Introduction
Benign lipomatous tumors are classified into five
types, lipoma, variants of lipoma, heterotopic lipomas,
hamartomatous lesions, infiltrating or diffuse neoplastic or
non-neoplastic proliferations of mature fat and hibernoma.
Angiolipoma is a variant of lipoma (1). Infiltrating angiolipoma (IAL) is a
rare lesion, and is a clinicopathological variant of angiolipoma,
characterized by infiltration of the surrounding structures,
particularly skeletal muscle. Angiolipoma accounts for 5–17% of all
lipomas and predominantly presents as subcutaneous nodules in young
adults, which are tender or painful on palpation, particularly
during the initial growth period. Angiolipoma exists in two forms,
circumscribed and diffuse. Diffuse tumors are considered to be IAL
with infiltration of the surrounding muscles. IAL has a high risk
of recurrence following surgical excision. Furthermore, IAL may
behave in a similar manner to that of a local aggressive neoplasm.
In 1966, the tumor was characterized as a clinicopathological
entity by Gonzalez-Crussi et al (2), who reviewed the previous literature
and identified cases which were consistent with this diagnosis.
According to the English literature, the tumor
rarely occurs in the head and neck and is extremely rare in the
oral cavity (3). Wide resection is
recommended for the treatment of infiltrating angiolipomas, due to
the risk of recurrence (4). The
current study reports the case of a 74-year-old female with IAL of
the lower lip. Following the surgical excision of the tumor, the
mRNA expression levels of the vascular endothelial growth factor
(VEGF) family members in the tumor were investigated. To the best
of our knowledge, this is the first case of IAL arising in the
lower lip to be reported. Written informed consent was obtained
from the patient’s family.
Case report
Patient and case history
In April 2000, a 74-year-old female was referred to
the Second Department of Oral and Maxillofacial Surgery, Osaka
Dental University (Osaka, Japan) with a painless mass in the lower
lip, which had been present for approximately four months. The
patient had no history of facial trauma. Clinical examination
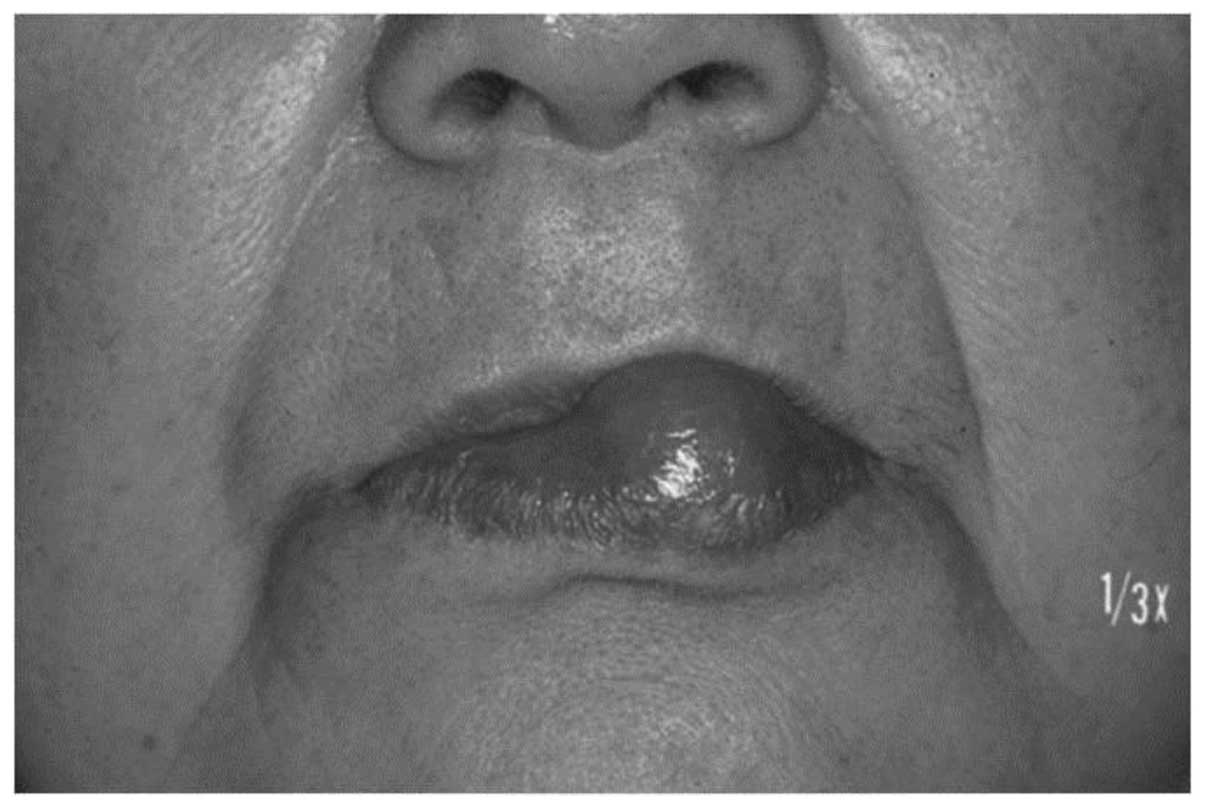
revealed a relatively circumscribed soft-tissue mass of 20×19 mm in
diameter in the lower lip (Fig. 1).
The overlying mucosa was intact. Hematological and biochemical
parameters were within the normal limits. White blood cell count,
68.6×102/μl (normal range, 35.8–80.0
×102/μl); red blood cell count, 483×104/μl
(normal range, 380–480×104/μl); hemaglobin level, 15.2
g/dl (normal range, 11.3–15.2 g/dl); hematocrit level, 44.0%
(normal range, 34.0–43.0%); platelet count, 23.4×104/μl
(normal range, 15.0–35.0×104/μl); glutamic oxaloacetic
transaminase level, 22 U/l (normal range, 7–38 U/l); glutamic
pyruvic transaminase level, 30 U/l (normal range, 4–44 U/l);
γ-glutamyl transpeptidase, 30 U/l (normal range 9–35 U/l); creatine
phosphokinase, 96 U/l (normal range, 32–187 U/l); blood urea
nitrogen level, 17.5 mg/dl (normal range, 8.0–20.0 mg/dl);
creatinine level, 0.51 mg/dl (normal range, 0.44–0.75 mg/dl);
c-reactive protein level, 0.06 mg/dl (normal range, 0.00–0.30
mg/dl). A diagnosis of a benign tumor was determined
preoperatively. Ultrasound examination was performed, however,
detailed information could not be obtained due to the size of the
tumor. Subsequently, an excisional biopsy was performed under local
anesthesia. As the tumor was unencapsulated, the normal tissue
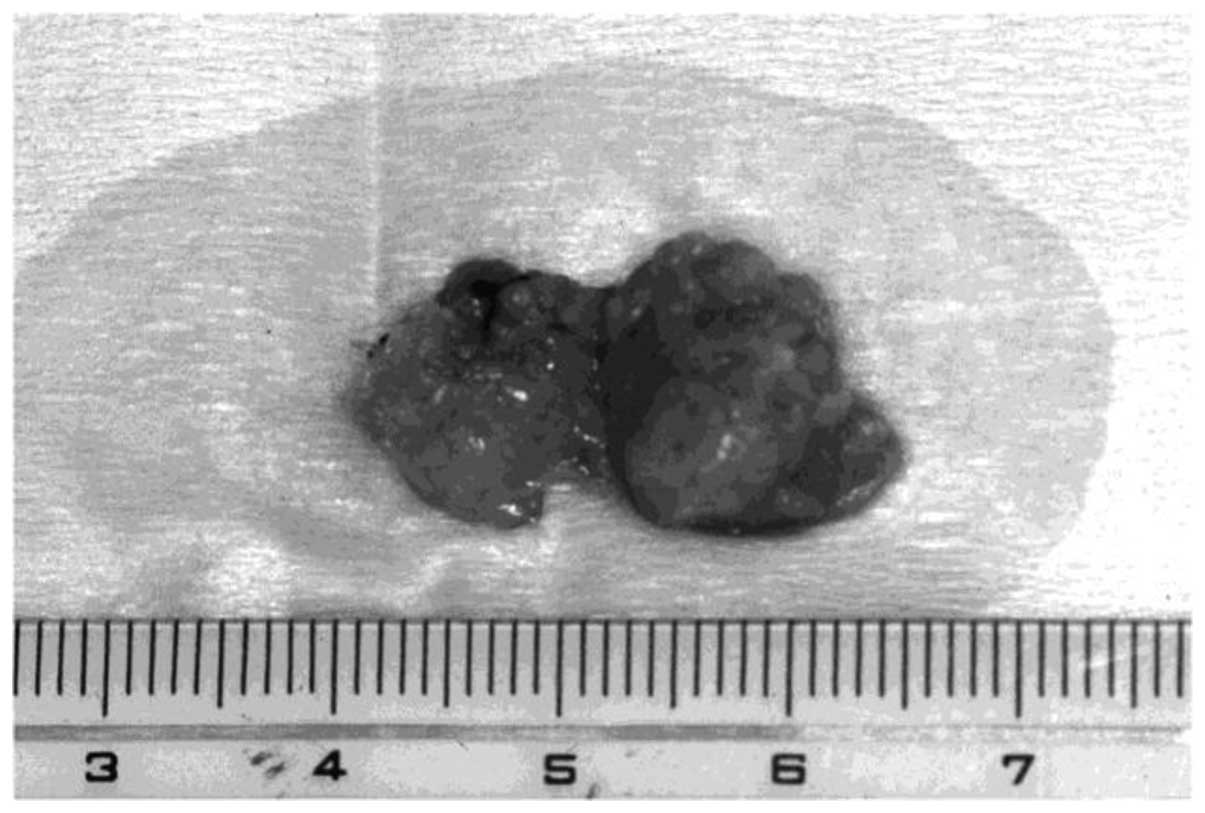
surrounding the tumor was extirpated. The excised specimen revealed
a solid soft-tissue mass (20×19×10 mm) with a dark yellow surface
(Fig. 2).
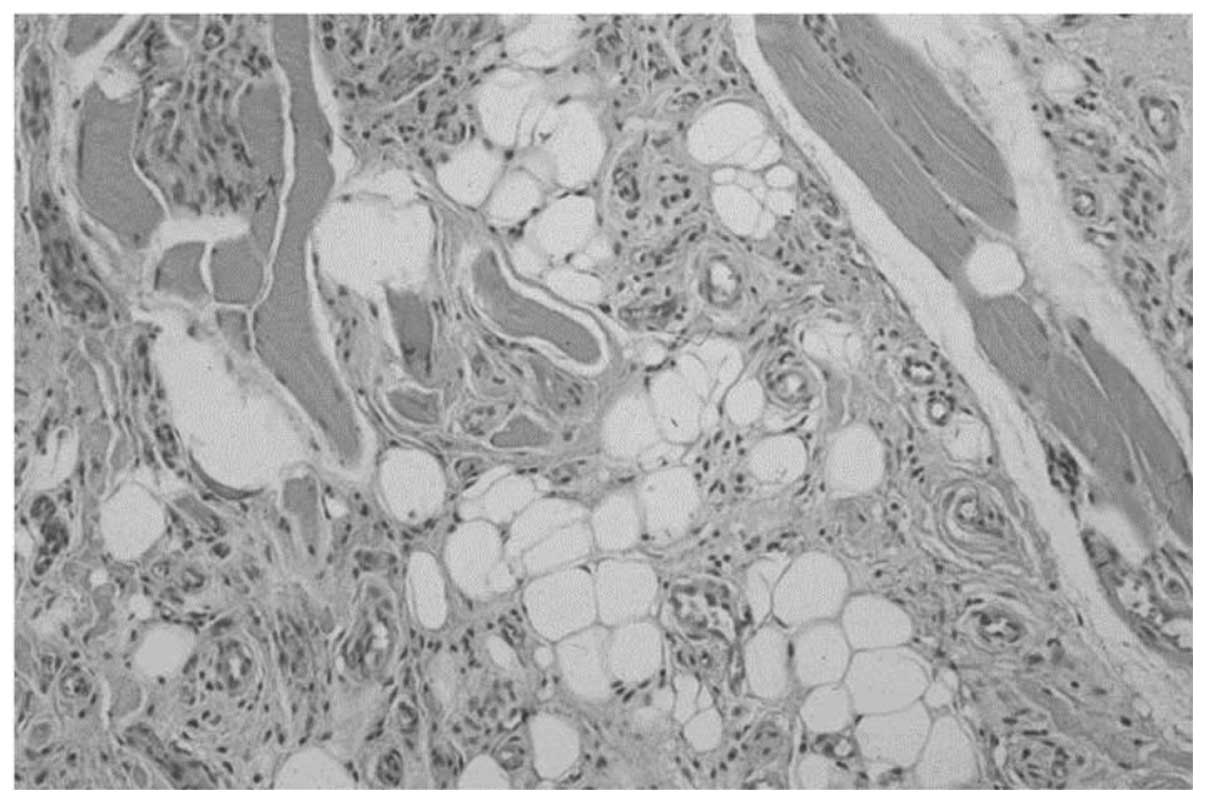
Microscopically, the specimen was unencapsulated and
mature lipocytes were separated by a branching network of
proliferating small vessels that infiltrated the adjacent tissues,
and muscle fibers partially existed in the tumor. It was composed
of proliferating mature lipocytes and numerous small blood vessels
containing microthrombi under the epithelium (Fig. 3). Cellular atypia was not observed,
therefore, the pathological diagnosis of this lesion was IAL
arising in the lower lip. No evidence of recurrence has been
identified during four years of follow up.
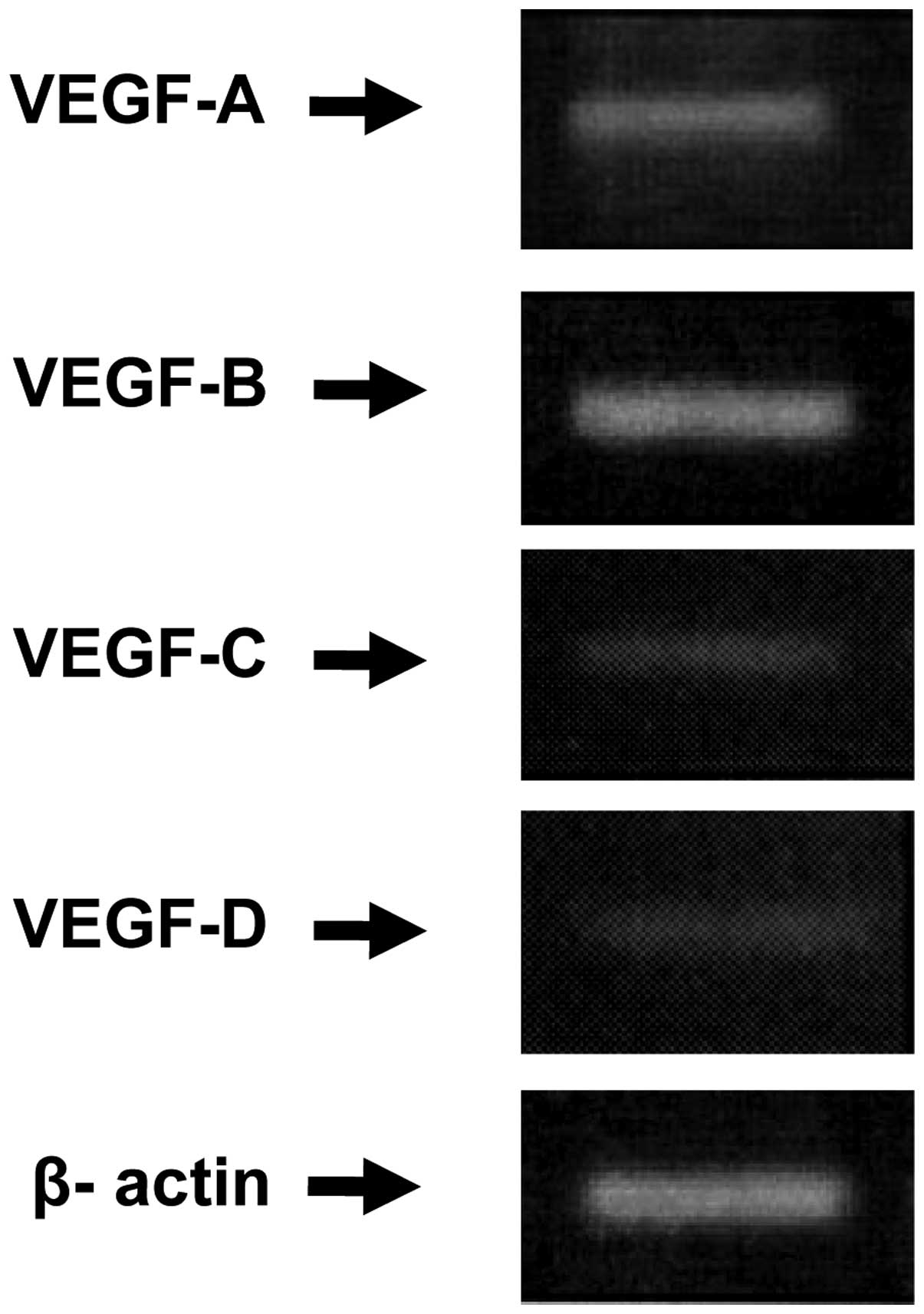
The expression of mRNA of all VEGF family members
was detected in the tumor by reverse transcription (RT)-polymerase
chain reaction (PCR) analysis, as shown in Fig. 4. However, the relative expression
level of each of the VEGF family members differed significantly. A
higher relative expression level of VEGF-A and -B were observed,
when compared with VEGF-C and -D, which exhibited extremely low
expression levels.
RNA preparation and RT-PCR
Total RNA was isolated from the tissues of the
patient using TRIZOL reagent (Invitrogen Life Technologies, Inc.,
Carlsbad, CA, USA), immediately after resection, according to the
manufacturer’s instructions. A total of 10 μl RT buffer (3 mM
MgCl2, 10 mM Tris-HCl, 75 mM KCl, 1 mM bovine serum
albumin; pH 8.3) containing 1 μg RNA, 0.2 μg oligo-dT primers, 0.5
mM dNTP, 5 U of RNasin and 100 U of Moloney murine leukemia virus
reverse transcriptase (Invitrogen Life Technologies, Inc.) was
incubated at 37°C for 60 min and a section of each RT product was
amplified by PCR, using a thermo cycler (Takara PCR Thermal Cycler
Dice Gradient TP600; Takara Bio, Inc., Otsu, Japan).
The size and sequences of the primers used are shown
in Table I. The PCR reactions
conditions were as follows: 40 cycles of denaturation at 94°C for 1
min, annealing at 52°C for 2 min, and chain extension with Taq
polymerase (Invitrogen Life Technologies, Inc.) at 72°C for 1 min,
followed by a final extension step at 72°C for 20 min. Following
amplification, the PCR reaction mixture was analyzed by 2% agarose
gel electrophoresis and stained with ethidium bromide (Invitrogen
Life Technologies, Inc.).
 | Table IPCR primers sequences used for
reverse-transcription-PCR. |
Table I
PCR primers sequences used for
reverse-transcription-PCR.
| Gene | Product size, bp | Primer sequence |
|---|
| VEGF-A | 212 |
5′-GCAGAATCATCACGAAGTGG-3′
5′-GCATGGTGATGTTGGACTCC-3′ |
| VEGF-B | 246 |
5′-CCTTGACTGTGGAGCTCATG-3′
5′-TGTCTGGCTTCACAGCACTG-3′ |
| VEGF-C | 435 |
5′-AGACTCAATGCATGCCACG-3′
5′-TTGAGTCATCTCCAGCATCC-3′ |
| VEGF-D | 313 |
5′-GCTGTTGCAATGAAGAGAGC-3′
5′-TCTTCTGTTCCAGCAAGTGG-3′ |
| β-actin | 610 |
5′-TGACGGGGTCACCCACACTGTGCCCATCTA-3′
5′-CTAGAAGCATTTGCGGTGGACGATGGAGGG-3′ |
Discussion
Infiltrating lipoma is a rare variant of lipoma. In
1853, Paget reported the case of a lipoma that infiltrated the
trapezius muscle (2). In 1946,
Regan et al (5) reviewed
several cases and defined this entity. The tumor is most commonly
identified in the deep muscles of the buttock, shoulder, thigh and
extremities (6). Clinically, oral
infiltrating lipoma presents as a painless solitary submucosal
swelling. On palpation, the tumor is semi-firm and rubbery, with
poorly defined margins. It is usually identified in the deeper
tissues. Dionne and Seemayer (7)
reviewed 20 patients with infiltrating lipoma and observed a
recurrence rate of 62.5%.
Angiolipoma accounts for 5–17% of all lipomas and
predominantly presents as subcutaneous nodules in young adults,
which are tender or painful on palpation, particularly during the
initial growth period (8).
Microscopically, the tumor presents as yellow nodules, which
consist of mature fat cells separated by a branching network of
small vessels. The proportion of fatty tissue and vascular channels
varies (9). The vessels commonly
contain fibrin thrombi, without evidence of necrosis due to the
extensive collateral circulation (8). Angiolipoma exists in two forms,
circumscribed and diffuse. The circumscribed variants, with a few
exceptions, are limited to the subcutis. Diffuse angiolipoma arises
in the deep soft tissues and infiltrates adjacent structures and
thus, complete excision is difficult (8). In the present case, the tumor was
considered to be IAL and may be termed intramuscular angiolipoma,
with infiltration of the surrounding muscles.
IAL occurs most commonly in the trunk and
extremities (7–9), however, it is extremely rare in the
oral cavity. To the best of our knowledge, only four cases of IAL
of the oral cavity, including our case, have been reported in the
English literature (Table II). The
first and second cases of oral IAL were found in the tongue of a
49-year-old male (10) and the
mucolabial fold of a 74-year-old male, respectively (11).
 | Table IIInfiltrating angiolioima of the oral
cavity. |
Table II
Infiltrating angiolioima of the oral
cavity.
| Authors (ref) | Age, years | Gender | Location | Size, cm |
|---|
| Lin et al
(10) | 49 | Male | Tongue | 3.0×2.0×2.5 |
| Sugiura et al
(11) | 74 | Male | Mucolabial fold | 1.0×1.0×2.0 |
| Dalambiris et
al (3) | 56 | Female | Upper labial | 1.0×1.2×0.5 |
| Present case | 74 | Female | Lower lip | 2.0×1.9×1.0 |
Lipoma is usually well encapsulated with a smooth or
lobulated surface and thus, it may be resected easily, however, IAL
is not encapsulated and complete excision is difficult due to the
infiltration of the surrounding tissues, particularly the muscle.
The recurrence rate of the tumor following surgical extirpation is
35–50% (7). The probable causes of
recurrence are inaccurate preoperative estimation of the extent of
the tumor and the obscure demarcation encountered during surgery
(8,12). In order to clarify the extent of
tumor demarcation, ultrasound provides information regarding the
extent of the tumor and the infiltration of other anatomical
structures. In the present case, the tumor could not be
differentiated from the muscle by ultrasound. Angiography may
provide more detailed information with regard to association
between the vascular supply and the major vessels (13). However, magnetic resonance imaging
has been reported to be more valuable than angiography or computed
tomography in determining the extent of the tumor and asserting a
preoperative diagnosis (12). Wide
excision of the tumor has been previously reported, however, this
often results in significant morbidity (14). Ida-Yonemochi et al (15) hypothesized that an operating
microscope may be utilized for total removal of an IAL in order to
minimize damage to the normal tissues during total extirpation of
cerebral arteriovenous malformations (12). In the present study, the tumor was
unencapsulated and infiltrated the muscle of the lower lip and
thus, the tumor was extirpated carefully, including the normal
tissue surrounding the tumor. A number of studies have reported the
use of radiotherapy for the treatment recurrences (2,9,16). In
this case, the tumor was completely excised and therefore, no
further treatment was required and no recurrence was observed
during the four years of postoperative follow up.
Matsuoka et al (12) revealed that mast cells surrounding
blood vessels expressed high levels of VEGF, which is known to be
an essential growth factor for endothelial cells in vasculogenesis.
This result indicates that mast cell-derived VEGF may be
responsible for the enhanced vascularity observed in this tumor. In
the present case, VEGF-A and -B, which are known to stimulate the
formation of blood vessels in tumors, were expressed. However,
VEGF-C and -D, which may promote the development of lymphatic
vessels in tumors and entry of tumor cells into lymphatic vessels,
were expressed at extremely low levels. We hypothesize that VEGF-C
and -D expression was low due to the benign nature of the
tumor.
Ida-Yoncmochi et al (15) demonstrated that mast cells
surrounding blood vessels strongly expressed VEGF, which is known
to be an essential growth factor for endothelial cells in
vasculogenesis. Although in situ hybridization was not
performed in the present study, VEGF production by mast cells is
highly probable as there were no other inflammatory cells within
the tumor tissue. These result indicates that mast cell-derived
VEGF may be responsible for the enhanced vascularity of this tumor.
Therefore, we believe that IAL is associated with fat, rather than
with neoplasm. We therefore recommend careful extirpation with no
wide safety margin to be the procedure of choice, except in those
cases where the tumor has invaded irregularly into the muscles.
In conclusion, cases of infiltrating angiolipoma of
the oral cavity are extremely rare. Magnetic resonance imaging has
been reported to be valuable in determining the extent of the tumor
and asserting a preoperative diagnosis. Histopathology showing
mature fat cells and numerous capillaries invading surrounding
structure may verify the diagnosis. We consider careful extirpation
with no wide safety margin to be the procedure of choice, with the
exception of cases where the tumor invades irregularly into the
muscles.
References
|
1
|
Weiss SW and Goldblum JR: Benign
lipomatius tumors. Enzinger and Weiss’s Soft Tissue Tumors. 4th
edition. Mosby; St Louis, MO: pp. 571–639. 2001
|
|
2
|
Gozalez-Crussi F, Enneking WF and Arean
VM: Infiltrating angiolipoma. J Bone Joint Surg. 48:1111–1124.
1966.
|
|
3
|
Dalambiras S, Tilaveridis I, Iordanidis S,
Zaraboukas T and Epivatianos A: Infiltrating angiolipoma of a the
oral cavity: report of a case and literature review. J Oral
Maxillofac Surg. 68:681–683. 2010. View Article : Google Scholar
|
|
4
|
Hamakawa H, Hino H, Sumida T and Tanioka
H: Infiltrating angiolipoma of the cheek: a case report and a
review of the literature. J Oral Maxillofac Surg. 58:674–677. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Regan JM, Bickel WH and Brodes AC:
Infiltrating lipomas of the extremities. Surg Gynecol Obstet.
54:871946.
|
|
6
|
Kindblom LG, Angervall L, Stener B and
Wickbom I: Intermusclar and intramusclar lipomas and hidernomas: A
clinical, roentgenologic, histlogic and prognostic study of 46
cases. Cancer. 33:754–762. 1974. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Dionne GP and Seemayer TA: Infiltrating
lipomas and angiolipomas revisited. Cancer. 33:732–738. 1974.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lin JJ and Lin F: Two entities in
angiolipoma: A study of 459 cases of lipoma with review of
literature on infiltrating angiolipoma. Cancer. 34:720–727. 1974.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Enzinger FM and Weiss SW: Soft Tissue
Tumors. St Louis, MO: Mosby; pp. 301–345. 1988
|
|
10
|
Lin SC, Wang TY and Hahn LJ: Angiolipoma
of the tongue: Report of a case. Ann Dent. 48:37–38.
1989.PubMed/NCBI
|
|
11
|
Sugiura J, Fujiwara K, Kurahashi I and
Kimura Y: Infiltrating angiolipoma of the mucolabial fold: A case
report and review of the literature. J Oral Maxillofac Surg.
57:446–448. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Matsuoka Y, Kurose K, Nakagawa O and
Katsuyama J: Magnetic resonance imaging of infiltrating angiolipoma
of the neck. Surg Neurol. 29:62–66. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chew FS, Hudson TM and Hawkins IF:
Radiology of infiltrating angiolipoma. AJR Am J Roentgenol.
135:781–787. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Austin RM, Mack GR, Townsend CM and Lack
EE: Infiltrating (intramuscular) lipoma and angiolipomas. A
clinicopathologic study of six cases. Arch Surg. 115:281–284. 1980.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ida-Yoncmochi H, Swelam W, Saito C and
Saku T: Angiolipoma of the buccal mucosa: a possible role of mast
cell-derived VEGF in its enhanced vascularity. J Oral Pathol Med.
34:59–61. 2005. View Article : Google Scholar
|
|
16
|
Stimpson N: Infiltrating angiolipoma of
skeletal muscle. Br J Surg. 58:464–466. 1971. View Article : Google Scholar : PubMed/NCBI
|