Introduction
Hepatitis C virus (HCV) infection, one of the main
causes of chronic hepatitis, is estimated to affect 170 million
people worldwide (1). The natural
history of HCV infection is characterized by acute and eventually
chronic infection, and may progress from a long-lasting
asymptomatic condition to decompensated liver cirrhosis or
hepatocellular carcinoma (HCC) (2). However, the long-term impact of HCV
infection is highly variable; some patients with persistent HCV
infection exhibit persistently normal alanine aminotransferase
(ALT) levels (PNALT), which are associated with milder disease
activity and slower progression to cirrhosis (3). In addition, the differences between
PNALT patients and those with chronic hepatitis C (CHC) who exhibit
elevated ALT levels have not yet been fully elucidated (4).
Persico et al reported that the grade of
disease activity does not increase over a period of years, and that
progression to cirrhosis is slow or absent in patients with
HCV-related chronic hepatitis associated with PNALT (5). We have previously reported that the
ALT level is a predictor of HCV-associated HCC incidence in a
community-based population in Japan (6). In addition, a number of studies have
shown that interferon (IFN)-based therapy reduces HCC in patients
with CHC, even in those in whom HCV RNA remains detectable
(7,8). Continuous normalization of
aminotransferase and α-feto protein (AFP) for more than 1 year
during IFN therapy is associated with a reduced risk of HCC
development following the termination of the IFN therapy (9).
A recent application of proteomic technology has
identified a spectral pattern from the serum of patients with liver
disease (10–12), and proteomic techniques will be
able to identify serum biomarkers that are present in the serum of
patients with PNALT. Furthermore, a biomarker or biomarker panel
may also help to elucidate a possible mechanism for chronic
hepatitis from PNALT and could perhaps lead to the development of
more effective treatments for chronic hepatitis. However, proteomic
approaches focused on PNALT have not been previously explored.
In this study, we verified differentially expressed
protein in serum samples and showed that the level of the
complement component 4a (C4a) in serum was higher in HCV carriers
with PNALT compared to CHC patients or healthy volunteers. The
present study reveals that C4a increases with HCV infection, but
decreases with disease progression. Identification of these and
other proteins will help clarify the underlying mechanisms and
contribute to improved clinical outcomes for HCV carriers.
Patients and methods
Study population
Anti-HCV seropositive subjects with detectable HCV
core antigen (HCVcAg) or HCV RNA were considered to be persistently
infected with HCV and were classified as HCV carriers. ALT levels
>30 IU/l and platelet counts <15×104/μl were
considered to be abnormal. HCV carriers exhibiting persistently
normal ALT levels accompanied by normal platelet counts during the
observation period were defined as the PNALT group in this study
(13). Subjects who underwent oral
or intravenous administration of medical herbs or other palliative
therapies were not excluded from this study, but those who had
received IFN therapy were excluded. All subjects were negative for
hepatitis B virus surface antigen (HBsAg).
The first group of subjects, who were undergoing
hospital-based clinical evaluation, consisted of 39 HCV carriers.
Of these, 19 with PNALT (PNALT-1 group) and 20 with CHC and
abnormal ALT levels (CHC-1 group) were enrolled. HCV carriers with
PNALT (PNALT-1 group) were defined as those who had normal serum
ALT levels (≤30 U/l) over a 12-month period and on at least 3
different occasions, and platelet counts of ≥15×104/μl.
Blood samples from the PNALT-1 and CHC-1 groups were obtained
during the last observation period.
The second group of subjects was part of a larger
cohort being followed-up as part of a study on the natural history
of liver disease; data on these individuals were acquired from 1994
through 2005 (14). An analysis
was conducted of HCV carriers who had undergone at least 3
independent ALT measurements obtained during annual general health
examinations or liver disease screenings. In total, 37 HCV carriers
with persistently normal ALT levels and platelet counts
≥15×104 μl (PNALT-2 group) and 30 HCV carriers with
persistently abnormal ALT levels and platelet counts
<15×104/μl (CHC-2) were investigated. Blood samples
from PNALT-2 or CHC-2 subjects were obtained during the last
observation period in this study from 2002 to 2005. Serum samples
were also obtained from healthy volunteers without HCV infection
(n=12).
After the blood samples were collected, serum was
stored at −80°C. Written informed consent was obtained from each
subject and the study protocol was approved by the Ethics Committee
of Kagoshima University Hospital; the Faculty of Medicine,
University of Miyazaki and Kyoto Prefectural Medical School.
Serum pre-treatment with ClinProt
magnetic beads
Serum samples (5 μl) were purified and concentrated
using magnetic bead-based weak cation exchange chromatography
resins (WCX) (Bluker Daltonics, Bremen, Germany).
2-Cyano-4-hydroxycinnamic acid (CHCA) matrix solution (Bluker
Daltonics) was diluted to 0.3 g/l in an ethanol:acetone (2:1)
solution. Purified serum samples and diluted CHCA solutions were
mixed (1:9), and 1 μl of the solution was applied onto a
matrix-assisted laser desorption/ionization time-of-flight
(MALDI-TOF) AnchorChip.
Mass spectrometry (MS) and peptide
identification
The AnchorChip target plate was placed in an
AutoFlex II TOF/TOF mass spectrometer (Bluker Daltonics). Spectra
were acquired in the positive linear mode in a molecular mass range
from 1,000 to 3,000 Da. The MALDI-TOF MS spectrum was subjected to
a Mascot database search (Matrix Science, Boston, MA, USA) using
the SwissProt database.
Serum markers
The presence of serum anti-HCV antibody (Ab) was
determined using a commercially available third-generation
enzyme-linked immunosorbent assay (ELISA). Serum levels of HCVcAg
were determined by a chemiluminescence enzyme immunoassay (HCV core
protein; SRL, Tokyo, Japan), with a detection threshold of 20
fmol/l. The serologically defined HCV genotype (HCV serotype) was
tested with a serological genotyping assay kit (Immunocheck F-HCV
Grouping, International Reagents Co., Tokyo, Japan). In some
patients, the HCV genotype was examined (HCV Core Genotype, SRL,
Tokyo, Japan). HCV genotype 1b was included with serotype I, and
genotypes 2a and 2b with serotype II. No other HCV genotype was
detected in this study population.
The serum concentration of C4a was determined using
a C4a ELISA kit (Human C4a ELISA kit, BD Biosciences, San Diego,
CA, USA).
Statistical analysis
The results are presented as the means ± standard
deviation (SD). All spectra in MALDI-TOF MS were analyzed using
Bluker Daltonics FlexAnalysis 2.2 software and ClinProTools 2.0
software. Statistical analysis of other clinical data was performed
using StatView 4.5 software (Abacus Concepts, Berkeley, CA, USA) or
SPSS software (SPSS Inc., Chicago, IL, USA). Differences were
evaluated by the Mann-Whitney U test, the Fisher’s exact test or
the Chi-square test as appropriate. Any probability value <0.05
was considered to indicate a statistically significant
difference.
Results
Profiling sera from patients with PNALT
and chronic hepatitis C using MALDI-TOF MS analysis
In the first hospital-based group, serum levels of
ALT, aspartate aminotransferase (AST), and γ-glutamyltranspeptidase
(γ-GTP) were lower and platelet counts and total cholesterol were
higher in PNALT-1 patients than in CHC-1 patients (Table I). In this group, the sera of
patients were analyzed to identify protein peaks that differed most
between patient subsets. Serum proteomics revealed that 6 serum
protein peaks with mass-to-charge ratios ranging from 1,000 to
3,000 differed significantly between PNALT-1 and CHC-1 groups
(Table II). In these protein
peaks, a 1738-m/z peak protein was identified as a fragment of C4,
with the sequence NGFKSHALQLNNRQI.
 | Table IPatient characteristics in the
hospital-based group. |
Table I
Patient characteristics in the
hospital-based group.
| Characteristics | PNALT-1 (n=19) | CHC-1 (n=20) | P-valuea |
|---|
| Age | 55.2±15.1 | 52.3±11.7 | 0.17 |
| Gender
(male/female) | 3/16 | 7/13 | 0.27 |
| HCV core antigen
(fmol/l) | 1163±803 | 1072±669 | 0.72 |
| HCV serotype
(I/II/UD) | 9/5/5 | 11/5/4 | 0.87 |
| Platelet count
(x104/μl) | 21.3±5.5 | 14.8±4.0 | <0.001 |
| AST (IU/l) | 25.4±3.9 | 73.6±37.5 | <0.001 |
| ALT (IU/l) | 23.5±5.1 | 90.9±54.8 | <0.001 |
| γ-GTP (IU/l) | 18.6±8.5 | 89.4±77.9 | <0.001 |
| Total cholesterol
(mg/dl) | 200.0±24.4 | 180.1±27.4 | 0.03 |
| Albumin (g/dl) | 4.5±0.2 | 4.5±0.4 | 0.76 |
 | Table IIProtein peaks expressed differentially
between HCV carriers with persistent normal ALT levels (PNALT) and
chronic hepatitis C (CHC) patients with abnormal ALT levels. |
Table II
Protein peaks expressed differentially
between HCV carriers with persistent normal ALT levels (PNALT) and
chronic hepatitis C (CHC) patients with abnormal ALT levels.
| Peak intensity | |
|---|
|
| |
|---|
| m/z | PNALT-1 (n=19) | CHC-1 (n=20) | P-valuea |
|---|
| 1738 | 109.4±67.1 | 83.9±54.0 | <0.01 |
| 1896 | 105.0±64.8 | 111.3±63.3 | <0.05 |
| 1943 | 191.3±149.5 | 139.6±73.6 | <0.01 |
| 2858 | 104.0±34.3 | 85.3±25.6 | <0.001 |
| 2928 | 31.8±9.7 | 64.0±28.9 | <0.001 |
| 2947 | 59.3±34.9 | 80.7±36.2 | <0.001 |
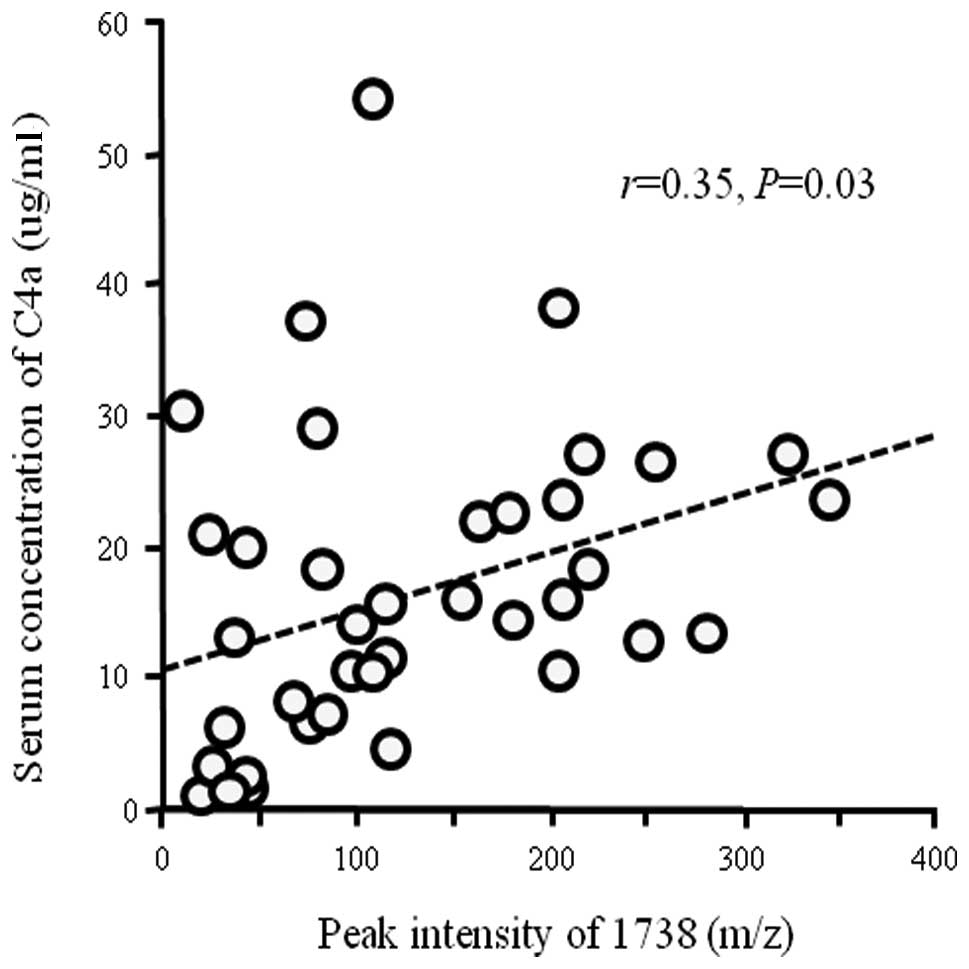
Correlation between the protein peak of
1738 m/z and serum levels of C4a determined by ELISA
Although the identified C4 fragment is part of C4c,
serum concentrations of C4c could not be determined by commercially
available methods, such as ELISA. By contrast, the recalibrated
peak intensity of this fragment significantly correlated with the
serum level of C4a, which could be determined with a commercially
available assay kit (Fig. 1). In
addition, serum concentrations of C4a were significantly higher in
PNALT-1 subjects [means ± SD (μg/ml), 20.6±11.9] compared with
those in CHC-1 subjects (12.2±10.2) (P=0.01).
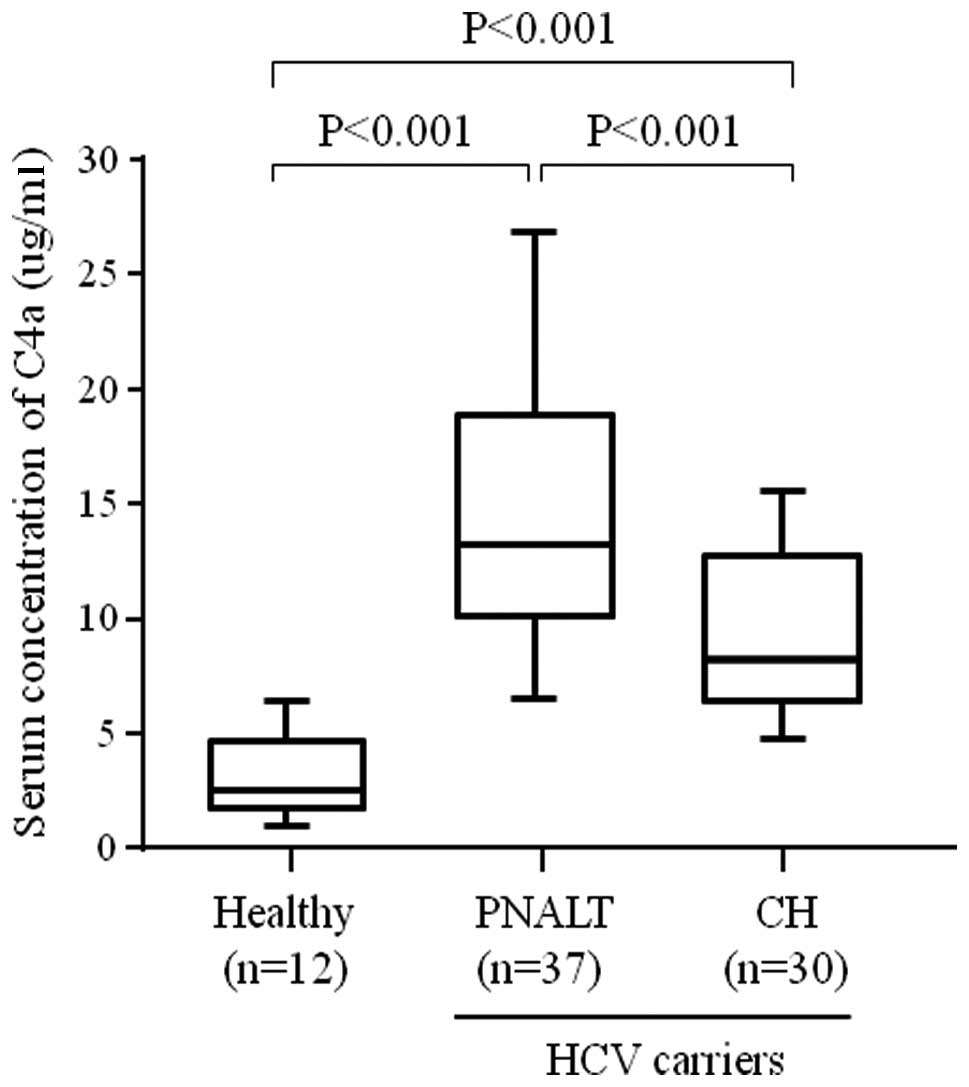
Serum levels of C4a determined by ELISA
in the second group
In the cohort-based population, age, the prevalence
of females and of serotype II, platelet counts, and serum albumin
and total cholesterol levels were significantly higher in the
PNALT-2 group than in the CHC-2 group. By contrast, serum AST, ALT
and γ-GTP levels were lower in the PNALT-2 group (Table III). In the cohort-based
population, serum concentrations of C4a, as determined by ELISA,
were significantly higher in the PNALT-2 group than in the CHC-2
group and healthy controls (Fig.
2).
 | Table IIIPatient characteristics in
cohort-based population with HCV infection. |
Table III
Patient characteristics in
cohort-based population with HCV infection.
| Characteristics | PNALT-2 (n=37) | CHC-2 (n=30) | P-valuea |
|---|
| Age | 75.6±6.5 | 70.4±6.6 | <0.01 |
| Gender
(male/female) | 8/29 | 15/15 | 0.02 |
| HCV core antigen
(fmol/l) | 6,042±4,295 | 4,553±3,546 | 0.27 |
| HCV serotype
(I/II) | 14/23 | 24/6 | <0.001 |
| Platelet count
(x104/μl) | 22.3±5.3 | 11.8±3.8 | <0.001 |
| AST (IU/l) | 28.3±8.0 | 100.1±81.8 | <0.001 |
| ALT (IU/l) | 19.5±6.0 | 96.9±81.8 | <0.001 |
| γ-GTP (IU/l) | 15.1±10.6 (n=31) | 56.2±46.7 (n=20) | <0.001 |
| T-Cho (mg/dl) | 181.8±29.3
(n=31) | 158.8±27.1
(n=29) | 0.02 |
| Albumin (g/dl) | 4.4±0.3 (n=33) | 4.1±0.5 (n=29) | <0.01 |
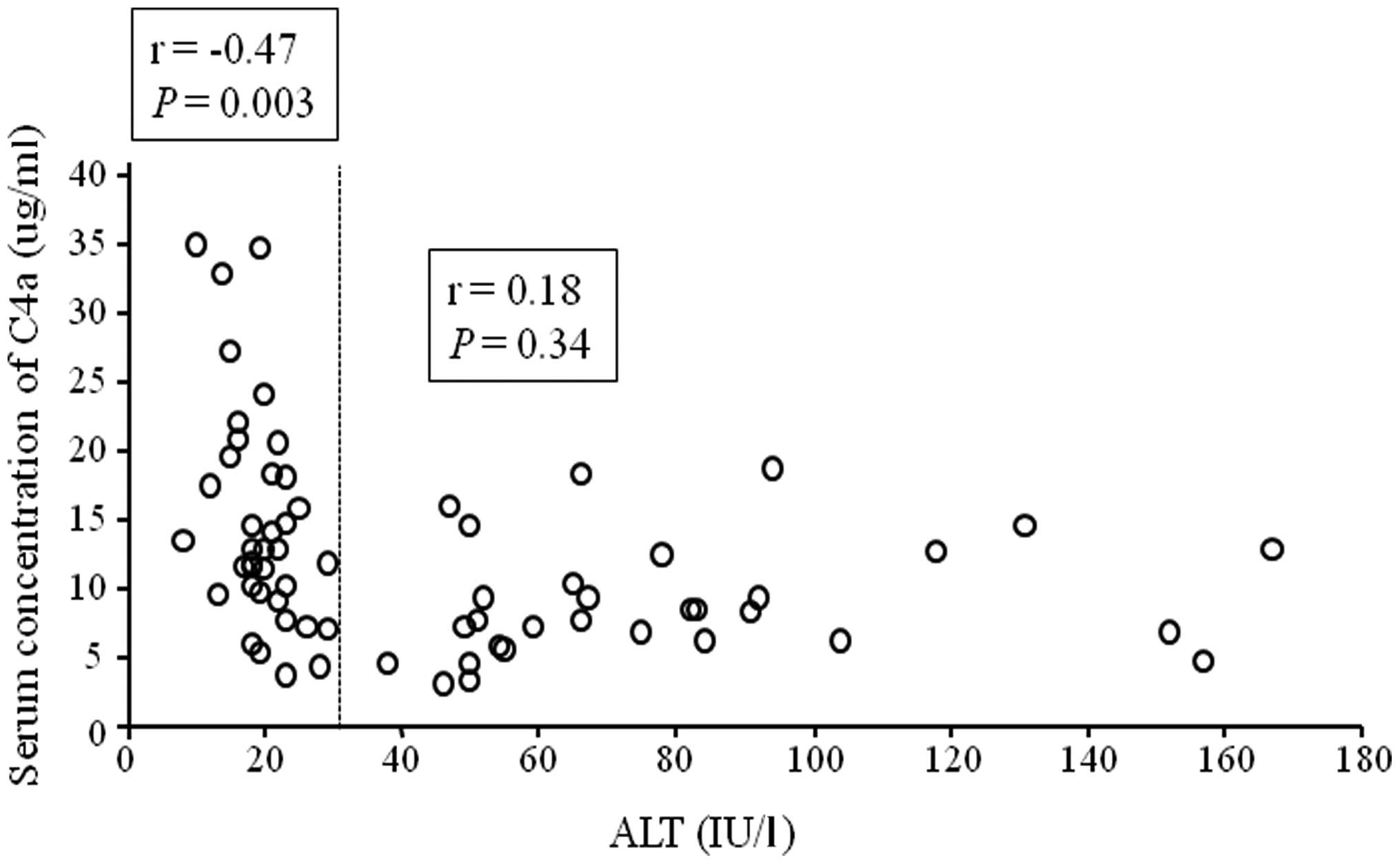
Serum C4a levels in the PNALT-2 group correlated
significantly with serum AST and ALT levels, but not with HCVcAg
levels or other blood laboratory parameters (Table IV). In addition, a significant
negative correlation between serum C4a and ALT levels was observed
in the population as a whole (Fig.
3, r=−0.35, P=0.03) and in PNALT patients (Table IV; ALT ≤30 IU/l; Fig. 3), but not in CHC-2 patients (ALT
>30 IU/l; Fig. 3).
 | Table IVCorrelation between serum C4a levels
and blood laboratory parameters in PNALT subjects. |
Table IV
Correlation between serum C4a levels
and blood laboratory parameters in PNALT subjects.
| Parameter | Correlation
coefficient | P-valuea |
|---|
| HCV core antigen | 0.06 | 0.73 |
| White blood
cell | −0.06 | 0.72 |
| Hematocrit | −0.06 | 0.12 |
| Platelet | 0.12 | 0.51 |
| Albumin | 0.03 | 0.88 |
| γ-globulin | −0.05 | 0.77 |
| AST | −0.39 | 0.02 |
| ALT | −0.47 | <0.01 |
|
Total-bilirubin | −0.07 | 0.69 |
| Total
cholesterol | 0.05 | 0.76 |
| Ferritin | −0.18 | 0.30 |
| Hyaluronic
acid | −0.23 | 0.17 |
| Type IV
collagen | −0.04 | 0.82 |
| α-fetoprotein | −0.29 | 0.09 |
| DCP | −0.07 | 0.69 |
Discussion
HCV is not thought to be directly cytopathic to
hepatocytes, and a T helper (Th)1-type or cytotoxic T lymphocyte
(CTL) response is critically involved in HCV-mediated liver injury
(15). Therefore, it is
conceivable that various suppressor mechanisms exist against
Th1-type immune responses in HCV carriers with PNALT, which may be
distinct from those in CHC patients with active liver inflammation
(16). However, few studies have
focused on PNALT using a serum proteomic approach. In this study of
HCV carriers, a number of proteins were detected which were
differentially expressed between PNALT and CHC patients. Of these,
the C4 fragment was identified by peptide mass fingerprint (PMF)
methods following a Mascot search, and serum levels of C4a, which
correlated with the protein peak of the identified C4 fragment,
were higher in PNALT than in CHC patients, as determined by ELISA.
In addition, serum C4a levels correlated with ALT levels in the
PNALT but not CHC patients.
Following acute HCV infection, approximately 70% of
individuals remain positive for both anti-HCV Ab and HCV RNA, and
are defined as HCV carriers. By contrast, approximately 30% of
acutely infected individuals clear the HCV and remain positive for
anti-HCV Ab but negative for HCV RNA. We confirmed that serum C4a
levels in those who cleared the virus were similar to healthy
controls (data not shown). Serum C4a levels were higher in CHC
patients and individuals with PNALT compared to healthy controls.
Therefore, serum C4a levels appear to be at least elevated by
existing HCV infection, although serum C4a levels did not correlate
with serum HCVcAg levels in individuals with PNALT (Table IV). It was previously reported
that serum L-ficolin levels were increased in HCV patients, and
that this protein only recognized and bound to glycoproteins E1 and
E2 of the HCV envelope, but also activated the complement lectin
pathway-mediated cytolytic activity in HCV-infected hepatocytes
(17). In the lectin pathway,
mannose-binding lectin (MBL)-associated serine protease-2 (MASP-2)
cleaves C4, releasing C4a and generating C4b (18). Thus, C4a levels should increase in
HCV carriers compared to healthy controls by post-translational
mechanisms.
Previous studies have reported decreased serum C4
levels in patients with CHC (19,20).
Recently, the HCV core protein and non-structural 5A protein (NS5A)
were reported to transcriptionally downregulate C4 expression by
modulating the expression of upstream stimulating factor 1 and IFN
regulatory factor 1, respectively (21). Thus, serum C4 protein levels are
decreased in HCV patients compared to healthy controls as a result
of altered transcriptional regulation (22). Although the mechanism of C4a
variation in HCV carriers has not been elucidated, our study
suggests that serum C4a levels in HCV carriers with PNALT should be
dominantly affected by post-translational mechanisms, but patients
with CHC may be affected by both translational (downregulation) and
post-translational (upregulation) mechanisms.
In CHC, decreased specific C4 activity without C3
consumption suggests complement activation leading to the
N-terminal cleavage of C4 with the production of C4a (20). Another study demonstrated increased
C4a levels in CHC patients without a significant increase in the
levels of C3a (23). Avirutnan
et al reported that flaviviruses, such as dengue virus, use
their non-structural protein, NS1, to attenuate complement
activation by directly interacting with C4, leading to viral
persistence (24). Although the
mechanisms responsible for HCV persistence or PNALT in HCV carriers
are not well understood, the interactions between HCV and the host
immune system are thought to play a pivotal role in patients with
HCV infection.
The majority of individuals with PNALT have minimal
or mild inflammation and absent or minimal fibrosis, and follow-up
studies have shown disease stability with minimal fibrosis
progression over the years, leading to a favorable prognosis.
However, cirrhosis and HCC are occasionally observed in HCV
carriers with normal ALT levels (25). In addition, some patients with
PNALT may develop ALT elevation over time (4), and these individuals may be at
increased risk of the significant progression of fibrosis.
Long-term observation or liver biopsy has not always been
performed; serum C4a levels may be a diagnostic marker for advanced
fibrosis or a predictor for ALT elevation in PNALT. These issues
should be subject to further analysis.
It has been reported that in HCV carriers with PNALT
or normal ALT levels, IFN-based therapy is safe and efficacious
(26,27). However, the decision whether or not
to treat HCV carriers should be made with the specific clinical
setting in mind (28). In
addition, it is recommended that serum ALT levels be kept <30
IU/l to prevent the occurrence of HCC (29). If serum C4a levels are an indicator
of disease prognosis, HCV carriers with low serum C4a levels may
have to be treated despite ALT elevation. More significantly,
elucidating the mechanism underlying the association between serum
C4a and ALT levels should lead to new approaches to the treatment
of HCV carriers with PNALT.
In conclusion, host factors such as C4a differ
between HCV carriers with PNALT and CHC patients with elevated ALT
levels. Proteomic approaches could greatly contribute to elucidate
the host factor in PNALT patients as more differences are
discovered. Identification of these and other proteins will help
clarify the mechanism and may improve clinical outcomes of HCV
carriers.
Acknowledgements
We thank Keiko Nakase, Ayaka Hamabe and Yuko
Morinaga for their technical assistance. This study was supported
by a grant (No. CA87982) from the United States National Institutes
of Health, a Grant-in-Aid for Research on Hepatitis and BSE from
the Ministry of Health, Labour and Welfare of Japan, Scientific
Research from the Ministry of Education, Culture, Sports, Science
and Technology of Japan, and a grant from the Miyazaki Prefecture
Collaboration of Regional Entities for the Advancement of
Technological Excellence (Japan Science and Technology Corp.).
References
|
1
|
No authors listed. Global surveillance and
control of hepatitis C. Report of a WHO consultation organized in
collaboration with the Viral Hepatitis Prevention Board, Antwerp,
Belgium. J Viral Hepat. 6:35–47. 1999.
|
|
2
|
Lavanchy D: The global burden of hepatitis
C. Liver Int. 29:74–81. 2009.
|
|
3
|
Persico M, Perrotta S, Persico E,
Terracciano L, Folgori A, Ruggeri L, Nicosia A, Vecchione R, Mura
VL, Masarone M and Torella R: Hepatitis C virus carriers with
persistently normal ALT levels: biological peculiarities and update
of the natural history of liver disease at 10 years. J Viral Hepat.
13:290–296. 2006.
|
|
4
|
Uto H, Kurogi J, Takahama Y, Kusumoto K,
Hayashi K, Ido A, Kohara M, Stuver SO, Moriuchi A, Hasegawa S,
Oketani M and Tsubouchi H: Alanine aminotransferase flare-up in
hepatitis C virus carriers with persistently normal alanine
aminotransferase levels in a hyperendemic area of Japan. J
Gastroenterol. 42:673–680. 2007.
|
|
5
|
Persico M, Persico E, Suozzo R, Conte S,
De Seta M, Coppola L, Palmentieri B, Sasso FC and Torella R:
Natural history of hepatitis C virus carriers with persistently
normal aminotransferase levels. Gastroenterology. 118:760–764.
2000.
|
|
6
|
Suruki R, Hayashi K, Kusumoto K, Uto H,
Ido A, Tsubouchi H and Stuver SO: Alanine aminotransferase level as
a predictor of hepatitis C virus-associated hepatocellular
carcinoma incidence in a community-based population in Japan. Int J
Cancer. 119:192–195. 2006.
|
|
7
|
Nishiguchi S, Kuroki T, Nakatani S,
Morimoto H, Takeda T, Nakajima S, Shiomi S, Seki S, Kobayashi K and
Otani S: Randomised trial of effects of interferon-alpha on
incidence of hepatocellular carcinoma in chronic active hepatitis C
with cirrhosis. Lancet. 346:1051–1055. 1995.
|
|
8
|
Ikeda K, Arase Y, Saitoh S, Kobayashi M,
Suzuki Y, Suzuki F, Tsubota A, Chayama K, Murashima N and Kumada H:
Interferon beta prevents recurrence of hepatocellular carcinoma
after complete resection or ablation of the primary tumor-A
prospective randomized study of hepatitis C virus-related liver
cancer. Hepatology. 32:228–232. 2000.
|
|
9
|
Arase Y, Ikeda K, Suzuki F, Suzuki Y,
Kobayashi M, Akuta N, Hosaka T, Sezaki H, Yatsuji H, Kawamura Y,
Kobayashi M and Kumada H: Interferon-induced prolonged biochemical
response reduces hepatocarcinogenesis in hepatitis C virus
infection. J Med Virol. 79:1485–1490. 2007.
|
|
10
|
Kanmura S, Uto H, Kusumoto K, Ishida Y,
Hasuike S, Nagata K, Hayashi K, Ido A, Stuver SO and Tsubouchi H:
Early diagnostic potential for hepatocellular carcinoma using the
SELDI ProteinChip system. Hepatology. 45:948–956. 2007.
|
|
11
|
Kanmura S, Uto H, Sato Y, Kumagai K,
Sasaki F, Moriuchi A, Oketani M, Ido A, Nagata K, Hayashi K, Stuver
SO and Tsubouchi H: The complement component C3a fragment is a
potential biomarker for hepatitis C virus-related hepatocellular
carcinoma. J Gastroenterol. 45:459–467. 2010.
|
|
12
|
Uto H, Kanmura S, Takami Y and Tsubouchi
H: Clinical proteomics for liver disease: a promising approach for
discovery of novel biomarkers. Proteome Sci. 8:702010.
|
|
13
|
Okanoue T, Makiyama A, Nakayama M, Sumida
Y, Mitsuyoshi H, Nakajima T, Yasui K, Minami M and Itoh Y: A
follow-up study to determine the value of liver biopsy and need for
antiviral therapy for hepatitis C virus carriers with persistently
normal serum aminotransferase. J Hepatol. 43:599–605. 2005.
|
|
14
|
Uto H, Stuver SO, Hayashi K, Kumagai K,
Sasaki F, Kanmura S, Numata M, Moriuchi A, Hasegawa S, Oketani M,
Ido A, Kusumoto K, Hasuike S, Nagata K, Kohara M and Tsubouchi H:
Increased rate of death related to presence of viremia among
hepatitis C virus antibody-positive subjects in a community-based
cohort study. Hepatology. 50:393–399. 2009.
|
|
15
|
Schirren CA, Jung MC, Gerlach JT, Worzfeld
T, Baretton G, Mamin M, Hubert Gruener N, Houghton M and Pape GR:
Liver-derived hepatitis C virus (HCV)-specific CD4(+) T cells
recognize multiple HCV epitopes and produce interferon gamma.
Hepatology. 32:597–603. 2000.
|
|
16
|
Itose I, Kanto T, Kakita N, Takebe S,
Inoue M, Higashitani K, Miyazaki M, Miyatake H, Sakakibara M,
Hiramatsu N, Takehara T, Kasahara A and Hayashi N: Enhanced ability
of regulatory T cells in chronic hepatitis C patients with
persistently normal alanine aminotransferase levels than those with
active hepatitis. J Viral Hepat. 16:844–852. 2009.
|
|
17
|
Liu J, Ali MA, Shi Y, Zhao Y, Luo F, Yu J,
Xiang T, Tang J, Li D, Hu Q, Ho W and Zhang X: Specifically binding
of L-ficolin to N-glycans of HCV envelope glycoproteins E1 and E2
leads to complement activation. Cell Mol Immunol. 6:235–244.
2009.
|
|
18
|
Ambrus G, Gál P, Kojima M, Szilágyi K,
Balczer J, Antal J, Gráf L, Laich A, Moffatt BE, Schwaeble W, Sim
RB and Závodszky P: Natural substrates and inhibitors of
mannan-binding lectin-associated serine protease-1 and -2: a study
on recombinant catalytic fragments. J Immunol. 170:1374–1382.
2003.
|
|
19
|
Poljacki M, Gajinov Z, Ivkov M, Matić M
and Golusin Z: Skin diseases and hepatitis virus C infection. Med
Pregl. 53:141–145. 2000.
|
|
20
|
Dumestre-Perard C, Ponard D, Drouet C,
Leroy V, Zarski JP, Dutertre N and Colomb MG: Complement C4
monitoring in the follow-up of chronic hepatitis C treatment. Clin
Exp Immunol. 127:131–136. 2002.
|
|
21
|
Banerjee A, Mazumdar B, Meyer K, Di
Bisceglie AM, Ray RB and Ray R: Transcriptional repression of C4
complement by hepatitis C virus proteins. J Virol. 85:4157–4166.
2011.
|
|
22
|
Wallis R, Dodds AW, Mitchell DA, Sim RB,
Reid KB and Schwaeble WJ: Molecular interactions between MASP-2,
C4, and C2 and their activation fragments leading to complement
activation via the lectin pathway. J Biol Chem. 282:7844–7851.
2007.
|
|
23
|
Pfeifer PH, Brems JJ, Brunson M and Hugli
TE: Plasma C3a and C4a levels in liver transplant recipients: a
longitudinal study. Immunopharmacology. 46:163–174. 2000.
|
|
24
|
Avirutnan P, Fuchs A, Hauhart RE, Somnuke
P, Youn S, Diamond MS and Atkinson JP: Antagonism of the complement
component C4 by flavivirus nonstructural protein NS1. J Exp Med.
207:793–806. 2010.
|
|
25
|
Zapata R: Clinical approach to the patient
with chronic hepatitis C infection and normal aminotransferases.
Ann Hepatol. 9(suppl): 72–79. 2010.
|
|
26
|
Zeuzem S, Diago M, Gane E, Reddy KR,
Pockros P, Prati D, Shiffman M, Farci P, Gitlin N, O’Brien CB,
Lamour F and Lardelli P; PEGASYS Study NR16071 Investigator Group.
Peginterferon alfa-2a (40 kilodaltons) and ribavirin in patients
with chronic hepatitis C and normal aminotransferase levels.
Gastroenterology. 127:1724–1732. 2004.
|
|
27
|
Hiramatsu N, Kurosaki M, Sakamoto N,
Iwasaki M, Sakamoto M, Suzuki Y, Sugauchi F, Tamori A, Kakinnuma S,
Matsuura K and Izumi N: Pretreatment prediction of anemia
progression by pegylated interferon alpha-2b plus ribavirin
combination therapy in chronic hepatitis C infection: decision-tree
analysis. J Gastroenterol. 46:1111–1119. 2011.
|
|
28
|
Puoti C: Hepatitis C virus with normal
transaminase levels. Dig Dis. 25:277–278. 2007.
|
|
29
|
Izumi N, Nishiguchi S, Hino K, Suzuki F,
Kumada H, Itoh Y, Asahina Y, Tamori A, Hiramatsu N, Hayashi N and
Kudo M: Management of hepatitis C: report of the consensus meeting
at the 45th annual meeting of the Japan Society of Hepatology
(2009). Hepatol Res. 40:347–368. 2010.
|