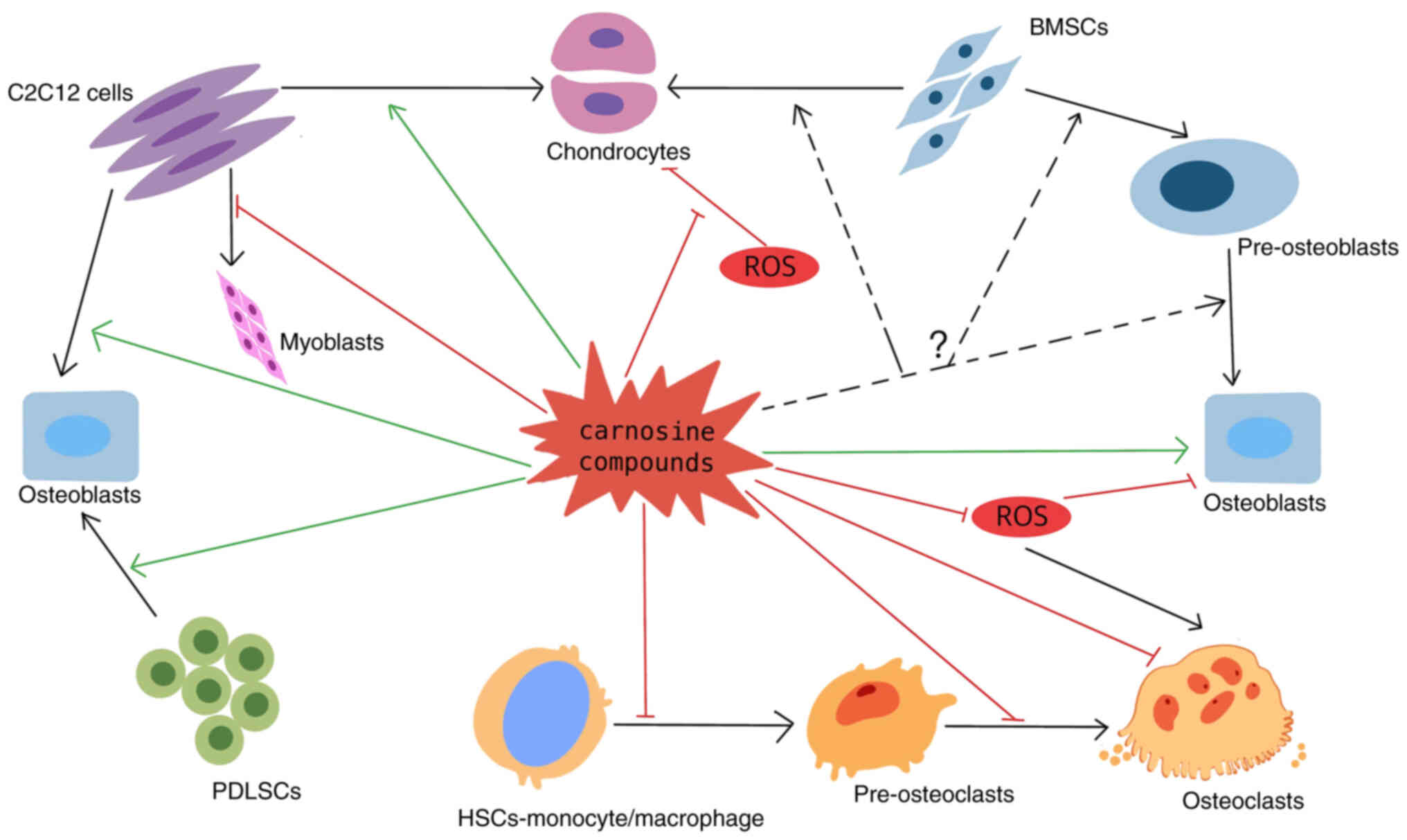
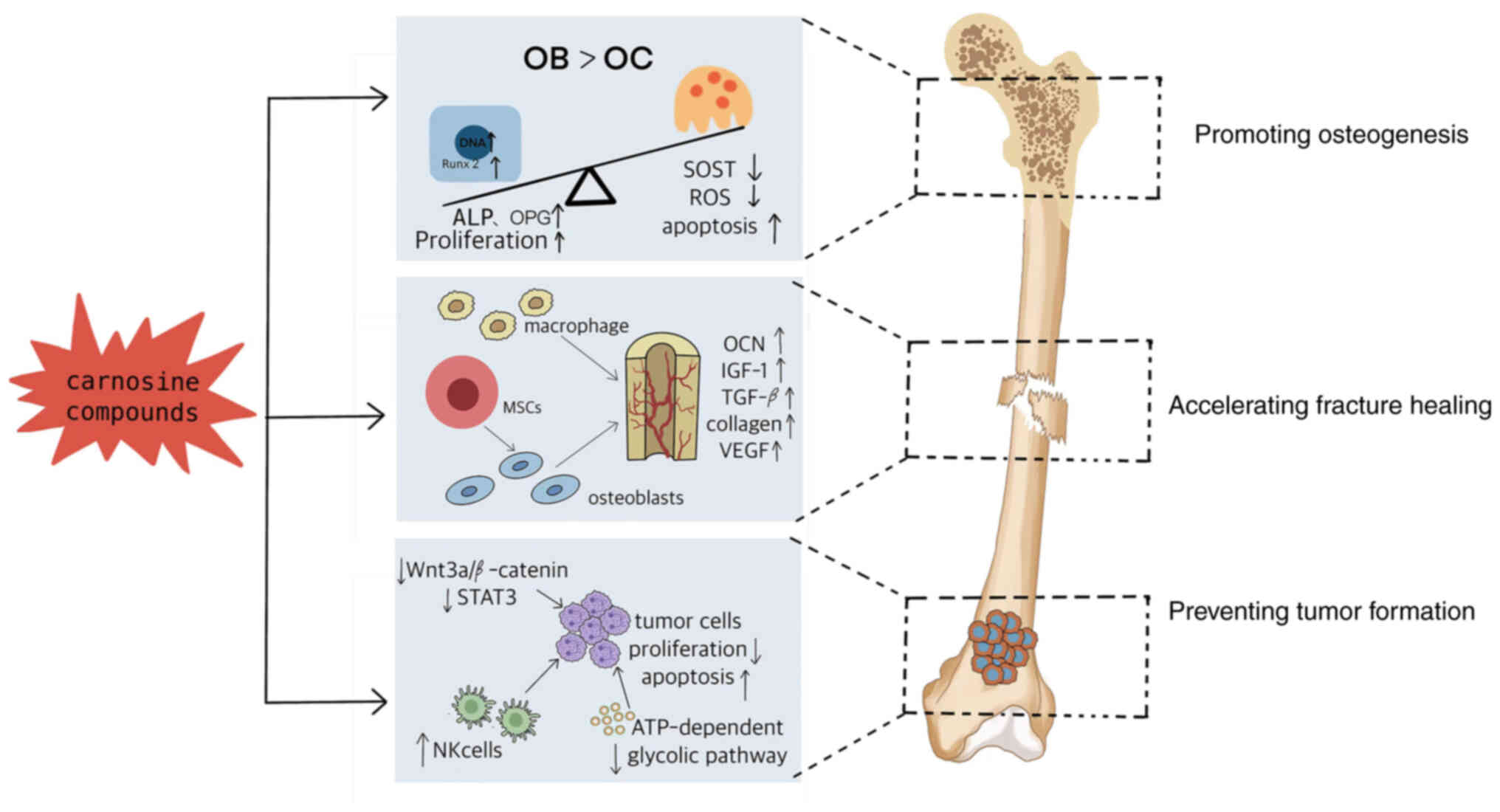
Introduction
Bone homeostasis is a dynamic lifelong process
involving all body systems. Aging, hypogonadism, inflammation,
malnutrition and alcohol abuse are some of the known risk factors
for osteoporosis or other bone-related diseases. These risk factors
cause an imbalance of osteoclastic bone resorption and osteoblastic
bone formation, leading to abnormal bone mass (1–3). A
recent meta-analysis showed that the commonest bone
metabolism-related disease is primary osteoporosis, which includes
postmenopausal and senile osteoporosis (4).
Carnosine is a water-soluble, small-molecule
dipeptide with anti-inflammatory, antioxidant, antiaging and
antitumor functions (5). In
vitro and in vivo studies suggest that carnosine could
promote the synthesis of new bone proteins by osteoblasts, increase
alkaline phosphatase activity, induce differentiation of
pluripotent mesenchymal stem cells into osteoblasts and
chondrocytes, not myoblasts, inhibit bone resorption activity by
osteoclasts without reducing their numbers and enhance protein
formation in fracture healing (6,7).
Carnosine could also reduce the damage of deleterious factors on
the bone microenvironment and maintain local and systemic bone
metabolism (8).
Therefore, in vivo carnosine and carnosine
supplements are indispensable for bone homeostasis. They may be an
effective method to prevent and improve multifactorial bone
metabolic disorders such as those caused by aging or oxidative
stress. The present review focused on the physiological role of
carnosine in bone metabolism and its potential therapeutic use in
different bone-related diseases (9,10).
Carnosine and its compounds
Carnosine is a natural bioactive dipeptide abundant
in skeletal muscles and the nervous system consisting of β-alanine
and L-histidine, under the co-catalysis of carnosine synthase and
ATP, which synthesize a variety of carnosine-related compounds such
as homocarnosine and acetyl carnosine. Carnosine chelates zinc ions
in a way that is not toxic to the human body, performing a positive
effect on bone metabolism (11),
and the bioactivity of zinc carnosine (ZnC) is stronger than that
of zinc sulfate or other carnosine chelates such as
N-acetyl-β-alanyl-L-histidine, and its mobilization is reactive
oxygen species (ROS)-mediated (12,13). In addition, due to the unique
binding characteristics of metals and hydroxyapatite in bone
tissue, the zinc-chelating carnosine is absorbed easily in the
intestinal tract and carnosine enhances the bioavailability of zinc
ions by raising its intracellular uptake in bone tissue, which
stimulates bone cells and collagen synthesis (14,15). A number of experimental studies
found that the alanyl group in ZnC improved the activity,
mineralization, enzyme activity and nucleic acid metabolism of
osteoblasts, and the activity and concentration of alkaline
phosphatase (ALP) significantly increased in a time-dependent
manner in culture media containing zinc carnosine (11).
Accumulated evidence shows the anti-oxidative stress
and anti-inflammation effects of carnosine. On the one hand, it can
enhance the scavenging and phagocytic activity of M1 macrophage
(9), increase the expression of
TGF-β, activate nuclear factor (erythroid-derived 2)-like 2 (Nrf2)
signaling pathway and scavenge ROS (16); On the other hand, carnosine
reduces the oxidative stress reaction by suppressing the release of
inflammatory cytokines IL-1β and IL-6, TNF-α and inhibiting NF-κB
in inhibiting the expression of inflammatory mediators NO and
inducible nitric oxide synthase (iNOS) in
lipopolysaccharide-induced RAW264.7 macrophages (17). Carnosine combines with advanced
glycation end products (AGEs) and 4-hydroxynonenal (4-HNE) to
reduce the expression of lipid and protein peroxidation (18). In addition, carnosine can also
promote wound healing and chelate heavy metal ions (Zn2+
and Cu2+), thus decreasing intracellular metal-induced
toxicity (19), as well as
reducing lactic acid accumulation and buffering the pH values of
muscles, thus increasing muscle endurance and alleviating fatigue
(20).
ZnC also known as polaprezinc, or PepZin GI, is a
new zinc peptide and the first zinc-related drug approved in Japan
(21). Current clinical evidence
confirms that ZnC and carnosine can both prevent diseases
characterized by oxidative stress and/or neurodegenerative changes,
such as diabetes and its concurrent kidney and neuropathy,
depression, cerebral ischemia, Alzheimer's disease (AD), peptic
ulcer and Helicobacter pylori-related gastritis (22). Studies have also shown a
significant therapeutic effect of the non-hydrolyzed form of
L-carnosine on influenza virus-induced pneumonia via NO and
cytokine regulation (3,23). In addition, carnosine can also be
used as an over-the counter food supplement to improve muscle
tolerance or as a major component of cosmetics to exert its
antioxidant and anti-aging effects (24). Although a large number of
preclinical studies have been conducted in vivo and ZnC has
been authorized by the FDA (6),
the safety and efficacy of carnosine in bone metabolism-related
diseases needs further verification.
Cell-specific effects of carnosine compounds
on bone
Osteoblasts
Carnosine and its compounds serve critical roles in
bone metabolism and mineralization. First, a series of short-term
MC3T3-El cell cultures showed that ZnC could markedly increase the
expression of target genes such as runt-related protein 2 (Runx2)
and osterix, promote the DNA synthesis of osteoblasts and
simultaneously increase the collagen and calcium content in the
matrix based on the newly synthesized bone proteins (25). This indicates that ZnC might be
involved in nuclear transcription and protein translation. After a
long-term MC3T3-El cell culture, ALP, a biochemical marker of
osteoblast differentiation, increased significantly and its
expression and activity changed in a time- and dose-dependent
manner, which indicates that ZnC contributes to the proliferation,
differentiation of osteoblasts (26). Second, bone resorption stimulating
factors such as parathyroid hormone (PTH), IL-1 and prostaglandin
E2 (PGE2) were shown to be effective inhibitors of bone formation
by osteoblasts (27). ZnC
(10−6−10−4 M) did not completely inhibit the
effect of these bone resorption stimulators, but they could block
the reduction of acid phosphatase and alkaline phosphatase
activities and prevent intracellular glucose depletion and lactic
acid accumulation (28).
At the molecular level, carnosine and its compounds
at the same dose (10−6−10−4 M), could rescue
the transcription of osteoblasts by decreasing the inhibition of
NF-κB signaling pathway by ROS or by enhancing the phosphorylation
motifs of cyclic adenosine monophosphate (cAMP)/protein kinase A
(PKA) (29); carnosine increases
the expression of osteoblast-specific transcription factor
Runx2/core-binding factor α1 (Cbfa1) through active TGF-β/bone
morphogenetic proteins (BMPs) and insulin-like growth factor 1
(IGF-1)/protein kinase C (PKC)/mitogen-activated protein kinases
(MAPK) transduction signaling pathway and upregulates the mRNA
expression levels of target genes osterix, osteocalcin (OCN),
osteoprotegerin (OPG), IGF-1 and TGF-β, finally promoting the
proliferation and differentiation of osteoblasts (6,30).
Autocrine of TGF-β could also stimulate the differentiation of
early osteoblasts, promote the recruitment of osteocytes and the
expression of their matrix protein, regulate the formation of
osteoblast and bone remodeling in coordination with PTH and
regulate coupling in bone formation and bone resorption (31).
In conclusion, study results support the idea that
carnosine compounds could directly and specifically promote the
proliferation and differentiation of osteoblasts in vivo and
in vitro. Probably, the mechanism is partly mediated by
increasing protein kinase and protein phosphatase in osteoblasts,
stimulating newly synthesized bone proteins in osteoblasts,
enhancing the activity of aminoacyl-tRNA synthetase in
zinc-activated translation and partially stimulating bone formation
and calcification (Fig. 1).
However, there are relatively few clues about the early stages of
osteoblast differentiation, which require further research.
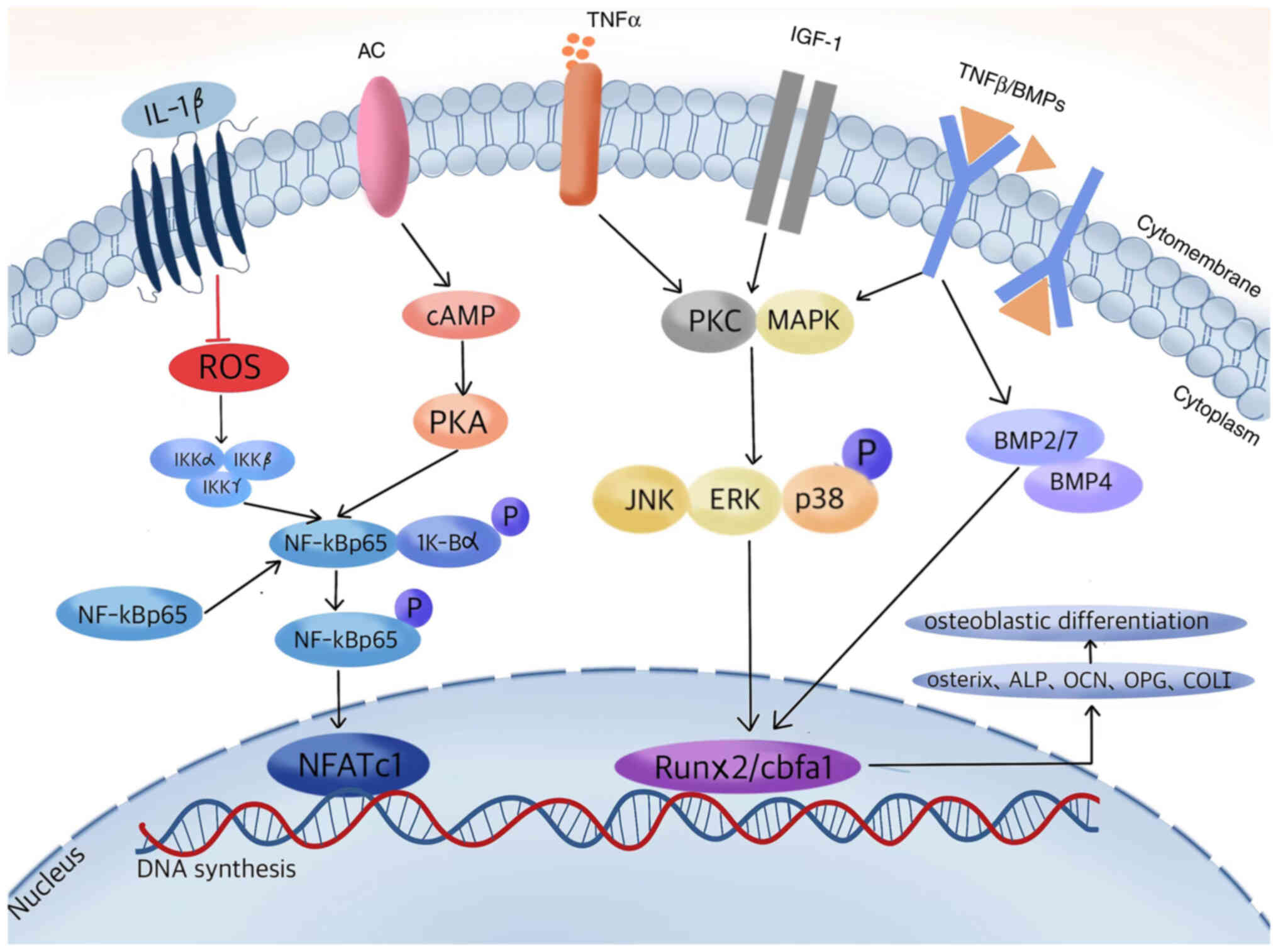 | Figure 1.Proposed signaling transduction
cascades of carnosine compounds in regulating osteoblastic
differentiation. A series of carnosine-regulated related cytokines
and molecules interact within osteoblasts to promote protein kinase
phosphorylation and DNA synthesis, increasing the expression of
downstream target genes and bone proteins, thereby stimulating bone
formation. AC, adenylyl cyclases; IGF-1, insulin-like growth factor
1; BMP, bone morphogenetic protein; ROS, reactive oxygen species;
NFATc1, nuclear factor of activated T cells 1; cAMP, cyclic
adenosine monophosphate; PKA, protein kinase A; PKC, protein kinase
C; Runx2, runt-related protein 2; cbfa1, core-binding factor α1;
ALP, alkaline phosphatase; OCN, osteocalcin; OPG, osteoprotegerin;
COL I, collagen I. |
Osteoclastogenesis
Osteoclasts are multinucleated cells originating in
mature monocyte-/macrophage-lineage cells and needed for bone
growth and remodeling, maintenance of the bone structure and bone
calcium metabolism throughout their life cycle. A number of bone
resorptive factors can stimulate bone marrow macrophages (BMMs) to
differentiate into osteoclasts, concomitant with an evident
increase in the number of osteoclast-like cells and the decrease of
bone calcium content. Under pro-inflammatory conditions, the uptake
of carnosine by macrophages is markedly increased (32). A dose of 20 mM carnosine can
reduce the release of pro-inflammatory cytokines and exert its
antioxidant effect by changing the balance and polarization state
of macrophage M1/M2 (33).
However, further studies are required to elucidate whether
carnosine serves a protected role in abnormal bone loss by
regulating the ‘liquid’ state of macrophage polarization.
Unlike osteoblasts, ZnC
(10−6−10−4 M) directly inhibit the early
stage of BMMs differentiation into osteoclast precursors, block the
formation of pre-osteoclasts and its receptor activation of NK-κB
(RANK) expression and suppresses osteoclastogenesis without
obviously inhibiting the function of osteoclasts (34). An experiment with mature
osteoclasts showed that ZnC (10−5 M) could inhibit TNF-α
production by osteoclasts and attenuate its stimulating effect on
osteoclast differentiation. The results indicated that the
inhibitory effect of ZnC on PTH-induced osteoclast cell formation
was through PKC-mediated protein kinase activation (28,35). An in vitro study with
cultured mouse marrow demonstrated that both zinc sulfate
(10−6 M) and zinc-chelating dipeptide (10−6
M) could inhibit the TGF-β induced formation of tartrate-resistant
acid phosphatase-positive cells, whereas these inhibitory effects
were abrogated by egtazic acid (EGTA) (36). It further revealed that ZnC could
inhibit the stimulatory effects of PTH on osteoclast-like
multinucleated cell formation of mouse marrow cells, through
Ca2+-calcineurin-nuclear factor of activated T cells 1
(NFATc1) dependent activation of protein kinase C (PKC) (37).
By contrast, another in vitro study
demonstrated that a dose of 50 µM polaprezinc produced the opposite
result, promoting BMMs and RAW264.7 cell differentiation into
osteoclasts by enhancing the mRNA expression levels of NFATc1 and
cathepsin K in osteoclasts as well as the transcriptional activity
of yes-associate protein (38).
However, this is in complete contrast to the effect of ZnC on
osteoclasts confirmed in a previous study (34). The authors do not provide further
statement on this point (38), so
whether ZnC exerts dose- or condition-specific effects on
osteoclastogenesis needs to be further studied.
Interaction between osteoblast-lineage
cells and osteoclast-lineage cells
The interactions between osteoblasts and
osteoclasts, as well as their precursors, serve a major role in
determining the balance between bone formation and resorption. In
addition to regulating the differentiation and function of
osteoblast and osteoclast, as aforementioned, carnosine compounds
are also reported to be involved in the interaction between
osteoblast-lineage cells and osteoclast-lineage cells, exhibiting
beneficial effects on preserving bone mass.
An in vitro study confirmed that carnosine
compounds could stimulate osteoblast-lineage cells obtained from
the calvaria of weanling rats (3-week-old males) to secrete decoy
receptor OPG and block the receptor activator of NF-κB ligand
(RANKL)-RANK interaction between osteoclast precursors and
osteoclasts by upregulating the OPG/RANKL ratio (13). This effectively inhibits the
formation, differentiation and apoptosis of osteoclasts and
stabilizes bone mass. In addition, RANKL secreted by mature bone
cells is involved in a series of cascade reactions of
osteoclastogenesis and bone resorption (21). It has been clarified that when
RANK is activated, it leads to phosphorylation of P65 and its
upstream inhibitor protein IκB, increased recruitment of TNF
receptor associated factor 6 and nuclear translocation,
subsequently promoting osteoclastogenesis (39). However, carnosine compounds
negatively regulate the osteoclast process by inhibiting ROS
production during this process (Fig.
2).
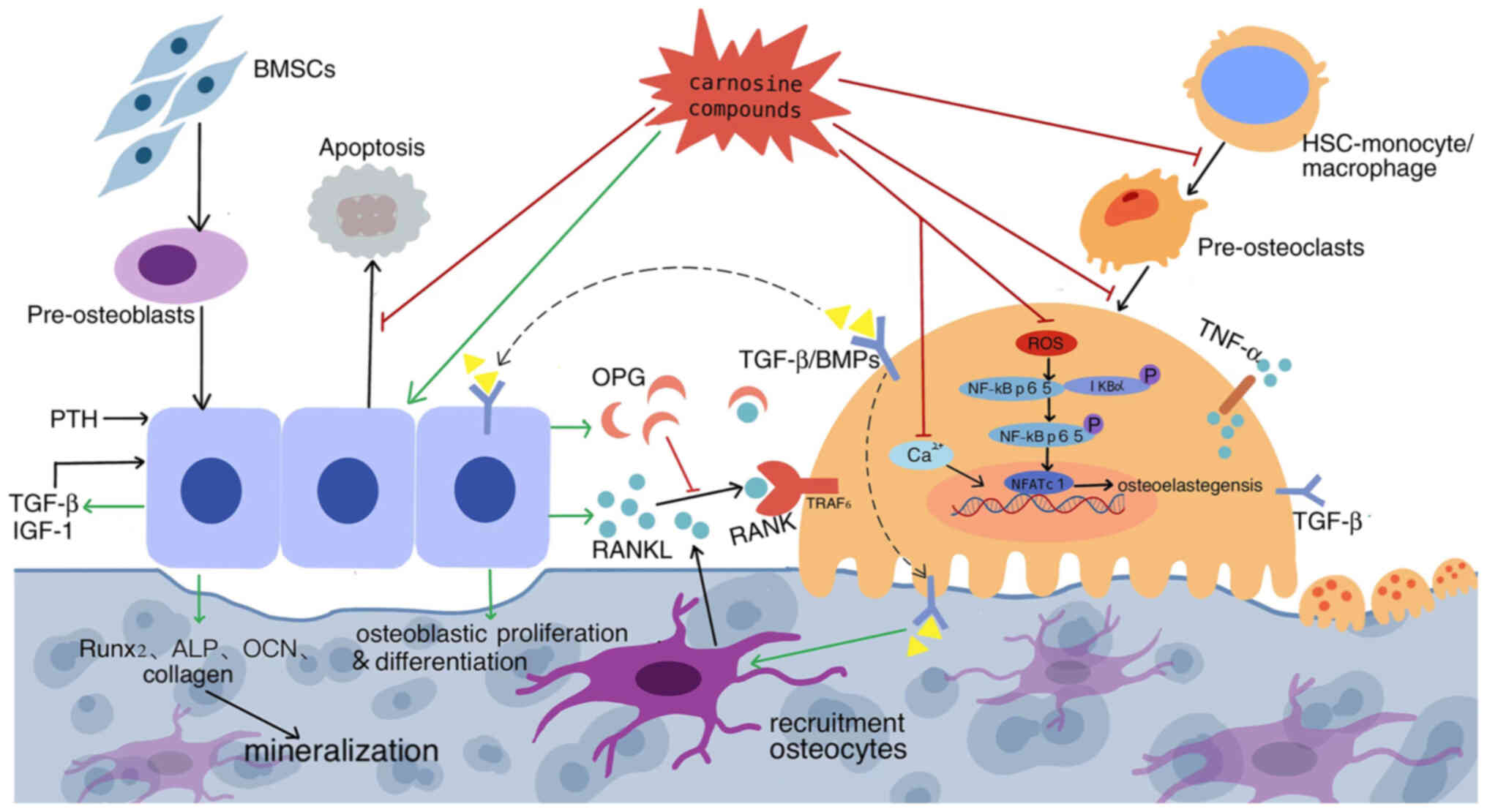 | Figure 2.Dual regulation of carnosine
compounds on cellular interactions between osteoblast lineage and
osteoclast linage cells. Carnosine stimulates osteoblast
proliferation and differentiation, inhibits apoptosis, activates
bone progenitor cell enrichment, whereas inhibiting BMM
differentiation and pre-osteoclast migration to reduce
osteoclastogenesis and through regulating the RANKL-RANK ratio,
thus effectively inhibiting the formation, differentiation and
apoptosis of osteoclasts and stabilizes bone mass. Its promoting
effect is marked with green arrows and inhibiting effect with red.
Dotted line indicate a proposed rather than confirmed effect. BMM,
bone marrow macrophage; RANKL, receptor activator of NF-κB ligand;
RANK, receptor activation of NK-κB; BMSCs, bone marrow mesenchymal
stem cells; PTH, parathyroid hormone; IGF-1, insulin-like growth
factor 1; OPG, osteoprotegerin; BMP, bone morphogenetic protein;
HSCs, hematopoietic stem cells; NFATc1, nuclear factor of activated
T cells 1. |
Consequently, under different physiological and
pathological conditions, carnosine compounds at the dose of
10−6−10−4 M could promote bone marrow
mesenchymal stem cells (BMSCs) to differentiate into osteoblasts,
stimulate osteoblast activity and activate osteoprogenitor
enrichment, inhibit BMMs differentiation into osteoclasts and
pre-osteoclast migration, reduce osteoclast formation and directly
or indirectly affect bone resorption of osteoclasts through cell
communication between osteoblasts/stromal cells and osteoclasts
progenitor cells. While the recently published study (38) drives debate on their effects on
osteoclastogenesis, more studies are needed to clarify this
point.
Mesenchymal stem cells
Mesenchymal stem cells (MSCs) are a subset of cells
derived from different tissues. BMSCs and periodontal ligament stem
cells (PDLSCs) are involved in bone formation and regeneration.
MSCs secrete cytokines through a paracrine and immune response.
These cytokines are used in the treatment of a number of diseases
(39).
Maeno et al (6) confirmed that carnosine compounds
stimulate PDLSCs to produce more BMPs, including BMP-2, BMP-4 and
BMP-7. This promotes the differentiation of MSCs into osteoblasts
and/or chondrocytes. PDLSCs, derived from teeth, have been proven
to differentiate into osteoblasts or fibroblasts. They maintain
periodontal tissue by regulating bone formation and bone resorption
and have stronger proliferative and anti-apoptotic abilities than
BMSCs (40). Carnosine compounds
also mediate the mesenchymal differentiation of PDLSCs into
osteoblasts through an autocrine process. The expression of the
osteogenic differentiation-related transcription factor RUNX2/Cbfa1
and the activity of ALP in PDLSCs were increased in a
time-dependent manner with carnosine (6).
In vitro studies by Takada et al
(41) showed that carnosine
compounds could increase the expressions of Runx2/Cbfa1,
transcription factor SOX9 and type X collagen (Col X, the marker of
complete differentiation of chondrocytes) by mediating cartilage
differentiation in C2C12 cells of mesenchymal myoblasts (42). By contrast, the expression of
myogenic-related markers MyoD and desmin decreased significantly.
This indicates that carnosine induces C2C12 myogenic stem cells to
differentiate into osteoblasts and/or chondrocytes and inhibits
their transformation into myoblasts (Fig. 3).
Mechanism and the potential effect of
carnosine compounds on bone-related diseases
Osteoporosis
Postmenopausal osteoporosis
Osteoporosis is the most common type of systemic
degenerative bone disease. It emerges at all ages but it is
predominantly diagnosed in postmenopausal women and older men.
Postmenopausal osteoporosis is characterized by a progressive
decrease in bone mineral density (BMD), deterioration of bone
microarchitecture and an imbalance of bone formation and resorption
regulated by osteoblasts and osteoclasts, respectively. This is
accompanied by the accumulation of bone marrow fat resulting in
compromised bone strength and an increased propensity for fragility
fractures (43).
Postmenopausal osteoporosis may be based on the
interaction of local cellular and molecular factors, including the
deficiency of estrogen and the change of immune status of
postmenopausal women. For example, T lymphocyte subtypes express
TNFα, which increases the apoptosis of osteoblasts and indirectly
stimulates the generation of osteoblasts through RANKL produced by
B cells, as well as the changes of chronic inflammation phenotypes
and cytokine expression (44).
Excessive accumulation of AGEs and age-related obesity lead to an
increased bone turnover rate and decreased bone trabecula and
cortical bone density, resulting in continuous bone destruction
(31). Studies refer to ‘the
three-way regulation’ model of bone metabolism, which consists of
the imbalance of regulation of the coupling of bone vessels,
osteoclasts and osteogenesis, which leads to a decrease in cortical
thickness and an increase in cortical porosity (45,46). The histomorphology and cytokines
related to bone metabolism in ovariectomized rats confirmed that
the oral administration of ZnC (10–100 mg/kg/day) for 6 weeks can
significantly increase zinc accumulation, bone protein synthesis
and collagen contents in bone, completely block the bone loss of
femoral trabecula and prevent the deterioration of cortical bone
(47). Comparatively low doses of
ZnC (10 and 30 mg/kg/day) possess the same ameliorating actions
(48). Carnosine compounds have
been proved to inhibit bone resorption in tissue culture, so
reasonably, carnosine may partially inhibit osteoclast resorption.
These results indicate that carnosine compounds have a direct
stimulating effect on bone formation and calcification and
effectively prevent BMD reduction in ovariectomized rats.
From the perspective of systemic nerve-bone axis
regulation, the dysfunction of the autonomic nervous system (made
up of the sympathetic and parasympathetic systems) is also frequent
in elderly or postmenopausal osteoporosis patients (49). Some authors found that under the
conditions of aging or menopause (estrogen deficiency), the
imbalance of sympathetic and parasympathetic nerves in bones
stimulates bone cells to overproduce neuropeptide Y, inhibits
cAMP/PKA/CREB signaling pathway, weakens osteogenic differentiation
and enhances the adipogenic differentiation of BMSC, leading to
bone-lipid imbalance and bone marrow obesity (49,50). Notably, carnosine is hypothesized
to inhibit sympathetic nerve activity and promote parasympathetic
nerve activity (51) and its
effect may be to inhibit sympathetic nerve activity through the
natural killer (NK) cells activity of spleen cells or regulate the
autonomic nervous system through histidine H3 receptor (52,53). In brief, carnosine may provide new
research avenues for postmenopausal and age-related bone metabolism
through neuro-bone axis regulation.
Carnosine compounds are important because they could
exert a broad-spectrum effect on the bone remodeling of osteoblasts
and osteoclasts and inhibit ovariectomy-induced osteoclastogenesis.
They could be a natural and novel treatment for postmenopausal
osteoporosis.
Senile osteoporosis
Aging is another important risk factor for primary
osteoporosis. At the cellular level, an increase in the level of
ROS shortens the life span of osteoblasts and inhibits their
formation, reduces BMSCs differentiation and stimulates the
formation of damaged DNA, proteins and lipids, leading to cell
apoptosis (54). On the other
hand, the levels of RANKL and sclerostin (SOST, an antagonist of
Wnt intracellular signal transduction) in osteoclasts are
diminished (55). RANKL can
stimulate ROS production. Conversely, the formation, activation and
bone-resorbing function of osteoclasts also need ROS stimulation
(56). When the balance between
osteoblasts and osteoclasts is disordered, osteoporosis usually
results.
At the molecular level, aging mainly reduces the
reactivity of insulin and estrogen, which decreases bone formation
and differentiation. Research shows that the level of carnosine
gradually decreases with age (57). Due to carnosine's effect on
glycolysis, it has been used to inhibit cell aging in cultured
human fibroblasts (9). Other
experiments in elderly (10-month-old) female rats found that when
the bone tissues were cultured for 24 h in a medium containing ZnC
(10−5 M), 4.5% glucose, 0.25% bovine serum albumin and
antibiotics, the activity of ALP in osteoblasts was significantly
increased and prolonged, thus lengthening the cell cycle. ZnC could
also activate aminoacyl-tRNA synthetase, a key enzyme in the
process of bone protein biosynthesis and translation, stimulate
bone formation and mineralization and alleviate senile osteoporosis
(47,58).
Disuse osteoporosis
Mounting evidence shows that bone disuse inhibits
bone formation and increases bone resorption. These, coupled with
the low dynamic and insulin-mediated gravity loading effect,
eventually lead to osteoporosis (59).
It has also been observed that weightlessness
damages the diaphyseal part more than the metaphyseal part in the
model of hindlimb suspension in rats (60). Additionally, weightlessness or
long-term physical fixation does not affect the intestinal
absorption of zinc, but it partially inhibits the transfer of serum
zinc to bone tissue. To compensate, zinc ions overflow from the
bone leading to a decrease in zinc level in the bone matrix and
cell components, which inhibits bone formation. Some researchers
have suggested that inflammation from bone disuse could reflect
periarticular osteopenia. This uncoupled state of bone resorption
and formation also leads to periarticular osteoporosis in patients
with rheumatoid arthritis (61).
In vitro, ZnC (10−5 M) in a
culture medium with bone tissue could stimulate the proliferation
and differentiation of stem cells, the production of IGF-1, TGF-β
and osteocalcin and the peptization of aminoacyl-tRNA synthetase in
osteoblasts, thus increasing ALP activity and aminoacyl-tRNA
content of protein in bone tissues. IGF-1 stimulated by carnosine
compounds could both increase bone trabecula formation and the
proliferation of osteoblasts and have an anabolic effect on bone
metabolism with or without bone disuse (62).
Other secondary osteoporosis
Long-term use of glucocorticoids can reduce bone
formation by directly inhibiting the proliferation and
differentiation of osteoblasts (inhibiting Runx2 and Collagen I)
and increasing apoptosis and bone absorption by negative feedback
gonadotropin secretion, or decreasing calcium absorption and
increasing calcium excretion. This eventually leads to a negative
calcium balance in the body, decreased bone density and secondary
development of glucocorticoid-induced osteoporosis (63). A study of hydrocortisone-induced
osteopathy showed that carnosine compounds prevent the increase of
PTH levels and the decrease of ALP activity and increase the DNA
and zinc contents. These effects prevent disordered bone metabolism
and architectural deterioration (64).
In clinical studies of peripheral osteoporosis
caused by autoimmune arthritis, inflammation caused by ROS
oxidative stress is the key cause of osteoporosis (65,66), whereas carnosine compounds block
the decrease of ALP activity induced by IL-1β, attenuate the
decrease of DNA and calcium contents, partly improve the immune
response triggered by the release of matrix degradation products,
inhibit inflammatory osteolysis and the loss of trabecula and
collagen and alleviate the continuous destruction of bone
microstructure and mechanical strength (67).
Vitamin D has extensive endocrine and autocrine
functions that could promote the proliferation of osteoblasts, the
differentiation and activity of osteoclasts, bone formation,
absorption and coupling (68),
which are essential for bone development and maintenance. Some
evidence shows that 1,25 (OH)2D3 is an effective promoter of
osteoblasts-osteocytes transformation including promoting
osteoblasts mineralization and inducing expression of bone cell
markers in pluripotent stem cells (iPSop) in vitro (69). Notably, deficiency of vitamin D or
calcium caused by various factors in the body leads to osteoporosis
(69). A study by Segawa et
al (70) observed that an
oral administration of 100 mg/kg/day of ZnC for 14 days could
partially increase ALP activity and calcium contents, which
directly stimulated bone formation. This was independent of the
change in serum mineral homeostasis, thus it could prevent the
deterioration of bone tissue caused by malnutrition.
Taken together, though the pathologic process of the
aforementioned types of osteoporosis vary, in terms of hormone
levels, cellular response and involved signaling pathways, they all
exhibit impacted cellular function, abnormal bone turnover and
somewhat enhanced lipogenesis and oxidative stress, which provide
targets for carnosine to protect bone. In this context and based on
the current evidence, carnosine complexes are more conducive to
postmenopausal osteoporosis, with a potential application value in
the secondary osteoporosis.
Fracture healing
Fracture healing is a complex process based on the
interaction between the inflammatory and hematopoietic stem cells
and is associated with rapid osteogenic and chondrogenic
differentiation of periosteal cells and continued vascular
angiogenesis and ingrowth. This is subsequently followed by the
remodeling of immature bone into the well-structured lamellar bone
and other biochemical signal cascades (71). Animal experiments have
demonstrated that macrophages are essential for both
intramembranous and endochondral ossification of bone after a
fracture. They contribute to the deposition of woven bone and the
formation of the soft callus for endochondral ossification
(72,73).
Studies have confirmed that the use of carnosine
compounds could significantly promote the expression of protein
components and the release of cytokines from osteoblasts in the
periosteum and endosteum during bone remodeling following a
fracture (7,38,74). These include bone formation
stimulating factors such as PTH, IGF-1, TGF-β, OCN and 66-kDa
albumin that enhance bone synthesis (75). A large amount of IGF-1 deposited
in the bone matrix participates in local anabolism cooperating with
PTH. The positive feedback induces bone tissues to produce more
albumin, which has a synergistic effect in early and late fracture
healing (76). This contributes
to maintaining bone mass and bone homeostasis during bone
reconstruction. The MAPKs inhibitor PD98059 or the PKC inhibitor
staurosporine can completely inhibit zinc ions, thus weakening
their effect on fracture healing and reducing carnosine compounds
available in the body. The bioactive effect of carnosine may be
partly exerted through the MAPK signaling pathway or PKC-mediated
activation of albumin synthesis in bone tissue (74).
In vitro research by Hughes et al
(77) supports this point of
view. By using protein synthesis-promoting dietary supplements, the
albumin, muscle content and BMD in fracture healing tissue and the
expressions of IGF-1, IGF-2, actin, myosin and vascular endothelial
growth factor (VEGF) mRNA in muscles were significantly increased.
Ko et al (38) found that
polaprezinc can enhance the activity of both osteoblasts and
osteoclasts, thus accelerating the healing process of fractured
bone.
Overall, although the specific mechanism of
promoting fracture healing is unclear, there is no doubt that
therapeutic or dietary supplementation of carnosine compounds
benefits soft tissue repair, muscle mass recovery and bone
remodeling.
Osteoarthritis (OA)
OA is the most prevalent chronic joint disease,
characterized by cartilage degeneration, subchondral bone
osteosclerosis and synovium inflammation.
It has been proved that oral carnosine supplement
could reduce the expression of IL-1α and TNF-α in serum and
synovial tissues, inhibit MMPs (MMP3, MMP13) and thrombospondin
type 1 motif in the extracellular matrix, the nuclear translocation
of NF-κB p65 and finally reverse the inflammatory response
specifically through the ROS/NF-κB signaling pathway, showing a
protective effect on diabetes-induced OA (29). In a canine OA model induced by
anterior cruciate ligament transection (ACLT), carnosine prevented
and trapped lipid peroxide 4-HNE (18), inhibiting the production of
catabolites in chondrocytes and reducing the expression of
inflammatory cytokines. In addition, carnosine could markedly
prevent arthritis and chondrocytes injury, through
anti-inflammatory and anti-oxidation effects (78). A recent study by Lanza et
al (79) further showed that
a selected molecular synergy enhanced its biological activity and
resistance, in which the carnosine-hyaluronic acid conjugate
reduced oxidative damage to chondrocytes and cartilage degradation
in OA.
A recent study from Busa et al (8) confirmed that carnosine can alleviate
knee osteoarthritis pain, improve synovial protection and decrease
cartilage degradation in vivo. In IL-1β stimulated
fibroblast-like synoviocyte cells, carnosine reduce the expression
levels of cyclooxygenase-2, MMP-3 and MMP-13, ROS level and
mitochondrial membrane permeability by activating the Nrf2/heme
oxygenase-1 signaling pathway.
Thus, considering the aforementioned protective
effects of carnosine in against cartilage degeneration, abnormal
subchondral bone remodeling and synovitis, it may become a
potential choice for OA treatment, particularly when chemically
conjugated with some selected agent with enhanced antioxidative and
anti-inflammation effects.
Bone tumors
Bone is the main target organ for tumor metastasis
and the presence of bone metastases is generally a sign of poor
prognosis for advanced malignant tumors. Preclinical and clinical
studies show that carnosine has active antitumor and
immunomodulatory effects (22)
that may directly or indirectly affect the proliferation and
metastasis of bone tumor cells, in which carnosine inhibits the
glycolytic pathway and/or possibly participates in the regulation
of biological activity mediated by mTOR, hypoxia-inducible factor
α, STAT3 and MAPK (80). Primary
bone tumors are highly invasive and strongly dependent on ATP
produced by glycolysis. They can penetrate and destroy the bone
cortex and expand into adjacent soft tissues, finally, affecting
the whole bone (81). The
viability of tumor cells is reduced in the presence of carnosine.
Carnosine inhibits tumor growth, is anti-antigenic, improves
genomic stability, reduces telomere impairment and protects cells
from DNA damage induced by genotoxic substances (82).
Additionally, carnosine selectively inhibits the
growth of transformed cells and ATP-dependent glycolysis activity,
thus inhibiting the growth of tumor cells. Relevant studies have
confirmed that carnosine activates the STAT3 signaling pathway,
inhibits neuronal cell apoptosis after acute cerebral ischemia,
increases the level of p27 (a cell cycle regulator) and shortens
the growth cycle and proliferation of tumor cells (83,84). However, the overexpression of
STAT3 activated by carnosine is related to the poor prognosis of
osteosarcoma. A study by Gao et al (85) indicated that carnosine could
inhibit the proliferation and invasion of osteosarcoma cells,
promote cell apoptosis and suppress the growth and activity of
tumors by mediating the Wnt3a/β-catenin signaling pathway. Hwang
et al (86) demonstrated
that carnosine could be anti-antigenic by inhibiting the
ERK/AKT/eNOS signaling pathway and MMP-2 mediated by VEGF, thus
inhibiting the viability and activity of bladder cancer cells.
Further studies show that the implantation of bone
metastases from malignant tumors may induce the imbalance of bone
homeostasis and the change of the mechanical stress of osteoblasts,
leading to osteoblastic and osteolytic lesions, thus accelerating
the progression of tumor. Hsieh et al (87) found that carnosine could inhibit
the migration and invasion of human colorectal HCT-116 cells by
inhibiting pro-inflammatory cytokines, oxidative stress, NF-κB and
MAPK pathway activities and regulating MMPs and
epithelial-mesenchymal transition related molecules such as
E-cadherin, Twist-1 and MMP-9. Another important study found that
the immunomodulatory action of carnosine may significantly inhibit
the sympathetic nerve activity of the spleen, thereby inhibiting
cancer cell proliferation by increasing NK cell activity (52).
Last but certainly not least, from the perspective
of antitumor therapy options, the current evidence shows that
carnosine mitigates cyclophosphamide (CTX) induced G2/M
arrest in bone marrow cells to inhibit its apoptosis and
genotoxicity, downregulates the expressions of cytochrome c,
Bcl-2-associated X protein (BAX) and p21, inhibits DNA damage and
ROS production, protects bone marrow cells from CTX-induced
oxidative stress and partly restores CTX-induced suppression of
hematopoietic function (10).
Additionally, the combination of carnosine and 5-fluorouracil
decreases hypoxia-inducible factor 1 and multi-drug resistant
protein MDR1-pg expression, overcomes the acquired resistance of
some chemotherapy drugs and continually improves the anti-tumor
effect of standard chemotherapy (88). Adding carnosine to adjuvant
chemotherapy and radiotherapy could increase its efficiency and
alleviate radiation-induced skin damage and decreased immunity. In
addition, it does not have any toxic effects and could therefore
act as a ‘security guard’ for normal tissues.
Carnosine could minimize the adverse effects of
antitumor therapy in both experimental and clinical research.
Exploring further the molecular mechanisms of carnosine in
antitumor growth and bone metastasis may provide a new preventive
agent for bone tumor therapy.
Conclusions
Taken together, carnosine and its compounds could
not only activate the synthesis of protein in osteoblasts, promote
the proliferation and differentiation of osteoblasts and
chondrocytes, but also regulate the differentiation of bone marrow
macrophages into osteoclasts precursors and the activity of
osteoclasts, thus enhancing the osteogenesis and promoting fracture
healing, as well ameliorating the bone damage in the bone tumor
(Fig. 4). Carnosine compounds
should be considered a beneficial agent in the occurrence,
development and process of bone reconstruction. Although further
clinical experimental studies are needed to verify their benefits
on bone and the mechanism are far from fully elucidated, it is
reasonable to hypothesize that carnosine combined with standard
anti-osteoporosis and anti-osteosarcoma therapies might improve its
curative effects on a series disease with abnormal bone remodeling,
as well promote fracture healing. Ultimately, it may become a
promising therapeutic strategy for bone metabolism-related
diseases.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Nation Nature Science
Foundation of China (grant no. NSFC81874029), Nature Science
Foundation of Hebei Province (grant no. H2020209266), Basic
Scientific Research Funds Program of Universities in Hebei Province
(grant no. JYG2021005); Central Government-guided Local Science and
Technology Development Foundation of Hebei Province (grant no.
226Z7709G) and Youth Talent Support Program of Hebei Province
(grant no. JI-2016-10).
Availability of data and materials
Not applicable.
Authors' contributions
HY conceived and wrote the manuscript. XH conducted
the formal literature search and analysis. FT made substantial
contributions to conception and design. LX and FT critically
revised the manuscript. Data authentication is not applicable. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Brown C: Osteoporosis: Staying strong.
Nature. 550:S15–S17. 2017. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Choi JY: Healthy bone tissue homeostasis.
Exp Mol Med. 52:11652020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Babizhayev MA and Deyev AI: Management of
the virulent influenza virus infection by oral formulation of
nonhydrolized carnosine and isopeptide of carnosine attenuating
proinflammatory cytokine-induced nitric oxide production. Am J
Ther. 19:e25–e47. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hu J, Zheng W, Zhao D, Sun L, Zhou B, Liu
J, Wang O, Jiang Y, Xia W, Xing X and Li M: Health-related quality
of life in men with osteoporosis: A systematic review and
meta-analysis. Endocrine. 74:270–280. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Boldyrev AA, Aldini G and Derave W:
Physiology and pathophysiology of carnosine. Physiol Rev.
93:1803–1845. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Maeno M, Ito-Kato E, Suzuki N, Takada T,
Takayama T, Ito K and Otsuka K: Effect of beta-alanyl-L-histidinato
zinc on the differentiation pathway of human periodontal ligament
cells. Life Sci. 74:2493–2504. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Igarashi A and Yamaguchi M: Great increase
in bone 66 kDa protein and osteocalcin at later stages with healing
rat fractures: effect of zinc treatment. Int J Mol Med. 11:223–228.
2003.PubMed/NCBI
|
|
8
|
Busa P, Lee SO, Huang N, Kuthati Y and
Wong CS: Carnosine alleviates knee osteoarthritis and promotes
synoviocyte protection via activating the Nrf2/HO-1 signaling
pathway: An in-vivo and in-vitro study. Antioxidants (Basel).
11:12092022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li X, Yang K, Gao S, Zhao J, Liu G, Chen
Y, Lin H, Zhao W, Hu Z and Xu N: Carnosine stimulates
macrophage-mediated clearance of senescent skin cells through
activation of the AKT2 signaling pathway by CD36 and RAGE. Front
Pharmacol. 11:5938322020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Deng J, Zhong YF, Wu YP, Luo Z, Sun YM,
Wang GE, Kurihara H, Li YF and He RR: Carnosine attenuates
cyclophosphamide-induced bone marrow suppression by reducing
oxidative DNA damage. Redox Biol. 14:1–6. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yamaguchi M: Role of nutritional zinc in
the prevention of osteoporosis. Mol Cell Biochem. 338:241–254.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yamaguchi M and Ozaki K:
Beta-alanyl-L-histidinato zinc prevents the toxic effect of
aluminium on bone metabolism in weanling rats. Pharmacology.
41:338–344. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yamaguchi M and Kishi S: Effect of
zinc-chelating dipeptide on bone metabolism in weanling rats:
Comparison with beta-alanyl-L-histidinato zinc-related compounds.
Peptides. 15:671–673. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Udechukwu MC, Collins SA and Udenigwe CC:
Prospects of enhancing dietary zinc bioavailability with
food-derived zinc-chelating peptides. Food Funct. 7:4137–4144.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
O'Connor JP, Kanjilal D, Teitelbaum M, Lin
SS and Cottrell JA: Zinc as a therapeutic agent in bone
regeneration. Materials (Basel). 13:22112020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ooi TC, Chan KM and Sharif R: Zinc
L-carnosine suppresses inflammatory responses in
lipopolysaccharide-induced RAW 264.7 murine macrophages cell line
via activation of Nrf2/HO-1 signaling pathway. Immunopharmacol
Immunotoxicol. 39:259–267. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Caruso G, Fresta CG, Martinez-Becerra F,
Antonio L, Johnson RT, de Campos RPS, Siegel JM, Wijesinghe MB,
Lazzarino G and Lunte SM: Carnosine modulates nitric oxide in
stimulated murine RAW 264.7 macrophages. Mol Cell Biochem.
431:197–210. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shi Q, Abusarah J, Zaouter C, Moldovan F,
Fernandes JC, Fahmi H and Benderdour M: New evidence implicating
4-hydroxynonenal in the pathogenesis of osteoarthritis in vivo.
Arthritis Rheumatol. 66:2461–2471. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Spaas J, Franssen WMA, Keytsman C,
Blancquaert L, Vanmierlo T, Bogie J, Broux B, Hellings N, van
Horssen J, Posa DK, et al: Carnosine quenches the reactive carbonyl
acrolein in the central nervous system and attenuates autoimmune
neuroinflammation. J Neuroinflammation. 18:2552021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chmielewska K, Dzierzbicka K,
Inkielewicz-Stępniak I and Przybyłowska M: Therapeutic potential of
carnosine and its derivatives in the treatment of human diseases.
Chem Res Toxicol. 33:1561–1578. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hu Y, Li X, Zhi X, Cong W, Huang B, Chen
H, Wang Y, Li Y, Wang L, Fang C, et al: RANKL from bone marrow
adipose lineage cells promotes osteoclast formation and bone loss.
EMBO Rep. 22:e524812021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jukić I, Kolobarić N, Stupin A, Matić A,
Kozina N, Mihaljević Z, Mihalj M, Šušnjara P, Stupin M, Ćurić ŽB,
et al: Carnosine, small but mighty-prospect of use as functional
ingredient for functional food formulation. Antioxidants (Basel).
10:10372021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xu T, Wang C, Zhang R, Xu M, Liu B, Wei D,
Wang G and Tian S: Carnosine markedly ameliorates H9N2 swine
influenza virus-induced acute lung injury. J Gen Virol.
96:2939–2950. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Caruso G: Unveiling the hidden therapeutic
potential of carnosine, a molecule with a multimodal mechanism of
action: A position paper. Molecules. 27:33032022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Seo HJ, Cho YE, Kim T, Shin HI and Kwun
IS: Zinc may increase bone formation through stimulating cell
proliferation, alkaline phosphatase activity and collagen synthesis
in osteoblastic MC3T3-E1 cells. Nutr Res Pract. 4:356–361. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hashizume M and Yamaguchi M: Effect of
beta-alanyl-L-histidinato zinc on differentiation of osteoblastic
MC3T3-E1 cells: Increases in alkaline phosphatase activity and
protein concentration. Mol Cell Biochem. 131:19–24. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yamaguchi M and Hashizume M: Effect of
parathyroid hormone and interleukin-1 alpha in osteoblastic
MC3T3-E1 cells: Interaction with beta-alanyl-L-histidinato zinc.
Peptides. 15:633–636. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yamaguchi M: beta-Alanyl-L-histidinato
zinc and bone resorption. Gen Pharmacol. 26:1179–1183. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang Y, Wang Y, Kong Y, Zhang X, Zhang H,
Gang Y and Bai L: Carnosine prevents type 2 diabetes-induced
osteoarthritis through the ROS/NF-κB pathway. Front Pharmacol.
9:5982018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yamaguchi M, Goto M, Uchiyama S and
Nakagawa T: Effect of zinc on gene expression in osteoblastic
MC3T3-E1 cells: Enhancement of Runx2, OPG, and regucalcin mRNA
expressions. Mol Cell Biochem. 312:157–166. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sanguineti R, Puddu A, Mach F, Montecucco
F and Viviani GL: Advanced glycation end products play adverse
proinflammatory activities in osteoporosis. Mediators Inflamm.
2014:9758722014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Fresta CG, Hogard ML, Caruso G, Melo Costa
EE, Lazzarino G and Lunte SM: Monitoring carnosine uptake by RAW
264.7 macrophage cells using microchip electrophoresis with
fluorescence detection. Anal Methods. 9:402–408. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Caruso G, Benatti C, Musso N, Fresta CG,
Fidilio A, Spampinato G, Brunello N, Bucolo C, Drago F, Lunte SM,
et al: Carnosine protects macrophages against the toxicity of
Aβ1-42 oligomers by decreasing oxidative stress. Biomedicines.
9:4772021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yamaguchi M and Kishi S: Zinc compounds
inhibit osteoclast-like cell formation at the earlier stage of rat
marrow culture but not osteoclast function. Mol Cell Biochem.
158:171–177. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yamaguchi M and Kishi S: Inhibitory effect
of zinc-chelating dipeptide on parathyroid hormone-stimulated
osteoclast-like cell formation in mouse marrow cultures:
Involvement of calcium signaling. Peptides. 16:629–633. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yamaguchi M and Kishi S: Differential
effects of transforming growth factor-beta on osteoclast-like cell
formation in mouse marrow culture: Relation to the effect of
zinc-chelating dipeptides. Peptides. 16:1483–1488. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kishi S and Yamaguchi M: Inhibitory effect
of zinc compounds on osteoclast-like cell formation in mouse marrow
cultures. Biochem Pharmacol. 48:1225–1230. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ko EA, Park YJ, Yoon DS, Lee KM, Kim J,
Jung S, Lee JW and Park KH: Drug repositioning of polaprezinc for
bone fracture healing. Commun Biol. 5:4622022. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Thomas S and Jaganathan BG: Signaling
network regulating osteogenesis in mesenchymal stem cells. J Cell
Commun Signal. 16:47–61. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Seo BM, Miura M, Gronthos S, Bartold PM,
Batouli S, Brahim J, Young M, Robey PG, Wang CY and Shi S:
Investigation of multipotent postnatal stem cells from human
periodontal ligament. Lancet. 364:149–155. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Takada T, Suzuki N, Ito-Kato E, Noguchi Y,
Ito M, Maeno M and Otsuka K: Effect of beta-alanyl-L-histidinato
zinc on the differentiation of C2C12 cells. Life Sci. 76:509–520.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ito-Kato E, Suzuki N, Maeno M, Takada T,
Tanabe N, Takayama T, Ito K and Otsuka K: Effect of carnosine on
runt-related transcription factor-2/core binding factor alpha-1 and
Sox9 expressions of human periodontal ligament cells. J Periodontal
Res. 39:199–204. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Eastell R, O'Neill TW, Hofbauer LC,
Langdahl B, Reid IR, Gold DT and Cummings SR: Postmenopausal
osteoporosis. Nat Rev Dis Primers. 2:160692016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Fischer V and Haffner-Luntzer M:
Interaction between bone and immune cells: Implications for
postmenopausal osteoporosis. Semin Cell Dev Biol. 123:14–21. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Xie H, Cui Z, Wang L, Xia Z, Hu Y, Xian L,
Li C, Xie L, Crane J, Wan M, et al: PDGF-BB secreted by
preosteoclasts induces angiogenesis during coupling with
osteogenesis. Nat Med. 20:1270–1278. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Lorenzo J: From the gut to bone:
Connecting the gut microbiota with Th17 T lymphocytes and
postmenopausal osteoporosis. J Clin Invest. 131:e1466192021.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kisi S and Yamaguchi M: Stimulatory effect
of beta-alanyl-L-histidinato zinc on alkaline phosphatase activity
in bone tissues from elderly rats: Comparison with zinc sulfate
action. Biol Pharm Bull. 17:345–347. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Kishi S, Segawa Y and Yamaguchi M:
Histomorphological confirmation of the preventive effect of
beta-alanyl-L-histidinato zinc on bone loss in ovariectomized rats.
Biol Pharm Bull. 17:862–865. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhang Y, Chen CY, Liu YW, Rao SS, Tan YJ,
Qian YX, Xia K, Huang J, Liu XX, Hong CG, et al: Neuronal induction
of bone-fat imbalance through osteocyte neuropeptide Y. Adv Sci
(Weinh). 8:e21008082021. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Siddappa R, Martens A, Doorn J, Leusink A,
Olivo C, Licht R, van Rijn L, Gaspar C, Fodde R, Janssen F, et al:
cAMP/PKA pathway activation in human mesenchymal stem cells in
vitro results in robust bone formation in vivo. Proc Natl Acad Sci
USA. 105:7281–7286. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Nagai K, Niijima A, Yamano T, Otani H,
Okumra N, Tsuruoka N, Nakai M and Kiso Y: Possible role of
L-carnosine in the regulation of blood glucose through controlling
autonomic nerves. Exp Biol Med (Maywood). 228:1138–1145. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Horii Y, Shen J, Fujisaki Y, Yoshida K and
Nagai K: Effects of L-carnosine on splenic sympathetic nerve
activity and tumor proliferation. Neurosci Lett. 510:1–5. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Nagai K, Misonou Y, Fujisaki Y, Fuyuki R
and Horii Y: Topical application of L-carnosine to skeletal muscle
excites the sympathetic nerve innervating the contralateral
skeletal muscle in rats. Amino Acids. 51:39–48. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Cararo JH, Streck EL, Schuck PF and
Ferreira Gda C: Carnosine and related peptides: Therapeutic
potential in age-related disorders. Aging Dis. 6:369–379. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Koide M, Yamashita T, Murakami K, Uehara
S, Nakamura K, Nakamura M, Matsushita M, Ara T, Yasuda H, Penninger
JM, et al: Sclerostin expression in trabecular bone is
downregulated by osteoclasts. Sci Rep. 10:137512020. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Manolagas SC and Almeida M: Gone with the
Wnts: beta-catenin, T-cell factor, forkhead box O, and oxidative
stress in age-dependent diseases of bone, lipid, and glucose
metabolism. Mol Endocrinol. 21:2605–2614. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Tallon MJ, Harris RC, Maffulli N and
Tarnopolsky MA: Carnosine, taurine and enzyme activities of human
skeletal muscle fibres from elderly subjects with osteoarthritis
and young moderately active subjects. Biogerontology. 8:129–137.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Yamaguchi M and Ozaki K: Effect of the new
zinc compound beta-alanyl-L-histidinato zinc on bone metabolism in
elderly rats. Pharmacology. 41:345–349. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Yamaguchi M and Ehara Y: Zinc decrease and
bone metabolism in the femoral-metaphyseal tissues of rats with
skeletal unloading. Calcif Tissue Int. 57:218–223. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Uddin SMZ and Qin YX: Dynamic acoustic
radiation force retains bone structural and mechanical integrity in
a functional disuse osteopenia model. Bone. 75:8–17. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Deal C: Bone loss in rheumatoid arthritis:
Systemic, periarticular, and focal. Curr Rheumatol Rep. 14:231–237.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Boudignon BM, Bikle DD, Kurimoto P,
Elalieh H, Nishida S, Wang Y, Burghardt A, Majumdar S, Orwoll BE,
Rosen C and Halloran BP: Insulin-like growth factor I stimulates
recovery of bone lost after a period of skeletal unloading. J Appl
Physiol (1985). 103:125–131. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Chotiyarnwong P and McCloskey EV:
Pathogenesis of glucocorticoid-induced osteoporosis and options for
treatment. Nat Rev Endocrinol. 16:437–447. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Segawa Y, Tsuzuike N, Itokazu Y, Tagashira
E and Yamaguchi M: beta-Alanyl-L-histidinato zinc prevents
hydrocortisone-induced disorder of bone metabolism in rats. Res Exp
Med (Berl). 192:317–322. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Wang Y, Li Y, Khabut A, Chubinskaya S,
Grodzinsky AJ and Önnerfjord P: Quantitative proteomics analysis of
cartilage response to mechanical injury and cytokine treatment.
Matrix Biol. 63:11–22. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Holmdahl R, Sareila O, Olsson LM, Backdahl
L and Wing K: Ncf1 polymorphism reveals oxidative regulation of
autoimmune chronic inflammation. Immunol Rev. 269:228–247. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Sugiyama T, Tanaka H and Kawai S:
Improvement of periarticular osteoporosis in postmenopausal women
with rheumatoid arthritis by beta-alanyl-L-histidinato zinc: A
pilot study. J Bone Miner Metab. 18:335–338. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Atkins GJ, Anderson PH, Findlay DM,
Welldon KJ, Vincent C, Zannettino ACW, O'Loughlin PD and Morris HA:
Metabolism of vitamin D3 in human osteoblasts: Evidence for
autocrine and paracrine activities of 1 alpha,25-dihydroxyvitamin
D3. Bone. 40:1517–1528. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Kato H, Ochiai-Shino H, Onodera S, Saito
A, Shibahara T and Azuma T: Promoting effect of 1,25(OH)2 vitamin
D3 in osteogenic differentiation from induced pluripotent stem
cells to osteocyte-like cells. Open Biol. 5:1402012015. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Segawa Y, Tsuzuike N, Tagashira E and
Yamaguchi M: Preventive effect of beta-alanyl-L-histidinato zinc on
bone metabolism in rats fed on low-calcium and vitamin D-deficient
diets. Res Exp Med (Berl). 192:213–219. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Majidinia M, Sadeghpour A and Yousefi B:
The roles of signaling pathways in bone repair and regeneration. J
Cell Physiol. 233:2937–2948. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Vi L, Baht GS, Whetstone H, Ng A, Wei Q,
Poon R, Mylvaganam S, Grynpas M and Alman BA: Macrophages promote
osteoblastic differentiation in-vivo: Implications in fracture
repair and bone homeostasis. J Bone Miner Res. 30:1090–1102. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Sandberg OH, Tätting L, Bernhardsson ME
and Aspenberg P: Temporal role of macrophages in cancellous bone
healing. Bone. 101:129–133. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Igarashi A and Yamaguchi M:
Characterization of the increase in bone 66 kDa protein component
with healing rat fractures: Stimulatory effect of zinc. Int J Mol
Med. 9:503–508. 2002.PubMed/NCBI
|
|
75
|
Igarashi A and Yamaguchi M: Increase in
bone protein components with healing rat fractures: Enhancement by
zinc treatment. Int J Mol Med. 4:615–620. 1999.PubMed/NCBI
|
|
76
|
Xian L, Wu X, Pang L, Lou M, Rosen CJ, Qiu
T, Crane J, Frassica F, Zhang L, Rodriguez JP, et al: Matrix IGF-1
maintains bone mass by activation of mTOR in mesenchymal stem
cells. Nat Med. 18:1095–1101. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Hughes MS, Kazmier P, Burd TA, Anglen J,
Stoker AM, Kuroki K, Carson WL and Cook JL: Enhanced fracture and
soft-tissue healing by means of anabolic dietary supplementation. J
Bone Joint Surg Am. 88:2386–2394. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Ponist S, Drafi F, Kuncirova V, Mihalova
D, Rackova L, Danisovic L, Ondrejickova O, Tumova I, Trunova O,
Fedorova T and Bauerova K: Effect of carnosine in experimental
arthritis and on primary culture chondrocytes. Oxid Med Cell
Longev. 2016:84705892016. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Lanza V, Greco V, Bocchieri E, Sciuto S,
Inturri R, Messina L, Vaccaro S, Bellia F and Rizzarelli E:
Synergistic effect of L-carnosine and hyaluronic acid in their
covalent conjugates on the antioxidant abilities and the mutual
defense against enzymatic degradation. Antioxidants (Basel).
11:6642022. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Hipkiss AR and Gaunitz F: Inhibition of
tumour cell growth by carnosine: Some possible mechanisms. Amino
Acids. 46:327–337. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Ritter J and Bielack SS: Osteosarcoma. Ann
Oncol. 21 (Suppl 7):vii320–vii325. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Ooi TC, Chan KM and Sharif R: Antioxidant,
anti-inflammatory, and genomic stability enhancement effects of
zinc l-carnosine: A potential cancer chemopreventive agent? Nutr
Cancer. 69:201–210. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Lu R, Wang Q, Li J and Miao D: P27
deletion enhances hematopoiesis by paracrine action of IL22
secreted from bone marrow mesenchymal stem cells. Am J Transl Res.
12:787–799. 2020.PubMed/NCBI
|
|
84
|
Wang JP, Yang ZT, Liu C, He YH and Zhao
SS: L-carnosine inhibits neuronal cell apoptosis through signal
transducer and activator of transcription 3 signaling pathway after
acute focal cerebral ischemia. Brain Res. 1507:125–133. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Gao K, Zhang Y, Niu J, Nie Z, Liu Q and Lv
C: Zinc promotes cell apoptosis via activating the Wnt-3a/β-catenin
signaling pathway in osteosarcoma. J Orthop Surg Res. 15:572020.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Hwang B, Shin SS, Song JH, Choi YH, Kim WJ
and Moon SK: Carnosine exerts antitumor activity against bladder
cancers in vitro and in vivo via suppression of angiogenesis. J
Nutr Biochem. 74:1082302019. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Hsieh SL, Hsieh S, Lai PY, Wang JJ, Li CC
and Wu CC: Carnosine suppresses human colorectal cell migration and
intravasation by regulating EMT and MMP expression. Am J Chin Med.
47:477–494. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Iovine B, Guardia F, Irace C and
Bevilacqua MA: l-Carnosine dipeptide overcomes acquired resistance
to 5-fluorouracil in HT29 human colon cancer cells via
downregulation of HIF1-alpha and induction of apoptosis. Biochimie.
127:196–204. 2016. View Article : Google Scholar : PubMed/NCBI
|