Introduction
Smokeless tobacco (SLT) poses a significant public
health concern in the Indian subcontinent, with India being widely
regarded as the global epicenter of SLT usage (1). SLT, which does not involve combustion,
is commonly employed intranasally or intraorally, predominantly in
the form of snuffs or by chewing tobacco leaves (2). Specific varieties of SLT differ across
various regions, resulting in a plethora of associated health
risks. Presently, a multitude of SLT options are available,
featuring diverse flavors and chewing habits, such as betel quid,
khaini, mawa, pan masala plain and gutka (3). The prevalence and utilization of
tobacco among young adults, specifically those aged ~30 years, are
substantial. These demographics account for ~12% of global
tobacco-related deaths, including the use of tobacco products and
smokeless alternatives (4). Notably,
the latter is considered to be a comparatively safe option for
continuous cigarette smoking. It is crucial to recognize that
tobacco is a significant risk factor for various chronic ailments,
such as cancer, respiratory conditions and cardiovascular diseases
(5).
The International Agency for Research on Cancer
(IARC) has classified SLT as a group 1 carcinogen (6). Notably, tobacco-specific
N-nitrosamines, namely
4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone, nitrosonornicotine
(NNN), N-nitrosoanatabine and N-nitrosoanabasine play a pivotal
role in the production of free radicals (6). Concerning their corresponding oxidative
stress actions, SLT extract is more harmful and results in
oxidative tissue damage and apoptosis. Furthermore, it has been
noted that the alkaline conditions present during betel nut chewing
promote the generation of free radicals (7). At low or moderate levels, reactive
oxygen species (ROS) and reactive nitrogen species (RNS) play
crucial roles in the maturation of cellular structures and serve as
tools for the host defense system. However, the excessive
production of free radicals and oxidants leads to a phenomenon
known as oxidative stress, which is a detrimental process capable
of significantly modifying cell membranes and various structures,
including proteins, lipids, lipoproteins and DNA (8). The administration of SLT at a low dose
over a prolonged period of time can trigger oxidative stress,
leading to detrimental effects on bodily tissues. These effects may
play a role in the toxicity and carcinogenicity associated with the
use of SLT (9).
Glutathione (GSH), an ubiquitous tripeptide thiol,
is a vital intracellular and extracellular protective antioxidant.
The intracellular and whole blood concentrations of GSH are in the
millimolar range, whereas the plasma concentration is in the
micromolar range accounting for ~0.4% of the total blood GSH levels
(10). Of note, GSH is detected at
high concentrations (5 mM) in the majority of cells. It plays a
crucial role in shielding cellular macromolecules from endogenous
and exogenous ROS and RNS. GSH directly scavenges diverse oxidants,
such as superoxide anions, hydroxyl radicals, nitric oxide and
carbon radicals (11). Studies have
indicated that antioxidants exert their safeguarding effects by
reducing oxidative DNA impairment and inhibiting the initiation and
progression of carcinogenesis (12,13).
This notion serves as a significant gauge for evaluating the
mechanisms of antioxidant defense in the context of malignancy.
In a blood sample with a pH of 7.4, ~69% of nicotine
exists in an ionized state, 31% remains unionized and <5%
attaches to plasma proteins (14).
The occurrence of hypoalbuminemia in patients with oral cancer may
be attributed to the effects of free radical-mediated protein
oxidation and the subsequent reduction in the protective
antioxidant defense mechanism (15).
The levels of oxidized proteins in plasma are noteworthy indicators
of oxidative stress originating from free radicals. Protein
oxidation plays a crucial role in the pathogenesis of oral cancers.
Hyperproteinemia, which manifests as cachexia, is a common
occurrence in oral malignancies. Therefore, serum proteins could
potentially function as pivotal diagnostic and prognostic markers
for oral premalignant diseases and oral malignancies. Researchers
have examined the associations between enzymes, proteins and
glycoproteins, and have found significant changes in the levels of
protein biomarkers in blood serum (16). Additionally, SLT use leads to a
significant decrease in serum albumin levels and alterations in
liver enzyme levels. This is likely due to the damage and
destruction of the liver tissue caused by the components of SLT,
which has been proven to activate microsomal enzymes in liver cells
(2). Therefore, the aim of the
present study was to evaluate and compare the levels of blood GSH,
total plasma protein and albumin in SLT users with precancerous and
cancerous lesions, compared to non-tobacco users.
Patients and methods
The present cross-sectional observational study was
conducted at the Department of Public Health Dentistry, Jawahar
Medical Foundation's ACPM Dental College (Dhule, India) between
January, 2022 and December, 2022. Approval was obtained from the
Institutional Ethics Committee of Jawahar Medical Foundation
Annasaheb Chudaman Patil Memorial Dental College Dhule
(EC/NEW/INST/2022/2959/Y22/212). Written informed consent was
obtained from all participants after the study protocol was
explained to them. The present study was conducted in accordance
with the principles of the Declaration of Helsinki and followed the
Strengthening the Reporting of Observational studies (STROBE)
guidelines.
Sample size calculation
The sample size was calculated using G*Power
software (latest ver. 3.1.9.7; Heinrich-Heine-Universität
Düsseldorf, Düsseldorf, Germany). The standard deviation was taken
from a previous study, to detect the effect size of 0.5 mg/dl in of
GSH in the oral cancer and control groups (17). The power of the study was 80%, with a
95% confidence interval, and type 1 error of 5%. The sample size
was calculated as 60 patients per group.
Selection criteria
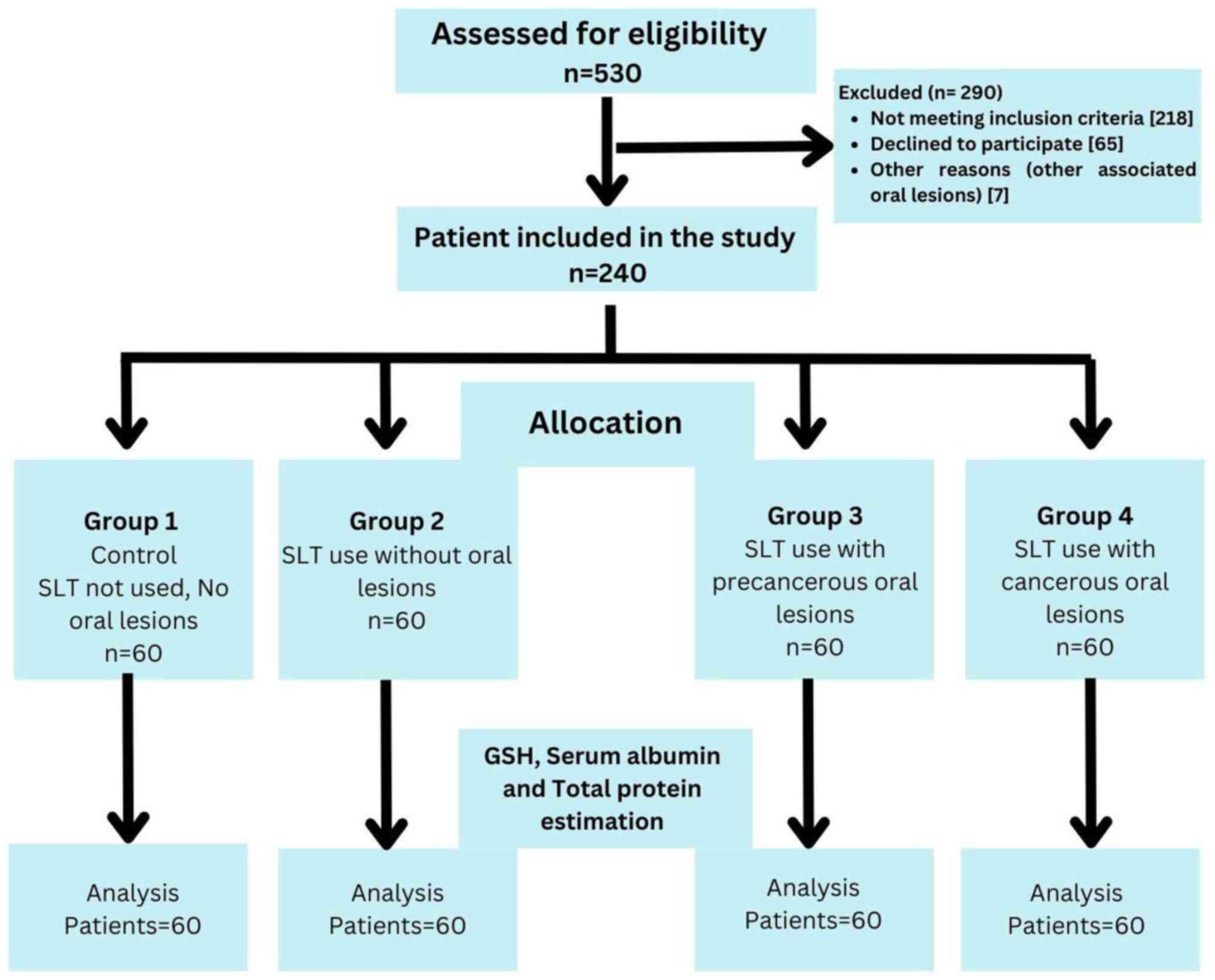
A total of 1,500 patients were screened in cancer
awareness camps in the district of Dhule to select a sample of 240
patients aged 30-60 years, based on the selection criteria of the
study. In total, 240 patients were divided into four groups as
follows: Group 1 (control group) comprised 60 healthy individuals
with no history of SLT use; group 2 comprised 60 healthy
individuals with at least a 1-year history of SLT use, but without
the occurrence of any oral precancerous or cancerous lesions; group
3 comprised 60 clinically and histopathologically confirmed
individuals with oral precancerous lesions who had not received any
prior treatment with at least a 1-year history of SLT use; and
group 4 comprised 60 clinically and histopathologically confirmed
individuals with oral squamous cell carcinoma without metastasis,
who had not received any prior treatment with at least a year
history of SLT use. Individuals >30 years of age who were
willing to participate in the study and had provided written
informed consent were included in the study. Individuals with any
systemic disease, cancer patients undergoing treatment or
radiotherapy, pregnant or lactating females, individuals using
tobacco smoking, those taking any medications for >3 months,
those with a previous history of malignancy or a history of
antioxidant medication, and those taking corticosteroids over the
past 6 months were excluded from the study. A flow diagram of the
study groups and selection process is presented in Fig. 1.
Procedure
The participants were instructed to fast overnight.
The following day, between 7 and 9 a.m., a total of 5 ml blood was
drawn from the mid-cubital vein with the necessary aseptic
precautions in a 5-ml disposable syringe and transferred to sterile
tubes containing ethylenediaminetetraacetic acid (EDTA) to prevent
coagulation. The blood was centrifuged at 1,007 x g for 7 min and
at a temperature of 8 to 12˚C in the centrifuge. The plasma and
buffy coat were separated, and plasma was used for the estimation
of glutathione, whereas serum was used for the estimation of total
protein and albumin. GSH, total protein and albumin levels were
estimated by a trained pathologist (the author BA) at the Pathology
Laboratory of ACPM Medical College, Dhule, India. Routine blood
investigations were performed in all patients.
Estimation of GSH
The GSH levels were estimated using the method
described in the study by Beutler et al (18), which is a simple and accurate method
for determining GSH levels in blood. This method is based on the
development of a relatively stable yellow color produced by the
reaction of Ellman's reagent [5,5'-dithiobis-(2-nitrobenzoic acid)
or DTNB reagent (MerckMillipore)] with GSH to form TNB chromophore,
which has a maximum absorbance at 412 nm. The rate of TNB
formation, measured at 412 nm, was proportional to the
concentration of GSH in the sample. The rate of change in
absorbance (∆A412 nm/min) was linear for the convenience and
consistency of the measurement, and was linearly proportional to
the total concentration of GSH. The optical density of the solution
was measured at 412 nm using a spectrophotometer (Manti Lab
Solutions), and the value of GSH was computed as mg/g Hb (18).
Estimation of serum protein
levels
The serum protein levels were estimated using the
Biuret method (19). Biuret reagent
(MerckMillipore) comprised of sodium hydroxide and hydrated copper
sulfate, together with potassium sodium tartrate, the latter of
which was added to chelate and thus stabilized the cupric ions. The
reaction of the cupric ions with the nitrogen atoms involved in
peptide bonds led to displacement of the peptide hydrogen atoms
under alkaline conditions. A tri- or tetra-dentate chelation with
the peptide nitrogen produced a characteristic violet color. The
intensity of the color, which had a maximum absorption at 540 nm
using a spectrophotometer (Manti Lab Solutions), was proportional
to the protein concentration.
Estimation of serum albumin
levels
The serum albumin levels were estimated using the
spectrophotometric method described by Rodkey (20). In this method, the serum was diluted
with a solution of bromocresol green (MerckMillipore) at a
sufficient concentration to allow an essentially linear change in
absorbance with the albumin concentration. Measurements were made
at 615 nm with spectrophotometer (Manti Lab Solutions), where the
absorbance of hemoglobin or bilirubin did not interfere.
Statistical analysis
Data were analyzed using SPSS software version 22
(IBM Corp.). The Shapiro-Wilk test was used to assess the normal
distribution of the data. As the data were normally distributed,
parametric tests, such as one-way analysis of variance (ANOVA),
followed by post hoc analysis with Tukey's test, were used to
assess the differences between the groups. For ordinal data, the
Chi-squared test and Fisher's exact test were used. Regression
analysis was used to assess the associations between variables, and
receiver operating curves (ROC) were used to evaluate biomarker
performance. A value of P≤0.05 was considered to indicate a
statistically significant difference.
Results
Descriptive analysis
The descriptive analysis in the present study did
not reveal any significant differences in the mean age of the
patients in the different the groups (P>0.05), whereas
statistically significant differences were observed in the duration
of SLT use between groups 2, 3 and 4. The maximum duration of SLT
use was 14.15±5.81 years, observed in SLT users who developed oral
cancer (group 4), whereas the minimum duration of SLT use was
6.15±2.35 years, observed in SLT users with precancerous oral
lesions. All groups had non-significant distributions of males and
females, although the habit of SLT use was more prevalent in males
than in females. Gutka chewing was the most common form of SLT,
followed by betel quid chewing. The frequency of SLT chewing/per
day was the highest in group 4, followed by groups 3 and 2. The
majority of the participants in group 2 chewed SLT for <5 min
(70%), whereas the majority of the participants in group 4 chewed
SLT for >5 min (63%), as depicted in Table I. Post hoc analysis revealed that
there were statistically notable disparities in the duration of SLT
use between groups 2 and 3 in comparison to group 4. Nevertheless,
there were no significant differences between groups 2 and 3.
Statistically significant disparities were observed among all
experimental groups in terms of the frequency of SLT use (Table II).
 | Table IDemographic details of the study
participants. |
Table I
Demographic details of the study
participants.
| Variable | Group 1 (Control, no
SLT) | Group 2 (SLT use, no
lesions) | Group 3 (SLT use,
precancerous lesions) | Group 4 (SLT use,
cancerous lesions) | P-value |
|---|
| Age, years (mean ±
SD)a | 44.9±5.67 | 44.4±4.27 | 45.6±5.84 | 45.5±5.87 | 0.595 (NS) |
| Sex, n
(%)b | | | | | |
|
Male | 39 (65%) | 40 (66%) | 42 (70%) | 43 (71%) | 0.856 (NS) |
|
Female | 21 (35%) | 20 (34%) | 18 (30%) | 17 (29%) | |
| Type of SLT use, n
(%)c | | | | | |
|
Betel
quid | NA | 18 (30%) | 12 (20%) | 20 (33%) | 0.7710 (NS) |
|
Gutka | NA | 30 (50%) | 36 (60%) | 32 (53%) | |
|
Mawa | NA | 5 (9%) | 4 (6%) | 2 (4%) | |
|
Naswar | NA | 4 (6%) | 6 (10%) | 4 (6%) | |
|
Other | NA | 3 (5%) | 2 (4%) | 2 (4%) | |
| Duration of SLT
use, years (mean ± SD)a | NA | 7.05±2.15 | 6.15±2.35 | 14.51±5.81 | 0.0001d |
| Frequency of SLT
use (per day) (mean ± SD)a | NA | 5.78±3.32 | 7.38±2.12 | 8.96±2.08 | 0.0001d |
| Retention of SLT, n
(%)b | | | | | |
|
<5
min | NA | 42 (70%) | 28 (47%) | 22 (37%) | 0.009d |
|
>5
min | NA | 18 (30%) | 32 (53%) | 38 (63%) | |
 | Table IIPost hoc analysis of the duration and
frequency of SLT use in groups 2, 3 and 4. |
Table II
Post hoc analysis of the duration and
frequency of SLT use in groups 2, 3 and 4.
| Parameter and
pairwise comparison | P-value |
|---|
| Duration of SLT use
(years; mean ± SD) | |
|
Group 2 vs.
group 3 | 0.403 (NS) |
|
Group 2 vs.
group 4 | 0.001a |
|
Group 3 vs.
group 4 | 0.001a |
| Frequency of SLT
use (per day; mean ± SD) | |
|
Group2 vs.
group 3 | 0.002a |
|
Group 2 vs.
group 4 | 0.002a |
|
Group 3 vs.
group 4 | 0.002a |
The most common precancerous lesion in group 3 was
leukoplakia (60%), followed by oral submucosal fibrosis (23%) and
erythroplakia (17%). The majority of the participants in group 4
had well-differentiated oral squamous cell carcinoma (52%)
(Table III). The majority of oral
lesions were present on the buccal mucosa, labial mucosa and
gingivobuccal corridor.
 | Table IIIHistopathological details of lesions
in groups 3 and 4. |
Table III
Histopathological details of lesions
in groups 3 and 4.
| Group | Histopathological
grade |
|---|
| Group 3 (SLT use,
precancerous lesions; n=60) | Mild dysplasia | Moderate
dysplasia | Severe
dysplasia | Carcinoma in
situ |
|
Leukoplakia
(n=36, 60%) | n=24 (66%,
40%) | n=8 (23%, 12%) | n=3 (8%, 5%) | n=1 (3%, 2%) |
|
Erythroplakia
(n=10, 17%) | n=3 (0.3%, 5%) | n=4 (0.4%, 7%) | n=2 (0.2%, 3%) | n=1 (0.1%, 2%) |
|
Oral
submucous fibrosis (n=14, 23%) | n=9 (64%, 15%) | n=4 (29%, 7%) | n=1 (7%, 2%) | n=0 (0%) |
| |
Well-differentiated | Moderately
differentiated | Poorly
differentiated | Metastasis |
| Group 4 (SLT use,
cancerous lesions; n=60) | | | | |
|
Squamous
cell carcinoma (n=60) | 32 (52%) | 20 (34%) | 8 (14%) | 0 (0%) |
Hematological analysis
Statistically significant differences were observed
in all hematological parameters between the groups. Group 4
exhibited a significant decrease in mean hemoglobin levels, total
platelet count and total leukocyte count, followed by groups 3 and
2 compared to the control group. The total red blood cell (RBC)
count, erythrocyte sedimentation rate, mean corpuscular hemoglobin
and high-sensitivity C-reactive protein (hs-CRP) levels were
significantly higher in group 4, followed by groups 3 and 2,
compared with the control group (Table
IV). Post hoc analysis revealed significant differences between
groups 1 and 4 in terms of Hb levels. Moreover, there were
significant differences between all groups, except for groups 3 and
4, in terms of the total RBC count. Additionally, significant
differences were found between groups 1 and 3, groups 1 and 4, and
groups 2 and 4 in terms of total platelet count. Furthermore, there
were significant differences between all groups, except groups 2
and 3, as well as groups 2 and 4, in terms of the total leukocyte
count. Moreover, significant differences were found between all
groups, except for groups 2 and 3, as well as groups 3 and 4, in
terms of MCH. Lastly, there were significant differences between
all groups, except for Groups 1 and 2, in terms of hs-CRP levels
(Table IV).
 | Table IVComparison of hematological
investigations of the study groups using one-way ANOVA followed by
post hoc analysis. |
Table IV
Comparison of hematological
investigations of the study groups using one-way ANOVA followed by
post hoc analysis.
| Parameter | Group | Mean ± SD | P-value | Pairwise
comparison | P-value |
|---|
| Hb% | Group 1 | 14.20±3.02 | 0.015a | Group 1 vs. group
2 | 0.740 |
| | Group 2 | 13.74±1.98 | | Group 1 vs. group
3 | 0.130 |
| | Group 3 | 13.20±1.94 | | Group 1 vs. group
4 | 0.010a |
| | Group 4 | 12.80±2.92 | | Group 2 vs. group
3 | 0.640 |
| | | | | Group 2 vs. group
4 | 0.170 |
| | | | | Group 3 vs. group
4 | 0.820 |
| Total RBCs
(106/µl) | Group 1 | 4.81±0.02 | 0.001a | Group 1 vs. group
2 | 0.001a |
| | Group 2 | 5.14±0.57 | | Group 1 vs. group
3 | 0.001a |
| | Group 3 | 5.67±0.62 | | Group 1 vs. group
4 | 0.001a |
| | Group 4 | 5.82±0.32 | | Group 2 vs. group
3 | 0.001a |
| | | | | Group 2 vs. group
4 | 0.001a |
| | | | | Group 3 vs. group
4 | 0.265 |
| ESR (mm/h) | Group 1 | 13.24±6.24 | 0.183 | Group 1 vs. group
2 | 0.320 |
| | Group 2 | 15.43±7.21 | | Group 1 vs. group
3 | 0.540 |
| | Group 3 | 14.95±6.96 | | Group 1 vs. group
4 | 0.160 |
| | Group 4 | 15.91±7.76 | | Group 2 vs. group
3 | 0.980 |
| | | | | Group 2 vs. group
4 | 0.980 |
| | | | | Group 3 vs. group
4 | 0.870 |
| Total platelets
(lakhs/mm3) | Group 1 | 3.24±1.21 | 0.001a | Group 1 vs. group
2 | 0.801 |
| | Group 2 | 2.91±2.14 | | Group 1 vs. group
3 | 0.001a |
| | Group 3 | 1.98±2.32 | | Group 1 vs. group
4 | 0.001a |
| | Group 4 | 1.89±2.12 | | Group 2 vs. group
3 | 0.054 |
| | | | | Group 2 vs. group
4 | 0.028a |
| | | | | Group 3 vs. group
4 | 0.994 |
| TLC
(103/mm3) | Group 1 | 9.23±1.91 | 0.001a | Group 1 vs. group
2 | 0.007 |
| | Group 2 | 8.24±1.22 | | Group 1 vs. group
3 | 0.001a |
| | Group 3 | 7.91±1.61 | | Group 1 vs. group
4 | 0.001a |
| | Group 4 | 7.23±1.84 | | Group 2 vs. group
3 | 0.699 |
| | | | | Group 2 vs. group
4 | 0.005a |
| | | | | Group 3 vs. group
4 | 0.117 |
| MCH (pg) | Group 1 | 29.62±1.92 | 0.001a | Group 1 vs. group
2 | 0.001a |
| | Group 2 | 32.58±1.41 | | Group 1 vs. group
3 | 0.001a |
| | Group 3 | 33.41±1.86 | | Group 1 vs. group
4 | 0.001a |
| | Group 4 | 34.06±1.84 | | Group 2 vs. group
3 | 0.052 |
| | | | | Group 2 vs. group
4 | 0.001a |
| | | | | Group 3 vs. group
4 | 0.186 |
| hs-CRP (mg/l) | Group 1 | 2.56±1.53 | 0.001a | Group 1 vs. group
2 | 0.238 |
| | Group 2 | 3.63±1.98 | | Group 1 vs. group
3 | 0.001a |
| | Group 3 | 12.21±3.43 | | Group 1 vs. group
4 | 0.001a |
| | Group 4 | 16.89±4.56 | | Group 2 vs. group
3 | 0.001a |
| | | | | Group 2 vs. group
4 | 0.001a |
| | | | | Group 3 vs. group
4 | 0.001a |
Inferential analysis
Analysis between the groups using ANOVA revealed
statistically significant differences in the mean values of GSH,
serum protein and albumin levels (P<0.001). The maximum levels
of reduced GSH (9.61±0.86 mg/Hb) were observed in healthy
individuals with no history of SLT use (group 1), and the minimum
levels (2.63±0.43) were found in group 4, as shown in Table V. Post hoc analysis using Tukey's
test revealed a statistically significant difference in serum GSH
levels between all groups (P<0.001; Table VI). No significant differences were
observed in the serum protein levels between groups 1 and 2, groups
1 and 4, and groups 2 and 4, whereas no significant differences
were observed in the serum albumin levels between groups 2 and 3,
as shown in Table VI.
 | Table VComparative analysis of the different
variables using ANOVA. |
Table V
Comparative analysis of the different
variables using ANOVA.
| Group | Average level of
GSH in blood, mg/Hb (mean ± SD) | Average serum level
of total protein, g/dl (mean ± SD) | Average serum level
of albumin, g/dl (mean ± SD) |
|---|
| Group 1 (n=60) | 9.61±0.86 | 6.56±0.41 | 4.25±0.29 |
| Group 2 (n=60) | 6.56±0.70 | 6.46±0.34 | 3.81±0.28 |
| Group 3 (n=60) | 4.68±0.74 | 5.74±0.42 | 3.68±0.37 |
| Group 4 (n=60) | 2.63±0.43 | 6.51±0.44 | 2.97±0.35 |
| Value from
ANOVA | 0.001a | 0.001a | 0.001a |
 | Table VIIntra-group comparisons of different
variables using Tukey's post-hoc test. |
Table VI
Intra-group comparisons of different
variables using Tukey's post-hoc test.
| Groups | Average serum level
of GSH mg/Hb | Average serum level
of total protein g/dl | Average serum level
of albumin g/dl |
|---|
| Group 1 vs. group
2 | 0.001a | 0.595 (NS) | 0.001a |
| Group 1 vs. group
3 | 0.001a | 0.001a | 0.001a |
| Group 1 vs. group
4 | 0.001a | 0.908 (NS) | 0.001a |
| Group 2 vs. group
3 | 0.001a | 0.001a | 0.124 (NS) |
| Group 2 vs. group
4 | 0.001a | 0.938 (NS) | 0.001a |
| Group 3 vs. group
4 | 0.001a | 0.001a | 0.001a |
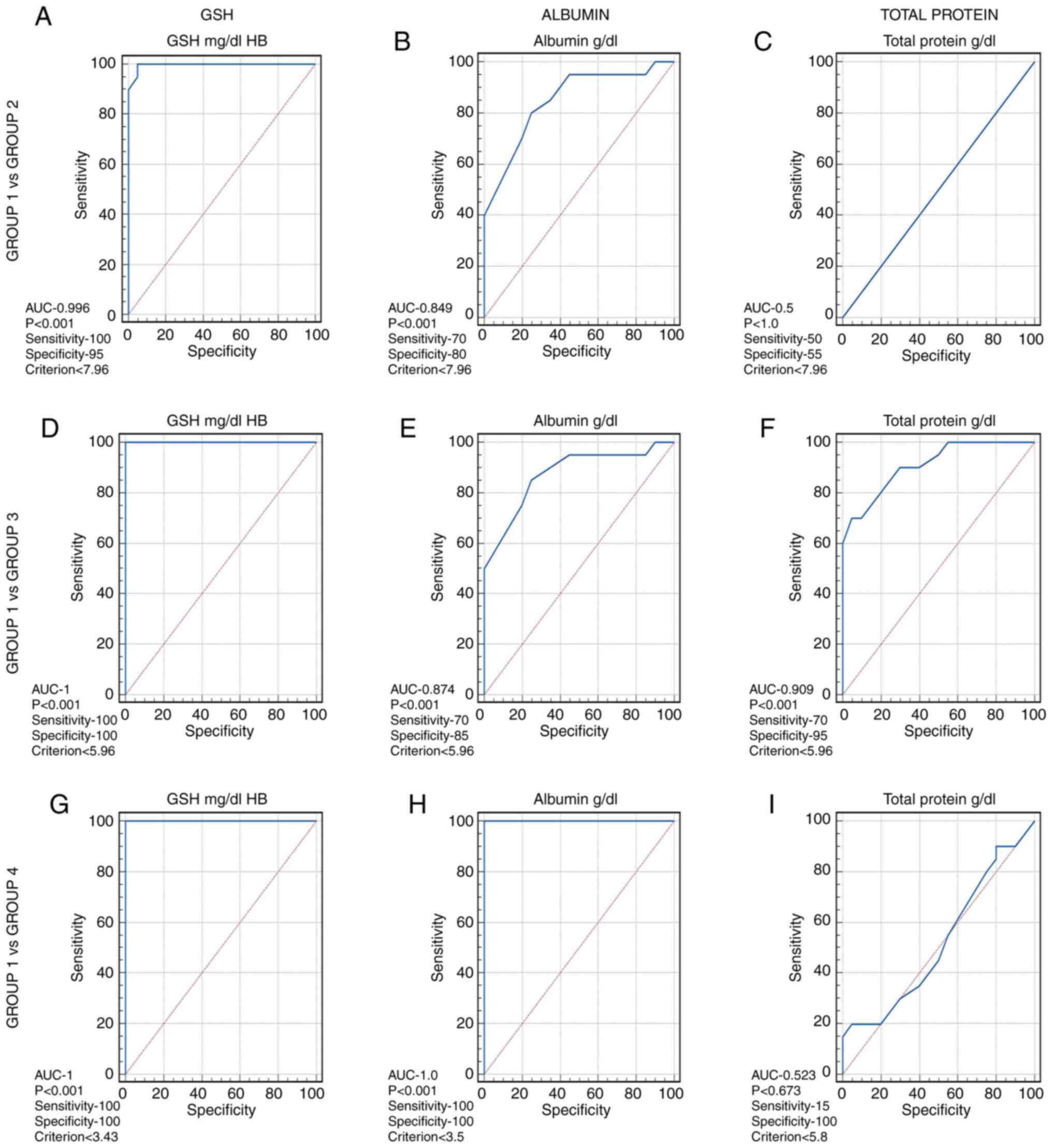
Results of ROC analysis
ROC analysis was conducted to determine the
sensitivity and specificity of GSH in the blood and albumin and
total protein levels in the serum at different thresholds. The
optimum threshold values of GSH was found to be ≤7.96 for group 2
(Fig. 2A), ≤5.96 for group 3
(Fig. 2D) and ≤3.43 for group 4
(Fig. 2G), and having a sensitivity
and specificity of 100%. Albumin had a threshold value of ≤7.9 for
group 2 with 70% sensitivity and 80% specificity (Fig. 2B), ≤5.9 for group 3 with 70%
sensitivity and 85% specificity (Fig.
2E), and ≤3.5 for group 4 (Fig.
2H), with 100% sensitivity and 100% specificity. The threshold
values of total serum protein levels were >7.9 for group 2
(Fig. 2C) at 50% sensitivity and 55%
specificity, ≤5.96 for group 3 (Fig.
2F) at 70% sensitivity and specificity of 95%, and ≤5.81, at
15% sensitivity and 100% specificity for group 4 (Fig. 2I). This indicates that GSH and
albumin levels are reliable diagnostic biomarkers, whereas the
total protein content is a weak biomarker.
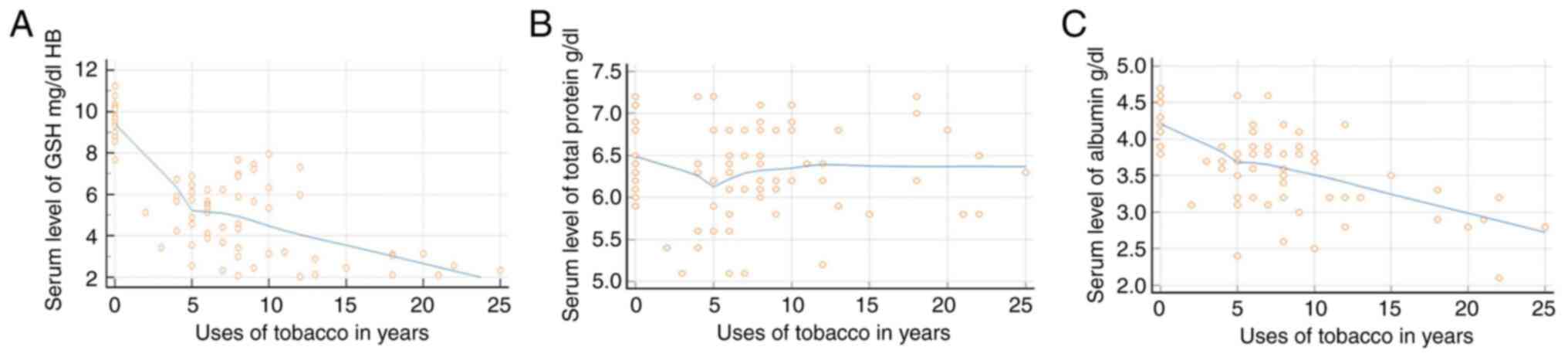
Regression analysis
Regression analysis revealed that the GSH levels in
the blood and serum albumin levels decreased as the duration of SLT
use increased; however, the total protein level only exhibited
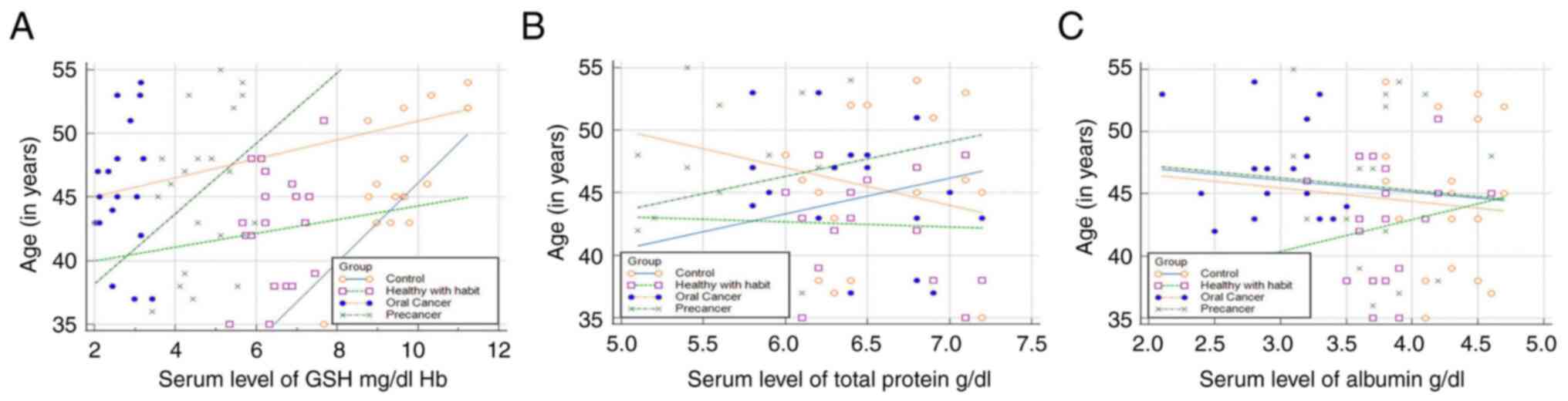
slight fluctuations in the initial phases of tobacco use (Fig. 3). The GSH levels exhibited an
increasing trend with age, indicating that the GSH levels increased
in all groups as the age of the patients increased. The total
protein level decreased in group 4, whereas groups 3 exhibited an
increasing trend with age. The serum albumin levels exhibited an
increasing trend with age only in group 2 (Fig. 4).
Discussion
In healthy humans, a state of equilibrium is
maintained between oxidants and antioxidants. Nevertheless, in an
atypical state, an over-abundance of oxidizing agents is generated,
leading to the inhibition of antioxidant defenses. Consequently,
this results in a deviation in the ratio favoring pro-oxidants
(8). SLT alters the activity of
antioxidants in saliva. Oxidative stress can function as a
biomarker for the diagnosis and prediction of damage and the
abnormal growth of oral tissues. Additionally, it can serve as an
early indicator to prevent abnormal changes in the oral cavity
(9).
The present study found the increased use of SLT in
males aged 45 years, which was in agreement with the findings of
previous studies (1,2,4,7). There is substantial evidence to
indicate that the enactment of smoke-free laws, the augmentation of
smoking taxes and the prevalence of a favorable societal attitude
towards SLT during working hours have been positively associated
with the rate of SLT consumption among males (4). Substances such as lime and catechu,
which are employed in the formulation of SLT commodities,
participate in the generation of ROS within the cellular
environment (14).
In the present study, GSH levels were higher in
non-tobacco users than in tobacco users. Tobacco snuffs can
generate free radicals, causing protein nitration, lipid
peroxidation and DNA adduct formation. Exposure to SLT leads to the
generation of ROS, which are considerably higher in SLT users than
in non-tobacco users (12,13). GSH, a vital antioxidant that
dissolves in water, is produced through the synthesis of the amino
acids, glycine, glutamate and cysteine. GSH exhibits an elevated
redox potential and effectively operates as a potent antioxidant
(10). It facilitates the
detoxification and breakdown of ROS, thereby inhibiting cellular
oxidative damage and cancer development.
Herein, GSH levels were found to be decreased in SLT
users with precancerous and cancerous lesions. The lowest GSH
levels were observed in SLT users with oral cancer. The findings of
the present study are consistent with those of previous studies
(5,17,21,22).
Glutathione consists of a reduced form known as GSH and a form that
has been oxidized, referred to as GSH disulfide (GSSG). The ability
of GSH to be regenerated is directly related to the redox state of
the GSSG-GSH couple (GSSG/2GSH). As a result, GSH provides primary
support for intracellular ‘redox homeostasis’ or ‘redox buffering’
capacity (22). When subjected to
excessive xenobiotics, including carcinogens, there is an increased
utilization of GSH for conjugation, which in turn leads to
detoxification. This process results in a decrease in the GSH/GSSG
ratio, rendering GSH less available, and consequently reducing the
body's defense against free radicals. Notably, the depletion of GSH
is adequate to sensitize cancer cells to both oxidative and
nitrative stress. This sensitization leads to DNA damage.
Consequently, DNA degeneration can occur, which can activate
carcinogens and ultimately initiate and progress into cancer.
Depletion of GSH may sensitize tumors to chemotherapy and
radiotherapy (10,16). ROC analysis revealed GSH to be a
reliable biomarker for predicting oral cancer. In the present
study, the mean GSH levels decreased with age and increased with
the duration of SLT use, which is in agreement with the findings of
previous studies (23,24).
Serum proteins have been widely acknowledged for
their antioxidant characteristics owing to the presence of unbound
thiol groups. Of all the proteins, albumin stands out as the most
efficacious and abundant extracellular antioxidant. In the present
study, it was found to be a reliable biomarker. The findings of the
present study indicated a significant decrease in serum albumin
levels in SLT users compared to healthy controls, and it decreased
further in patients with precancerous and cancerous lesions. This
finding is in accordance with that of previous studies (9,15,25). The
effect of free radicals formed by SLT has been demonstrated to
induce modifications to DNA bases, causing fractures in the DNA
strand, impairing the integrity of tumor suppressor genes and
promoting the expression of proto-oncogenes. Additionally, these
free radicals can disrupt antioxidant defense mechanisms. Albumin
is considered to possess the ability to function as an antioxidant,
which may be attributed to the abundant presence of sulfhydryl and
free thiol groups within the molecular structure of albumin. Oral
cancers are associated with inflammatory mediators, such as IL-6
and TNF, which potentially exert their effects through dual
mechanisms, namely, by enhancing the extravasation of albumin
across the capillary endothelium at the tumor site and dampening
the hepatic production of albumin. A decrease in serum albumin
levels can lead to a reduction in the sequestration of unbound
reactive molecules, thereby enhancing the probability of
deleterious cellular damage that can induce cancer development
(26).
In the present study, serum protein levels also
decreased with the use of SLT and during the precancerous phase.
However, there was no significant difference in serum protein
levels between the control group and SLT users with oral cancer.
This finding is in agreement with the findings of previous studies
(13,27). The decrease in the serum protein
concentration can potentially be elucidated in relation to the
inflammatory response associated with oral malignancies. The total
serum protein level was found to be a weak biomarker in the present
study.
The findings of the present study revealed that
reduced glutathione and serum albumin are reliable biomarkers for
the early diagnosis and the assessment of the prognosis of oral
premalignant and malignant lesions, and for planning specific
treatment strategies for their prevention and management. These
findings are of particular importance for subjects who have the
habit of using SLT. They should be encouraged to quit these habits,
and awareness campaigns should be organized where these biomarkers
can be used efficiently for mass screening.
The altered hematological parameters in SLT users
suggest the selective toxicity of SLT and its components. The
increase in the RBC count in SLT users observed herein indicated
erythropoiesis, which may be due to insufficient pulmonary function
with the long-term use of SLT. Elevated hs-CRP levels also suggest
the presence of chronic inflammation and an increased risk of
cardiovascular disease in SLT users (28).
A limitation of the present study was the evaluation
of GSH, albumin and serum proteins in the blood samples of
patients, but not in salivary samples. The primary challenge
hindering the development of a salivary diagnostic protocol lies in
the fact that although numerous biomarkers identified in the blood
serum can also be detected in saliva, their concentrations are so
minimal that they offer limited value to the diagnostic process.
Moreover, saliva collection is highly sensitive and affects the
salivary biomarker levels. The present study also did not consider
variations in biomarker levels at the different stages of oral
squamous cell carcinoma. The present study evaluated GSH levels in
the blood samples of patients; however, the estimation of GSH
levels in blood and tissue samples of patients could have provided
more information on oxidative stress. As GSH levels have been
associated with oxidative stress (29), future studies should be conducted to
assess ROS, and enzymes such as glutathione-peroxidase, and
catalase at the cellular level. The present study measured the
total serum protein levels, which were found to be weak biomarkers.
Therefore, future prospective studies need to be conducted to
evaluate specific proteins in salivary or serum samples from
patients at different stages of oral cancer.
In conclusion, the findings of the present study
draw a conclusion regarding the impact of oral premalignant and
malignant tumors, on the concentration of GSH and proteins in the
sera in comparison with individuals without any health issues or
tobacco habits, indicating the possibility of utilizing these
changes in GSH and protein profiles to diagnose and predict the
course of oral cancer.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
AN conceived and designed the study. BA and AV
recruited the patients and assessed the variables. PV performed the
data analysis. ABW and AAP assessed the laboratory data and drafted
the manuscript. All authors have reviewed the manuscript, and all
authors have read and approved the final manuscript. AV and PV
confirm the authenticity of all the raw data.
Ethical approval and consent to
participate
Ethical approval for the study was obtained from the
Institutional Ethical Committee at Jawahar Medical Foundation's
Annasaheb Chudaman Patil Memorial Dental College Dhule
(EC/NEW/INST/2022/2959/Y22/212) and the study was conducted in
accordance with the ethical principles outlined in the Declaration
of Helsinki. Written informed consent was obtained from all
participants after the study protocol was explained to them.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Acharya S, Singh S and Bhatia SK:
Association between smokeless tobacco and risk of malignant and
premalignant conditions of oral cavity: A systematic review of
Indian literature. J Oral MaxillofacPathol. 25(371)2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Diaz MC, Kierstead EC, Edwards D, Kim Y,
Rose SW, Emery S, Khatib B, Liu M and Kostygina G: Online tobacco
advertising and current chew, dip, snuff and snus use among youth
and young adults, 2018-2019. Int J Environ Res Public Health.
19(4786)2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Niaz K, Maqbool F, Khan F, Bahadar H,
Hassan FI and Abdollahi M: Smokeless tobacco (paan and
gutkha) consumption, prevalence, and contribution to oral
cancer. Epidemiol Health. 9(e2017009)2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Gupta PC and Ray CS: Smokeless tobacco and
health in India and south Asia. Respirology. 8:419–431.
2003.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Garg S, Pandeshwar P and Padmashree S:
Estimation of serum superoxide dismutase and glutathione peroxidase
levels in tobacco chewers and smokers: A comparative study. J Oral
Med Oral Surg Oral Pathol Oral Radiol. 4:147–154. 2018.
|
|
6
|
IARC Working Group on the Evaluation of
Carcinogenic Risks to Humans. Smokeless tobacco and some
tobacco-specific N-nitrosamines. IARC Monogr Eval Carcinog Risks
Hum. 89:1–592. 2007.PubMed/NCBI
|
|
7
|
Tilashalski K, Rodu B and Cole P: A pilot
study of smokeless tobacco in smoking cessation. Am J Med.
104:456–458. 1998.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Young I and Woodside J: Antioxidants in
health and disease. J Clin Pathol. 54:176–186. 2001.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bagchi M, Bagchi D, Hassoun EA and Stohs
SJ: Subchronic effects of smokeless tobacco extract (STE) on
hepatic lipid peroxidation, DNA damage and excretion of urinary
metabolites in rats. Toxicology. 127:29–38. 1998.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zitka O, Skalickova S, Gumulec J, Masarik
M, Adam V, Hubalek J, Trnkova L, Kruseova J, Eckschlager T and
Kizek R: Redox status expressed as GSH:GSSG ratio as a marker for
oxidative stress in paediatric tumour patients. Oncol Lett.
4:1247–1253. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Pizzorno J: Glutathione! Integr Med
(Encinitas). 13:8–12. 2014.PubMed/NCBI
|
|
12
|
Lobo V, Patil A, Phatak A and Chandra N:
Free radicals, antioxidants and functional foods: Impact on human
health. Pharmacogn Rev. 4:118–126. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Gupta N, Verma K, Nalla S, Kulshreshtha A,
Lall R and Prasad S: Free radicals as a double-edged sword: The
cancer preventive and therapeutic roles of curcumin. Molecules.
25(5390)2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Benowitz NL: Smokeless tobacco as a
nicotine delivery device: Harm or harm reduction? Clin Pharmacol
Ther. 90:491–493. 2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kosova F, Cetin B, Akinci M, Aslan S, Ari
Z, Sepici A, Altan N and Cetin A: Advanced oxidation protein
products, ferrous oxidation in xylenol orange, and malondialdehyde
levels in thyroid cancer. Ann Surg Oncol. 14:2616–2620.
2007.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Patidar KA, Parwani RN and Wanjari SP:
Correlation of salivary and serum IgG, IgA levels with total
protein in oral submucous fibrosis. J Oral Sci. 53:97–102.
2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Rasool M, Khan SR, Malik A, Khan KM, Zahid
S, Manan A, Qazi MH and Naseer MI: Comparative studies of salivary
and blood sialic acid, lipid peroxidation and antioxidative status
in oral squamous cell carcinoma (OSCC). Pak J Med Sci. 30:466–471.
2014.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Beutler E, Duron O and Kelly BM: Improved
method for the determination of blood glutathione. J Lab Clin Med.
61:882–888. 1963.PubMed/NCBI
|
|
19
|
Wokes F and Still BM: The estimation of
protein by the biuret and Greenberg methods. Biochem J. 36:797–806.
1942.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Rodkey FL: Direct spectrophotometric
determination of albumin in human serum. Clin Chem. 11:478–487.
1965.PubMed/NCBI
|
|
21
|
Sharma M, Rajappa M, Kumar G and Sharma A:
Oxidant-antioxidant status in Indian patients with carcinoma of
posterior one-third of tongue. Cancer Biomark. 5:253–260.
2009.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Mohideen K, Sudhakar U, Jeddy N, Sankari
SL, Radhika T and Vani N: Assessment of the anti-oxidant reduced
glutathione in oral squamous cell carcinoma-Systematic review and
meta-analysis. J Oral Maxillofac Pathol. 26(592)2022.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhu Y, Carvey PM and Ling Z: Age-related
changes in glutathione and glutathione-related enzymes in rat
brain. Brain Res. 1090:35–44. 2006.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Begum SF, Nagajothi G, Latha KS, Sandeep
G, Sreekanth B, Kumar CS, Rajendra W and Maddu N: Possible role of
nicotine and cotinine on nitroxidative stress and antioxidant
content in saliva of smokeless tobacco consumers. Pract Lab Med.
12(e00105)2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Metgud R and Patel S: Serum and salivary
levels of albumin as diagnostic tools for oral pre-malignancy and
oral malignancy. Biotech Histochem. 89:8–13. 2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Mohanty V, Subbannayya Y, Patil S, Abdulla
R, Ganesh MS, Pal A, Ray JG, Sidransky D, Gowda H, Prasad TSK and
Chatterjee A: Molecular alterations in oral cancer between tobacco
chewers and smokers using serum proteomics. Cancer Biomark.
31:361–373. 2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
More CB, Shah PH and Venkatesh R:
Estimation of serum protein in oral potentially malignant disorders
and oral malignancy-A cross-sectional study. J Clin Diagn Res.
11:ZC17–ZC19. 2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Shukla AK, Khaitan T, Gupta P and Naik SR:
Smokeless tobacco and its adverse effects on hematological
parameters: A cross-sectional study. Adv Prev Med.
2019(3182946)2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Cheng SB, Liu HT, Chen SY, Lin PT, Lai CY
and Huang YC: Changes of oxidative stress, glutathione, and its
dependent antioxidant enzyme activities in patients with
hepatocellular carcinoma before and after tumor resection. PLoS
One. 12(e0170016)2017.PubMed/NCBI View Article : Google Scholar
|