Introduction
Aspergillus fumigatus (A fumigatus) is an
environmentally ubiquitous, spore-forming mould saprophyte that
causes disease in individuals with poor immunity (1). The incidence of invasive fungal
infections is increased in patients receiving bone marrow and organ
transplantation, chemotherapy for cancer, or treatment with
glucocorticoids or broad-spectrum use of antibiotics (2). Aspergillosis is the second most
common type of fungal infection following candidiasis (3). The use of antifungal agents improves
the prognosis of patients to a certain extent; however, the
mortality rate of invasive aspergillosis remains high due to the
limited efficacy of currently available drugs (4). Infection-mediated immune injury is an
important cause of death (5).
Immunological diagnosis, prevention and reconstitution are crucial
to improving the prognosis of patient recovery (6). Improved understanding of the
regulatory mechanisms underlying the host immune response following
invasive aspergillosis may aid in improving the prognosis of
patients.
Alveolar macrophages serve an important role in
innate immunity in the lung tissue, as they exhibit the capacity to
identify, process and present antigens, inducing specific immune
responses, and are known as an outpost of natural immunity
(7). They also serve a role in
immune homeostasis via negative feedback regulation of the local
microenvironment, preventing excessive immune damage and
maintaining the stability of the internal environment (8). A previous study reported that A.
fumigatus enhanced the expression of dectin-2 and cluster of
differentiation (CD)206 in stimulated macrophages, indicating that
the fungus induces a certain regulatory effect on the phenotype and
function of macrophages (9).
Together, natural killer cells (NK cells) and
macrophages build the first immune defence line in the body for the
removal of invasive microbes (10,11).
The activation or inhibition of NK cells is mainly regulated by the
expression of activating or inhibitory receptors on the cell
surface (12). When NK cells are activated, a large number of
cytokines and chemokines are secreted, such as interferon-γ (IFN-γ)
and tumor necrosis factor-α (TNF-α), which regulate the immune
state of the host (13). NK cells have a direct killing
effect on invasive pathogens, including bacteria, parasites and
yeast (14–16). The effects of NK cells and IFN-γ on
A. fumigatus were first reported in mice (17). The enrichment of NK cells was an
important mechanism to prevent further infection of A.
fumigatus in a mouse model of neutropenia. Additionally,
clinical studies have also demonstrated that IFN-γ serves an
important role in inhibiting A. fumigatus (18).
The interaction between macrophages and NK cells has
attracted widespread attention, particularly in the study of tumor
occurrence and development, and microbial invasion and infection.
Macrophages and NK cells infiltrate numerous infected or cancerous
tissues (19).
Monocytes/macrophages serve an important role in immune
surveillance and immunoregulation depending on their functions in
phagocytosis and antigen presentation (20). Peripheral monocytes frequently
differentiate into different subtypes of macrophages depending on
the tissue microenvironment (21).
The Type 1 T helper (Th1) cytokine IFN-γ and lipopolysaccharide
(LPS), the ligand of Toll-like receptor 4, polarize monocytes
towards a classically activated (M1) macrophage phenotype; M1
macrophages produce proinflammatory cytokines, such as TNF-α and
interleukin (IL)-12, subsequently facilitating the clearance of
pathogens (22). Conversely,
following exposure to Type 2 T helper cytokines, such as IL-4 and
IL-13, monocytes differentiate into alternatively activated
macrophages (M2 macrophages) inducing the production of
anti-inflammatory mediators, including IL-10, which promote
anti-inflammation and wound healing (23). Macrophages are affected by
phagocytic antigens and the immune microenvironment, express
specific receptors or ligands, and secrete large quantities of
cytokines and chemokines that regulate NK cell function (24).
Galectin-9, a β-galactoside binding lectin, is
present in various tissues and is particularly abundant in the
liver (25). It is a type of
eosinophil chemoattractant and is a member of the galactose
agglutinin family. Galectin-9 possesses a variety of biological
functions, including contributions to cell differentiation,
maturation, adhesion, chemotaxis and apoptosis. It also serves
important roles in chronic inflammation, acute inflammation,
allergy and autoimmune diseases (26). A recent study reported that
macrophages regulated the function of NK cells by secreting
Galectin-9 following hepatitis C infection (27).
The present study investigated the regulatory
effects of Galectin-9 on the interaction between macrophages and NK
cells in aspergillosis. It suggested novel directions for further
study into the mechanisms underlying the roles of macrophages and
NK cells in inflammatory infection caused by A. fumigatus
and potential strategies to control the progression of
inflammation.
Materials and methods
Strains of A. fumigatus, activation of
spores and culture conditions
The A. fumigatus strain was obtained from the
Medical Mycology Research Center of Chiba University. A.
fumigatus resting conidia were cultivated for 3 days on beer
mash plates at 28°C. The conidia were detached from the plate using
endotoxin-free sterile water and filtered through a cell strainer
with a 40-µm nylon mesh pore membrane to obtain a single-fungal
cell suspension. The swelling and synchronization of fungal growth
was achieved by cultivating the conidia in SDA culture medium (cat.
no. HB0235-10; Qingdao Hope Bio-Technology Co., Ltd.) at room
temperature under continuous agitation at 200 RPM overnight. The
spore morphology was imaged by an Olympus microscope
(magnification, ×200; CKX41-A32PH; Olympus Corporation). The plates
and dishes were obtained from Corning Inc.
Acquisition of macrophages
Peripheral blood mononuclear cells (PBMCs) were
freshly isolated from the peripheral blood of 3 healthy individuals
at the Changchun Blood Center between May 2017 and September 2017.
The 3 donors were all male (24–32 years old). The donors were all
negative for hepatitis B, hepatitis C and human immunodeficiency
virus infection. PBMCs were isolated via Ficoll density gradient
separation at 500 × g for 30 min at room temperature. The study was
approved by the Ethics Committee of Changchun Blood Center. All
experiments were conducted in accordance with the approved
guidelines and regulations (24).
Written informed consent for the use of haemocytes in research was
obtained from all participants. Monocytes were then purified by
magnetic-activated cell sorting with CD14+ microbeads
(Miltenyi Biotec, Inc.) with 3×3 ml of buffer flow through the
column (Miltenyi Biotec, Inc.) under the action of gravity. The
purity of the cells was ≥95% as determined by flow cytometry.
Monocytes were seeded in 96-well plates at a density of
0.2×106 cells/well in full RPMI-1640 medium (Thermo
Fisher Scientific, Inc.), with 1% penicillin and streptomycin and
10% FBS (all from Gibco; Thermo Fisher Scientific, Inc.). Then, 400
IU/ml granulocyte-macrophage colony-stimulating factor (GM-CSF;
cat. no. 300-03-100; PeproTech, Inc.) was added to the culture
medium, and the cells were grown at 37°C in a 5% CO2
incubator for 5 days of culture.
Macrophage stimulation experiment
Macrophages were collected following digestion by
0.2% EDTA at 37°C for 5 min (cat. no. 02-032-1ACS; Biological
Industries). The cells were reseeded into 12-well plates at a
density of 0.2×106 cells/well following three washes
with warm PBS buffer (cat. no. 02-024-1ACS; Biological Industries).
Swelling conidia were added to the macrophage culture plates, with
a ratio of macrophages to conidia of 1:5. Treatment with full
RPMI-1640 medium (as above) was used as the control group. Cells
and culture supernatant were collected for subsequent experiments
following co-culture at 37°C for 24 h.
NK cell stimulation experiment
PBMCs were freshly isolated from the peripheral
blood of healthy individuals as aforementioned. NK cells were then
purified by MACS using an NK Cell Isolation kit (cat. no.
130-092-657; Miltenyi Biotec, Inc.) according to the manufacturer's
protocols and were seeded into 12-well plates at a density of
0.2×106 cells/well. Swelling conidia were added to NK
cell culture plates, with a ratio of NK cells to conidia of 1:5.
Treatment with full RPMI-1640 medium was used as the control group.
Cells and culture supernatant were collected for subsequent
experiments following co-culture at 37°C for 24 h.
Macrophage and NK cell co-culture
Macrophages were collected following digestion by
0.2% EDTA at 37°C for 5 min and were reseeded into 12-well plates
at a density of 0.2×106 cells/well following three
washes with warm PBS buffer. An equal quantity of autologous
purified NK cells was added to the plates, which were then were
mixed softly. Swelling conidia were added to the macrophage/NK cell
co-culture plates, with a ratio of macrophages to conidia of 1:5.
To investigate the contact dependence of the interaction,
macrophages and NK cells were separated by a membrane (0.4-µm pore
size) in Transwell plates (cat. no. CLS3464-48EA; Costar; Corning
Inc.). The swelling conidia were able to contact the macrophages
but not the NK cells. Macrophages with conidia or NK cells with
conidia were used as controls. The cells and culture supernatant
were collected for subsequent experiments following co-culture at
37°C for 24 h. In the antibody neutralization test, macrophages
were stimulated by A. fumigatus conidia (1:5) for 24 h and
then co-cultured with the NK cells from the same individual for a
further 12 h, in the presence or absence of Galectin-9 (cat. no.
AF2045; R&D Systems, Inc.)-, IL-18 (cat. no. D044-3; R&D
Systems, Inc.)- or TNF-α (cat. no. AF-410-NA; R&D Systems,
Inc.)-specific antibodies when NK cells were added. The antibody
working concentrations were all 10 µg/ml.
Enzyme-linked immunosorbent assay
(ELISA)
Cell culture supernatants were collected following
24 h of incubation in each experimental condition. The
concentrations of TNF-α (cat. no. BMS223-4; Invitrogen; Thermo
Fisher Scientific, Inc.), IL-18 (cat. no. BMS267-2; Invitrogen;
Thermo Fisher Scientific, Inc.), Galectin-9 (cat. no. DGAL90;
R&D Systems, Inc.), IL-12 (cat. no. BMS238; Invitrogen; Thermo
Fisher Scientific, Inc.) and IL-10 (cat. no. BMS215-2; Invitrogen;
Thermo Fisher Scientific, Inc.) were measured by ELISA. The ELISA
experiments were performed according to the manufacturer's
protocols. The absorbance was detected using a Synergy™ H1 Full
Function Enzyme Marker (BioTek Instruments, Inc.).
Reverse transcription-quantitative PCR
(RT-qPCR)
RT-qPCR analysis was performed using standard
procedures. Total RNA was isolated using a RNeasy kit (Qiagen,
Inc.), 5 µg of each sample was reverse-transcribed using the M-MLV
first-stand synthesis system (Invitrogen; Thermo Fisher Scientific,
Inc.) and cDNAs were analyzed in triplicate with the MJ Real-Time
PCR System (Bio-Rad Laboratories, Inc.). The sequences of the
gene-specific and GAPDH primers used for qPCR are presented in
Table I. Amplification was
performed for 40 cycles with a denaturation temperature of 94°C for
30 sec, annealing temperature of 58°C for 30 sec and extension
temperature of 74°C for 45 sec in a Veriti thermal cycler (Applied
Biosystems; Thermo Fisher Scientific, Inc.). The PCR products were
200 bp in length. RT-qPCR was performed using the Power SYBR Green
Master Mix (Takara Bio, Inc.) and an ABI 7300 Real-Time PCR system
(Applied Biosystems, Thermo Fisher Scientific, Inc.). All primers
were synthesized by Invitrogen (Thermo Fisher Scientific, Inc.).
Fold changes in expression of each gene were calculated by a
comparative threshold cycle (Cq) method using the formula
2−ΔΔCq (28). Data were
collected from three independent experiments.
 | Table I.Primer sequences for reverse
transcription-quantitative PCR. |
Table I.
Primer sequences for reverse
transcription-quantitative PCR.
| Gene | Forward primer
(5′-3′) | Reverse primer
(5′-3′) |
|---|
| GAPDH |
CGGATTTGGTCGTATTGGG |
TCTCGCTCCTGGAAGATGG |
| TNF-α |
ATCCTGGGGGACCCAATGTA |
AAAAGAAGGCACAGAGGCCA |
| iNOS |
GAGGAGCAGGTCGAGGACTAT |
TCTTCGCCTCGTAAGGAAATAC |
| CD80 |
AAAAGACAGCTACGTGGGTGA |
GCCATGTTCTATCGGGTACTTC |
| CD86 |
TAGGGCTCCGGATATCTTTG | TCTTGAGGGTCCTTTC
TCCA |
| CD163 |
TAGGGCTCCGCTTTGGATAT |
TCTTGAGGGTCTCCACCTTT |
| CD206 |
CAAATCCACGATCAAACCTGTG |
AGAACCCTTCATAAGACCACC |
| IL-10 |
GGGAGAACCTGAAGACCCTCA |
TGCTCTTGTTTTCACAGGGAAG |
Flow cytometry
Cultured macrophages and/or NK cells were
resuspended in staining buffer (1% FBS in PBS) and were
preincubated with Fc receptor blocking reagent (cat. no.
130-059-901; Miltenyi Biotec, Inc.) for 15 min at 4°C. Macrophages
were stained with phycoerythrin-conjugated mouse anti-human CD80
(cat. no. 560925; BD Biosciences) and FITC-conjugated mouse
anti-human CD86 (cat. no. 560958; BD Biosciences). NK cells were
stained with allophycocyanin-conjugated mouse anti-human CD69 (cat.
no. 560967; BD Biosciences) on the cell surface. All the antibody
were 1:100 dilutions, and incubated in 4°C for 15 min. The
detection of BUV395-conjugated IFN-γ (cat. no. 565971; BD
Biosciences) expression in NK cells was performed following cell
surface staining. The IFN-γ antibody was diluted 1:100 and
incubated in 4°C for 30 min. Brefeldin A (Sigma-Aldrich; Merck
KGaA) was subsequently added for a final concentration of 5 µg/ml.
A BD LSRIIFortesa™ flow cytometer (BD Biosciences) was used to
perform the experiments, and the acquired data were analyzed with
FlowJo version 10 (FlowJo LLC).
RNA interference experiment
Monocytes were purified as aforementioned. The
monocytes were seeded into 96-well plates at a density of
0.2×106 cells/well in full RPMI-1640 medium with 400
IU/ml GM-CSF. The cells were cultured as 37°C in a 5%
CO2 incubator for 5 days. Galectin-9-specific small
interfering RNA (siRNA) reagent (Sense: 5′GUGCAGAGCUCAGAUUUCATT-3′
Antisense: 5′-UGAAAUCUGAGCUCUGCACTT-3′, Shanghai GenePharma Co.,
Ltd.) and the scramble control (Sense: 5′-GCUCAGAUUUCATTGUGCAGA-3′
Antisense: 5′-UGAGCUCUGCACTUGAAAUCT3′, Shanghai GenePharma Co.,
Ltd.) was added to the plates. The cells were transfected with 50
nM of siRNA in Lipofectamine® 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.). GFP was transfected together with the
siRNA treatment to determine transfection efficiency. Galectin-9
expression was detected using a fluorescence microscope
(magnification, ×200) and western blotting. Subsequent experiments
were performed 48 h following transfection.
Western blotting
Cell lysates were extracted using RIPA buffer
(Beyotime Institute of Biotechnology) supplemented with a cocktail
protease inhibitor (Roche Molecular Diagnostics), and the protein
concentration was determined using a BCA protein assay kit
(Beyotime Institute of Biotechnology) according to the
manufacturer's protocol. A total of 5–40 µg cell total protein was
separated by 10% SDS-PAGE. Proteins from macrophages treated with
the Galectin-9-siRNA reagent were separated via 10% SDS-PAGE under
nonreducing conditions. The proteins were then transferred onto
PVDF membranes (GE Healthcare Life Sciences) by electroblotting.
The membranes were blocked at 37°C for 1 h with 5% skimmed milk in
Tris-buffered saline (TBS) with Tween-20 (0.1%) and were then
incubated for overnight at 4°C with antibodies against Galectin-9
(1:1,000; cat. no. AF2045, R&D Systems, Inc.) and β-actin
(1:1,000; cat. no. 4967, Cell Signaling Technology, Inc.). The
membranes were washed with TBS washing buffer six times, and then
incubated with horseradish peroxidase-conjugated goat anti-mouse
(cat no. TA130001) or goat anti-rabbit (cat no. TA130015) secondary
antibodies (1:2,000; OriGene Technologies, Inc.) at 37°C for 1 h.
Following further washing, the protein bands were visualized by
Thermo Scientific Pierce ECL (cat. no. 32106, Thermo Fisher
Scientific, Inc.) using an enhanced chemiluminescent system (Thermo
Fisher Scientific, Inc.). The western blot images were quantified
by optical density analysis (Image J version 1.8.0, National
Institutes of Health).
Statistical analysis
All data and results were calculated from at least
three replicate measurements and are presented as the mean ±
standard deviation. Significant differences between experimental
groups and the control group were determined using two-way analysis
of variance followed by a Dunnett's t-test, mean values were
compared using paired t-tests (two groups) followed by the
Bonferroni correction for multiple comparison tests. using SPSS
version 20.0 (IBM Corp.). P<0.05 was considered to indicate a
statistically significant difference.
Results
A. fumigatus induces macrophages to
polarize into the M1 type
Macrophages were cultured with activated A.
fumigatus conidia for 24 h, and the cells and conidia
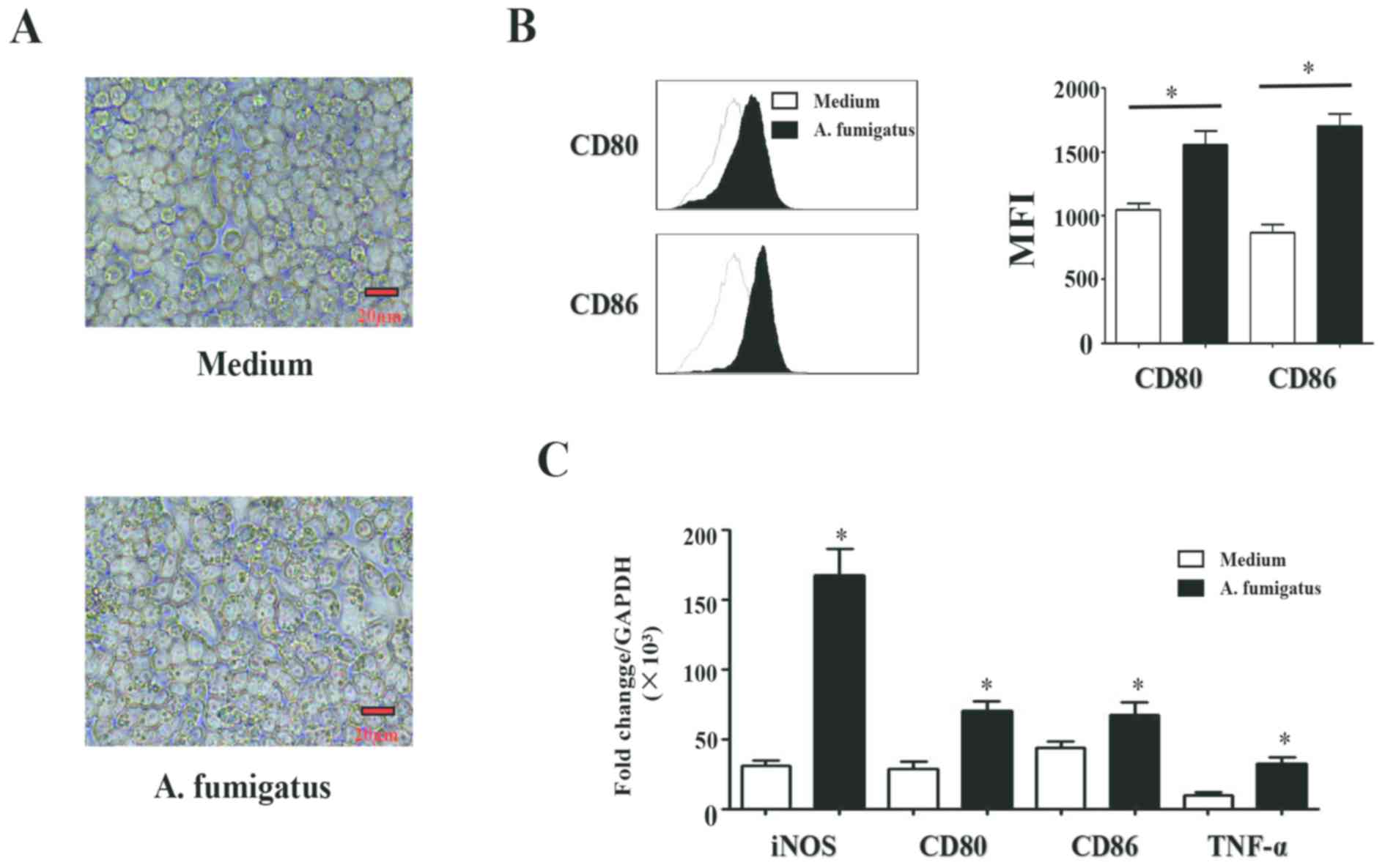
morphologies are presented in Fig.
1A. It was demonstrated that the swelling conidia of A.
fumigatus significantly increased the expression of CD80
[Median Fluorescence Intensity, (MFI), 1,000±125 vs. 1,652±145;
P<0.05] and CD86 (MFI, 950±136 vs. 1,552±245; P<0.05) in the
macrophages (Fig. 1B). RT-qPCR
analysis revealed that the mRNA expression levels of TNF-α
(P<0.01), iNOS (P<0.05), CD80 (P<0.05) and CD86
(P<0.05) were significantly upregulated in the group treated
with swelling conidia of the A. fumigatus compared with the
control (Fig. 1C). The expression
of the M2 type macrophages cell surface markers, CD163 and CD206,
and IL-10 and arginase-1 gene expression were detected; however,
there were no significant differences reported between the A.
fumigatus-treated and control groups (data not shown).
Collectively, the results suggested that A. fumigatus
induced macrophages to polarize into the M1 phenotype.
A. fumigatus promotes M1 macrophages
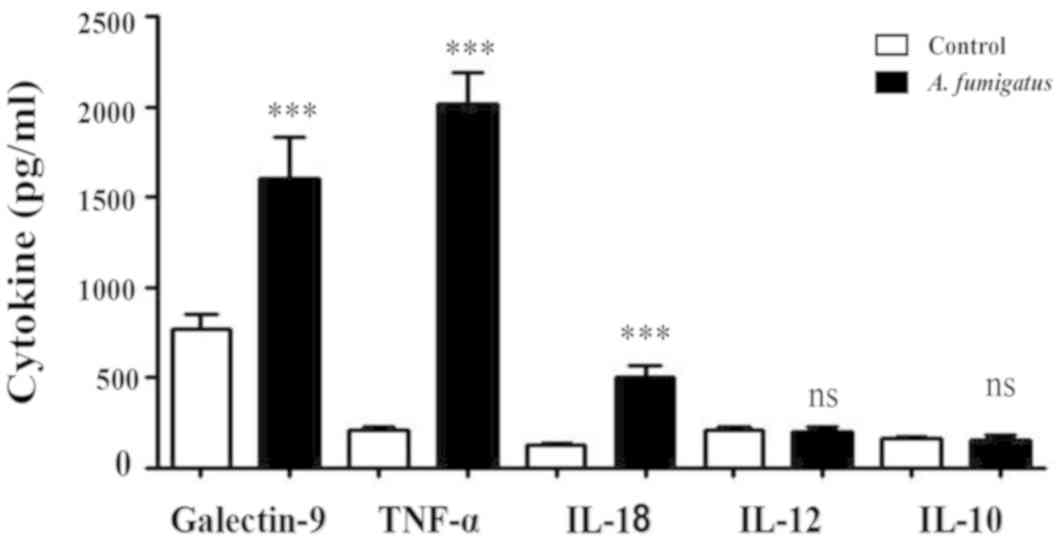
to secrete Galectin-9, TNF-α and IL-18
Galectin-9, TNF-α, IL-18, IL-12 and IL-10 levels in
the culture supernatants of macrophages treated with swelling
conidia of A. fumigatus or control were detected via ELISA.
The results revealed that A. fumigatus induced increased
macrophage secretion of Galectin-9 (1,700±130 vs. 800±120 pg/ml;
P<0.05), TNF-α (1,950±300 vs. 250±55 pg/ml; P<0.001) and
IL-18 (550±110 vs. 200±45 pg/ml; P<0.05) compared with the
control; however, there were no significant differences in the
concentrations of IL-12 or IL-10 (P>0.05; Fig. 2).
A. fumigatus activates macrophages,
which then upregulate CD69 expression and IFN-γ secretion in NK
cells
Swelling conidia of A. fumigatus stimulated
NK cells to secrete IFN-γ Intracellular Staining (IC), 5.3±1.1 vs.
1.1±0.4%; ELISA, 1,050±100 vs. 150±55 pg/ml; P<0.05) in the
A. fumigatus and NK cell co-culture system. IFN-γ expression
was significantly increased when A. fumigatus-stimulated
macrophages were added (IC, 5.3±1.1 vs. 12.3±1.3%; ELISA, 1,050±100
vs. 2,450±300 pg/ml; P<0.05; Fig.
3A-C). There was no significant difference in IFN-γ expression
in the NK cells when NK cells and monocytes were co-cultured in a
Transwell setting (P>0.05). The relative expression of CD69 on
the NK cell surface between the various groups was similar to that
of IFN-γ (Fig. 3D and E). The
results indicate that the interactions between the macrophages and
NK cells were not dependent on cell-cell contact.
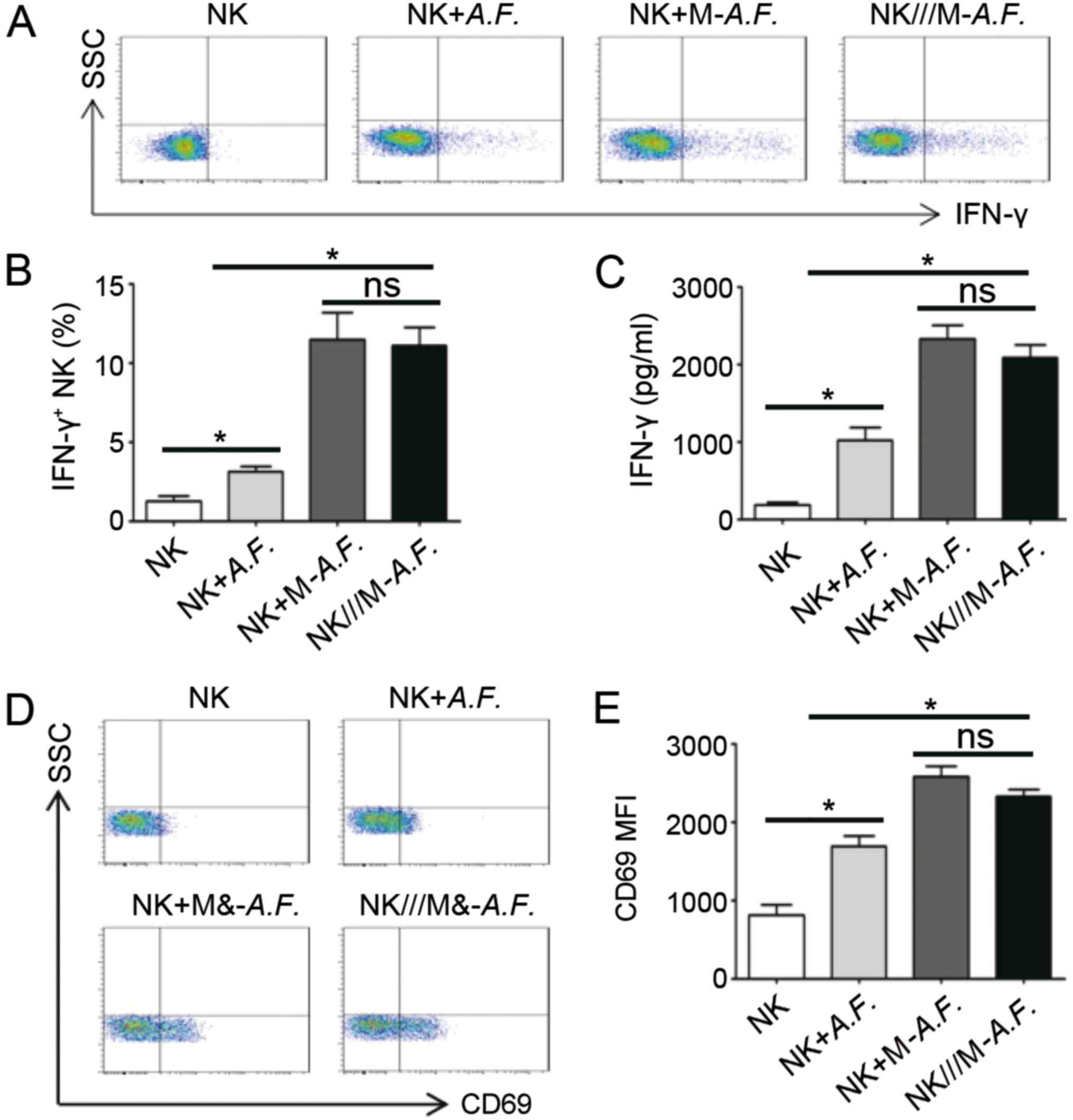 | Figure 3.A.F. stimulates macrophages,
which activate NK cells and upregulate IFN-γ and CD69. Macrophages
were stimulated with A.F. conidia (1:5) for 24 h and then
co-cultured with NK cells from the same individual under various
conditions for a further 12 h. (A) IFN-γ intracellular staining in
NK cells. (B) IFN-γ+ NK cell percentage (n=3). (C) ELISA
analysis of the supernatants of the cell culture and co-cultures
for IFN-γ (n=3). (D) CD69 expression on NK cells and (E) analysis
of the MFI (n=3). *P<0.05, as indicated. A.F., Aspergillus
fumigatus; CD, cluster of differentiation; SSC, side scatter;
MFI, median fluorescence intensity; interferon-γ, IFN-γ; M-,
macrophage; NK cell, natural killer cell; ns, not significant; ///,
separated by Transwell insert. |
Macrophages treated with A. fumigatus
regulate NK cells in a Galectin-9-dependent manner
To directly investigate the macrophage signals that
induce NK cells to produce IFN-γ, antibodies against Galectin-9,
TNF-α and IL-18 were added to the macrophage/NK cell co-cultures.
The cell surface expression of CD69 and IFN-γ levels were detected.
It was observed that IFN-γ secretion was significantly decreased
following treatment with Galectin-9 antibody (IC, 12.3±1.3 vs.
4.3±1.1%; ELISA, 2,350±320 vs. 1,040±120 pg/ml; P<0.05; Fig. 4A and B). CD69 expression was also
inhibited (P<0.05; Fig. 4C);
however, the TNF-α and IL-18 antibodies did not affect IFN-γ or
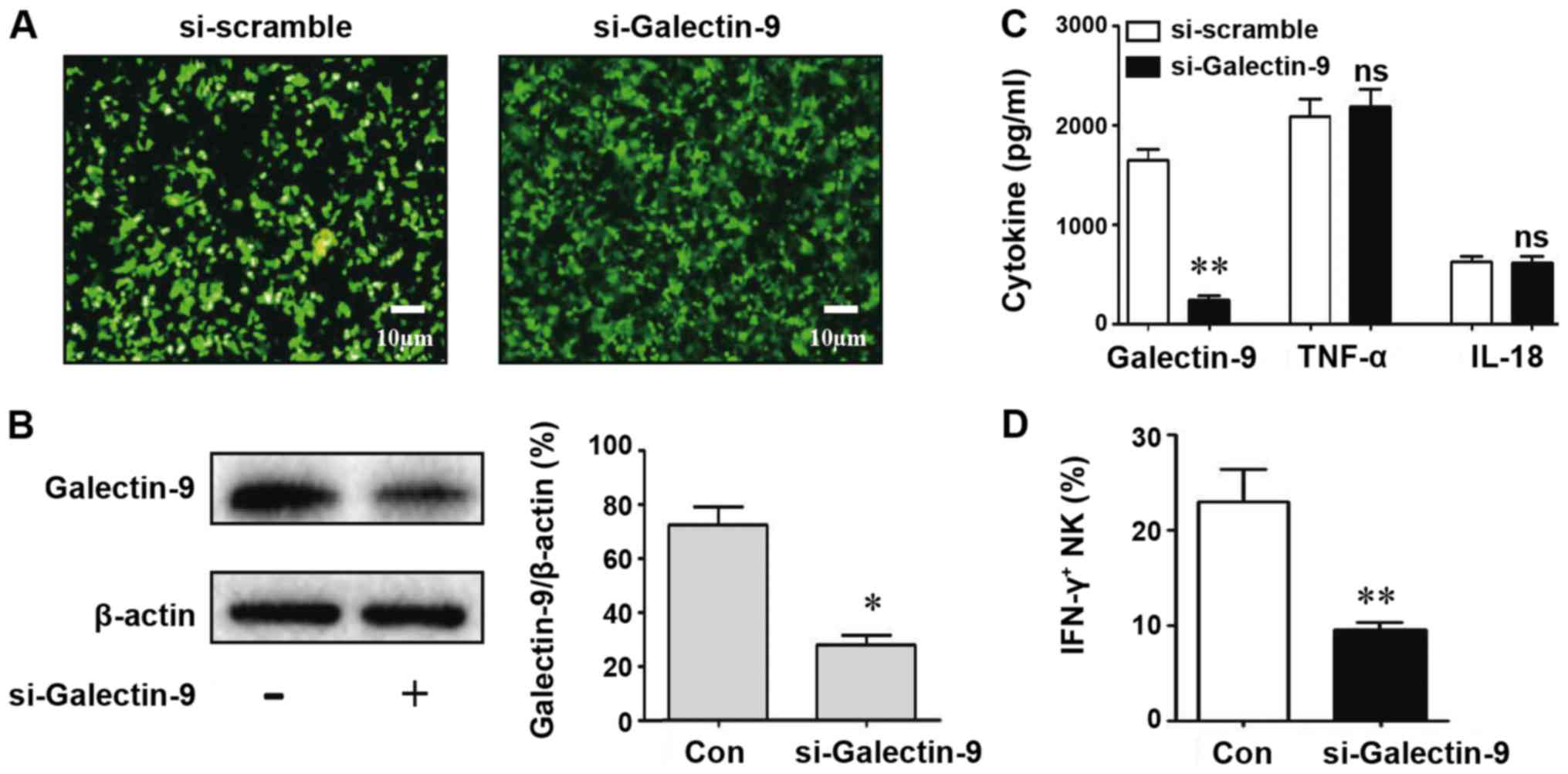
CD69 expression (P>0.05). To further demonstrate the important
role of Galectin-9 in the macrophage activation of NK cells, cells
were transfected with si-Galectin-9 to decrease the expression of
Galectin-9 in macrophages. Western blotting was performed to detect
the Galectin-9 silencing efficiency in the macrophages. As
presented in Fig. 5A and B,
Galectin-9 was significantly downregulated by >50% in
si-Galectin-9-transfected macrophages compared with the control.
Additionally, the levels of Galectin-9 (P<0.05) were decreased
significantly in the si-Galectin-9-treated group supernatant
compared with the control; however, the levels of TNF-α (P>0.05)
and IL-18 (P>0.05) were not affected (Fig. 5C). The number of IFN-γ-positive NK
cells decreased significantly following Galectin-9 silencing in
macrophages (Fig. 5D).
Collectively, the results indicated that A. fumigatus
induced macrophages to polarize into M1 macrophages by secreting
Galectin-9, which then promoted NK cell activity and IFN-γ
secretion.
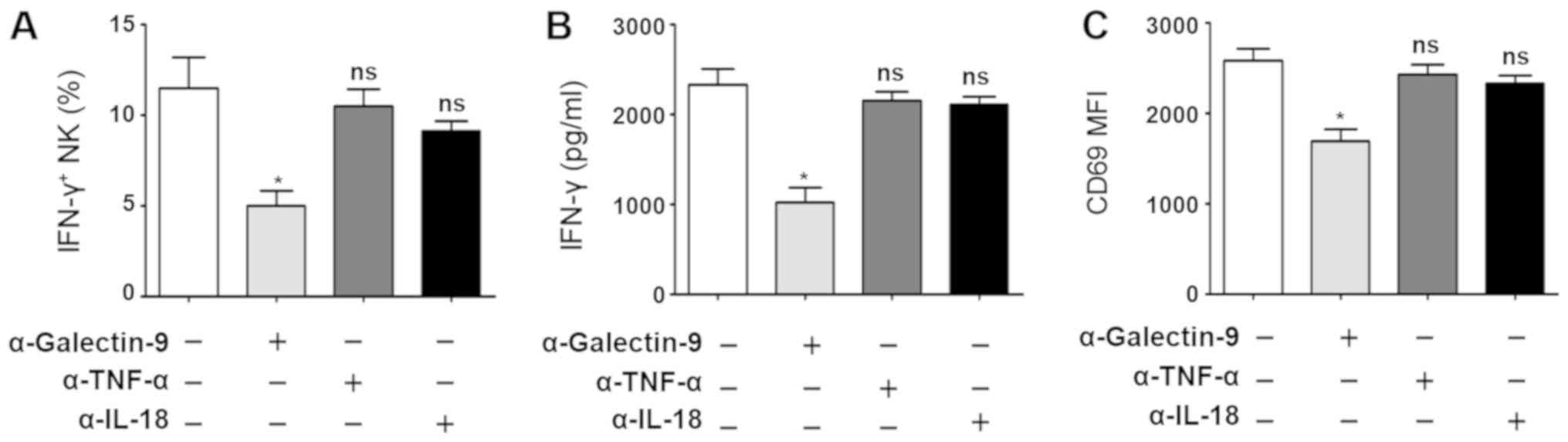 | Figure 4.Galectin-9 is involved in the
crosstalk between NK cells and macrophages in response to A.
fumigatus. Macrophages were stimulated by A. fumigatus
conidia (1:5) for 24 h and were then co-cultured with the NK cells
from the same individual for a further 12 h, in the presence or
absence of Galectin-9-, IL-18- or TNF-α-specific antibodies when
added to NK cells. (A) IFN-γ+ NK cell percentage (n=3).
(B) IFN-γ cytokine levels as determined by ELISA (n=3). (C) CD69
expression on NK cells (n=3). *P<0.05 vs. control. CD, cluster
of differentiation; IL, interleukin; interferon-γ, IFN-γ; NK cell,
natural killer cell; ns, not significant; TNF-α, tumor necrosis
factor-α; A. fumigatus, Aspergillus fumigatus; MFI, median
fluorescence intensity. |
Discussion
A. fumigatus is a ubiquitous opportunistic
fungus. The incidence of infection caused by A. fumigatus in
immunocompromised patients has increased, and the mortality rate is
at a high level (29). Following
inhalation of A. fumigatus, the ciliated cells of the
tracheal mucosa can remove A. fumigatus conidia
automatically following indraft in a normal host; however, in an
immunodeficient host, A. fumigatus conidia can colonize in
the lungs and germinate into hyphae, eventually leading to the
onset of invasive pulmonary aspergillosis (30).
Macrophages serve an important role in the
elimination of invasive microorganisms, effectively capturing and
removing A. fumigatus conidia by releasing acidified
phagosomes and reactive oxygen intermediates (31). There are two major subtypes of
macrophages, including the classically activated M1 macrophages and
the alternatively activated M2 macrophages (22). M1 macrophages secrete
proinflammatory cytokines, in contrast to M2 macrophages (23). The present findings revealed that
the swelling conidia of A. fumigatus significantly increased
the expression of CD80 and CD86 on macrophages, along with the
expression of iNOS and TNF-α. Additionally, the swelling conidia of
Aspersions fumigatus promoted the secretion of Galectin-9,
TNF-α and IL-18. TNF-α and IL-18 are two important Th1-type
cytokines that exhibit strong immunomodulatory functions. They
activate T and NK cells in vivo to contribute towards
antitumor or anti-infection immune responses (32,33).
The body's immune system is a net structure composed
of a variety of immune cells (24). Therefore, the interaction between
immune cells has received increasing attention. Activated
macrophages interact with other immune cells via surface molecules
(24). Intrahepatic macrophages
reduce inflammatory injury and serve an antiviral role in the liver
by interacting with NK cells (34). Monocytes regulate the functions of
other cells by secreting a variety of cytokines and expressing
various surface proteins (10).
The interaction between NK cells and monocytes is present in
various tissues and organs, and peripheral blood (35); however, to the best of our
knowledge, there has been limited investigation into the
interaction between NK cells and monocytes in fungal infection.
T-cell immunoglobulin and mucin-domain containing-3 (Tim-3) and
4-1BB are known receptors of Galectin-9 on the surface of T cells
and NK cells (35). Tim-3 was
reported to act as a marker of activation or maturation of NK
cells. Recombinant Galectin-9 enhanced the ability of NK cells to
secrete cytokines and induce cytotoxicity in vitro (27). In the present study, it was
observed that Galectin-9 signaling regulated the function of NK
cells following infection by Aspersions fumigatus; however,
the effects of Galectin-9 signaling downstream molecules such as
Tim-3 on NK cell function requires further investigation.
Macrophage-derived Galectin-9 increased NK cell cytotoxicity in HCV
infection, which may be associated with liver injury and persistent
infection (24). It is important
to understand whether there are interactions between natural immune
cells, and to identify which key molecules serve an important role
in the fungi and the host.
It has been reported that A. fumigatus
activates NK cells, but the function of A. fumigatus in the
interactions between macrophages and NK cells remains unclear
(36). In the present study, it
was observed that A. fumigatus conidia induced macrophages
to polarize into M1 macrophages and secrete substantial quantities
of Galectin-9. It was further revealed using antibody
neutralization and gene silencing experiments that Galectin-9
served an important role in the interaction between macrophages and
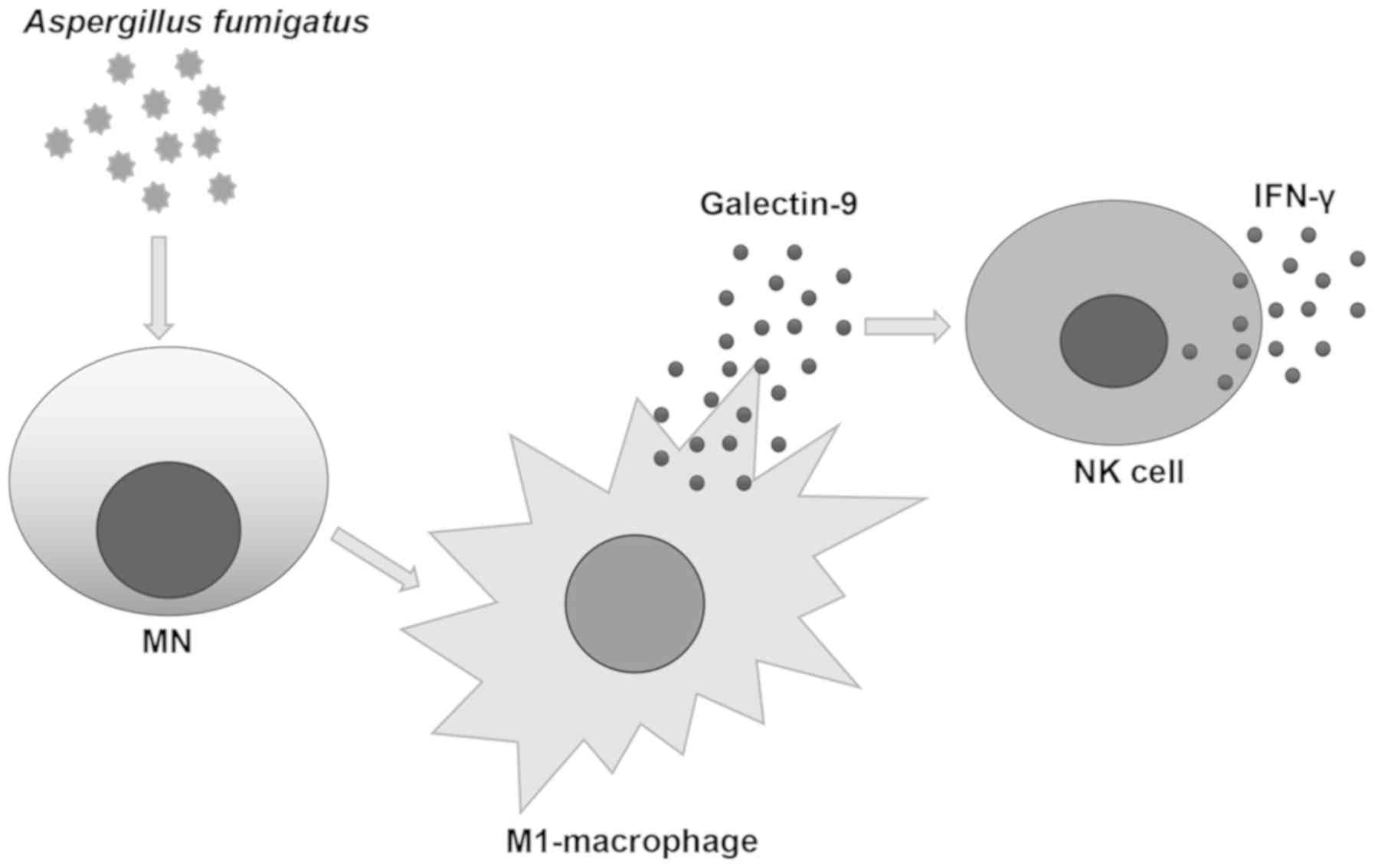
NK cells regulated by A. fumigatus. Collectively, a
mechanism via which A. fumigatus polarized M1 macrophages by
secreting Galectin-9 was identified, with downstream activation of
NK cells and secretion of IFN-γ (Fig.
6). These findings suggested a novel direction for the study of
macrophages and NK cells in the inflammatory response to the A.
fumigatus infection, and may aid the identification of
potential strategies to control the progression of
inflammation.
Acknowledgements
The authors would like to thank the staff of Jilin
University Mycology Research Center for their assistance.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant no. 81271802) and the
National Science and Technology Major Project of the Ministry of
Science and Technology of China (grant no. 2013ZX10004612-006).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XZ performed the laboratory experiments and drafted
the manuscript. DH analyzed the experimental data and revised the
manuscript. SG and YW were involved in conducting the laboratory
experiments. LW designed the project and supervised this study.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Changchun Blood Center, Jilin, China and conducted in
accordance with the approved guidelines for the ‘Use of haemocytes
in research’. Written informed consent for the use of haemocytes in
research was obtained from all participants
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interest.
References
|
1
|
Dewi IMW, van de Veerdonk FL and Gresnigt
MS: The multifaceted role of T-helper responses in host defense
against Aspergillus fumigatus. J Fungi (Basel). 3(pii): E552017.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hellmann AM, Lother J, Wurster S, Lutz MB,
Schmitt AL, Morton CO, Eyrich M, Czakai K, Einsele H and Loeffler
J: Human and murine innate immune cell populations display common
and distinct response patterns during their in vitro interaction
with the pathogenic mold Aspergillus fumigatus. Front Immunol.
8:17162017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schmidt S, Tramsen L, Schneider A,
Schubert R, Balan A, Degistirici Ö, Meisel R and Lehrnbecher T:
Impact of human mesenchymal stromal cells on antifungal host
response against Aspergillus fumigatus. Oncotarget. 8:95495–95503.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chakrabarti A, Chatterjee SS, Das A and
Shivaprakash MR: Invasive aspergillosis in developing countries.
Med Mycol. 49 (Suppl 1):S35–S47. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bao GQ, He L, Lee D, D'Angelo J and Wang
HC: An ongoing search for potential targets and therapies for
lethal sepsis. Mil Med Res. 2:202015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Osherov N: Interaction of the pathogenic
mold Aspergillus fumigatus with lung epithelial cells. Front
Microbiol. 3:3462012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hussell T and Bell TJ: Alveolar
macrophages: Plasticity in a tissue-specific context. Nat Rev
Immunol. 14:81–93. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rosowski EE, Raffa N, Knox BP, Golenberg
N, Keller NP and Huttenlocher A: Macrophages inhibit Aspergillus
fumigatus germination and neutrophil-mediated fungal killing. PLoS
Pathog. 14:e10072292018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sun H, Xu XY, Shao HT, Su X, Wu XD, Wang Q
and Shi Y: Dectin-2 is predominately macrophage restricted and
exhibits conspicuous expression during Aspergillus fumigatus
invasion in human lung. Cell Immunol. 284:60–67. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Messlinger H, Sebald H, Heger L, Dudziak
D, Bogdan C and Schleicher U: Monocyte-derived signals activate
human natural killer cells in response to Leishmania parasites.
Front Immunol. 9:242018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rückerl D, Campbell SM, Duncan S,
Sutherland TE, Jenkins SJ, Hewitson JP, Barr TA, Jackson-Jones LH,
Maizels RM and Allen JE: Macrophage origin limits functional
plasticity in helminth-bacterial co-infection. PLoS Pathog.
13:e10062332017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ewen EM, Pahl JHW, Miller M, Watzl C and
Cerwenka A: KIR downregulation by IL-12/15/18 unleashes human NK
cells from KIR/HLA-I inhibition and enhances killing of tumor
cells. Eur J Immunol. 48:355–365. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Massawe R, Drabo L and Whalen M: Effects
of pentachlorophenol and dichlorodiphenyltrichloroethane on
secretion of interferon gamma (IFNγ) and tumor necrosis factor
alpha (TNFα) from human immune cells. Toxicol Mech Methods.
27:223–235. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Spörri R, Joller N, Albers U, Hilbi H and
Oxenius A: MyD88-dependent IFN-gamma production by NK cells is key
for control of Legionella pneumophila infection. J Immunol.
176:6162–6171. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hansen DS, D'Ombrain MC and Schofield L:
The role of leukocytes bearing natural killer complex receptors and
killer immunoglobulin-like receptors in the immunology of malaria.
Curr Opin Immunol. 19:416–423. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ma LL, Wang CL, Neely GG, Epelman S,
Krensky AM and Mody CH: NK cells use perforin rather than
granulysin for anticryptococcal activity. J Immunol. 173:3357–3365.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Espinosa V, Dutta O, McElrath C, Du P,
Chang YJ, Cicciarelli B, Pitler A, Whitehead I, Obar JJ, Durbin JE,
et al: Type III interferon is a critical regulator of innate
antifungal immunity. Sci Immunol. 2(pii): eaan53572017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Morton CO, Bouzani M, Loeffler J and
Rogers TR: Direct interaction studies between Aspergillus fumigatus
and human immune cells; what have we learned about pathogenicity
and host immunity? Front Microbiol. 3:4132012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shi J, Zhao Y, Wang Y, Gao W, Ding J, Li
P, Hu L and Shao F: Inflammatory caspases are innate immune
receptors for intracellular LPS. Nature. 514:187–192. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Plebanek MP, Angeloni NL, Vinokour E, Li
J, Henkin A, Martinez-Marin D, Filleur S, Bhowmick R, Henkin J,
Miller SD, et al: Pre-metastatic cancer exosomes induce immune
surveillance by patrolling monocytes at the metastatic niche. Nat
Commun. 8:13192017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kochiyama T, Li X, Nakayama H, Kage M,
Yamane Y, Takamori K, Iwabuchi K and Inada E: Effect of propofol on
the production of inflammatory cytokines by human polarized
macrophages. Mediators Inflamm. 2019:19195382019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou D, Huang C, Lin Z, Zhan S, Kong L,
Fang C and Li J: Macrophage polarization and function with emphasis
on the evolving roles of coordinated regulation of cellular
signaling pathways. Cell Signal. 26:192–197. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang Q, Wang Y, Zhai N, Song H, Li H,
Yang Y, Li T, Guo X, Chi B, Niu J, et al: HCV core protein inhibits
polarization and activity of both M1 and M2 macrophages through the
TLR2 signaling pathway. Sci Rep. 6:361602016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li H, Zhai N, Wang Z, Song H, Yang Y, Cui
A, Li T, Wang G, Niu J, Crispe IN, et al: Regulatory NK cells
mediated between immunosuppressive monocytes and dysfunctional T
cells in chronic HBV infection. Gut. 67:2035–2044. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liang CC, Li CS, Weng IC, Chen HY, Lu HH,
Huang CC and Liu FT: Galectin-9 is critical for mucosal adaptive
immunity through the T Helper 17-IgA Axis. Am J Pathol.
188:1225–1235. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Golden-Mason L and Rosen HR: Galectin-9:
Diverse roles in hepatic immune homeostasis and inflammation.
Hepatology. 66:271–279. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nishio A, Tatsumi T, Nawa T, Suda T,
Yoshioka T, Onishi Y, Aono S, Shigekawa M, Hikita H, Sakamori R, et
al: CD14+ monocyte-derived galectin-9 induces natural
killer cell cytotoxicity in chronic hepatitis C. Hepatology.
65:18–31. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gabrielli E, Fothergill AW, Brescini L,
Sutton DA, Marchionni E, Orsetti E, Staffolani S, Castelli P,
Gesuita R and Barchiesi F: Osteomyelitis caused by Aspergillus
species: A review of 310 reported cases. Clin Microbiol Infect.
20:559–565. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Miceli MH and Kauffman CA: Aspergillus
Galactomannan for diagnosing invasive aspergillosis. JAMA.
318:1175–1176. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xu S and Shinohara ML: Tissue-resident
macrophages in fungal infections. Front Immunol. 8:17982017.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Timoteo RP, da Silva MV, Miguel CB, Silva
DA, Catarino JD, Rodrigues Junior V, Sales-Campos H and Freire
Oliveira CJ: Th1/Th17-related cytokines and chemokines and their
implications in the pathogenesis of pemphigus vulgaris. Mediators
Inflamm. 2017:71512852017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kaplanski G: Interleukin-18: Biological
properties and role in disease pathogenesis. Immunol Rev.
281:138–153. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tu Z, Bozorgzadeh A, Pierce RH, Kurtis J,
Crispe IN and Orloff MS: TLR-dependent cross talk between human
Kupffer cells and NK cells. J Exp Med. 205:233–244. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Golden-Mason L, McMahan RH, Strong M,
Reisdorph R, Mahaffey S, Palmer BE, Cheng L, Kulesza C, Hirashima
M, Niki T and Rosen HR: Galectin-9 functionally impairs natural
killer cells in humans and mice. J Virol. 87:4835–4845. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bouzani M, Ok M, McCormick A, Ebel F,
Kurzai O, Morton CO, Einsele H and Loeffler J: Human NK cells
display important antifungal activity against Aspergillus
fumigatus, which is directly mediated by IFN-γ release. J Immunol.
187:1369–1376. 2011. View Article : Google Scholar : PubMed/NCBI
|