Introduction
Splenosis is a rare benign condition occurring after
trauma, splenectomy, or other procedures involving splenic tissue.
It occurs when splenic sinus cells are transplanted directly into
different compartments of the abdominal cavity or into the thorax
(1). Distant transplantation of
splenic tissue can occur by hematogenous spread to different
organs, such as the liver, skin, or brain (2). Intrahepatic splenosis (IHS) is a
condition in which splenic tissue is embedded within the liver
parenchyma, and it was first described in the literature in
1939(3). It is usually detected
incidentally during a physical examination or on imaging, some
cases presented with diarrhea, pain, or bowel obstruction. The
average interval between the initial trauma and detection is 16
years (range: 5 months to 32 years) (4), and cases of up to 60 years interval
have been reported (5).
Radiological findings are usually non-specific (6-10).
Typical findings are hypodense areas on abdominal ultrasonography
and non-contrast CT. Following contrast administration, IHS is
hyperdense in the arterial phase, and hypodense in the delayed
phase (3). On MRI, IHS is
described as areas of homogeneous hypo-intensity in T1WI and
hyperintensity in T2WI (10).
Clinically, it is sometimes preoperatively diagnosed as a malignant
tumor and surgical treatment should be inevitable. We report our
experience with a rare case of IHS in which the imaging pattern was
suggestive of hepatocellular carcinoma (HCC).
Case report
A 46-year-old man with a previous treatment history
of a left lateral segmentectomy of the liver and splenectomy for a
road traffic injury 30 years earlier presented to our emergency
department with acute abdominal pain. The patient had no weight
loss and no history of hepatitis or heavy drinking. The routine
blood tests, including liver and renal function, were normal, and
the serological biomarkers of hepatitis B and C virus infections
were negative. Serum tumor marker levels for alpha-fetoprotein,
CEA, CA19-9, and CA-125 were normal.
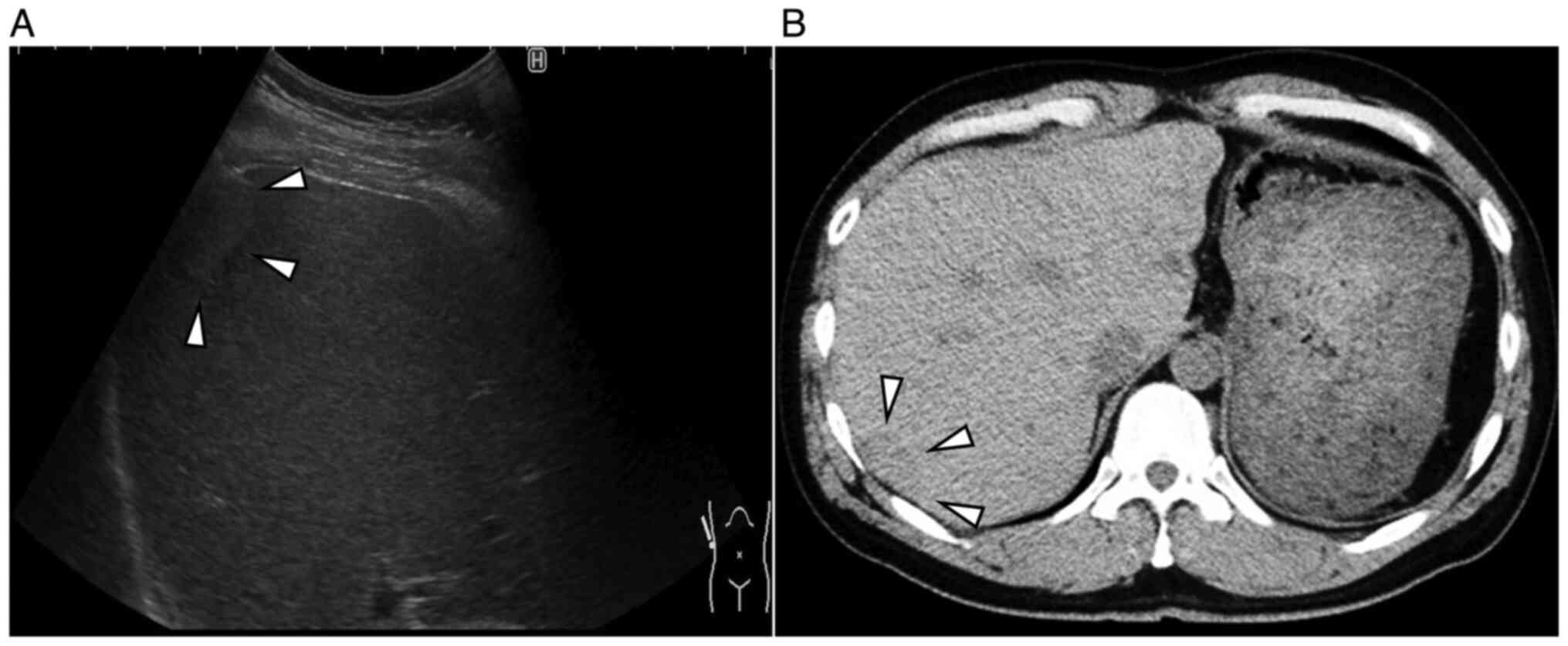
Abdominal ultrasonography (US) revealed a 2.4x1.4 cm
isoechoic lesion with a hypoechoic zone at the margins in segment 7
of the right lobe of the liver (Fig.
1A). Abdominal plain-computed tomography (CT) revealed a
hypodense mass measuring 2.5x1.7 cm (Fig. 1B). Abdominal magnetic resonance
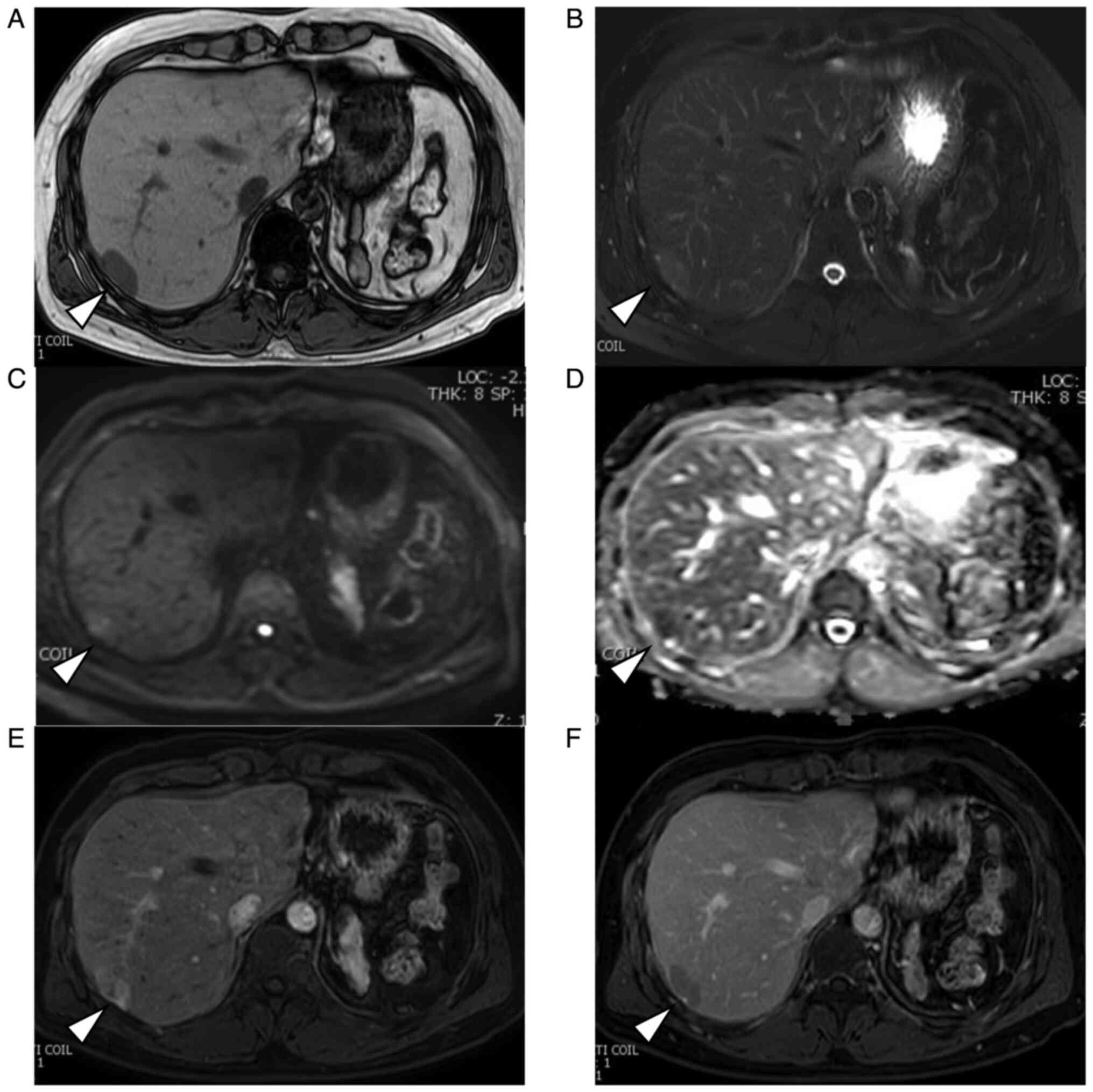
imaging (MRI) revealed a homogeneously hypointense mass in
T1-weighted images and hyperintensity in T2-weighted images
(Fig. 2A and B). The mass showed a heterogeneous
hyperintensity in diffusion-weighted images and signal reduction in
apparent diffusion coefficient. (Fig.
2C and D). After injecting
gadoxetic acid, the lesion appeared strongly heterogeneous and
hyperintense during the early phase and relatively hypointense
during delayed phase that is ‘washout pattern’ (Fig. 2E and F). An indication of a pseudocapsule was
also seen.
Radiographic features suggested a differential
diagnosis of HCC, hepatic hemangioma, and hepatocellular adenoma.
Surgical intervention was proposed, and the patient decided to
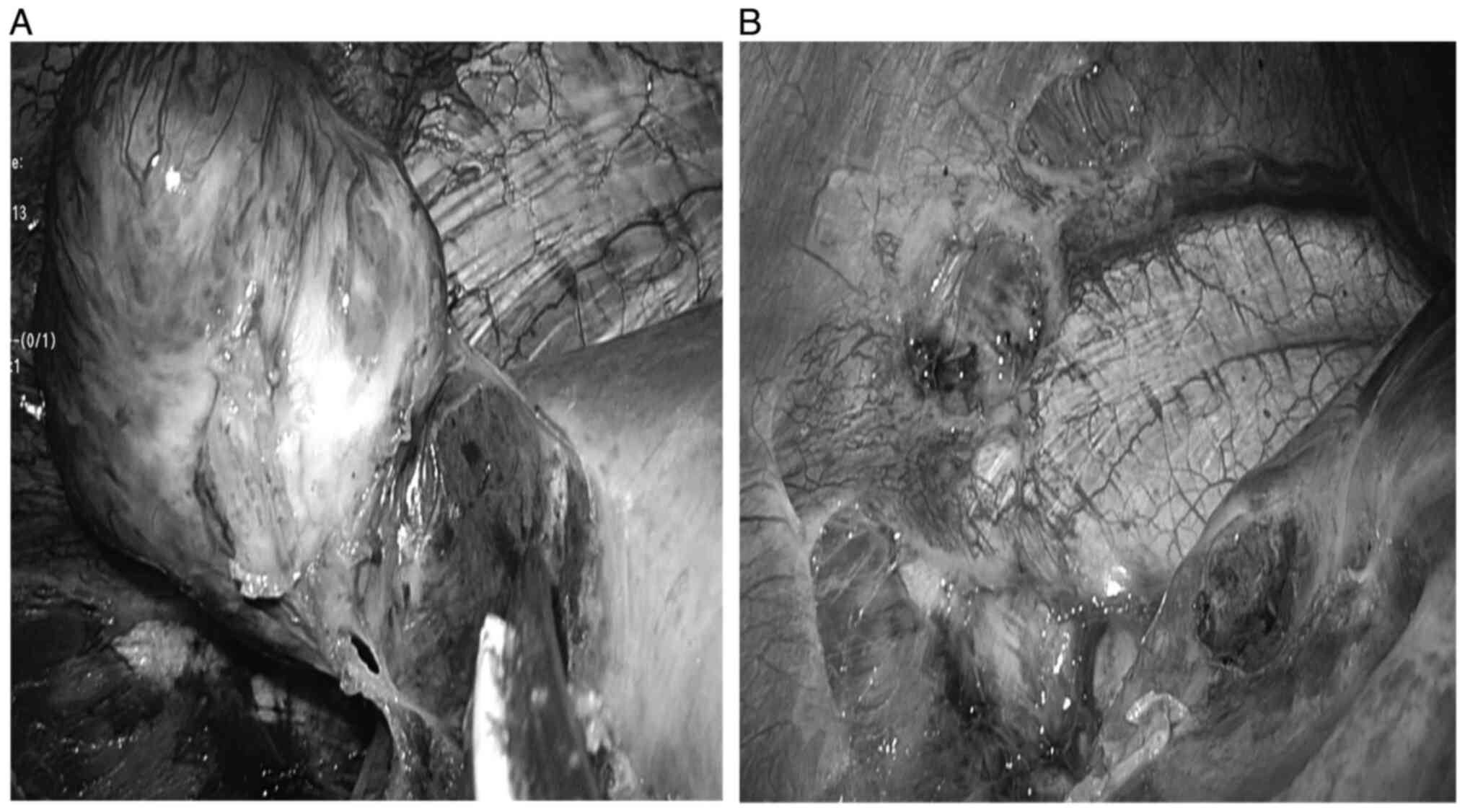
undergo a laparoscopic surgery. During surgery, the tumor was found
in segment 7 of the right lobe (Fig.
3A). The tumor's feeding artery and drainage vessel were found
to be from the inferior phrenic artery and vein (Fig. 3B). Liver tumor was successfully
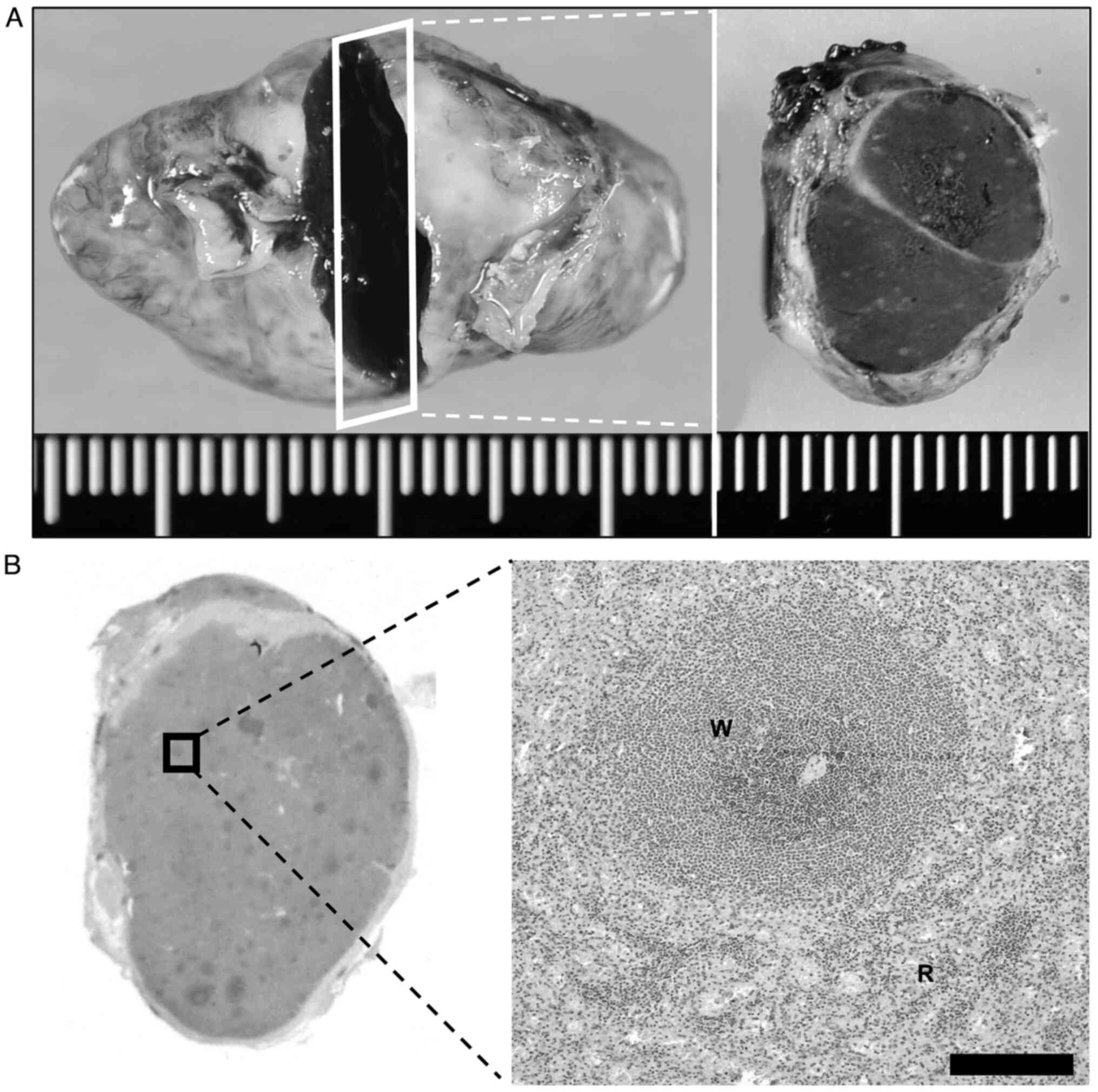
resected laparoscopically. Then, histopathologic examination of the
resected specimen revealed that the tumor was totally consisted of
splenic tissue surrounded by a fibrous capsule (Fig. 4A and B). The postoperative diagnosis was
confirmed as IHS. The patient recovered uneventfully and was
discharged on the fourth postoperative day. The patient visited
outpatient care for 2 years of postoperative follow-up without any
trouble.
Discussion
Splenosis is usually caused by heterotopic
autotransplantation or implantation of splenic tissue after
elective splenectomy or traumatic splenic rupture. Approximately
70% of patients with IHS are reported to have a history of splenic
rupture or surgery (11). IHS is
rare because splenosis usually occurs in the mesentery, omentum, or
peritoneum in the left upper abdomen (12).
After a literature search, using the search term
‘intrahepatic splenosis’ on the PubMed, we identified and reviewed
52 cases. Of these, 14 cases were excluded because of lacking
clinical details, and the remaining 38 cases were included in our
review (Table SI). There were 31
(81.5%) male and 7 (18.4%) female patients, with a mean age of 47.9
(±13) years. There was a previous history of abdominal trauma in
89% of the patients, and 97% had a history of splenectomy, which is
consistent with a previous report (12). Seventy-three percent of patients
were asymptomatic on admission. The mean tumor size on imaging was
3.7 (±1.2) cm. Fifty-eight percent of the patients underwent
surgery, including hepatectomy, with the preoperative diagnosis of
HCC or liver metastases (3,7,10,13).
The radiographic appearance of IHS generally varies.
Typical findings are hypodense areas on non-contrast CT. Following
contrast administration, the lesions are hyperdense in the arterial
phase, iso-dense in the portal venous phase, and hypodense in the
delayed phase (3,14). On MRI, IHS is usually described as
areas of homogeneous hypo-intensity in T1WI and hyperintensity in
T2WI (10). Following contrast
administration, IHS shows a heterogeneous enhancement in the
arterial phase, which becomes homogeneous in the later phases
(8,9,15).
In the delayed phase, the signal intensity of IHS may be lower than
that of the liver parenchyma (10). The imaging features are similar to
the signal and enhancement patterns of the spleen, usually
described as geographic or zebra patterns of enhancement. In the
previous studies, 61.5% of patients had typical radiological
features. In our case, the enhancement pattern of the tumor in the
enhanced MRI showed early enhancement in the arterial phase and
washout in the delayed phase that was similar to the radiographic
features of moderately differentiated HCC.
There are currently reported to be two dominant
mechanisms of IHS occurring. One hypothesis is that an invagination
or an exophytic growth of splenic tissue directly seeded into the
liver capsule. With this mechanism, the most frequent site of IHS
is the area surrounded by the left lobe and the diaphragm because
it can be easily seeded with splenic tissue during splenectomy
(14,16-18).
The other is that hematogenous spread due to the entry of an
erythrocyte progenitor cell into the portal venous system. In the
latter pattern, the margin between IHS and liver parenchyma is
usually unclear (2,19,20).
In our case, tumor happened to metastasize in the right lobe near
the inferior phrenic vessels without seeding at the most frequent
site, and it was extrahepatically fed by these vessels. The tumor
showed early enhancement in the arterial phase and washout in the
delayed phase that was characteristic imaging pattern for HCC,
although IHS shows a heterogeneous enhancement in the arterial
phase, and homogeneous in the later phases (8,9,15).
In our case, IHS was extrahepatically fed, so that the radiological
findings were typical for HCC. It was difficult to make a correct
diagnosis of IHS preoperatively. Surgical procedure was
successfully performed.
We reported a rare case of IHS mimicking HCC. With a
history of traumatic rupture of the spleen and splenectomy, even
when the tumor has typical radiological patterns for HCC, IHS might
be a different diagnosis.
Supplementary Material
Previous reports of patients with
intrahepatic splenosis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
IU, HT, KM, AK, SM, RK, KN, SN, YY and AM
participated in the diagnosis and treatment of the patient and
wrote the first draft of this manuscript. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient to publish this case report and any accompanying
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Gandhi D, Sharma P, Garg G, Songmen S,
Solanki S and Singh T: Intrahepatic splenosis demonstrated by
diffusion weighted MRI with histologic confirmation. Radiol Case
Rep. 15:602–606. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Menth M, Herrmann K, Haug A, Raziorrouh B,
Zachoval R, Jung CM and Otto C: Intra-hepatic splenosis as an
unexpected cause of a focal liver lesion in a patient with
hepatitis C and liver cirrhosis: A case report. Cases J.
2(8335)2009.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Toh WS, Chan KS, Ding CSL, Tan CH and
Shelat VG: Intrahepatic splenosis: A world review. Clin Exp
Hepatol. 6:185–198. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Rickert CH, Maasjosthusmann U,
Probst-Cousin S, August C and Gullotta F: A unique case of cerebral
spleen. Am J Surg Pathol. 22:894–896. 1998.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Baldari G, Sindoni A, Belletti A, Baldari
S and Ruffini L: (99m)Tc disphosphonate uptake due to splenosis.
Incidental finding 60 years after splenectomy. Clin Nucl Med.
40:533–535. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
De Vuysere S, Van Steenbergen W, Aerts R,
Van Hauwaert H, Van Beckevoort D and Van Hoe L: Intrahepatic
splenosis: Imaging features. Abdom Imaging. 25:187–189.
2000.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Yeh ML, Wang LY, Huang CI, Hsieh MY, Lin
ZY, Chuang WL, Chang WT, Wu CC and Chen CY: Abdominal splenosis
mimicking hepatic tumor: A case report. Kaohsiung J Med Sci.
24:602–606. 2008.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Lin WC, Lee RC, Chiang JH, Wei CJ, Chu LS,
Liu RS and Chang CY: MR features of abdominal splenosis. AJR Am J
Roentgenol. 180:493–496. 2003.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Inchingolo R, Peddu P and Karani J:
Hepatic splenosis presenting as arterialised liver lesion in a
patient with NASH. Eur Rev Med Pharmacol Sci. 17:2853–2856.
2013.PubMed/NCBI
|
|
10
|
Sato N, Abe T, Suzuki N, Waragai M,
Teranishi Y, Takano Y, Sato A, Azami A and Gotoh M: Intrahepatic
splenosis in a chronic hepatitis C patient with no history of
splenic trauma mimicking hepatocellular carcinoma. Am J Case Rep.
15:416–420. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Case records of the Massachusetts general
hospital. Case 29-1995: A 65-year-old man with mediastinal
Hodgkin's disease and a pelvic mass. N Engl J Med. 333:784–791.
1995.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Mescoli C, Castoro C, Sergio A, Ruol A,
Farinati F and Rugge M: Hepatic spleen nodules (HSN). Scand J
Gastroenterol. 45:628–632. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kang KC, Cho GS, Chung GA, Kang GH, Kim
YJ, Lee MS, Kim HK and Park SJ: Intrahepatic splenosis mimicking
liver metastasis in a patient with gastric cancer. J Gastric
Cancer. 11:64–68. 2011.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Tsitouridis I, Michaelides M, Sotiriadis C
and Arvaniti M: CT and MRI of intraperitoneal splenosis. Diagn
Interv Radiol. 16:145–149. 2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Pekkafali Z, Karsli AF, Silit E, Başekim
CC, Narin Y, Mutlu H and Kizilkaya E: Intrahepatic splenosis: A
case report. Eur Radiol. 12:S62–S65. 2002.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Liu K, Liang Y, Liang X, Yu H, Wang Y and
Cai X: Laparoscopic resection of isolated hepatic splenosis
mimicking liver tumors: Case report with a literature review. Surg
Laparosc Endosc Percutan Tech. 22:e307–e311. 2012.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Teles GNS, Monteiro PEZ and Raphe R:
Intrahepatic splenosis mimicking hepatic neoplasia. Int J Surg Case
Rep. 44:47–50. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Choi GH, Ju MK, Kim JY, Kang CM, Kim KS,
Choi JS, Han KH, Park MS, Park YN, Lee WJ and Kim BR: Hepatic
splenosis preoperatively diagnosed as hepatocellular carcinoma in a
patient with chronic hepatitis B: A case report. J Korean Med Sci.
23:336–341. 2008.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Seguchi S, Yue F, Asanuma K and Sasaki K:
Experimental splenosis in the liver and lung spread through the
vasculature. Cell Tissue Res. 360:287–296. 2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Kwok CM, Chen YT, Lin HT, Su CH, Liu YS
and Chiu YC: Portal vein entrance of splenic erythrocytic
progenitor cells and local hypoxia of liver, two events cause
intrahepatic splenosis. Med Hypotheses. 67:1330–1332.
2006.PubMed/NCBI View Article : Google Scholar
|