Introduction
Glioblastoma is a common type of malignant tumor in
the central nervous system, characterized by aggressive behavior,
rapid progression, high recurrence rates and poor prognosis. The
median survival of patients with glioblastoma patients is <1
year (1–4). Although progress has been made in the
biological research into and treatment of glioblastoma, the primary
treatment for glioblastoma is surgery combined with postoperative
radiotherapy, chemotherapy and immunotherapy; however, the overall
therapeutic effect remains poor. Knowledge about this malignant
tumor is limited, and its exact pathogenesis remains unclear.
Therefore, it is of importance to examine the molecular mechanism
underlying the progression of glioblastoma.
The mammalian synaptotagmin (SYT) family consists of
17 isoforms. SYT proteins are sensors of calcium signaling in
cellular secretions (5–8). SYT consists of a transmembrane helix
close to the N-terminus, a flexible region of variable length, and
two C2 domains (9), with an
average length of ~135 amino acid residues, and each of them is
composed of eight β-folding and three ring structures. SYT1-SYT7
are all expressed in the brain, particularly SYT1, SYT2, SYT3 and
SYT7 (10). SYT7 is widely
distributed in different types of biological tissue, and it may be
spliced to >12 SYT7 variants with differing genetic structures.
SYT7 is located in core vesicles, opposite the reticular structures
of the Golgi apparatus and lysosomes in PC12 cells (11). SYT is able to interact with soluble
N-ethylmaleimide-sensitive fusion protein attachment protein
receptor (SNARE) to regulate fusion of the vesicle membrane and
plasma membrane, which serves an important role in cell secretion
(12). A previous study
demonstrated that SYT7 is able to regulate apoptosis, cell
differentiation, cytokine activity, amine biosynthesis,
transcriptional regulation and vesicular transport (13). SYT7 is involved in
lysosome-mediated repair mechanisms and induces apoptosis (14), and it is associated with prostate
cancer and serves an important role in prostate cancer development
(15). However, the roles of SYT7
in the progression of glioma remain unknown.
The present study demonstrated that SYT7 was
expressed in glioblastoma, and that the proliferation of U87MG
cells was inhibited as SYT7 was downregulated by a lentiviral
vector expressing SYT7 short hairpin (sh)RNA, via the promotion of
cellular apoptosis. The results of the present study demonstrated
that downregulation of SYT7 may inhibit glioblastoma growth by
promoting cellular apoptosis. These results enhance the possibility
that SYT7 may be a therapeutic target for glioblastoma
treatment.
Materials and methods
Cell culture and reagents
The human glioma cell lines A172 (cat. no. TCHu171),
U251 (cat. no. TCHu58) and U87 (cat. no. TCHu138), and human
epithelial 293T cells (cat. no. GNHu17) (all from Cell Bank of the
Chinese Academy of Sciences, Shanghai, China) were cultured in
Dulbecco's modified Eagle's medium (DMEM; Hyclone; GE Healthcare
Life Sciences, Logan, UT, USA) supplemented with 10%
heat-inactivated fetal bovine serum (Hyclone; GE Healthcare Life
Sciences) at 37°C in an atmosphere which contains 5%
CO2. The U87 cell line employed in the present study is
known to be misidentified/cross-contaminated with an unknown cell
line, which is most likely also a glioblastoma cell line (16).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) assay
Total RNA was extracted from the three glioma cell
lines (A172, U251 and U87) using the TRIzol total RNA reagent
(Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA).
cDNA synthesis was performed using 2 µg total RNA using the
RevertAid™ H Minus First Strand cDNA Synthesis kit
(Takara Bio, Inc., Otsu, Japan). The SYT7 primers were obtained
from Shanghai GeneChem Co., Ltd. (Shanghai, China), and GAPDH was
used as an internal control. The primers were as follows: SYT7
forward, 5′-ACTCCATCATCGTGAACATCATC-3′ and reverse,
5′-TCGAAGGCGAAGGACTCATTG-3′; GAPDH forward,
5′-TGACTTCAACAGCGACACCCA-3′ and reverse,
5′-CACCCTGTTGCTGTAGCCAAA-3′. qPCR was performed using the SYBR
PrimeScript qPCR kit (Takara Bio, Inc.) on an Applied Biosystems
7300 Fluorescent Quantitative PCR System (Applied Biosystems;
Thermo Fisher Scientific, Inc.). The reaction mixtures were
incubated at 95°C for 30 sec, with 45 amplification cycles at 95°C
for 5 sec, 60°C for 30 sec and 72°C for 60 sec, followed by a final
extension of 72°C for 7 min. The PCR products of SYT7 and GAPDH
were 178 and 121 bp, respectively. The relative expression of SYT7
mRNA was calculated via the 2−ΔΔCq method (17), using the GAPDH mRNA expression
level for normalization.
Construction of the shRNA lentiviral
vector and transfection
The lentiviruses expressing shRNA targeting the
sequence of the SYT7 gene (5′-TCACCGTGAAGATCATGAA-3′) and negative
control shRNA were purchased from Shanghai GeneChem Co., Ltd. The
shRNAs were subsequently cloned into a pGCSIL-green fluorescent
protein lentiviral vector with AgeI/EcoRI sites to
generate recombinant the lentiviral shRNA expression vectors.
Lentiviral vectors and packaging vectors were transfected into 293T
cells using Lipofectamine 2000, according to manufacturer's
instructions. Lentiviral particles were purified using
ultracentrifugation, and an endpoint dilution assay was performed
to determine the titer of the lentiviruses. The U87MG glioblastoma
cells, seeded in 6-well plates at a density of 4.0×105
cells/well, were infected with shSYT7-lentivirus (4×108
TU/ml × 2.5 µl) or negative control (8×108 TU/ml × 1.25
µl) lentivirus. The cells were observed under a fluorescence
microscope (MicroPublisher 3.3RTV; Olympus, Tokyo, Japan) 72 h
subsequently. Following 5 days of infection, the knockdown
efficiency of the shRNA-SYT7 vectors was investigated via qPCR
analysis.
Cell growth assay
Cell growth was measured using a Celigo Image
Cytometer (Nexcelom Bioscience LLC, Lawrence, MA, USA). U87MG cells
at the logarithmic phase following infection with the shCtrl
lentivirus or shSYT7 lentivirus were seeded at 2×103
cells/well in 96-well plates, 3 duplicates/group, and incubated at
37°C for 5 days. The cells in the plates were counted using the
Celigo Image Cytometer for each day's analysis.
Caspase-3/7 assay
U87MG cells at the logarithmic phase following
infection with the shCtrl lentivirus or shSYT7 lentivirus were
seeded in a 96-well plate and incubated at 37°C for 5 days in a 5%
CO2 incubator. Caspase-Glo3/7 (Promega, Madison, WI,
USA) buffer and powder were mixed to generate the Caspase-Glo
reaction buffer and, following cell counting, the cell suspension
was adjusted to a concentration of 1×104 cells/well at
room temperature. Cells were seeded in a new 96-well plate and a
control group was designated; control cells were plated in the same
manner however, they were not treated with reaction buffer. A total
of 100 µl Caspase-Glo reaction buffer was added to each well, the
cells were incubated for 2 h at 37°C, and the signal intensity was
measured using a Multiskan Spectrum microplate reader at 480 nm
(Tecan Group Ltd., Männedorf, Switzerland). All experiments were
performed in triplicate.
Apoptosis assay using flow
cytometry
Annexin V-allophycocyanin (APC) single labeling was
performed to detect cellular apoptosis. U87MG cells were infected
with shSYT7 or shCtrl lentiviruses and incubated at 37°C. U87MG
cells were harvested by trypsinization and rinsed in 1X binding
buffer, followed by the addition of 10 µl Annexin V-APC buffer
(eBioscience; Thermo Fisher Scientific, Inc.) for 10 min staining
at room temperature. A FACSCalibur II cell sorter and CellQuest
Research Software (version 5.1; BD Biosciences, San Diego, CA, USA)
were used for the assay. All experiments were performed in
triplicate.
MTT cell proliferation assay
An MTT Cell Proliferation Assay kit (American Type
Culture Collection, Manassas, VA, USA) was used for the cell
proliferation assay, according to the manufacturer's instructions.
Cells were rinsed with PBS at the end of the culture period and the
MTT reagents were added to the 96-well plate according to the
manufacturer's instructions. DMSO was then used to dissolve the
formazan and the absorbance was measured using a microplate reader
at 570 nm. All experiments were repeated three times.
Western blot analysis
The cells were harvested at 48 h following
transfection and lysed in radioimmunoprecipitation assay buffer
(Beyotime Institute of Biotechnology, Shanghai, China) for 30 min
at 4°C. Proteins were extracted and the concentration was
determined using a Bicinchoninic Acid Protein Assay kit (Pierce;
Thermo Fisher Scientific, Inc.). Equal amounts of total protein (40
µg) of each group was separated by SDS-PAGE on a 12.5% gel
according to the Laemmli method, as described by Liu et al
(18), and transferred to
polyvinylidene fluoride membranes. Membranes were blocked in TBST
buffer (containing 0.1% Tween-20) with 5% skim milk at room
temperature for 1 h, and then incubated with the following primary
antibodies overnight at 4°C: Polyclonal mouse anti-SYT7 (cat. no.
SAB1412773, 1:2,000; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany)
and anti-GAPDH antibody (cat. no. sc-47724, 1:2,000; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA). Goat anti-mouse
immunoglobulin G conjugated with horseradish peroxidase (cat. no.
sc-2005; Santa Cruz Biotechnology, Inc.) was used as the secondary
antibody at a 1:2,000 dilution for a 2 h incubation at room
temperature. The bound antibodies were detected using an Enhanced
Chemiluminescence Plus Western Blotting Detection System (GE
Healthcare Life Sciences, Little Chalfont, UK). GAPDH was used as
an internal control to normalize the SYT7 expression levels.
Statistical analysis
Data were statistically analyzed using a
χ2 test (for PCR, caspase-3/7 assay and FACS analysis)
and one-way analysis of variance with Scheffe's post hoc test,
using the SPSS 20.0 statistical software package (IBM Corp.,
Armonk, NY, USA). Data are presented as the mean ± standard
deviation. P<0.05 was considered to indicate a statistically
significant difference.
Results
Expression of SYT7 in three human
glioma cell lines
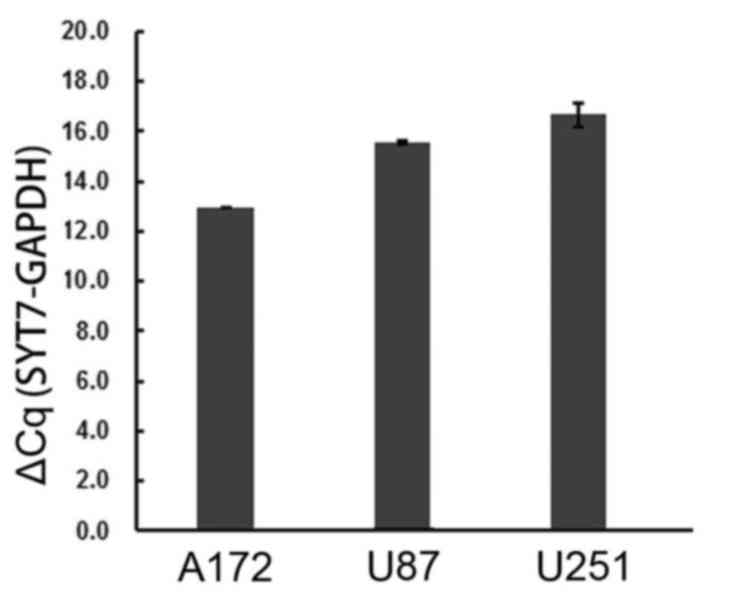
The expression levels of SYT7 were examined in three
glioma cell lines (A172, U251 and U87). The expression levels of
SYT7 mRNA were evaluated by RT-qPCR analysis. The results
demonstrated that SYT7 was expressed in all three human glioma cell
lines (Fig. 1). GAPDH was used as
an internal control.
Knockdown efficiency in 293T cells as
determined by western blotting
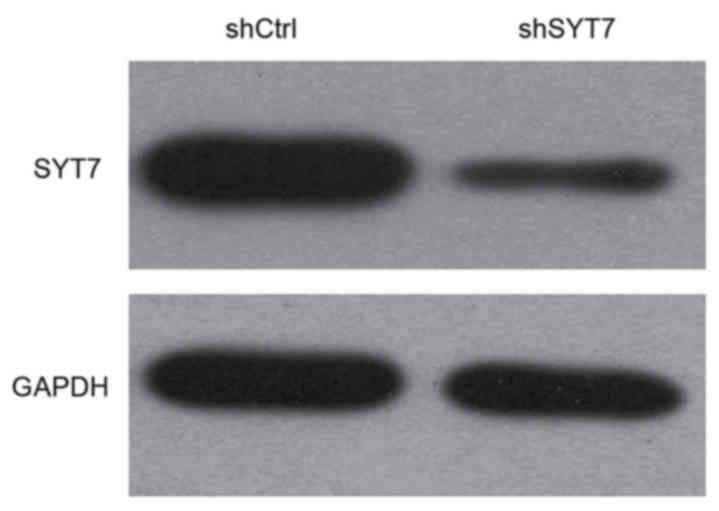
In order to determine the target knockdown
efficiency and exogenous selection, 293T cells were infected with
shSYT7 lentivirus and shCtrl lentivirus, and the SYT7 protein
expression levels were measured by western blotting. It was
observed that the protein expression level of SYT7 was decreased by
shSYT7 compared with the shCtrl group (Fig. 2).
shSYT7 works efficiently in U87MG
cells
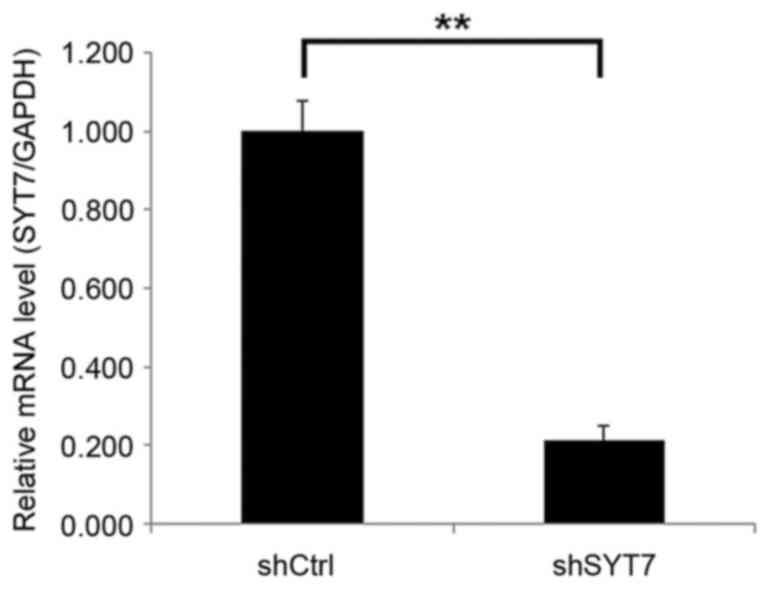
U87MG cells were infected with shSYT7 lentivirus and
shCtrl lentivirus in order to demonstrate the efficacy of shSYT7 in
U87MG cells. The mRNA expression level of SYT7 was significantly
downregulated by shSYT7; treatment with shRNA decreased the level
of SYT7 mRNA to 20% of the control value (Fig. 3), as determined by qPCR
analysis.
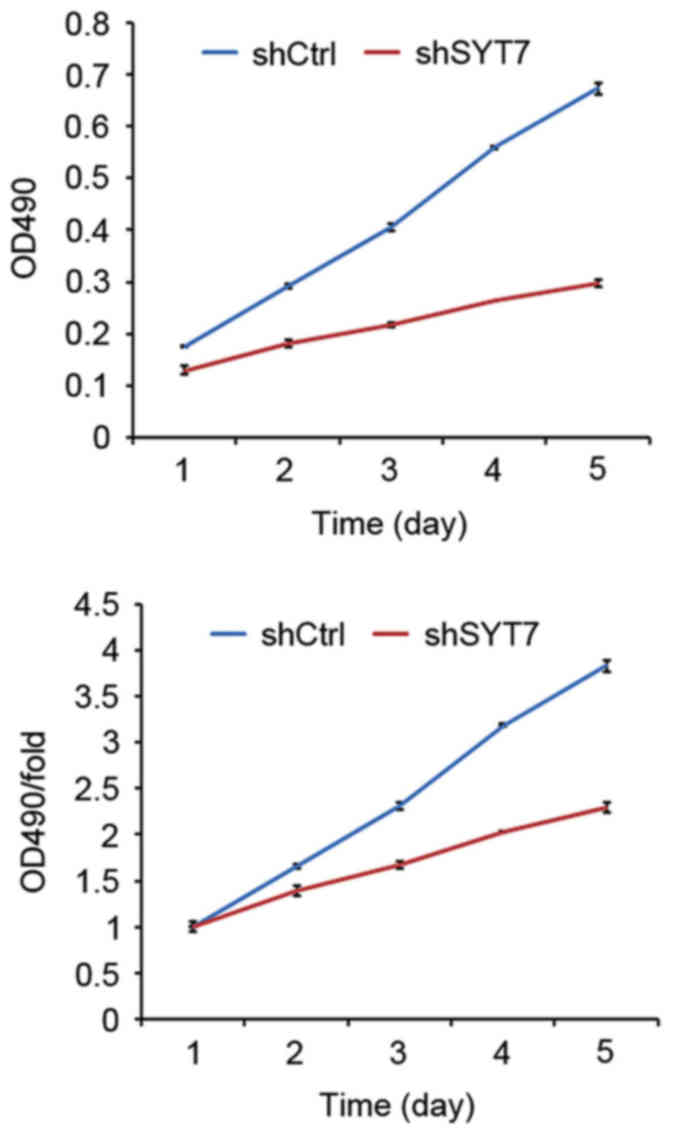
Cell proliferation is inhibited by
shSYT7 by promoting cellular apoptosis
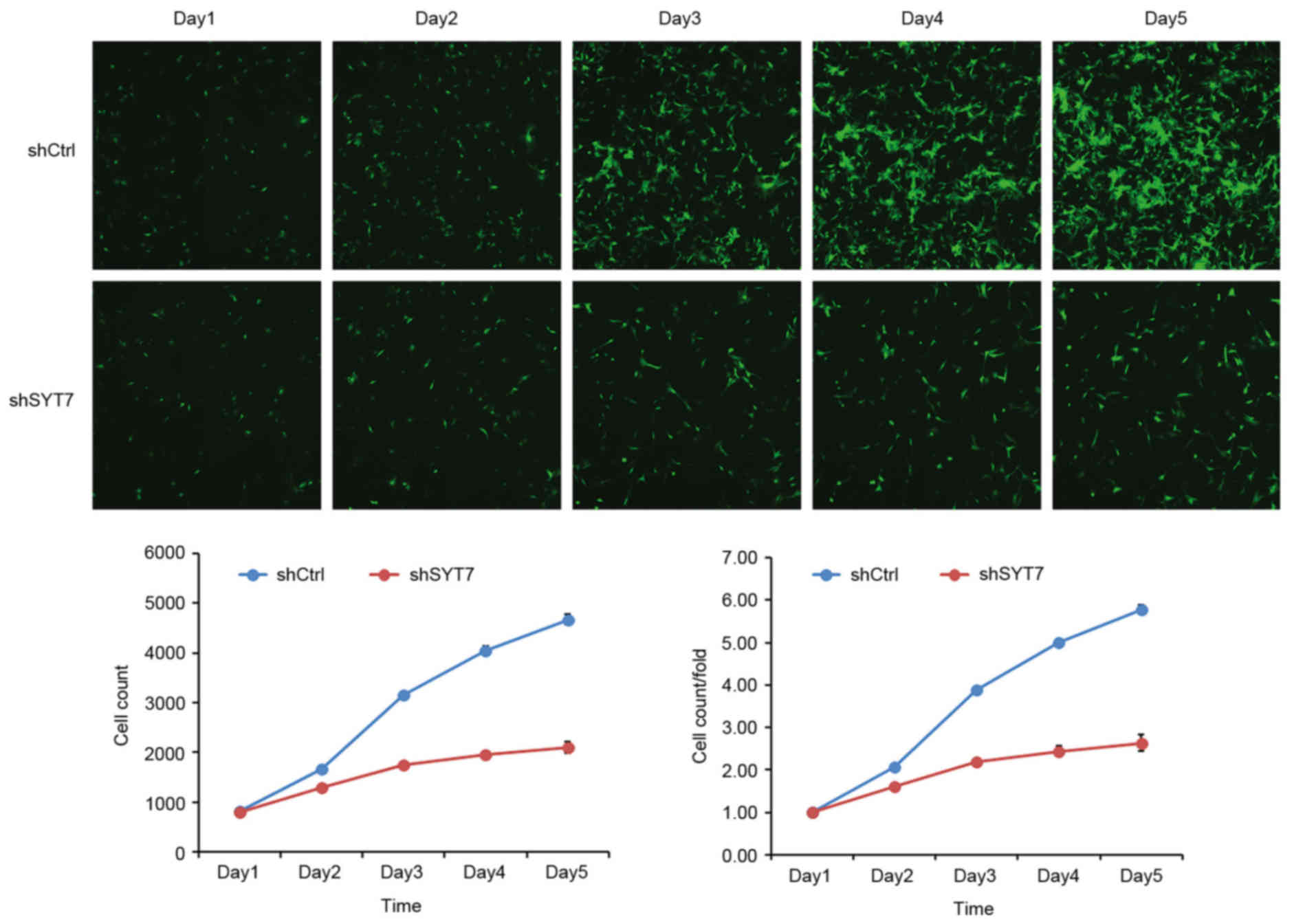
To examine the effect of SYT7 on cell proliferation,
U87MG cells were infected with shSYT7 and shCtrl lentiviruses.
Cells were seeded at 2×103 cells/well in 96-well plates
and counted using the Celigo Image Cytometer for 5 days. It was
observed that U87MG cell growth was inhibited by shSYT7 compared
with the shCtrl group (Fig. 4). In
addition, an MTT cell proliferation assay was performed 3 days
subsequent to infection. It was observed that the results were
consistent with the Celigo Image Cytometer Analysis. U87MG cell
proliferation was markedly inhibited by shSYT7 compared with the
shCtrl group, with a relative survival of 33.8% of the control
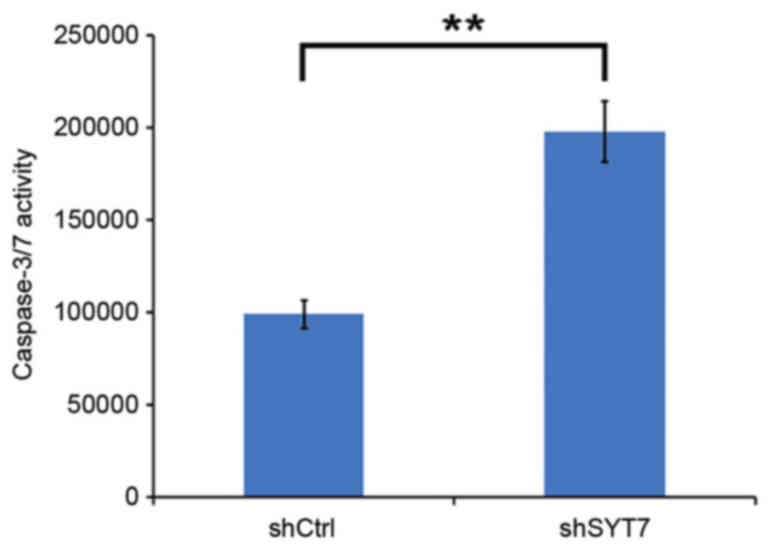
(Fig. 5). To examine the mechanism
underlying cell growth inhibition, a caspase-3/7 assay and an
apoptosis assay using flow cytometry were performed. It was
observed that the activity of caspase-3/7 was significantly
increased in the shSYT7 group compared with the shCtrl group
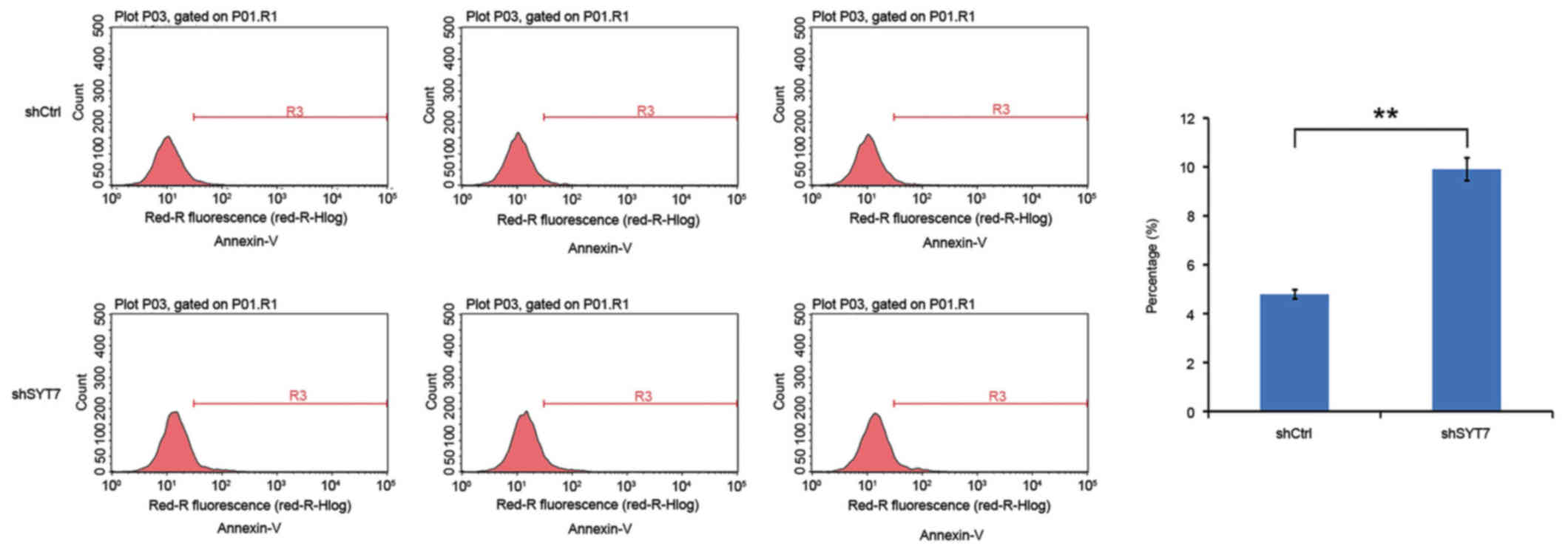
(Fig. 6). The results of the flow
cytometry analysis indicated that U87MG cellular apoptosis was
significantly increased in the shSYT7 group compared with the
shCtrl group (Fig. 7). Therefore,
the results of the present study demonstrated that the
downregulation of SYT7 suppressed cell proliferation by promoting
cellular apoptosis.
Discussion
In the present study, it was demonstrated that the
downregulation of SYT7 inhibited glioblastoma growth by promoting
cellular apoptosis. The expression levels of SYT7 were examined in
three glioma cell lines (A172, U251 and U87), and it was observed
that SYT7 was expressed in all the cell lines; however, only the
mRNA levels were assayed, and the expression of SYT7 in
glioblastoma multiforme or high-grade glioma tumor samples requires
further study. Western blotting or an immunohistochemistry assay
may be used to determine the SYT7 expression. In the present study,
the target knockdown efficiency in 293T cells was determined using
western blotting. The SYT7 protein level was downregulated by
shSYT7, and shSYT7 worked efficiently in U87MG cells. An MTT assay
and Celigo Image Cytometer Analysis were performed for the
examination of cell proliferation, and it was observed that U87MG
cell proliferation was significantly inhibited by shSYT7. In
addition, a caspase-3/7 assay and an apoptosis assay by flow
cytometry were performed to examine the mechanism of cell growth
inhibition. It was observed that the activity of caspase-3/7 and
U87MG cellular apoptosis was significantly increased in shSYT7
group. However, it is important to note that these conclusions are
taken from a single cell line and only the potential role of SYT7
in cellular apoptosis was assessed; further in vitro assays
are required to determine the biological role of SYT7 in
mitochondrial function, for example.
Glioma is the most common type of human cerebral
tumor, with a highly malignant character and poor prognosis
(19). The primary treatment for
glioblastoma is surgery combined with postoperative radiotherapy,
chemotherapy and immunotherapy. As progress in the biological
research and treatment of glioblastoma has been made, increasing
numbers of gene therapies have been trialed in glioblastoma,
including anti-angiogenesis (20),
hypoxic tumor-targeting therapy (21) and RNA interference treatment
(22), which have achieved
promising results.
SYT is a protein sensor of calcium signaling
(5–8); it is able to interact with SNARE to
regulate the fusion of vesicle membranes and the plasma membrane,
serving important roles in cellular secretion (12). The expression of SYT7 is associated
with prostate cancer and serves an important role in prostate
cancer development (15). However,
the SYT family has numerous members which are widely distributed
and exhibit complex functions (23). SYT 7 is considered to be the main
Ca2+ sensor of synaptic vesicles (13,24).
Lysosomes are an essential component of plasma membrane repair. The
exocytosis of lysosomes is induced by Ca2+ and depends
on the function of SYT7 (14). The
possible mechanism of action of SYT7 in glioma involves the
downregulation of SYT7 which leads to the inhibition of
lysosome-dependent plasma membrane repair, thereby inducing
apoptosis and inhibiting cell proliferation. However, the molecular
mechanism requires further clarification. The intracellular
localization of other subtypes of SYT remains to be elucidated, and
their physiological function and interactions with other proteins
remain unclear. Further research is required into the physiological
function and molecular mechanisms of SYT proteins.
In conclusion, the present study demonstrated that
the downregulation of SYT7 inhibited glioblastoma growth by
promoting cellular apoptosis. SYT7 may be considered to be a novel
therapeutic target for glioblastoma. However, the mechanism
underlying the effects of SYT7 in glioblastoma remains to be
elucidated, and more in-depth research is required.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (grant no. 81660420), the
Construction Plan of the Superior Science and Technology Innovation
Team of Jiangxi Province (grant no. 20152BCB24009), the Foreign
Science and Technology Cooperation Plan of Jiangxi Province (grant
no. 20151BDH80009), the Traditional Chinese Medicine Scientific
Research Project of Jiangxi Province Health and Family Planning
Commission (grant no. 2014A103) and the Guiding Science and
Technology Project of Nanchang [grant no. 2014 (196)].
References
|
1
|
Krex D, Klink B, Hartmann C, von Deimling
A, Pietsch T, Simon M, Sabel M, Steinbach JP, Heese O, Reifenberger
G, et al: Long-term survival with glioblastoma multiforme. Brain.
130:2596–2606. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Xiao B, Zhou X, Ye M, Lv S, Wu M, Liao C,
Han L, Kang C and Zhu X: MicroRNA-566 modulates vascular
endothelial growth factor by targeting Von Hippel-Landau in human
glioblastoma in vitro and in vivo. Mol Med Rep. 13:379–385. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wu M, Fan Y, Lv S, Xiao B, Ye M and Zhu X:
Vincristine and temozolomide combined chemotherapy for the
treatment of glioma: A comparison of solid lipid nanoparticles and
nanostructured lipid carriers for dual drugs delivery. Drug Deliv.
23:2720–2725. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Alves TR, Lima FR, Kahn SA, Lobo D, Dubois
LG, Soletti R, Borges H and Neto VM: Glioblastoma cells: A
heterogeneous and fatal tumor interacting with the parenchyma. Life
Sci. 89:532–539. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rickman C, Craxton M, Osborne S and
Davletov B: Comparative analysis of tandem C2 domains from the
mammalian synaptotagmin family. Biochem J. 378:681–686. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sugita S, Shin OH, Han W, Lao Y and Südhof
TC: Synaptotagmins form a hierarchy of exocytotic Ca(2+) sensors
with distinct Ca(2+) affinities. EMBO J. 21:270–280. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rizo J, Chen X and Araç D: Unraveling the
mechanisms of synaptotagmin and SNARE function in neurotransmitter
release. Trends Cell Biol. 16:339–350. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gustavsson N and Han W: Calcium-sensing
beyond neurotransmitters: Functions of synaptotagmins in
neuroendocrine and endocrine secretion. Biosci Rep. 29:245–259.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vasquez JK, Chantranuvatana K, Giardina
DT, Coffman MD and Knight JD: Lateral diffusion of proteins on
supported lipid bilayers: Additive friction of synaptotagmin 7
C2A-C2B tandem domains. Biochemistry. 53:7904–7913. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Osborne SL, Herreros J, Bastiaens PI and
Schiavo G: Calcium-dependent oligomerization of synaptotagmins I
and II. Synaptotagmins I and II are localized on the same synaptic
vesicle and heterodimerize in the presence of calcium. J Biol Chem.
274:59–66. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Andrews NW and Chakrabarti S: There's more
to life than neurotransmission: The regulation of exocytosis by
synaptotagmin VII. Trends Cell Biol. 15:626–631. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yoshihara M and Montana ES: The
synaptotagmins: Calcium sensors for vesicular trafficking.
Neuroscientist. 10:566–574. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Dlugosz A, Muschiol S, Zakikhany K, Assadi
G, D'Amato M and Lindberg G: Human enteroendocrine cell responses
to infection with Chlamydia trachomatis: A microarray study. Gut
Pathog. 6:242014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Divangahi M, Chen M, Gan H, Desjardins D,
Hickman TT, Lee DM, Fortune S, Behar SM and Remold HG:
Mycobacterium tuberculosis evades macrophage defenses by inhibiting
plasma membrane repair. Nat Immunol. 10:899–906. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Walker MG, Volkmuth W, Sprinzak E, Hodgson
D and Klingler T: Prediction of gene function by genome-scale
expression analysis: Prostate cancer-associated genes. Genome Res.
9:1198–1203. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Allen M, Bjerke M, Edlund H, Nelander S
and Westermark B: Origin of the U87MG glioma cell line: Good news
and bad news. Sci Transl Med. 8:354re32016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu H, Liang S, Yang X, Ji Z, Zhao W, Ye X
and Rui J: RNAi-mediated RPL34 knockdown suppresses the growth of
human gastric cancer cells. Oncol Rep. 34:2267–2272. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xiong J, Bing Z, Su Y, Deng D and Peng X:
An integrated mRNA and microRNA expression signature for
glioblastoma multiforme prognosis. PLoS One. 9:e984192014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tanaka T, Manome Y, Wen P, Kufe DW and
Fine HA: Viral vector-mediated transduction of a modified platelet
factor 4 cDNA inhibits angiogenesis and tumor growth. Nat Med.
3:437–442. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dupont F, Avalosse B, Karim A, Mine N,
Bosseler M, Maron A, van den Broeke AV, Ghanem GE, Burny A and
Zeicher M: Tumor-selective gene transduction and cell killing with
an oncotropic autonomous parvovirus-based vector. Gene Ther.
7:790–796. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kang CS, Pu PY, Li YH, Zhang ZY, Qiu MZ,
Huang Q and Wang GX: An in vitro study on the suppressive effect of
glioma cell growth induced by plasmid-based small interference RNA
(siRNA) targeting human epidermal growth factor receptor. J
Neurooncol. 74:267–273. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hui E, Bai J, Wang P, Sugimori M, Llinas
RR and Chapman ER: Three distinct kinetic groupings of the
synaptotagmin family: Candidate sensors for rapid and delayed
exocytosis. Proc Natl Acad Sci USA. 102:pp. 5210–5214. 2005;
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Maximov A and Südhof TC: Autonomous
function of synaptotagmin 1 in triggering synchronous release
independent of asynchronous release. Neuron. 48:547–554. 2005.
View Article : Google Scholar : PubMed/NCBI
|