Introduction
Leukemia is a heterogeneous group of hematopoietic
malignancies that include a number of diverse and biologically
distinct subgroups, of which chronic myelogenous leukemia (CML) and
acute promyelocytic leukemia (APL) are two types. The former rarely
affects children, while the latter affects adults and children.
Thus, the treatment and prevention of leukemia is of great
importance. Leukemic cells are often a type of overgrown immature
cell. Thus, leukemia may be treated by the inhibition of cell
growth proliferation, induction apoptosis and terminal
differentiation. As medical science progresses, there is an
increasing amount of research being performed on the antitumor
function in natural medicine.
As an antioxidant, chlorogenic acid (CA) is a type
of ester that is widely studied in edible and medicinal plants. It
is isolated from Eucommiaceae plants and honeysuckle and has marked
antioxidant and anti-inflammatory effects. In addition, it may
prevent diabetes and cardiovascular disease (1–3).
Recent studies have demonstrated that CA has the activity of
induced human hepatoma and glioma cell apoptosis (4,5). CA
reportedly possesses antitumor activity via the induction of
apoptosis in human CML cell lines, including U937 and K562 cells
(6,7). However, the effects of CA on human
APL HL-60 cells remain unknown.
The current study aimed to investigate whether CA
inhibits proliferation and induces apoptosis in human leukemia
HL-60 cells. The results suggest that CA may be available for the
clinical treatment of human acute promyelocytic leukemia.
Materials and methods
Chemical reagents and cells
HL-60 cells were obtained from the China Center for
Type Culture Collection (Wuhan, China). CA was purchased from
Sigma-Aldrich (St. Louis, MO, USA) and was dissolved at a
concentration of 0.1 M in DMSO as a stock solution stored at −70°C.
Fetal bovine serum (FBS) was purchased from Gibco-BRL (Grand
Island, NY, USA). Hoechst and PI staining, DNA Ladder Extraction,
Trypan Blue Staining Cell Viability assay kits, propidium iodide
(PI) and RNase were purchased from Beyotime Institute of
Biotechnology (Shanghai, China). Giemsa stain was purchased from
the Beijing Dingguo Changsheng Biotechnology Co., Ltd. (Beijing,
China).
Cell culture
HL-60 cells were maintained in RPMI-1640 (Gibco-BRL)
medium supplemented with 10% heat-inactivated FBS, at 37°C in a 5%
CO2 humidified incubator. Cells were harvested by
centrifugation at 38 × g for 5 min and resuspended in fresh medium
every two days.
Cell viability analysis
Cells in logarithmic phase growth were seeded at a
concentration of 1×105 cells/ml in a 24-well plate
(Corning Incorporated, New York, NY, USA) and treated with 0, 1, 5
and 10 μM CA, respectively. Following 24, 48, 72, 96 and 120 h, the
cells were harvested, diluted by trypan blue working solution and
counted with an automated cell counter (Bio-Rad Laboratories, Inc.,
Hercules, CA, USA) to allow for growth curve construction.
Wright-Giemsa staining
Cells treated with CA at the indicated
concentrations for 48 h were harvested and collected on slides,
air-dried, stained with Wright for 5 min at room temperature and
one drop of Giemsa was added. The staining solution was diluted
with PBS to form a water break-free surface. After 15 min, the
slides were rinsed in deionized water and air-dried. The cells were
observed by light microscopy (Olympus Corporation, Tokyo, Japan)
and images were captured by digital camera (Olympus) (8).
Evaluation of apoptosis by nuclear
morphology
Cells in logarithmic phase growth were seeded at a
concentration of 1×105 cells/ml in a 24-well plate and
treated with CA (1, 5 and 10 μM, respectively). After 48 h, the
cells were incubated for 15 min at 37°C with the DNA-specific dye
Hoechst 33342 and propidium iodide (PI). Nuclear morphology was
investigated by fluorescence microscopy (Olympus) and images were
captured using a digital camera (Olympus).
DNA ladder analyze apoptosis
Cells treated with CA at the indicated
concentrations for 48 h were harvested and washed with PBS (pH
7.4). DNA was then extracted from the cells according to the DNA
Ladder Extraction kit instructions. Samples were separated by
electrophoresis on 1.5% (w/v) agarose gels containing GoldView and
subsequently the separated DNA ladders were visualized using a UV
transilluminator (Beijing Liuyi Instrument Factory, Beijing,
China). The size of the DNA ladders was determined by comparison
with DL2000 DNA markers (Takara Biotechnology Inc., Dalian,
China).
Cell cycle analysis
Cells treated with CA for 48 h were harvested and
washed twice with ice-cold PBS. The cells were fixed for 30 min at
4°C in ice-cold 70% ethanol. The cells were washed twice with PBS
and resuspended in 50 μg/ml DNase-free RNase (Sigma-Aldrich) at
37°C for 30 min. PI (25 μg/ml; Sigma -Aldrich) was added at 4°C for
30 min in the dark. The cells were then analyzed using a flow
cytometer (FACSCalibur, Becton-Dickinson, San Jose, CA, USA) and
histograms were analyzed by ModFit software (Becton-Dickinson).
Statistical analysis
Data obtained from experiments are presented as
means ± SEM from at least three independent experiments.
Statistical analyses were performed by one-way analysis of variance
followed by the Student’s t-test. P<0.05 and P<0.01 were
considered to indicate a statistically significant difference.
Results
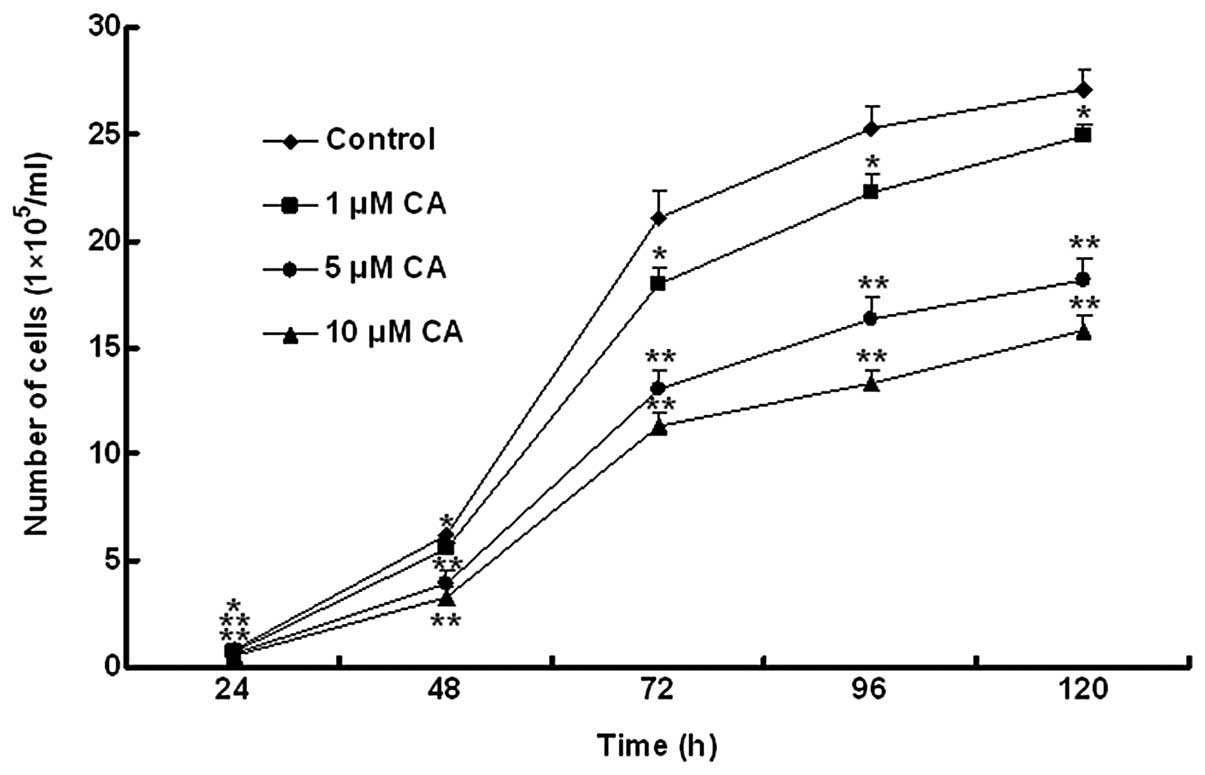
Cell viability analysis
Cell proliferation is a crucial marker for the
promotion and progression of carcinogenesis. As a result, to
investigate cell viability and the effect of CA on the
proliferation of HL-60 cells, cells treated with specific doses of
CA were examined using a growth curve constructed using trypan blue
staining. Cell viability in the control increased in a
time-dependent manner, reached a plateau at 72 h and persisted
until at least 120 h. This same change was observed in CA-treated
groups. However, when compared with the control, cell viability in
CA-treated groups decreased significantly at 72 h and cell
viability decreased with an increasing dose of CA. The results
indicated that growth of HL-60 cells reached a plateau at 72 h and
when treated with CA, cell growth was inhibited significantly in a
dose- and time-dependent manner compared with the control (Fig. 1). According to the growth curve,
the optimal HL-60 cell growth was identified at 48 h. In the
subsequent experiments, induction of apoptosis and inhibition of
cell proliferation were estimated, respectively, following 48-h
cell treatment with various doses of CA.
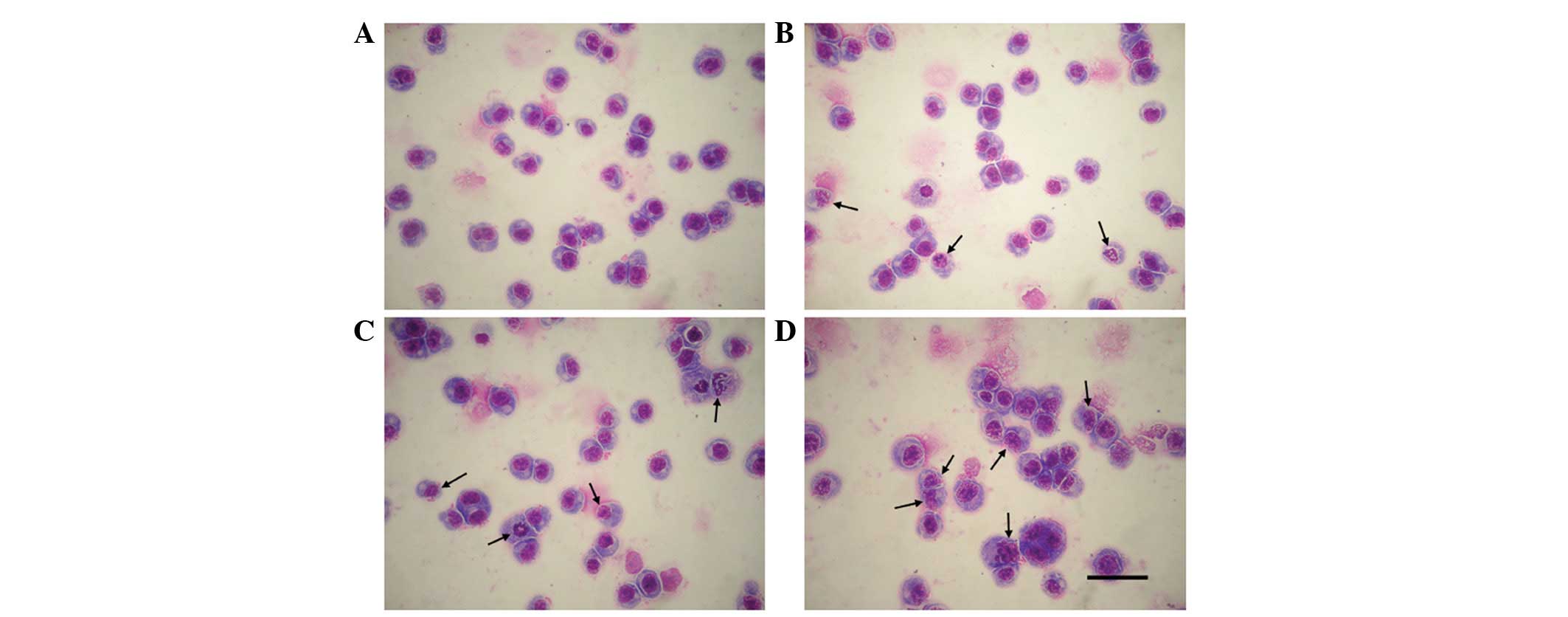
Effects of CA on the morphological
changes of HL-60 cells
As morphological changes of cells are key for the
detection of cell apoptosis, the morphological changes of HL-60
cells following 48 h of CA treatment were observed using
Wright’s-Giemsa staining. Following treatment for 48 h, the
irregular changes in morphology, including shrinkage of the cell
membrane were detected in the 1 μM CA group. As the concentration
of CA increased, apoptotic cells significantly increased and
generated more apoptotic bodies when compared with the control
(Fig. 2).
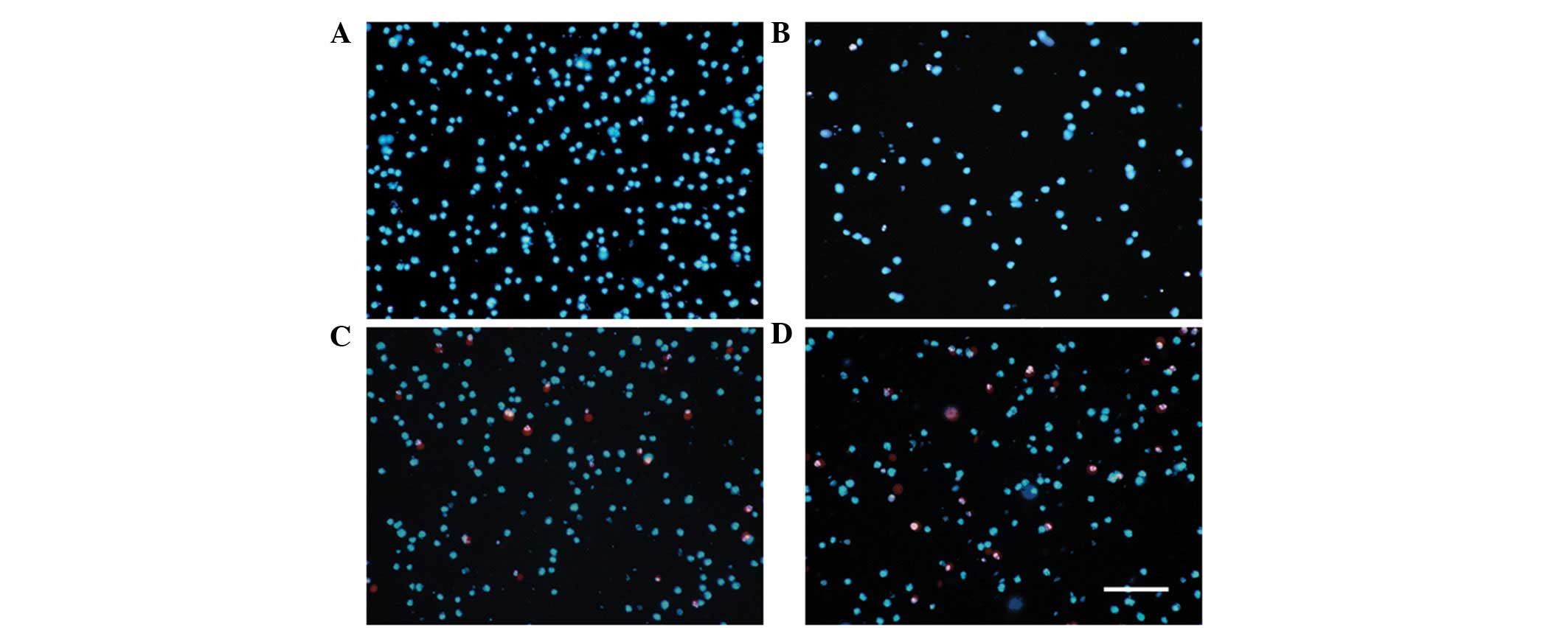
Apoptosis and necrosis assay
To detect the apoptosis and necrosis of HL-60 cells
following 48 h treatment with CA, the cells were stained with
Hoechst and PI. The results indicated that, in the CA-treated
groups, early apoptotic nuclei had an appearance of bright blue
fluorescence accompanied with cell nuclear morphological changes
and the late apoptotic cell membrane was damaged, which was dyed
red by PI, whereas the normal cells showed a faint blue. Following
48-h treatment with CA, the results revealed that together with the
concentration of CA increasing, the number of late apoptotic cells
increased in a dose-dependent manner (Fig. 3).
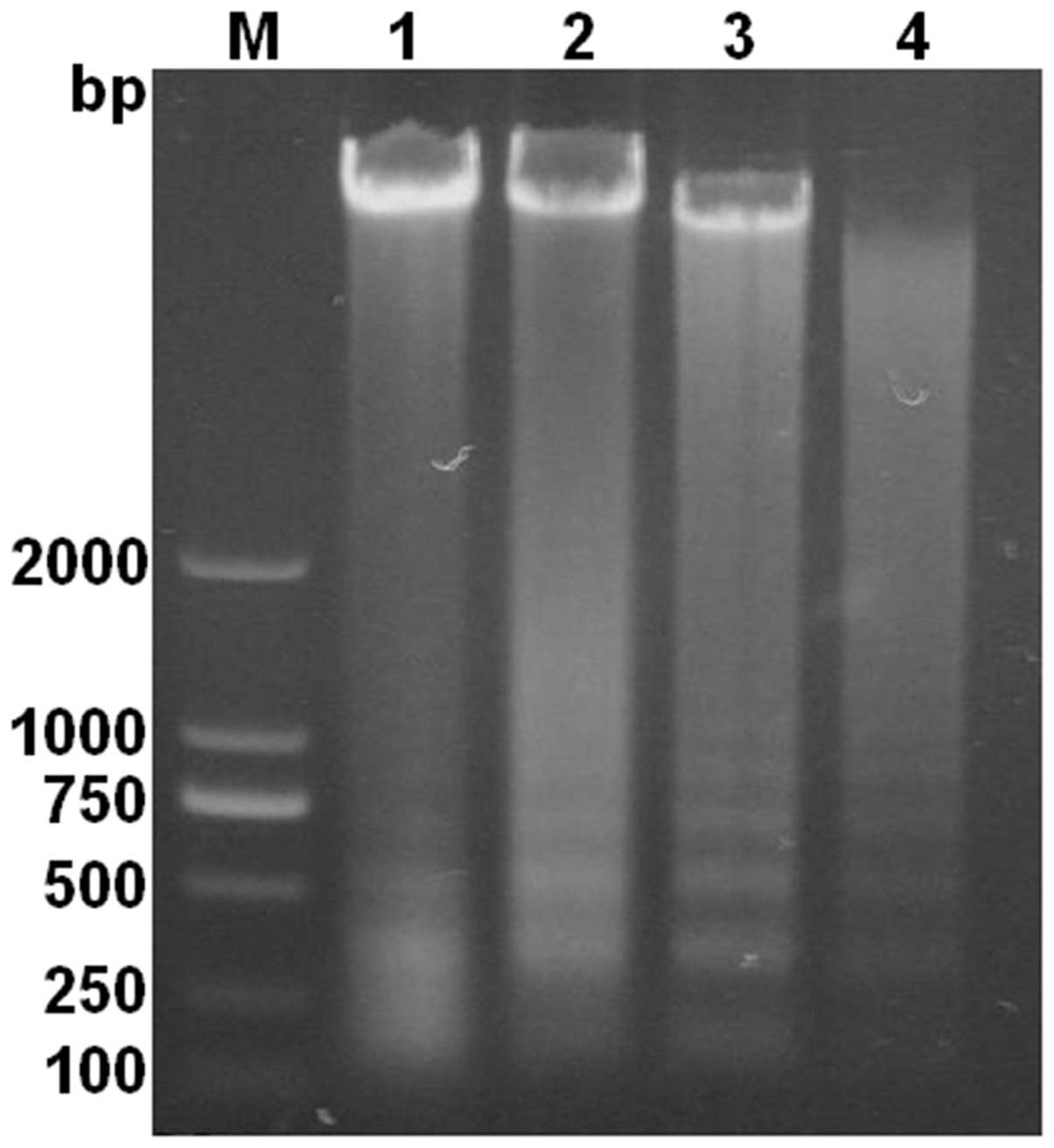
DNA ladder analysis apoptosis
Since the DNA ladder is a significant marker for the
study of apoptosis, to analyze the apoptosis of HL-60 cells at the
molecular level, agarose gel electrophoresis of DNA samples from
the cells treated with CA for 48 h was performed. A characteristic
DNA ladder was observed along with an increasing concentration of
CA (Fig. 4). The results also
showed that, following 48 h of CA treatment, apoptotic cells
increased in a dose-dependent manner.
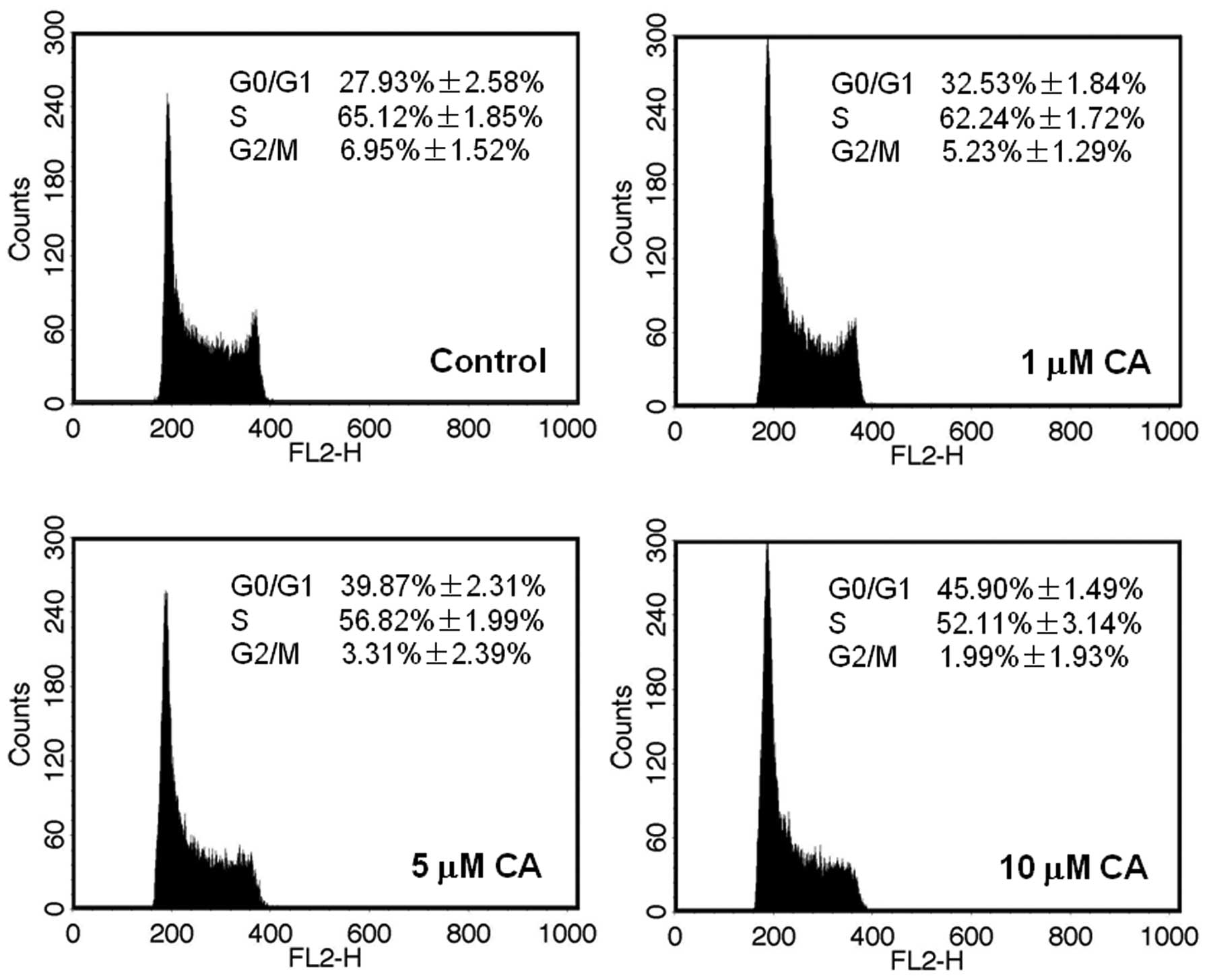
Cell cycle arrest by CA
Cell cycle arrest is a regulatory process that halts
progression through the cell cycle during normal phases (G1, S, G2
and M). To investigate whether the growth inhibitory effect of CA
resulted from growth arrest, the present study analyzed the cell
cycle following 48 h of treatment with 0, 1, 5 and 10 μM of CA. The
results indicated that the G0/G1 phase of HL-60 cells treated with
CA, increased significantly in concordance with a decrease of the S
and G2/M phases and CA induced an increased G0/G1 phase arrest in a
concentration-dependent manner (Fig.
5). When cells were treated with 1 μM of CA a slight
accumulation (~32.53%) of cells in G0/G1 phase was observed.
However, when 5 μM of CA was added to cells, a significant
accumulation (~39.87%) of cells in G0/G1 phase was observed. Cells
treated with 10 μM of CA resulted in a more significant (~45.90%)
enhancement of cells in G0/G1 phase.
Discussion
It is well established that cancer cells evade
apoptosis by the accumulation of a number of genetic and epigenetic
changes (9). Thus, leukemia may be
treated by the inhibition of APL cell growth proliferation and
induction of apoptosis. An increasing number of studies have
focused on natural drugs to treat leukemia. A number of previous
studies have shown that Artemisinin (10), Isoliquiritigenin (11), Pycnogenol (12) and other natural drugs have been
shown to effectively inhibit APL cell growth proliferation, induce
apoptosis and differentiation. Thus, it is crucial to identify
safer drugs that yield the same or improved effects, but with fewer
side effects.
A previous study revealed that a number of natural
drugs from plants were found to inhibit APL cell growth
proliferation and induce apoptosis in human APL cells. CA is a type
of phenolic antioxidant, with low toxicity and less side effects,
with a marked antioxidant and antibacterial effect. CA may prevent
colon (13) and liver cancer
(14). It reportedly possesses
antitumor activity via the induction apoptosis of CML cell lines,
including U937 and K562 cells. However, no study is currently
available on the use of CA treatment on APL HL-60 cells. Thus, the
current study investigated the ability of CA to inhibit HL-60 cell
growth proliferation and to induce apoptosis.
To clarify the effects of CA on growth proliferation
and apoptosis in human APL HL-60 cells, the present study first
investigated cell viability and the effect of CA on the
proliferation of HL-60 cells using a growth curve constructed using
trypan blue staining. According to the growth curve, the growth of
HL-60 cells reached a plateau at 72 h. When treated with CA, the
cell growth was inhibited significantly in a dose- and
time-dependent manner and the optimal time of HL-60 cell growth was
identified at 48 h (Fig. 1). Thus,
in the subsequent experiments, the inhibition of proliferation and
induction of apoptosis were estimated following 48 h treatment with
various doses of CA. Cell apoptosis in cell development, growth,
maturation and differentiation is important in the process of
carcinogenesis and is significant in the research and treatment of
tumor development. In the current study, Wright-Giemsa staining was
implemented to observe the morphological changes of HL-60 cells
following 48 h CA treatment. The cells treated with CA revealed the
characteristics of apoptosis (Fig.
2). Thus, this result suggests that CA may induce apoptosis in
HL-60 cells. The distinct morphological features of apoptosis in
HL-60 cells treated with CA were observed in a
concentration-dependent manner, when cells were observed by
fluorescence microscopy following staining with Hoechst and PI
(Fig. 3). A significant
characteristic DNA ladder was observed with an increased CA
concentration, which further suggests that CA has specific leukemia
cell apoptosis-inducing activity in HL-60 cells at the molecular
level (Fig. 4).
Cancer cells evade programmed cell death, thus
increasing their life span, which is different from normal cells
(9). A number of previous studies
have shown that numerous anticancer drugs achieve cell cycle
regulation through specific blockage of the cell cycle in the
two-phase transformation point of G0/G1 to S and G2 to M (15–17).
In the present study, the cell cycle was analyzed using flow
cytometry and the results showed that CA may induce HL-60 cell
G0/G1 phase arrest with a decrease of S and G2/M phase. The current
study has found that CA may induce HL-60 cell G0/G1 phase arrest
and the result suggests that CA also has specific HL-60 cell growth
inhibition-inducing activity.
The Bcl-2 gene family is significant in the
regulation of cell apoptosis. It has been demonstrated that
flavonoid compounds, including baicalin may induce apoptosis in
HL-60 cell lines by downregulating the expression of Bcl-2
(18). Besides the Bcl-2 gene
family, the caspase family is also crucial in apoptosis. As the
most important member of the caspase family, caspase-3 has been
widely studied. Under a variety of apoptotic stimuli, procaspase-3
is hydrolyzed to active caspase-3 and cleaved with PARP
(poly(ADP-ribose) polymer-rase). As a result, the activity of the
PARP-negatively regulated Ca2+/Mg2+-dependent
endonuclease increased to cleave the DNA between nucleosomes and
induce apoptosis (19).
Phosphatidylinositol-3-kinase (PI3K) is an important intracellular
signaling pathway associated with apoptosis and Akt is a potent
kinase for Bad, which is an apoptotic member of the Bcl-2 family
which may displace Bax from binding to Bcl-2 and Bcl-XL, resulting
in cell death (20). Thus, it is
hypothesized that CA may induce cell apoptotis by downregulating
Akt expression in Hl-60 cells. CA is also hypothesized to inhibit
proliferation and promote the apoptosis of the HL-60 cells with a
possible mechanism involving the downregulation of the expression
of Akt, which further downregulates apoptosis-associated proteins,
including Bcl-2 and caspase-3. Therefore, a detailed mechanisms of
the inhibition of proliferation and promotion of apoptosis in HL-60
cells treated with CA should be further examined.
In conclusion, to the best of our knowledge, the
current study provided evidence, for the first time, that CA may
inhibit growth proliferation and induce apoptosis in human acute
promyelocytic leukemia HL-60 cells. The molecular mechanism of this
process requires further investigation in future studies. The
results of this study provides a new theoretical basis and
therapeutic strategy for CA in the clinical treatment of acute
promyelocytic leukemia.
Acknowledgements
This study was supported by a grant from the Top
Innovative Talents Training Fund for College Students from
Chongqing University of Technology (no. BC201205).
References
|
1
|
Gallus S, Tavani A, Negri E and La Vecchia
C: Does coffee protect against liver cirrhosis? Ann Epidemiol.
12:202–205. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Namba T and Matsuse T: A historical study
of coffee in Japanese and Asian countries: focusing the medicinal
uses in Asian traditional medicines. Yakushigaku Zasshi. 37:65–75.
2002.(In Japanese).
|
|
3
|
Phan TT, Sun L, Bay BH, Chan SY and Lee
ST: Dietary compounds inhibit proliferation and contraction of
keloid and hypertrophic scar-derived fibroblasts in vitro:
therapeutic implication for excessive scarring. J Trauma.
54:1212–1224. 2003. View Article : Google Scholar
|
|
4
|
Belkaid A, Currie JC, Desgagnés J and
Annabi B: The chemopreventive properties of chlorogenic acid reveal
a potential new role for the microsomal glucose-6-phosphate
translocase in brain tumor progression. Cancer Cell Int. 6:72006.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Granado-Serrano AB, Martin MA,
Izquierdo-Pulido M, et al: Molecular mechanisms of (−)-epicatechin
and chlorogenic acid on the regulation of the apoptotic and
survival/proliferation pathways in a human hepatoma cell line. J
Agr Food chem. 55:2020–2027. 2007.
|
|
6
|
Bandyopadhyay G, Biswas T, Roy KC, et al:
Chlorogenic acid inhibits Bcr-Abl tyrosine kinase and triggers p38
mitogen-activated protein kinase-dependent apoptosis in chronic
myelogenous leukemic cells. Blood. 104:2514–2522. 2004. View Article : Google Scholar
|
|
7
|
Yang JS, Liu CW, Ma YS, et al: Chlorogenic
acid induces apoptotic cell death in U937 leukemia cells through
caspase- and mitochondria-dependent pathways. In Vivo. 26:971–978.
2012.PubMed/NCBI
|
|
8
|
Zhang XM, Gao N, Chen RX, Xu HZ and He QY:
Characteristics of boningmycin induced cellular senescence of human
tumor cells. Yao Xue Xue Bao. 45:589–594. 2010.(In Chinese).
|
|
9
|
Klein G: Cancer, apoptosis, and nonimmune
surveillance. Cell Death Differ. 11:13–17. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim SH, Chun SY and Kim TS:
Interferon-alpha enhances artemisinin-induced differentiation of
HL-60 leukemia cells via a PKC alpha/ERK pathway. Eur J Pharmacol.
587:65–72. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li D, Wang Z, Chen H, et al:
Isoliquiritigenin induces monocytic differentiation of HL-60 cells.
Free Radical Bio Med. 46:731–736. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Huang WW, Yang JS, Lin CF, Ho WJ and Lee
MR: Pycnogenol induces differentiation and apoptosis in human
promyeloid leukemia HL-60 cells. Leuk Res. 29:685–692. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tavani A, Pregnolato A, La Vecchia C, et
al: Coffee and tea intake and risk of cancers of the colon and
rectum: a study of 3,530 cases and 7,057 controls. Int J Cancer.
73:193–197. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Larsson SC and Wolk A: Coffee consumption
and risk of liver cancer: a meta-analysis. Gastroenterology.
132:1740–1745. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen CJ, Wen YF, Huang PT, et al:
2-(1-Hydroxethyl)-4,8-dihydrobenzo[1,2-b:5,4-b′]dithiophene-4,8-dione
(BTP-11) enhances the ATRA-induced differentiation in human
leukemia HL-60 cells. Leuk Res. 33:1664–1669. 2009.PubMed/NCBI
|
|
16
|
Guney I, Wu S and Sedivy JM: Reduced c-Myc
signaling triggers telomere-independent senescence by regulating
Bmi-1 and p16(INK4a). Proc Natl Acad Sci USA. 103:3645–3650. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu LL, Chen N, Yuan X, et al: The
mechanism of alteronol inhibiting the proliferation of human
promyelocytic leukemia HL-60 cells. Yao Xue Xue Bao. 47:1477–1482.
2012.(In Chinese).
|
|
18
|
Shieh DE, Cheng HY, Yen MH, et al:
Baicalin-induced apoptosis is mediated by Bcl-2-dependent, but not
p53-dependent, pathway in human leukemia cell lines. Am J Chin Med.
34:245–261. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ivana Scovassi A and Diederich M:
Modulation of poly(ADP-ribosylation) in apoptotic cells. Biochem
Pharmacol. 68:1041–1047. 2004.PubMed/NCBI
|
|
20
|
Zheng J, Hu JD, Chen YY, et al: Baicalin
induces apoptosis in leukemia HL-60/ADR cells via possible
down-regulation of the PI3K/Akt signaling pathway. Asian Pac J
Cancer Prev. 13:1119–1124. 2012. View Article : Google Scholar : PubMed/NCBI
|