Introduction
Colorectal cancer is the third most common
malignancy worldwide and the second most common cause of
cancer-related mortality (1).
Global projections predict an increasing number of colorectal
cancer cases with an alarming trend of rising incidence among
younger adults (2). Advances in
understanding of the pathophysiology of the disease has increased
the available treatment options for local and advanced disease
(3). The 5-year survival rate has
risen over the past several decades and exceeds 60% (4). As more prognostic and predictive
factors related to the tumor biology, including tumor sidedness,
RAS and BRAF mutations and microsatellite instability (MSI), are
validated, it is becoming increasingly evident that colorectal
cancer is a highly heterogeneous disease with different outcomes
across patient subgroups (5).
Further research into prognostic markers is warranted to guide
appropriate treatment decisions.
The translationally controlled tumor protein (TCTP)
is a highly conserved protein present in virtually all eukaryotic
organisms (6). TCTP is involved in
a variety of normal cell functions and disease processes. TCTP
levels are upregulated in mitotically active tissues, which implies
a crucial role in cell growth and proliferation (7). Various growth signals and cytokines
induce TCTP synthesis (8,9). The structural similarity to specific
chaperone proteins, the ability to affect microtubule dynamics and
cell morphology and its anti-apoptotic properties suggest a
cytoprotective function of TCTP (10-12).
Increasing evidence supports an association between
TCTP and oncogenic transformation. TCTP expression levels tend to
be higher in tumors compared with the corresponding normal tissue
(12,13). TCTP was found to bind the tumor
suppressor p53 and repress its transcription thus preventing
apoptosis in tumor cells and promoting malignant transformation
(14,15). Inhibition of TCTP expression
suppresses the malignant phenotype in cancer cells (13) and renders them more sensitive to
cell death due to oxidative and metabolic stress (16). In addition, increased TCTP levels
are associated with the chemoresistance of cancer cells (17,18).
TCTP is found to be involved in cancer progression via inhibition
of apoptosis, acceleration of mitotic exit and induction of
invasion and metastasis (19).
Tumor reversion is the process in which tumor cells
lose their malignant phenotype (20). During the process of tumor
reversion, a significant downregulation of TCTP has been
demonstrated, which increases the interest in TCTP as a potential
therapeutic target (21).
Knockdown of TCTP in colon cancer cells inhibits invasion and
migration in vitro and liver metastasis in vivo
(22).
TCTP is overexpressed in various types of cancers
(23,24). TCTP expression is identified in the
cytoplasm, the nuclei and extracellularly (25,26).
The prognostic value of TCTP expression has not been extensively
investigated (27). In colorectal
cancer, high TCTP expression is associated with a higher
pathological grade and a metastatic stage at diagnosis but little
evidence is present about survival outcomes (26,27).
The present retrospective study evaluated the
performance of TCTP expression in the primary tumor as a prognostic
marker for patients with colon cancer.
Materials and methods
Patient selection
A retrospective analysis of the UMHAT Sveta Marina
Clinic of Medical Oncology (Varna, Bulgaria) database was
conducted. All consecutive colon cancer patients who were diagnosed
and treated between 1 January 2015 and 31 December 2015 and met the
following predefined eligibility criteria were included in the
analysis. Adult patients with a histologically verified diagnosis
of colon cancer who underwent resection of the primary tumor and
initiated systemic anticancer therapy within the prespecified
period were included in the study. Only patients requiring systemic
treatment were included: Stage IV patients and patients with a high
risk of relapse in whom adjuvant chemotherapy was indicated (Stage
III and high-risk Stage II). Patients were excluded if the archived
primary tumor sample was unavailable for analysis or if the
database did not include information about key study variables such
as date of diagnosis, disease-free survival (DFS), progression-free
survival (PFS) or overall survival (OS). Patient data which
included demographic information, disease characteristics,
treatment regimens, response to therapy and clinical outcomes were
retrospectively obtained from the hospital's archive of patients'
files. Patient characteristics are given in Table I. The present study was performed
in line with the principles of the Declaration of Helsinki. This
study was approved by the Medical University Varna Ethics Review
Committee (approval no. 34/13.11.2014). The patients provided
written informed consent to participate.
 | Table IPatient characteristics in cytoplasmic
and nuclear TCTP expression subgroups. |
Table I
Patient characteristics in cytoplasmic
and nuclear TCTP expression subgroups.
| | TCTP cytoplasmic
expression | TCTP nuclear
expression |
|---|
| Clinicopathological
characteristics | Low (%) | High (%) | P-value | Low (%) | High (%) | P-value |
|---|
| Age | | | 0.056 | | | 0.621 |
|
≤64 | 24 (75.0) | 8 (25.0) | | 21 (65.6) | 11 (34.4) | |
|
>64 | 22 (54.4) | 20 (47.6) | | 30 (71.4) | 12 (28.6) | |
| Sex | | | 0.323 | | | 1 |
|
Male | 31 (67.4) | 15 (32.6) | | 32 (69.6) | 14 (30.4) | |
|
Female | 15 (53.6) | 13 (46.4) | | 19 (67.9) | 9 (32.1) | |
| ECOG | | | 0.622 | | | 0.606 |
|
0 | 30 (65.2) | 16 (34.8) | | 33 (71.7) | 13 (28.3) | |
|
1 | 16 (57.1) | 12 (42.9) | | 18 (64.3) | 10 (35.7) | |
| G | | | 0.757 | | | 0.173 |
|
2 | 39 (62.9) | 23 (37.1) | | 45 (72.6) | 17 (27.4) | |
|
3 | 7 (58.3) | 5 (41.7) | | 6 (50.0) | 6 (50.0) | |
| RAS | | | 0.630 | | | 0.802 |
|
Wild-type | 24 (58.5) | 17 (41.5) | | 29 (70.7) | 12 (29.3) | |
|
Mutant | 22 (66.7) | 11 (33.3) | | 22 (66.7) | 11 (33.3) | |
| Primary tumor
location | | | 0.229 | | | 0.612 |
|
Left
colon | 24 (55.8) | 19 (44.2) | | 31 (72.1) | 12 (27.9) | |
|
Right
colon | 22 (71.0) | 9 (29.0) | | 20 (64.5) | 11 (35.5) | |
| Stage groups at
diagnosis | | | 0.280 | | | 0.266 |
|
Non-metastatic | 10(50) | 10(50) | | 16(80) | 4(20) | |
|
Metastatic | 36 (66.7) | 18 (33.3) | | 35 (64.8) | 19 (35.2) | |
| Stage at
diagnosis | | | 0.420a | | | 0.447a |
|
Stage
II | 1(50) | 1(50) | | 2(100) | 0 (0) | |
|
Stage
III | 9(50) | 9(50) | | 14 (77.8) | 4 (22.2) | |
|
Stage
IV | 36 (66.7) | 18 (33.3) | | 35 (64.5) | 19 (35.5) | |
| Best response to
first-line therapy | | | 0.277a | | | 0.784a |
|
CR | 1 (33.3) | 2 (66.7) | | 3(100) | 0 (0) | |
|
PR | 6 (85.7) | 1 (14.3) | | 5 (71.4) | 2 (28.6) | |
|
SD | 15 (53.6) | 13 (46.4) | | 19 (67.9) | 9 (32.1) | |
|
PD | 22 (66.7) | 11 (33.3) | | 21 (63.6) | 12 (36.4) | |
| DCR | | | 0.474 | | | 0.613 |
|
CR+PR+SD | 22 (57.9) | 16 (42.1) | | 27 (71.1) | 11 (28.9) | |
|
PD | 22 (66.7) | 11 (33.3) | | 21 (63.6) | 12 (36.4) | |
Immunohistochemical staining
Archival samples from the primary resected tumor of
all colon cancer patients in the study were obtained. All tissue
samples were fixed with a 10% neutral buffered formalin for 24 h.
Tissue sections (5-µm thick) were cut from the paraffin blocks and
placed on glass slides. Sections were deparaffinized with xylene
and rehydrated in a graded series of ethanol to deionized water.
Antigen retrieval was performed in pre-heated EnVision FLEX Target
Retrieval Solution (Agilent Technologies, Inc.) in PT Link tanks
and incubated for 30 min at 97˚C in a medium at pH 9. After
cooling, the slides were placed in a diluted FLEX Wash Buffer (20x;
Agilent Technologies, Inc.) for 1-5 min. Sections were stained
using the FLEX protocol in Dako Autostainer/Autostainer Plus (Dako;
Agilent Technologies, Inc.). Samples were incubated with polyclonal
rabbit antibody against TCTP (cat. no. ABIN701089;
antibodies-online GmbH). The antibody (anti-TCTP, diluted 1:400)
was incubated for 20 min. Analysis of expression levels of TCTP was
performed using the UltraVision detection system with the
anti-polyvalent HRP/diaminobenzidine (DAB). The reaction was
developed with the appropriate substrate-chromogen (DAB) reagent.
Counterstaining was performed using Mayer's hematoxylin for 1 min
at room temperature. Digital images were obtained using the Leica
Aperio ScanScope AT2 device (Leica Microsystems, Inc.) and further
analyses of the scanned images were performed with ImageScope
v12.1.0.5029 (Leica Microsystems, Inc.).
H-score assessment
A semiquantitative assessment of TCTP levels in the
cells of primary tumors was performed. Two independent pathologists
blinded from the clinical data calculated separate TCTP H-scores in
the cytoplasm and the nuclei of the tumor cells. Staining intensity
was classified into four grades: 0 (no staining), 1 (weak staining,
yellow), 2 (moderate staining, deep yellow) and 3 (strong staining,
brown). The H-score was defined as the percentage of cells with
weak stain intensity, plus two times the percentage of cells
with-moderate stain intensity, plus three times the percentage of
cells with strong stain intensity (28). The mean values of the H-score
assessments of both pathologists for each sample were used in the
analyses.
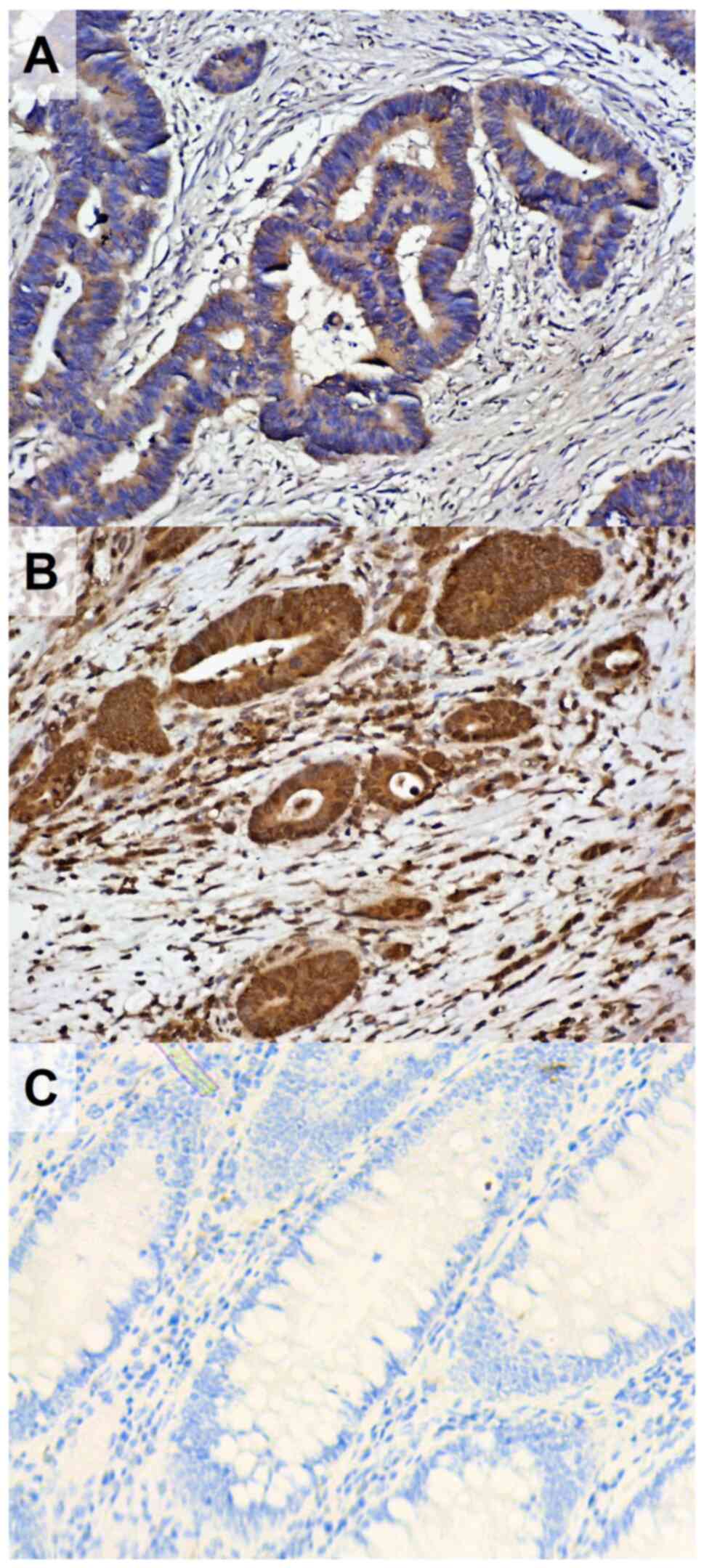
The median cytoplasmic TCTP H-score in our patient
population was 180. The median was used to categorize tumors with
low (≤180) and high (>180) cytoplasmic H-score for subsequent
categorical statistical analyses. The median nuclear TCTP H-score
in the sample was 0. Patients were stratified into two groups;
those with negative nuclear TCTP expression (H-score=0) and
patients with positive nuclear TCTP expression (H-score>0;
Fig. 1). Cytoplasmic and nuclear
TCTP H-score assessments were performed on adjacent healthy tissue
taken at the surgical resection margins for each analyzed sample
following the same procedures.
Endpoints
Disease-free survival (DFS) was defined as the time
to relapse or mortality, from any cause, in patients who received
initial curative treatment in the adjuvant treatment setting.
Progression-free survival (PFS) was defined as the time elapsed
between initiation of first-line treatment for metastatic disease
and tumor progression or mortality from any cause. Overall survival
(OS) was defined as the time between the initial diagnosis of the
disease and mortality. Radiologic tumor response to treatment was
assessed according to the Response Evaluation Criteria in Solid
Tumors v1.1(29). Radiographic
assessments were regularly performed with a tomographic imaging
method such as computerized tomography (CT) and/or positron
emission tomography-CT during follow-ups of patients. Disease
control rate (DCR) was defined as the proportion of patients
achieving complete response, partial response, or stable disease
during the first line of treatment for metastatic disease.
Statistical design and analysis
IBM SPSS software version 23 (IBM Corp.) was used to
perform the statistical analyses. Patient characteristics between
different nuclear and cytoplasmic TCTP expression subgroups were
compared with the Chi-squared test or Fisher's exact test as
appropriate based on the number of observations within each
subgroup. Paired samples t-test was used for the comparison of TCTP
expression between tumor cells and adjacent healthy tissue. The
Mann-Whitney U test and Jonckheere-Terpstra test were used to
compare and identify associations between TCTP levels in the
primary tumor and clinicopathological characteristics. The
probability of survival was estimated using the Kaplan-Meier method
and differences in survival in each subgroup were evaluated with a
log-rank test. Survival data were censored at the time of analysis.
Cox proportional hazards regression was used to investigate
associations between patient survival and clinicopathological
characteristics including TCTP levels in the primary tumor with the
calculation of hazard ratios (HRs). P<0.05 was considered to
indicate a statistically significant difference.
Results
Patient characteristics
A total of 74 patients with colon cancer were
included in the present retrospective study. The mean age of the
patients was 64.9±9.0 years. Forty-six patients were male (62.2%)
and 28 were female (37.8%). The number of patients with RAS
wild-type tumors was 41 (55.4%), whereas 33 (44.6%) patients had a
mutation in the RAS genes. According to histopathological tumor
grading, 62 (83.8%) patients had moderately differentiated tumors
(G2) and 12 (16.2%) had poorly differentiated tumors (G3).
According to the stage at diagnosis, 54 (73%)
patients were diagnosed with primary metastatic disease and 20
(27%) patients were diagnosed with nonmetastatic disease. Among the
latter group, 17 patients further progressed to stage IV while
three patients remained disease-free at the time of analysis. All
patients diagnosed with nonmetastatic colon cancer were treated
with adjuvant chemotherapy. Primary metastatic patients and those
who relapsed received at least one line of chemotherapy in the
metastatic setting.
Associations between TCTP levels and
demographic and clinicopathological characteristics
Chi-squared tests of independence showed that there
were no significant associations between TCTP cytoplasmic and
nuclear expression subgroups and patients' age, sex, Eastern
Cooperative Oncology Group (ECOG) PS, tumor grade, RAS status,
primary tumor location, stage at diagnosis and best response to
first-line chemotherapy. The associations between TCTP expression
and clinicopathological characteristics are summarized in Table I. Patients with higher nuclear TCTP
H-score tended to have a higher number of metastatic sites at
diagnosis (P=0.059; Jonckheere-Terpstra test). A higher nuclear
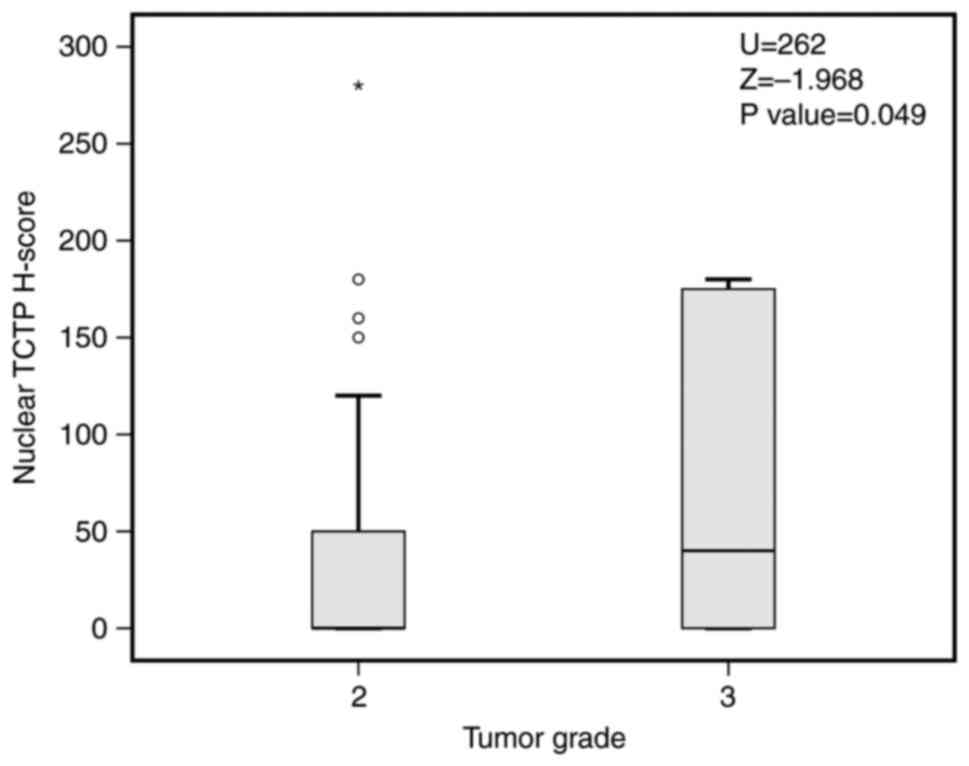
TCTP H-score was associated with a higher tumor grade (P=0.049,
Mann-Whitney; Fig. 2). Mean
cytoplasmic and nuclear TCTP H-scores in tumor cells were
significantly higher compared with adjacent normal colon tissue
(P=0.004 and P<0.001; paired samples t-test).
Effects of TCTP expression on survival
outcomes
Cytoplasmic expression levels of TCTP had no
statistically significant association with DFS (P=0.723), PFS
(P=0.377) and OS (P=0.990).
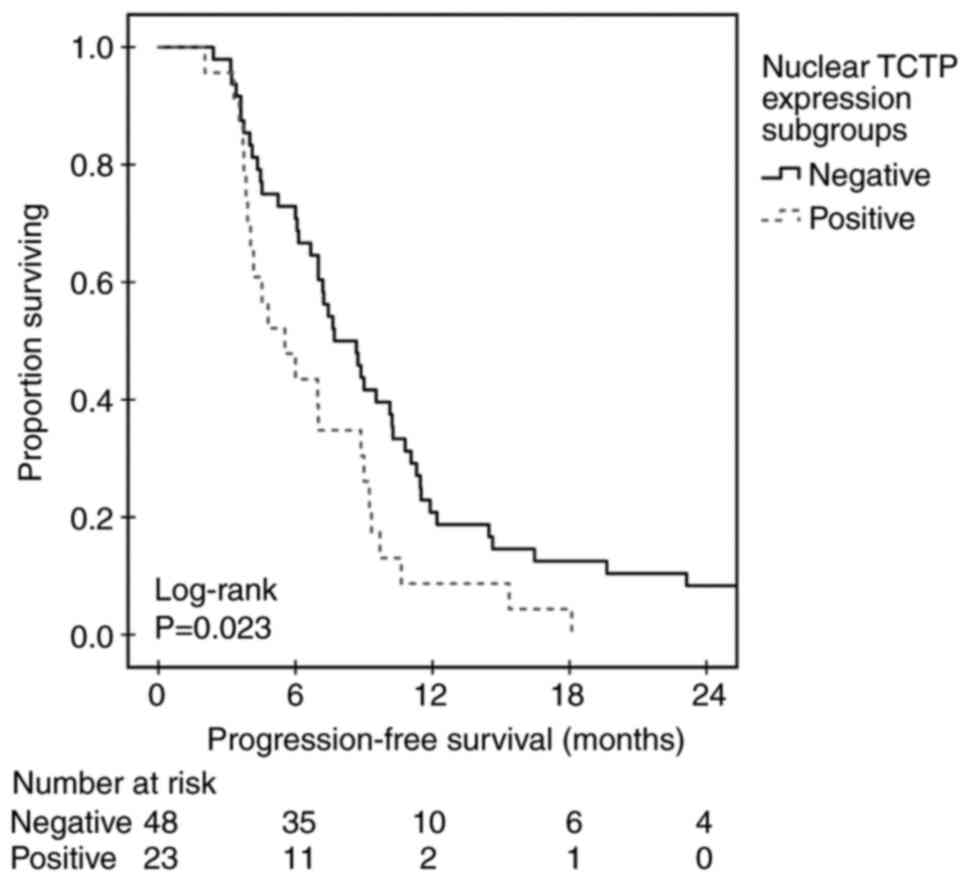
Patients whose primary tumors had a negative nuclear
TCTP H-score had statistically significant improved clinical
outcomes. The PFS for the negative nuclear TCTP expression group
was 7.7 months [95% confidence interval (CI), 5.8-9.5] compared
with 5.5 months (95% CI, 3.2-7.8) in the group with positive
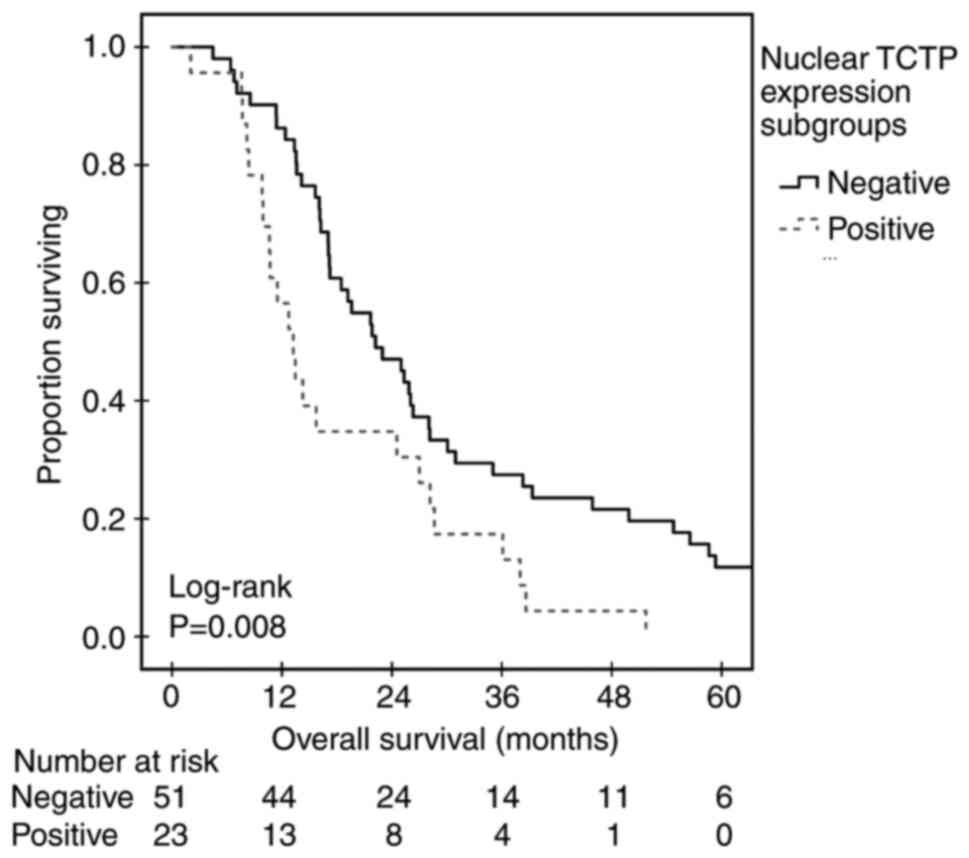
nuclear expression (P=0.023; Mantel-Cox log-rank; Fig. 3). Patients with a negative nuclear
expression of TCTP also had a significantly higher median OS (22.2
months; 95% CI, 16.1-28.3) compared with those with positive TCTP
nuclear expression (median 13.2 months; 95% CI, 10.1-16.3; P=0.008;
Mantel-Cox log-rank; Table II;
Fig. 4). In univariate Cox
regression analysis, a positive nuclear TCTP H-score was a
statistically significant risk factor for worse PFS [hazard ratio
(HR) 1.797; 95% CI, 1.073-3.010; P=0.026] and OS (HR 1.995; 95% CI,
1.189-3.348; P=0.009; Table
III). In a multivariate Cox regression model, a positive
nuclear TCTP H-score was an independent risk factor for worse PFS
and OS (Table IV). The 1-year OS
rate in the group with negative nuclear TCTP expression was 86.3%
compared with 56.5% in patients with positive nuclear TCTP
expression (P=0.008). There was a statistically significant
negative correlation between nuclear TCTP H-score and OS (ρ=-0.287;
P=0.013). The DFS in patients diagnosed with nonmetastatic disease
did not differ among nuclear TCTP expression subgroups
(P=0.813).
 | Table IIMedian progression-free and overall
survival in nuclear TCTP H-score subgroups. |
Table II
Median progression-free and overall
survival in nuclear TCTP H-score subgroups.
| | Progression-free
survival | Overall
survival |
|---|
| | | 95% Confidence
interval | | 95% Confidence
interval |
|---|
| Nuclear TCTP
H-Score | Median | Lower bound | Upper bound | Median | Lower bound | Upper bound |
|---|
| Negative | 7.700 | 5.852 | 9.548 | 22.200 | 16.069 | 28.331 |
| Positive | 5.533 | 3.238 | 7.829 | 13.233 | 10.155 | 16.311 |
 | Table IIIUnivariate Cox regression analysis
for predicting progression-free and overall survival. |
Table III
Univariate Cox regression analysis
for predicting progression-free and overall survival.
| | PFS | OS |
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Age | | | 0.682 | | | 0.606 |
|
≤64 | 1 | - | | 1 | - | |
|
>64 | 1.105 | 0.685-1.782 | | 1.134 | 0.703-1.828 | |
| Sex | | | 0.326 | | | 0.286 |
|
Male | 1 | - | | 1 | - | |
|
Female | 1.284 | 0.779-2.117 | | 1.301 | 0.802-2.111 | |
| ECOG | | | 0.109 | | | 0.012 |
|
0 | 1 | - | | 1 | - | |
|
1 | 1.489 | 0.915-2.423 | | 1.871 | 1.148-3.047 | |
| G | | | 0.311 | | | 0.006 |
|
2 | 1 | - | | 1 | - | |
|
3 | 1.387 | 0.736-2.613 | | 2.491 | 1.293-4.799 | |
| RAS | | | 0.485 | | | 0.002 |
|
Wild-type | 1 | - | | 1 | - | |
|
Mutant | 1.184 | 0.737-1.900 | | 2.113 | 1.306-3.420 | |
| Primary tumor
location | | | 0.301 | | | 0.154 |
|
Left
colon | 1 | - | | 1 | - | |
|
Right
colon | 1.287 | 0.798-2.075 | | 1.422 | 0.877-2.305 | |
| Stage groups at
diagnosis | | | 0.318 | | | <0.001 |
|
Non-metastatic | 1 | - | | 1 | - | |
|
Metastatic | 1.331 | 0.759-2.334 | | 3.174 | 1.775-5.673 | |
| TCTP-cytoplasmic
expression | | | 0.379 | | | 0.990 |
|
Low | 1 | - | | 1 | - | |
|
High | 1.249 | 0.761-2.049 | | 1.003 | 0.619-1.626 | |
| TCTP-nuclear
expression | | | 0.026 | | | 0.009 |
|
Low | 1 | - | | 1 | - | |
|
High | 1.797 | 1.073-3.010 | | 1.995 | 1.189-3.348 | |
 | Table IVMultivariate Cox regression analysis
for predicting PFS and OS. |
Table IV
Multivariate Cox regression analysis
for predicting PFS and OS.
| | PFS | OS |
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Sex | | | 0.456 | | | 0.024 |
|
Male | 1 | - | | 1 | - | |
|
Female | 1.225 | 0.719-2.088 | | 1.845 | 1.085-3.137 | |
| G | | | 0.290 | | | 0.003 |
|
2 | 1 | - | | 1 | - | |
|
3 | 1.432 | 0.736-2.785 | | 3.092 | 1.481-6.456 | |
| RAS | | | 0.809 | | | 0.009 |
|
Wild-type | 1 | - | | 1 | - | |
|
Mutant | 1.063 | 0.646-1.750 | | 2.050 | 1.196-3.512 | |
| ECOG | | | 0.097 | | | 0.043 |
|
0 | 1 | - | | 1 | - | |
|
1 | 1.570 | 0.921-2.676 | | 1.703 | 1.017-2.851 | |
| Stage groups at
diagnosis | | | 0.853 | | | 0.005 |
|
Non-metastatic | 1 | - | | 1 | - | |
|
Metastatic | 1.060 | 0.571-1.967 | | 2.352 | 1.290-4.286 | |
| TCTP nuclear
expression | | | 0.042 | | | 0.040 |
|
Low | 1 | - | | 1 | - | |
|
High | 1.743 | 1.021-2.975 | | 1.799 | 1.027-3.151 | |
Discussion
Despite advances in screening, diagnosis and
treatment, the prognosis for colorectal cancer remains poor,
particularly in the advanced stages of the disease. Therefore,
there is a need to identify new prognostic markers that can predict
patient outcomes and guide treatment decisions.
The current retrospective study discovered
significant differences in survival outcomes in patients with colon
cancer depending on nuclear TCTP expression levels. Despite having
similar clinical and pathological characteristics (age, sex, ECOG
PS, tumor grade, stage at diagnosis and RAS status), the median PFS
and OS were significantly lower in patients with higher nuclear
TCTP expression. In addition, multivariate analysis demonstrated
that nuclear TCTP expression was an independent risk factor for
worse PFS and OS in the present patient population. No prognostic
value of cytoplasmic TCTP levels was found.
The prognostic value of TCTP expression in different
types of cancer was previously evaluated. Previous research on
breast cancer found that higher TCTP expression with predominantly
nuclear staining was associated with a higher pathological grade
and high Ki-67 expression as markers of disease aggressiveness
(21). In another study on
hepatocellular carcinoma high TCTP expression was determined to be
an independent poor prognostic factor associated with higher
disease stage and shorter OS (30). In patients with glioma, high TCTP
expression was associated with advanced pathological grade and
shorter OS (23). TCTP
overexpression was associated with higher FIGO stage and tumor
grade and lower OS in patients with epithelial ovarian carcinoma
(31).
Information about the prognostic significance of
TCTP expression in patients with colon cancer is limited, at least
to the best of the authors' knowledge (26,27).
Fischer et al (27)
investigated the role of TCTP mRNA and protein expression levels as
prognostic factors in different types of cancer. In their colon
cancer dataset, no association between TCTP mRNA expression and OS
was observed but, similarly to the present study, nuclear TCTP
expression was associated with a higher tumor grade. In another
study, Xiao et al (26)
documented higher TCTP expression in patients with higher
pathological grades and metastatic stage of the disease. The same
study also reported that high TCTP expression correlated with poor
metastasis-free survival.
The role of TCTP in carcinogenesis was inferred
since its discovery and it received particular attention during the
investigation of tumor reversion as a process (32). TCTP gene expression levels are
significantly higher in malignant cells than in revertant cells
(13). TCTP is shown to control
tumorigenesis mainly through p53 degradation and subsequent
inhibition of apoptosis (14).
Targeting TCTP by inhibition or gene silencing promotes apoptosis
in cancer cell lines and reduces tumor cell viability (33). TCTP knockdown improves treatment
response to 5-fluorouracil and oxaliplatin in colon cancer cell
lines (34).
While the potential of TCTP as a prognostic marker
in colorectal cancer is promising, there are several challenges for
the clinical translation of this marker. One challenge is the lack
of standardization in the methods used to measure TCTP expression.
Most studies have used immunohistochemistry to measure TCTP
expression, but there is significant variability in the antibodies
and scoring systems used. Standardization of these methods will be
necessary to ensure that TCTP expression can be reliably measured
and compared across studies. To the best of the authors' knowledge,
most studies to date have been small and retrospective and there is
a need for larger studies that include diverse patient populations
that control for other prognostic factors.
The present research had several limitations. The
main limitation was its retrospective nature. Selection bias was
minimized by analyzing all consecutive patients with colon cancer
within the prespecified period who met the eligibility criteria.
However, information about more recently established prognostic and
predictive markers such as the presence of BRAF mutations or MSI
was unavailable. Thus, it was not possible to analyze possible
associations between TCTP expression levels and routinely used
prognostic markers in present clinical practice. Another possible
limitation of the present study was the lack of concurrent p53
mutational status analysis.
While previous reports focus on the suppression of
p53 by TCTP, it has been shown that TCTP mediates the process of
tumor reversion in a pleiotropic manner (i.e., suppression of p53,
acting as a transcription factor, regulation of translation and
secretion of proteins and regulation of the cytoskeleton) (20). Other limitations of the present
study include the small sample size and the single-center design of
the study which did not allow for inclusion of a diverse patient
population in terms of race and ethnicity. On the other hand, the
long follow-up allowed for statistical analyses of survival
outcomes.
In conclusion, the present study revealed that
nuclear TCTP expression level is a potential prognostic marker for
clinical outcomes in patients with colon cancer. A positive nuclear
TCTP expression is associated with a higher tumor grade and worse
PFS and OS. To the best of the authors' knowledge, this is the
first study to demonstrate the value of nuclear TCTP expression as
a prognostic marker for PFS and OS in patients with colon cancer.
These findings may help to identify patients with more aggressive
tumor biology and worse survival outcomes who are candidates for
tailored intensified therapy.
Acknowledgements
Preliminary data from this study were presented at
the 2023 Annual Meeting of the American Society of Clinical
Oncology May 31-June 4, 2023 in Chicago, IL and published as
abstract no. e15513 in Journal of Clinical Oncology 41 (Suppl 16):
2023.
Funding
Funding: No funding was received.
Availability of data and materials
The data used and analyzed during the current study
are available from the corresponding author on reasonable
request.
Authors' contributions
DS and ID performed the experiments and analyzed the
data. DS, NC and MP wrote the manuscript. DS and NC confirm the
authenticity of all the raw data. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The present study was performed in line with the
principles of the Declaration of Helsinki. This study was approved
by the Medical University Varna Ethics Review Committee (approval
no. 34/13.11.2014). The patients provided written informed consent
to participate.
Patient consent for publication
Not applicable.
Authors' information
Dragomir Svetozarov Stoyanov ORCID:
0000-0003-2064-9475
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Xi Y and Xu P: Global colorectal cancer
burden in 2020 and projections to 2040. Transl Oncol.
14(101174)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM
and Wallace MB: Colorectal cancer. Lancet. 394:1467–1480.
2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Siegel RL, Miller KD, Goding Sauer A,
Fedewa SA, Butterly LF, Anderson JC, Cercek A, Smith RA and Jemal
A: Colorectal cancer statistics, 2020. CA Cancer J Clin.
70:145–164. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Punt CJA, Koopman M and Vermeulen L: From
tumour heterogeneity to advances in precision treatment of
colorectal cancer. Nat Rev Clin Oncol. 14:235–246. 2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bommer UA and Thiele BJ: The
translationally controlled tumour protein (TCTP). Int J Biochem
Cell Biol. 36:379–385. 2004.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Thiele H, Berger M, Skalweit A and Thiele
BJ: Expression of the gene and processed pseudogenes encoding the
human and rabbit translationally controlled tumour protein (TCTP).
Eur J Biochem. 267:5473–5481. 2000.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Bommer UA, Borovjagin AV, Greagg MA,
Jeffrey IW, Russell P, Laing KG, Lee M and Clemens MJ: The mRNA of
the translationally controlled tumor protein P23/TCTP is a highly
structured RNA, which activates the dsRNA-dependent protein kinase
PKR. RNA. 8:478–496. 2002.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Nielsen HV, Johnsen AH, Sanchez JC,
Hochstrasser DF and Schiøtz PO: Identification of a basophil
leukocyte interleukin-3-regulated protein that is identical to
IgE-dependent histamine-releasing factor. Allergy. 53:642–652.
1998.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Thaw P, Baxter NJ, Hounslow AM, Price C,
Waltho JP and Craven CJ: Structure of TCTP reveals unexpected
relationship with guanine nucleotide-free chaperones. Nat Struct
Biol. 8:701–704. 2001.PubMed/NCBI View
Article : Google Scholar
|
|
11
|
Gachet Y, Tournier S, Lee M,
Lazaris-Karatzas A, Poulton T and Bommer UA: The growth-related,
translationally controlled protein P23 has properties of a tubulin
binding protein and associates transiently with microtubules during
the cell cycle. J Cell Sci. 112:1257–1271. 1999.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Li F, Zhang D and Fujise K:
Characterization of fortilin, a novel antiapoptotic protein. J Biol
Chem. 276:47542–47549. 2001.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Tuynder M, Susini L, Prieur S, Besse S,
Fiucci G, Amson R and Telerman A: Biological models and genes of
tumor reversion: Cellular reprogramming through tpt1/TCTP and
SIAH-1. Proc Natl Acad Sci USA. 99:14976–14981. 2002.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Rho SB, Lee JH, Park MS, Byun HJ, Kang S,
Seo SS, Kim JY and Park SY: Anti-apoptotic protein TCTP controls
the stability of the tumor suppressor p53. FEBS Lett. 585:29–35.
2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Amson R, Pece S, Lespagnol A, Vyas R,
Mazzarol G, Tosoni D, Colaluca I, Viale G, Rodrigues-Ferreira S,
Wynendaele J, et al: Reciprocal repression between P53 and TCTP.
Nat Med. 18:91–99. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Lucibello M, Gambacurta A, Zonfrillo M,
Pierimarchi P, Serafino A, Rasi G, Rubartelli A and Garaci E: TCTP
is a critical survival factor that protects cancer cells from
oxidative stress-induced cell-death. Exp Cell Res. 317:2479–2489.
2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Sinha P, Kohl S, Fischer J, Hütter G, Kern
M, Köttgen E, Dietel M, Lage H, Schnölzer M and Schadendorf D:
Identification of novel proteins associated with the development of
chemoresistance in malignant melanoma using two-dimensional
electrophoresis. Electrophoresis. 21:3048–3057. 2000.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Graidist P, Phongdara A and Fujise K:
Antiapoptotic protein partners fortilin and MCL1 independently
protect cells from 5-fluorouracil-induced cytotoxicity. J Biol
Chem. 279:40868–40875. 2004.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Chan THM, Chen L and Guan XY: Role of
translationally controlled tumor protein in cancer progression.
Biochem Res Int. 2012(369384)2012.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Telerman A and Amson R: The molecular
programme of tumour reversion: The steps beyond malignant
transformation. Nat Rev Cancer. 9:206–216. 2009.PubMed/NCBI View
Article : Google Scholar
|
|
21
|
Lucibello M, Adanti S, Antelmi E, Dezi D,
Ciafrè S, Carcangiu ML, Zonfrillo M, Nicotera G, Sica L, De Braud F
and Pierimarchi P: Phospho-TCTP as a therapeutic target of
Dihydroartemisinin for aggressive breast cancer cells. Oncotarget.
6:5275–5291. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Ma Q, Geng Y, Xu W, Wu Y, He F, Shu W,
Huang M, Du H and Li M: The role of translationally controlled
tumor protein in tumor growth and metastasis of colon
adenocarcinoma cells. J Proteome Res. 9:40–49. 2010.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Miao X, Chen YB, Xu SL, Zhao T, Liu JY, Li
YR, Wang J, Zhang J and Guo GZ: TCTP overexpression is associated
with the development and progression of glioma. Tumour Biol.
34:3357–3361. 2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Amson R, Pece S, Marine JC, Di Fiore PP
and Telerman A: TPT1/TCTP-regulated pathways in phenotypic
reprogramming. Trends Cell Biol. 23:37–46. 2013.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhang D, Li F, Weidner D, Mnjoyan ZH and
Fujise K: Physical and functional interaction between myeloid cell
leukemia 1 protein (MCL1) and Fortilin. The potential role of MCL1
as a fortilin chaperone. J Biol Chem. 277:37430–37438.
2002.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Xiao B, Chen D, Luo S, Hao W, Jing F, Liu
T, Wang S, Geng Y, Li L, Xu W, et al: Extracellular translationally
controlled tumor protein promotes colorectal cancer invasion and
metastasis through Cdc42/JNK/MMP9 signaling. Oncotarget.
7:50057–50073. 2016.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Fischer N, Saeed MEM, Lippe E, Roth W and
Efferth T: High TCTP expression as prognostic factor in different
cancer types. World Acad Sci J. 3(3)2021.
|
|
28
|
McCarty K Jr, Miller LS, Cox EB, Konrath J
and McCarty K Sr: Estrogen receptor analyses. Correlation of
biochemical and immunohistochemical methods using monoclonal
antireceptor antibodies. Arch Pathol Lab Med. 109:716–721.
1985.PubMed/NCBI
|
|
29
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Chan THM, Chen L, Liu M, Hu L, Zheng BJ,
Poon VK, Huang P, Yuan YF, Huang JD, Yang J, et al: Translationally
controlled tumor protein induces mitotic defects and chromosome
missegregation in hepatocellular carcinoma development. Hepatology.
55:491–505. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Chen C, Deng Y, Hua M, Xi Q, Liu R, Yang
S, Liu J, Zhong J, Tang M, Lu S, et al: Expression and clinical
role of TCTP in epithelial ovarian cancer. J Mol Histol.
46:145–156. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Tuynder M, Fiucci G, Prieur S, Lespagnol
A, Géant A, Beaucourt S, Duflaut D, Besse S, Susini L, Cavarelli J,
et al: Translationally controlled tumor protein is a target of
tumor reversion. Proc Natl Acad Sci USA. 101:15364–15369.
2004.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Gnanasekar M, Thirugnanam S, Zheng G, Chen
A and Ramaswamy K: Gene silencing of translationally controlled
tumor protein (TCTP) by siRNA inhibits cell growth and induces
apoptosis of human prostate cancer cells. Int J Oncol.
34:1241–1246. 2009.PubMed/NCBI
|
|
34
|
Bommer UA, Vine KL, Puri P, Engel M,
Belfiore L, Fildes K, Batterham M, Lochhead A and Aghmesheh M:
Translationally controlled tumour protein TCTP is induced early in
human colorectal tumours and contributes to the resistance of
HCT116 colon cancer cells to 5-FU and oxaliplatin. Cell Commun
Signal. 15(9)2017.PubMed/NCBI View Article : Google Scholar
|