Introduction
Acute pancreatitis is a severe disease with a high
risk of morbidity and mortality. The pathophysiology of acute
pancreatitis exhibits edema, perivascular infiltration, acinar
necrosis and hemorrhage in the pancreas (1). Inflammatory mediators are synthesized
and secreted by pancreatic cells, thus entering the blood
circulation, spreading to other organs. These inflammatory
mediators give rise to systemic inflammatory response syndrome
(SIRS), which is the leading cause of high mortality of the disease
(2,3).
Previous studies have not completely revealed the
molecular mechanisms of acute pancreatitis. Auto digestion of the
gland is widely accepted, however, the initiating factor, which
turns pro enzymes into their active forms, is still obscure.
Previous study has focused on investigating the initiators of this
systemic disease, including interleukin (IL)-1, interleukin-6
(IL-6) and tumor necrosis factor (TNF)-α. IL-1 and TNF-α, which are
the primary inducers of IL-6, are the primary components of the
inflammatory cytokine family. Understanding the pathway of the
activation of proinflammatory cytokines is the key to explain the
disease pathogenesis in the early phase.
In previous years, several signaling pathways have
been identified to serve a critical role in the process of acute
pancreatitis (4–6). Mitogen-activated protein kinases
(MAPKs) are known to be activated by numerous of stimuli, such as
cytokines, neurotransmitters, growth factors and many different
kinds of stress (7). NF-κB is a
key regulator of the immune system (8), presenting in the cytoplasm, p65 and
p50 are the most common heterodimer complexes of NF-κB, and p65 is
the functional component involving the activation of NF-κB.
However, their roles in regulation of cytokines like IL-1, IL-6 and
TNF-α in acute pancreatitis have not been completely
elucidated.
Melatonin (N-acetyl-5-methoxytryptamine) was
first isolated from the pineal gland successfully by Lerner et
al (9). It is primarily known
for its regulation of circadian and annual cycles (10). Subsequent research reveal melatonin
not only as a potent antioxidant, but also a non-enzymatic free
radical scavenger (11). Moreover,
melatonin serves an important role in protecting lipid membranes
against lipid peroxidation phenomenon (12).
Melatonin has been identified as a potential
molecule in the therapy of inflammatory diseases (13,14).
In the present study, the authors aimed to estimate its protective
effects and possible mechanisms of exogenous administration of
melatonin in the rat models of acute pancreatitis induced by
taurocholate.
Materials and methods
Animals
A total of 64 male Sprague-Dawley rats, weighing
200–250 g, were obtained from the Shanghai Laboratory Animal
Center, Chinese Academy of Sciences (Shanghai, China). They were
kept at constant room temperature of 25°C in a 12 h light-dark
cycle with access to standard rat pellets and water chow ad
libitum. All animals were acclimatized in the laboratory for at
least one week and were deprived of food for 12 h prior to the
experiment initiation, but water was allowed free access. All
procedures were performed in accordance with the Guidelines for
Animal Experiments of Wenzhou Medical University. All the animal
studies complied with current ethical considerations with the
approval of the Laboratory Animal Ethics Committee of Wenzhou
Medical University (Wenzhou, China).
Induction of severe acute
pancreatitis
All rats were anesthetized by administration of 4%
chloral hydrate (300 mg/kg; Beijing Solarbio Science and Technology
Co., Ltd., Beijing, China). The rats were randomly divided into the
sham operation group (SO, n=16), the severe acute pancreatitis
group (SAP, n=16), the melatonin treatment group (MLT, n=16) and
the SB203580 treatment group (SB, n=16). In the SAP, SB and MLT
groups, the biliopancreatic duct through the papilla was reached
and cannulated by penetrating with a 24-gauge catheter after a
micro clip clamping the hepatic duct. Acute pancreatitis was
induced by 5% taurocholate (Sigma-Aldrich; Merck KGaA, Darmstadt,
Germany) through retrograde infusion of a dose of 1 ml/kg via a
microinjection pump at a rate of 0.2 ml/min. Rats in the SO group
were underwent to the same surgery but without infusion. The MLT
group was treated with 50 mg/kg of melatonin (Sigma-Aldrich; Merck
KGaA) intraperitoneally 30 min prior to operation. The SB group
received 0.5 mg/kg SB203580 via caudal vein 30 min before
operation. Following each operation, the abdomen was closed in two
layers. All procedures were conducted using sterile techniques.
Sample collection and detection of
serum amylase and ELISA
At 6 and 12 h following SAP induction, rats were
anesthetized with 4% chloral hydrate (300 mg/kg body weight).
Abdominal aorta puncture was used to obtain 6 ml blood from each
animal, 2 ml of each sample was collected and stored in tubes
without anticoagulants (Generay Biotechnology Co., Ltd., Shanghai,
China). Blood samples (1 in 2 ml) was centrifuged at 1,200 × g for
5 min, the serum was collected for determination of amylase
activity with a fully automatic biochemical analyzer (Hitachi
Corporation, Tokyo, Japan). The other 1 ml of each sample was
centrifuged at 3,000 × g for 15 min, and the serum undiluted was
for the ELISA assay. The remaining 4 ml blood was treated with
sodium heparin anticoagulant and used to isolate peripheral blood
mononuclear cells (PBMCs). The pancreatic tissues were harvested
immediately, then placed in 40 g/l paraformaldehyde and prepared
for routine paraffin embedding prior to pathological examination.
The rats were anesthetized with 4% chloral hydrate (300 mg/kg body
weight) and subsequently euthanized by exsanguination after the
experiments.
Histological analysis and pathological
scores of pancreatic tissues
Samples of pancreatic tissues were fixed in 40 g/l
paraformaldehyde, dehydrated embedded in paraffin. Paraffin
sections were cut at 4 µm-thick sections. Pancreas sections were
stained with hematoxylin and eosin, and observed using light
microscopy (Nikon Corporation, Tokyo, Japan). Using the standards
set by Schmidt et al (1),
tissues were examined by two experienced histologists blinded to
the experimental protocol. The pancreatic sections, presenting a
minimum of six fields, were examined for each sample and scored on
a scale of 0–3 (0 being normal and 3 being severe) on the basis of
edema, inflammatory cell infiltration, acinar cell degeneration and
parenchymal hemorrhage.
Isolation of PBMCs
PBMC isolation was performed with density gradient
centrifugation. The blood sample (4 ml) was diluted with an equal
volume of PBS. Then, the diluted blood was carefully laid over 4 ml
ficoll (Beijing Solarbio Science and Technology Co., Ltd.). The
PBMCs layered on the white band at the medium interface after
centrifugation, were carefully removed into a new tube. The
harvested PBMCs were diluted in PBS and the cells were pelleted by
another centrifugation. Then PBMCs were immediately stored in
liquid nitrogen until use.
ELISA
ELISAs for TNF-α, IL-1 and IL-6 (R&D Systems
Inc., Minneapolis, MN, USA) were carried out in duplicate with 40
µl of samples in each well of 96-well plates (Nunc, Roskilde,
Denmark), which had been coated with 100 µl aliquots of either
anti-rat IL-1 (cat. no. MAB501), IL-6 (cat. no. MAB506) or TNF-α
(cat. no. MAB510; all 1:500; all from R&D Systems Inc.,
Minneapolis, MN, USA) antibodies in PBS (at pH 7.4) at 4°C
overnight. The plates were washed in PBS containing 0.05% Tween-20
and blocked with PBS containing 10% fetal bovine serum for 2 h.
Following additional washes, the standards and the serum were added
to the plates and incubated at room temperature for 3 h. Following
incubation, the wells were washed and a goat anti-mouse secondary
IgG HRP conjugated secondary fluorescent antibody [cat. no. GAM007,
1:5,000; Hangzhou MultiSciences (Lianke) Biotech, Co., Ltd.,
Hangzhou, China] was added to each well. Incubation continued at
room temperature for 1 h. Following washing the wells again, the
TMB substrate [Hangzhou MultiSciences (Lianke) Biotech, Co., Ltd.]
was added. Each were had sulfuric acid (2 mol/l) added to terminate
the reaction. Color development was measured with an automated
microplate ELISA reader at 450 nm. Standard curves were obtained
for each sample through serial dilutions of recombinant IL-1, IL-6
or TNF-α.
Western blot analysis
Western blotting was used to measure the protein
expression of β-actin, phospho-specific p38 and p65, total p38 and
p65. PBMCs were completely lysed in radioimmunoprecipitation assay
buffer supplemented with protease and phosphatase inhibitors (Roche
Diagnostics GmbH, Basel, Switzerland) at a cell density of
1×107 cells/100 ml. Protein concentration was determined
by the bicinchoninic acid assay method (BCA kit; Beyotime Institute
of Biotechnology, Haimen, China). From each sample, 50 mg of total
protein separated by 12% SDS-PAGE and the resolved proteins were
transferred to a polyvinylidene difluoride membrane (EMD Millipore,
Billerica, MA, USA). After being blocked with 5% BSA in TBS-0.1%
Tween-20 (TBST) for 2 h at room temperature, membranes were
incubated overnight at 4°C with specific primary antibodies. The
following antibodies were used: p-p38 (Thr180/Tyr185 Rabbit mAb,
cat. no. 9215); p38 (D13E1 Rabbit mAb, cat. no. 8690); p-p65
(Ser536 rabbit mAb, cat. no. 3033); p65 (D14E12 rabbit mAb, cat.
no. 8242); β-actin (13E5 rabbit mAb, cat. no. 4970; all 1:1,000;
all from Cell Signaling Technology, Inc., Danvers, MA, USA). Then
the membranes were washed three times for 10 min each with TBST,
then incubated with a goat anti-rabbit secondary IgG HRP conjugated
anti-rabbit secondary fluorescent antibody [cat. no. GAR0072,
1:1,000; Hangzhou MultiSciences (Lianke) Biotech, Co., Ltd.] for 1
h at room temperature. After another 3 times washing with TBST, the
enhanced chemiluminescence method (WesternBright Enhanced
Chemiluminescence Substrate kit; Advansta Inc., Menlo Park, CA,
USA) was used as recommended by the manufacturer to measure the
quantity of protein. Protein expression levels were normalized
against β-actin. In western blotting, the authors randomly selected
three samples in each group to repeat the experiment in triplicate.
Images of the blots were captured using the ChemiDoc MP system and
the density of specific bands was quantified using Image Lab
software version 4.1 (both from Bio-Rad Laboratories, Inc.,
Hercules, CA, USA).
Statistical analysis
Statistical analyses were carried out using the SPSS
software (version 15.0; SPSS Inc., Chicago, IL, USA). All data are
expressed as means ± standard deviation. One-way analysis of
variance followed by Newmann-Keuls post hoc test was used to
identify differences between means. P<0.05 was considered to
indicate a statistically significant difference.
Results
Effect of melatonin on serum
amylase
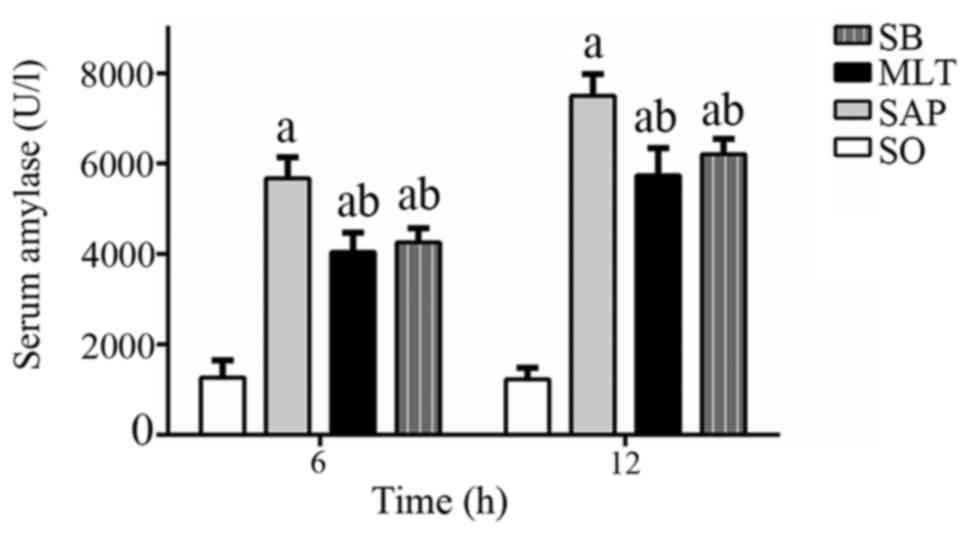
Serum amylase levels were substantially elevated
following the induction of pancreatitis, which further confirmed
the diagnosis of pancreatitis. The levels of serum amylase in SAP
groups were significantly increased at 6 and 12 h compared with SO
groups (P<0.01). Pretreatment of pancreatitis rats with 50 mg/kg
melatonin (MLT groups) suppressed the amylase elevation at both 6
and 12 h compared with SAP groups (Fig. 1). SB groups (0.5 mg/kg) either
declined the levels of serum amylase vs. SAP groups at both
time-points. Though the levels were decreased in both MLT and SB
groups, but were still higher compared to SO groups at both
time-points.
Effect of melatonin on pancreatic
histopathology
Normal tissue morphology of pancreas was observed in
SO groups. Edema, parenchyma hemorrhage and inflammatory
infiltration of neutrophils in pancreatic tissues were observed in
SAP group at 6 h and the changes became more severe as time
prolonged. While more mild edema, parenchyma hemorrhage and
inflammatory infiltration of neutrophils into the pancreatic
tissues were identified in MLT groups and SB groups (Fig. 2A), the pathological scores of
pancreas tissues declined considerably in both groups (Fig. 2B). The mean pathological scores for
pancreatic tissues in the MLT group were significantly higher
(P<0.01) than those for samples in the SO group (5.44±0.50 vs.
0.31±0.46 following 6 h; 8.31±0.46 vs. 0.75±0.54 following 12 h),
but were significantly lower (P<0.01) than those for samples in
the SAP group at each time-point (5.44±0.50 vs. 7.31±0.70 following
6 h; 8.31±0.46 vs. 11.13±0.58 following 12 h).
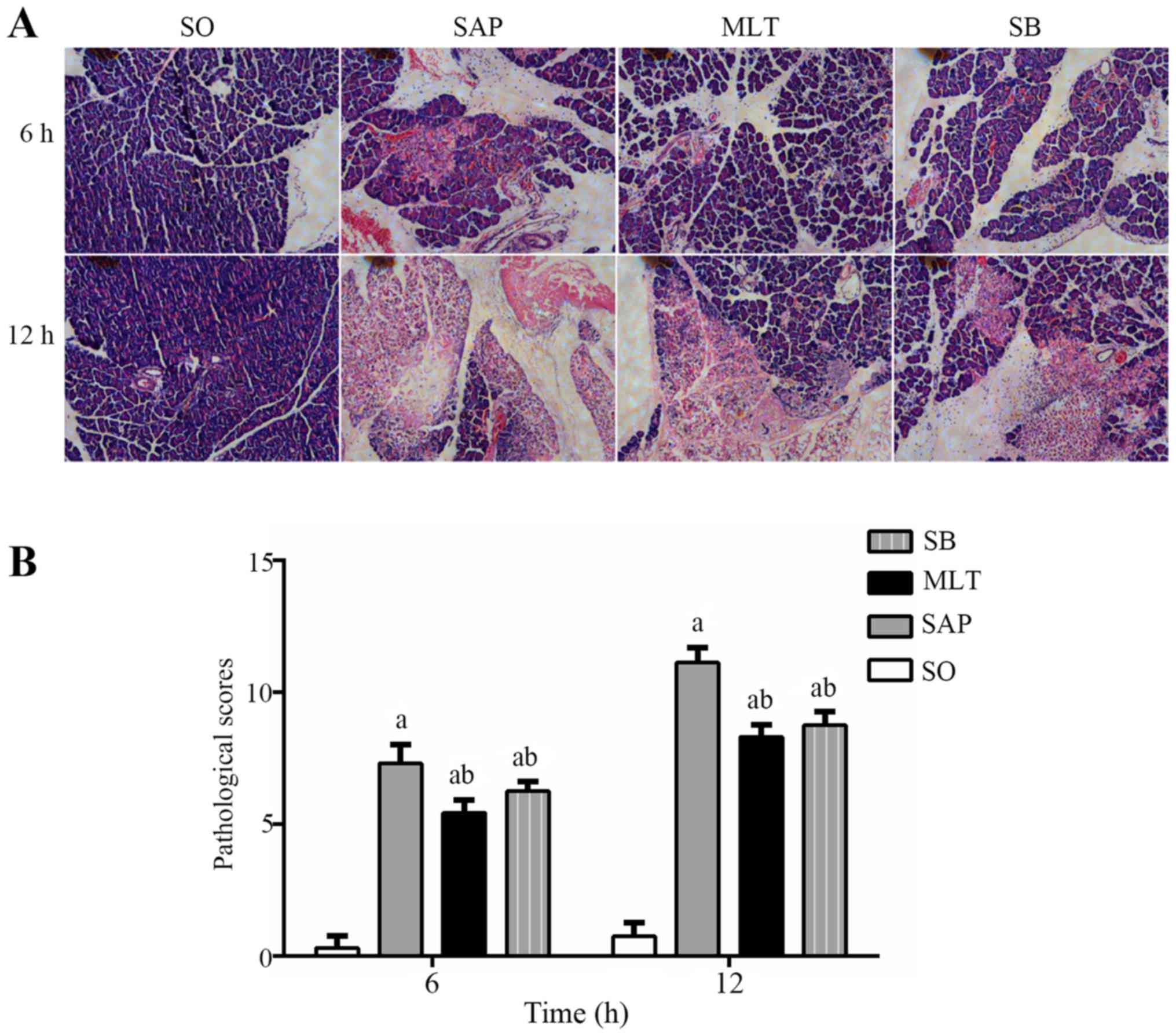 | Figure 2.Protective effect of melatonin on
pancreatic morphology. (A) SO groups present the normal structures
of pancreas tissues at 6 and 12 h using hematoxylin and eosin
staining magnification, ×400. Pancreatic edema, parenchyma
hemorrhage and neutrophil infiltration were observed in SAP groups
at 6 h, and became worse at 12 h (magnification, ×400). The MLT and
SB groups had more mild edema, parenchyma hemorrhage and neutrophil
infiltration (magnification, ×400). (B) The pathological scores of
pancreas tissues. Data are presented as mean ± standard deviation
(n=6). aP<0.01 vs. SO group; bP<0.01
vs. SAP group. SO, sham operation group; SAP, severe acute
pancreatitis group; MLT, melatonin treatment group; SB, SB203580
treatment group. |
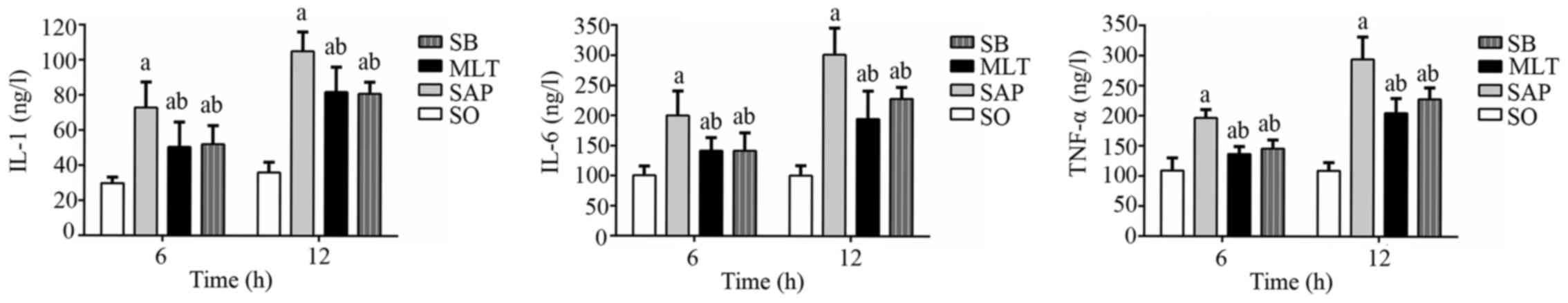
Effect of melatonin on IL-1, IL-6 and
TNF-α production
Serum IL-1, IL-6 and TNF-α levels all increased in
the SAP group at 6 h compared with the SO group and did not have a
significant difference as time prolonged. Compared to the SAP
group, both MLT and SB groups had significantly lower levels of
IL-1, IL-6 and TNF-α at both 6 and 12 h, nonetheless, were still
higher at both time-points compared with SO group (Fig. 3).
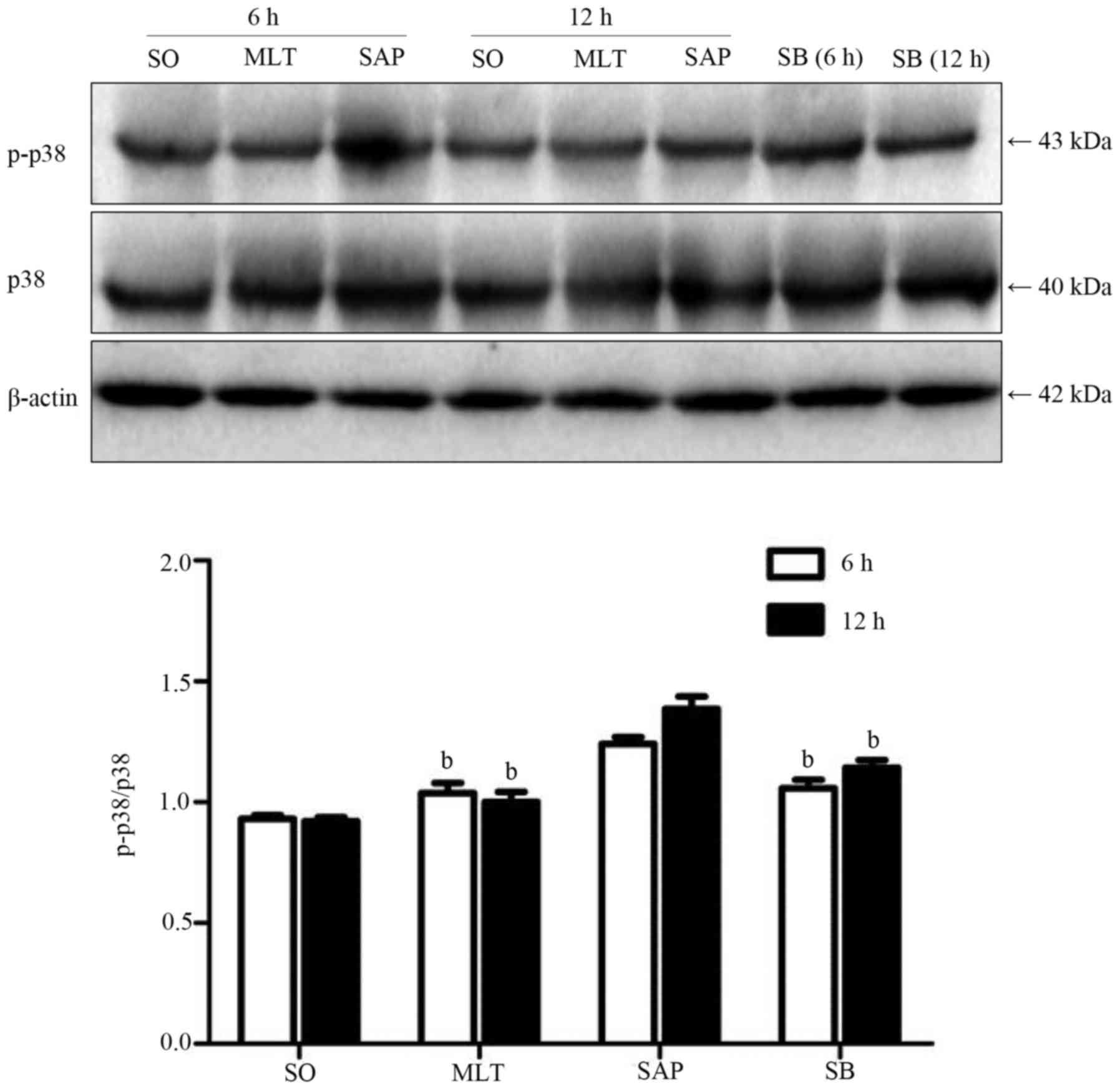
Effect of melatonin on activation of
p38 in PBMCs
Phosphorylation of p38 was observed following the
induction of acute pancreatitis. However, both melatonin and the
p38 inhibitor had a significant suppression on the activation of
p38 MAP kinase when compared with SAP group at 6 and 12 h, the
phosphorylation of p38 was remarkably subdued in MLT and SB groups
at both time-points (Fig. 4).
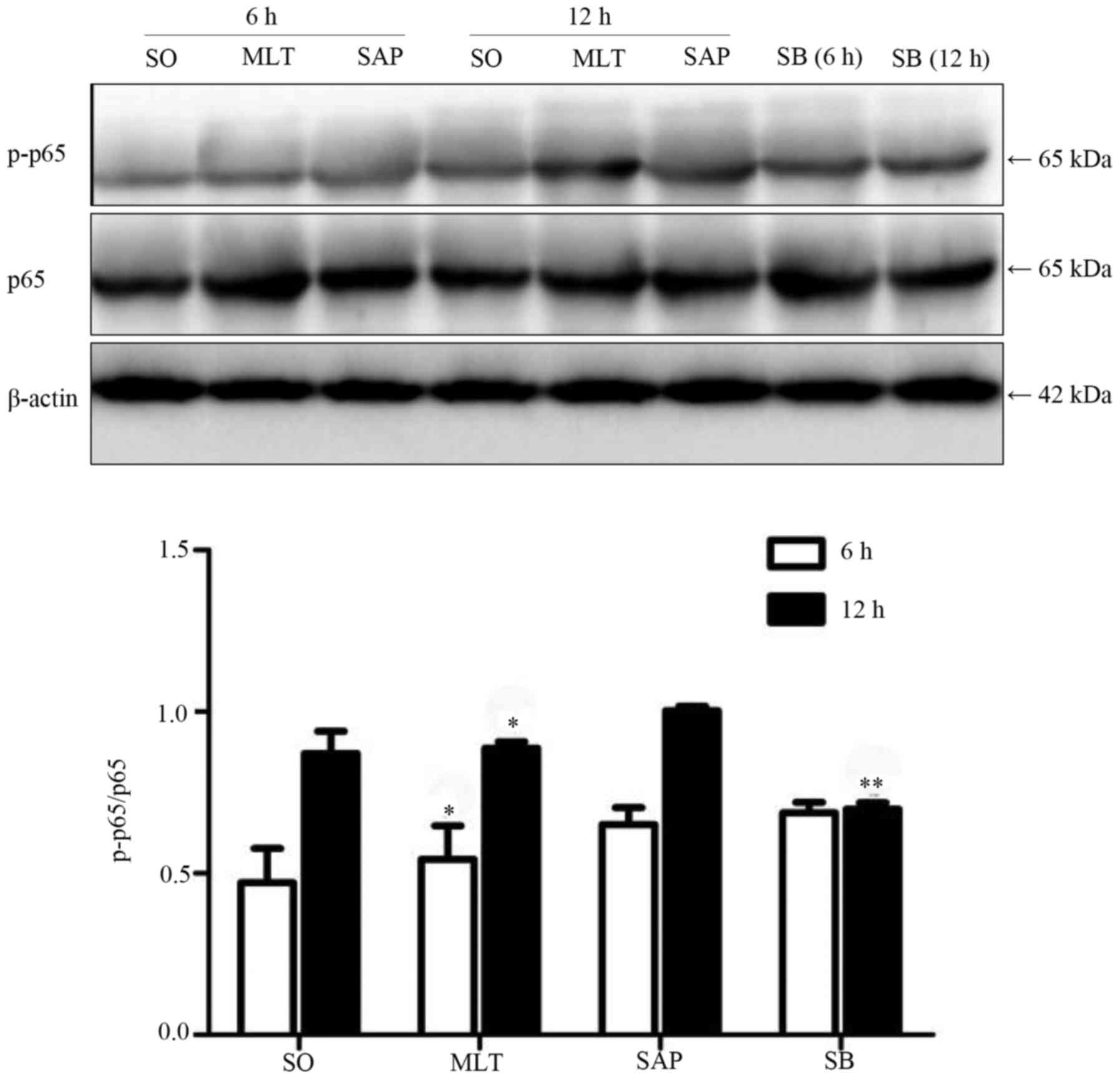
Effect of melatonin on NF-κB in
PBMCs
Phosphorylation of p65 was found after the induction
of acute pancreatitis. Melatonin had significantly subdued the
phosphorylation of p65 when compared with the SAP group at both
time-points. Pretreatment with the p38 inhibitor suppressed the
expression of phosphorylated p65 at 12 h, but not at 6 h (Fig. 5).
Discussion
In the present study, the authors have investigated
the effect of melatonin on suppressing the development of
taurocholate-induced AP. Administration of exogenous melatonin
significantly inhibited serum amylase production, IL-1, IL-6 or
TNF-α levels. Furthermore, melatonin inhibited the activation of
p38 and NF-κB. Jung et al (15,16)
indicated that the role of NF-κB suppression in response to
melatonin treatment. But they primarily researched oxidative liver
injury and pancreas samples in AP. In the current study, the
authors have discussed NF-κB in peripheral blood mononuclear cells
(PBMCs) in SAP.
Pancreatic digestive enzymes including amylase and
lipase contribute to necrosis of acinar cells at an early stage,
and consequently, lead to the inflammation of the pancreas.
Nevertheless, cytokines and oxidative stress may lead to the major
factors for development of SIRS (17). Therefore, blockade of IL-1, IL-6
and TNF-α receptors soon after induction of pancreatitis are
associated with decreased production of inflammatory cytokines and
alleviated pathological changes (18). Previously, accumulating evidences
suggested that melatonin suppresses inflammatory cytokines
expression such as IL-1, IL-6 and TNF-α in rats with
ventilator-induced lung injury and azoxymethane/dextran sodium
sulfate-induced large bowel oncogenesis (19,20).
In an experimental model of pancreatitis, pretreatment with
melatonin prevented the increased levels of amylase in serum
(Fig. 1) and the elevations of
IL-1, IL-6 and TNF-α were inhibited (Fig. 3), suggesting that melatonin may be
effective in controlling excessive exocrine enzyme secretion and
inflammatory cytokines during severe acute pancreatitis.
The MAPKs are a large family of serine/threonine
kinases, regulating kinases (ERKs), c-Jun N-terminal kinases (JNKs)
and p38 MAPKs involved in cell proliferation, differentiation and
death (21). NF-κB is a
transcription factor, which serves a critical role in immunity,
inflammation, cell proliferation and cell death. Activation of
NF-κB target genes requires phosphorylation of NF-κB proteins; p65
is the functional component involving the activation of NF-κB
(22). Oxidative stress and
proinflammatory cytokines trigger common signal transduction
pathways that result in an inflammatory cascade, especially through
activation of MAPKs, as well as NF-κB (23). The potential contaminating source
to induce cellular cytokine productions may serve an important role
in the activation of p38 MAPK and NF-κB in various pathological
conditions, such as acute pancreatitis (24). In addition, it has been also
reported that the SB203580, a selective p38 MAPK inhibitor,
virtually prevents cerulein-induced cell death and generation of
inflammatory cytokines, suggesting that p38 MAPK may have
detrimental role in cerulein-induced AP (25). Kim et al and Bae et
al (26,27) reported that the inhibition of p38
MAPK and NF-κB may inhibit IL-1, IL-6 and TNF-α productions.
Meanwhile, melatonin has been reported to inhibit p38 MAPK and
NF-κB in various animal models (28–30).
In the current study, the models of taurocholate-induced AP
exhibited increased serum amylases, IL-1, IL-6 and TNF-α levels,
yet p38 MAPK and NF-κB activation were detected in the PBMCs
(Figs. 4 and 5). Therefore, the authors concluded that
the levels of inflammatory cytokines in taurocholate-induced SAP
were inhibited by suppression of p38 MAPK and NF-κB activation in
PBMCs.
In conclusion, the present study demonstrated that
melatonin attenuates the severity of taurocholate-induced AP may
through suppressing pro-inflammatory cytokine production by
deactivating p38 MAPK and NF-κB in PBMCs. Therefore, melatonin
exerts potent anti-inflammatory effects in AP.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Science and
Technology Bureau of Wenzhou, Zhejiang Province, China (grant nos.
2014S0192 and Y20150158), and The Public Projects of Zhejiang
Province (grant no. 2016C33215).
Availability of data and materials
The analyzed data sets generated during the study
are available from the corresponding author on reasonable
request.
Authors' contributions
YC performed the experiments and wrote the
manuscript; JW designed the study; QZ and QC analyzed the data; YZ
and BS collected the data; and YJ advised on assay performance. All
authors evaluated and approved the final manuscript.
Ethics approval and consent to
participate
All procedures were performed in accordance with the
Guidelines for Animal Experiments of Wenzhou Medical University.
All the animal studies complied with current ethical considerations
with the approval of the Laboratory Animal Ethics Committee of
Wenzhou Medical University (Wenzhou, China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Schmidt J, Rattner DW, Lewandrowski K,
Compton CC, Mandavilli U, Knoefel WT and Warshaw AL: A better model
of acute pancreatitis for evaluating therapy. Ann Surg. 215:44–56.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Norman J: The role of cytokines in the
pathogenesis of acute pancreatitis. Am J Surg. 175:76–83. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Büchler MW, Gloor B, Müller CA, Friess H,
Seiler CA and Uhl W: Acute necrotizing pancreatitis: Treatment
strategy according to the status of infection. Ann Surg.
232:619–626. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Xiao WQ, Yin GJ, Fan YT, Qiu L, Cang XF,
Yu G, Hu YL, Xing M, Wu DQ, Wang XP, et al: Catalpol ameliorates
sodium taurocholate-induced acute pancreatitis in rats via
inhibiting activation of nuclear factor kappa B. Int J Mol Sci.
15:11957–11972. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wei S, Huang Q, Li J, Liu Z, You H, Chen Y
and Gong J: Taurine attenuates liver injury by downregulating
phosphorylated p38 MAPK of Kupffer cells in rats with severe acute
pancreatitis. Inflammation. 35:690–701. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu HS, Pan CE, Liu QG, Yang W and Liu XM:
Effect of NF-kappaB and p38 MAPK in activated monocytes/macrophages
on pro-inflammatory cytokines of rats with acute pancreatitis.
World J Gastroenterol. 9:2513–2518. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pagès G, Lenormand P, L'Allemain G,
Chambard JC, Meloche S and Pouysségur J: Mitogen-activated protein
kinases p42mapk and p44mapk are required for fibroblast
proliferation. Proc Natl Acad Sci USA. 90:pp. 8319–8323. 1993;
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li Q and Verma IM: NF-kappaB regulation in
the immune system. Nat Rev Immunol. 2:725–734. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lerner AB, Case JD and Takahashi Y:
Isolation of melatonin and 5-methoxyindole-3-acetic acid from
bovine pineal glands. J Biol Chem. 235:1992–1997. 1960.PubMed/NCBI
|
|
10
|
Berger J: A two-clock model of circadian
timing in the immune system of mammals. Pathol Biol (Paris).
56:286–291. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Galano A, Tan DX and Reiter RJ: Melatonin
as a natural ally against oxidative stress: A physicochemical
examination. J Pineal Res. 51:1–16. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
García JJ, Reiter RJ, Pié J, Ortiz GG,
Cabrera J, Sáinz RM and Acuña-Castroviejo D: Role of pinoline and
melatonin in stabilizing hepatic microsomal membranes against
oxidative stress. J Bioenerg Biomembr. 31:609–616. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen CF, Wang D, Reiter RJ and Yeh DY:
Oral melatonin attenuates lung inflammation and airway
hyperreactivity induced by inhalation of aerosolized pancreatic
fluid in rats. J Pineal Res. 50:46–53. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Motilva V, García-Mauriño S, Talero E and
Illanes M: New paradigms in chronic intestinal inflammation and
colon cancer: Role of melatonin. J Pineal Res. 51:44–60. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jung KH, Hong SW, Zheng HM, Lee DH and
Hong SS: Melatonin downregulates nuclear erythroid 2-related factor
2 and nuclear factor-kappaB during prevention of oxidative liver
injury in a dimethylnitrosamine model. J Pineal Res. 47:173–183.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jung KH, Hong SW, Zheng HM, Lee HS, Lee H,
Lee DH, Lee SY and Hong SS: Melatonin ameliorates cerulein-induced
pancreatitis by the modulation of nuclear erythroid 2-related
factor 2 and nuclear factor-kappaB in rats. J Pineal Res.
48:239–250. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pereda J, Sabater L, Aparisi L, Escobar J,
Sandoval J, Viña J, López-Rodas G and Sastre J: Interaction between
cytokines and oxidative stress in acute pancreatitis. Curr Med
Chem. 13:2775–2787. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xue DB, Zhang WH, Yun XG, Song C, Zheng B,
Shi XY and Wang HY: Regulating effects of arsenic trioxide on cell
death pathways and inflammatory reactions of pancreatic acinar
cells in rats. Chin Med J (Engl). 120:690–695. 2007.PubMed/NCBI
|
|
19
|
Pedreira PR, García-Prieto E, Parra D,
Astudillo A, Diaz E, Taboada F and Albaiceta GM: Effects of
melatonin in an experimental model of ventilator-induced lung
injury. Am J Physiol Lung Cell Mol Physiol. 295:L820–L827. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tanaka T, Yasui Y, Tanaka M, Tanaka T,
Oyama T and Rahman KM: Melatonin suppresses AOM/DSS-induced large
bowel oncogenesis in rats. Chem Biol Interact. 177:128–136. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Torres M and Forman HJ: Redox signaling
and the MAP kinase pathways. Biofactors. 17:287–296. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Viatour P, Merville MP, Bours V and
Chariot A: Phosphorylation of NF-kappaB and IkappaB proteins:
Implications in cancer and inflammation. Trends Biochem Sci.
30:43–52. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang X, Martindale JL, Liu Y and Holbrook
NJ: The cellular response to oxidative stress: Influences of
mitogen-activated protein kinase signalling pathways on cell
survival. Biochem J. 333:291–300. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Blinman TA, Gukovsky I, Mouria M,
Zaninovic V, Livingston E, Pandol SJ and Gukovskaya AS: Activation
of pancreatic acinar cells on isolation from tissue: Cytokine
upregulation via p38 MAP kinase. Am J Physiol Cell Physiol.
279:C1993–C2003. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Choi SB, Bae GS, Jo IJ, Seo SH, Kim DG,
Shin JY, Hong SH, Choi BM, Park SH, Song HJ and Park SJ: Protective
effects of lithospermum erythrorhizon against cerulein-induced
acute pancreatitis. Pancreas. 44:31–40. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kim TH, Bae GS, Oh HJ, Kim MS, Park KC,
Koo BS, Kim BJ, Yang YS, Park DE, Lee JH, et al:
2′,4′,6′-Tris(methoxymethoxy) chalcone (TMMC) attenuates the
severity of cerulein-induced acute pancreatitis and associated lung
injury. Am J Physiol Gastrointest Liver Physiol. 301:G694–G706.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bae GS, Kim MS, Jeong J, Lee HY, Park KC,
Koo BS, Kim BJ, Kim TH, Lee SH, Hwang SY, et al: Piperine
ameliorates the severity of cerulein-induced acute pancreatitis by
inhibiting the activation of mitogen activated protein kinases.
Biochem Biophys Res Commun. 410:382–388. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang S, Li W, Gao Q and Wei T: Effect of
melatonin on the generation of nitric oxide in murine macrophages.
Eur J Pharmacol. 501:25–30. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Aparicio-Soto M, Alarcón-de-la-Lastra C,
Cárdeno A, Sánchez-Fidalgo S and Sanchez-Hidalgo M: Melatonin
modulates microsomal PGE synthase 1 and NF-E2-related
factor-2-regulated antioxidant enzyme expression in LPS-induced
murine peritoneal macrophages. Br J Pharmacol. 171:134–144. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jung KH, Hong SW, Zheng HM, Lee HS, Lee H,
Lee DH, Lee SY and Hong SS: Melatonin ameliorates cerulein-induced
pancreatitis by the modulation of nuclear erythroid 2-related
factor 2 and nuclear factor-kappaB in rats. J Pineal Res.
48:239–250. 2010. View Article : Google Scholar : PubMed/NCBI
|