Introduction
Granular cell tumor (GCT), which was first reported
by Abriksoosoff in 1926 (1), is an
uncommon and usually benign tumor (2). It occurs in almost all organs, but
mostly in the skin or soft tissues. GCT may occur in one tissue,
and in certain cases in multi-organs simultaneously or
asynchronously (2). Currently, it
is widely accepted that GCT derives from neurogenic Schwann cells
(3). GCT occurring in the
gastrointestinal tract accounts for only 8% of all GCTs (2). Esophageal GCT, which was also first
reported by Abriksoosoff in 1931 (4), is the most common type of GCT in the
gastrointestinal tract. It is estimated that esophageal GCT
accounts for approximately one-third of all GCTs in the
gastrointestinal tract and ~1% of all esophageal benign tumors
(5–7). Esophageal GCTs exhibit almost the same
clinicopathological characteristics as GCTs of other organs.
Although the diagnosis of GCT is relatively straightforward,
deciding on an appropriate treatment strategy is often complex.
Previously surgical resectioning, including wide excision, was the
recommended treatment strategy (8).
However, in recent years this technique has been gradually
abandoned (5). Currently, the
majority of GCT patients undergo endoscopic resectioning, after
which, the majority of tumors are removed and relapse is rare
either in situ or in other locations even after several
years. However, relapse seldom affects the patients’ lifespan
(5).
Since the first report of GCT, ~300 esophageal GCTs
have been reported in literature (3). The aim of the present study is to
describe their clinical, endoscopic and histological features.
Materials and methods
All the data were collected in our endoscopic
centers (Digestive Endoscopy Center, Drum Tower Hospital Affiliated
Medical College of Nanjing University, Nanjing, China; and
Digestive Endoscopy Center, Renmin Hospital, Hubei University of
Medicine, Shiyan, China) between January 2005 and June 2013.
Following the identification of esophageal GCT, patient demographic
data, which includes age, gender, indications for endoscopy,
therapeutic methods, the records of colonoscopy and endoscopic
ultrasound (EUS), were carefully collected. Endoscopic and EUS
images were reviewed again by two of our authors. The follow-ups
were conducted by calling the patient and asking for their health
condition, including if there were new tumors, relapse or
metastasis of the primary tumor. All the patient slides were
checked again for histological and immunohistochemical stains. The
protocol of the present study was prepared according to the
Declaration of Helsinki and approved by the ethics committee of the
Medical School of Nanjing University and Hubei University of
Medicine (China).
Results
During the survey period, 19 cases (11 female and
eight male) of esophageal GCTs were identified. The median age at
the time of diagnosis was 42.0 years old (range, 24–71 years old).
For all the cases, the tumor was solitary. The majority of patient
indications for endoscopy were non-specific. Two patients with a
tumor diameter of ≥1.5 cm complained of dysphagia. Following the
removal of the tumor, the symptom of dysphagia was relieved.
Endoscopic therapy was effectively performed in 17 cases, and no
complications occurred during the procedure or in the following
period. Colonoscopy was undertaken in nine patients and no colonic
GCT was identified. The median follow-up period was 45 months
(range, 7–95 months). During the follow-up period, one patient was
lost to follow-up and one confirmed another esophageal GCT after 12
months. An endoscopic forcep biopsy was obtained from five of the
19 patients and three confirmed the diagnosis of GCTs. The general
clinical information is presented in Table I.
 | Table IClinical characteristics of the 19
esophageal GCT cases. |
Table I
Clinical characteristics of the 19
esophageal GCT cases.
| Clinical
characteristics | n | % |
|---|
| Gender |
| Female | 11 | 57.9 |
| Male | 8 | 42.1 |
| Indications for
endoscopy |
| Epigastric
discomfort | 5 | 26.3 |
| Abdominal
distention | 3 | 15.8 |
| Heart-burn | 6 | 31.6 |
| Epigastric pain | 3 | 15.8 |
| Dysphagia | 2 | 10.5 |
| Therapeutic
methods |
| Endoscopic
polypectomy | 12 | 63.2 |
| Endoscopic mucosal
resection | 2 | 10.5 |
| Removed by biopsy
forcep | 3 | 15.8 |
| Surgery | 1 | 5.3 |
| Untreated | 1 | 5.3 |
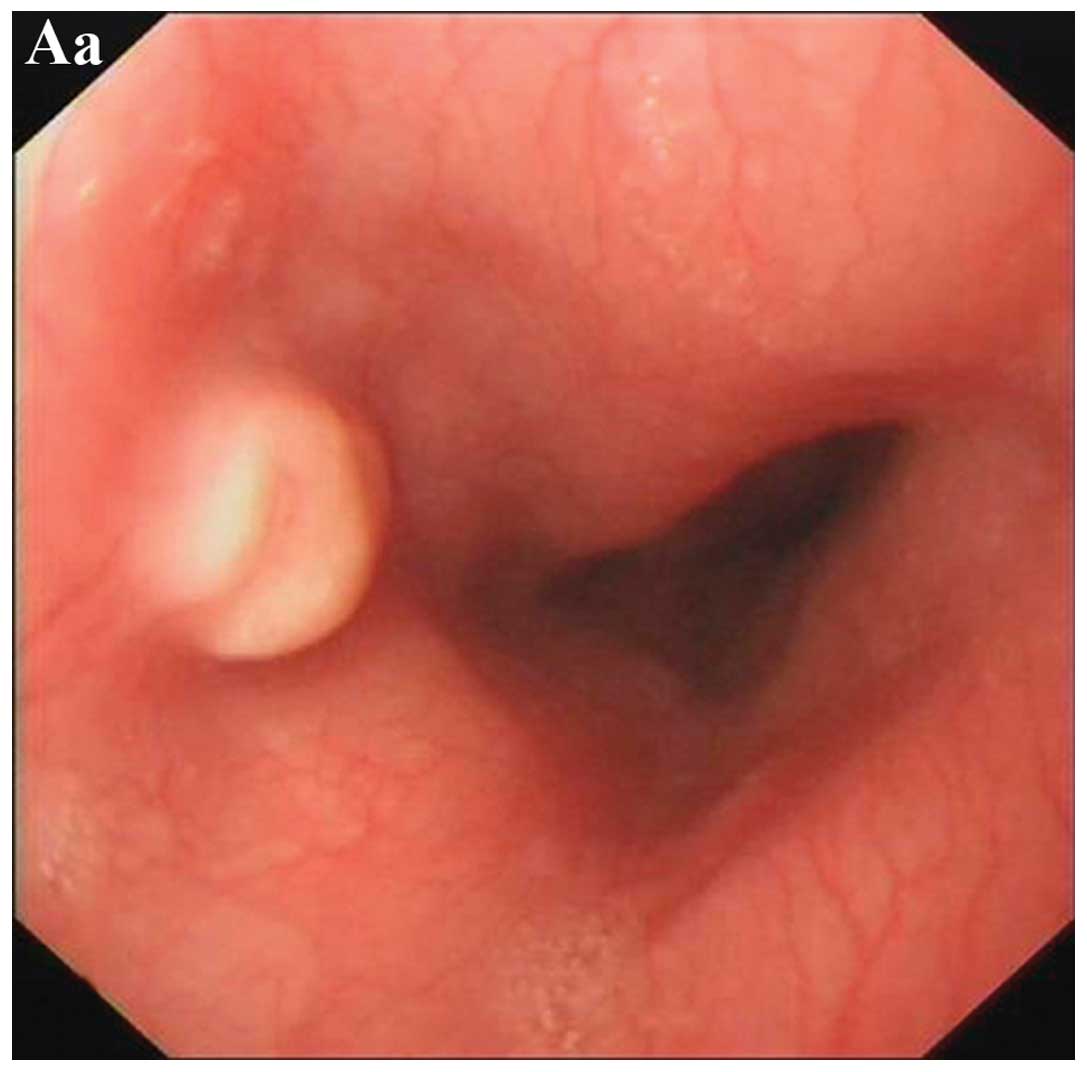
The endoscopic appearance of esophageal GCT was
variable. All 20 tumor surfaces were smooth and white to gray
(n=9), pink (n=7) or yellow (n=4) in color (Fig. 1). The primary endoscopic diagnoses
were GCT (n=7), polyp (n=6), leiomyoma (n=4), lipoma (n=2) and
interstitialoma (n=1). Tumors were located in the upper, middle and
lower segment of the esophagus in four, eight and eight cases
respectively. The tumor size ranged from 0.4 to 2.0 cm, with a
median size of 0.7 cm. One patient had the occurrence of two
tumors, which were located in the middle segment of the esophagus
with similar endoscopic features.
Nine patients underwent EUS by UM-20-29R (12 MHz) or
UM-2R (20 MHz), eight of which demonstrated a smooth margin with
intracavity growth features. Tumors were derived from the mucosa,
muscularis mucosa, submucosa layer and muscularis propria layer in
two, four, two and one cases, respectively (Fig. 1). The case that derived from the
muscularis propria layer indicated an irregular margin and intra-
and extra-cavity growth features. The majority of the tumors
demonstrated hypoechoic (n=8) echostructures. A total of six and
three cases indicated homogenous and heterogeneous echoic features,
respectively. No necrosis or fibrosis was found inside the tumors
with heterogeneous echoic by histological examination.
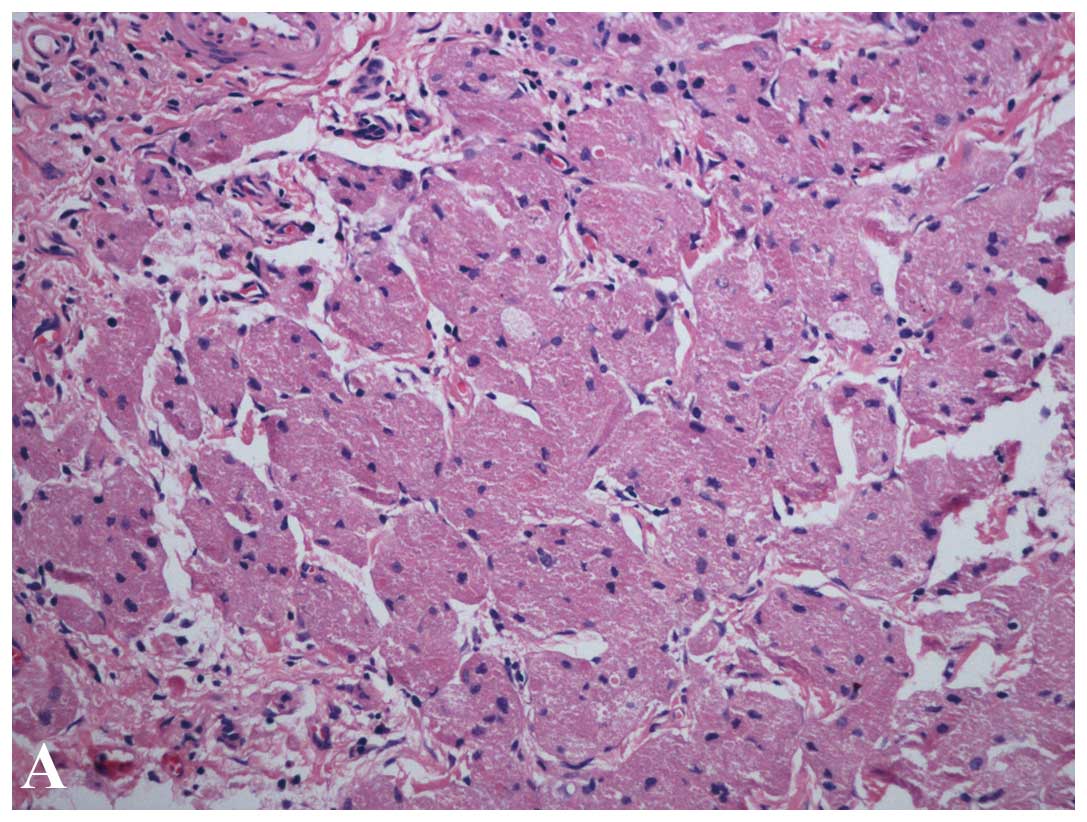
Histologically, the tumors demonstrated a nest
appearance, which was separated by fibers with non-capsulated
margins and obscure boundaries. The tumor cells could be spherical,
polygon or fusifom in shape with infiltration of the surrounding
layers. The cytoplasm contained abundant eosinophilic granules and
a tiny round nucleus located centrally. No necrosis or nuclear
fission was determined in the tumor cells (Fig. 2). Pleomorphism was identified in
three cases and 16 of the 19 cases underwent immunohistochemical
staining. All 16 cases indicated positive staining for S-100,
periodic acid-Schiff-diastase (PAS-D) and nestin, and negative
staining for cluster of differentiation 117 (CD117), CD34, desmin,
cytokeratin (CK) and smooth muscle antibody (SMA) (Fig. 2). A total of 12 of the 16 cases were
CD68-positive. Of the three cases that indicated pleomorphism, the
percentage of Ki-67-positive cells was >2%. All the other cases
were Ki-67-negative. The two tumors that occurred in the same
patient showed the same histological and immunohistochemical stain
features.
Discussion
GCTs, once referred to as Abrikossoff’s tumors or
granular cell myoblastomas (1,2), are
relatively uncommon and esophageal GCT is much rarer (4). With the widespread use of gastroscopy,
more frequent detection of this tumor type has become possible and
endoscopic tumor excision is becoming more frequent (5,8).
Esophageal GCT could occur at any age, but it is
more common in 40- to 60-year-old patients (9,10) and
in females compared with males (5,10–12).
The present study reconfirmed these results. Thirty years ago,
studies of esophageal GCTs were mainly derived from the symptom of
dsyphagia and autopsy (11).
However, the majority of esophageal GCTs have been diagnosed
incidentally in more recent years, due to the widespread usage of
endoscopy (2,12). If the tumor size is ≥1 cm, the
patients may complain of dysphagia (5,11).
Similar to most studies in recent years, the majority of cases in
the present survey were not diagnosed due to dysphagia. In the 2
patients who complained of dysphagia, the tumor size was ≥1.5
cm.
GCTs may occur multiple times in one tissue type and
in multiple organs (2). It has been
previously reported that ~5–30% of esophageal GCTs were multiple
(2,5,12). In
the present study, there were no multiple esophageal GCTs or GCTs
in other organs. Only one new esophageal GCT was identified during
the 12 months follow-up in one patient. Zhong et al
(5) reported that ≥80% (19/23) of
patients could be diagnosed by endoscopic forcep biopsy. At the
Digestive Endoscopy Centers of Drum Tower Hospital Affiliated
Medical College of Nanjing University and Renmin hospital Hubei
University of Medicine, biopsies were only performed when the tumor
size was ≤0.6 cm. If the tumor size was >0.6 cm, EUS and
endoscopic resection were recommended. Ultimately, in the present
study only five patients underwent biopsy and three were diagnosed
with GCT.
Typically, the endoscopic feature of esophageal GCT
is an elevated lesion with a white-to-gray appearance (3,5).
However, certain tumors may show a pink or yellow appearance
(3,5). The surface is usually smooth and, in
certain cases, coincides with ulceration or necrosis (3). The tumors are usually located in the
middle and lower segments of the esophagus. The results of the
present study showed similar endoscopic features to such previous
studies.
The first and largest study reporting the EUS
features of esophageal GCT was published in 1997 (12). It concluded that esophageal GCTs
show a hypoechoic and homogeneous echostructure that usually
derives from mucosa and muscularis mucosa layers, and has smooth
edges (12). Various results were
reported in other studies (3,4,12,13).
Hyperechoic, besides one case in the present study, has also been
reported in certain cases (5). This
type of feature usually causes the misdiagnosis of lipoma,
particularly in patients with tumors that have a yellow surface.
Even though homogeneous echostructure and smooth margins are often
reported (12,13), heterogeneous echostructure and
irregular margins have also been encountered (5). The causes of these differences remain
unknown. Due to all these atypical features, a negative attitude is
held for the advantages of EUS in surveillance of esophageal GCTs
(12). However, considering
extra-cavity growth (one case in the present study) and safety of
resection, simple endoscopic surveillance may not be sufficient.
EUS surveillance should be undertaken during follow-up and prior to
endoscopic resection. Allowing for the fact that the majority of
GCTs grow extremely slowly, if no new symptoms appear then a
two-year interval is adequate.
The first report of endoscopic therapy for
esophageal GCT was in 1979 (14).
Since then, endoscopic therapy, which includes endoscopic
diathermic electrosurgical snare, endoscopic polypectomy,
endoscopic mucosal resection and endoscopic submucosal dissection,
for esophageal GCTs has become increasingly popular (3,5). In
the present study, 17 of the 19 cases underwent endoscopic therapy
safely. Even though it is a benign disease, certain tumors may
undergo a malignant change and some tumors may reoccur following
resection (15,16). Follow-up was indicated for all the
patients, independent of tumor resection (15,16).
In the largest series as yet, specific cases left untreated showed
either a stable tumor size or regression of the tumor (9).
Histological features of esophageal GCTs could mimic
esophageal squamous cancers (17),
particularly spindle-cell squamous cancers. In certain cases these
two diseases could co-exist in the same patient (18,19).
The most differentiating points between them are the
nuclear-cytoplasmic ratios and nuclear fission. For esophageal GCT
the nuclear-cytoplasmic ratios are usually low and nuclear fission
is rare. However, for malignant esophageal GCT, it is extremely
difficult to differentiate it from esophageal squamous cancers.
Besides, GCT can also mimic gastrointestinal stromal tumor (GIST)
(20) or leiomyoma. In the present
study, two of the five patients who underwent biopsy were
misdiagnosed prior to resection; one was misdiagnosed as GIST and
the other as leiomyoma.
If histological features cannot aid the diagnosis,
immunohistochemical staining may be helpful. Since the positive
staining of S-100 was first reported in 1986 (21), other positive markers have also been
reported, including PAS (22),
neuron-specific enolase (19) and
nestin (7). Negative markers, which
include CD117, CD34, desmin, CK, SMA, glial fibrillary acidic
protein, inhibin-α, myoglobin, fibronectin and carcinoembryonic
antigen, have also been reported (7,23–25).
All these markers will aid the differential diagnosis of GIST,
leiomyoma and squamous cancers. Ki-67, which is a nuclear
proliferation-associated antigen and is considered as a reliable
marker of cell proliferation, was reported negative in the majority
of cases, however, in certain cases it was locally positive
(25,26). In the present study, Ki-67 was
identified as locally positive in three cases and, in these cases,
pleomorphism was observed. Thus, it is indicated that Ki-67
staining should be undertaken in all GCTs to identify the
differential degree of the tumor.
In conclusion, esophageal GCTs occurred mostly in
females between 40 and 60 years old, and almost all were solitary
in the present study. The endoscopic and EUS features varied widely
and EUS may be necessary in the surveillance. Immunohistochemical
staining of tumor markers will aid the diagnosis and differential
diagnosis.
Acknowledgements
This study was supported by Nanjing Medical Science
and the Technique Development Foundation.
Abbreviations:
|
GCT
|
granular cell tumor
|
|
EUS
|
endoscopic ultrasound
|
|
SMA
|
smooth muscle antibody
|
References
|
1
|
Abrikossoff A: Myomas originating from
transversely striated voluntary musculature. Virchows Arch A Pathol
Anat Histol. 260:215–233. 1926.(In German).
|
|
2
|
Morrison JG, Gray GF Jr, Dao AH and Adkins
RB Jr: Granular cell tumors. Am Surg. 53:156–160. 1987.
|
|
3
|
De Rezende L, Lucendo AJ and
Alvarez-Argüelles H: Granular cell tumors of the esophagus: report
of five cases and review of diagnostic and therapeutic techniques.
Dis Esophagus. 20:436–443. 2007.
|
|
4
|
Abrikossoff A: Further investigations on
myoblastomas. Virchows Arch Pathol Anat Physiol Klin Med.
280:723–40. 1931.(In German).
|
|
5
|
Zhong N, Katzka DA, Smyrk TC, Wang KK and
Topazian M: Endoscopic diagnosis and resection of esophageal
granular cell tumors. Dis Esophagus. 24:538–543. 2011.
|
|
6
|
Johnston J and Helwig EB: Granular cell
tumors of the gastrointestinal tract and perianal region: a study
of 74 cases. Dig Dis Sci. 26:807–816. 1981.
|
|
7
|
Parfitt JR, McLean CA, Joseph MG,
Streutker CJ, Al-Haddad S and Driman DK: Granular cell tumours of
the gastrointestinal tract: expression of nestin and
clinicopathological evaluation of 11 patients. Histopathology.
48:424–430. 2006.
|
|
8
|
Fujiwara Y, Watanabe T, Hamasaki N, Wada
T, Shiba M, Uchida T, et al: Endoscopic resection of two granular
cell tumours of the oesophagus. Eur J Gastroenterol Hepatol.
11:1413–1416. 1999.
|
|
9
|
Voskuil JH, van Dijk MM, Wagenaar SS, van
Vliet AC, Timmer R and van Hees PA: Occurrence of esophageal
granular cell tumors in The Netherlands between 1988 and 1994. Dig
Dis Sci. 46:1610–1614. 2001.
|
|
10
|
Goldblum JR, Rice TW, Zuccaro G and
Richter JE: Granular cell tumors of the esophagus: a clinical and
pathologic study of 13 cases. Ann Thorac Surg. 62:860–865.
1996.
|
|
11
|
Patel RM, DeSota-LaPaix F, Sika JV,
Mallaiah LR and Purow E: Granular cell tumor of the esophagus. Am J
Gastroenterol. 76:519–523. 1981.
|
|
12
|
Palazzo L, Landi B, Cellier C, Roseau G,
Chaussade S, Couturier D and Barbier J: Endosonographic features of
esophageal granular cell tumors. Endoscopy. 29:850–853. 1997.
|
|
13
|
Garrido E, Marín E, González C, Juzgado D,
Boixeda D and Vázquez-Sequeiros E: Endoscopic mucosal resection of
Abrikosoff’s tumor of the esophagus. Gastroenterol Hepatol.
31:572–575. 2008.(In Spanish).
|
|
14
|
Schröder G and Kohlmann HW: Therapeutic
endoscopy of an Abrikosov-tumor of the esophagus. Z Gastroenterol.
17:281–286. 1979.(In German).
|
|
15
|
Orlowska J, Pachlewski J, Gugulski A and
Butruk E: A conservative approach to granular cell tumors of the
esophagus: four case reports and literature review. Am J
Gastroenterol. 88:311–315. 1993.
|
|
16
|
Radaelli F and Minoli G: Granular cell
tumors of the gastrointestinal tract: Questions and answers.
Gastroenterol Hepatol. 11:798–800. 2009.
|
|
17
|
Lack EE, Worsham GF, Callihan MD, Crawford
BE, Klappenbach S, Rowden G and Chun B: Granular cell tumor: a
clinicopathologic study of 110 patients. J Surg Oncol. 13:301–316.
1980.
|
|
18
|
Szumilo J, Dabrowski A, Skomra D and
Chibowski D: Coexistence of esophageal granular cell tumor and
squamous cell carcinoma: a case report. Dis Esophagus. 15:88–92.
2002.
|
|
19
|
Saito K, Kato H, Fukai Y, Kimura H,
Miyazaki T, Kashiwabara K, et al: Esophageal granular cell tumor
covered by intramucosal squamous cell carcinoma: report of a case.
Surg Today. 38:651–655. 2008.
|
|
20
|
Prematilleke IV, Sujendran V, Warren BF,
Maynard ND and Piris J: Granular cell tumour of the oesophagus
mimicking a gastrointestinal stromal tumour on frozen section.
Histopathology. 44:502–503. 2004.
|
|
21
|
Miwa K, Hattori T, Hosokawa Y, Nakamura Y,
Isobe Y, Fujisawa K and Nakagawara G: Granular cell tumor of the
esophagus. Gastroenterol Jpn. 21:508–512. 1986.
|
|
22
|
Szumiło J, Skomra D, Zinkiewicz K and
Zgodziński W: Multiple synchronous granular cell tumours of the
esophagus: a case report. Ann Univ Mariae Curie Sklodowska Med.
56:253–256. 2001.
|
|
23
|
Ohmori T, Arita N, Uraga N, Tabei R, Tani
M and Okamura H: Malignant granular cell tumor of the esophagus. A
case report with light and electron microscopic, histochemical, and
immunohistochemical study. Acta Pathol Jpn. 37:775–783. 1987.
|
|
24
|
John BK, Dang NC, Hussain SA, Yang GC,
Cham MD, Yantiss R, et al: Multifocal granular cell tumor
presenting as an esophageal stricture. J Gastrointest Cancer.
39:107–113. 2008.
|
|
25
|
Maekawa H, Maekawa T, Yabuki K, Sato K,
Tamazaki Y, Kudo K, et al: Multiple esophagogastric granular cell
tumors. J Gastroenterol. 38:776–780. 2003.
|
|
26
|
Mitomi H, Matsumoto Y, Mori A, Arai N,
Ishii K, Tanabe S, et al: Multifocal granular cell tumors of the
gastrointestinal tract: Immunohistochemical findings compared with
those of solitary tumors. Pathol Int. 54:47–51. 2004.
|