Introduction
Adenovirus infection often occurs in children aged 6
months to 2 years (1,2). Some children have severe clinical
manifestations, extrapulmonary complications, and chronic airway or
lung diseases. According to local and international reports,
neonatal adenovirus pneumonia is rare (3,4).
The present study describes the case of a fatal
adenovirus infection in a neonate with coagulation disorders and
hypoxemia, that rapidly progressed to multiple organ failure
syndrome. Th patient succumbed to the disease despite receiving
extracorporeal supportive treatment.
Case report
A 15-day-old full-term male infant was admitted to a
local maternity hospital with fever and anorexia. The neonate was
born vaginally at 40(+2) weeks of gestation. The birth weight was
3,300 g. The Apgar score yielded a score of 9 in 1 min and a score
of 10 in 5 min. The infant's mother was 27 years old, gravida 2,
para 2. The infant was in a good physical condition until 8 days
prior to hospital admission. Following a physical examination and
clinical analysis, he was diagnosed with pneumonia and received
oxygen supplementation, intravenous maintenance fluids,
ceftriaxone, clarithromycin and nebulization therapy. The patient
was transferred to another hospital, where he was intubated and
mechanically ventilated, for increased respiratory distress and a
poor response to treatment. However, his general condition
continued to deteriorate, and the respiratory distress worsened. He
remained on high frequency oscillatory ventilation (HFOV) with a
mean airway pressure (MAP) of 17 cmH2O, and the fraction
of inspired oxygen (FiO2) was increased up to 1.0 to
maintain pulse blood oxygen saturation (SpO2)
>85-88%. A chest radiograph revealed exacerbated pneumonia. The
patient was in a poor condition and was transferred to the
Children's Hospital of Soochow University.
Upon arrival to the neonatal intensive care unit
(NICU), Children's Hospital of Soochow University, his
SpO2 was 80-85% on HFOV ventilation, with a MAP of 20
cmH2O and FiO2 of 1.0. The laboratory
analyses revealed a white blood cell count of 8.00x109/l
(neutrophils, 59.8%), a hemoglobin count of 109 g/l, platelet count
of 106x109/l and c-reactive protein levels of 6.11 mg/l;
the prothrombin time (PT) and activated partial thromboplastin time
(APTT) were >170 and 112.5 seconds, respectively. The D-dimer
levels were 3470 µg/l, and the infant tested negative for blood
culture, TORCH, influenza, parainfluenza, respiratory syncytial
virus, syphilis and mycoplasma antibodies. However, the infant
tested positive for adenovirus in sputum. Further analyses revealed
glutamic pyruvic transaminase levels of 213 U/l,
glutamic-oxalacetic transaminase levels of 1147 U/l, bilirubin
levels of 20.7 µmol/l, direct bilirubin levels of 17.3 µmol/l,
creatinine levels of 28.8 µmol/l, urea nitrogen levels of 1.49
mmol/l and N-terminal-pro-B-brain natriuretic peptide levels of
>35,000 pg/ml. The humoral immune indices were as follows:
Complement C3, 0.61 g/l; complement C4, 0.07 g/l; IgA, 0.95 g/l;
IgG, 11.53 g/l; and IgM, 1.3 g/l. The cellular immune indices were:
CD3+ cells, 76.5%; CD4+ cells, 52.3%;
CD8+ cells, 23.3%; CD4+/CD8+
cells, 2.2%; CD19+ and CD23+ cells, 1.9%.
After 1 day, the patient continued to have low oxygen saturation
levels. His oxygenation index was 95 (MAP x FiO2
x100/PaO2), which lasted for >4, and MAP increased to
30 cmH2O. At 3 days following his hospital admission,
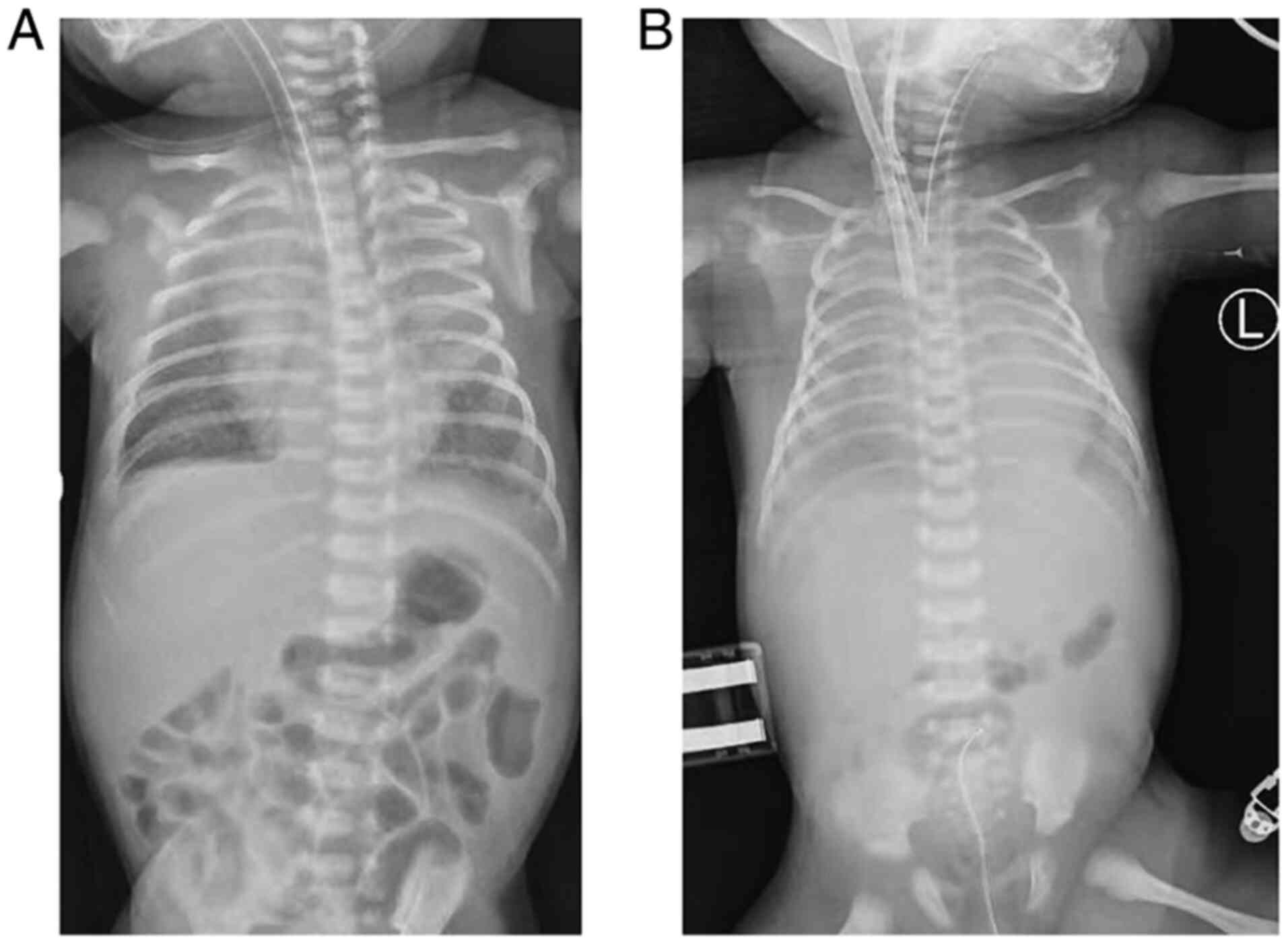
extracorporeal membrane oxygenation (ECMO) was administered
(Fig. 1A). The patient was placed on
veno-arterial ECMO to comply with a lung protective ventilation
strategy and prevent the progression of pneumomediastinum. The
medical practitioner inserted a 14F drainage cannula into the right
internal jugular vein and an 8F return cannula into the right
common carotid artery. Following cannulation, the blood flow of
ECMO was titrated to 0.48 l/min to achieve optimal gas exchange
(Fig. 1B). The patient received
heparin for anticoagulation, maintaining the activated clotting
time between 180-220 sec and the APTT between 60-80 sec. The
ultra-protective strategy was used for mechanical ventilation using
the pressure control ventilation model with a peak inspiratory
pressure of 15 cmH2O, positive end expiratory pressure
of 4 cmH2O, a respiratory rate of 15/min and a
FiO2 of 30%. The patient's respiratory function and
coagulation function progressively deteriorated. Diagnostic
bronchoscopy with broncho-alveolar lavage fluid collection was
performed on day 3 of ECMO, and adenovirus was identified.
Peritoneal dialysis was performed on day 3 of ECMO, following which
the patient experienced hypotension and oliguria. During this
period, the patient was administered a large dose of noradrenaline
(0.5 µg/kg/min) and anti-inflammatory drugs (methylprednisolone, 3
mg/kg/day). Bilateral closed thoracic drainage was performed on day
5 of ECMO, following pleural effusion. Diagnostic bronchoscopy was
repeated on day 7 of ECMO which revealed bronchitis, blocked
bronchi, and atelectasis. The coagulation function worsened, and
the patient developed gastrointestinal bleeding and respiratory
hemorrhage. Moreover, there was no notable improvement in the
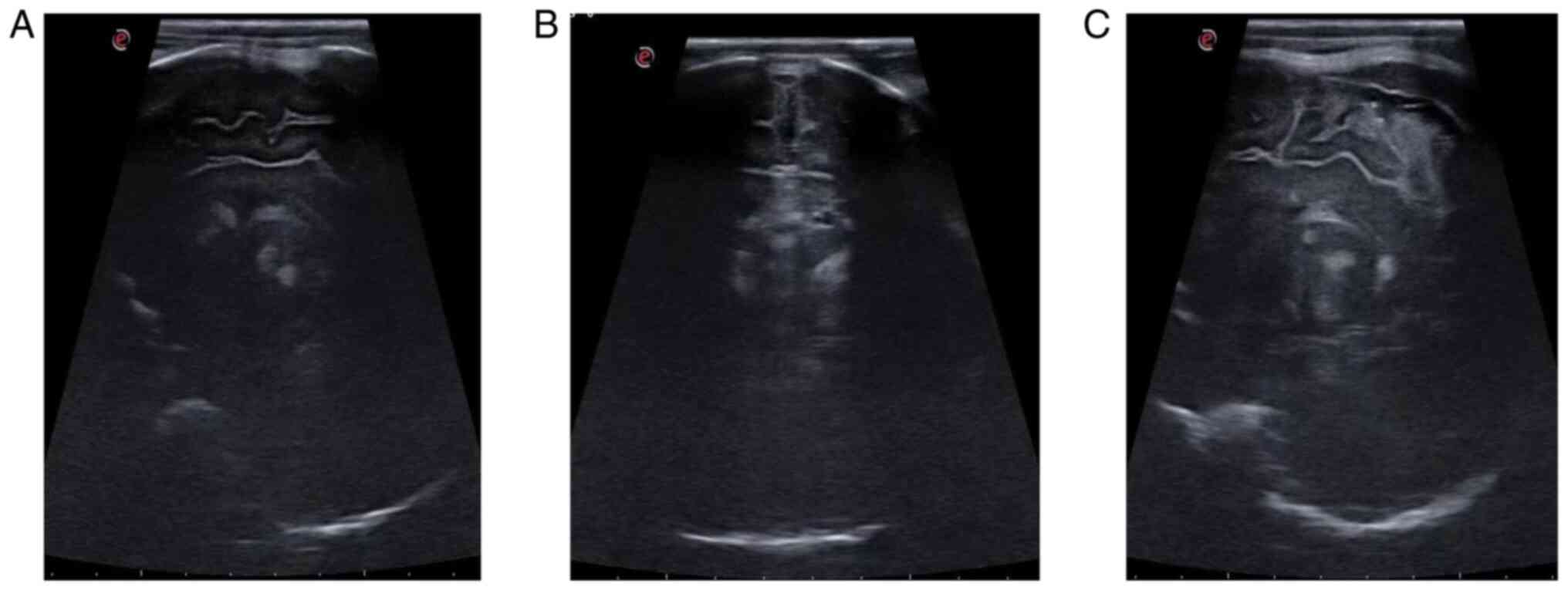
pulmonary symptoms. A cranial ultrasonography suggested massive
intracranial hemorrhage (Fig. 2),
and the patient succumbed after 11 days of ECMO.
Discussion
With continuous developments in the field of
perinatal medicine, a variety of anti-infective drugs have been
used, and notable progress has been made in the treatment of
infectious diseases. Pneumonia remains the leading cause of
mortality in infants, and distinguishing the pathogenic
microorganisms at an early is a difficult task (5). The present study describes the clinical
case of a neonate who received antimicrobial and ECMO therapy for
respiratory distress syndrome. Subsequently, the patient was
diagnosed with adenoviral infection based on blood and sputum
sample analyses. The patient's clinical status continuously
deteriorated, ultimately resulting in death following 11 days of
ECMO treatment.
Neonatal adenovirus infection is uncommon, and the
mortality rate of severe adenovirus disseminated infection in
neonates is almost 67% (6). The lack
of specific symptoms, early identification and effective treatment
strategies all contribute to the high mortality rate and fatal
outcome in the majority of cases.
The main clinical manifestations of adenovirus
infection are similar to those of respiratory tract infections
caused by a variety of other pathogens (7,8). Due to
the lack of specific clinical features, the early diagnosis of
neonatal adenovirus infection is difficult. The majority of these
patients present with dyspnea and coagulation disorders, and
rectifying hypoxemic respiratory failure may be difficult. Some
patients develop refractory hypoxemia despite intubation and
treatment with invasive mechanical ventilation.
Respiratory viral infections in the NICU remain
under-recognized, Common viruses that cause neonatal respiratory
diseases include respiratory syncytial virus, parainfluenza virus,
influenza virus, rhinoviruses, varicella virus, coronavirus and
human metapneumovirus. To date, a routine test for detecting
viruses is not available in the NICU due to the following reasons:
i) The clinical symptoms and signs of viral infections are
non-specific and overlap with other conditions of critically ill
neonates; ii) laboratory examinations to confirm viral infection is
difficult; and iii) effective treatment interventions are lacking
(9).
The clinical manifestations of adenovirus infection
are mainly a persistently high fever, cough, wheezing, and at
times, respiratory failure and acute respiratory distress syndrome.
Moreover, infants and neonates are susceptible to more severe
complications, often with extrapulmonary dissemination (10).
Adenovirus is a non-enveloped double-stranded
deoxyribonucleic acid virus. In total, there are 55 serotypes of
adenovirus that are sub-divided into seven species (A to G), which
may cause disease in humans. Types 3 and 7 are the most common
pathogens causing pneumonia (11),
and the majority of these infections occur in infants <2 years
of age, typically resulting in gastrointestinal and respiratory
tract manifestations. The adenovirus strain is easily isolated from
nasopharyngeal secretions. In the present study, human adenovirus
of species D-53 (HAdV-D53) was identified from bronchoalveolar
lavage fluid and blood in this patient. HAdV-D53 is known as the
cause of epidemic acute conjunctivitis (12). In addition, in the present study,
HAdV-D53 was also found in the breast milk of the patient's mother;
furthermore, the medical worker also had severe acute
conjunctivitis during the treatment period for this patient. It is
considered that the virus is highly pathogenic and infectious.
ECMO has been applied more widely in the treatment
of critically ill patients with respiratory and circulatory failure
with the implementation of the pulmonary protective ventilation
strategy (13). The success rate of
treatment with ECMO is higher in children, particularly for
neonatal respiratory diseases (14).
Although the use of ECMO in patients with severe respiratory
failure has been shown to lead to improved outcomes (15), there are very few cases in which ECMO
therapy was administered for severe adenovirus pneumonia in
neonates with poor coagulation function. ECMO in patients with
refractory hypoxemia caused by adenovirus may increase the risk of
bleeding due to the need of anticoagulation for circuit patency.
Bleeding is considered one of the most severe complications of
ECMO; thus, its use should perhaps be avoided as much as possible
by pediatricians. However, the patient described herein presented
with coagulation disorders at the beginning of ECMO, and several
efforts were made to maintain clotting function following ECMO.
Cranial sonography revealed hemorrhagic tracts in the brain. There
is limited evidence or clinical experience available for successful
treatment in neonates. Antiviral therapy, such as the use of
ribavarin and cidofovir for adenoviral infection have been
previously; however, no evident beneficial effect on the clinical
status was observed (16,17).
In conclusion, adenovirus pneumonia in neonates is
frequently disseminated and fatal, and needs to be differentiated
from other conditions, such as sepsis and pneumonia caused by other
pathogens. Rapid tests for detecting respiratory viruses are
currently available and are crucial in the diagnosis of viral
infections in high-risk infants. ECMO may provide an opportunity
and time frame for neonates infected with adenovirus though which
to improve. However, extrapulmonary complications significantly
affect the prognosis.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by grants from the
National Natural Science Foundation of China (grant nos. 81971423
and 81771626).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HW, TP, XL and XZ contributed to the acquisition,
analysis and interpretation of the data; they made substantial
contributions to the conception and design of the study, and
supervised and substantially revised the study. All authors had
equal contributions and equal participation in this article. HW and
XZ confirm the authenticity of all the raw data. All authors have
read and agreed to the published version of the manuscript.
Ethics approval and consent to
participate
The present study was reviewed and approved by the
Ethics Committee of the Children's Hospital of Soochow University
(Suzhou, China). Written informed consent was obtained from the
parents of the patient.
Patient consent for publication
Written informed consent was obtained from the
parents of the patient for the publication of the present case
report and the accompanying images. A copy of the signed consent
form is available for review by the editor of this journal.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Tural-Kara T, Özdemir H, Yıldız N, Aldemir
Kocabaş B, Erat T, Yahşi A, Doğu F, Tutar E, İnce E and Çiftçi E:
Underlying diseases and causative microorganisms of recurrent
pneumonia in children: A 13-year study in a University Hospital. J
Trop Pediatr. 65:224–230. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Lin GL, Lu CY, Chen JM, Lee PI, Ho SY,
Weng KC, Huang LM and Chang LY: Molecular epidemiology and clinical
features of adenovirus infection in Taiwanese children, 2014. J
Microbiol Immunol Infect. 52:215–224. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Wang X, Tan X and Li Q: The difference in
clinical features and prognosis of severe adenoviral pneumonia in
children of different ages. J Med Virol: Feb 25, 2022 (Epub ahead
of print). doi: https://doi.org/10.1002/jmv.27680.
|
|
4
|
Lim LM, Woo YY, de Bruyne JA, Nathan AM,
Kee SY, Chan YF, Chiam CW, Eg KP, Thavagnanam S and Sam IC:
Epidemiology, clinical presentation and respiratory sequelae of
adenovirus pneumonia in children in Kuala Lumpur, Malaysia. PLoS
One. 13(e0205795)2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wardlaw T, Salama P, Johansson EW and
Mason E: Pneumonia: The leading killer of children. Lancet.
368:1048–1050. 2006.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Moallem M, Song E, Jaggi P, Conces MR,
Kajon AE and Sánchez PJ: Adenovirus and ‘Culture-Negative Sepsis’
in a Preterm Neonate. AJP Rep. 6:e417–e420. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hung KH and Lin LH: Adenovirus pneumonia
complicated with acute respiratory distress syndrome: A case
report. Medicine (Baltimore). 94(e776)2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Foong Ng K, Kee Tan K, Hong Ng B, Nair P
and Ying Gan W: Epidemiology of adenovirus respiratory infections
among hospitalized children in Seremban, Malaysia. Trans R Soc Trop
Med Hyg. 109:433–439. 2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Calvo C, García-García ML, Sanchez-Dehesa
R, Román C, Tabares A, Pozo F and Casas I: Eight years prospective
study of adenoviruses infections in hospitalized children.
Comparisons with other respiratory viruses. PLoS One.
10(e0132162)2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Baserga M and Chan B: Hematochezia and
thrombocytopenia in a 3-day-old infant: Congenital adenoviral
infection. J Neonatal Perinatal Med. 11:335–338. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Schmitz H, Wigand R and Heinrich W:
Worldwide epidemiology of human adenovirus infections. Am J
Epidemiol. 117:455–466. 1983.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Li J, Lu X, Jiang B, Du Y, Yang Y, Qian H,
Liu B, Lin C, Jia L, Chen L and Wang Q: Adenovirus-associated acute
conjunctivitis in Beijing, China, 2011-2013. BMC Infect Dis.
18(135)2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Nasr VG, Raman L, Barbaro RP, Guner Y,
Tonna J, Ramanathan K, Pappalardo F, Thiagarajan RR and Alexander
PMA: ELSO Registry Scientific Oversight Committee. Highlights from
the Extracorporeal Life Support Organization Registry: 2006-2017.
ASAIO J. 65:537–544. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Stentz MJ, Kelley ME, Jabaley CS,
O'Reilly-Shah V, Groff RF, Moll V and Blum JM: Trends in
extracorporeal membrane oxygenation growth in the United States,
2011-2014. ASAIO J. 65:712–717. 2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Prodhan P, Bhutta AT, Gossett JM, Stroud
MH, Rycus PT, Bratton SL and Fiser RT: Extracorporeal membrane
oxygenation support among children with adenovirus infection: A
review of the Extracorporeal Life Support Organization registry.
ASAIO J. 60:49–56. 2014.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ronchi A, Doern C, Brock E, Pugni L and
Sánchez PJ: Neonatal adenoviral infection: A seventeen year
experience and review of the literature. J Pediatr.
164:529–535.e1-4. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Gavin PJ and Katz BZ: Intravenous
ribavirin treatment for severe adenovirus disease in
immunocompromised children. Pediatrics. 110(e9)2002.PubMed/NCBI View Article : Google Scholar
|