Introduction
Contrast-induced encephalopathy (CIE) is an acute
and reversible transient neurological disorder induced by a
contrast agent. This occurs as a rare neurological complication
following the intravascular injection of a contrast agent. The
incidence of CIE is ~0.3-1%; however, this increases to 4% when
hypertonic contrast agents are used (1). The clinical manifestations of CIE vary,
such as intracranial hemorrhage, cortical blindness, epileptic
seizure, etc.; however, cognitive dysfunction has not been reported
to date, at least to the best of our knowledge. The present study
describes the case of a patient with cognitive decline as the main
clinical manifestation of CIE.
Case report
The patient described herein was a 57-year-old
female, who presented with recurrent headaches for >1 month and
was admitted to the Guangdong Second Provincial General Hospital.
Although she had a 4-year history of hypertension, she was not
taking anti-hypertensive drugs regularly. In 2016, she underwent
right internal carotid artery stent implantation and left internal
carotid aneurysm embolization in another hospital. Moreover, a left
internal carotid artery dissection aneurysm was found without any
positive signs upon a hospital physical examination. Subsequently,
an auxiliary examination after admission revealed that the
creatinine level was 79 µmol/l and the alanine aminotransferase
level was 41 U/l. An abdominal ultrasonography revealed fatty liver
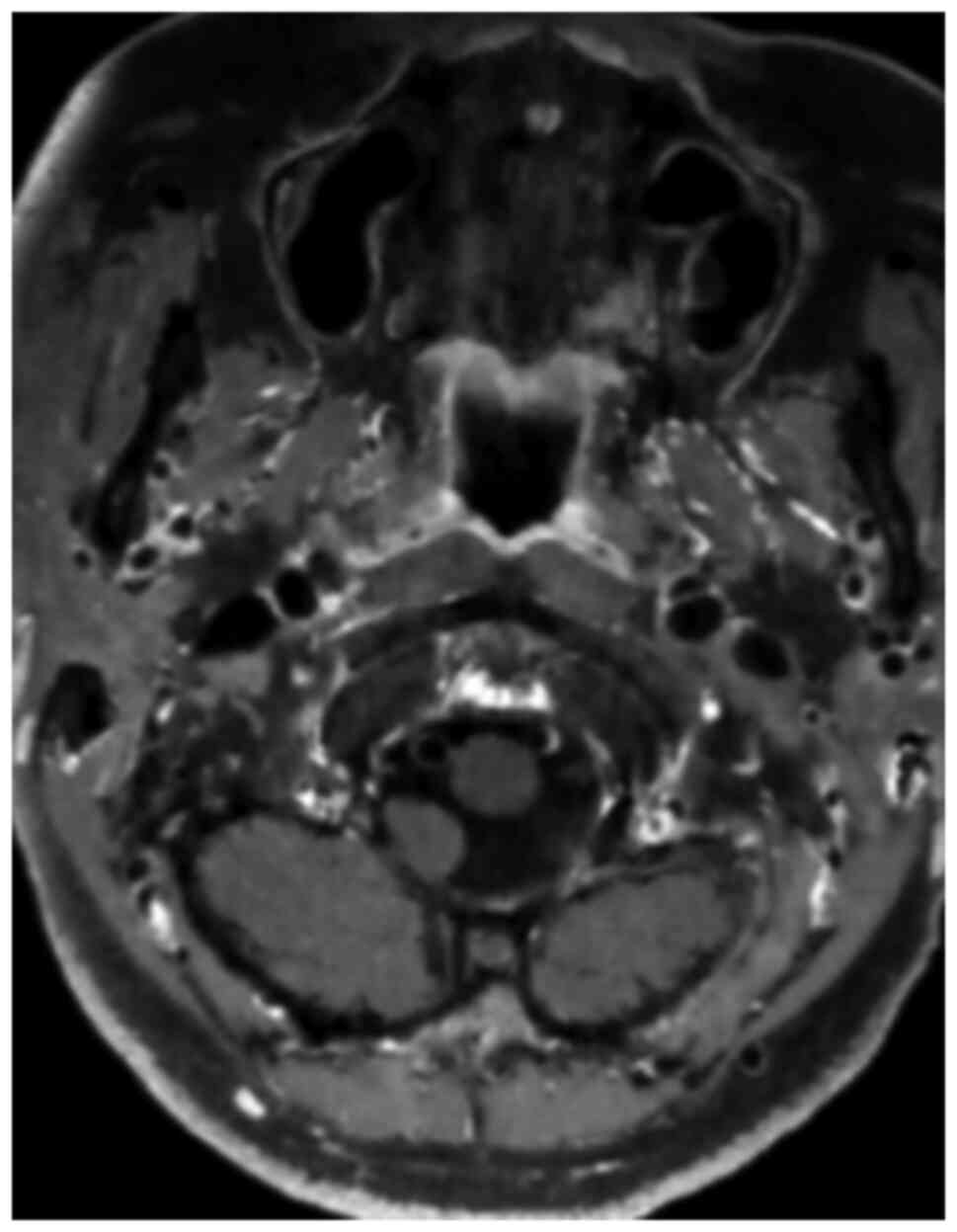
and slight effusion in the right kidney. Cerebral vascular wall
imaging demonstrated the bilateral internal carotid artery with a
double lumen in the C1 segment (Fig.
1) and a left internal carotid artery C6 segment aneurysm
surgery was performed. A summary of the general information of the
patient is presented in Table I.
 | Table IGeneral information of the
patient. |
Table I
General information of the
patient.
| Characteristic |
Features/information |
|---|
| Sex | Female |
| Age | 57 years old |
| Physical
examination | No positive
signs |
| Laboratory tests | Creatinine level, 79
µmol/l |
| | Alanine
aminotransferase level, 41 U/l |
| Imaging | Cerebral vascular
wall imaging revealed a bilateral internal carotid artery with a
double lumen in the C1 segment |
| Medical history | |
|
Chief
complaint | Recurrent headaches
for >1 month |
|
Previous
history | She underwent right
internal carotid artery stenting and left internal carotid artery
aneurysm embolization 4 years prior |
Nonetheless, bilateral internal carotid artery
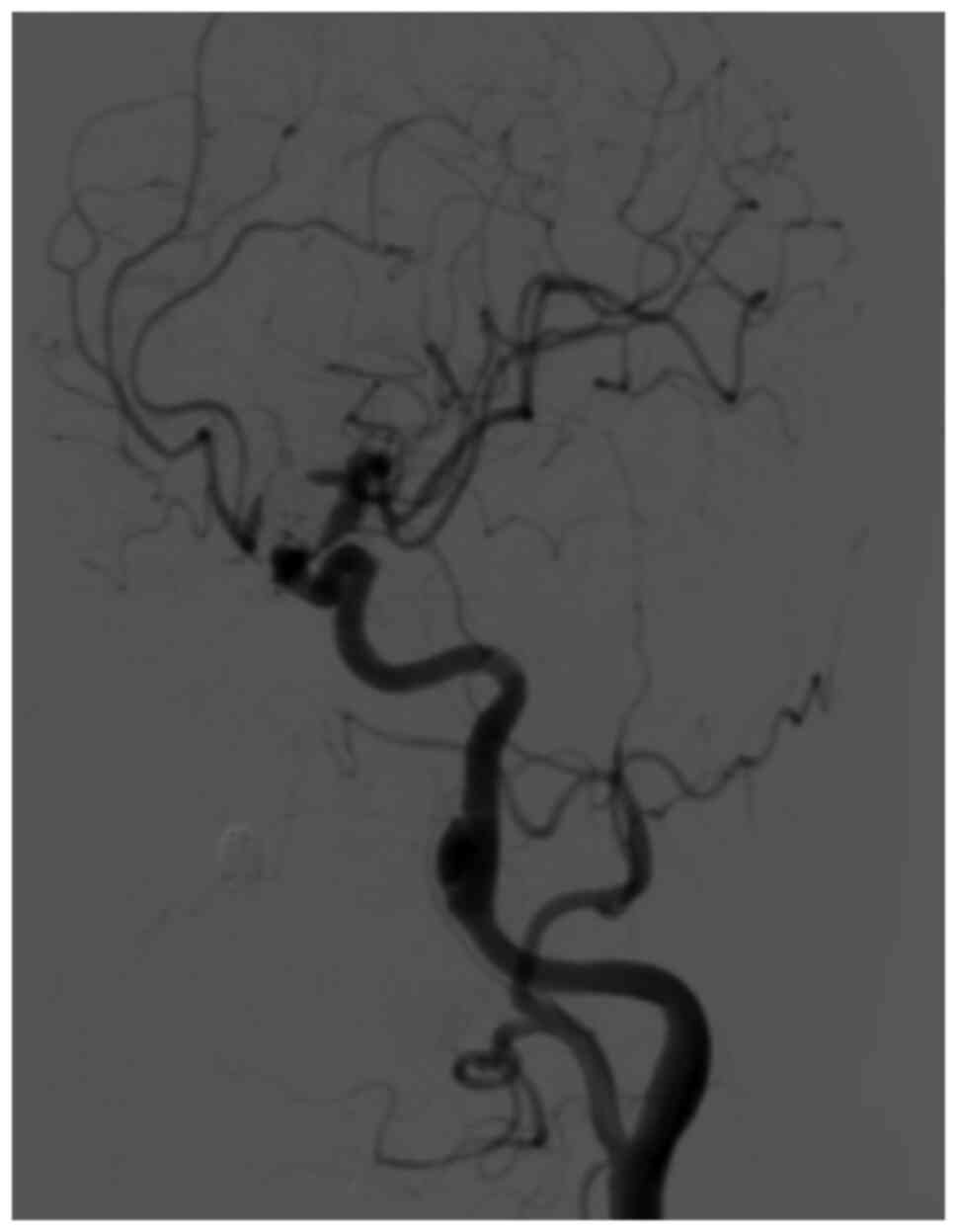
dissection was not excluded. A cerebral angiography was performed
to determine whether there was dissection and whether further
interventional treatment was required; the patient then underwent a
cerebral angiography with 130 ml iohexol (the iodine concentration
of the iohexol was 300 mg/ml) under local anesthesia, which
revealed a dissecting aneurysm in the C1 segment of the left
internal carotid artery (Fig. 2).
The surgery was successfully completed; however, the patient then
suffered from headaches, vomiting and visual impairment, with a
slight decrease in cognitive function 5 min later. A physical
examination revealed that the visual acuity of both eyes had
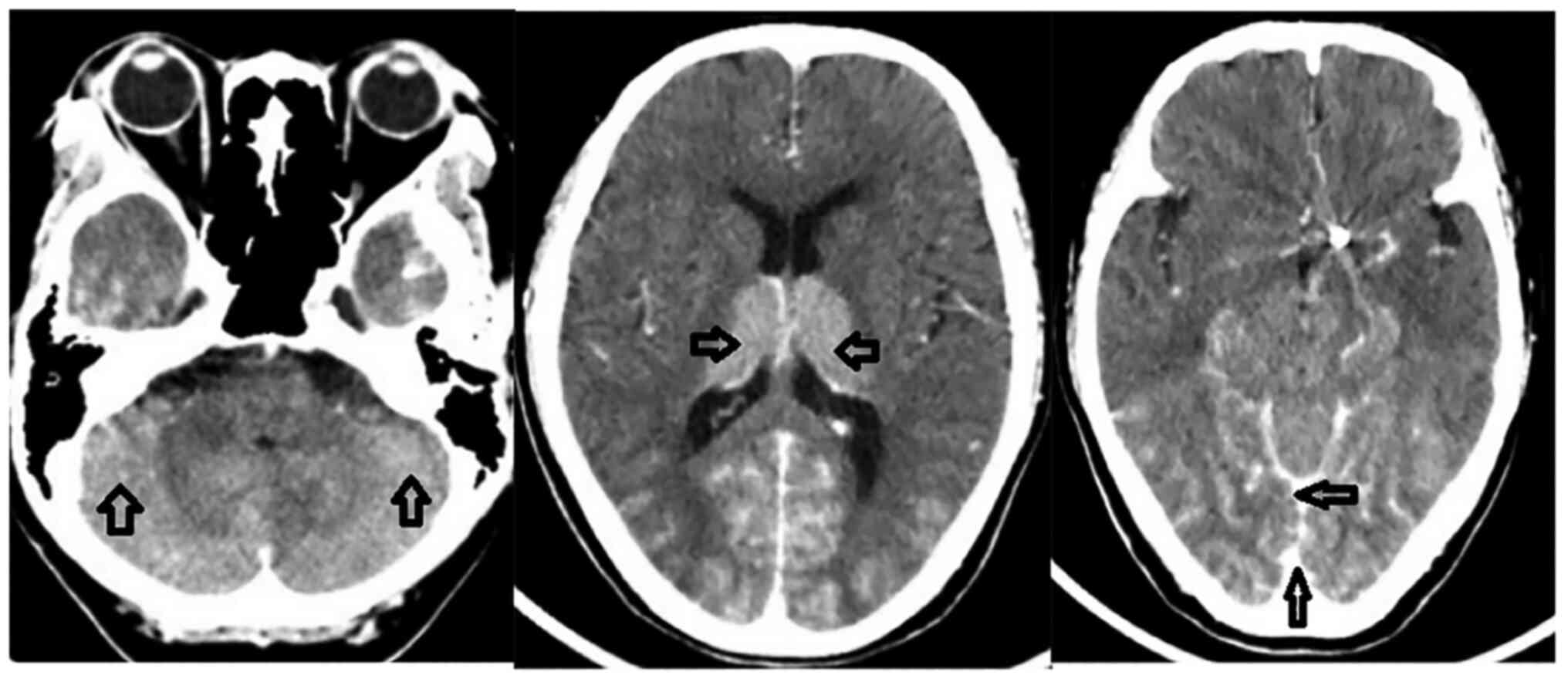
notably decreased, with only light sensation. The emergency
craniocerebral CT scan then revealed an increased density in the
bilateral cerebellum, thalamus, sulcus and cistern (Fig. 3). The probability of CIE was thus
considered, due to the symptoms, signs and CT examination. Thus,
the patient was administered anti-emetic (metoclopramide, 10 mg),
anti-inflammatory (dexamethasone, 5 mg), vasospasm relief
(nimodipine, 10 mg) and intracranial pressure reduction (mannitol,
25 g) treatment. The head CT scan then revealed the resolution of
high-density in the aforementioned areas (Fig. 4) at 24 h after the surgery. On the
third day after surgery, the visual acuity of the patient had
gradually recovered; however, the development of cognitive function
was not evident, which was manifested by occasional incomplete
answers. The Mini-Mental State Examination score (2) was 16 points, and combined with the
condition of illiteracy, the patient could be diagnosed with mild
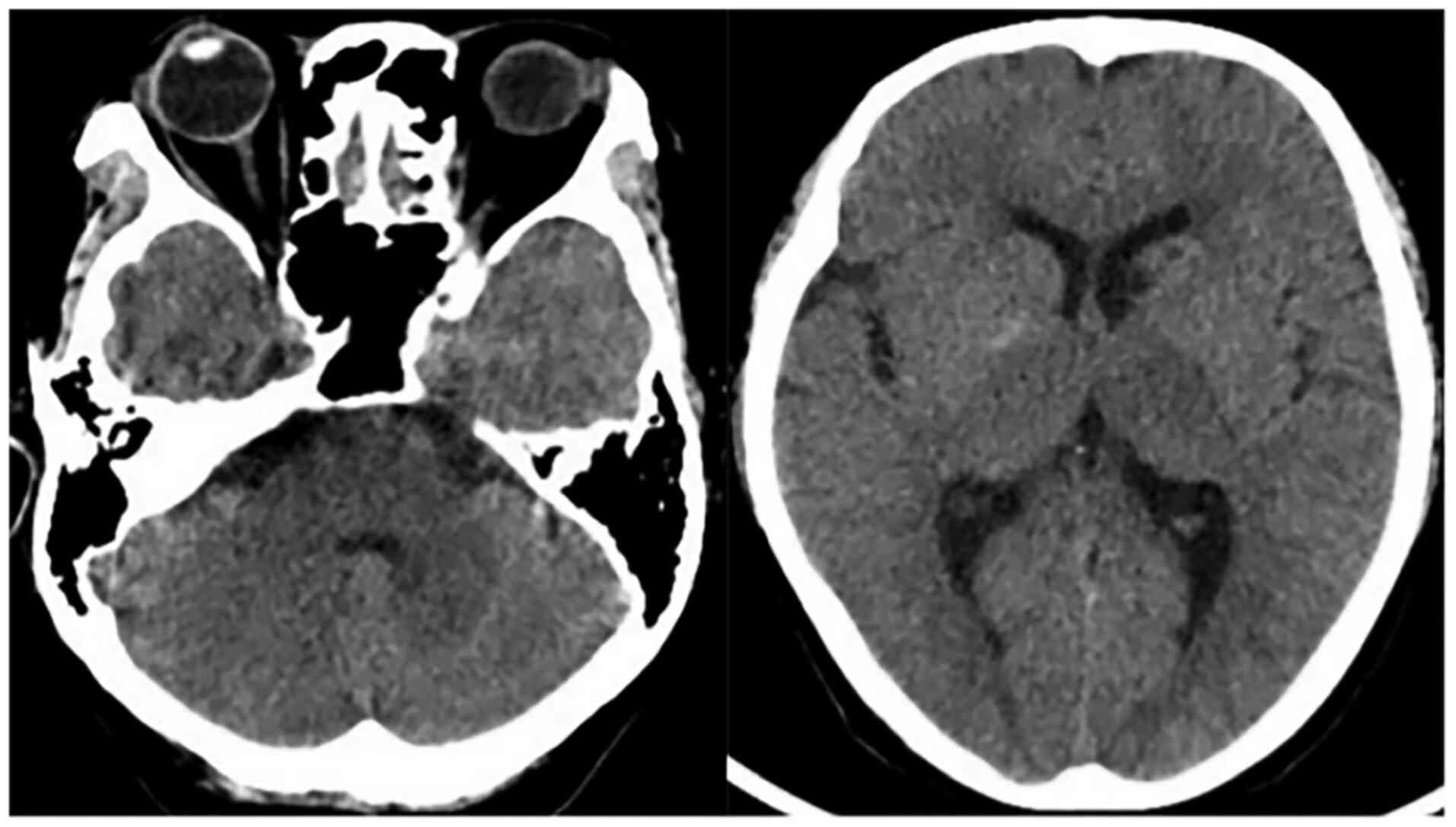
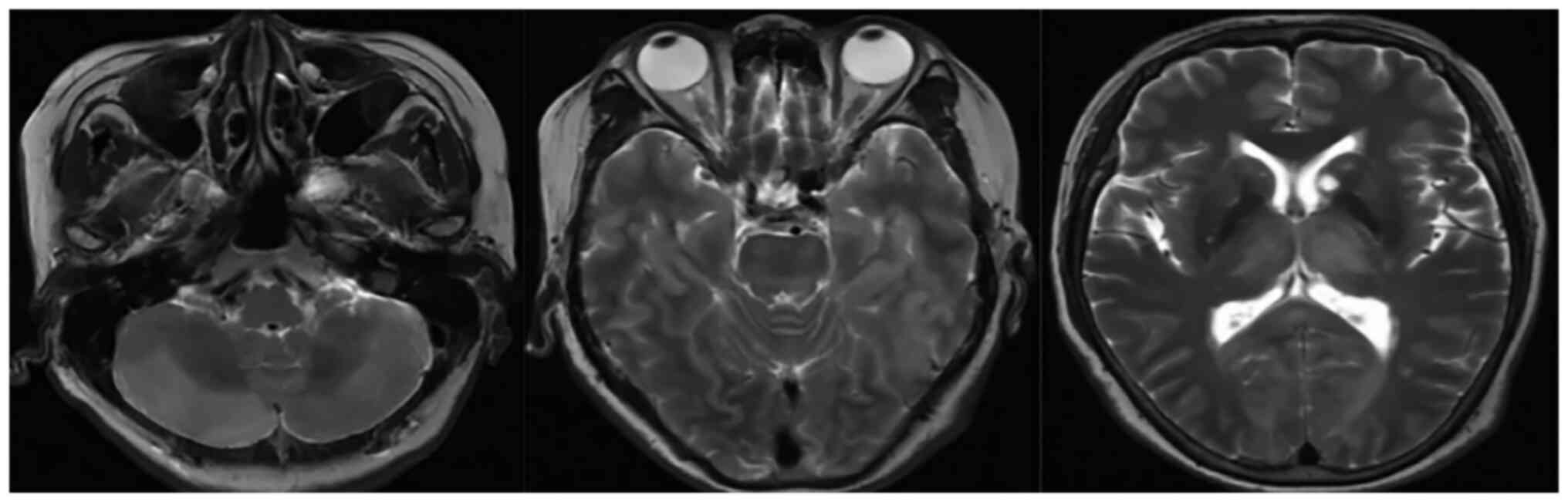
dementia. The MRI examination of the brain revealed multiple
abnormal signal shadows in the bilateral thalamus,
occipito-temporal lobe, corpus callosum, brainstem and bilateral
cerebellar hemispheres (Fig. 5).
Therefore, the administration of an intra-arterial injection of
metoclopramide (10 mg) for anti-emetic treatment, dexamethasone (5
mg) for reducing the inflammatory response, nimodipine (10 mg) pump
for vasospasm relief, mannitol (25 g) for intracranial pressure
reduction and other treatments was continued. At 17 days after the
surgery, the cognitive function of the patient gradually recovered
and she was discharged from the hospital.
Discussion
Contrast encephalopathy is an unusual neurological
complication and its pathogenesis remains unclear. According to
previous studies (3-5) the
widely accepted theoretical mechanism is set around the destruction
of the blood-brain barrier and the chemical properties of the
contrast agents which may be as follows: i) the contrast medium
disrupts the blood-brain barrier temporarily and enters the
cerebrospinal fluid, increasing the osmotic pressure of the
cerebrospinal fluid, and leading to impaired brain function. The
majority of contrast agents have an osmolality range of 1.2-1.8
mOsm/l, compared with 0.3 mOsm/l for normal blood, which may open
the normal endothelial cell tight junctions of the blood-brain
barrier (6,7). ii) Contrast agents exert a direct toxic
effect on nerve cells and affect their function. iii) Iodine
contrast agents affect the secretion of functional substances
regulating vasomotor and causes vasospasm. In this case, the
cognitive decline of the patient is considered to be related to the
neurological deficits caused by the edema of nerve cells. Moreover,
the risk factors of CIE are also not clear. Some studies have
suggested that the most common factors related to the occurrence of
CIE include the male sex, an advanced age, hypertension, diabetes,
renal function impairment, the contrast agent dose, etc. (8,9). CIE can
occur at any age; however, patients who are older are more likely
to suffer from this condition, although the incidence for each age
group has not yet been determined. At the same time, the male sex
and hypertension may be the main risk factors for CIE.
CIE may occur within minutes or hours of the
application of the contrast agent. Generally, normal conditions
will resume within 1-3 days; however, a small portion of patients
do not return to a normal state until several weeks after the
application of the contrast agent (8). There is as yet no clear explanation
available for this phenomenon. In addition, very few fatal CIE
cases have been reported (10). The
clinical manifestations of CIE are diverse, such as intracranial
hemorrhage, meningitis, cortical blindness, epileptic seizure, etc.
(11). The typical imaging
manifestations of CIE are brain parenchyma, subarachnoid contrast
enhancement and brain edema (11);
the majority of patients gradually return to a normal state. In the
case presented herein, bilateral cortical blindness and cognitive
decline occurred within a few minutes following digital subtraction
angiography, and cortical blindness gradually recovered within 3
days after the surgery; however, the recovery of cognitive function
was not evident. The occurrence of cortical blindness has been
demonstrated in previous reports of CIE cases (8), although the occurrence of cognitive
decline is very rare. In the present study, combined with the MRI
results of the patient, it was considered that this cognitive
decline was caused by neurological dysfunction. The head CT
examination revealed that the density of the brain parenchyma,
sulcus and cistern had increased; however, the head CT
re-examination 24 h after the surgery indicated that the
high-density shadow in the aforementioned areas was attenuated or
had disappeared, which was in line with the typical imaging
manifestations of CIE.
Although the majority of patients with CIE have a
favorable outcome, often with a complete resolution of symptoms,
once a diagnosis of CIE is made, aggressive treatment should be
administered immediately to prevent irreversible events. Current
treatment options include the promotion of contrast agent
excretion, the reduction of cerebral edema, and anti-inflammatory
and symptomatic supportive therapy (8). In the case described herein, the
clinical and imaging manifestations of the patient gradually
improved following the aforementioned combination of supportive
treatments.
In conclusion, CIE is a rare neurological
complication caused by contrast agents and generally presents as a
transient, reversible neurologic disorder. The earlier diagnosis
and treatment are crucial for the better prognosis of patients.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YL designed the study, and wrote, edited and
reviewed the manuscript, and processed the figures. XL designed the
study. HL identified the disease and provided guidance. FW provided
the imaging data. ZZ collected the surgical data. SL managed the
patient and provided the clinical data. All authors have read and
approved the final manuscript. XL and FW confirm the authenticity
of all the raw data.
Ethics approval and consent to
participate
The institutional Review Board of Guangdong Second
Provincial General Hospital approved the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Potsi S, Chourmouzi D, Moumtzouoglou A,
Nikiforaki A, Gkouvas K and Drevelegas A: Transient contrast
encephalopathy after carotid angiography mimicking diffuse
subarachnoid haemorrhage. Neurol Sci. 33:445–448. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Folstein MF, Folstein SE and McHugh PR:
‘Mini-mental state’. A practical method for grading the cognitive
state of patients for the clinician. J Psychiatr Res. 12:189–198.
1975.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Yu J and Dangas G: Commentary: New
insights into the risk factors of contrast-induced encephalopathy.
J Endovasc Ther. 18:545–546. 2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Junck L and Marshall WH: Neurotoxicity of
radiological contrast agents. Ann Neurol. 13:469–484.
1983.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Rapoport S, Bookstein JJ, Higgins CB,
Carey PH, Sovak M and Lasser EC: Experience with metrizamide in
patients with previous severe anaphylactoid reactions to ionic
contrast agents. Radiology. 143:321–325. 1982.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Rapoport SI and Levitan H: Neurotoxicity
of X-ray contrast media. Relation to lipid solubility and
blood-brain barrier permeability. Am J Roentgenol Radium Ther Nucl
Med. 122:186–193. 1974.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Grainger R: Osmolality of intravascular
radiological contrast media. Br J Radiol. 53:739–746.
1980.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Liu MR, Jiang H, Li XL and Yang P: Case
report and literature review on low-osmolar, non-ionic iodine-based
contrast-induced encephalopathy. Clin Interv Aging. 15:2277–2289.
2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chu YT, Lee KP, Chen CH, Sung PS, Lin YH,
Lee CW, Tsai LK, Tang SC and Jeng JS: Contrast-induced
encephalopathy after endovascular thrombectomy for acute ischemic
stroke. Stroke. 51:3756–3759. 2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zhao W, Zhang J, Song Y, Sun L, Zheng M,
Yin H, Zhang J, Wang W and Han J: Irreversible fatal
contrast-induced encephalopathy: A case report. BMC Neurol.
19(46)2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Leong S and Fanning NF: Persistent
neurological deficit from iodinated contrast encephalopathy
following intracranial aneurysm coiling. A case report and review
of the literature. Interv Neuroradiol. 18:33–41. 2012.PubMed/NCBI View Article : Google Scholar
|