Introduction
Prostate cancer (PCa) is a significant global health
concern, with an estimated 1.4 million new cases and >375,000
associated mortalities annually in 2020 (1). In the United States, PCa is the most
commonly diagnosed cancer among men, with ~290,000 new cases and
35,000 mortalities reported annually (2). The incidence of PCa has also been
increasing in Asia, reflecting the growing global burden of this
disease (1). This increasing trend
underscores the importance of understanding the mechanisms driving
PCa progression and treatment resistance to improve patient
outcomes.
The treatment landscape for PCa has evolved
significantly, with androgen deprivation therapy (ADT) remaining
the cornerstone of treatment for advanced disease (3,4).
While ADT initially elicits favorable clinical responses, most
patients eventually progress to castration-resistant prostate
cancer (CRPC) within 3 years (2).
Some patients develop highly aggressive small-cell neuroendocrine
carcinoma, which is often characterized by resistance to ADT, a
relative lack of clinical treatment strategies and a high mortality
rate (5-year survival rate is only ~10%) (5–7).
The gut microbiota, a complex microbial community in
the gastrointestinal tract, plays a crucial role in human health
and disease. Research has shown that the gut microbiota can
influence cancer development, progression and treatment response
through multiple mechanisms, including drug metabolism, immune
modulation and metabolite production, thereby affecting tumor
biology (8–10). For instance, the gut microbiota can
directly metabolize anticancer drugs, thereby altering their
efficacy and contributing to treatment resistance (8,11–14).
Specifically, in PCa, the gut microbiota may influence disease
progression and treatment resistance through androgen metabolism
and the production of procarcinogenic metabolites (8,15,16).
Gut microbiota-derived metabolites constitute a
critical but underappreciated axis in the development of treatment
resistance in advanced PCa, acting through convergent pathways
involving androgen receptor (AR) signaling, immune modulation and
metabolic adaptation. The present review summarizes the current
status of treatment resistance in PCa and examines the effects of
gut microbiota on treatment resistance in PCa through direct and
indirect pathways. The aim of the current study is to integrate
existing knowledge and explore novel approaches (for example,
probiotics, prebiotics, fecal microbiota transplantation) in order
to improve the understanding of the impact of gut microbiota on
treatment resistance in PCa, paving the way for improved treatment
strategies.
Gut microbiota and treatment resistance
The gut microbiota, a complex and dynamic community
of microorganisms residing in the gastrointestinal tract, has
emerged as a critical determinant of the efficacy and resistance to
cancer treatments across various malignancies. Accumulating
evidence suggests that the composition and function of the gut
microbiota can modulate host immune responses, drug metabolism and
tumor microenvironments, thereby playing a pivotal role in
determining the success of therapeutic interventions. In this
context, understanding the intricate interplay between gut
microbiota and cancer treatments is essential for developing novel
strategies to overcome resistance and improve patient outcomes.
Butyric acid produced by Clostridium
butyricum can reverse the anti-programmed cell death protein 1
(PD-1) resistance in patients with non-small cell lung cancer
caused via the use of proton pump inhibitors by upregulating the
expression of perforin and granzyme B in CD8+ T cells
(17). Similarly, study on
pan-cancer have revealed that the effect of indole-3-carboxaldehyde
derived from Lactobacillus is bidirectional. By activating
the aryl hydrocarbon receptor (AhR), indole-3-carboxaldehyde can
maintain the stem cell-like phenotype of T cells and enhance the
efficacy of immune checkpoint inhibitors in breast cancer and
melanoma. However, indole-3-carboxaldehyde can also upregulate the
expression of ATP-binding cassette subfamily B member 1
(P-glycoprotein) (ABCB1) in tumor cells through AhR activation,
promoting cross-resistance of breast cancer to taxane drugs
(18). In addition, dysbiosis
caused by a high-fat diet leads to excessive production of leucine
derived from the Clostridium genus in the intestine,
activating mammalian target of rapamycin (mTOR) signaling in
myeloid-derived suppressor cells (MDSCs) (19). This weakens the efficacy of
cyclin-dependent kinases 4 and 6 inhibitors in breast cancer and
promotes immune escape of estrogen receptor-positive tumors
(19). In liver cancer,
Fusobacterium nucleatum binds to E-cadherin in tumor cells
through its surface Fusobacterium adhesin A, thereby
activating β-catenin signaling pathway to upregulate multidrug
resistance protein 1 transcription and promote acquired resistance
to sorafenib (20). Bacteroides
fragilis directly binds to the neurogenic locus notch homolog
protein 1 (NOTCH1) receptor on tumor cells via its surface
proteins, activating the NOTCH1 signaling pathway and inducing
epithelial-mesenchymal transition (EMT) and a stem cell-like
phenotype, thereby promoting resistance to 5-fluorouracil and
oxaliplatin in colorectal cancer (21). In PCa, the decreased abundance of
Akkermansia muciniphila is associated with enzalutamide
resistance (22). Based on the
cross-cancer hypothesis-the concept that molecular mechanisms or
resistance pathways observed in one cancer type may be universally
present and functionally relevant across histologically distinct
malignancies-the mechanism of enzalutamide resistance may be
related to reducing the production of short-chain fatty acids
(SCFAs) (especially butyrate) and relieving regulatory T cell
(Treg)-mediated immunosuppression (23). However, the transferability of
mechanisms between different cancer types remains controversial,
and direct evidence in PCa is lacking.
Multiple recent systematic reviews and meta-analyses
have provided important evidence-based insights into the role of
the microbiome in PCa. A meta-analysis by Huang et al
(24) encompassing seven studies
(including 250 PCa patients and 192 controls), demonstrated that
the α-diversity of the gut microbiota was notably lower in patients
with PCa compared with healthy controls. Regarding microbial
abundance, patients with PCa exhibited markedly higher relative
abundances of Proteobacteria, the class Bacteroidia, the class
Clostridia, as well as the genera Prevotella,
Escherichia-Shigella and Faecalibacterium (24). Conversely, the abundances of
Actinobacteria, Firmicutes and the genus Veillonella were
notably reduced (24). Another
systematic review, covering 42 studies, further elucidated the
multifaceted role of the microbiome in PCa diagnosis, prognosis and
treatment response (25). The
urinary microbiota demonstrated potential diagnostic value
(sensitivity 58–82%). Enrichment of the class
Betaproteobacteria in the gut was associated with earlier
progression to CRPC, with a median time to progression shortened by
5.2 months (hazard ratio, 1.8; 95% confidence interval, 1.3–2.5)
(25). Furthermore, ADT-induced
dysbiosis (for example, overgrowth of Klebsiella species)
was associated with a 2.1-fold increased risk of resistance, while
responders to immunotherapy exhibited enrichment of Akkermansia
muciniphila (25). This body
of cross-cancer and PCa-specific evidence indicates that the gut
microbiota can influence cancer detection, progression and
therapeutic efficacy through both direct and indirect mechanisms,
thereby laying a theoretical foundation for the development of
microbiota-targeted intervention strategies.
Current status of PCa treatment
resistance
ADT is the cornerstone treatment for metastatic PCa,
but resistance is a major challenge, with most patients progressing
to CRPC within 18–24 months (26).
In North America and Europe, 10–20% of patients develop CRPC within
5 years of starting ADT, while in Asia, the progression rate is
similar, but overall survival outcomes are worse due to differences
in healthcare infrastructure and treatment availability (27). Chemotherapy, particularly with
docetaxel, is the mainstay treatment for metastatic CRPC (mCRPC);
however, resistance is common (26). In North America, 30–40% of patients
with mCRPC do not respond to initial docetaxel treatment, and in
Asia, up to 45% of patients may not benefit from docetaxel-based
therapy (27). Immunotherapy,
including immune checkpoint inhibitors, has shown promise in
treating advanced PCa; however, resistance remains a significant
issue (26). In North America, the
response rate to immune checkpoint inhibitors is 15–20%, whereas in
Asia, the response rate is slightly lower, at 10–15% (26,27).
The landscape of PCa treatment has evolved
significantly, yet the emergence of treatment resistance remains a
formidable barrier to achieving durable therapeutic success.
Previous investigations have unveiled the multifaceted mechanisms
underlying resistance to ADT, immunotherapy and chemotherapy
(Table I; Fig. 1). Elucidating these mechanisms is
essential for devising more effective and targeted therapeutic
strategies. This section provides an in-depth overview of the
current status of treatment resistance in PCa, with a focus on the
biological, genetic and microenvironmental factors that contribute
to therapeutic failure.
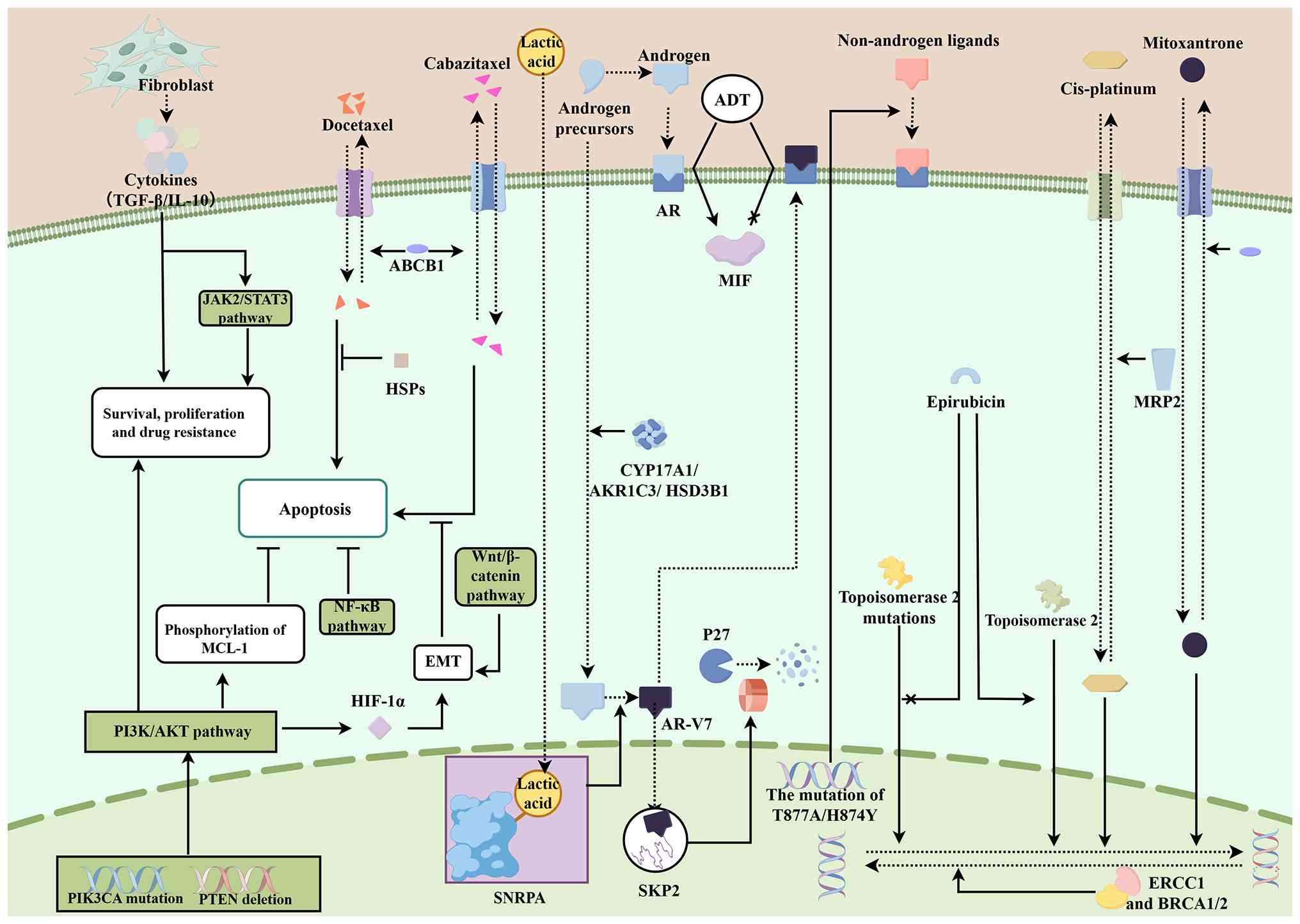 | Figure 1.Current resistance mechanisms of ADT,
chemotherapy and immunotherapy in PCa. This figure elucidates the
resistance mechanisms of PCa to ADT, immunotherapy and chemotherapy
(Figdraw; www.figdraw.com; ID, UPPYYb6b66). It
summarizes key mechanisms underlying treatment resistance,
including AR alterations, intratumoral androgen synthesis,
activation of survival pathways (PI3K/AKT, NF-κB), and tumor
microenvironment contributions. Specific resistance mechanisms for
chemotherapeutic agents (such as docetaxel, cisplatin and
mitoxantrone) are also depicted. ABCB1, atp-binding cassette
subfamily b member 1 (p-glycoprotein); ADT, androgen deprivation
therapy; AKR1C3, aldo-keto reductase family 1 member c3; AR,
androgen receptor; AR-V7, androgen receptor splicing variant 7;
BRCA1/2, breast cancer susceptibility gene 1/2; CYP17A1, cytochrome
p450 family 17 subfamily a member 1; EMT, epithelial-mesenchymal
transition; ERCC1, excision repair cross-complementation group 1;
H874Y, androgen receptor mutation at position 874 (histidine to
tyrosine); HIF-1α, hypoxia-inducible factor 1α; HSD3B1,
hydroxysteroid 3β-dehydrogenase type 1; HSPs, heat shock proteins;
IL-10, interleukin-10; JAK2, Janus kinase 2; STAT3, signal
transducer and activator of transcription 3; MIF, macrophage
migration inhibitory factor; MRP2, multidrug resistance-associated
protein 2; NF-κB, nuclear factor κ-light-chain-enhancer of
activated B cells; P27, cyclin-dependent kinase inhibitor 1b;
PI3K/AKT, phosphoinositide 3-kinase/protein kinase b; PI3KCA,
phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit α;
PTEN, phosphatase and tensin homolog; SKP2, S-phase
kinase-associated protein 2; SNRPA, small nuclear ribonucleoprotein
polypeptide a; T877A, androgen receptor mutation at position 877
(threonine to alanine); TGF-β, transforming growth factor-β; Wnt,
wingless-related integration site. |
 | Table I.Mechanisms of therapeutic resistance
in PCa. |
Table I.
Mechanisms of therapeutic resistance
in PCa.
| A, ADT |
|---|
|
|---|
| Resistance
mechanism category | Specific resistance
mechanism | (Refs.) |
|---|
| Alterations in
AR | AR gene
amplification and overexpression, allowing cancer cells to maintain
AR signaling despite low androgen levels. AR gene mutations lead to
receptor activation by non-androgenic ligands or antagonists (T877A
and H874Y mutations). | (27–29) |
| Intratumoral
androgen synthesis | PCa cells
synthesize androgens intratumorally via upregulation of enzymes
such as CYP17A1, AKR1C3 and HSD3B1, bypassing the need for
circulating androgens. | (30,31) |
| Other
mechanisms | The aberrant
activation of the PI3K/AKT pathway bypasses AR signaling;
ADT-induced metabolic reprogramming generates the AR-V7 splice
variant; AR-V7 drives senescence escape; upregulation of MIF and
bidirectional signaling of BCL2 promote proliferation; along with
neuroendocrine differentiation, collectively drive castration
resistance. | (32–38) |
|
| B,
Immunotherapy |
|
| Resistance
mechanism category | Specific
resistance mechanism | (Refs.) |
|
| Tumor
microenvironment and immunosuppression | Immunosuppressive
cells (Tregs, MDSCs, TAMs) create an immunosuppressive niche in the
tumor microenvironment, hindering the efficacy of immune checkpoint
inhibitors. | (40–42) |
| Genetic and
epigenetic alterations | Mutations in DNA
repair genes (BRCA2, ATM) lead to genomic instability; epigenetic
changes alter the expression of immune-related genes, contributing
to resistance. | (43–45) |
| Immune checkpoint
inhibitors and resistance mechanisms | Low tumor
mutational burden and lack of PD-L1 expression in patients with
PCa; immunosuppressive cytokines (TGF-β, IL-10) inhibit the
antitumor immune response. | (46–48) |
|
| C,
Chemotherapy |
|
| Resistance
mechanism category | Specific
resistance mechanism | (Refs.) |
|
| Docetaxel
resistance | Overexpression of
ABC transporters (ABCB1) effluxes cytotoxic drugs, reducing
intracellular drug concentration; alterations in microtubule
dynamics and upregulation of survival pathways to (PI3K/AKT/mTOR)
promote cell survival. Activation of the AKT pathway stabilizes
anti-apoptotic protein MCL-1, inhibiting apoptosis induced by
docetaxel; heat shock proteins protect cancer cells from
docetaxel-induced stress. | (49–53) |
| Cabazitaxel
resistance | Overexpression of
ABC transporters and activation of survival pathways; AR gene
mutations lead to constitutive receptor activation independent of
androgen levels. Upregulation of the Wnt/β-catenin signaling
pathway enhances cell survival and promotes an EMT phenotype,
associated with increased metastatic potential and drug
resistance. | (54–56) |
| Cisplatin
resistance | Increased DNA
repair capacity (upregulation of ERCC1 and BRCA1/2), reduces drug
uptake and activation of survival pathways (NF-κB pathway).
Expression of multidrug resistance-associated protein 2 reduces
intracellular cisplatin levels by mediating drug efflux. | (57–59) |
| Mitoxantrone
resistance | Overexpression of
ABC transporters (ABCB1) increases drug efflux, reducing
intracellular drug concentration; defects in the DNA damage
response (mutations in BRCA2) enhance repair of
mitoxantrone-induced DNA damage. | (60,61) |
| Epirubicin
resistance | Overexpression of
multidrug resistance proteins and breast cancer resistance protein
reduces intracellular epirubicin levels; alterations in
topoisomerase II impair the drug ability to induce DNA strand
breaks. | (62,63) |
| Vinblastine
resistance | Changes in tubulin
dynamics (mutations or overexpression of β-tubulin isotypes) reduce
vinblastine ability to inhibit microtubule polymerization;
activation of prosurvival pathways (MAPK pathway) promotes cell
survival. | (64,65) |
| Tumor
microenvironment | Cancer-associated
fibroblasts secrete cytokines and growth factors (IL-6) that
activate survival pathways (for example, JAK2/STAT3) in cancer
cells, promoting resistance. Hypoxic microenvironment induces
HIF-1α expression, promoting EMT and activating survival pathways,
contributing to chemotherapy resistance. | (66,67) |
ADT resistance
ADT, which targets the AR signaling pathway, has
long been the cornerstone of treatment for advanced PCa. However,
the development of ADT resistance, which leads to CRPC, is a key
concern. Understanding the mechanisms underlying this resistance is
critical for developing effective treatment strategies.
Alterations in the AR
AR gene amplification and subsequent upregulation
are common resistance mechanisms. Studies have shown that AR
amplification occurs in an important proportion of patients with
CRPC, allowing cancer cells to maintain AR signaling despite low
androgen levels (28,29). Mutations in the AR gene can lead to
receptor activation by non-androgenic ligands or antagonists. For
example, mutations such as T877A and H874Y have been identified,
which enable the receptor to be activated by alternative ligands
(30). This alteration highlights
the adaptability of PCa cells to maintain AR signaling and disease
progression under conditions of ADT.
Intratumoral androgen synthesis
PCa cells can develop the ability to synthesize
androgens intratumorally, thereby bypassing the need for
circulating androgens. This is achieved through upregulation of
enzymes involved in androgen biosynthesis, such as cytochrome P450
family 17 subfamily A member 1, aldo-keto reductase family 1 member
C3 and hydroxysteroid 3β-dehydrogenase type 1. These enzymes
convert precursor molecules into active androgens within the tumor
microenvironment, sustaining AR activation (31,32).
Other mechanisms
The phosphatidylinositol 3-kinase (PI3K)/protein
kinase B (AKT)/mTOR pathway can be activated by PIK3CA mutation,
loss of phosphatase and tensin homolog (PTEN), or other mechanisms
(e.g., PIK3CB mutation, PIK3CB mutation, AKT1 mutation, TSC1/TSC2
inactivating mutation, MTOR mutation) to promote PCa cell survival
and proliferation independent of AR signaling (33). ADT drives metabolic reprogramming
of adenomatosis polyposis coli downregulated 1-positive
cancer-associated fibroblasts (CAFs) via activation of the
PI3K/AKT/hypoxia-inducible factor-1α (HIF-1α) signaling cascade.
Lactate is then exported to the microenvironment and transported
into cancer cells, where it induces the lactylation of small
nuclear ribonucleoprotein polypeptide A, thereby regulating
splicing of AR and generating AR splicing variant 7 (ARV7)
(34). Although ADT can induce
androgen-sensitive PCa cells to enter the senescent state, a
previous study has shown that the continuous increase of active
ARV7 is associated with the escape of PCa from cell senescence
(35). ARV7 can directly bind to
the SKP2 promoter and activate its transcription, promoting the
proteasomal degradation of p27 protein and subsequent
G1/S transition, thereby achieving aging escape
(36). Li et al (37) revealed that ADT, by inhibiting AR
signaling, leads to the upregulation of macrophage migration
inhibitory factor (MIF) expression, which in turn promotes PCa cell
proliferation by upregulating AMP deaminase 2 expression. Although
AR negatively regulates MIF expression, its splice variant ARV7
does not (37). A study has found
that B-cell lymphoma 2 (BCL2) is almost universally upregulated in
ADT treatment castration-sensitive PCa cells, and BCL2 in turn
mediates bidirectional signaling between AR-BCL2 and PI3K pathway
through non-classical functions, driving the transformation of
hormone sensitive to castration resistant phenotype (38). Neuroendocrine differentiation is
another mechanism, where PCa cells transition to a neuroendocrine
phenotype, which is less dependent on androgen signaling (39).
Immunotherapy resistance
Immunotherapy, including immune checkpoint
inhibitors, has shown promise in the treatment of advanced PCa, but
its efficacy is limited by factors such as drug resistance
(40). Further research is needed
to identify novel biomarkers and therapeutic strategies to enhance
the efficacy of immune checkpoint inhibitors in
castration-resistant PCa.
Tumor microenvironment and
immunosuppression
Immunosuppressive cells such as Tregs, MDSCs and
tumor-associated macrophages are often enriched in the tumor
microenvironment, creating an immunosuppressive niche that hinders
the efficacy of immune checkpoint inhibitors (41–43).
Novysedlak et al (41)
highlighted the role of MDSCs in promoting an immunosuppressive
environment in PCa, which can limit the effectiveness of immune
checkpoint inhibitors.
Genetic and epigenetic
alterations
Genetic mutations and epigenetic modifications can
drive resistance to immunotherapy. For instance, mutations in DNA
repair genes, such as breast cancer susceptibility gene (BRCA) 2
and ataxia telangiectasia-mutated gene (ATM), can lead to genomic
instability, which may influence the immune landscape of prostate
tumor (44–46).
Immune checkpoint inhibitors and
resistance mechanisms
PD-1/programmed death-ligand 1 (PD-L1) immune
checkpoint inhibitors have shown promise in a variety of cancers;
however, their efficacy has been limited in PCa. PCa resistance
mechanisms include low tumor mutational burden and lack of PD-1
expression, which are often observed in patients (47,48).
In addition, the presence of immunosuppressive cytokines, such as
transforming growth factor-β (TGF-β) and IL-10, contribute to the
resistance of prostate cancer to immune checkpoint inhibitors by
constructing an immunosuppressive tumor microenvironment and
inhibiting the function of effector T cells (49).
Resistance to chemotherapy
Chemotherapy remains a cornerstone in the management
of advanced PCa; however, resistance to chemotherapeutic agents is
a significant challenge that hampers treatment efficacy. This
section delves into the mechanisms underlying chemotherapy
resistance in PCa, with a focus on specific drugs and associated
signaling pathways.
Docetaxel resistance
Docetaxel is the first-line chemotherapeutic agent
for mCRPC. However, resistance to docetaxel is common and driven by
multiple mechanisms. One key factor is the overexpression of
ATP-binding cassette (ABC) transporters, such as ABCB1
(P-glycoprotein), which efflux cytotoxic drugs from cancer cells,
thereby reducing intracellular drug concentration and efficacy
(50). Alterations in microtubule
dynamics and upregulation of survival pathways, such as the
PI3K/AKT/mTOR pathway, contribute to docetaxel resistance by
promoting cell survival and proliferation (51). A study has shown that activation of
the AKT pathway can phosphorylate and stabilize myeloid cell
leukemia 1, an anti-apoptotic protein, thereby inhibiting apoptosis
induced by docetaxel (52).
Furthermore, the expression of heat shock proteins can stabilize
oncogenic proteins and protect cancer cells from docetaxel-induced
stress (53). Chemotherapy-induced
neurotoxicity remains a major clinical issue, with increasing
evidence pointing to the role of cellular stress and survival
pathways in mediating resistance (54). These findings suggest that
strategies targeting these pathways may provide new therapeutic
opportunities to overcome chemotherapy resistance in PCa.
Cabazitaxel resistance
Cabazitaxel is used as second-line therapy for
patients who have progressed to docetaxel. Resistance to
cabazitaxel can arise through mechanisms similar to docetaxel
resistance, including overexpression of ABC transporters and
activation of survival pathways (55). Additionally, genetic alterations in
the AR pathway contribute to cabazitaxel resistance. For example,
mutations in the AR gene can lead to constitutive activation of the
receptor, promoting cell survival and proliferation independently
of androgen levels (56).
Moreover, upregulation of the Wnt/β-catenin signaling pathway has
been implicated in cabazitaxel resistance by enhancing cell
survival and promoting an EMT phenotype, which is associated with
increased metastatic potential and drug resistance (57).
Cisplatin resistance
Cisplatin is used in combination regimens for
aggressive PCa variants. Resistance to cisplatin can be attributed
to several mechanisms, including increased DNA repair capacity,
reduced drug uptake and the activation of survival pathways.
Specifically, upregulation of DNA repair proteins, such as excision
repair cross-complementation group 1 and BRCA 1/2, can enhance the
repair of cisplatin-induced DNA damage, thereby reducing the
cytotoxic effects of the drug (58). Additionally, activation of the
nuclear factor kappa-B (NF-κB) pathway can promote cell survival by
upregulating anti-apoptotic genes, thereby contributing to
cisplatin resistance (59).
Similarly, the expression of multidrug resistance-associated
protein 2 can reduce intracellular cisplatin levels by effluxing
the drug, thereby diminishing its efficacy (60).
Mitoxantrone resistance
Mitoxantrone is an anthracycline derivative widely
used in the treatment of mCRPC. Resistance to mitoxantrone has been
linked to alterations in the drug efflux and DNA repair pathways.
Specifically, upregulation of ABC transporters, such as ABCB1
(P-glycoprotein), can lead to increased drug efflux, reducing
intracellular drug concentration and efficacy (61). Additionally, mitoxantrone
resistance may be associated with defects in DNA damage response.
For instance, mutations or overexpression of genes involved in the
DNA repair machinery, such as BRCA2, can promote resistance by
enhancing the repair of mitoxantrone-induced DNA damage (62).
Epirubicin resistance
Epirubicin, another anthracycline antibiotic, is
used in combination regimens for PCa treatment. Resistance to
epirubicin often involves mechanisms similar to those of
mitoxantrone. Overexpression of multidrug resistance proteins and
breast cancer resistance protein notably reduces intracellular
epirubicin levels (63). Moreover,
alterations in topoisomerase II, which is the primary target of
epirubicin, can lead to resistance. Mutations or downregulation of
topoisomerase II can impair the ability of the drug to induce DNA
strand breaks, thereby reducing its cytotoxic effects (64).
Vinblastine resistance
Vinblastine, a vinca alkaloid, is used in various
chemotherapy regimens for PCa treatment. Vinblastine resistance is
often associated with changes in tubulin dynamics. Specifically,
mutations or overexpression of β-tubulin isotypes can affect the
binding affinity of vinblastine for tubulin, thereby reducing its
ability to inhibit microtubule polymerization (65). Additionally, similar to docetaxel,
the activation of pro-survival pathways such as the
mitogen-activated protein kinase (MAPK) pathway can contribute to
resistance by promoting cell survival and proliferation (66).
Mechanisms involving tumor
microenvironment
The tumor microenvironment also plays a crucial role
in resistance to chemotherapy. The presence of CAFs can promote
resistance by secreting cytokines and growth factors that activate
the survival pathways in cancer cells. For example, CAFs secrete
IL-6, which activates the Janus kinase 2 (JAK2) signal transducer
and activator of transcription 3 (STAT3) pathway, leading to
increased cell survival and resistance to chemotherapy (67). Additionally, the hypoxic
microenvironment within tumors can induce the expression of HIF-1α,
which promotes EMT and activates survival pathways, thereby
contributing to chemotherapy resistance (68).
Mechanisms of gut microbiota in PCa
treatment resistance
Gut microbiota has emerged as a critical player in
the modulation of treatment resistance in PCa. Accumulating
evidence suggests that specific metabolites produced by gut
microbiota can influence the efficacy of ADT, chemotherapy and
immunotherapy, thereby contributing to treatment resistance
(Table II; Fig. 2). This section will delve into the
detailed mechanisms by which key metabolites from the gut
microbiota impact treatment resistance in PCa, with a particular
focus on distinguishing between ADT, chemotherapy and immunotherapy
resistance mechanisms.
 | Figure 2.Mechanism of gut microbiota
metabolites in PCa treatment resistance. This diagram illustrates
the role of gut microbiota metabolites in modulating PCa treatment
resistance (Figdraw; www.figdraw.com; ID, PISAAda91d). It illustrates how
gut microbiota-derived metabolites (SCFAs, TMAO, I3C, LPC, PAG,
bile acids and histamine) modulate resistance to ADT, chemotherapy
and immunotherapy through AR signaling, DNA repair, immune cell
function, and key pathways including Wnt/β-catenin, PI3K/AKT and
p38/HMOX1. ACSS2, acetyl-coa synthetase 2; AR, androgen receptor;
ARV7, androgen receptor splicing variant 7; ATM/ATR, ataxia
telangiectasia-mutated/ataxia telangiectasia and rad3-related
protein; CCL20, c-c motif chemokine ligand 20; CCR6, c-c chemokine
receptor type 6; CB2, cannabinoid receptor 2; CCNG2, cyclin g2;
CoA, coenzyme a; c-Myc, myc proto-oncogene; DNA-PKcs, DNA-dependent
protein kinase catalytic subunit; FXR, farnesoid × receptor; Glo1,
glyoxalase 1; H1/2R, histamine h1 receptor/h2 receptor; HDAC,
histone deacetylase; HMOX1, heme oxygenase-1; I3C (DIM),
indole-3-carbinol (3,3’-diindolylmethane); JAK2, Janus kinase 2;
STAT3, signal transducer and activator of transcription 3; Nrf2,
nuclear factor erythroid 2-related factor 2; Glo1, glyoxalase 1;
LPC, lysophosphatidylcholine; LPCAT1, lysophosphatidylcholine
acyltransferase 1; NF-κB, nuclear factor κB; MAPK,
mitogen-activated protein kinase; HMOX1, heme oxygenase-1; PAG,
phenylacetylglutamine; PC, phosphatidylcholine; PI3K,
phosphoinositide 3-kinase; PSA, prostate-specific antigen; RAD51,
DNA repair protein RAD51 homolog 1; SCFAs, short-chain fatty acids;
TGR5, g protein-coupled bile acid receptor 1; TLR3, toll-like
receptor 3; TMAO, trimethylamine n-oxide; Wnt, wingless-related
integration site. |
 | Table II.Mechanisms of gut microbiota
metabolites in PCa treatment resistance. |
Table II.
Mechanisms of gut microbiota
metabolites in PCa treatment resistance.
| A, Short chain
fatty acids |
|---|
|
|---|
| Impact on
treatment | Pathway
involved | Outcome | (Refs.) |
|---|
| Promotes ADT
resistance | 1. HDAC inhibition
leads to histone hyperacetylation at AR gene promoter, which leads
to upregulation of AR and ARV7. | 1. Drives CRPC
progression under low-androgen conditions. | (69–71) |
|
| 2. Increases
peripheral androgen synthesis. |
|
|
|
| 2. Upregulates
17β-HSD and 3β-HSD in intestinal epithelial cells and
hepatocytes. |
|
|
| Counteracts ADT
resistance | Inhibits the
JAK2/STAT3/Nrf2/Glo1 pathway, thereby increasing methylglyoxal
production. | Induces
mitochondrial damage and apoptosis in PCa cells
(concentration-dependent). | (72) |
| Promotes
chemotherapy resistance | 1. Activates
ATM/ATR-mediated DNA damage repair pathway. | Reduces
docetaxel-induced apoptosis; enhances tolerance to | (73,74) |
|
| 2. Upregulates
acetyl-CoA synthetase 2, which enhances acetyl-CoA production and
promotes neuroendocrine differentiation. | chemotherapeutic
agents. |
|
| Promotes
immunotherapy resistance | 1. Induces
protective autophagy in PCa cells. |
|
|
|
| 2. Activates
Toll-like receptor 3/NF-κB/MAPK pathway, which | Establishes
immunosuppressive microenvironment; reduces | (69) |
|
| upregulates CCL20
and recruits CCR6+ macrophages, thereby suppressing
CD8+ T cell function. | efficacy of immune
checkpoint inhibitors. |
|
|
| B,
Trimethylamine N-oxide |
|
| Impact on
treatment | Pathway
involved | Outcome | (Refs.) |
|
| Promotes ADT
resistance | Upregulates
p38/HMOX1 pathway, which increases AR and prostate-specific antigen
expression. | Enhances
antioxidant adaptation under androgen deficiency; promotes cell
proliferation. | (76,77) |
| Promotes
chemotherapy resistance | Reduces apoptotic
response of PCa cells to chemotherapeutic agents. | Decreases
chemosensitivity. | (76) |
|
| C, I3C |
|
| Impact on
treatment | Pathway
involved | Outcome | (Refs.) |
|
| Anti-tumor (via
DIM) | 1. Acts as CB2
receptor agonist. | 1. Modulates tumor
immune microenvironment; inhibits | (82–85) |
|
| 2. Inhibits
PI3K/AKT and NF-κB pathways. | proliferation. |
|
|
| 3. Reprograms
glycolysis, TCA cycle and lipid metabolism in | 2. Induces
apoptosis; suppresses invasion. |
|
|
| prostate
tissue. | 3. Inhibits PCa
cell energy metabolism and biosynthesis. |
|
|
| D, LPC |
|
| Impact on
treatment | Pathway
involved | Outcome | (Refs.) |
|
| Promotes ADT
resistance | Upregulates LPCAT1
expression. | Enhances DNA repair
pathways; promotes survival under low-androgen conditions. | (93) |
|
| E,
Phenylacetylglutamine |
|
| Impact on
treatment | Pathway
involved | Outcome | (Refs.) |
|
| Potential to
enhance treatment sensitivity | Inhibits
Wnt/β-catenin signaling pathway by upregulating CCNG2
expression. | Reduces expression
of downstream target genes, such as c-Myc and cyclin D1. | (94) |
|
| F, Bile
acids |
|
| Impact on
treatment | Pathway
involved | Outcome | (Refs.) |
|
| Associated with PCa
treatment | Mechanism remains
to be determined. | May modulate immune
microenvironment and lipid metabolism; role in treatment resistance
unclear. | (109–111) |
|
| G,
Histamine |
|
| Impact on
treatment | Pathway
involved | Outcome | (Refs.) |
|
| Promotes PCa
progression | Activates H1
receptor. | Promotes tumor
growth under high-fat diet conditions. | (113) |
| Potential anti-PCa
activity | Long-term H2
receptor antagonist use associated with reduced PCa risk. | Mechanism remains
to be determined. | (117) |
SCFAs
SCFAs, including acetate, propionic acid and butyric
acid, are produced by gut microbiota such as Firmicutes and
Bacteroidetes by fermenting dietary fiber (69). These metabolites regulate cellular
metabolism and signaling pathways, thereby influencing PCa
progression and treatment response. Recent studies on metabolic
reprogramming have shown that tumor cells often rewire their
metabolism to resist immune responses, similar to findings observed
in lung cancer (70). In PCa,
SCFAs may influence disease progression and treatment resistance
through androgen metabolism and the production of procarcinogenic
metabolites.
ADT is the cornerstone therapy for treating advanced
PCa. However, a number of patients eventually develop resistance to
ADT, leading to the progression of CRPC. An in vitro study
revealed that treatment with butyrate and acetate markedly
upregulated both the protein and mRNA levels of the AR and its
splice variant ARV7 in PCa cells, suggesting that SCFAs may
directly regulate AR gene transcription (69). Subsequent histone deacetylase
(HDAC) activity assays and chromatin immunoprecipitation analyses
further confirmed that butyrate and acetate function as HDAC
inhibitors, inducing histone hyperacetylation at the AR gene
promoter region and thereby directly enhancing the expression of AR
and its splice variants (69).
Furthermore, Bui et al (71) demonstrated that treatment of
intestinal epithelial cells or hepatocytes with SCFAs, particularly
butyrate, notably upregulated the expression and activity of
17β-hydroxysteroid dehydrogenase and 3β-hydroxysteroid
dehydrogenase. These two enzymes are the key rate-limiting enzymes
responsible for converting precursors into active androgens.
Subsequent animal experiments further confirmed that SCFAs,
particularly butyrate, contributes to peripheral androgen
synthesis, thereby promoting ADT resistance in PCa (71). By contrast, Hsia et al
(72) demonstrated that butyrate
(2 to 4 mM) increases methylglyoxal production by inhibiting the
JAK2/STAT3/nuclear factor erythroid 2-related factor 2/glyoxalase 1
pathway, thereby inducing mitochondrial damage and subsequent
apoptosis in PCa cells. This inhibitory effect on PCa was
concentration-dependent, and no pro-proliferative effect was
observed at lower concentrations. This finding contrasts with
previous reports (69,71) revealing the bidirectional role of
SCFAs, particularly butyrate, in PCa therapy. We hypothesize that
the absence of a pro-proliferative effect at low concentrations may
be attributed to a lower threshold for this functional switch.
Future studies aimed at identifying the molecular switch that
precisely regulates this threshold hold promise for transforming
butyrate into a manipulable and precise therapeutic tool.
Chemotherapy is another important treatment modality
for PCa, particularly in patients with metastatic disease. However,
resistance to chemotherapy is a major challenge limiting the
effectiveness of this treatment. SCFAs have been shown to influence
the sensitivity of PCa cells to chemotherapeutic agents through
various mechanisms. Zhong et al (73) demonstrated that butyrate
pretreatment markedly reduced docetaxel-induced apoptosis in PCa
cells, concomitant with activation of the ATM/ataxia telangiectasia
and Rad3-related protein (ATR) pathway. Notably, the research team
experimentally showed that pharmacological inhibition of the
ATM/ATR signaling axis markedly reversed butyrate-induced docetaxel
resistance (73). These findings
indicate that butyrate confers docetaxel resistance in PCa cells by
activating the ATM/ATR-mediated DNA damage repair pathway. As
demonstrated by Gao et al (74) through both in vitro cell
models and in vivo experiments, acetate treatment notably
promotes neuroendocrine differentiation in PCa cells, a critical
phenotype associated with acquired therapeutic resistance (74). The study further revealed that
acetate upregulates the expression of acetyl-CoA synthetase 2,
thereby enhancing acetyl-coenzyme A production and subsequently
activating transcriptional programs linked to neuroendocrine
differentiation. This cascade ultimately leads to enhanced tumor
tolerance to chemotherapeutic agents such as docetaxel (74). SCFAs promote the formation of
chemotherapy resistance in PCa through multiple independent yet
complementary signaling pathways. Rather than acting through a
single mechanism, SCFAs establish a multifaceted defense system
that synergistically enhances tumor cell tolerance to
chemotherapeutic agents such as docetaxel.
Immunotherapy, including immune checkpoint
inhibitors, has shown promise for the treatment of advanced PCa.
However, the response rate to immunotherapy remains low and many
patients develop resistance (2).
SCFAs have emerged as important modulators of the immune system,
particularly in PCa immunotherapy. A study utilizing co-culture
systems and a PCa mouse model systematically investigated the
effects of SCFAs on the tumor immune microenvironment (69). The findings demonstrated that SCFAs
not only directly induced protective autophagy in PCa cells,
thereby shielding them from immune attack, but also, more
importantly, stimulated the expression and secretion of C-C motif
chemokine ligand 20 (CCL20) in PCa cells via the Toll-like receptor
3/NF-κB/MAPK signaling pathway. The secreted CCL20 subsequently
recruited C-C chemokine receptor type 6-expressing macrophages into
the tumor microenvironment (69).
These polarized macrophages further suppress the infiltration and
cytotoxic function of CD8+ T cells through the secretion
of inhibitory cytokines, thereby establishing an immunosuppressive
microenvironment. This mechanism is speculated to be a key factor
contributing to the poor response of PCa to immune checkpoint
inhibitors such as PD-1/PD-L1 antibodies (69). Based on bioinformatical analyses,
Matsushita et al (75)
hypothesized that gut microbiota-derived butyrate may promote the
progression of high-risk PCa by facilitating Tregs differentiation
and subsequently suppressing antitumor immune responses. However,
the conclusions drawn from the bioinformatics analysis still
require further experimental research to clarify the specific
pathways and effects.
SCFAs exert a bidirectional regulatory role in PCa.
On one hand, they contribute to therapeutic resistance through
multiple mechanisms, including enhancing AR signaling, promoting
androgen synthesis, activating DNA damage repair pathways and
inducing immunosuppression, thereby compromising the efficacy of
endocrine therapy, chemotherapy and immunotherapy. On the other
hand, specific SCFAs such as butyrate, at appropriate
concentrations, can induce apoptosis in PCa cells, exerting
antitumor effects. Future research should focus on elucidating the
molecular switch governing this functional duality, with the goal
of transitioning SCFAs from risk factors to precise therapeutic
tools.
Trimethylamine N-oxide (TMAO)
TMAO is produced by Escherichia coli,
Enterobacter aerogenes and other gut microbiota by metabolizing
dietary choline and carnitine (73). This metabolite can influence
cellular stress responses and inflammation, thereby promoting
resistance to treatment.
Zhou et al (76) treated PCa cell lines with varying
concentrations of TMAO and observed that cell proliferation
markedly increased in a concentration-dependent manner. Through
mechanistic investigation using molecular biology techniques, the
study revealed that TMAO treatment upregulated the p38/heme
oxygenase-1 (HMOX1) pathway and increased the expression of AR and
its downstream target genes, such as prostate-specific antigen
(76). Activation of HMOX1 confers
antioxidant adaptation in cells under androgen deficiency
conditions and enhances the resistance of PCa cells to ADT
treatment (77). Furthermore, the
study examined the impact of TMAO pretreatment on chemosensitivity,
demonstrating that higher concentrations of TMAO (≥200 µM) notably
reduced the apoptotic response of PCa cells to chemotherapeutic
agents, including docetaxel (76).
Studies on other cancer types have shown that TMAO
can affect cellular metabolism and signaling pathways associated
with drug resistance. For example, TMAO has been shown to affect
mitochondrial function and reactive oxygen species levels, which
are key determinants of sensitivity (69). Given the metabolic changes
associated with TMAO, this metabolite may affect therapeutic
efficacy in PCa in a similar manner by modulating the cellular
redox status and survival pathways. TMAO can promote the
polarization of macrophages to the M2 phenotype, leading to an
immunosuppressive tumor microenvironment, thereby reducing the
efficacy of immunotherapy (78).
Simultaneously, TMAO impairs the function of dendritic cells,
reducing their ability to present antigens and activate T cells,
leading to a weakened adaptive immune response (79). Since immune cells are key players
in the treatment of PCa, we hypothesize that the effects of TMAO on
immune cells may play the same role in the treatment of PCa.
However, further studies are needed to confirm whether these
effects play a role in PCa.
Although direct clinical investigations into the
role of TMAO in promoting resistance are still in their preliminary
stages, existing evidence from population-based studies and
multi-omics analyses suggests a potential link. A prospective
analysis of the PLCO cancer screening trial cohort by Reichard
et al (80) demonstrated
that elevated circulating TMAO levels were markedly associated with
an increased risk of lethal PCa, suggesting that TMAO may actively
participate in disease progression and lethal transformation.
Indole-3-carbinol (I3C)
I3C, derived from cruciferous vegetables and
produced by Bacteroides fragilis and Clostridium
sporogenes, affects the expression of genes involved in cell cycle
regulation and apoptosis (81).
The in vivo activity of I3C is largely attributed to its
dimeric derivative, 3,3′-diindolylmethane (DIM). To the best of our
knowledge, Tucci et al (82) were the first to report that DIM
acts as a cannabinoid receptor 2 (CB2) agonist in both
androgen-dependent and androgen-independent PCa cell models. The
experiment revealed that activation of the CB2 receptor can
modulate the tumor immune microenvironment and inhibit tumor cell
proliferation, suggesting that DIM exerts its anti-PCa effects
through the CB2 signaling axis (82).
DIM has been shown to inhibit multiple oncogenic
pathways, including PI3K/AKT and NF-κB, thereby inducing apoptosis
and suppressing invasion (83,84).
Although various carcinogenic pathways have been identified in PCa,
it remains to be further investigated whether the multiple
carcinogenic pathway inhibition mediated by DIM can be effective in
PCa. An animal study has revealed that dietary supplementation with
I3C notably alters the metabolic profile of mouse prostate tissue,
involving reprogramming of intermediates related to glycolysis, the
tricarboxylic acid cycle and lipid metabolism (85). Such metabolic modulation may impact
the energy metabolism and biosynthesis of PCa cells, thereby
inhibiting tumor growth. In addition, I3C has been demonstrated to
upregulate PTEN expression (86)
and inhibit the Wnt/β-catenin pathway (87) in other tumor types (for example,
colorectal and esophageal cancers). Among the multiple oncogenic
pathways inhibited by I3C, mechanistic pathways, including the
PI3K/AKT, NF-κB and Wnt/β-catenin pathways, were also activated in
PCa. Among the multiple oncogenic pathways inhibited by I3C,
mechanistic pathways including PI3K/AKT, NF-κB and Wnt/β-catenin
pathways were also activated in PCa. The present study hypothesizes
that I3C may enhance the efficacy of existing therapies through
multiple pathways.
As naturally occurring dietary compounds, I3C and
its derivatives offer favorable safety profiles and accessibility.
Future research should systematically evaluate the antitumor
activity of I3C/DIM in CRPC and different molecular subtypes of PCa
models, elucidate their interplay with AR signaling, lipid
metabolism and the immune microenvironment, and explore their
synergistic effects and potential to overcome resistance when
combined with ADT, novel endocrine agents and immunotherapy.
Lysophosphatidylcholine (LPC)
Bacteria such as Escherichia, Bilophila,
Enterorhabdus and Gordonibacter may produce LPC by
secreting phospholipases (for example, phospholipase D or
phospholipase A2), which catalyze the hydrolysis of dietary or
host-derived phosphatidylcholine (PC) as a substrate (88). Buszewska-Forajta et al
(89) reviewed the application of
lipidomics in PCa diagnosis and highlighted the potential of LPC as
a diagnostic biomarker. Subsequently, the same group utilized
matrix-assisted laser desorption/ionization time-of-flight mass
spectrometry (MALDI-TOF/MS) to analyze urinary metabolites from
patients with PCa and established a diagnostic model based on LPC
and other lipid metabolites, demonstrating favorable discriminatory
performance (90). Similarly, Li
et al (91) employed the
acidified Bligh-Dyer method to extract lipids from urine samples,
followed by detection and analysis using MALDI-TOF/MS. Their
results demonstrated that the urinary PCs/LPC ratio was
significantly higher in patients with PCa than in those with benign
prostatic hyperplasia in both the discovery and validation cohorts
(P<0.001) (91). These findings
reveal that urinary LPC and its ratio to PC (PC/LPC) may serve as
potential non-invasive biomarkers for the diagnosis and metabolic
stratification of PCa.
Lysophosphatidylcholine acyltransferase 1 (LPCAT1)
catalyzes the conversion of LPC to PC and serves as a key enzyme in
maintaining the homeostasis of membrane phospholipids (92). Using fecal microbiota
transplantation (FMT), Liu et al (93) demonstrated that transferring fecal
microbiota from patients with mCRPC into mice led to an increased
abundance of intestinal Ruminococcus, marked upregulation of
LPCAT1 expression in prostate tissues, and elevated levels of the
DNA repair proteins (DNA repair protein RAD51 homolog 1 and
DNA-dependent protein kinase catalytic subunit). Lipidomic analysis
further revealed markedly increased fecal levels of LPC and PC
following CRPC microbiota transplantation, suggesting that gut
microbiota dysbiosis may induce reprogramming of intestinal lipid
metabolism (93). This study
provides preliminary evidence for the involvement of the ‘gut
microbiota-LPCAT1-DNA repair axis’ in PCa resistance; however, the
precise molecular mechanisms through which LPCAT1 regulates DNA
repair, as well as the role of its substrate LPC in this process,
warrant further investigation.
Collectively, these findings position LPC and its
metabolic regulator LPCAT1 at the intersection of microbial
ecology, lipid remodeling and DNA repair in PCa. While LPC-related
biomarkers show promise for non-invasive stratification, the
functional role of LPC within the tumor microenvironment,
particularly whether it actively modulates treatment response,
remains unclear. Future studies integrating spatial metabolomics
with microbiota targeted interventions are warranted to determine
if LPC represents not only a diagnostic tool but also a potential
therapeutic target.
Phenylacetylglutamine (PAG)
PAG is an end product of phenylalanine metabolism
dependent on gut microbiota. It is generated by intestinal
microorganisms converting dietary phenylalanine to phenylacetic
acid, which combines with glutamine in the liver to form PAG and is
released into the circulation (94). In the field of cardiovascular
diseases, PAG activates adrenergic receptors, promoting high
platelet reactivity and inflammatory responses, notably increasing
the risks of complications such as myocardial infarction, stroke
and heart failure (95–98).
In the field of oncology, the role of PAG presents a
different aspect. The latest research has revealed that it exerts a
protective effect in PCa. Lv et al (94) demonstrated through in vitro
cell experiments and in vivo animal models that the gut
microbiota-derived metabolite PAG inhibits PCa progression by
upregulating cyclin G2 expression, thereby suppressing the
Wnt/β-catenin signaling pathway and its downstream target genes,
including c-Myc and cyclin D1 (94). The abnormal activation of the
Wnt/β-catenin pathway has been confirmed to be involved in the
formation of castration-resistant PCa, and is closely related to
the resistance to various treatments (such as AR signal inhibitors
and chemotherapy) (99,100). Therefore, the inhibition of the
Wnt pathway mediated by PAG may enhance the sensitivity of existing
treatment methods by weakening the characteristics of tumor stem
cells and reversing EMT, thereby delaying the occurrence of drug
resistance.
In recent years, the role of PAG in tumor biology
has gradually attracted attention. However, the expression level of
PAG in PCa tissues and its association with patient prognosis
remain to be clarified. Future studies need to further elucidate
the receptor targets of PAG in the PCa microenvironment, evaluate
the correlation between its serum or tumor local levels and the
patient treatment response and drug resistance time, and explore
the feasibility of modulating the intestinal microbiota or
supplementing PAG to enhance existing treatments while preventing
the risk of cardiovascular diseases.
Bile acids
Bile acids are amphipathic steroidal compounds
synthesized from cholesterol in the liver. The gut microbiota
enzymatically converts primary bile acids into secondary bile acids
through deconjugation via bile salt hydrolases and subsequent
7α-dehydroxylation. Beyond their role in lipid metabolism, these
secondary bile acids function as signaling molecules that activate
the farnesoid X receptor (FXR) and the Takeda G protein-coupled
receptor 5 (TGR5), thereby participating in the regulation of host
metabolism, immunity and inflammation (101–103).
In colorectal cancer, the abnormal bile acid
metabolism mediated by the gut microbiota can also inhibit the
Wnt/β-catenin pathway (104).
This is similar to the role of PAG, but it is still unknown whether
it can inhibit the treatment resistance of PCa. Furthermore, bile
acids and their metabolites also play a significant role. For
instance, 3-oxolithocholic acid inhibits colorectal cancer
progression by modulating T cell differentiation (105); microbiota-derived bile acids
activate TGR5 to induce the infiltration of MDSCs into the liver,
thereby promoting colorectal cancer liver metastasis (106); conjugated bile acids impair
CD8+ T cells function in hepatocellular carcinoma
(107); aldo-keto reductase
family 1 member D1-mediated bile acid metabolism enhances natural
killer (NK) cell cytotoxicity and suppresses hepatocellular
carcinoma progression (108).
Therefore, bile acids have shown a significant impact on immune
cells in gastrointestinal tumors. The immune microenvironment is a
crucial aspect in cancer treatment. Bile acids are expected to
become an important factor in regulating the immune
microenvironment of PCa. However, further research is still needed
to explore its effects on the immune cells in PCa tissues.
Previous studies have emphasized the significance of
bile acids as signaling molecules in the treatment of PCa (109,110). The study by Kure et al
(111) indicates that ADT
treatment markedly alters the abundance of bacteria involved in
bile acid metabolism in the gut microbiota. This discovery provides
indirect evidence that bile acids are involved in the efficacy and
drug resistance formation of ADT.
We hypothesize that, on one hand, bile acids may
modulate lipid metabolism and inflammatory responses through
nuclear receptors such as FXR or TGR5, as well as other pathways,
with lipid metabolic reprogramming being a characteristic feature
of PCa progression and castration resistance. On the other hand,
the immunomodulatory effects of bile acids on immune cells,
including T cells and MDSCs, may reshape the immunosuppressive
tumor microenvironment in PCa. However, their role in PCa therapy
remains to be elucidated. Moving forward, it is essential to
systematically characterize the bile acid profiles of patients with
PCa and integrate functional experiments to clarify the specific
mechanisms by which they modulate AR signaling. It is expected to
become a key factor in the treatment of PCa.
Histamine
Histamine is a multifunctional bioactive amine
synthesized from histidine via catalysis by histidine decarboxylase
(HDC), and exerts a broad spectrum of physiological and
pathological effects through four G protein-coupled receptors
(H1-H4 receptors) (112).
Matsushita et al (113) demonstrated that high-fat diet
feeding notably increased histamine levels in mouse prostate
tissues, and that histamine promotes PCa cell proliferation and
tumor growth through activation of the H1 receptor. Notably,
administration of the H1 receptor antagonist loratadine or genetic
ablation of HDC markedly suppressed high-fat diet-induced tumor
promotion. The study links histamine signaling to lipid metabolic
reprogramming and diet-associated tumor progression, suggesting
that histamine serves as a key node connecting environmental
factors with the biological behavior of PCa (113). Furthermore, a case-control study
by Wang et al (114)
suggested that long-term use of H2 receptor antagonists is
associated with a reduced risk of PCa. This raises the possibility
that antihistamines may possess potential anti-PCa activity,
although whether the underlying mechanism depends on histamine
receptor blockade remains to be determined.
In recent years, numerous researchers have studied
the role of histamine receptors in oncology. However, whether they
can have an impact on the treatment of PCa requires further
experimental verification. Lauretta et al (115) reviewed the therapeutic potential
of the H3 receptor in oncology, noting that it may influence the
tumor microenvironment by modulating neurotransmitter release and
immune cell function. Li et al (116) revealed that the allergic mediator
histamine induces PD-L1 expression and T helper 2 cell-type
inflammation via activation of the H1 receptor on tumor-associated
macrophages, thereby conferring resistance to cancer immunotherapy.
These findings suggest that the functions of histamine receptors
are subtype-specific.
Research on the role of histamine signaling in PCa
therapy is still in its infancy. Future efforts should
systematically characterize the expression and function of each
histamine receptor subtype in PCa cells and the tumor
microenvironment, and elucidate their crosstalk with AR signaling
and lipid metabolism. Concurrently, leveraging the safety and
accessibility of existing antihistamines, it is worthwhile
exploring their clinical translational potential in combination
with ADT, novel endocrine agents or immunotherapy to enhance
sensitivity and overcome resistance.
Potential therapeutic strategies
Due to the possible role of the gut microbiota in
PCa treatment resistance, an increasing number of potential
therapeutic measures related to the gut microbiota have become the
focus of research.
Probiotics
Several probiotic strains have been identified for
their potential benefits in modulating gut microbiota and
influencing PCa treatment outcomes.
Lactobacillus acidophilus
This strain produces lactic acid, which lowers the
pH of the gut and inhibits the growth of harmful bacteria. It also
enhances the production of SCFAs such as butyrate, which have
anti-inflammatory and anticancer properties (24,79).
Lactobacillus rhamnosus
This strain has been shown to improve gut barrier
function and reduce the translocation of harmful bacteria and their
metabolites into systemic circulation. It also enhances the
activity of NK cells and Tregs, which play crucial roles in immune
surveillance and cancer cell elimination (117).
Bifidobacterium bifidum
This strain produces antimicrobial substances that
inhibit the growth of pathogenic bacteria. It also modulates the
immune system by promoting the production of cytokines that enhance
the function of the immune cells (118).
Bifidobacterium longum
This strain has been shown to produce SCFAs and
other metabolites that influence the tumor microenvironment and
reduce inflammation, thereby potentially improving PCa outcomes
(119).
Streptococcus thermophilus
This strain is commonly used in combination with
other probiotics (for example, Bifidobacterium and
Bacteroides dorei) (120).
It enhances the production of lactic acid and SCFAs, which can
inhibit the growth of harmful bacteria and promote healthy gut
microbiota balance (121).
Future research should focus on identifying
specific probiotic strains and combinations. Personalized probiotic
therapies based on the characteristics of an individual's gut
microbiota may be a promising approach. In conclusion, probiotics
offer a novel and promising therapeutic strategy for regulating the
gut microbiota of patients with PCa by combining probiotics with
other treatments, such as immunotherapy or ADT, which may enhance
their therapeutic benefit in patients with PCa. Probiotics have the
potential to improve prognosis and reduce PCa treatment resistance
by enhancing immune function, reducing inflammation and improving
treatment effectiveness.
Prebiotics
Modulating the gut microbiota using prebiotics is a
promising therapeutic strategy to mitigate this resistance.
Prebiotics are non-digestible food components that selectively
stimulate the growth and activity of beneficial gut bacteria,
thereby improving the health of the host. This section explores the
potential mechanisms and therapeutic applications of specific
prebiotics in the context of resistance to PCa treatment.
Inulin is a fructan that selectively promotes the
growth of beneficial bacteria, such as Bifidobacterium and
Lactobacillus. These bacteria produce SCFAs such as
butyrate, which have anti-inflammatory and anticancer properties
(122). Inulin has been shown to
reduce the levels of pro-inflammatory cytokines in patients with
PCa, potentially lowering the risk of disease progression (79).
Fructooligosaccharides (FOS) selectively stimulate
the growth of beneficial bacteria, leading to the increased
production of SCFAs and enhanced gut barrier function. This reduces
the translocation of harmful bacteria and their metabolites into
systemic circulation (116). FOS
enhance the function of Tregs and NK cells, which play crucial
roles in immune surveillance and cancer cell elimination (118).
Galactooligosaccharides (GOS) promote the growth of
beneficial bacteria, particularly Bifidobacterium species,
which produce SCFAs and other metabolites that modulate the immune
system (24). GOS improve gut
barrier function and reduce systemic inflammation, potentially
enhancing the efficacy of other treatments such as ADT and
immunotherapy (79).
Future research should focus on identifying
specific prebiotic formulations and doses that most effectively
regulate the gut microbiota and improve PCa prognosis. Personalized
prebiotic therapy is also very important, according to the
different selection of different prebiotics in the patient's gut
flora, which may achieve a good effect. Similarly, combining
prebiotics with other treatments such as immunotherapy or ADT may
enhance their therapeutic effects.
FMT
FMT has emerged as a promising therapeutic strategy
for modulating the gut microbiota to overcome treatment resistance
in PCa. Previous studies have highlighted the critical role of the
gut microbiota in influencing the immune response and tumor
progression, suggesting that altering the microbial composition
could enhance the efficacy of existing treatments and mitigate
resistance mechanisms.
Preclinical studies have begun to elucidate the
mechanisms by which FMT may exert antitumor effects. Pernigoni
et al (15) demonstrated,
through metagenomic sequencing, a marked enrichment of bacterial
strains (for example, certain Ruminococcaceae species)
capable of encoding enzymes involved in androgen biosynthesis, such
as androstenedione, in the fecal microbiota of men with CRPC.
Transplantation of this ‘resistance-associated microbiota’ into
murine models enabled tumors to proliferate despite castration
levels of androgens (15).
Conversely, antibiotic-mediated microbiota depletion or
intervention with specific probiotics restored tumor sensitivity to
ADT (15). This provides direct
evidence supporting the rationale for using FMT to replace a
‘resistant’ microbiota. Further research indicates that
combinations of specific compounds, such as icaritin and curcumol,
can inhibit PCa progression by modulating the gut
microbiota-enriching beneficial taxa, which in turn suppresses DNA
methyltransferase 1 expression and activates CD8+ T
cell-mediated antitumor immunity (123). This suggests that FMT may exert
synergistic effects through epigenetic modulation and immune
remodeling. FMT offers a comprehensive and direct approach to
microbiota modulation compared with other interventions targeting
the gut microbiota. Probiotics introduce specific beneficial
bacteria, but may not address overall dysbiosis as effectively as
FMT (124). Additionally, FMT has
the potential to influence a broader range of microbial species and
their interactions, which are essential for restoring a balanced
microbiota ecosystem. By contrast, dietary interventions, although
effective in some cases, may take longer to achieve notable changes
in microbiota composition (79).
Despite the potential of FMT, its clinical
translation remains hindered by multiple obstacles. The foremost
challenge lies in inter-individual variability and the consequent
lack of reproducibility. Host dietary habits and genetic background
may influence the colonization efficacy of FMT, leading to
divergent therapeutic outcomes even when the same donor microbiota
is transplanted into different recipients. Second, safety concerns
cannot be overlooked. If donor screening for FMT is not rigorous,
transplantation of microbiota containing an overabundance of
SCFAs-producing bacteria or harboring pro-tumorigenic functional
genes could paradoxically exacerbate disease progression.
Furthermore, FMT in cancer patients carries the inherent risk of
introducing opportunistic pathogens, particularly in
immunocompromised individuals with advanced disease. Finally,
regulatory and standardization barriers impede clinical
implementation. Currently, the application of FMT in oncology lacks
unified criteria for donor selection, standardized protocols for
fecal material preparation, and consensus on administration routes
and dosages (125). Moreover,
regulatory policies vary considerably across countries and remain
ambiguous, hindering the conduct of large-scale clinical studies
(126).
An ongoing FMT clinical trial (NCT05273255) being
conducted at the University Hospital Zurich (Zurich, Switzerland),
encompassing multiple solid tumors including PCa, aims to restore
treatment sensitivity in recipients by altering their gut
microbiota, thereby identifying specific microbial strains that
could be utilized for future personalized therapies.
In summary, FMT, as a gut microbiota-targeted
intervention strategy, holds promise for reversing treatment
resistance in PCa. Future research should prioritize: i) Validating
its efficacy and safety through multicenter clinical trials; ii)
integrating metagenomic and metabolomic approaches to develop
functionally defined synthetic microbial consortia as alternatives
to whole fecal transplantation, thereby enhancing therapeutic
reproducibility; and iii) elucidating the complex
microbiota-host-tumor interplay to establish a foundation for
precision FMT-based therapies.
Challenges and future perspectives
Despite growing evidence highlighting the critical
role of gut microbiota in PCa treatment resistance, several
challenges remain in translating these findings into clinical
practice. Addressing these challenges and exploring innovative
strategies are essential for advancing our understanding and
improving patient outcomes.
Complexity of the gut microbiota
Gut microbiota is an incredibly complex ecosystem,
with thousands of microbial species and their metabolites
interacting in ways that are not yet fully understood. This
complexity makes it difficult to pinpoint the specific microbial
signatures or pathways that drive treatment resistance in PCa.
While studies have identified certain bacterial taxa associated
with resistance to ADT and immunotherapy, the mechanisms by which
these bacteria exert their effects remain unclear. Future research
should focus on high-resolution microbiome analyses, integrating
metagenomics, metatranscriptomics and metabolomics, to unravel the
intricate interactions between the gut microbiota and PCa.
Clinical translation
Translating microbiome research from the laboratory
to the clinic has several challenges. One major issue is the lack
of standardized protocols for microbiome analysis and intervention.
The preparation and administration of FMT have not yet been
standardized, and rigorous clinical trials are needed to establish
its safety and efficacy in PCa. Additionally, regulatory hurdles
and ethical considerations regarding the use of live microbial
therapies must be addressed. Collaborative efforts among
researchers, clinicians and regulatory bodies are essential to
develop guidelines and frameworks for the safe and effective use of
microbiome-based therapies.
Immune and metabolic interactions
Gut microbiota influences PCa through multiple
pathways, including immune modulation and metabolic alterations.
However, the precise mechanisms by which these pathways interact to
drive treatment resistance remain unclear. For example, while
certain gut bacteria can enhance the efficacy of immune checkpoint
inhibitors by promoting T cell infiltration, others may produce
metabolites that suppress antitumor immunity. Understanding these
complex interactions will require interdisciplinary research
combining immunology, microbiology and bioinformatics. Developing
combination therapies that target both the microbiota and the tumor
microenvironment may offer a more effective approach to overcoming
resistance.
Longitudinal studies and biomarker
development
Most current studies on gut microbiota and PCa are
cross-sectional, providing snapshots of the microbiota at a single
time point. Longitudinal studies are needed to track changes in
microbiota over time and to correlate these changes with treatment
response and disease progression. Additionally, robust biomarkers
that can predict treatment resistance and monitor therapeutic
efficacy are needed. Metabolomic and proteomic approaches may help
identify biomarkers that can guide clinical decision making and
improve patient outcomes.
Novel therapeutic approaches
Emerging therapeutic strategies, such as
probiotics, prebiotics and synthetic microbiota, hold promise for
modulating gut microbiota to overcome treatment resistance. Further
research is required to identify the most effective strains and
formulations. Although certain probiotic strains have shown
potential benefits in modulating the immune system and reducing
inflammation, their long-term effects and interactions with other
treatments remain unclear. Similarly, prebiotics and synthetic
microbiota offer innovative approaches; however, their mechanisms
of action and optimal dosing need to be further researched.
Conclusion
Gut microbiota and their metabolites represent a
hidden but critical factor driving the formation of treatment
resistance in advanced PCa. Metabolites such as SCFAs, TMAO and LPC
are deeply involved in the evolution of resistance to ADT,
chemotherapy and immunotherapy through the regulation of AR
signaling, drug efflux and immune evasion. Future research must
shift from ‘descriptive association’ to ‘mechanistic intervention’,
aiming to target the gut microbiota-metabolite network and thereby
establish a new therapeutic paradigm that improves upon the
traditional tumor-centric view.
Acknowledgements
Not applicable.
Funding
This research was funded by the Shandong Province Medical and
Health Science and Technology Development Plan Project (grant no.
202304051613) and the Science and Technology Program of Yantai
Affiliated Hospital of Binzhou Medical University (grant no.
YTFY2024KYQD01).
Availability of data and materials
Not applicable.
Authors' contributions
JS, YX, HS and ZZ were responsible for the
conception of this study. JS, YX, ZZ and HS were responsible for
literature retrieval, screening and evaluation. JS, HC, PY, YuL,
GZ, YaL, AT and JC were involved in the creation of the images and
the language editing of this article. JS, HC and PY were mainly
responsible for the writing of the article. All authors have read
and approved the final manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ABCB1
|
ATP-binding cassette subfamily B
member 1 (P-glycoprotein)
|
|
ADT
|
androgen deprivation therapy
|
|
AhR
|
aryl hydrocarbon receptor
|
|
AKT
|
protein kinase B
|
|
AR
|
androgen receptor
|
|
ARV7
|
androgen receptor splicing variant
7
|
|
ATM
|
ataxia telangiectasia-mutated
gene
|
|
ATR
|
ataxia telangiectasia and
Rad3-related protein
|
|
BCL2
|
B-cell lymphoma 2
|
|
BRCA2
|
breast cancer susceptibility gene
2
|
|
CAFs
|
cancer-associated fibroblasts
|
|
CB2
|
cannabinoid receptor 2
|
|
CCL20
|
C-C motif chemokine ligand 20
|
|
CRPC
|
castration-resistant prostate
cancer
|
|
mCRPC
|
metastatic CRPC
|
|
DIM
|
3,3′-diindolylmethane
|
|
EMT
|
epithelial-mesenchymal transition
|
|
FMT
|
fecal microbiota transplantation
|
|
FOS
|
fructooligosaccharides
|
|
FXR
|
farnesoid X receptor
|
|
GOS
|
galactooligosaccharides
|
|
HDAC
|
histone deacetylase
|
|
HDC
|
histidine decarboxylase
|
|
HIF-1α
|
hypoxia-inducible factor-1α
|
|
HMOX1
|
heme oxygenase-1
|
|
I3C
|
indole-3-carbinol
|
|
JAK2
|
Janus kinase 2
|
|
LPC
|
lysophosphatidylcholine
|
|
LPCAT1
|
lysophosphatidylcholine
acyltransferase 1
|
|
MALDI-TOF/MS
|
matrix-assisted laser
desorption/ionization time-of-flight mass spectrometry
|
|
MAPK
|
mitogen-activated protein kinase
|
|
MDSCs
|
myeloid-derived suppressor cells
|
|
MIF
|
macrophage migration inhibitory
factor
|
|
mTOR
|
mammalian target of rapamycin
|
|
NF-κB
|
nuclear factor κ-light-chain-enhancer
of activated B cells
|
|
NK
|
natural killer
|
|
NOTCH1
|
neurogenic locus notch homolog
protein 1
|
|
PAG
|
phenylacetylglutamine
|
|
PC
|
phosphatidylcholine
|
|
PCa
|
prostate cancer
|
|
PD-1
|
programmed cell death protein 1
|
|
PD-L1
|
programmed death-ligand 1
|
|
PI3K
|
phosphatidylinositol 3-kinase
|
|
PTEN
|
phosphatase and tensin homolog
|
|
SCFAs
|
short-chain fatty acids
|
|
STAT3
|
signal transducer and activator of
transcription 3
|
|
TGR5
|
Takeda G protein-coupled receptor
5
|
|
TMAO
|
trimethylamine N-oxide
|
|
Tregs
|
regulatory T cells
|
References
|
1
|
Ye GC, Peng H, Xiang JC, Miao LT, Liu CZ,
Wang SG and Xia QD: Comprehensive analysis of the interaction
microbiome and prostate cancer: An initial exploration from
multi-cohort metagenome and GWAS studies. J Transl Med. 23:1302025.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lyu A, Fan Z, Clark M, Lea A, Luong D,
Setayesh A, Starzinski A, Wolters R, Arias-Badia M, Allaire K, et
al: Evolution of myeloid-mediated immunotherapy resistance in
prostate cancer. Nature. 637:1207–1217. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shore ND, Cookson MS and Efstathiou E: The
landscape of androgen deprivation therapies for the treatment of
advanced prostate cancer. Future Oncol. 20:3351–3354. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jazayeri SB, Cooley LF, Srivastava A and
Shore N: Hormonal intensification should start at the Low-risk
stage in metastatic prostate cancer. Eur Urol Open Sci. 45:38–40.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mohanty SK, Lobo A, Williamson SR, Shah
RB, Trpkov K, Varma M, Sirohi D, Aron M, Kandukari SR, Balzer BL,
et al: Reporting trends, practices, and resource utilization in
neuroendocrine tumors of the prostate gland: A survey among
Thirty-nine genitourinary pathologists. Int J Surg Pathol.
31:993–1005. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Baralo B, Schneider M and Baralo I:
Survival analysis of small cell carcinomas of the genitourinary
system. Proc (Bayl Univ Med Cent). 36:8–14. 2023.PubMed/NCBI
|
|
7
|
Abbott T, Ng K, Nobes J and Muehlschlegel
P: Small-cell carcinoma of the prostate challenges of diagnosis and
treatment: A next of kin and physician perspective piece. Onco
Ther. 11:291–301. 2023. View Article : Google Scholar
|
|
8
|
Cruz-Lebrón A, Faiez TS, Hess MM and
Sfanos KS: Diet and the microbiome as mediators of prostate cancer
risk, progression, and therapy response. Urol Oncol. 43:209–220.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Weersma RK, Zhernakova A and Fu J:
Interaction between drugs and the gut microbiome. Gut.
69:1510–1519. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wong CC and Yu J: Gut microbiota in
colorectal cancer development and therapy. Nat Rev Clin Oncol.
20:429–452. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Attwaters M: Gut bugs disrupt cancer
drugs. Nat Rev Microbiol. 20:7042022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cheng WY, Wu CY and Yu J: The role of gut
microbiota in cancer treatment: Friend or foe? Gut. 69:1867–1876.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Panebianco C, Andriulli A and Pazienza V:
Pharmacomicrobiomics: Exploiting the drug-microbiota interactions
in anticancer therapies. Microbiome. 6:922018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Garajová I, Balsano R, Wang H, Leonardi F,
Giovannetti E, Deng D and Peters GJ: The role of the microbiome in
drug resistance in gastrointestinal cancers. Expert Rev Anticancer
Ther. 21:165–176. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pernigoni N, Zagato E, Calcinotto A,
Troiani M, Mestre RP, Calì B, Attanasio G, Troisi J, Minini M,
Mosole S, et al: Commensal bacteria promote endocrine resistance in
prostate cancer through androgen biosynthesis. Science.
374:216–224. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Daisley BA, Chanyi RM, Abdur-Rashid K, Al
KF, Gibbons S, Chmiel JA, Wilcox H, Reid G, Anderson A, Dewar M, et
al: Author Correction: Abiraterone acetate preferentially enriches
for the gut commensal Akkermansia muciniphila in
castrate-resistant prostate cancer patients. Nat Commun.
11:63942020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tomita Y, Goto Y, Sakata S, Imamura K,
Minemura A, Oka K, Hayashi A, Jodai T, Akaike K, Anai M, et al:
Clostridium butyricum therapy restores the decreased
efficacy of immune checkpoint blockade in lung cancer patients
receiving proton pump inhibitors. Oncoimmunology. 11:20810102022.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jia D, Wang Q, Qi Y, Jiang Y, He J, Lin Y,
Sun Y, Xu J, Chen W, Fan L, et al: Microbial metabolite enhances
immunotherapy efficacy by modulating T cell stemness in pan-cancer.
Cell. 187:1651–1665.e21. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen J, Liu X, Zou Y, Gong J, Ge Z, Lin X,
Zhang W, Huang H, Zhao J, Saw PE, et al: A high-fat diet promotes
cancer progression by inducing gut microbiota-mediated leucine
production and PMN-MDSC differentiation. Proc Natl Acad Sci USA.
121:e23067761212024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li X, Yi Y, Wu T, Chen N, Gu X, Xiang L,
Jiang Z, Li J and Jin H: Integrated microbiome and metabolome
analysis reveals the interaction between intestinal flora and serum
metabolites as potential biomarkers in hepatocellular carcinoma
patients. Front Cell Infect Microbiol. 13:11707482023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ding X, Ting NL, Wong CC, Huang P, Jiang
L, Liu C, Lin Y, Li S, Liu Y, Xie M, et al: Bacteroides
fragilis promotes chemoresistance in colorectal cancer, and its
elimination by phage VA7 restores chemosensitivity. Cell Host
Microbe. 33:941–56.e10. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Terrisse S, Zitvogel L and Kroemer G:
Effects of the intestinal microbiota on prostate cancer treatment
by androgen deprivation therapy. Microb Cell. 9:202–206. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhu X, Li K, Liu G, Wu R, Zhang Y, Wang S,
Xu M, Lu L and Li P: Microbial metabolite butyrate promotes
anti-PD-1 antitumor efficacy by modulating T cell receptor
signaling of cytotoxic CD8 T cell. Gut Microbes. 15:22491432023.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Huang H, Liu Y, Wen Z, Chen C, Wang C, Li
H and Yang X: Gut microbiota in patients with prostate cancer: A
systematic review and meta-analysis. BMC Cancer. 24:2612024.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Distante A, Garino D, Cerrato C,
Perez-Ardavin J, Flores FQ, Lopetuso L and Mir MC: The role of the
human microbiome in prostate cancer: A systematic review from
diagnosis to treatment. Prostate Cancer Prostatic Dis. Nov
18–2025.(Epub ahead of print). doi: 10.1038/s41391-025-01028.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rehman LU, Nisar MH, Fatima W, Sarfraz A,
Azeem N, Sarfraz Z, Robles-Velasco K and Cherrez-Ojeda I:
Immunotherapy for prostate cancer: A current systematic review and
patient centric perspectives. J Clin Med. 12:14462023. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li M, Yao J, Zhang H, Ge Y and An G:
Geographic heterogeneity in the outcomes of patients receiving
immune checkpoint inhibitors for advanced solid tumors: A
meta-analysis. Transl Cancer Res. 10:310–326. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li C, Cheng D and Li P: Androgen receptor
dynamics in prostate cancer: From disease progression to treatment
resistance. Front Oncol. 15:15428112025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Khorasanchi A, Hong F, Yang Y, Singer EA,
Wang P, Li M, Zheng L, Monk P, Mortazavi A and Meng L: Overcoming
drug resistance in castrate-resistant prostate cancer: Current
mechanisms and emerging therapeutic approaches. Cancer Drug Resist.
8:92025.PubMed/NCBI
|
|
30
|
Huang Q, Mitchell C, Theodoulou E, Lee ACK
and Brown J: Implementation of fracture risk assessment in men with
prostate cancer requiring long-term androgen deprivation therapy: A
systematic scoping review using the i-PARIHS implementation
framework. J Cancer Surviv. 20:399–414. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Matsukawa A, Rajwa P, Kawada T, Bekku K,
Laukhtina E, Klemm J, Pradere B, Mori K, Karakiewicz PI, Kimura T,
et al: Impact of disease volume on survival efficacy of triplet
therapy for metastatic hormone-sensitive prostate cancer: A
systematic review, meta-analysis, and network meta-analysis. Int J
Clin Oncol. 29:716–725. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Miszczyk M, Soeterik T, Marra G, Matsukawa
A and Shariat SF: Metastasis-directed therapy in oligometastatic
prostate cancer. Curr Opin Urol. 34:178–182. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shorning BY, Dass MS, Smalley MJ and
Pearson HB: The PI3K-AKT-mTOR pathway and prostate cancer: At the
crossroads of AR, MAPK, and WNT signaling. Int J Mol Sci.
21:45072020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhao D, Mo Z, Zhang T, Cai X, Yang Z, Chen
D, Zhao J, Li Y, Zhou F, Li Z, et al: Lactate derived from
cancer-associated fibroblasts promotes alternative splicing and
castration resistance in prostate cancer. Sci Adv. 12:eady53242026.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Carpenter V, Saleh T, Min Lee S, Murray G,
Reed J, Souers A, Faber AC, Harada H and Gewirtz DA:
Androgen-deprivation induced senescence in prostate cancer cells is
permissive for the development of castration-resistance but
susceptible to senolytic therapy. Biochem Pharmacol.
193:1147652021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhuang D, Kang J, Luo H, Tian Y, Liu X and
Shao C: ARv7 promotes the escape of prostate cancer cells from
androgen deprivation therapy-induced senescence by mediating the
SKP2/p27 axis. BMC Biol. 23:662025. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li C, He C, Pan J, Feng Y, Tian D, Meng J,
Qi Z, Li C and Yang K: MIF facilitates resistance to androgen
deprivation therapy by regulating AMPD2 expression in prostate
cancer cells. Prostate. 86:53–64. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hirani R, Nandakumar S, Zaman N,
Prabhakaraalva P, King SA, Kalidindi TM, Ghale R, Rajanala SH and
Fidele DC: BCL2 drives castration resistance in
castration-sensitive prostate cancer by orchestrating reciprocal
crosstalk between oncogenic pathways. Cell Rep. 44:1157792025.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Maylin ZR, Smith C, Classen A, Asim M,
Pandha H and Wang Y: Therapeutic exploitation of neuroendocrine
transdifferentiation drivers in prostate cancer. Cells.
13:19992024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhang B, Lau LY, Chen Y and Xie R:
Bibliometric analysis of immune-related acute kidney injury induced
by cancer immunotherapy (2000–2025). Naunyn Schmiedebergs Arch
Pharmacol. 399:2647–2662. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Novysedlak R, Guney M, Al Khouri M,
Bartolini R, Koumbas Foley L, Benesova I, Ozaniak A, Novak V,
Vesely S, Pacas P, et al: The immune microenvironment in prostate
cancer: A comprehensive review. Oncology. 103:521–545. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
San-Jose Manso L, Alfranca A, Moreno-Pérez
I, Ruiz-Vico M, Velasco C, Toquero P, Pacheco M, Zapatero A, Aldave
D, Celada G, et al: Immunome profiling in prostate cancer: A guide
for clinicians. Front Immunol. 15:13981092024. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Liu Q, Guan Y and Li S: Programmed death
receptor (PD-)1/PD-ligand (L)1 in urological cancers the
‘all-around warrior’ in immunotherapy. Mol Cancer. 23:1832024.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Jiang C, Hu C, Hao J, Wang W, Sun J and
Mao X: Roles of CDK12 mutations in PCa development and treatment.
Biochim Biophys Acta Rev Cancer. 1880:1892472025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Grypari IM, Tzelepi V and Gyftopoulos K:
DNA damage repair pathways in prostate cancer: A narrative review
of molecular mechanisms, emerging biomarkers and therapeutic
targets in precision oncology. Int J Mol Sci. 24:114182023.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Li B, Lin R, Hua Y, Ma B and Chen Y:
Single-cell RNA sequencing reveals TMEM71 as an immunomodulatory
biomarker predicting immune checkpoint blockade response in breast
cancer. Discov Oncol. 16:12562025. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wu Z, Zhang J, Li L, Wang Z and Yang C:
Biomarkers in metastatic castration-resistant prostate cancer for
efficiency of immune checkpoint inhibitors. Ann Med.
57:24267552025. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Hong X and Zhang Y, Chi Z, Xu Q, Lin W,
Huang Y, Lin T and Zhang Y: Efficacy and safety of programmed
Death-1 (PD-1)/Programmed Death-Ligand 1 (PD-L1) Checkpoint
inhibitors in patients with metastatic Castration-resistant
prostate cancer: A systematic review and Meta-analysis. Clin Oncol
(R Coll Radiol). 36:e20–e30. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Rebuzzi SE, Rescigno P, Catalano F,
Mollica V, Vogl UM, Marandino L, Massari F, Pereira Mestre R,
Zanardi E, Signori A, et al: Immune checkpoint inhibitors in
advanced prostate cancer: Current data and future perspectives.
Cancers (Basel). 14:12452022. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Chen F, Wu S, Kuang N, Zeng Y, Li M and Xu
C: ABCB1-mediated docetaxel resistance reversed by erastin in
prostate cancer. FEBS J. 291:3249–3266. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Brandt MP, Vakhrusheva O, Hackl H, Daher
T, Tagscherer K, Roth W, Tsaur I, Handle F, Eigentler A, Culig Z,
et al: Inhibition of the sterol regulatory element binding protein
SREBF-1 overcomes docetaxel resistance in advanced prostate cancer.
Am J Pathol. 194:2150–2162. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Wang J and Nie D: Interaction of pregnane
X receptor with hypoxia-inducible factor-1 regulates
chemoresistance of prostate cancer cells. Cancer Drug Resist.
6:378–389. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Souchek JJ, Laliwala A, Houser L, Muraskin
L, Vu Q and Mohs AM: Fatty acid synthase inhibitors enhance
Microtubule-stabilizing and Microtubule-destabilizing drugs in
Taxane-resistant prostate cancer cells. ACS Pharmacol Transl Sci.
6:1859–1869. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Zhang B, Lau LY, Wu Z and Chen Y:
Chemotherapy induced neurotoxicity in cancer survivors assessed
through a dual database bibliometric analysis from 2005 to 2025.
Discov Oncol. 16:19612025. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Slootbeek PHJ, Kloots ISH, van Oort IM,
Kroeze LI, Schalken JA, Bloemendal HJ and Mehra N: Cross-Resistance
between Platinum-Based chemotherapy and PARP inhibitors in
castration-resistant prostate cancer. Cancers (Basel). 15:28142023.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zhang Y, Duan H, Zhao H, Qi L, Liu Y,
Zhang Z, Liu C, Chen L, Jin M, Guan Y, et al: Development and
evaluation of a PSMA-Targeted nanosystem Co-Packaging docetaxel and
androgen receptor siRNA for Castration-resistant prostate cancer
treatment. Pharmaceutics. 14:9642022. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Jiang X, Guo S, Wang S, Zhang Y, Chen H,
Wang Y, Liu R, Niu Y and Xu Y: EIF4A3-Induced circARHGAP29 promotes
aerobic glycolysis in Docetaxel-resistant prostate cancer through
IGF2BP2/c-Myc/LDHA signaling. Cancer Res. 82:831–845. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Guo Z, Lu X, Yang F, Qin L, Yang N, Wu J
and Wang H: Docetaxel chemotherapy plus androgen-deprivation
therapy in high-volume disease metastatic hormone-sensitive
prostate cancer in Chinese patients: An efficacy and safety
analysis. Eur J Med Res. 27:1482022. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
White RE III, Bannister M, Day A, Bergom
HE, Tan VM, Hwang J, Dang Nguyen H and Drake JM: Saracatinib
synergizes with enzalutamide to downregulate AR activity in CRPC.
Front Oncol. 13:12104872023. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Dyshlovoy SA, Shubina LK, Makarieva TN,
Hauschild J, Strewinsky N, Guzii AG, Menshov AS, Popov RS, Grebnev
BB, Busenbender T, et al: New diterpenes from the marine sponge
Spongionella sp. overcome drug resistance in prostate cancer by
inhibition of P-glycoprotein. Sci Rep. 12:135702022. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Wang H, Cui C, Li W, Wu H, Sha J, Pan J
and Xue W: AGD1/USP10/METTL13 complexes enhance cancer stem cells
proliferation and diminish the therapeutic effect of docetaxel via
CD44 m6A modification in castration resistant prostate cancer. J
Exp Clin Cancer Res. 44:122025. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Walmsley CS, Jonsson P, Cheng ML, McBride
S, Kaeser C, Vargas HA, Laudone V, Taylor BS, Kappagantula R, Baez
P, et al: Convergent evolution of BRCA2 reversion mutations under
therapeutic pressure by PARP inhibition and platinum chemotherapy.
NPJ Precis Oncol. 8:342024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Packeiser EM, Engels L, Nolte I,
Goericke-Pesch S and Murua Escobar H: MDR1 inhibition reverses
Doxorubicin-resistance in six Doxorubicin-resistant canine prostate
and bladder cancer cell lines. Int J Mol Sci. 24:81362023.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Li S, Xiong S, Li Z, Yang L, Yang H, Xiong
J, Pan W, Guo J, Xu S and Fu B: USP3 promotes DNA damage response
and chemotherapy resistance through stabilizing and
deubiquitinating SMARCA5 in prostate cancer. Cell Death Dis.
15:7902024. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Chen X, Ma J, Wang X, Zi T, Qian D, Li C
and Xu C: CCNB1 and AURKA are critical genes for prostate cancer
progression and castration-resistant prostate cancer resistant to
vinblastine. Front Endocrinol (Lausanne). 13:11061752022.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Che P, Jiang S, Zhang W, Zhu H, Hu D and
Wang D: A comprehensive gene expression profile analysis of
prostate cancer cells resistant to paclitaxel and the potent target
to reverse resistance. Hum Exp Toxicol. 41:96032712211298542022.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Tohidast M, Memari N, Amini M, Hosseini
SS, Jebelli A, Doustvandi MA, Baradaran B and Mokhtarzadeh A:
MiR-145 inhibits cell migration and increases paclitaxel
chemosensitivity in prostate cancer cells. Iran J Basic Med Sci.
26:1350–1359. 2023.PubMed/NCBI
|
|
68
|
Kim CJ, Gonye AL, Truskowski K, Lee CF,
Cho YK, Austin RH, Pienta KJ and Amend SR: Nuclear morphology
predicts cell survival to cisplatin chemotherapy. Neoplasia.
42:1009062023. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Liu Y, Zhou Q, Ye F, Yang C and Jiang H:
Gut microbiota-derived short-chain fatty acids promote prostate
cancer progression via inducing cancer cell autophagy and M2
macrophage polarization. Neoplasia. 43:1009282023. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Chen Y, Bai M, Liu M, Zhang Z, Jiang C, Li
K, Chen Y, Xu Y and Wu L: Metabolic reprogramming in lung cancer:
Hallmarks, mechanisms, and targeted strategies to overcome immune
resistance. Cancer Med. 14:e713120257
|
|
71
|
Bui NN, Li CY, Wang LY, Chen YA, Kao WH,
Chou LF, Hsieh JT, Lin H and Lai CH: Clostridium scindens
metabolites trigger prostate cancer progression through androgen
receptor signaling. J Microbiol Immunol Infect. 56:246–256. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Hsia YJ, Lin ZM, Zhang T and Chou TC:
Butyrate increases methylglyoxal production through regulation of
the JAK2/Stat3/Nrf2/Glo1 pathway in castration-resistant prostate
cancer cells. Oncol Rep. 51:712024. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Zhong W, Wu K, Long Z, Zhou X, Zhong C,
Wang S, Lai H, Guo Y, Lv D, Lu J and Mao X: Gut dysbiosis promotes
prostate cancer progression and docetaxel resistance via activating
NF-κB-IL6-STAT3 axis. Microbiome. 10:942022. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Gao D, Shen Y, Xu L, Sun Y, Hu H, Xu B,
Wang Z and Xu H: Acetate utilization promotes hormone therapy
resistance in prostate cancer through neuroendocrine
differentiation. Drug Resist Updat. 77:1011582024. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Matsushita M, Fujita K, Motooka D, Hatano
K, Fukae S, Kawamura N, Tomiyama E, Hayashi Y, Banno E, Takao T, et
al: The gut microbiota associated with high-Gleason prostate
cancer. Cancer Sci. 112:3125–3135. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Zhou Y, Lv J, Jin S, Fu C, Liu B, Shen Y,
Li M, Zhang Y and Feng N: Gut microbiota derived metabolite
trimethylamine N-oxide influences prostate cancer progression via
the p38/HMOX1 pathway. Front Pharmacol. 15:15260512025. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Luo XH, Liu JZ, Wang B, Men QL, Ju YQ, Yin
FY, Zheng C and Li W: KLF14 potentiates oxidative adaptation via
modulating HO-1 signaling in castrate-resistant prostate cancer.
Endocr Relat Cancer. 26:181–195. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Lin YH, Chen YT and Tsai HY: Revisiting
the impact of antibiotics on prostate cancer risk: Beyond the gut
microbiota. Int J Uro. 31:3332024. View Article : Google Scholar
|
|
79
|
Lachance G, Robitaille K, Laaraj J,
Gevariya N, Varin TV, Feldiorean A, Gaignier F, Julien IB, Xu HW,
Hallal T, et al: The gut microbiome-prostate cancer crosstalk is
modulated by dietary polyunsaturated long-chain fatty acids. Nat
Commun. 15:34312024. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Reichard CA, Naelitz BD, Wang Z, Jia X, Li
J, Stampfer MJ, Klein EA, Hazen SL and Sharifi N: Gut
Microbiome-dependent metabolic pathways and risk of lethal prostate
cancer: Prospective analysis of a PLCO cancer screening trial
cohort. Cancer Epidemiol Biomarkers Prev. 31:192–199. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wu Y, Li RW, Huang H, Fletcher A, Yu L,
Pham Q, Yu L, He Q and Wang TTY: Inhibition of tumor growth by
dietary Indole-3-Carbinol in a prostate cancer xenograft model may
be associated with disrupted gut microbial interactions. Nutrients.
11:4672019. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Tucci P, Brown I, Bewick GS, Pertwee RG
and Marini P: The plant derived 3–3′-diindolylmethane (DIM) behaves
as CB(2) receptor agonist in prostate cancer cellular models. Int J
Mol Sci. 24:36202023. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Reyes-Hernández OD, Figueroa-González G,
Quintas-Granados LI, Gutiérrez-Ruíz SC, Hernández-Parra H,
Romero-Montero A, Del Prado-Audelo ML, Bernal-Chavez SA, Cortés H,
Peña-Corona SI, et al: 3,3′-Diindolylmethane and indole-3-carbinol:
Potential therapeutic molecules for cancer chemoprevention and
treatment via regulating cellular signaling pathways. Cancer Cell
Int. 23:1802023. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Shilpa G, Lakshmi S, Jamsheena V,
Lankalapalli RS, Prakash V, Anbumani S and Priya S: Studies on the
mode of action of synthetic diindolylmethane derivatives against
triple negative breast cancer cells. Basic Clin Pharmacol Toxicol.
131:224–240. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Peter RM, Sarwar MS, Wang L, Chou P, Wang
C, Wang Y, Su X and Kong AN: Dietary phytochemical
indole-3-carbinol regulates metabolic reprogramming in mouse
prostate tissue. Pharm Res. 42:237–247. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Chen H, Gao B, Li J, Liu L, Zhang Y, Shuai
M and Ji Y: Indole-3-carbinol prevented tumor progression and
potentiated PD1ab therapy by upregulating PTEN in colorectal
cancer. Discov Oncol. 16:2242025. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Chen Q, Jiang C and Li H:
Indole-3-Carbinol promotes apoptosis and inhibits the metastasis of
esophageal squamous cell carcinoma by downregulating the
Wnt/β-Catenin signaling pathway. Nutr Cancer. 76:543–551. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Tang X, Wang W, Hong G, Duan C, Zhu S,
Tian Y, Han C, Qian W, Lin R and Hou X: Gut microbiota-mediated
lysophosphatidylcholine generation promotes colitis in intestinal
epithelium-specific Fut2 deficiency. J Biomed Sci. 28:202021.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Buszewska-Forajta M, Pomastowski P,
Monedeiro F, Walczak-Skierska J, Markuszewski M, Matuszewski M,
Markuszewski MJ and Buszewski B: Lipidomics as a diagnostic tool
for prostate cancer. Cancers (Basel). 13:20002021. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Buszewska-Forajta M, Pomastowski P,
Monedeiro F, Król-Górniak A, Adamczyk P, Markuszewski MJ and
Buszewski B: New approach in determination of urinary diagnostic
markers for prostate cancer by MALDI-TOF/MS. Talanta.
236:1228432022. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Li X, Nakayama K, Goto T, Kimura H,
Akamatsu S, Hayashi Y, Fujita K, Kobayashi T, Shimizu K, Nonomura
N, et al: High level of
phosphatidylcholines/lysophosphatidylcholine ratio in urine is
associated with prostate cancer. Cancer Sci. 112:4292–302. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Wang B and Tontonoz P: Phospholipid
remodeling in physiology and disease. Annu Rev Physiol. 81:165–188.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Liu Y, Yang C, Zhang Z and Jiang H: Gut
microbiota dysbiosis accelerates prostate cancer progression
through increased LPCAT1 expression and enhanced DNA repair
pathways. Front Oncol. 11:6797122021. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Lv J, Jin S, Zhou Y, Fu C, Shen Y, Liu B,
Li J, Li M, Zhang Y and Feng N: Gut microbiota-derived metabolite
phenylacetylglutamine inhibits the progression of prostate cancer
by suppressing the Wnt/β-catenin signaling pathway. Front
Pharmacol. 16:15280582025. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Zhou Q, Zhao JT, He XS, Chen WK, Hu FM,
Zhang FR, Lu L and Lu QY: Phenylacetylglutamine, anaemia and
outcomes in ischaemic heart failure: A cohort study. BMJ Open.
15:e0979222025. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Zhao P, Yu Z, Song W, Zhao S, Jin C, Liu
X, Yu B and Tian J: Associations of plasma metabolite
phenylacetylglutamine on coronary plaque characterization in
patients with ST-segment elevation myocardial infarction.
Cardiovasc Toxicol. 25:1903–1913. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Wei M, Huang Q, Yu F, Luo YF, Feng X, Liao
D, Li J, Zhang B, Liu ZY and Xia J: Elevated phenylacetylglutamine
caused by gut dysbiosis associated with type 2 diabetes increases
neutrophil extracellular traps formation and exacerbates brain
infarction. Clin Sci (Lond). 139:717–736. 2025.PubMed/NCBI
|
|
98
|
Fei J, Chang X, Yang W, He Y, Liu Y, Shi
M, Liu Y, Wang X, Peng H, Bu X, et al: Plasma phenylacetylglutamine
and cognitive impairment after ischemic stroke. J Am Heart Assoc.
14:e0429552025. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Gao Z, Xiong Z, Tao Y, Wang Q, Guo K, Xu K
and Huang H: LGR5 interacts with HSP90AB1 to mediate enzalutamide
resistance by activating the WNT/β-catenin/AR axis in prostate
cancer. Chin Med J. 138:3184–3194. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Chen H, Li Z, Yue Y, Zhu X, Wang J, Chen
Y, Wang Y, Luo Z and Liu H: CAF-mediated regulation of prostate
cancer stem cell stemness via the Wnt/β-catenin and SDF-1/CXCR4
pathways in castration-resistant prostate cancer. Front Cell Dev
Biol. 13:16172002025. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Lin BS, Yin MY, Xie SA, Li P and Li X:
Pleiotropic immunoregulation by bile acids in pathophysiology.
Front Immunol. 17:17190922026. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Lin X, Xia L, Zhou Y, Xie J, Tuo Q, Lin L
and Liao D: Crosstalk between bile acids and intestinal epithelium:
Multidimensional roles of farnesoid X receptor and takeda G protein
receptor 5. Int J Mol Sci. 26:42402025. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Tong Y and Lou X: Interplay between bile
acids, gut microbiota, and the tumor immune microenvironment:
Mechanistic insights and therapeutic strategies. Front Immunol.
16:16383522025. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Wang Z, Liu Z, Cui L, Sun J, Bu C, Tang M,
Li M, Gao S, Chen W and Tao X: Disturbance of bile acids profile
aggravates the diarrhea induced by capecitabine through inhibiting
the Wnt/β-catenin pathway. J Adv Res. 72:591–604. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Sun F, Wang K, Dong X, Secaira-Morocho H,
Hui A, Cai C, Sze JJ, Low B, Udgata S, Pasch CA, et al: The
microbial bile acid metabolite 3-Oxo-LCA inhibits colorectal cancer
progression. Cancer Res. 85:4937–4957. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Li C, Xing X, Li M, Liu Y, Huang S, Zhu T,
Gu W and Yan B: Bile acids produced by gut microbiota activate TGR5
to promote colorectal liver metastasis progression by inducing
MDSCs infiltration in liver. Int Immunopharmacol. 158:1148292025.
View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Detwiler Z and Chaudhari SN: BAAT away
liver cancer: Conjugated bile acids impair T cell function in
hepatocellular carcinoma immunotherapy. Immunometabolism (Cobham).
7:e000622025. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Wei H, Suo C, Gu X, Shen S, Lin K, Zhu C,
Yan K, Bian Z, Chen L, Zhang T, et al: AKR1D1 suppresses liver
cancer progression by promoting bile acid metabolism-mediated NK
cell cytotoxicity. Cell Metab. 37:1103–1118.e7. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Pernigoni N, Guo C, Gallagher L, Yuan W,
Colucci M, Troiani M, Liu L, Maraccani L, Guccini I, Migliorini D,
et al: The potential role of the microbiota in prostate cancer
pathogenesis and treatment. Nat Rev Urol. 20:706–718. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Lin G, Zhang F, Weng X, Hong Z, Ye D and
Wang G: Role of gut microbiota in the pathogenesis of
castration-resistant prostate cancer: A comprehensive study using
sequencing and animal models. Oncogene. 43:2373–2388. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Kure A, Tsukimi T, Ishii C, Aw W, Obana N,
Nakato G, Hirayama A, Kawano H, China T, Shimizu F, et al: Gut
environment changes due to androgen deprivation therapy in patients
with prostate cancer. Prostate Cancer Prostatic Dis. 26:323–330.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Conti P, Gallenga CE, Annicchiarico C,
Coppola A, Pellegrino R, Conti MJ and Mastrangelo F: Mast cells
accumulate in the stroma of breast adenocarcinoma and secrete
Pro-Inflammatory cytokines and Tumor-damaging mediators: Could
IL-37 and IL-38 play an Anti-tumor role? Int J Mol Sci. 27:8242026.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Matsushita M, Fujita K, Hatano K, Hayashi
T, Kayama H, Motooka D, Hase H, Yamamoto A, Uemura T, Yamamichi G,
et al: High-fat diet promotes prostate cancer growth through
histamine signaling. Int J Cancer. 151:623–636. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Wang SF, Liu YC, Nguyen PA, Lin GL, Huang
CW, Rahmanti AR and Yang HC: Association between Long-term Use of
H2 receptor antagonists and prostate cancer risk: A Case-control
study in taiwan. J Cancer. 17:419–426. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Lauretta P, Martinez Vivot R, Velazco A
and Medina VA: Histamine H3 receptor: An emerging target for cancer
therapy? Inflamm Res. 74:972025. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Li H, Xiao Y, Li Q, Yao J, Yuan X, Zhang
Y, Yin X, Saito Y, Fan H, Li P, et al: The allergy mediator
histamine confers resistance to immunotherapy in cancer patients
via activation of the macrophage histamine receptor H1. Cancer
Cell. 40:36–52.e9. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Duan H, Wang L, Huangfu M and Li H: The
impact of microbiota-derived short-chain fatty acids on macrophage
activities in disease: Mechanisms and therapeutic potentials.
Biomed Pharmacother. 165:1152762023. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Matsushita M, Fujita K, Hatano K, De
Velasco MA, Uemura H and Nonomura N: Connecting the dots between
the Gut-IGF-1-Prostate axis: A role of IGF-1 in prostate
carcinogenesis. Front Endocrinol (Lausanne). 13:8523822022.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Lin PH, Howard L and Freedland SJ: Weight
loss via a low-carbohydrate diet improved the intestinal
permeability marker, zonulin, in prostate cancer patients. Ann Med.
54:1221–1225. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Nam EH, Lee M, Kim H, Kim D, Lee Y, Jung
YH, Yang J and Shin M: The impact of Streptococcus thermophilus
IDCC 2201 on gut microbiota and its potential as a prophylactic
agent for colorectal cancer. Sci Rep. 15:371402025. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Li JKM, Wang LL, Lau BSY, Tse RTH, Cheng
CKL, Leung SCH, Wong CYP, Tsui SKW, Teoh JYC, Chiu PKF and Ng CF:
Oral antibiotics perturbation on gut microbiota after prostate
biopsy. Front Cell Infect Microbiol. 12:9599032022. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Sevcikova A, Martiniakova M, Omelka R,
Stevurkova V and Ciernikova S: Gut dysbiosis impacts the immune
system and promotes prostate cancer. Immunol Lett. 268:1068832024.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Xu W, Li Y, Liu L, Xie J, Hu Z, Kuang S,
Fu X, Li B, Sun T, Zhu C, et al: Icaritin-curcumol activates CD8+ T
cells through regulation of gut microbiota and the DNMT1/IGFBP2
axis to suppress the development of prostate cancer. J Exp Clin
Cancer Res. 43:1492024. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Thomas LV, Suzuki K and Zhao J:
Probiotics: A proactive approach to health. A symposium report. Br
J Nutr. 114 (Suppl 1):S1–S15. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Jia D and Wang L: Opportunities and
challenges in applying microbiota to clinical cancer immunotherapy.
Trends Microbiol. 34:367–377. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Ebrahimi R, Shahrokhi Nejad S, Fekri M and
Nejadghaderi SA: Advancing prostate cancer treatment: The role of
fecal microbiota transplantation as an adjuvant therapy. Curr Res.
9:1004202025.PubMed/NCBI
|
















