Introduction
The prevalence of bladder dysfunction increases with
age and it strongly influences the quality of life of the elderly
population. Aging male patients with prostate hyperplasia are prone
to suffer from lower urinary tract symptoms (LUTS), and elderly
female patients are susceptible to overactive bladder with absence
of bladder outlet obstruction (BOO) (1,2).
Therefore, BOO is not the only factor causing bladder dysfunction.
Transurethral resection of the prostate is an effective approach to
relieve obstruction in males, but complete bladder function is not
recovered following prostate resection (3), which may be due to the irreversible
bladder damage mediated by aging. Therefore, female rats without
BOO were used in the present study. The urodynamics and bladder
morphology of aging rats requires investigation. It was speculated
that a molecular mechanism associated with the aging process may
explain these alterations (4,5).
Age-associated alterations have been investigated
more extensively in previous studies, but these have focused
primarily on bladder contraction in vivo and in vitro
(5) The exact pathophysiological
mechanisms of bladder aging remain to be elucidated (6). Inflamm-aging, which offers new
insights into the aging process, involves chronic inflammatory
cytokine production and functional decline (7). Accumulating evidence demonstrates
that aging is closely associated with chronic low-level
inflammation. Meanwhile, a recent study confirmed that activation
of the NACHT, LRR and PYD domains-containing protein 3 (NLRP3)
inflammasome, which includes cleaved Caspase1, is regulated through
the NAD-dependent protein deacetylase sirtuin-3, mitochondrial
(SIRT3)-superoxide dismutase 2, mitochondrial (SOD2) signaling
pathway (8). Our previous study
demonstrated that the NLRP3 inflammasome is involved in endothelial
cellular senescence (9). Compared
with the majority of other types of epithelial cells, the
urothelium may underlie this mechanism in the bladder. However, the
involvement of NLRP3 in urothelial alterations and bladder
dysfunction with advancing age remains poorly understood.
The proposed etiologies of LUTS involve a number of
factors, including myogenic and neurogenic factors, but remodeling
of the urothelium serves an equal role in the progression of LUTS
(10). A previous study
demonstrated that the urotheliogenic factor and its interactions
with the detrusor muscle and neurons may explain the mechanism
underlying bladder dysfunction with advancing age (11). The urothelium, which is
characterized by sensory innervation, serves a critical role in
regulating micturition (12). The
expression level of NLRP3, which is principally located in the
urothelium, is induced by bladder injury from noxious stimuli in
the urine and may trigger the urothelial inflammatory response
(13). Aging process was
demonstrated to be accelerated by senescent cells (14) and urothelial senescence may be
responsible for bladder degradation. However, there is no evidence
to confirm that alterations in the urothelium are associated with a
decline in bladder function with age. It was hypothesized that
inflamm-aging may serve an important role in the bladder,
particularly in the urothelium, with advancing age. Therefore, in
order to validate this hypothesis in the present study, the
senescence marker p21 (15) was
identified by immunohistochemical staining, and differences in
inflammasome expression were determined by immunofluorescence and
western blot analysis between young and old rats. Cystometry was
used to assess detrusor activity.
Materials and methods
Animals and sample preparation
The animal experiments were approved by the Ethics
Committee of Chengdu University (Chengdu, China). A total of 20
female Sprague-Dawley (SD) rats were obtained from The Dashuo
Laboratory Animal Co., Ltd. (Chengdu, China) and divided into the
following two groups (n=10 rats/group): 2-month-old group (young
group, 271±28 g), and 24-month-old group (old group, 412±47 g). A
total of two rats were housed in each cage at room temperature
(20±2°C) and saturated humidity (50±2%), with access to food and
water ad libitum. The room had a constant 12-h light/dark
cycle. The rats were anesthetized with 3% isoflurane inhalant, and
the peritoneal cavity was opened. The bladder was quickly excised
and the rat was subsequently sacrificed. The tissue from the upper
part of the bladder were placed in 10% formalin for fixation at
room temperature for 2 days, and used for histological analysis.
The remaining bladder tissues were cut into small pieces of ~5 mm
and placed in liquid nitrogen for western blot analysis.
Blood chemical measurement
In total, 1 ml blood was collected from the
abdominal aorta of 20 rats following bladder extraction and the
serum was extracted by centrifugation at 13,000 × g for 10 min at
4°C. The serum was stored at 4°C and the oxidative stress markers
including malondialdehyde and superoxide dismutase, and the level
of blood lipids were detected within 12 h. The biochemical data
were detected with a full-automatic biochemical analyzer (Chemray
240; Rayto Life and Analytical Sciences Co., Ltd., Shenzhen, China)
using routine enzymatic assays.
Urodynamic analysis
Rats from the young group and the old group (n=4
rats/group) were anesthetized via inhalation of 3% isoflurane. A
PE50 polyethylene catheter (internal diameter, 0.58 mm; outer
diameter, 0.96 mm) was implanted into the bladder dome and buried
subcutaneously at the back of the neck as described by Zhao et
al (4). A total of 3 days
after the surgery, cystometry was performed through the
polyethylene catheter that was connected to a pressure transducer
(Bonito XL; Laborie Medical Technologies Inc., Mississauga, ON,
Canada) and a syringe pump (Jian Yuan Medical Technology Co., Ltd.,
Changsha, China), which administered a warm saline infusion at a
rate of 10 ml/h. The rats remained awake without anesthetization
and were restricted to a small cage. Urodynamic parameters,
including the maximum bladder capacity (MBC), maximum voiding
pressure (MVP), bladder leak point pressure (BLPP), voiding volume
(VV), voiding time (VT) and residual volume (RV), were analyzed.
The BLPP was recorded when micturition occurred in a phase of
high-frequency oscillations.
Histomorphology and
immunohistochemical staining
The paraffin-embedded bladders were cut into
sections (5 µm) perpendicular to the longitudinal axis of the
bladder. Masson's trichrome staining was performed on a number of
the sections, according to the manufacturer's instructions
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany). The remaining
sections were deparaffinized and rehydrated in xylene and a
gradient alcohol series, and antigen retrieval was performed by
heating the sections at ~95°C for 8 min with sodium citrate antigen
retrieval solution (pH 6.0). Subsequently, endogenous peroxidase
was blocked with 3% hydrogen peroxide. PBS containing 3% bovine
serum albumin (BSA; cat. no. G5001; Wuhan Servicebio Technology
Co., Ltd., Wuhan, China) was used for blocking of non-specific
binding at room temperature for 30 min. The sections were incubated
with primary antibodies, including those for rabbit p21 (1:100;
cat. no. 10355-1-1AP; Wuhan Sanying Biotechnology, Wuhan, China)
and NLRP3 (1:100; cat. no. 19771-1-AP; Wuhan Sanying
Biotechnology), at 4°C overnight, and subsequently with secondary
antibodies (1:1,000; cat. no. GB23302; Wuhan Servicebio Technology
Co., Ltd.) incubated at room temperature for 50 min and labelled
with horseradish peroxidase. 3,3′-Diaminobenzidine chromogenic
reagent was used to stain the nucleus at room temperature until the
nuclei exhibited positive staining. Subsequently, the samples were
counterstained with hematoxylin at room temperature for 3 min.
Following dehydration, the sections were mounted with resin medium.
Samples were observed and imaged using an ortho-fluorescence
microscope (Nikon Corporation, Tokyo, Japan; magnification, 100× or
200×). The detrusor muscle and collagen content were quantified
using ImagePro Plus 6.0 software (Media Cybernetics, Inc.,
Rockville, MD, USA).
Western blot analysis
The bladder tissues were lysed with
radioimmunoprecipiation assay-150 lysis buffer and complete
protease inhibitor (Roche Applied Science, Penzberg, Germany). The
protein concentration was detected by bicinchoninic acid protein
assay kit; cat. no. CW0014; CoWin Biosciences Co., Ltd., Beijing,
China) and 50 µg total protein was loaded in each lane. Proteins
were separated on a 10% SDS-PAGE gel and transferred to a PVDF
membrane (EMD Millipore, Billerica, MA, USA). Subsequently, the
membrane was blocked with 5% BSA in TBS with Tween-20 for 1 h at
room temperature and incubated with the primary antibodies,
including interleukin (IL)1β (1:100; cat. no. 12242S; Cell
Signaling Technology, Inc., Danvers, MA, USA), Caspase1 (1:100;
cat. no. 3866S; Cell Signaling Technology, Inc.), SIRT3 (1:200;
cat. no. 1099-1-AP; Wuhan Sanying Biotechnology) and GAPDH
(1:1,000; cat. no. 10494-1-AP; Wuhan Sanying Biotechnology, Wuhan,
China) overnight at 4°C. The membranes were incubated with the
horseradish peroxidase-conjugated secondary antibodies (1:1,000;
cat. nos. 7074P2 and 7076P2; Cell Signaling Technology, Inc.) for 1
h at room temperature. The images were detected by a ChemiDoc XRS+
system (Bio-Rad Laboratories, Inc., Hercules, CA, USA) using an
enhanced chemiluminescence kit (Thermo Fisher Scientific, Inc.).
The densities of the western blot bands were analyzed using Image
Lab Software 5.2.1 (Bio-Rad Laboratories, Inc.).
Immunofluorescent staining
Sections from the paraffin-embedded bladders were
deparaffinized and rehydrated, and antigen retrieval was performed.
The slides were incubated with spontaneous fluorescence quenching
reagent for 5 min and subsequently with 5% BSA for 30 min. The
excess BSA was washed off and the sections were incubated with the
primary antibody anti-Caspase1 (1:200; cat. no. 1872; Abcam,
Cambridge, UK) overnight at 4°C. The sections were rinsed with PBS
(pH 7.4) and then covered with the secondary antibody for 50 min in
the dark. When the nuclei had been stained with a DAPI counterstain
for 10 min at room temperature, antifade mounting medium and a
coverslip were added to the slide. Samples were observed and imaged
using an ortho-fluorescent microscopy (Nikon Corporation, Tokyo,
Japan; magnification, 100× or 400×).
Statistical analysis
The data are presented as the mean ± standard
deviation. Each experiment was repeated at least three times.
Statistical analysis was performed using GraphPad Prism software
5.01 (GraphPad Software, Inc., La Jolla, CA, USA). Comparisons
between two groups were performed using Student's t-tests and
P<0.05 was considered to indicate a statistically significant
difference.
Results
Changes in bladder weight and
morphology
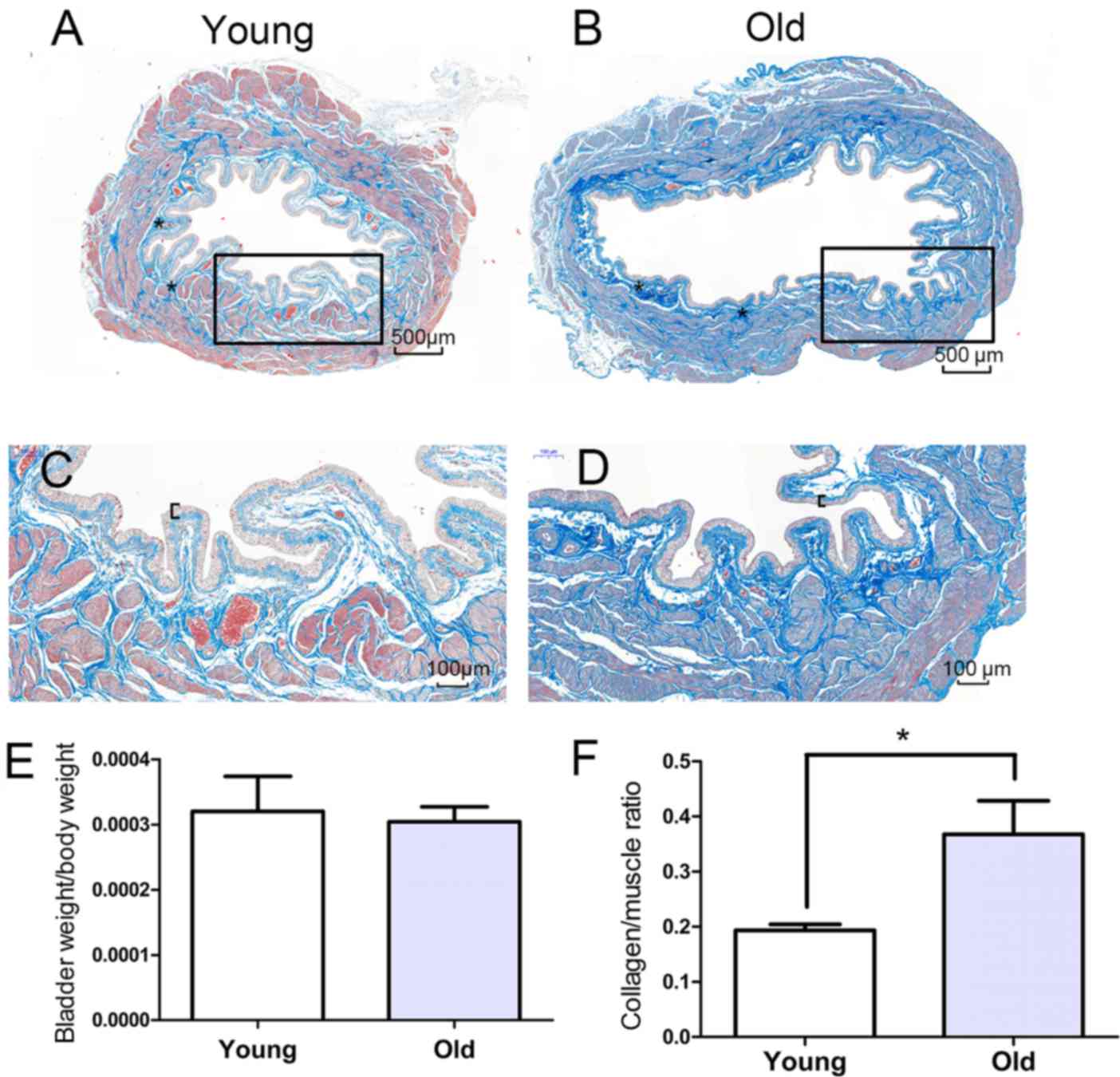
The morphological alterations in the bladders were
evaluated using Masson's trichrome staining, and representative
images are presented in Fig. 1A-D.
Compared with the young bladders, the old bladders displayed
pathological characteristics of aging, including increased collagen
deposition, urothelial thinning and muscle shrinkage, which
contribute to weakening of detrusor contraction. The weights of the
bladders in the old group (0.126±0.021 g) were significantly higher
compared with those of the young group (0.086±0.010 g).
Furthermore, the body weight was increased in the old group
compared with the young group and the ratio of bladder weight to
body weight was not significantly different (P>0.05) between the
two groups (Fig. 1E). The ratio of
collagen content to muscle was higher in the old group compared
with the young group (Fig.
1F).
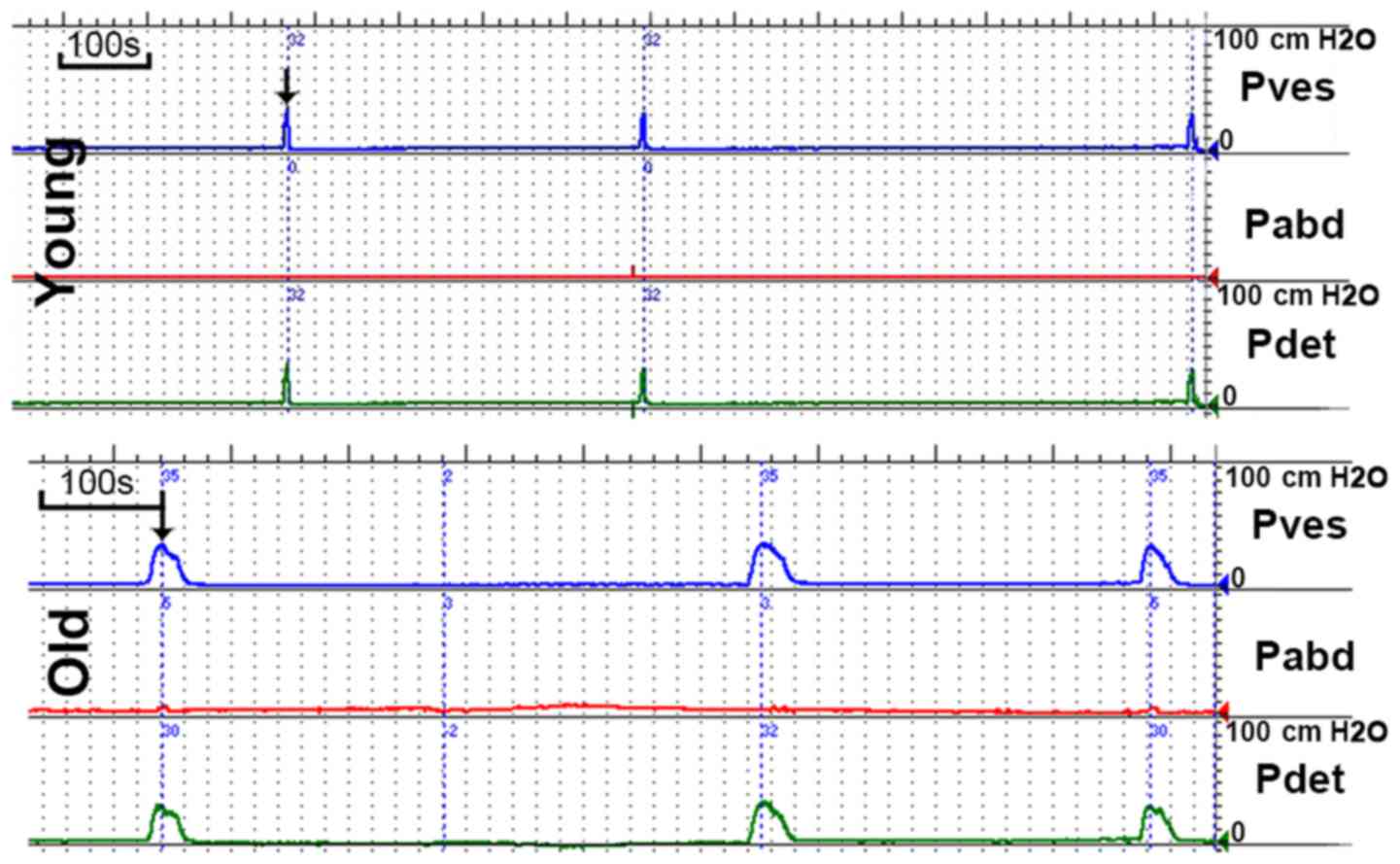
Urodynamic parameters
Representative cystometrogram recordings and
urodynamic parameters are presented in Fig. 2 and Table I, respectively. The results of the
cystometry indicated that the MVP and BLPP were similar in the two
groups. However, the MBC and RV were significantly increased in the
old group and the VT was prolonged in the old group compared to the
young group. Marked unstable contraction was not observed prior to
the voiding peak pressure.
 | Table I.Characteristics of oxidative stress
marker and urodynamic parameters in rats. |
Table I.
Characteristics of oxidative stress
marker and urodynamic parameters in rats.
| Parameter | Young | Old |
|---|
| MDA, nmol/ml | 4.12±0.93 |
8.24±2.31a |
| SOD, U/ml, | 360.44±32.63 |
314.98±13.94a |
| MBC, ml | 1.08±0.21 |
1.55±0.17a |
| MBC/BW | 11.72±1.23 | 12.04±0.97 |
| BLPP,
cmH2O | 23.62±3.85 | 24.94±5.32 |
| MVP,
cmH2O | 36.37±2.56 | 34.94±3.54 |
| VV, ml | 0.97±0.19 | 1.28±0.22 |
| RV, ml | 0.09±0.04 |
0.26±0.11a |
| VT, sec | 9.75±2.21 |
16.50±4.65a |
Identification of senescence markers
in the bladder tissues and oxidative stress markers in the
serum
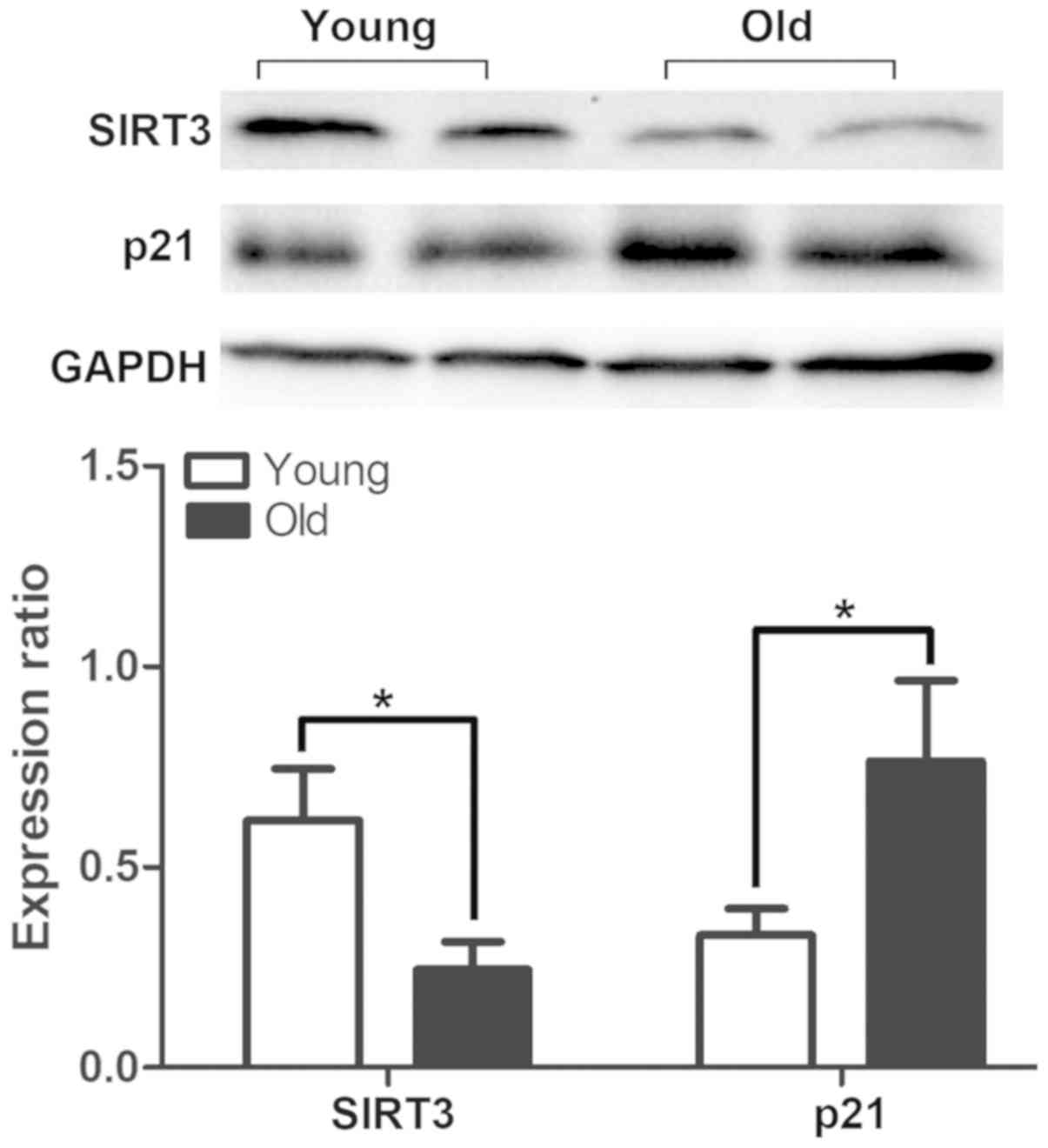
In order to determine the aging status of the
bladders, the expression of the senescence marker p21 was detected
by western blotting and immunohistochemical staining. The results
demonstrated that the expression of p21 was significantly increased
in the old group (Fig. 3), and
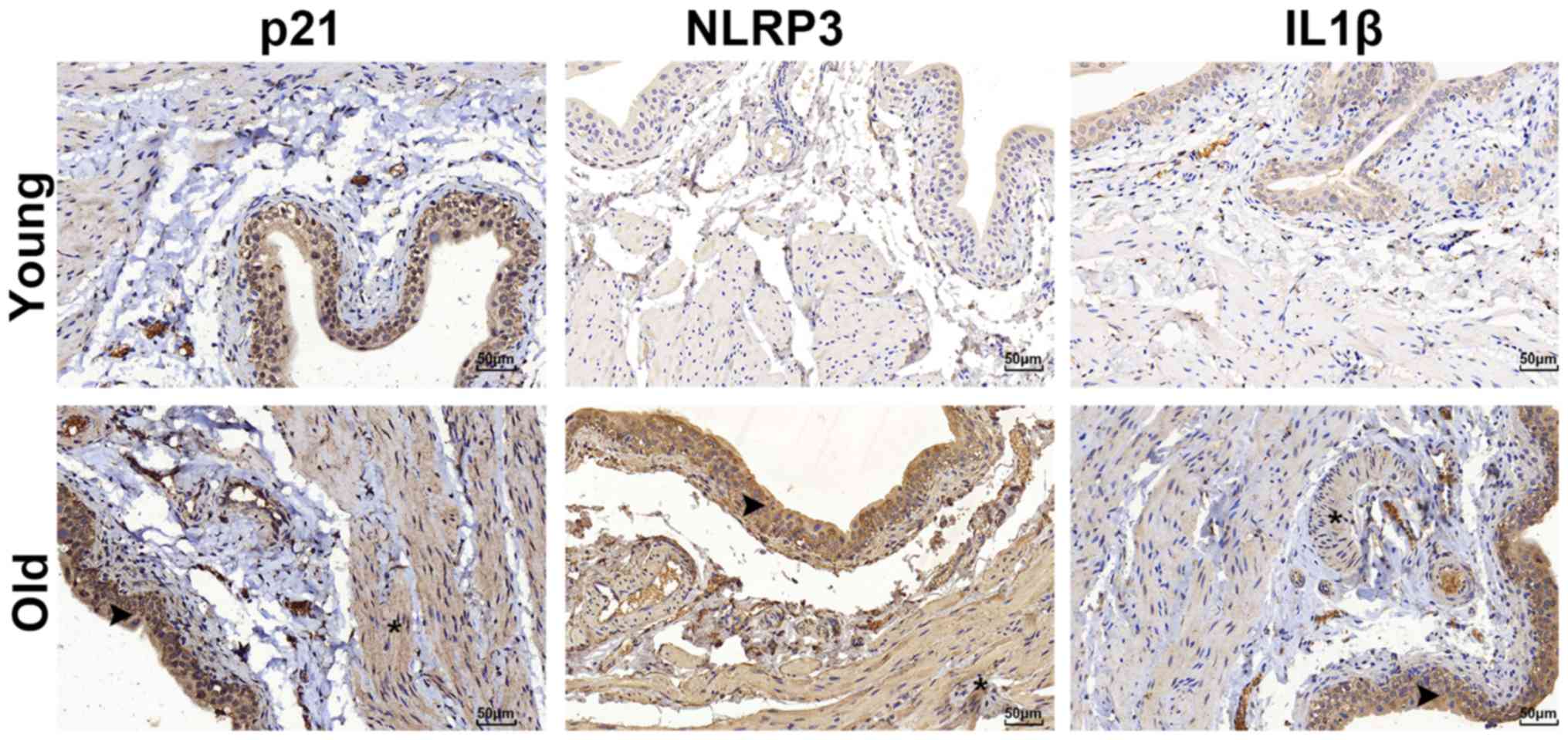
there was positive expression of p21 in the bladders of the old
group that was primarily located in the urothelium rather than the
muscle layer (Fig. 4).
Aging-associated degeneration of the mitochondrial protein SIRT3
was analyzed by western blotting, and the data indicated that the
expression was significantly decreased in the bladders of the old
group (Fig. 3). The serum
biomarkers of oxidative stress were analyzed and the results
demonstrated that the levels of malondialdehyde (MDA) were
increased, while those of the antioxidant marker SOD were decreased
in the old group compared with the young group (Table I). The level of blood lipids was
not altered among groups.
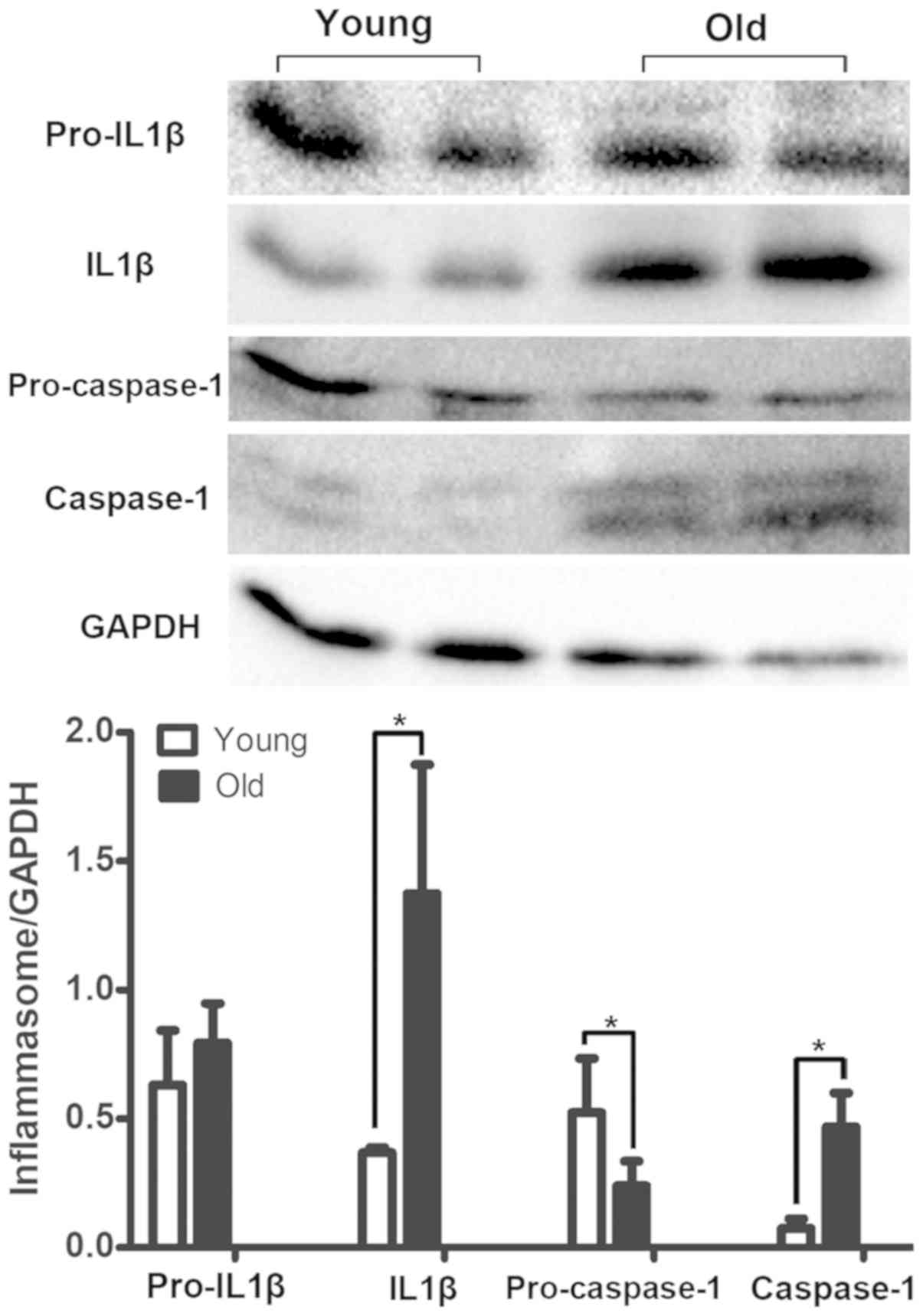
Differences in the expression of NLRP3
inflammasome components
The expression of NLRP3 inflammasome components
(NLRP3 and IL1β) was detected by immunohistochemical staining. The
results revealed that NLRP3 and IL1β were primarily located in the
urothelium and were markedly upregulated in the bladder tissues of
the old group (Fig. 4). However,
the inflammasome sensor NLRP3 was scarcely observed in the muscle
layer, as illustrated in Fig. 4.
Furthermore, the increased expression profile of the NLRP3
inflammasome components was examined using a western blot assay.
The bladders in the old group had significantly elevated levels of
Caspase1 and IL1β expression, while the expression levels of
pro-Caspase1 were decreased in the old group (Fig. 5). In addition, the expression of
pro-IL1β was not different between the two groups (Fig. 5).
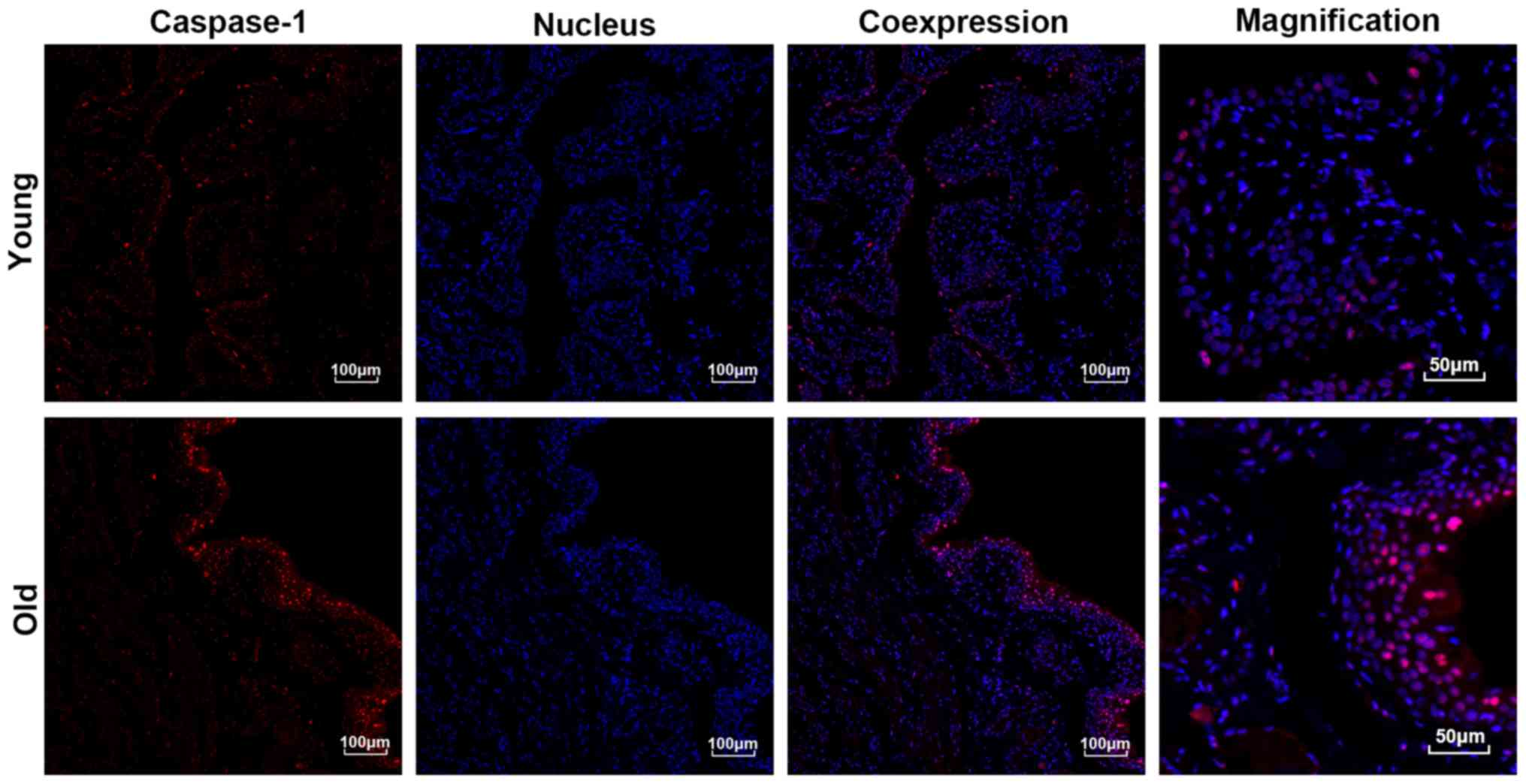
Expression and distribution of
Caspase1
As presented in Fig.
6, the results of the immunofluorescent staining demonstrated
that the expression levels of Caspase1, particularly in the
urothelium, were significantly higher in the bladders of the old
group compared with those in the young group. Moreover, the
expression of active Caspase1 was increased and primarily localized
to the nucleus. In accordance with the results of the
immunofluorescent staining, the western blotting also indicated
that the expression levels of Caspase1 were significantly increased
in the bladders of the old group compared with the young group
(Fig. 5).
Discussion
Aging mediates the progressive degenerative process
closely associated with low-level inflammation and exacerbates the
degenerative alterations that naturally occur. A previous study
documented that chronic inflammation serves a crucial role in
degenerative disorders in elderly patients (16). However, the exact mechanism behind
the accelerated aging process of the bladder has rarely been
studied. The aim of the present study was to examine the hypothesis
that inflamm-aging contributes to bladder dysfunction. In young
bodies, the immune response is rapid and transient, but aging
bodies fail to respond in time, leading to chronic low-grade
inflammation. NLRP3 is associated with chronic low-grade
inflammation leading to functional decline in the process of aging
(17). In addition, the bladder is
a metabolically active organ and is chronically exposed to the
toxic environment of urine, which readily induces bladder
inflamm-aging. The present findings indicated that there is an
association between the physiology of aging bladders and functional
alterations in the NLRP3 inflammasome. Therefore, the present study
focused on the molecular and morphological alterations associated
with bladder dysfunction, in order to elucidate the effect of
inflammation on aging of the bladder. The present study is the
first, to the best of our knowledge, to demonstrate a crucial
association between inflamm-aging and the aging process of
bladder.
The present study demonstrated that the levels of
serum MDA increased and those of antioxidative SOD decreased with
advancing age. These results are consistent with those of a
previous study, which reported that peroxidative injury is
associated with the aging process (18). A previous report indicated that the
generation of reactive oxygen species (ROS) is a crucial element in
the activation of the NLRP3 inflammasome (19). A previous study suggested that
atherosclerosis induces injury to larger vessels, which leads to
chronic bladder ischemia that produces ROS (20). However, in the present study, it
was observed that the rats without raised blood lipids (data not
shown) exhibited morphological alterations in the small vessels of
the bladder submucosa, which may also cause bladder ischemia and
hypoxia. This may induce oxidative injury that results in aging
bladder dysfunction, from hyperactivity to underactivity (21,22).
Subsequently, the expression of IL1β was detected in
the bladder tissues. The results indicated that the levels of IL1β
were distinctly increased in aged bladders compared with young
ones. Furthermore, Masson's trichrome staining demonstrated that
there was a mass of collagen fibers in the submucosa and detrusor
layer in the bladders of the old group, which is a typical feature
of age-induced bladder degeneration.
In a previous study, inflammasome activation was
exclusive to immune cells such as macrophages, but recent research
has confirmed that IL1β is secreted from the lung epithelium
(23) and renal tubular epithelium
(24). Compared with the majority
of other types of epithelial cells, the urothelium may be a source
of IL1β. In the present study, it was verified that IL1β comes from
the urothelium via immunohistochemical staining. Notably, this is
in accordance with the idea that IL1β infiltrates the laminar and
detrusor layer, where it triggers collagen deposition in BOO
(25). Thus, older bladders become
more rigid. Another previous study demonstrated that IL1β causes
denervation in the bladder to induce an overactive bladder and
mediate the detrusor-impaired contractility responsible for LUTS
(26). The NLRP3 inflammasome,
which is upstream of IL1β, is located in the urothelium and is not
expressed in the other layers of the bladder (27). Although this phenomenon has been
identified in BOO, the results of the present study demonstrated
that IL1β also has an impact on bladder dysfunction in the absence
of BOO (Fig. 7).
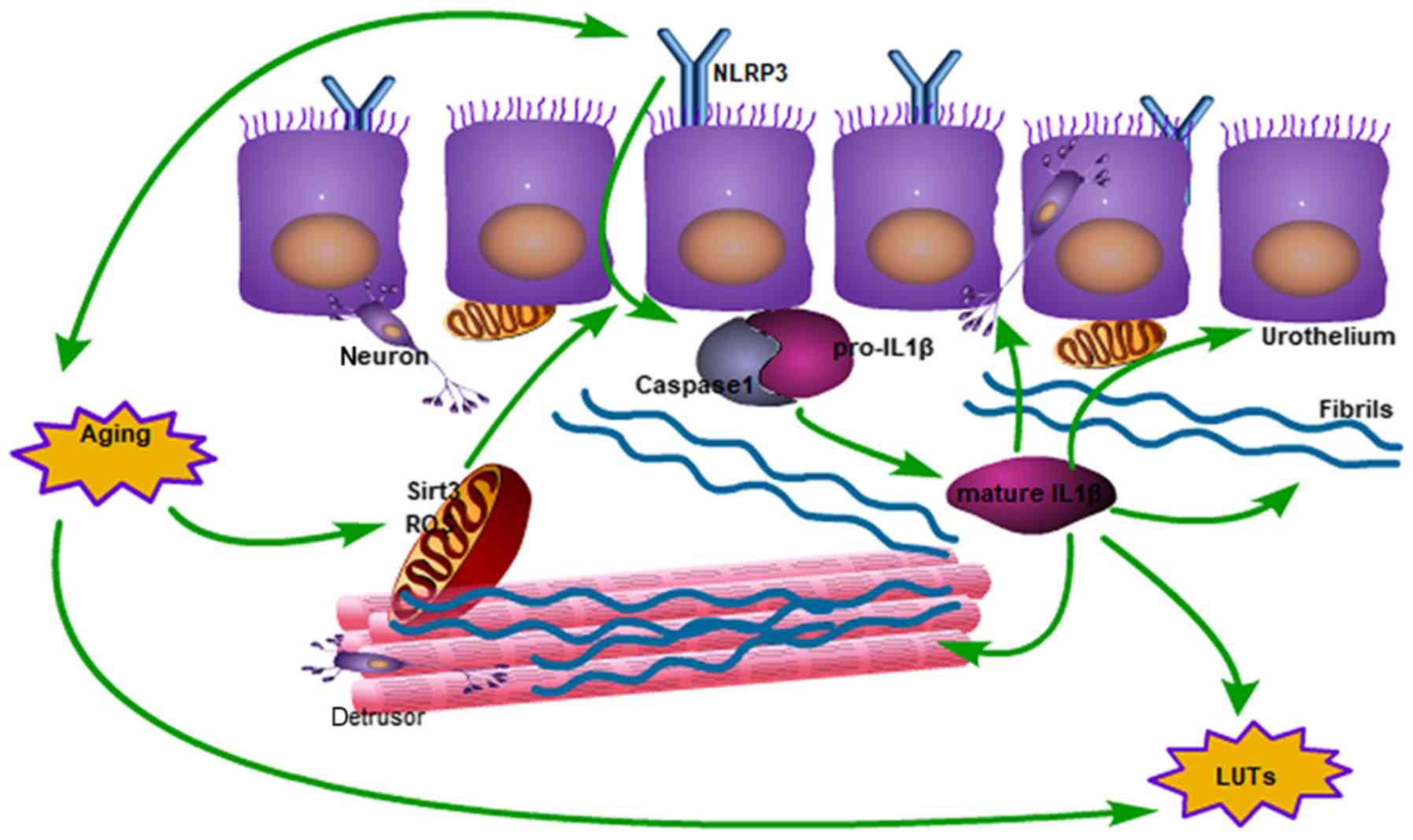 | Figure 7.Schematic diagram of the impact of
aging on bladder function degeneration, from mitochondrial damage
to activated IL1β. In the mitochondria, the expression of SIRT3
decreases and ROS increases, which results in activation of the
inflammasome. Subsequently, IL1β induces collagen deposition and
neurological disorders, which interacts with the aging process of
the bladder. ROS, reactive oxygen species; LUTs, lower urinary
tract symptoms; IL1β, interleukin 1β; SIRT3, NAD-dependent protein
deacetylase sirtuin-3, mitochondrial; NLRP3, NACHT, LRR and PYD
domains-containing protein 3. |
The present study evaluated the state of the older
bladders using western blotting and immunohistochemical staining
for p21, which was localized to the urothelium and muscle layer.
The difference between the young and old groups principally existed
in the urothelium rather than the detrusor, which may mean that the
urothelium is more prone to aging compared with the detrusor. In
other types of cells, p21 serves an important role in age-imposed
tissue degeneration (28). It is
thought that p21 may be used as a predictor of a senescent
bladder.
Activated NLRP3, as a pathogen sensor, is able to
cleave and promote Caspase1, which facilitates the processing of
pro-IL1β to mature IL1β (17). The
results of the present study demonstrated that Caspase1 serves a
crucial role in the urothelium. This is consistent with previous
research, which reported that NLRP3 is located in the urothelium
and is not expressed in other layers of the bladder (27). Furthermore, the urothelium
maintains a strong defensive barrier and serves a crucial role in
sensory nerve function, but this defense system is susceptible to
weakening with advancing age (29). The urothelium is exposed to the
toxic environment of urine, and it is vulnerable to aberrant
urothelial differentiation and a decline in urothelial function.
Daly et al (30)
demonstrated that degeneration of the bladder with age is due to
altered purinergic signaling in the urothelium, and afferent
neurons are affected. However, another study reported that IL1β
damages the afferent and efferent neurons that mediate detrusor
contraction due to urothelial damage and has a direct apoptotic
effect on neurons (31). The
present study demonstrated that IL1β expression is upregulated in
older bladders and is primarily located in the urothelium, which
demonstrates the crucial role of IL1β in the senescence process of
the bladder.
In the present study, it was observed that the MBC
and RV were increased in the old group compared with the young
group, suggesting that the bladder function had degenerated, which
was similar to the results of a previous study (4). However, catheter implantation may
destroy the integrity of the bladder, which decreases bladder
capacity and weakens contractility. This may explain the
differences in the maximum capacity and voiding pressure. In order
to make adjustments for a number of the urodynamic parameters,
bladder weight was taken into account when significantly different
changes in the MBC were evaluated. However, the results indicated
that there was no significant difference in the MBC to bladder
weight ratio between the young group and old group. Bladder
capacity may increase with bladder weight and collagen deposition,
which does not rapidly occur in SD female rats before 24 months of
age (4). The present findings
demonstrated that the VT was prolonged in old female rats without
BOO, which indicated that the bladder contractility is weakened in
old rats compared with young rats. In addition, unstable
contraction was not observed, although this has been reported in
male rats (32). This difference
may be explained by the fact that male rats have greater urethral
resistance compared with females.
SIRT3, which acts upstream of NLRP3, is located in
the mitochondria and regulates ROS generation, which serves an
important role in weakening the NLRP3 inflammasome (8). Notably, a study demonstrated that
SIRT3 is involved in longevity and neurodegeneration (33). The present study is the first, to
the best of our knowledge, to confirm that SIRT3 expression is
reduced in older bladders compared with young bladders. Brown et
al (34) reported that
aging-associated degeneration may be reversed via the SIRT3
pathway. Therefore, the association of SIRT3 with aging of the
bladder requires further investigation.
In conclusion, IL1β is triggered by cleaved Caspase1
in the process of bladder aging and appears to serve a crucial role
in the pathological alterations to the urothelium and detrusor,
which may be an important risk factor for LUTS. Taken together, the
present findings suggest a novel mechanism underlying the
alterations in aging bladders via the NLRP3 inflammasome. NLRP3
inhibitors may become a novel therapy for bladder dysfunction
triggered by inflamm-aging. However, a safe and effective NLRP3
inhibitor has not yet been produced to treat LUTS, and this may be
a focus of future studies.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant no. 81500577), the
Research Projects of Chengdu Science and Technology (grant no.
2015HM0100580SF), the Research Projects of the Department of
Science and Technology of Sichuan Province (grant no. 2017JY0097
and 19YYJC1427), the Research Projects of the Department of
Education of Sichuan Province (grant nos. 16ZA0383 and 16ZA0388),
the Research Projects of the Health and Family Planning Commission
of Chengdu (grant no. 2015123), and the Research Projects of
Sichuan Province Medical Association (grant no. S17002).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JY and LC were responsible for protocol/project
development. PH and LC performed the manuscript writing and data
analysis. YY and KW performed data collection. BA, AS, YZ, ZW, SX
and XL analyzed and interpreted the data, and edited the
manuscript, revising it critically for important intellectual
content.
Ethics approval and consent to
participate
The animal experiments were approved by the Ethics
Committee of Chengdu University (Chengdu, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Vahabi B, Wagg AS, Rosier PFWM, Rademakers
KLJ, Denys MA, Pontari M, Lovick T, Valentini FA, Nelson PP,
Andersson KE and Fry CH: Can we define and characterize the aging
lower urinary tract?-ICI-RS 2015. Neurourol Urodyn. 36:854–858.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Suskind AM: The aging overactive bladder:
A review of aging-related changes from the brain to the bladder.
Curr Bladder Dysfunct Rep. 12:42–47. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Oelke M, Baard J, Wijkstra H, de la
Rosette JJ, Jonas U and Hofner K: Age and bladder outlet
obstruction are independently associated with detrusor overactivity
in patients with benign prostatic hyperplasia. Eur Urol.
54:419–426. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhao W, Aboushwareb T, Turner C, Mathis C,
Bennett C, Sonntag WE, Andersson KE and Christ G: Impaired bladder
function in aging male rats. J Urol. 184:378–385. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lluel P, Palea S, Barras M, Grandadam F,
Heudes D, Bruneval P, Corman B and Martin DJ: Functional and
morphological modifications of the urinary bladder in aging female
rats. Am J Physiol Regul Integr Comp Physiol. 278:R964–R972. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Siroky MB: The aging bladder. Rev Urol.
1:S3–S7. 2004.
|
|
7
|
Baylis D, Bartlett DB, Patel HP and
Roberts HC: Understanding how we age: Insights into inflammaging.
Longev Healthspan. 2:82013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen ML, Zhu XH, Ran L, Lang HD, Yi L and
Mi MT: Trimethylamine-N-Oxide induces vascular inflammation by
activating the NLRP3 inflammasome through the SIRT3-SOD2-mtROS
signaling pathway. J Am Heart Assoc. 6:e0063472017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xing SS, Li J, Chen L, Yang YF, He PL, Li
J and Yang J: Salidroside attenuates endothelial cellular
senescence via decreasing the expression of inflammatory cytokines
and increasing the expression of SIRT3. Mech Ageing Dev. 175:1–6.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Birder L and Andersson KE: Urothelial
signaling. Physiol Rev. 93:653–680. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Roosen A, Chapple CR, Dmochowski RR,
Fowler CJ, Gratzke C, Roehrborn CG, Stief CG and Andersson KE: A
refocus on the bladder as the originator of storage lower urinary
tract symptoms: A systematic review of the latest literature. Eur
Urol. 56:810–819. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Birder LA, Ruggieri M, Takeda M, van
Koeveringe G, Veltkamp S, Korstanje C, Parsons B and Fry CH: How
does the urothelium affect bladder function in health and disease?
ICI-RS 2011. Neurourol Urodyn. 31:293–299. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Inouye BM, Hughes FM Jr, Sexton SJ and
Purves JT: The emerging role of inflammasomes as central mediators
in inflammatory bladder pathology. Curr Urol. 11:57–72. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
van Deursen JM: The role of senescent
cells in ageing. Nature. 509:439–446. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hudgins AD, Tazearslan C, Tare A, Zhu Y,
Huffman D and Suh Y: Age- and Tissue-Specific Expression of
Senescence Biomarkers in Mice. Front Genet. 9:592018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Howcroft TK, Campisi J, Louis GB, Smith
MT, Wise B, Wyss-Coray T, Augustine AD, McElhaney JE, Kohanski R
and Sierra F: The role of inflammation in age-related disease.
Aging (Albany NY). 5:84–93. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Youm YH, Grant RW, McCabe LR, Albarado DC,
Nguyen KY, Ravussin A, Pistell P, Newman S Carter R, Laque A, et
al: Canonical Nlrp3 inflammasome links systemic low-grade
inflammation to functional decline in aging. Cell Metab.
18:519–532. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Inal ME, Kanbak G and Sunal E: Antioxidant
enzyme activities and malondialdehyde levels related to aging. Clin
Chim Acta. 305:75–80. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tschopp J and Schroder K: NLRP3
inflammasome activation: The convergence of multiple signalling
pathways on ROS production? Nat Rev Immunol. 10:210–215. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Andersson KE, Boedtkjer DB and Forman A:
The link between vascular dysfunction, bladder ischemia, and aging
bladder dysfunction. Ther Adv Urol. 9:11–27. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nomiya M, Yamaguchi O, Akaihata H, Hata J,
Sawada N, Kojima Y and Andersson KE: Progressive vascular damage
may lead to bladder underactivity in rats. J Urol. 191:1462–1469.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sunagawa M, Wolf-Johnston A, Nomiya M,
Sawada N, Andersson KE, Hisamitsu T and Birder LA: Urinary bladder
mucosal responses to ischemia. World J Urol. 33:275–280. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Peeters PM, Perkins TN, Wouters EF,
Mossman BT and Reynaert NL: Silica induces NLRP3 inflammasome
activation in human lung epithelial cells. Part Fibre Toxicol.
10:32013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bakker PJ, Butter LM, Claessen N, Teske
GJ, Sutterwala FS, Florquin S and Leemans JC: A tissue-specific
role for Nlrp3 in tubular epithelial repair after renal
ischemia/reperfusion. Am J Pathol. 184:2013–2022. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hughes FM Jr, Sexton SJ, Jin H, Govada V
and Purves JT: Bladder fibrosis during outlet obstruction is
triggered through the NLRP3 inflammasome and the production of
IL-1beta. Am J Physiol Renal Physiol. 313:F603–F610. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lutolf R, Hughes FM Jr, Inouye BM, Jin H,
McMains JC, Pak ES, Hannan JL and Purves JT: NLRP3/IL-1beta
mediates denervation during bladder outlet obstruction in rats.
Neuroural Urodyn. 37:952–959. 2018. View Article : Google Scholar
|
|
27
|
Hughes FM Jr, Hill HM, Wood CM, Edmondson
AT, Dumas A, Foo WC, Oelsen JM, Rac G and Purves JT: The NLRP3
Inflammasome mediates inflammation produced by bladder outlet
obstruction. J Urol. 195:1598–1605. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li J, Han S, Cousin W and Conboy IM:
Age-specific functional epigenetic changes in P21 and p16 in
injury-activated satellite cells. Stem Cells. 33:951–961. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Perse M, Injac R and Erman A: Oxidative
status and lipofuscin accumulation in urothelial cells of bladder
in aging mice. PloS One. 8:e596382013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Daly DM, Nocchi L, Liaskos M, McKay NG,
Chapple C and Grundy D: Age-related changes in afferent pathways
and urothelial function in the male mouse bladder. J Physiol.
592:537–549. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
He P: Lutolf R, Hughes FM, Jr., Inouye BM,
Jin H, McMains JC, Pak ES, Hannan JL, Purves JT. NLRP3/IL-1beta
mediates denervation during bladder outlet obstruction in rats.
Neurourology and urodynamics 2017. Neurourol Urodyn. 37:15062018.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kohan AD, Danziger M, Vaughan ED Jr and
Felsen D: Effect of aging on bladder function and the response to
outlet obstruction in female rats. Urol Res. 28:33–37. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Haigis MC and Sinclair DA: Mammalian
sirtuins: Biological insights and disease relevance. Annu Rev
Pathol. 5:253–295. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Brown K, Xie S, Qiu X, Mohrin M, Shin J,
Liu Y, Zhang D, Scadden DT and Chen D: SIRT3 reverses
aging-associated degeneration. Cell Rep. 3:319–327. 2013.
View Article : Google Scholar : PubMed/NCBI
|