Introduction
Glioblastoma (GBM) is one of most common malignant
brain tumors in adults (1). The
incidence of GBM in China is also increasing (2). Despite developments in neurosurgery,
chemotherapy and radiation therapy, the prognosis of GBM has not
changed significantly in the last decade. Surgical resection is the
most effective method of therapy for GBM, however the high
frequency of tumor recurrence is the major obstacle for GBM
clinical therapy (3). As the
current treatment modalities of GBM are not satisfactory, new
agents for potential use in GBM clinical therapy have attracted
attention.
Herbal therapies are gradually being recognized and
adopted in clinical cancer therapy, and traditional Chinese
medicine has demonstrated effectiveness in cancer therapy (4–6), not
only by attenuating the side-effects of radical or chemical therapy
but also by exhibiting antitumor efficacy, inhibiting the
recurrence and metastasis of cancer, improving the quality of life
and aiding the recovery of patients.
Flavonoids are polyphenolic compounds which exist in
certain types of green vegetables, fruits and other botanicals. The
significant physiological functions of flavonoids are attracting an
increasing amount of attention. The results of an epidemiological
survey have demonstrated that the intake of vegetables and fruits
with high flavonoid contents reduces the incidence of cancer,
including lung cancer and colon cancer (7). As flavonoids have varied
bioactivities, efficient antitumor activity and low toxicity, the
development of flavonoids is of significant value in cancer therapy
and prevention (8).
Isoliquiritigenin (ISL) is a flavonoid compound with
a variety of bioactivities that is isolated from licorice (a
legume) and shallot (a liliaceae), which are widely used in
traditional Chinese medicine. The antitumor efficacy of ISL against
various types of cancer has previously been demonstrated in
vitro and in vivo(9–11).
In the current study, the antitumor activity of ISL on the glioma
cell line U87 was examined.
Materials and methods
Chemicals
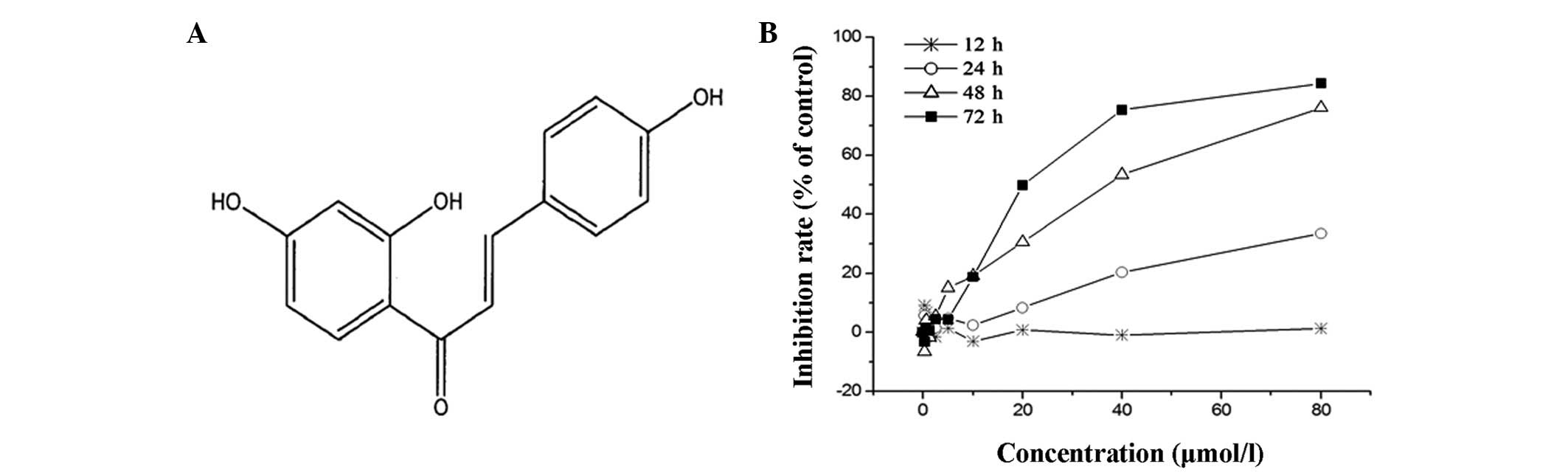
ISL (Fig. 1A) was
purchased from Sigma (St. Louis, MO, USA). It was dissolved in
dimethyl sulfoxide (DMSO) and diluted prior to use. The pan-caspase
inhibitor Z-VAD-FMK was purchased from Beyotime Institute of
Biotechnology (Jiangsu, China). Antibodies against p21, p27,
caspase-3 and β-actin were purchased from Santa Cruz Biotechnology
(Santa Cruz, CA, USA). Horseradish peroxidase (HRP)-conjugated goat
anti-mouse or anti-rabbit IgG were obtained from Beyotime Institute
of Biotechnology. RPMI-1640 medium and fetal bovine serum (FBS)
were supplied by Gibco (Grand Island, NY, USA). All other chemicals
used in this study were biologically pure.
Cell culture and drug treatment
The U87 human glioma cells (American Type Culture
Collection, ATCC; Manassas, VA, USA) were cultured in RPMI-1640
medium supplemented with 10% FBS, 100 U/ml penicillin and 100 μg/ml
streptomycin at 37°C in a humidified atmosphere containing 5%
CO2.
After the cells were grown to sub-confluence, they
were pre-treated with various concentrations of ISL (10–80 μM) or
pan-caspase inhibitor Z-VAD-FMK (25 μM) in RPMI-1640 medium with
10% FBS for different times. Thereafter, the cells were washed
twice with PBS and then cultivated for further assay.
Cell proliferation assay
U87 cells were plated in triplicate in 96-well
plates (Corning Inc., Corning, NY, USA) at a density of 5,000
cells/well. Following overnight cultivation, various concentrations
of ISL were added and cultivation of the cells was continued for
12, 24, 48 and 72 h. The effects on cell growth were determined by
methyl-thiazolyltetrazolium (MTT) assay. Briefly, 20 μl MTT
solution (5 mg/ml) was added to each well and the cells were
incubated for another 4 h at 37°C. Subsequently, 150 μl DMSO was
added to dissolve the MTT formazan and the absorbance was measured
using a microplate reader (Thermo Scientific, Rockford, IL, USA) at
a wavelength of 570 nm.
Cell cycle analysis
U87 cells were plated at a density of
1×106 cells/ml medium in 50-mm diameter dishes. ISL was
added after 24 h of cultivation. The cells were harvested from the
culture dishes 24 h later and stained with 70% ethanol at −2°C.
Prior to FACS analysis, the cells were washed with PBS twice,
resuspended and then incubated with 200 μg/ml RNase A (Sigma) for
60 min at 37°C. Propidium iodide (PI; Sigma) was added at 50 μg/ml
and the cells were incubated in the dark for 15–30 min. The samples
were analyzed using a fluorescence-activated cell sorter (BD
Calibur; BD Biosciences, San Jose, CA, USA).
FITC-Annexin V/PI apoptosis assay
U87 cells were plated at a density of
1×106 cells/ml medium in 50-mm diameter dishes. ISL was
added after 24 h of cultivation. The cells were harvested from the
culture dishes 24 h later by trypsinization. After washing twice
with PBS, the cells were resuspended in 500 μl binding buffer.
Then, 5 μl FITC-labeled Annexin V (Kaiji Biotechnology Ltd.,
Nanjing, China) and 5 μl PI were added. Upon incubation in the dark
for 15 min at room temperature, the samples were analyzed with a
FACScan instrument (BD Calibur).
Preparation of cell lysates
Following treatment with ISL for 24 h, the U87 cells
were washed twice with ice-cold PBS and then scraped and lysed with
RIPA lysis buffer (50 nM Tris, pH 7.4, 150 mM NaCl, 1% Triton
X-100, 1% sodium deoxycholate, 0.1% SDS and 0.05 mM EDTA). A
cocktail of protein inhibitor mix (25 mM NaF, 1 mM
Na3VO4, 20 mM β-glycerophosphate, 20 mM PNPP,
1 mM PMSF, 1 mg/ml leupeptin, 1 mg/ml aprotinin and 1 mg/ml
pepstatin A) was also added to the RIPA lysis buffer. The samples
were then centrifuged at 12,000 × g for 30 min at 4°C and the
supernatants were collected as the total cell extracts. Protein
concentrations were quantified using a BCA protein assay kit
(Pierce, Rockford, IL, USA)
Western blot analysis
Western blot analysis was used to detect p21, p27
and caspase-3, with β-actin as a control. An aliquot of cell lysate
containing 50 μg protein was separated on 12% sodium dodecyl
sulfate-polyacrylamide gel (SDS-PAGE), and then transferred to a
0.45-μm polyvinylidene fluoride membrane. The membranes were
blocked with 5% non-fat milk in Tris-HCl, pH 7.5, and 150 mmol/l
NaCl containing 0.1% Tween-20 for 1 h and then incubated with
individual primary antibodies overnight at 4°C. The blots were then
incubated with HRP-conjugated antibody. Signals were detected using
enhanced chemiluminescence reagents (Millipore, Billerica, MA, USA)
and densitometric analysis was performed with the use of X-ray
film. The expression levels were normalized to β-actin and the
control levels were set to 100%.
Statistical analysis
The results in this study are expressed as the mean
± SD. Treatment effects were compared using the Student's t-test
and P<0.05 was considered to indicate a statistically
significant result.
Results
Inhibitory effects of ISL on the
proliferation of glioma cells
The inhibitory effects of ISL on U87 glioma cells
were assessed by MTT assay. The U87 cells were treated with various
concentrations of ISL for different times and then examined by MTT
assay. As shown in Fig. 1B,
incubation with ISL for 12 h at concentrations of 0–80 μmol/l had
no effect on the viability of the U87 cells. Otherwise, the
inhibitory effects of ISL on the U87 cells increased as the time of
treatment increased.
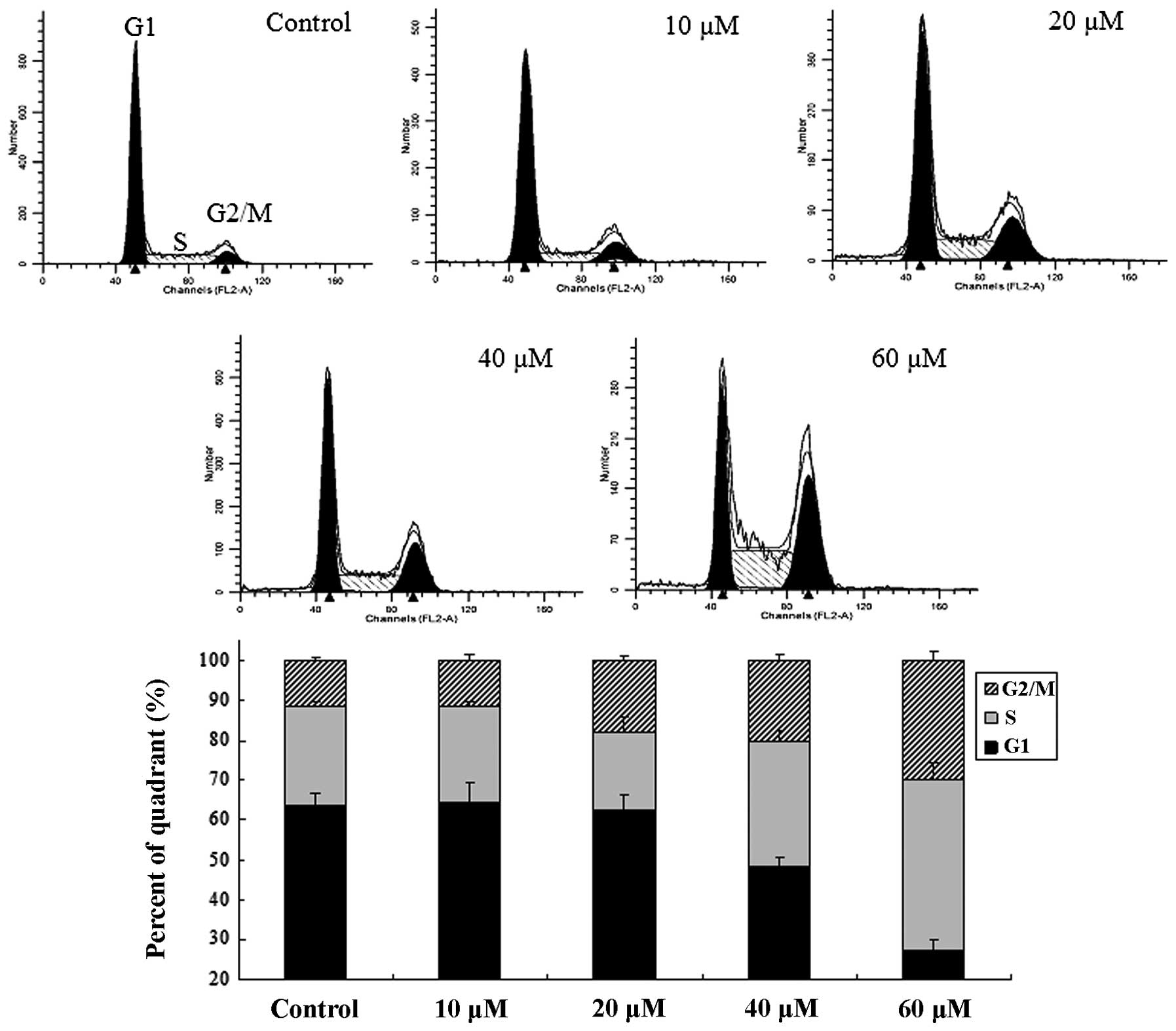
Cell cycle arrest induced by ISL
The effect of ISL on the cell cycle of the U87 cells
was determined by FACS analysis. As shown in Fig. 2, ISL markedly induced S and G2/M
phase arrest in a concentration-dependent manner. ISL at a
concentration of 10 μM had no evident effect on cell cycle arrest.
The levels of S and G2/M arrest increased as the ISL concentration
increased. As the ISL concentration increased from 0 to 60 μmol/l,
an increase in the percentage of cells in the G2/M phase from 11.55
to 29.91% and an increase in the percentage of cells in the S phase
from 24.87 to 42.62% was observed. By contrast, the percentage of
cells in the G1 phase decreased from 63.58 to 24.48% following
treatment with ISL at corresponding doses.
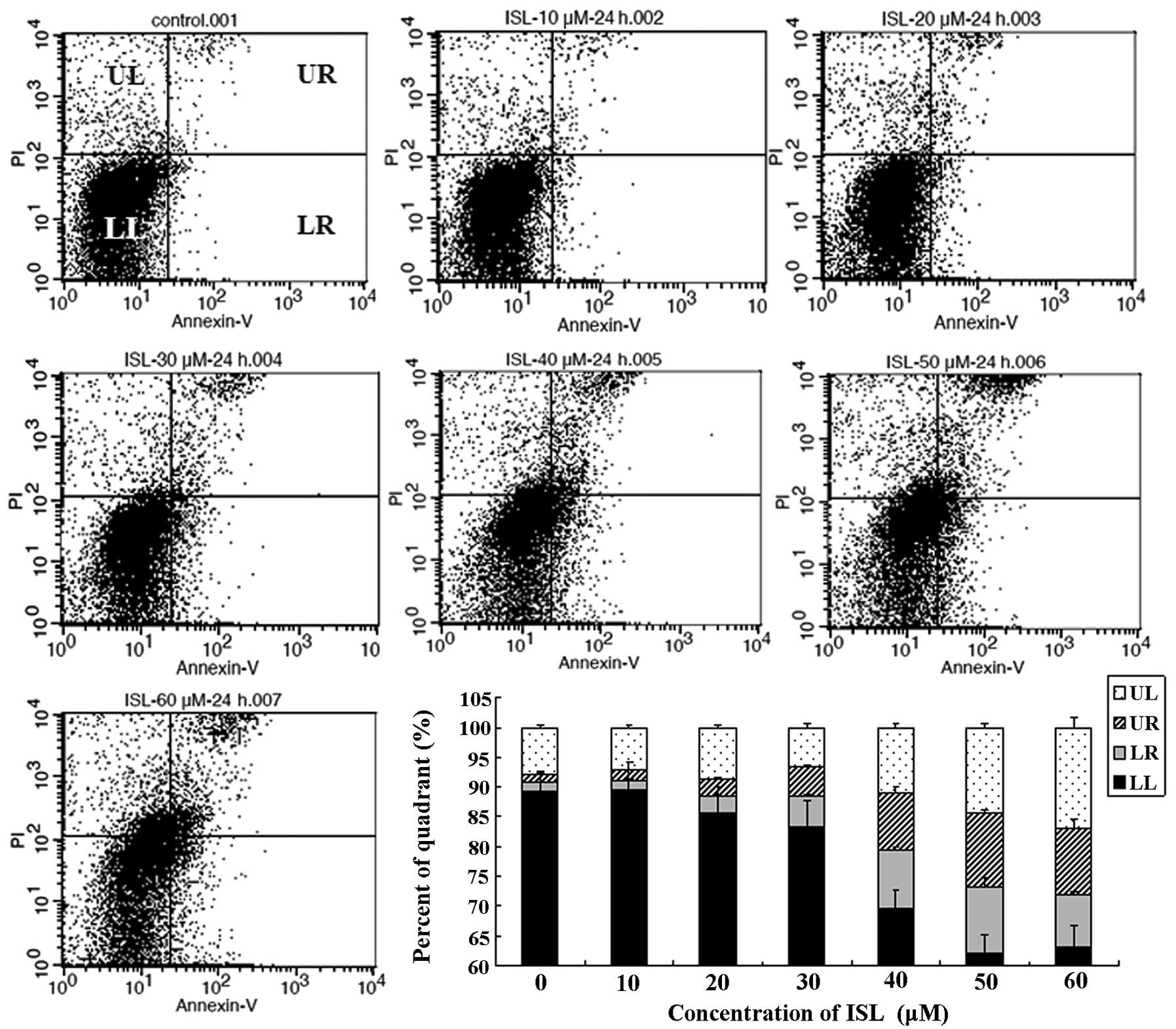
Apoptosis induced by ISL
The apoptosis of the U87 cells induced by ISL was
analyzed by FACS. Annexin V/PI analysis was used to examine the
apoptosis of the U87 cells after 24 h of culturing in the presence
of various concentrations of ISL. The results (Fig. 3) revealed that ISL induced
apoptosis and necrosis of the U87 cells. As the ISL concentration
increased, the percentages of apoptosis and necrosis were increased
dose-dependently. As shown in Fig.
3, as the ISL concentration increased from 0 to 60 μM, the
percentages of UR (secondary apoptotic or late stage necrotic), LR
(early apoptotic) and UL (non-apoptotic necrotic) cells increased
from 1.54 to 11.12%, 1.42 to 8.76% and 7.77 to 16.93%,
respectively. As observed from Fig.
3, the percentages of UR and LR cells did not markedly change
as the ISL concentration was increased from 40 to 60 μM, however
the percentage of UL cells increased as the ISL concentration
increased, which indicated that ISL was able to induce U87 cell
death by a non-apoptotic process.
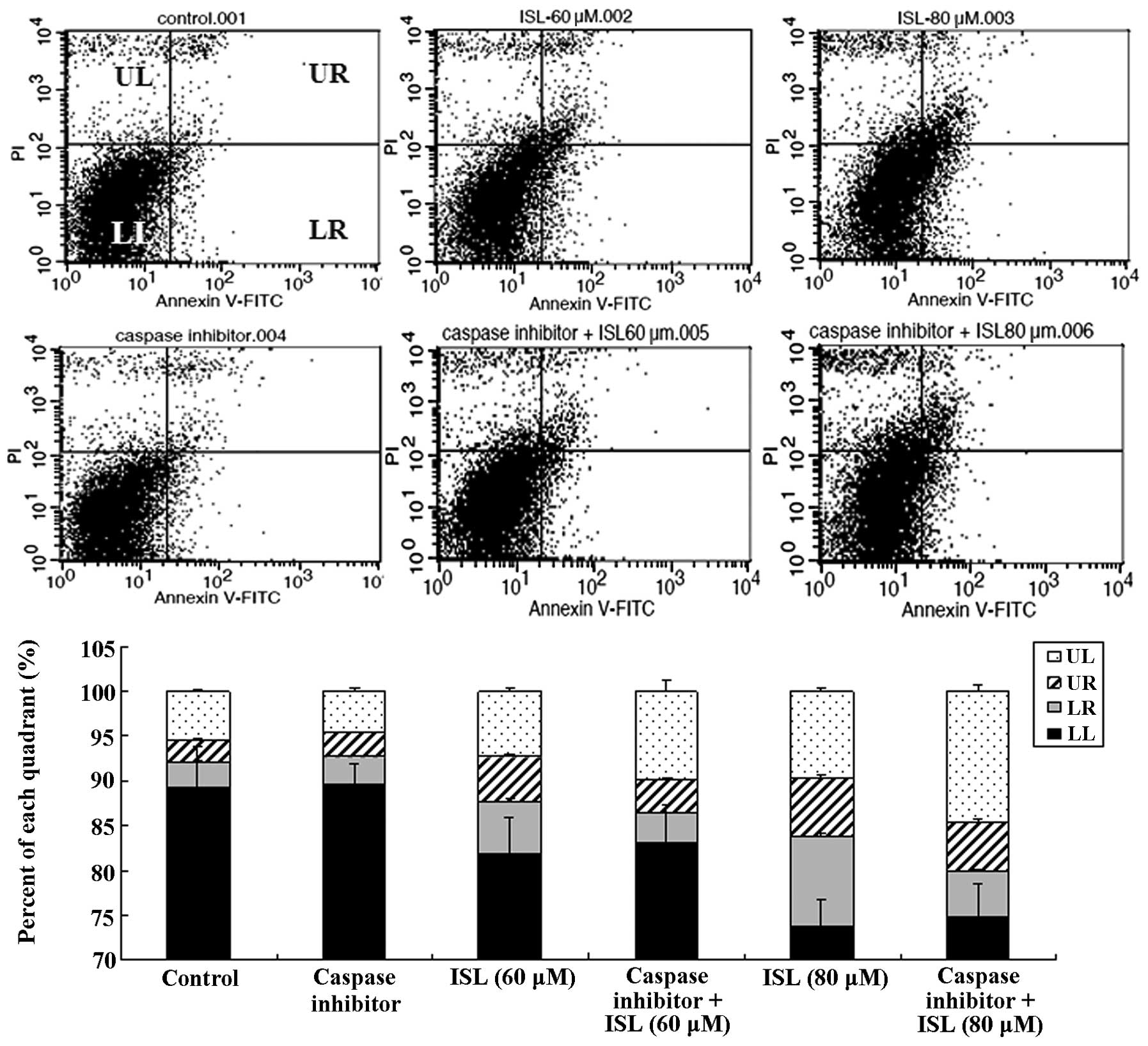
Apoptosis is induced by ISL via
apoptosis-dependent and non-apoptosis-dependent pathways
To investigate the detailed mechanism of the
ISL-induced apoptosis, the pan-caspase inhibitor Z-VAD-FMK was
added and FACS was used to detect the change of cell apoptosis.
Notably, the results of FACS (Fig.
4) revealed that the pan-caspase inhibitor Z-VAD-FMK decreased
the ISL-induced apoptotic cell death (LR and UR), however, the
percentages of the U87 glioma cells undergoing necrotic cell death
(UL) were not affected by the addition of the pan-caspase
inhibitor. This indicates that ISL was able to induce the death of
U87 cells via a non-apoptotic pathway. As shown in Fig. 4, the treatment with Z-VAD-FMK did
not alter the percentage of living cells. The results of western
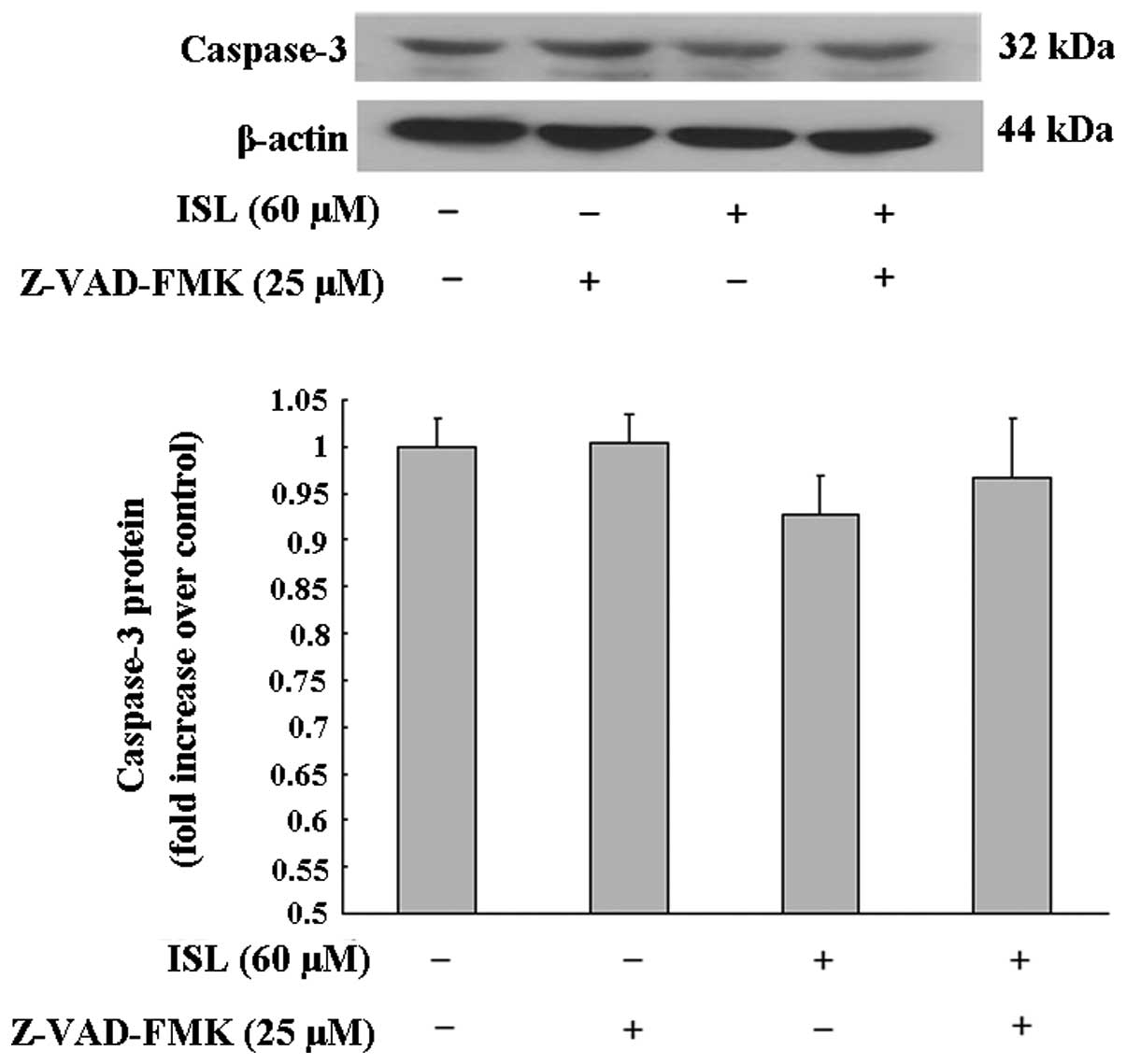
blot using caspase-3 antibody (Fig.
5) verified that treatment with ISL induced the activation of
caspase-3, and that the pan-caspase inhibitor Z-VAD-FMK at 25 M was
able to block the activation of caspase-3 and thereby reduce the
percentage of early apoptotic cells. However, the percentage of
total living cells was not altered by the treatment with Z-VAD-FMK,
and the percentage of nonapoptotic necrotic cells was increased
along with the ISL concentration.
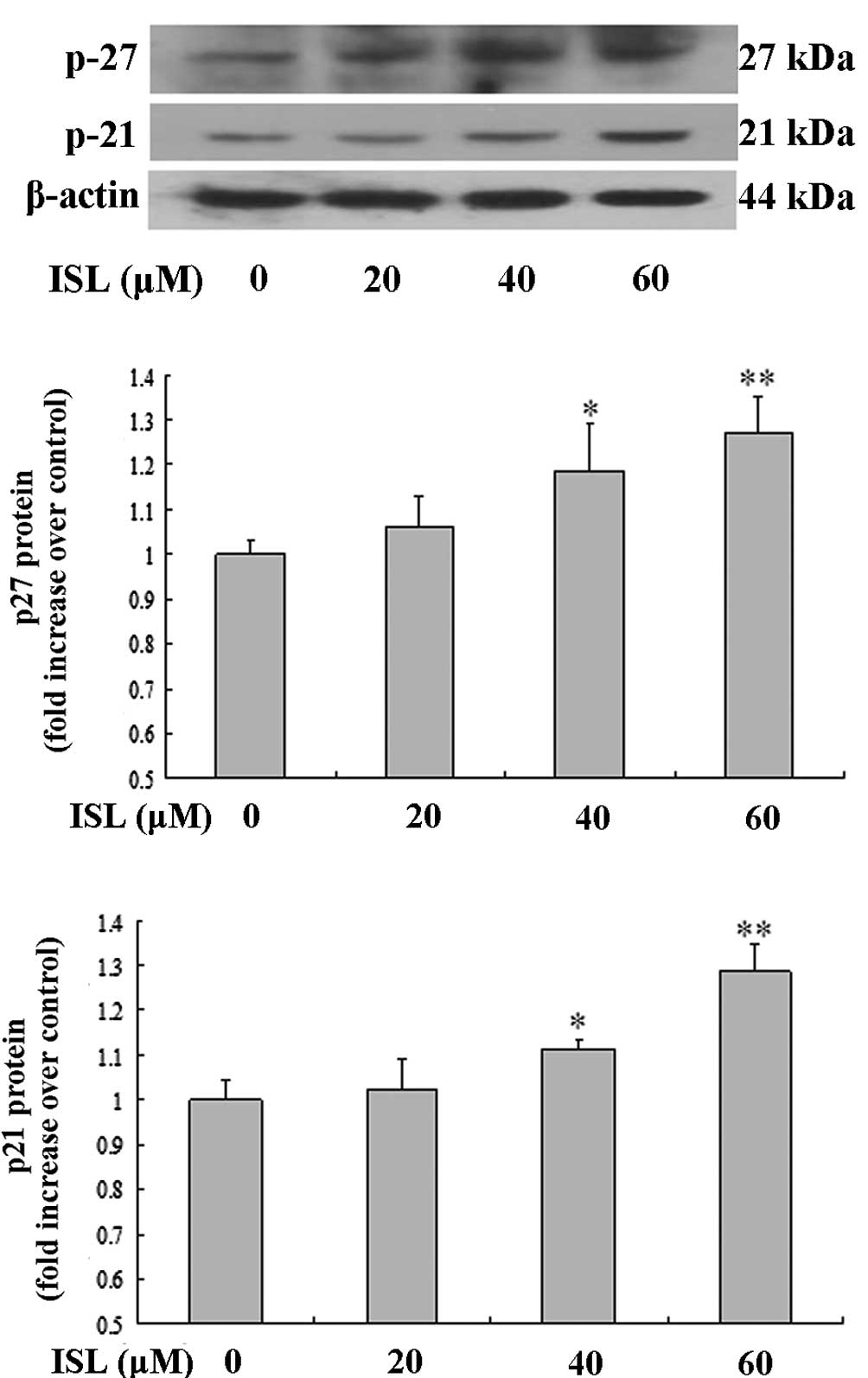
Effects of ISL on p27 and p21 expression
levels determined by western blot analysis
To further investigate the mechanism of the
ISL-induced cell cycle arrest and apoptosis, the protein expression
levels of p21 and p27 were determined by western blot analysis. As
shown in Fig. 6, the expression
levels of the p21 and p27 proteins were upregulated as the ISL
concentration increased.
Discussion
ISL has been considered as a potential therapeutic
agent for the treatment of cancer, and has demonstrated antitumor
efficacy in various types of cancer, including colon (9,12),
breast (13,14), gastric (15) and prostate cancer (16) and melanoma (17). However, the effects of ISL in
glioma cancer cells remain unknown. In this study, we examined the
effect and mechanism of ISL on human glioma cell growth. Our
results revealed that ISL inhibited the growth of U87 glioma cells
in a time-dependent and dose-dependent manner. Treatment of the U87
cells with ISL resulted in G2/M-phase and S-phase cell cycle arrest
and induced apoptosis in a dose-dependent manner. The blockade of
cell cycle progression by ISL was achieved through upregulation of
the p21/WAF1 and p27 pathways. In addition, the cell death of U87
cells induced by ISL progressed via apoptotic and necrotic
pathways.
The results of MTT assays revealed that ISL
inhibited the proliferation of U87 cells in a time- and
dose-dependent manner. However, the MTT assay system determined
only the total percentage of cell death and did not discriminate
between cell death occurring via apoptotic or necrotic pathways.
With Annexin V/PI doubled staining using FACS for phosphatidyl
serine detection, it was identified that ISL induced the death of
the U87 glioma cells in apoptotic and non-apoptotic manners. We
identified that the necrotic phenotype occurred in more cells than
the apoptotic phenotype, therefore, necrosis may be the prevalent
response to ISL treatment in U87 glioma cells. To further
investigate whether the underlying mechanism of cell death induced
by ISL was caspase-related or not, we blocked caspases with the
pan-caspase inhibitor Z-VAD-FMK and analyzed the cell death. The
results of western blot analysis revealed that caspase-3 activation
was effectively blocked, however, the percentage of living cells
did not increase, which suggests that ISL-induced cell death
occured in a caspase-independent manner. The results of FACS also
revealed that the percentage of necrotic cells increased with the
increase of ISL concentration and was not altered by treatment with
the pan-caspase inhibitor Z-VAD-FMK, indicating that the
non-apoptotic cell death induced by ISL was predominant compared
with the cell death occurring via an apoptotic pathway.
It was also identified that p21/WAF1 and p27 were
upregulated by the treatment with ISL, in accordance with the
results reported by others (18–20),
indicating that the G2/M arrest induced by ISL is a general effect
occurring in multiple cell lines. p21/WAF1 and p27 are universal
inhibitors of cyclin-dependent kinases and play important roles in
G2/M arrest. The upregulation of p21 and p27 may contribute to the
antiproliferative activity of ISL in U87 glioma cells.
In summary, the results of our study suggest that
ISL induced apoptotic and necrotic cell death of U87 glioma cells,
and that ISL-induced cell death occurred in the presence of the
caspase inhibitor Z-VAD-FMK. The ISL induced signaling involves the
upregulation of p21/WAF1 and p27. As ISL has demonstrated evident
bioactivity in cancer therapy and prevention, distinguishing the
onset of ISL-induced apoptosis or necrosis using an in vitro
cell culture system is likely to be important for the utilization
of this compound as a potential therapeutic agent in humans.
References
|
1
|
Lima FR, Kahn SA, Soletti RC, Biasoli D,
Alves T, da Fonseca AC, et al: Glioblastoma: Therapeutic
challenges, what lies ahead. Biochim Biophys Acta. 1826:338–349.
2012.PubMed/NCBI
|
|
2
|
Ma X, Lv Y, Liu J, Wang D, Huang Q, Wang
X, Li G, Xu S and Li X: Survival analysis of 205 patients with
glioblastoma multiforme: clinical characteristics, treatment and
prognosis in China. J Clin Neurosci. 16:1595–1598. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Davis ME and Stoiber AM: Glioblastoma
multiforme: enhancing survival and quality of life. Clin J Oncol
Nurs. 15:291–297. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lin H, Liu J and Zhang Y: Developments in
cancer prevention and treatment using traditional Chinese medicine.
Front Med. 5:127–133. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Efferth T, Li PC, Konkimalla VS and Kaina
B: From traditional Chinese medicine to rational cancer therapy.
Trends Mol Med. 13:353–361. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang S, Wu X, Tan M, Gong J, Tan W, Bian
B, Chen M and Wang Y: Fighting fire with fire: poisonous Chinese
herbal medicine for cancer therapy. J Ethnopharmacol. 140:33–45.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kandaswami C, Lee LT, Lee PP, Hwang JJ, Ke
FC, Huang YT and Lee MT: The antitumor activities of flavonoids. In
Vivo. 19:895–909. 2005.PubMed/NCBI
|
|
8
|
Weng CJ and Yen GC: Flavonoids, a
ubiquitous dietary phenolic subclass, exert extensive in vitro
anti-invasive and in vivo anti-metastatic activities. Cancer
Metastasis Rev. 31:323–351. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lee CK, Son SH, Park KK, Park JH, Lim SS
and Chung WY: Isoliquiritigenin inhibits tumor growth and protects
the kidney and liver against chemotherapy-induced toxicity in a
mouse xenograft model of colon carcinoma. J Pharmacol Sci.
106:444–451. 2008. View Article : Google Scholar
|
|
10
|
Orlikova B, Schnekenburger M, Zloh M,
Golais F, Diederich M and Tasdemir D: Natural chalcones as dual
inhibitors of HDACs and NF-κB. Oncol Rep. 28:797–805.
2012.PubMed/NCBI
|
|
11
|
Lee YM, Jeong GS, Lim HD, An RB, Kim YC
and Kim EC: Isoliquiritigenin 2-methyl ether induces growth
inhibition and apoptosis in oral cancer cells via heme oxygenase-1.
Toxicol In Vitro. 24:776–782. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Auyeung KK and Ko JK: Novel herbal
flavonoids promote apoptosis but differentially induce cell cycle
arrest in human colon cancer cell. Invest New Drugs. 28:1–13. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lau GT, Ye L and Leung LK: The licorice
flavonoid isoliquiritigenin suppresses phorbol ester-induced
cyclooxygenase-2 expression in the non-tumorigenic MCF-10A breast
cell line. Planta Med. 76:780–785. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ye L, Gho WM, Chan FL, Chen S and Leung
LK: Dietary administration of the licorice flavonoid
isoliquiritigenin deters the growth of MCF-7 cells overexpressing
aromatase. Int J Cancer. 124:1028–1036. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ma J, Fu NY, Pang DB, Wu WY and Xu AL:
Apoptosis induced by isoliquiritigenin in human gastric cancer
MGC-803 cells. Planta Med. 67:754–777. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang X, Yeung ED, Wang J, Panzhinskiy EE,
Tong C, Li W and Li J: Isoliquiritigenin, a natural anti-oxidant,
selectively inhibits the proliferation of prostate cancer cells.
Clin Exp Pharmacol Physiol. 37:841–847. 2010.PubMed/NCBI
|
|
17
|
Iwashita K, Kobori M, Yamaki K and
Tsushida T: Flavonoids inhibit cell growth and induce apoptosis in
B16 melanoma 4A5 cells. Biosci Biotechnol Biochem. 64:1813–1820.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hsu YL, Kuo PL, Chiang LC and Lin CC:
Isoliquiritigenin inhibits the proliferation and induces the
apoptosis of human non-small cell lung cancer A549 cells. Clin Exp
Pharmacol Physiol. 31:414–418. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li T, Satomi Y, Kotoh D, Shimada J, Baba
M, Okuyama T, Nishino H and Kitamura N: Induction of cell cycle
arrest and p21(CIP1/WAF1) expression in human lung cancer cells by
isoliquiritigenin. Cancer Lett. 207:27–35. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lee YM, Lim do Y, Choi HJ, Jung JL, Chung
WY and Park JH: Induction of cell cycle arrest in prostate cancer
cells by the dietary compound isoliquiritigenin. J Med Food.
12:8–14. 2009. View Article : Google Scholar : PubMed/NCBI
|