atypical
cells detected in the pathology and increased number of cells, high
IgG index, low carcinoembryonic antigen (CEA) and other concurrent
irAEs. Subsequent follow-up revealed no findings suggestive of
meningeal carcinomatosis or brain metastasis recurrence. In
addition, the symptoms may have appeared due to the effect after
the γ-knife irradiation; we judged that the effect was small,
because the head lesions were micronodules and no edema was
observed on MRI following irradiation.
Table II summarizes
the 15 reported cases of aseptic meningitis as an irAE following
treatment with nivolumab, pembrolizumab, atezolizumab or ipilimumab
(19-27).
Nine cases were associated with ipilimumab treatment, which was
administered for melanoma in 6 cases (19-22)
and renal cell carcinoma in 3 cases (23,24).
Three cases were treated with a combination of ipilimumab and
nivolumab (19,24), and cases 8 and 9 involved the same
patient, who experienced meningitis relapse following
re-administration of an ICI (24).
The majority of patients, except cases 7, 10 and 11, developed
aseptic meningitis after 1-4 cycles of ICI treatment. Seven cases
involved neurological symptoms (19,22,23,26,27).
By contrast, most patients initially exhibited non-specific
symptoms, such as headache or nausea. All patients underwent brain
MRI and lumbar puncture, which revealed lymphocyte-dominated
leukocyte elevation in 13 patients. However, the brain
contrast-enhanced MRI failed to reveal abnormal findings in most
cases; the only two findings were arachnoiditis during spinal MRI
(22) and abnormal enhancements
along the lines of the corpus callosum (26). A total of 13 patients received
steroid treatment, while a follow-up observation was only performed
in 2 patients (19). The steroid
treatment was ineffective for case 6, and intravenous
immunoglobulin (IVIg) was therefore administered in this case
(22). All patients ultimately
experienced an improvement in their symptoms. The tumor responses
were progressive disease in 2 cases (19,21),
stable disease in 1 case (19),
partial response in 3 cases (19,23,25),
and complete response in 3 cases (melanoma, renal cell carcinoma
and NSCLC) (22,24,27).
Two patients were treated again with ICIs (19,24)
and 1 patient experienced relapse of meningitis (24).
Several reports have described ipilimumab-induced
meningitis, which may be associated with the drug's affinity for
the cranial nervous system. Indeed, ipilimumab frequently induces
hypophysitis, which is thought to be caused by the expression of
CTLA-4 in the anterior pituitary cells and a resulting type II
hypersensitivity (28). There are
only a few reports of pembrolizumab-induced meningitis (27) and meningoencephalitis (14), although the increasing use of this
drug will presumably result in more cases being reported.
The main treatment for neurological irAEs involves
immunosuppression, with prednisone or methylprednisolone being
recommended for CTCAE grade 3 or higher meningitis (3). Aseptic meningitis as an irAE has a
good neurological prognosis, as it typically responds well to
steroid treatment (11).
Furthermore, steroid use is not associated with reduced overall
survival or time to ICI treatment failure (29). Steroid monotherapy is often
effective in this setting, although select patients may require
IVIg or plasma exchange (19).
Natalizumab is a monoclonal antibody that targets α4 integrin
(30) and might be effective in
treating central nervous system symptoms of neurological irAEs, as
it inhibits the transfer of lymphocytes to the central nervous
system by inhibiting the binding of lymphocyte integrins and VCAM-1
in the blood-brain barrier (30). A
previous report has indicated that, while steroid treatment was
ineffective, natalizumab was effective in treating autoimmune
encephalitis as an irAE, which was caused by the combination of
ipilimumab and nivolumab (31).
A prospective cohort study of 43 patients with
advanced NSCLC revealed that patients with irAEs after nivolumab
treatment had a higher objective response rate (37 vs. 17%) and
longer median progression-free survival (6.4 vs. 1.5 months)
(32). Of note, autoimmune
encephalitis as an irAE was also reportedly associated with an
increased response to pembrolizumab in NSCLC cases (33). It was therefore suspected that a
similar positive response may be observed in cases with central
nervous system irAEs (for example, meningitis). In addition to the
present case, 2 NSCLC cases had a good response rate to
pembrolizumab.
While repeat treatment using ICIs may be an
effective strategy for cases of aseptic meningitis as an irAE, it
is associated with a risk of meningitis relapse (24). A previous study suggested that the
risk-reward ratio for anti-PD or anti-PD-L1 re-challenge appeared
to be acceptable, although there was no evidence of prolonged
progression-free survival or overall survival outcomes (34). In the present case, platinum-based
treatment was selected for the recurrence, based on the presence of
other irAEs and the patient's opinion.
In conclusion, the present study reported a case of
nausea and vomiting that was ultimately diagnosed as aseptic
meningitis, as an irAE related to pembrolizumab treatment. Although
there are a few reports of meningitis as an irAE, this condition
may be underdiagnosed due to its non-specific symptoms. The
majority of patients will experience a good response to steroid
treatment, and a good response rate for ICIs has been observed in
patients with irAEs. A CSF examination is required to diagnose
aseptic meningitis as an irAE, which may be supported based on
lymphocytic inflammation findings, such as elevated lymphocyte or
ADA values. Since there are few reports of aseptic meningitis after
ICIs for lung cancer, an accumulation of further case reports is
desired to discern whether re-administration of ICIs is
acceptable.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
GI collected the data and wrote the paper. YF
treated the patient and revised the article for important
intellectual content as the corresponding author. HM and HT treated
the patient and provided advice on the paper. KA, KK, YuukiH, RK,
TN, KoheiY, YT, YS, TS and KosukeY collected, analyzed and
interpreted the clinical data. SK collected, analysed and
interpreted the neurological findings and provided advice on the
paper. YU, YasushiH and KN analyzed and interpreted the
pathological data and provided advice on the paper. MK and AY
critically revised the manuscript for important intellectual
content. All authors read and approved the manuscript and agree to
be accountable for all aspects of the research in ensuring that the
accuracy or integrity (including the collected data) of any part of
the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
This is to certify that the above case report was
approved for publication by Tottori University Ethics Review Board
(serial no. 22J002).
Patient consent for publication
As the patient was deceased, according to hospital
policy, request to publish the case report was published online,
and as no contest was made by any family member or other person,
the hospital provided approval to proceed with publication of the
case study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ettinger DS, Wood DE, Aggarwal C, Aisner
DL, Akerley W, Bauman JR, Bharat A, Bruno DS, Chang JY, Chirieac
LR, et al: NCCN guidelines insights: Non-small cell lung cancer,
version 1.2020. J Natl Compr Canc Netw. 17:1464–1472.
2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dalakas MC: Neurological complications of
immune checkpoint inhibitors: What happens when you ‘take the
brakes off’ the immune system. Ther Adv Neurol Disord.
11(1756286418799864)2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Brahmer JR, Lacchetti C, Schneider BJ,
Atkins MB, Brassil KJ, Caterino JM, Chau I, Ernstoff MS, Gardner
JM, Ginex P, et al: Management of immune-related adverse events in
patients treated with immune checkpoint inhibitor therapy: American
society of clinical oncology clinical practice guideline. J Clin
Oncol. 36:1714–1768. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Reck M, Rodriguez-Abreu D, Robinson AG,
Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe
S, et al: Pembrolizumab versus chemotherapy for PD-L1-positive
non-small-cell lung cancer. N Engl J Med. 375:1823–1833.
2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Socinski MA, Jotte RM, Cappuzzo F, Orlandi
F, Stroyakovskiy D, Nogami N, Rodriguez-Abreu D, Moro-Sibilot D,
Thomas CA, Barlesi F, et al: Atezolizumab for first-line treatment
of metastatic nonsquamous NSCLC. N Engl J Med. 378:2288–2301.
2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Gandhi L, Rodriguez-Abreu D, Gadgeel S,
Esteban E, Felip E, De Angelis F, Domine M, Clingan P, Hochmair MJ,
Powell SF, et al: Pembrolizumab plus chemotherapy in metastatic
non-small-cell lung cancer. N Engl J Med. 378:2078–2092.
2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Antonia SJ, Villegas A, Daniel D, Vicente
D, Murakami S, Hui R, Yokoi T, Chiappori A, Lee KH, de Wit M, et
al: Durvalumab after chemoradiotherapy in stage III non-small-cell
lung cancer. N Engl J Med. 377:1919–1929. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Ready N, Farago AF, de Braud F, Atmaca A,
Hellmann MD, Schneider JG, Spigel DR, Moreno V, Chau I, Hann CL, et
al: Third-line nivolumab monotherapy in recurrent SCLC: CheckMate
032. J Thorac Oncol. 14:237–244. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Horn L, Mansfield AS, Szczesna A, Havel L,
Krzakowski M, Hochmair MJ, Huemer F, Losonczy G, Johnson ML, Nishio
M, et al: First-line atezolizumab plus chemotherapy in
extensive-stage small-cell lung cancer. N Engl J Med.
379:2220–2229. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Waghdhare S, Kalantri A, Joshi R and
Kalantri S: Accuracy of physical signs for detecting meningitis: A
hospital-based diagnostic accuracy study. Clin Neurol Neurosurg.
112:752–757. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Blackmon JT, Viator TM and Conry RM:
Central nervous system toxicities of anti-cancer immune checkpoint
blockade. J Neurol Neuromedicine. 1:39–45. 2016.
|
|
12
|
Fujiwara S, Mimura N, Yoshimura H,
Fujimoto D, Ito M, Mori R, Ito J, Tomii K, Kawamoto M and Kohara N:
Elevated adenosine deaminase levels in the cerebrospinal fluid in
immune checkpoint inhibitor-induced autoimmune encephalitis. Intern
Med. 58:2871–2874. 2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Tuon FF, Higashino HR, Lopes MI, Litvoc
MN, Atomiya AN, Antonangelo L and Leite OM: Adenosine deaminase and
tuberculous meningitis-a systematic review with meta-analysis.
Scand J Infect Dis. 42:198–207. 2010.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yonenobu Y, Ishijima M, Toyooka K and
Fujimura H: A case of meningoencephalitis associated with
pembrolizumab treated for squamous cell lung cancer. Rinsho
Shinkeigaku. 59:105–108. 2019.PubMed/NCBI View Article : Google Scholar : (In Japanese).
|
|
15
|
Thwaites GE and Hien TT: Tuberculous
meningitis: Many questions, too few answers. Lancet Neurol.
4:160–170. 2005.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Akaishi T, Narikawa K, Suzuki Y, Mitsuzawa
S, Tsukita K, Kuroda H, Nakashima I, Fujihara K and Aoki M:
Importance of the quotient of albumin, quotient of immunoglobulin G
and Reibergram in inflammatory neurological disorders with
disease-specific patterns of blood-brain barrier permeability.
Neurol Clin Neurosci. 3:94–100. 2015.
|
|
17
|
Leitinger M, Varosanec MV, Pikija S, Wass
RE, Bandke D, Weis S, Studnicka M, Grinzinger S, McCoy MR, Hauer L
and Sellner J: Fatal necrotizing encephalopathy after treatment
with nivolumab for squamous non-small cell lung cancer: Case report
and review of the literature. Front Immunol. 9(108)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Glantz MJ, Cole BF, Glantz LK, Cobb J,
Mills P, Lekos A, Walters BC and Recht LD: Cerebrospinal fluid
cytology in patients with cancer: Minimizing false-negative
results. Cancer. 82:733–739. 1998.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Spain L, Walls G, Julve M, O'Meara K,
Schmid T, Kalaitzaki E, Turajlic S, Gore M, Rees J and Larkin J:
Neurotoxicity from immune-checkpoint inhibition in the treatment of
melanoma: A single centre experience and review of the literature.
Ann Oncol. 28:377–385. 2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Bot I, Blank CU, Boogerd W and Brandsma D:
Neurological immune-related adverse events of ipilimumab. Pract
Neurol. 13:278–280. 2013.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Voskens CJ, Goldinger SM, Loquai C, Robert
C, Kaehler KC, Berking C, Bergmann T, Bockmeyer CL, Eigentler T,
Fluck M, et al: The price of tumor control: An analysis of rare
side effects of anti-CTLA-4 therapy in metastatic melanoma from the
ipilimumab network. PLoS One. 8(e53745)2013.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Bompaire F, Mateus C, Taillia H, De
Greslan T, Lahutte M, Sallansonnet-Froment M, Ouologuem M, Renard
JL, Gorochov G, Robert C and Ricard D: Severe
meningo-radiculo-neuritis associated with ipilimumab. Invest New
Drugs. 30:2407–2410. 2012.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yang JC, Hughes M, Kammula U, Royal R,
Sherry RM, Topalian SL, Suri KB, Levy C, Allen T, Mavroukakis S, et
al: Ipilimumab (anti-CTLA4 antibody) causes regression of
metastatic renal cell cancer associated with enteritis and
hypophysitis. J Immunother. 30:825–830. 2007.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Takamatsu D, Furubayashi N, Negishi T,
Ieiri K, Inoue T, Tsukino K and Nakamura M: Relapse of aseptic
meningitis induced by ipilimumab and nivolumab therapy for
metastatic renal cell carcinoma: A case report. Mol Clin Oncol.
11:590–594. 2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Cordes LM, Davarpanah NN, Reoma LB, Gasmi
B, Quezado M, Khan OI, Nath A and Apolo AB: Neurotoxicities
associated with checkpoint inhibitors: Two case reports and a
review of the literature. Clin Case Rep. 8:24–32. 2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Toyozawa R, Haratake N, Toyokawa G,
Matsubara T, Takamori S, Miura N, Yamaguchi M, Takenoyama M and
Seto T: Atezolizumab-induced aseptic meningitis in patients with
NSCLC. JTO Clin Res Rep. 1(100012)2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Lima G, Kahn A, Sama S and Savage J:
Aseptic meningitis as an immune-related adverse event after
pembrolizumab. Case Rep Oncol Med. 2019(7183747)2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Tadano H and Torigoe T: Immune-related
adverse events of immune checkpoint inhibitors. Nihon Rinsho Meneki
Gakkai Kaishi. 40:102–108. 2017.PubMed/NCBI View Article : Google Scholar : (In Japanese).
|
|
29
|
Horvat TZ, Adel NG, Dang TO, Momtaz P,
Postow MA, Callahan MK, Carvajal RD, Dickson MA, D'Angelo SP, Woo
KM, et al: Immune-related adverse events, need for systemic
immunosuppression, and effects on survival and time to treatment
failure in patients with melanoma treated with ipilimumab at
memorial sloan kettering cancer center. J Clin Oncol. 33:3193–3198.
2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Polman CH, O'Connor PW, Havrdova E,
Hutchinson M, Kappos L, Miller DH, Phillips JT, Lublin FD,
Giovannoni G, Wajgt A, et al: A randomized, placebo-controlled
trial of natalizumab for relapsing multiple sclerosis. N Engl J
Med. 354:899–910. 2006.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Hottinger AF, de Micheli R, Guido V,
Karampera A, Hagmann P and Du Pasquier R: Natalizumab may control
immune checkpoint inhibitor-induced limbic encephalitis. Neurol
Neuroimmunol Neuroinflamm. 5(e439)2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Teraoka S, Fujimoto D, Morimoto T, Kawachi
H, Ito M, Sato Y, Nagata K, Nakagawa A, Otsuka K, Uehara K, et al:
Early immune-related adverse events and association with outcome in
advanced non-small cell lung cancer patients treated with
nivolumab: A prospective cohort study. J Thorac Oncol.
12:1798–1805. 2017.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Niki M, Nakaya A, Kurata T, Nakahama K,
Yoshioka H, Kaneda T, Kibata K, Ogata M and Nomura S:
Pembrolizumab-induced autoimmune encephalitis in a patient with
advanced non-small cell lung cancer: A case report. Mol Clin Oncol.
10:267–269. 2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Simonaggio A, Michot JM, Voisin AL, Le
Pavec J, Collins M, Lallart A, Cengizalp G, Vozy A, Laparra A,
Varga A, et al: Evaluation of readministration of immune checkpoint
inhibitors after immune-related adverse events in patients with
cancer. JAMA Oncol. 5:1310–1317. 2019.PubMed/NCBI View Article : Google Scholar
|